Applying Generic Water Quality Criteria to Cu and Zn in a Dynamic Aquatic Environment—The Case of the Brackish Water Formation Strömmen-Saltsjön
Abstract
1. Introduction
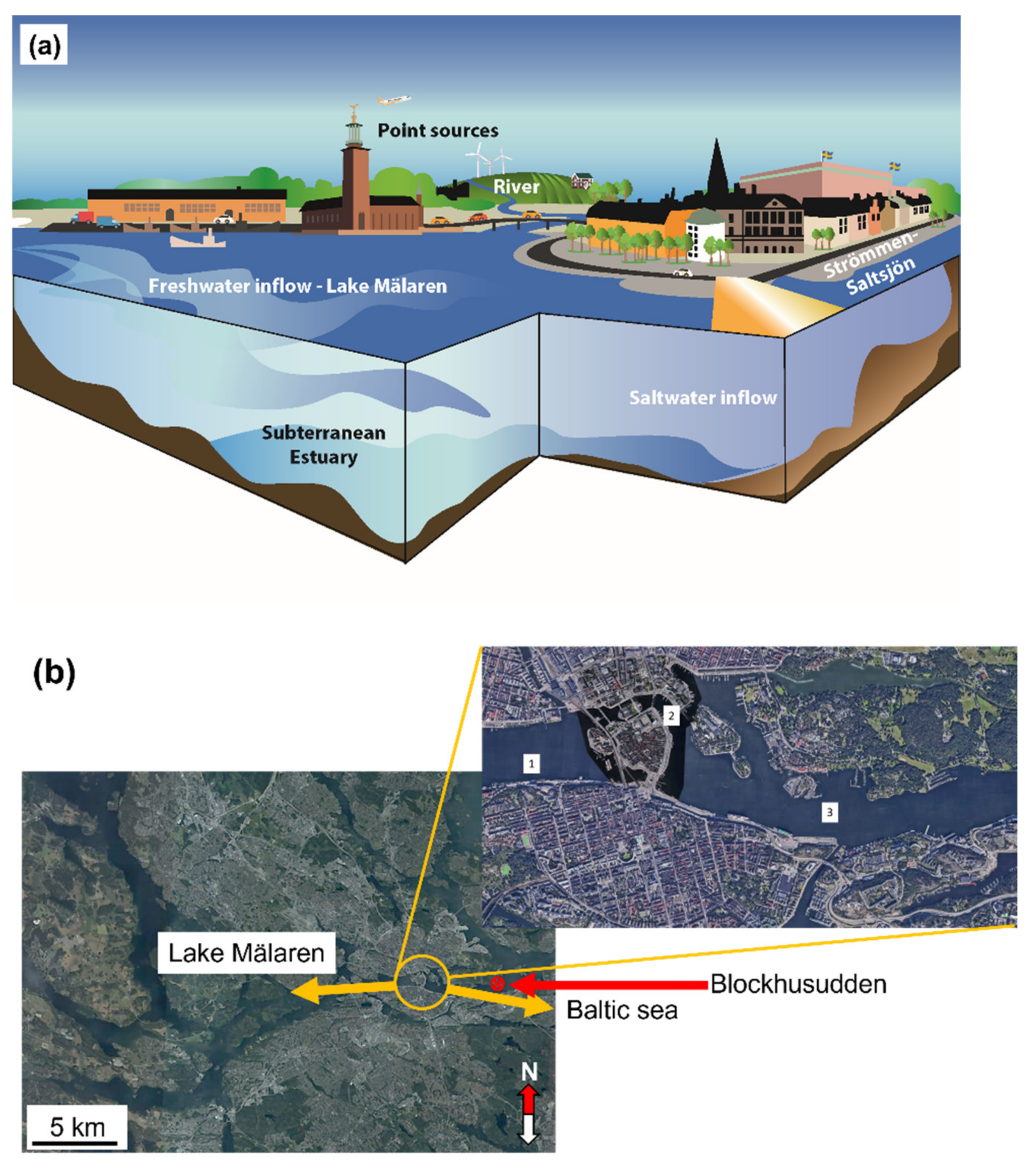
2. Strömmen-Saltsjön Water Body—A Case Study
2.1. Site Description
2.2. Water Chemistry and Flows of Cu and Zn to the Strömmen-Saltsjön Water Body
2.3. Aquatic Life of the Strömmen-Saltsjön Water Body
3. Results and Discussion
3.1. Current Status Classification
3.2. Water Classification Based on Bioavailability of Cu and Zn Is Different from Predictions Made Using Current National Classification Employing Generic Safety Factors
3.3. Sediment Concentrations of Cu and Zn Cannot Be Used as a Basis for Chemical and Ecological Status in Water Formations
3.4. Background Concentrations, Bioavailability and Organism Sensitivity in Waters of Dynamic Salinity Need to Be Considered in Risk Assessment
3.5. The Prevailing Benthic Fauna Should Be Considered in Risk Assessment
4. Conclusions
- It is possible to improve ecological status classification by making more locally suitable assessments of the ecotoxicity of Cu and Zn in low salinity brackish waters in Swedish estuaries.
- Suitable assessment grounds for fresh waters versus marine waters shall be chosen according to water characteristics rather than geographical location.
- Where metal variations are great, especially in sediments, monitoring the organism vitality and reproduction makes more sense.
- Background concentrations and acclimatisation are important aspects to consider for metals such as Cu and Zn, which are naturally occurring and essential nutrients for organisms.
- Overly conservative assessment factors and lack of assessment of the ability of biological uptake of metals will make risk assessments less reliable. This will result in higher administrative costs and difficulties for society to comply, while no improvement in the real status of the environment will be achieved.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Copper Institute Voluntary Risk Assessment Report (VRAR) for Copper and Copper Compound Submitted to ECHA Based on Industry Initiative to Follow the Risk Assessment Procedures of Existing Substance Regulation (EEC) No 793/93. Available online: https://echa.europa.eu/copper-voluntary-risk-assessment-reports (accessed on 10 December 2021).
- Available online: http://echa.europa.eu/documents/10162/d7248de0-eb5b-4a9b-83b9-042c4fd66998 (accessed on 1 January 2021).
- Foekema, E.M.; Kaag, N.H.B.M.; Kramer, K.J.M.; Long, K. Mesocosm validation of the marine No Effect Concentration of dissolved copper derived from a species sensitivity distribution. Sci. Total Environ. 2015, 521–522, 173–182. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Arnold, W.R.; Cotsifas, J.S.; Smith, D.S.; Le Page, S.; Gruenthal, K.M. A comparison of the copper sensitivity of two economically important saltwater mussel species and a review of previously reported copper toxicity data for mussels: Important implications for determining future ambient copper saltwater criteria in the USA. Environ. Toxicol. 2009, 24, 618–628. [Google Scholar] [CrossRef]
- Hall, L.W.; Anderson, R.D.; Lewis, B.L.; Arnold, W.R. The influence of salinity and dissolved organic carbon on the toxicity of copper to the estuarine copepod, Eurytemora affinis. Arch. Environ. Contam. Toxicol. 2008, 54, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Grosell, M.; Nielsen, C.; Bianchini, A. Sodium turnover rate determines sensitivity to acute copper and silver exposure in freshwater animals. Comp. Biochem. Physiol. Part. C Toxicol. Pharmacol. 2002, 133, 287–303. [Google Scholar] [CrossRef]
- Khan, F.R.; Bury, N.R.; Hogstrand, C. Copper and zinc detoxification in Gammarus pulex (L.). J. Exp. Biol. 2012, 215, 822–832. [Google Scholar] [CrossRef] [PubMed]
- De Souza Machado, A.A.; Spencer, K.; Kloas, W.; Toffolon, M.; Zarfl, C. Metal fate and effects in estuaries: A review and conceptual model for better understanding of toxicity. Sci. Total Environ. 2016, 541, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Wood, C.M.; Farrell, A.P.; Brauner, C.J. Homeostasis and Toxicology of Essential Metals; Elsevier: London, UK, 2012. [Google Scholar]
- Sunda, W.G.; Huntsman, S.A. Processes regulating cellular metal accumulation and physiological effects: Phytoplankton as model systems. Sci. Total Environ. 1998, 219, 165–181. [Google Scholar] [CrossRef]
- Grosell, M.; Blanchard, J.; Brix, K.V.; Gerdes, R. Physiology is pivotal for interactions between salinity and acute copper toxicity to fish and invertebrates. Aquat. Toxicol. 2007, 84, 162–172. [Google Scholar] [CrossRef]
- De Polo, A.; Scrimshaw, M.D. Challenges for the development of a biotic ligand model predicting copper toxicity in estuaries and seas. Environ. Toxicol. Chem. 2012, 31, 230–238. [Google Scholar] [CrossRef]
- Wright, D.A. Trace metal and major ion interactions in aquatic animals. Mar. Pollut. Bull. 1995, 31, 8–18. [Google Scholar] [CrossRef]
- Pinho, G.L.L.; Pedroso, M.S.; Rodrigues, S.C.; Souza, S.S.D.; Bianchini, A. Physiological effects of copper in the euryhaline copepod Acartia tonsa: Waterborne versus waterborne plus dietborne exposure. Aquat. Toxicol. 2007, 84, 62–70. [Google Scholar] [CrossRef]
- Pinho, G.L.L.; Bianchini, A. Acute copper toxicity in the euryhaline copepod Acartia tonsa: Implications for the development of an estuarine and marine biotic ligand model. Environ. Toxicol. Chem. 2010, 29, 1834–1840. [Google Scholar] [CrossRef]
- Pearson, H.B.C.; Comber, S.D.W.; Braungardt, C.B.; Worsfold, P.; Stockdale, A.; Lofts, S. Determination and Prediction of Zinc Speciation in Estuaries. Environ. Sci. Technol. 2018, 52, 14245–14255. [Google Scholar] [CrossRef]
- Monserrat, J.M.; Martínez, P.E.; Geracitano, L.A.; Lund Amado, L.; Martinez Gaspar Martins, C.; Lopes Leães Pinho, G.; Soares Chaves, I.; Ferreira-Cravo, M.; Ventura-Lima, J.; Bianchini, A. Pollution biomarkers in estuarine animals: Critical review and new perspectives. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2007, 146, 221–234. [Google Scholar] [CrossRef]
- Klerks, P.L.; Weis, J.S. Genetic adaptation to heavy metals in aquatic organisms: A review. Environ. Pollut. 1987, 45, 173–205. [Google Scholar] [CrossRef]
- Muyssen, B. Tolerance and acclimation to zinc of field-collected Daphnia magna populations. Aquat. Toxicol. 2002, 56, 69–79. [Google Scholar] [CrossRef]
- Johnson, H.L.; Stauber, J.L.; Adams, M.S.; Jolley, D.F. Copper and zinc tolerance of two tropical microalgae after copper acclimation. Environ. Toxicol. 2007, 22, 234–244. [Google Scholar] [CrossRef]
- Paquin, P.R.; Gorsuch, J.W.; Apte, S.; Batley, G.E.; Bowles, K.C.; Campbell, P.G.C.; Delos, C.G.; Di Toro, D.M.; Dwyer, R.L.; Galvez, F.; et al. The biotic ligand model: A historical overview. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2002, 133, 3–35. [Google Scholar] [CrossRef]
- Niyogi, S.; Wood, C.M. Biotic Ligand Model, a flexible tool for developing site-specific water quality guidelines for metals. Environ. Sci. Technol. 2004, 38, 6177–6192. [Google Scholar] [CrossRef]
- Nys, C.; Van Regenmortel, T.; Janssen, C.R.; Oorts, K.; Smolders, E.; De Schamphelaere, K.A.C. A framework for ecological risk assessment of metal mixtures in aquatic systems. Environ. Toxicol. Chem. 2018, 37, 623–642. [Google Scholar] [CrossRef]
- European Union. Directive 2008/105/EC of the European Parliament and of the Council of 16 December 2008 on environmental quality standards in the field of water policy, amending and subsequently repealing Council Directives 82/176/EEC, 83/513/EEC, 84/156/EEC, 84/491/EEC, 86/280/EEC and amending Directive 2000/60/EC of the European Parliament and of the Council. Off. J. Eur. Union 2018, L 348, 84–97. [Google Scholar]
- Arnold, W.R.; Santore, R.C.; Cotsifas, J.S. Predicting copper toxicity in estuarine and marine waters using the Biotic Ligand Model. Mar. Pollut. Bull. 2005, 50, 1634–1640. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Guidance document n.o 5. In Transitional and Coastal Waters, Typology, Reference Conditions and Classification Systems; Commission, European Communities: Luxembourg, Luxembourg, 2003. [Google Scholar]
- Pearson, H.B.C.; Comber, S.D.W.; Braungardt, C.; Worsfold, P.J. Predicting copper speciation in estuarine waters—Is dissolved organic carbon a good proxy for the presence of organic ligands? Environ. Sci. Technol. 2017, 51, 2206–2216. [Google Scholar] [CrossRef] [PubMed]
- The European Parliament and the Council of the European Union. The EU Water Framework Directive—Integrated River Basin Management for Europe. Available online: https://ec.europa.eu/environment/water/water-framework/index_en.html (accessed on 17 January 2021).
- Pradhan, A.; Ivarsson, P.; Ragnvaldsson, D.; Berg, H.; Jass, J.; Olsson, P.-E. Transcriptional responses of zebrafish to complex metal mixtures in laboratory studies overestimates the responses observed with environmental water. Sci. Total Environ. 2017, 584–585, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Peters, A.; Nys, C.; Merrington, G.; Verdonck, F.; Baken, S.; Cooper, C.A.; Van Assche, F.; Schlekat, C.; Garman, E. Demonstrating the reliability of bio-met for determining compliance with environmental quality standards for etals in Europe. Environ. Toxicol. Chem. 2020, 39, 2361–2377. [Google Scholar] [CrossRef] [PubMed]
- Palm Cousins, A.; Jönsson, A.; Iverfeldt, Å. Testing the Biotic Ligand Model for Swedish Surface Water Conditions—A Pilot Study to Investigate the Applicability of BLM in Sweden; IVL Report B1858; IVL Swedish Environmental Research Institute: Stockholm, Sweden, 2009. [Google Scholar]
- Foekema, E.M.; Kramer, K.J.M.; Kaag, N.H.B.M.; Sneekes, A.C.; Bierman, S.; Hoornsman, G.; Koelemij, E. Determination of the Biological Effects and Fate of Dissolved Zinc in Outdoor Marine Mesocosms; C108/12; Institute for Marine Resources & Ecosystem Studies: Yerseke, The Netherlands, 2012. [Google Scholar]
- Available online: https://www.aces.su.se/aces/wp-content/uploads/2018/10/Copper-EQS-data-overview-2018.pdf (accessed on 1 January 2021).
- Jönsson, A.; Gustafsson, Ö.; Axelman, J.; Sundberg, H. Global Accounting of PCBs in the Continental Shelf Sediments. Environ. Sci. Technol. 2003, 37, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Available online: http://havochvatten.diva-portal.org/smash/record.jsf?pid=diva2%3A1368668&dswid=2186 (accessed on 1 January 2021).
- Jönsson, A.; Lindström, M.; Carman, R.; Mörth, C.-M.; Meili, M.; Gustafsson, Ö. Evaluation of the Stockholm archipelago sediments, northwestern Baltic Sea proper, as a trap for freshwater runoff organic carbon. J. Mar. Syst. 2005, 56, 167–178. [Google Scholar] [CrossRef]
- Available online: https://www.stockholmvattenochavfall.se/globalassets/pdf1/rapporter/svoa/svoa-mr-2019_avlopp_20mb658.pdf (accessed on 1 January 2021).
- Havs-och Vattenmyndigheten; Vattenmyndigheterna. Länsstyrelserna VISS Strömmen—WA79755821/SE591920-180800. Available online: https://viss.lansstyrelsen.se/Waters.aspx?waterMSCD=WA79755821 (accessed on 13 January 2022). (In Swedish).
- Lücke, J. Undersökningar i Stockholms skärgård 2018. In Vattenkemi, Plankton och Bottenfauna; Stockholm Vatten och Avfall: Stockholm, Sweden, 2019. (In Swedish) [Google Scholar]
- Lindström, M.; Jonsson, A.; Brolin, A.A.; Håkanson, L. Heavy metal sediment load from the city of Stockholm. Water Air Soil Pollut. Focus 2001, 1, 103–118. [Google Scholar] [CrossRef]
- Jönsson, A. Ni, Cu, Zn, Cd and Pb in Sediments in the Citycentre of Stockholm, Sweden Origins, Deposition Rates and Bioavailability; IVl Report B2013; IVL Swedish Environmental Research Institute: Stockholm, Sweden, 2011. [Google Scholar]
- Benjamin, M.M.; Honeyman, B.D. Global biogeochemical cycle. In Global Biogeochemical Cycle; Butcher, S.S., Charlson, R.J., Orians, G.H., Wolfe, G.V., Eds.; Academic Press: London, UK, 1998. [Google Scholar]
- Turner, A. Trace-metal partitioning in estuaries: Importance of salinity and particle concentration. Mar. Chem. 1996, 54, 27–39. [Google Scholar] [CrossRef]
- European Commision. Revision of EC Sludge Directive Challenges Landspreading; ENDS Report 299; European Commision: Brussels, Belgium, 1999. [Google Scholar]
- Havs- och Vattenmyndigheten. Havs- och Vattenmyndighetens Föreskrifter om Klassificering och Miljökvalitetsnormer Avseende Ytvatten; HVMFS; Swedish Agency for Marine and Water Management: Gothenburg, Sweden, 2019. (In Swedish) [Google Scholar]
- Havs- och Vattenmyndigheten. Miljögifter i Vatten—Klassificering av Ytvattenstatus; Vägledning för Tillämpning av HVMFS; Swedish Agency for Marine and Water Management: Gothenburg, Sweden, 2013. (In Swedish) [Google Scholar]
- Available online: https://ec.europa.eu/health/ph_risk/committees/04_scher/docs/scher_o_115.pdf (accessed on 1 January 2021).
- Bodar, C.W.M. Environmental Risk Limits for Zinc; National Institute for Public Health and the Environment: Bilthoven, The Netherlands, 2007; Available online: https//www.rivm.nl/bibliotheek/rapporten/601782004.pdf (accessed on 1 January 2021).
- Visual Minteq, v.3.1; KTH Royal Institute of Technology: Stockholm, Sweden, 2013.
- County Administrative Board of Stockholm, Stockholm, Sweden, Monitoring Data for 2018 for Water Formation Strömmen, Blockhusudden, Part of Saltsjön. Available online: https://miljobarometern.stockholm.se/vatten/kustvatten/strommen/ (accessed on 23 January 2021).
- Miljödata-MVM Datavärdskap Sjöar Och Vattendrag, Samt Datavärdskap Jordbruksmark. Available online: https://miljodata.slu.se/MVM/Search (accessed on 5 November 2021). (In Swedish).
- Crommentuijn, T.; Polder, M.; Sijm, D.; De Bruijn, J.; Van De Plassche, E. Evaluation of the Dutch environmental risk limits for metals by application of the added risk approach. Environ. Toxicol. Chem. 2000, 19, 1692–1701. [Google Scholar] [CrossRef]
- Di Toro, D.M.; Mahony, J.D.; Hansen, D.J.; Berry, W.J. A model of the oxidation of iron and cadmium sulfide in sediments. Environ. Toxicol. Chem. 1996, 15, 2168–2186. [Google Scholar] [CrossRef]
- Peters, A.; Wilson, I.; Merrington, G.; Heijerick, D.; Baken, S. Assessing Compliance of European Fresh Waters for Copper: Accounting for Bioavailability. Bull. Environ. Contam. Toxicol. 2019, 102, 153–159. [Google Scholar] [CrossRef]
- Miljödata-MVM Datavärdskap Sjöar Och Vattendrag, Samt Datavärdskap Jordbruksmark. Available online: https://miljodata.slu.se/MVM/Search (accessed on 5 November 2020). (In Swedish).
- Stockholms Stad. Miljögifter i Ytvatten. Available online: https://miljobarometern.stockholm.se/vatten/kemisk-status-och-miljogifter/miljogifter-i-ytvatten (accessed on 5 November 2021). (In Swedish).
- Kumke, T.; Schoonderwaldt, A.; Kienel, U. Spatial variability of sedimentological properties in a large Siberian lake. Aquat. Sci. 2005, 67, 86–96. [Google Scholar] [CrossRef]
- Håkanson, L.; Jansson, M. Principles of Lake Sedimentology; Springer: Berlin/Heidelberg, Germany, 1983. [Google Scholar]
- Marin Miljöanalys. Rapport Ytgeologi Slussen; Stockholm City Council: Stockholm, Sweden, 2009. (In Swedish) [Google Scholar]
- Ankley, G.T. Evaluation of metal/acid-volatile sulfide relationships in the prediction of metal bioaccumulation by benthic macroinvertebrates. Environ. Toxicol. Chem. 1996, 15, 2138–2146. [Google Scholar] [CrossRef]
- Pesch, C.E.; Hansen, D.J.; Boothman, W.S.; Berry, W.J.; Mahony, J.D. The role of acid-volatile sulfide and interstitial water metal concentrations in determining bioavailability of cadmium and nickel from contaminated sediments to the marine polychaete Neanthes arenaceodentata. Environ. Toxicol. Chem. 1995, 14, 129–141. [Google Scholar] [CrossRef]
- Sundelin, B.; Eriksson, A.-K. Mobility and bioavailability of trace metals in sulfidic coastal sediments. Environ. Toxicol. Chem. 2001, 20, 748–756. [Google Scholar] [CrossRef]
- Kelderman, P.; Osman, A.A. Effect of redox potential on heavy metal binding forms in polluted canal sediments in Delft (The Netherlands). Water Res. 2007, 41, 4251–4261. [Google Scholar] [CrossRef]
- Van Den Berg, G.A.; Meijers, G.G.A.; Van Der Heijdt, L.M.; Zwolsman, J.J.G. Dredging-related mobilisation of trace metals: A case study in The Netherlands. Water Res. 2001, 35, 1979–1986. [Google Scholar] [CrossRef]
- Van Den Berg, G.A.; Loch, J.P.G.; Winkels, H.J. Effect of fluctuating hydrological conditions on the mobility of heavy metals in soils of a freshwater estuary in The Netherlands. Water Air Soil Pollut. 1998, 102, 377–388. [Google Scholar] [CrossRef]
- Van Den Berg, G.A.; Loch, J.P.G.; Zwolsman, J.J.G.; Van Der Heijdt, L.M. Non-steady state behaviour of heavy metals in contaminated freshwater sediments. Water Sci. Technol. 1998, 37, 39–46. [Google Scholar] [CrossRef]
- Mahony, J.D.; Di Toro, D.M.; Gonzalez, A.M.; Curto, M.; Dilg, M.; De Rosa, L.D.; Sparrow, L.A. Partitioning of metals to sediment organic carbon. Environ. Toxicol. Chem. 1996, 15, 2187–2197. [Google Scholar] [CrossRef]
- Sánchez-Marín, P.; Aierbe, E.; Lorenzo, J.I.; Mubiana, V.K.; Beiras, R.; Blust, R. Dynamic modeling of copper bioaccumulation by Mytilus edulis in the presence of humic acid aggregates. Aquat. Toxicol. 2016, 178, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Simpson, S.L.; King, C.K. Exposure-Pathway Models Explain Causality in Whole-Sediment Toxicity Tests. Environ. Sci. Technol. 2005, 39, 837–843. [Google Scholar] [CrossRef] [PubMed]
- King, C.; Simpson, S.; Smith, S.; Stauber, J.; Batley, G. Short-term accumulation of Cd and Cu from water, sediment and algae by the amphipod Melita plumulosa and the bivalve Tellina deltoidalis. Mar. Ecol. Prog. Ser. 2005, 287, 177–188. [Google Scholar] [CrossRef]
- Persson, J.; Jonsson, P. Historical Development of Laminated Sediments—An Approach to Detect Soft Sediment Ecosystem Changes in the Baltic Sea. Mar. Pollut. Bull. 2000, 40, 122–134. [Google Scholar] [CrossRef]
- Eckhéll, J.; Jonsson, P.; Meili, M.; Carman, R. Storm influence on the accumulation and lamination of sediments in deep areas of the Northwestern Baltic proper. AMBIO A J. Hum. Environ. 2000, 29, 238–245. [Google Scholar] [CrossRef]
- Jönsson, A.; Carman, R. Distribution of PCBs in sediment from different bottom types and water depths in Stockholm archipelago, Baltic Sea. AMBIO A J. Hum. Environ. 2000, 29, 277–281. [Google Scholar] [CrossRef]
- Leonardsson, K.; Blomqvist, M.; Rosenberg, R. Reducing spatial variation in environmental assessment of marine benthic fauna. Mar. Pollut. Bull. 2016, 104, 129–138. [Google Scholar] [CrossRef]
- Available online: https://pub.epsilon.slu.se/12590/7/herbert_r_etal_gamla_pb_150908.pdf (accessed on 1 December 2021).
- Ragnvaldsson, D. Bakgrundshalter av Metaller i Svenska Sjöar Och Vattendrag—Sammanställning av Halter för Ett Urval av Metaller Jämfört Med Bedömningsgrunder i HaVs Föreskrift; Report 2015:4; Envix: Umeå, Sweden, 2017. (In Swedish) [Google Scholar]
- Wahlgren, C.H.; Schoning, K.; Tenne, M.; Hansen, L.M. Stockholmsområdets Berggrund, Jordarter, Geologiska Utveckling och Erfarenheter Från Infrastrukturprojekt; Report 2018:8; SGU Geological Survey of Sweden: Uppsala, Sweden, 2018. [Google Scholar]
- Schoning, K.; Lundqvist, L. Hållbar Ballastförsörjning–förutsättningar i Stockholms och Uppsala Län; Report 2018:09; SGU Geological Survey of Sweden: Uppsala, Sweden, 2018. (In Swedish) [Google Scholar]
- Landner, L.; Reuther, R. Metals in Society and in the Environment. In A Critical Review of Current Knowledge on Fluxes, Speciation, Bioavailability and Risk for Adverse Effects of Copper, Chromium, Nickel and Zinc; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Bielmyer, G.K.; Bullington, J.B.; Decarlo, C.A.; Chalk, S.J.; Smith, K. The effects of salinity on acute toxicity of zinc to two euryhaline species of fish, Fundulus heteroclitus and Kryptolebias marmoratus. Integr. Comp. Biol. 2012, 52, 753–760. [Google Scholar] [CrossRef]
- Brown, R.J.; Galloway, T.S.; Lowe, D.; Browne, M.A.; Dissanayake, A.; Jones, M.B.; Depledge, M.H. Differential sensitivity of three marine invertebrates to copper assessed using multiple biomarkers. Aquat. Toxicol. 2004, 66, 267–278. [Google Scholar] [CrossRef]
- Figueira, E.; Branco, D.; Antunes, S.C.; Gonçalves, F.; Freitas, R. Are metallothioneins equally good biomarkers of metal and oxidative stress? Ecotoxicol. Environ. Saf. 2012, 84, 185–190. [Google Scholar] [CrossRef]
- Kumar, R.; Pradhan, A.; Khan, F.A.; Lindström, P.; Ragnvaldsson, D.; Ivarsson, P.; Olsson, P.-E.; Jass, J. Comparative analysis of stress induced gene expression in Caenorhabditis elegans following exposure to environmental and lab reconstituted complex metal mixture. PLoS ONE 2015, 10, e0132896. [Google Scholar] [CrossRef]


| Percentile | Freshwater | Estuarine Water | Marine Water | |||
|---|---|---|---|---|---|---|
| Log Kd | Kd (L/kg) | Log Kd | Kd (L/kg) | Log Kd | Kd (L/kg) | |
| 10th | 3.8 | 5.8 | 4.2 | 15.5 | 4.6 | 38.0 |
| 50th | 4.5 | 30.2 | 4.8 | 56.2 | 5.1 | 131.8 |
| 90th | 5.3 | 194.2 | 5.4 | 263.0 | 5.7 | 457.1 |
| Month (2018) | pH | DOC (mg/L) | Ca2+ (mg/L) | Cudiss (µg/L) | Zndiss (µg/L) |
|---|---|---|---|---|---|
| January | 7.8 | 6.6 | 27 | 1.5 | 0.8 |
| February | 7.9 | 6.9 | 23 | 1.7 | 1.7 |
| March | 7.8 | 6.0 | 22 | 1.9 | 1.3 |
| April | 7.9 | 7.1 | 28 | 1.7 | 1.3 |
| May | 8.2 | 6.2 | 32 | 1.7 | 3.4 |
| June | 8.5 | 5.2 | 51 | 1.6 | 1.1 |
| July | 8.3 | 4.9 | 70 | 1.8 | 2.1 |
| August | 8.1 | 5.3 | 74 | 1.6 | 2.4 |
| September | 7.9 | 4.5 | 78 | 0.7 | 3.2 |
| October | 7.8 | 4.5 | 77 | 1.3 | 4.4 |
| November | 7.7 | 4.4 | 91 | 1.4 | 4.5 |
| December | 7.7 | 4.5 | 80 | 1.5 | 3.8 |
| Average 2018 | 8.0 | 5.7 | 54.4 | 1.5 | 2.5 |
| PSU | Cl(I) | SO4(II) | Br(I) | F(I) | Br(I) | Na(I) | Mg(II) | Ca(II) | K(I) | Sr(II) | HCO3(II) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.25 | 137.6 | 19.30 | 0.48 | 0.009 | 0.03 | 76.6 | 9.20 | 2.90 | 2.80 | 0.06 | 1.00 |
| 5.23 | 2928.4 | 410.4 | 10.2 | 0.20 | 0.70 | 1629.6 | 195.2 | 62.4 | 60.4 | 1.20 | 21.5 |
| (a) Cu | Cudiss (Table 1) (µg/L) | CuBA (Cufree ions + Labile Complexes, VM) (µg/L) | CuBA (BM) (Cudiss µg/L) | General EQS (CuBA Fresh Water (µg/L, AF = 2) | CuBA Coastal Waters (CuBA = Cudiss/DOC/2)0.6136) | SWE EQS Coastal Water (Baltic Sea) CuBA (µg/L, AF = 6) | RCR Cu (RCR = cal. CuBA/SWE EQS Coastal Waters) | RCR Cu (RCR = CuBA VM/SWE Limit Value (0.87 µg/L)) | RCR Cu (BM) (RCR = CuBA/ General EQS |
| Strömmen-Saltsjön | 1.50 | 0.15 | 0.11 | 0.50 | 0.81 | 0.87 | 0.93 | 0.13 | 0.22 |
| Status class. 2018 | Good RCR < 1 no risk | RCR << 1 no risk | RCR << 1 no risk | ||||||
| (b) Zn | Zndiss (Table 1) (µg/L) | ZnBA (Znfree ions +Labile Complexes (VM) (µg/L) | ZnBA (BM) (Zndiss µg/L) | General EQS ZnBA Fresh Water (µg/L, AF = 2) | Local EQS (1.1 µg/L for Baltic Sea, AF = 5.5) + Ambient Background Conc. (2 µg/L) | RCR Zn Coastal Water (RCR = Calc. Zndiss/Calc. Local EQS) (µg/L) | RCR Zn Calc. for FreshWater (RCR = ZnBA VM/SWE General EQS) | RCR Zn ZnBA (BM; RCR = ZnBA /SWE EQS) | |
| Strömmen-Saltsjön | 2.50 | 0.42 | 0.56 | 5.50 | 3.1 | 0.81 | 0.08 | 0.1 | |
| Status class. 2018 | Good RCR < 1 no risk | RCR << 1 no risk | RCR << 1 no risk | ||||||
| Cu/Zn | RCR Coastal Waters Cu | RCR Freshwater Cu | RCR Coastal Waters Zn | RCR Freshwater Zn |
|---|---|---|---|---|
| Mean (n) | 1.21 (11) | 0.16 (43) | 2.16 (11) | 0.13 (39) |
| Median | 1.11 (11) | 0.16 (43) | 0.82 (11) | 0.08 (39) |
| Max | 1.95 | 0.37 | 6.47 | 0.55 |
| Min | 0.91 | 0.01 | 0.13 | 0.01 |
| Average 2018 (n) | 0.94 (3) | 0.14 (3) | 0.62 (3) | 0.13 (11) |
| Median 2018 (n) | 0.95 (3) | 0.14 (3) | 0.48 (3) | 0.08 (11) |
| Cu/Zn | 5th (µg/L) | 50th (Median) (µg/L) | 95th (µg/L) | 98th (µg/L) |
|---|---|---|---|---|
| Cu (lakes, n = 1851) | 0.2 | 0.7 | 4.0 | 6.8 |
| Cu (rivers, n = 13,874) | 0.1 | 0.7 | 2.9 | 4.0 |
| Cu (Ekoln-Vreta, n = 23) | 1.8 | 2.2 | 3.1 | 3.2 |
| Zn (lakes, n = 1851) | 0.7 | 4.7 | 14.0 | 19.0 |
| Zn (rivers, n = 12,266) | 0.5 | 3.1 | 12.0 | 19.0 |
| Zn (Ekoln-Vreta, n = 23) | 0.5 | 1.2 | 5.5 | 10.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ragnvaldsson, D.; Herting, G.; Jönsson, A.; Odnevall, I. Applying Generic Water Quality Criteria to Cu and Zn in a Dynamic Aquatic Environment—The Case of the Brackish Water Formation Strömmen-Saltsjön. Water 2022, 14, 847. https://doi.org/10.3390/w14060847
Ragnvaldsson D, Herting G, Jönsson A, Odnevall I. Applying Generic Water Quality Criteria to Cu and Zn in a Dynamic Aquatic Environment—The Case of the Brackish Water Formation Strömmen-Saltsjön. Water. 2022; 14(6):847. https://doi.org/10.3390/w14060847
Chicago/Turabian StyleRagnvaldsson, Daniel, Gunilla Herting, Anders Jönsson, and Inger Odnevall. 2022. "Applying Generic Water Quality Criteria to Cu and Zn in a Dynamic Aquatic Environment—The Case of the Brackish Water Formation Strömmen-Saltsjön" Water 14, no. 6: 847. https://doi.org/10.3390/w14060847
APA StyleRagnvaldsson, D., Herting, G., Jönsson, A., & Odnevall, I. (2022). Applying Generic Water Quality Criteria to Cu and Zn in a Dynamic Aquatic Environment—The Case of the Brackish Water Formation Strömmen-Saltsjön. Water, 14(6), 847. https://doi.org/10.3390/w14060847






