Biodiversity Studies for Sustainable Lagoon: Thermophilic and Tropical Fish Species vs. Endemic Commercial Species at Mellah Lagoon (Mediterranean, Algeria)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Study and Fish Collection
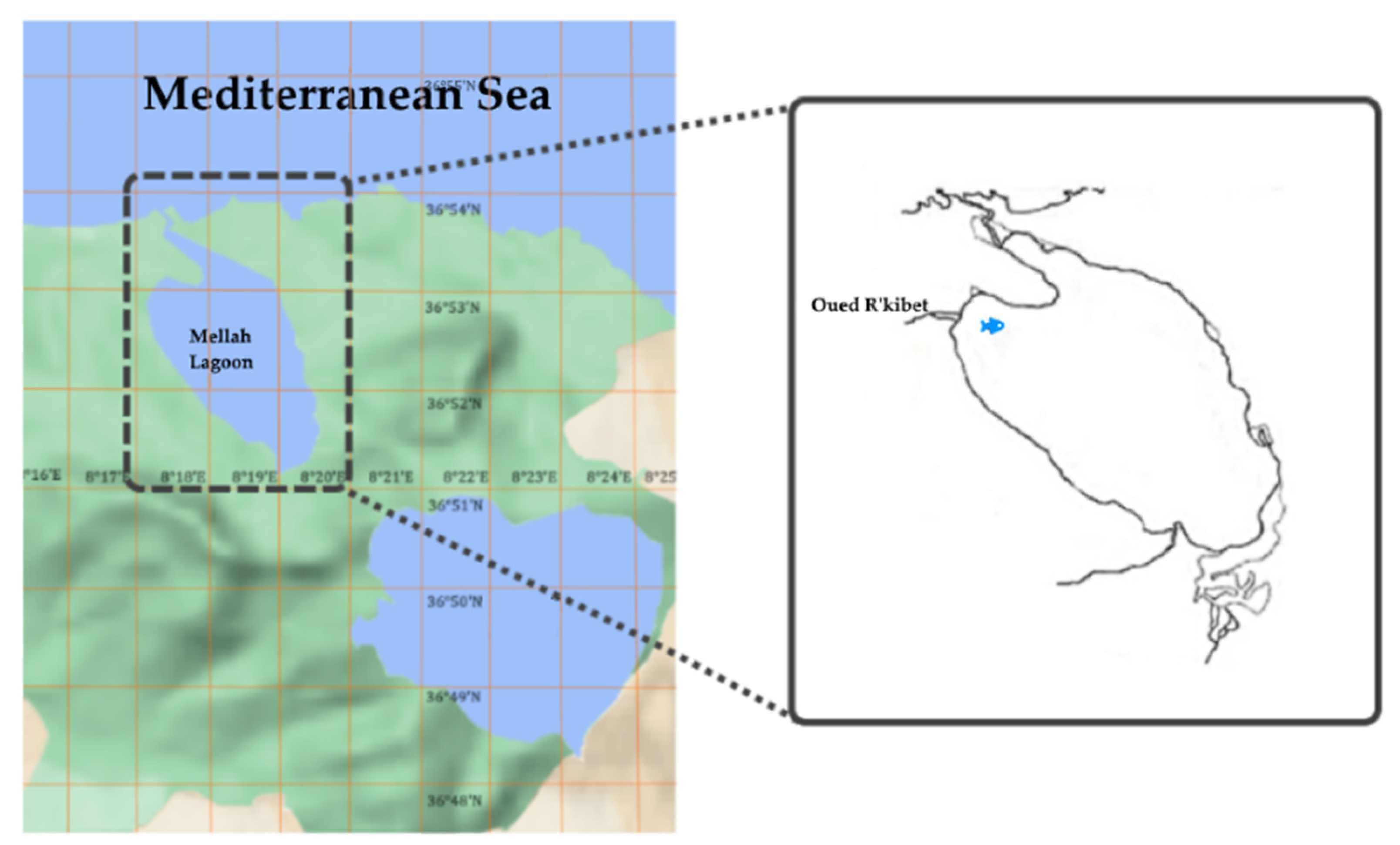
2.2. Fish Discrimination in Mellah Lagoon
2.3. Mediterranean Lagoon Fauna, Fauna Identification, Bioinvasion, and Lagoon Fauna Socio-Economic Value: Data Sources
3. Results
3.1. Fish Biodiversity at Mellah Lagoon
3.1.1. Morphological Identification

3.1.2. Barcoding
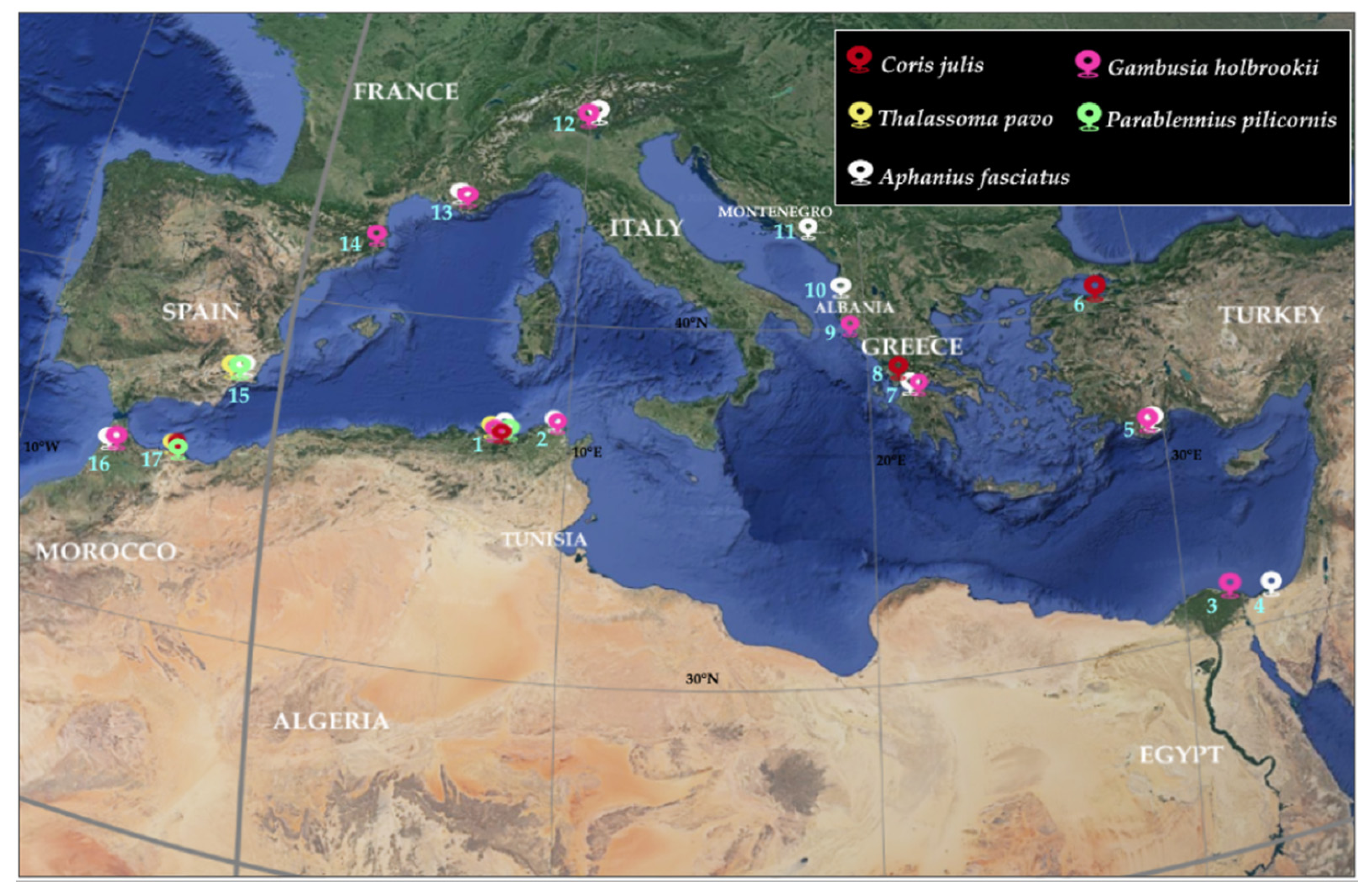
3.2. Patterns of Non-Indigenous Mellah Lagoon Fish in Mediterranean Lagoons
3.3. Mediterranean Lagoon Fish and Socio-Economical Use Analysis
4. Discussion
4.1. An Updated Overview on Fish Biodiversity at Mellah Lagoon
4.2. Pattern of Thermophilic and Tropical Fish Mellah Lagoon in Other Mediterranean Lagoons
4.3. Mediterranean Lagoon Sustainability
4.4. Mediterranean Lagoon Fish Conservation and Management: Thermophilic and Tropical Species vs. Endemic Commercial Species
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sorrell, S. Reducing energy demand: A review of issues, challenges and approaches. Renew. Sustain. Energy Rev. 2015, 47, 74–82. [Google Scholar] [CrossRef] [Green Version]
- Tollefson, J. How hot will Earth get by 2100? Nature 2020, 580, 443–445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guerriero, G.; Bassem, S.M.; Khalil, W.K.B.; Temraz, T.A.; Ciarcia, G.; Abdel-Gawad, F.K. Temperature changes and marine fish species (Epinephelus coioides and Sparus aurata): Role of oxidative stress biomarkers in toxicological food studies. Emir. J. Food Agric. 2018, 30, 205–211. [Google Scholar] [CrossRef] [Green Version]
- FAO. The State of the World’s Biodiversity for Food and Agriculture. In FAO Commission on Genetic Resources for Food and Agriculture Assessments; Bélanger, J., Pilling, D., Eds.; FAO: Rome, Italy, 2019; p. 576. [Google Scholar]
- Gentilucci, M.; Moustafa, A.; Abdel-Gawad, F.; Mansour, S.; Coppola, M.; Caserta, L.; Inglese, S.; Pambianchi, G.; Guerriero, G. Advances in Egyptian Mediterranean Coast Climate Change Monitoring. Water 2021, 13, 1870. [Google Scholar] [CrossRef]
- Dangi, T.B.; Jamal, T. An integrated approach to “sustainable community-based tourism”. Sustainability 2016, 8, 475. [Google Scholar] [CrossRef] [Green Version]
- Corrales, X.; Coll, M.; Ofir, E.; Heymans, J.J.; Steenbeek, J.; Goren, M.; Edelist, D.; Gal, G. Future scenarios of marine resources and ecosystem conditions in the Eastern Mediterranean under the impacts of fishing, alien species and sea warming. Sci. Rep. 2018, 8, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Servello, G.; Andaloro, F.; Azzurro, E.; Castriota, L.; Catra, M.; Chiarore, A.; Crocetta, F.; D’Alessandro, M.; Denitto, F.; Froglia, C.; et al. Marine alien species in Italy: A contribution to the implementation of descriptor D2 of the marine strategy framework directive. Mediterr. Mar. Sci. 2019, 20, 1–48. [Google Scholar] [CrossRef] [Green Version]
- Cataudella, S.; Crosetti, D.; Massa, F. Mediterranean coastal lagoons: Sustainable management and interactions among aquaculture, capture fisheries and the environment. In Studies and Reviews General Fisheries Commission for the Mediterranean; FAO: Rome, Italy, 2015; pp. 1–293. [Google Scholar]
- Newton, A.; Brito, A.C.; Icely, J.D.; Derolez, V.; Clara, I.; Angus, S.; Schernewski, G.; Inácio, M.; Lillebø, A.I.; Sousa, A.I.; et al. Assessing, quantifying and valuing the ecosystem services of coastal lagoons. J. Nat. Conserv. 2018, 44, 50–65. [Google Scholar] [CrossRef]
- Tsikliras, A.; Dinouli, A.; Tsiros, V.-Z.; Tsalkou, E. The Mediterranean and Black Sea Fisheries at Risk from Overexploitation. PLoS ONE 2015, 10, e0121188. [Google Scholar] [CrossRef] [Green Version]
- Guerriero, G.; Bassem, S.M.; Abdel Gawad, F.K. Biological responses of white sea bream (Diplodus sargus, Linnaeus 1758) and sardine (Sardine pilchardus, Walbaum 1792) exposed to heavy metal contaminated water. Emir. J. Food Agric. 2018, 30, 688–694. [Google Scholar] [CrossRef]
- Vassalli, Q.A.; Caccavale, F.; Avagnano, S.; Murolo, A.; Guerriero, G.; Fucci, L.; Ausió, J.; Piscopo, M. New Insights into Protamine-Like Component Organization inMytilus galloprovincialis’ Sperm Chromatin. DNA Cell Biol. 2015, 34, 162–169. [Google Scholar] [CrossRef]
- Scalici, M.; Traversetti, L.; Spani, F.; Malafoglia, V.; Colamartino, M.; Persichini, T.; Cappello, S.; Mancini, G.; Guerriero, G.; Colasanti, M. Shell fluctuating asymmetry in the sea-dwelling benthic bivalve Mytilus galloprovincialis (Lamarck, 1819) as morphological markers to detect environmental chemical contamination. Ecotoxicology 2017, 26, 396–404. [Google Scholar] [CrossRef]
- Occhipinti-Ambrogi, A.; Savini, D. Biological invasions as a component of global change in stressed marine ecosystems. Mar. Pollut. Bull. 2003, 46, 542–551. [Google Scholar] [CrossRef]
- Skutnik, J.E.; Otieno, S.; Khoo, S.K.; Strychar, K.B. Examining the Effect of Heat Stress on Montastraea cavernosa (Linnaeus 1767) from a Mesophotic Coral Ecosystem (MCE). Water 2020, 12, 1303. [Google Scholar] [CrossRef]
- Dobson, K.L.; Ferrier-Pagès, C.; Saup, C.M.; Grottoli, A.G. The Effects of Temperature, Light, and Feeding on the Physiology of Pocillopora damicornis, Stylophora pistillata, and Turbinaria reniformis Corals. Water 2021, 13, 2048. [Google Scholar] [CrossRef]
- Gentilucci, M.; Parisi, C.; Coppola, M.R.; Majdoubi, F.-Z.; Madonna, A.; Guerriero, G. Influence of Mediterranean Sea Temperature Increase on Gaeta Gulf (Tyrrhenian Sea) Biodiversity. Proc. Zool. Soc. 2020, 74, 91–103. [Google Scholar] [CrossRef]
- Boumendjel, M.; Taibi, F.; Hennouni, N.; Samar, M.F.; Abdesselem, A.; Slimane, B.; Alloui, B. Ecologie et polymormorphisme de l’Aphanius fasciatus, Valenciennes 1821 (Cyprinodontidae), dans le lac Bleu, parc national d’El-Kala (Nord-Est, Algerie). Sci. Technologie. C Biotechnol. 2015, 41, 9–21. [Google Scholar]
- Parisi, C.; Guerriero, G. Antioxidative Defense and Fertility Rate in the Assessment of Reprotoxicity Risk Posed by Global Warming. Antioxidants 2019, 8, 622. [Google Scholar] [CrossRef] [Green Version]
- El Mahrad, B.; Abalansa, S.; Newton, A.; Icely, J.D.; Snoussi, M.; Kacimi, I. Social-Environmental Analysis for the Management of Coastal Lagoons in North Africa. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- Chaoui, L.; Kara, M.H.; Faure, É.; Quignard, J.P. L’ichtyofaune de la lagune du Mellah (Algérie Nord-Est): Diversité, production et analyse des captures commerciales. Cybium 2006, 30, 123–132. [Google Scholar]
- Embarek, R.; Amara, R.; Kara, M.H. Fish assemblage structure in shallow waters of the Mellah Lagoon (Algeria): Seasonal and spatial distribution patterns and relation to environmental parameters. Acta Ichthyol. Piscat. 2017, 47, 133–144. [Google Scholar]
- Libralato, S.; Caccin, A.; Pranovi, F. Modelling species invasions using thermal and trophic niche dynamics underclimate change. Front. Mar. Sci. 2015, 2, 29. [Google Scholar] [CrossRef] [Green Version]
- Tramice, A.; Trifuoggi, M.; Ahmad, M.F.; Lam, S.S.; Iodice, C.; Velotto, G.; Giarra, A.; Inglese, S.; Cupo, A.; Guerriero, G.; et al. Comparative Fatty Acid Profiling of Edible Fishes in Kuala Terengganu, Malaysia. Foods 2021, 10, 2456. [Google Scholar] [CrossRef] [PubMed]
- Inkscape. Available online: https://inkscape.org/it/ (accessed on 2 May 2020).
- Di Finizio, A.; Guerriero, G.; Russo, G.L.; Ciarcia, G. Identification of Gadoid Species (Pisces, Gadidae) by Sequencing and PCR–RFLP Analysis of Mitochondrial 12S and 16S rRNA Gene Fragments. Eur. Food Res. Technol. 2007, 225, 337–344. [Google Scholar] [CrossRef]
- Madonna, A.; Alwany, M.A.; Rabbito, D.; Trocchia, S.; Labar, S.; Abdel-Gawad, F.K.; Ciarcia, G. Caves Biodiversity in the Marine Area of Riviera d’Ulisse Regional Park, Italy: Grotta del Maresciallo overview. J. Biodivers. Endanger. Species 2015, 3, 2. [Google Scholar] [CrossRef] [Green Version]
- GenBank. Available online: http://www.ncbi.nlm.nih.gov (accessed on 31 May 2020).
- Mount, D.W. Using a FASTA Sequence Database Similarity Search. Cold Spring Harb. Protoc. 2007, 2007, pdb.top16. [Google Scholar] [CrossRef]
- WoRMS—World Register of Marine Species. Available online: https://www.marinespecies.org/about.php (accessed on 10 May 2020).
- FishBase: A Global Information System on Fishes. Available online: http://www.fishbase.org/home.htm (accessed on 21 August 2020).
- FAO. Available online: http://www.fao.org/about/it/ (accessed on 28 November 2021).
- IUCN. Termophilic Species. Available online: http://www.iucn.it/ (accessed on 19 October 2021).
- Wikipedia. Available online: http://www.wikipedia.com (accessed on 18 October 2021).
- Benessere. Available online: http://www.http://www.benessere.com/alimentazione/ (accessed on 13 October 2021).
- AIAM. Available online: http://www.aiamitalia.it (accessed on 16 October 2021).
- IRCCS HUMANITAS. Available online: https://www.humanitas.it/ (accessed on 11 November 2021).
- Graphical Paint. Available online: https://paint-net.it.uptodown.com/windows (accessed on 23 October 2021).
- Icons. Available online: https://www.iconfinder.com/icons/2216357/location_map_map_pin_marker_pin_position_icon (accessed on 23 October 2021).
- Ayaz, A.; Özekinci, U.; Altınağaç, U.; Özen, Ö. An investigation on ghost fishing of circular fish traps used in Turkey. E.U. J. Fish. Aquat. Sci. 2006, 23, 351–354. [Google Scholar]
- Costa, M.J.; Cabral, H.N.; Drake, P.; Economou, A.N.; Fernandez-Delgado, C.; Gordo, L.; Marchand, J.; Thiel, R. Recruitment and Production of Commercial Species in Estuaries. In Fish in Estuaries; Elliot, M., Hemingway, K., Eds.; Blackwell Science: London, UK, 2002; pp. 54–123. [Google Scholar]
- Selfati, M.; El Ouamari, N.; Franco, A.; Lenfant, P.; Lecaillon, G.; Mesfioui, A.; Boissery, P.; Bazairi, H. Fish assemblages of the Marchica lagoon (Mediterranean, Morocco): Spatial patterns and environmental drivers. Reg. Stud. Mar. Sci. 2019, 32, 100896. [Google Scholar] [CrossRef]
- Zarrad, R.; Alemany, F.; Rodriguez, J.-M.; Jarboui, O.; Lopez-Jurado, J.-L.; Balbin, R. Influence of summer conditions on the larval fish assemblage in the eastern coast of Tunisia (Ionian Sea, Southern Mediterranean). J. Sea Res. 2013, 76, 114–125. [Google Scholar] [CrossRef]
- Akel, E.K. A comparative study on the catch characteristics of purseseine operating during the daytime in Abu-Qir and El-Mex Bays, Alexandria (Egypt). Egypt. J. Aquat. Res. 2005, 31, 357–372. [Google Scholar]
- Akel, E.H.; Karachle, P.K. The marine ichthyofauna of Egypt. Egypt. J. Aquat. Biol. Fish. 2017, 21, 81–116. [Google Scholar] [CrossRef] [Green Version]
- Soldo, A.; Bakiu, R. Checklist of marine fishes of Albania. Acta Adriat. Int. J. Mar. Sci. 2021, 62, 63–73. [Google Scholar] [CrossRef]
- Guidetti, P.; Terlizzi, A.; Fraschetti, S.; Boero, F. Spatio-temporal variability in fish assemblages associated with coralligenous formations in south eastern Apulia (SE Italy). Ital. J. Zool. 2002, 69, 325–331. [Google Scholar] [CrossRef]
- Lejeune, P. The effect of local stock density on social behavior and sex change in the Mediterranean labrid Coris julis. J. Appl. Phycol. 1987, 18, 135–141. [Google Scholar] [CrossRef]
- Bray, R.A.; Bartoli, P. A redescription of Lepidauchen stenostoma Nicoll, 1913 (Digenea), and a reassessment of the status of the genus Lepidauchen Nicoll, 1913. Syst. Parasitol. 1996, 33, 167–176. [Google Scholar] [CrossRef]
- Crec’Hriou, R.; Alemany, F.; Roussel, E.; Chassanite, A.; Marinaro, J.Y.; Mader, J.; Rochel, E.; Planes, S. Fisheries replenishment of early life taxa: Potential export of fish eggs and larvae from a temperate marine protected area. Fish. Oceanogr. 2010, 19, 135–150. [Google Scholar] [CrossRef]
- Pérez-Ruzafa, A.; Quispe-Becerra, J.I.; García-Charton, J.A.; Marcos, C. Composition, structure and distribution of the ichthyoplankton in a Mediterranean coastal lagoon. J. Fish Biol. 2004, 64, 202–218. [Google Scholar] [CrossRef]
- Milazzo, M.; Palmeri, A.; Falcón, J.M.; Badalamenti, F.; Garcìa-Charton, J.A.; Sinopoli, M.; Brito, A. Vertical distribution of two sympatric labrid fishes in the Western Mediterranean and Eastern Atlantic rocky subtidal: Local shore topography does matter. Mar. Ecol. 2011, 32, 521–531. [Google Scholar] [CrossRef]
- Karachle, P.K.; Stergiou, K.I. An update on the feeding habits of fish in the Mediterranean Sea (2002-2015). Mediterr. Mar. Sci. 2017, 18, 43–52. [Google Scholar] [CrossRef] [Green Version]
- Papadakis, O.; Tsirintanis, K.; Lioupa, V.; Katsanevakis, S. The neglected role of omnivore fish in the overgrazing of Mediterranean rocky reefs. Mar. Ecol. Prog. Ser. 2021, 673, 107–116. [Google Scholar] [CrossRef]
- Azzurro, E.; Pais, A.; Consoli, P.; Andaloro, F. Evaluating day–night changes in shallow Mediterranean rocky reef fish assemblages by visual census. Mar. Biol. 2007, 151, 2245–2253. [Google Scholar] [CrossRef]
- Encarnação, J.; Morais, P.; Baptista, V.; Cruz, J.; Teodósio, M.A. New Evidence of Marine Fauna Tropicalization off the Southwestern Iberian Peninsula (Southwest Europe). Diversity 2019, 11, 48. [Google Scholar] [CrossRef] [Green Version]
- Sala, E. Fish predators and scavengers of the sea urchin Paracentrotus lividus in protected areas of the north-west Mediterranean Sea. Mar. Biol. 1997, 129, 531–539. [Google Scholar] [CrossRef]
- Kraïem, M.M.; Chouba, L.; Ramdani, M.; Ahmed, M.H.; Thompson, J.R.; Flower, R.J. The fish fauna of three North African lagoons: Specific inventories, ecological status and production. Hydrobiologia 2009, 622, 133–146. [Google Scholar] [CrossRef]
- Sellami, R.; Chaouachi, B.; Hassine, O.K.B. Impacts anthropiques et climatiques sur la diversité ichtyque d’une lagune méditerranéenne (Ichkeul, Tunisie). Cybium 2010, 34, 5–10. [Google Scholar]
- Lotan, R.; Ben-Tuvia, A. Distribution and reproduction of killifish Aphanius dispar and A. fasciatus and their hybrids in the Bardawil Lagoon on the Mediterranean coast of Sinai, Egypt. Isr. J. Zool. 1996, 42, 203–213. [Google Scholar]
- Acarli, D.; Kara, A.; Bayhan, B. Length-weight relations for 29 fish species from Homa Lagoon, Aegean sea, Turkey. Acta Ichthyol. Piscat. 2014, 44, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Innal, D. Distribution and length-weight relationships of the Mediterranean banded killifish (Aphanius fasciatus) in Mediterranean brackish water systems of Turkey. Indian J. Geo-Mar. Sci. 2020, 49, 553–558. [Google Scholar]
- Nicolaidou, A.; Reizopoulou, S.; Koutsoubas, D.; Orfanidis, S.; Kevrekidis, T. Biological components of Greek lagoonal ecosystems: An overview. Mediterr. Mar. Sci. 2005, 6, 31–50. [Google Scholar] [CrossRef] [Green Version]
- Kottelat, M.; Barbieri, R.H.; Stoumboudi, M.T. Aphanius almiriensis, a new species of toothcarp from Greece (Teleostei: Cyprinodontidae). Rev. Suisse Zool. 2007, 114, 13–31. [Google Scholar] [CrossRef]
- Facca, C.; Cavraro, F.; Franzoi, P.; Malavasi, S. Lagoon resident fish species of conservation interest according to the habitat directive (92/43/CEE): A review on their potential use as ecological indicator species. Water 2020, 12, 2059. [Google Scholar] [CrossRef]
- Peja, N.; Vaso, A.; Miho, A.; Rakaj, N.; Crivelli, A.J. Characteristics of Albanian lagoons and their fisheries. Fish. Res. 1996, 27, 215–225. [Google Scholar] [CrossRef]
- Franco, A.; Pérez-Ruzafa, A.; Drouineau, H.; Franzoi, P.; Koutrakis, E.; Lepage, M.; Verdiell-Cubedo, D.; Bouchoucha, M.; López-Capel, A.; Riccato, F.; et al. Assessment of fish assemblages in coastal lagoon habitats: Effect of sampling method. Estuarine Coast. Shelf Sci. 2011, 112, 115–125. [Google Scholar] [CrossRef]
- Valdesalici, S.; Langeneck, J.; Barbieri, M.; Castelli, A.; Maltagliati, F. Distribution of natural populations of the killifish Aphanius fasciatus (Valenciennes, 1821) (Teleostei: Cyprinodontidae) in Italy: Past and current status, and future trends. Ital. J. Zool. 2015, 82, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Zucchetta, M.; Capoccioni, F.; Franzoi, P.; Ciccotti, E.; Leone, C. Fish Response to Multiple Anthropogenic Stressors in Mediterranean Coastal Lagoons: A Comparative Study of the Role of Different Management Strategies. Water 2021, 13, 130. [Google Scholar] [CrossRef]
- Kalogianni, E.; Giakoumi, S.; Zogaris, S.; Chatzinikolaou, Y.; Zimmerman, B.; Economou, A.N. Current distribution and ecology of the critically endangered Valencia letourneuxi in Greece. Biologia 2010, 65, 128–139. [Google Scholar] [CrossRef] [Green Version]
- Locatello, L.; Rasotto, M.B.; Adriaenssens, B.; Pilastro, A. Ejaculate traits in relation to male body size in the eastern mosquitofish Gambusia holbrooki. J. Fish Biol. 2008, 73, 1600–1611. [Google Scholar] [CrossRef]
- Le Direach, L.; Astruch, P.; Changeux, T.; Moussy, F.; Jehl, C.; Brodu, N.; Schohn, T. Favoring exchanges between the sea and the lagoons: A necessary support for the restoration of the functional role as fish nursery in the saltmarshes of Hyères (Provence, France). Vie Et Milieu/Life Environ. 2020, 70, 1–11. [Google Scholar]
- Srean, P.; Almeida, D.; Rubio-Gracia, F.; Luo, Y.; García-Berthou, E. Effects of size and sex on swimming performance and metabolism of invasive mosquitofish Gambusia holbrooki. Ecol. Freshw. Fish 2017, 26, 424–433. [Google Scholar] [CrossRef]
- Taybi, A.F.; Mabrouki, Y.; Doadrio, I. The occurrence, distribution and biology of invasive fish species in fresh and brackish water bodies of NE Morocco. Biology 2020, 18, 59–73. [Google Scholar] [CrossRef]
- Otero, M.; Cebrian, E.; Francour, P.; Galil, B.; Savini, D. Monitoring Marine Invasive Species in Mediterranean Marine Protected Areas (MPAs): A Strategy and Practical Guide for Managers; IUCN: Malaga, Spain, 2013; pp. 1–136. [Google Scholar]
- Pastor, J.; Francour, P. Occurrence and Distribution Range of Parablennius pilicornis (Actinopterygii: Perciformes: Blenniidae) Along the French Mediterranean Coast. Acta Ichthyol. Piscat. 2010, 40, 179–185. [Google Scholar] [CrossRef] [Green Version]
- Brondizio, E.S.; Settele, J.; Díaz, S.; Ngo, H.T. (Eds.) Global Assessment Report on Biodiversity and Ecosystem Services of the Intergovernmental Science—Policy Platform on Biodiversity and Ecosystem Services; IPBES Secretariat: Bonn, Germany, 2019. [Google Scholar]
- Halisçelik, E.; Soytas, M.A. Sustainable development from millennium 2015 to Sustainable Development Goals 2030. Sustain. Dev. 2019, 27, 545–572. [Google Scholar] [CrossRef]
- Guerriero, G.; Di Finizio, A.; Ciarcia, G. Biological Pollution: Molecular Identification of Non-Native Species in the Central Tyrrhenian Sea. Catrina 2010, 5, 41–47. [Google Scholar]
- Guerriero, G.; Rabbito, D.; Alwany, M.A.; Madonna, A.; Temraz, T.A.; Sulaiman, O.O.; Bassem, S.M.; Trocchia, S.; Abdel-Gawad, F.K.; Ciarcia, G. Fisheries and biodiversity along Mediterranean Sea: Italian and Egyptian coast overview. Euro-Mediterr. J. Environ. Integr. 2017, 2, 16. [Google Scholar] [CrossRef] [Green Version]
- El-Haddad, K.M.; Ali, A.-H.A.M.; Abdel-Rahman, M.S.; Mohammed, S.-Z.; Abdel-Gawad, F.; Guerriero, G.; Temraz, T.A. Herbivorous fish of Sinai marine protected areas (Gulf of Aqaba): Structure baseline for potential thermal stress impact management. BioRxiv 2021, 9, 460804. [Google Scholar]
- Azzurro, E.; Sbragaglia, V.; Cerri, J.; Bariche, M.; Bolognini, L.; Ben Souissi, J.; Moschella, P. Climate change, biological invasions, and the shifting distribution of Mediterranean fishes: A large-scale survey based on local ecological knowledge. Glob. Change Biol. 2019, 25, 2779–2792. [Google Scholar] [CrossRef]
- Yarra, A.N.; Magoulick, D.D. Modelling effects of invasive species and drought on crayfish extinction risk and population dynamics. Aquat. Conserv. Mar. Freshw. Ecosyst. 2018, 29, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Everett, R.A. Patterns and pathways of biological invasions. Trends Ecol. Evol. 2000, 15, 177–178. [Google Scholar] [CrossRef]
- Charles, H.; Dukes, J.S. Impacts of Invasive Species on Ecosystem Services. In Biological Invasions; Nentwig, W., Ed.; Part of the Ecological Studies Book Series; Springer: Berlin/Heidelberg, Germany, 2008; pp. 217–237. [Google Scholar]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.-I.; Knowler, D.J.; Lévêque, C.; Naiman, R.J.; Prieur-Richard, A.-H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater biodiversity: Importance, threats, status and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef]
- Miqueleiz, I.; Bohm, M.; Ariño, A.H.; Miranda, R. Assessment gaps and biases in knowledge of conservation status of fishes. Aquat. Conserv. Mar. Freshw. Ecosyst. 2019, 30, 225–236. [Google Scholar] [CrossRef]
- Darwall, W.; Smith, K. The Status and Distribution of the Freshwater Fish Endemic to the Mediterranean Basin; International Union for Conservation of Nature: Gland, Switzerland, 2006. [Google Scholar]
- Gardner, R.C.; Barchiesi, S.; Beltrame, C.; Finlayson, C.; Galewski, T.; Harrison, I. State of the World’s Wetlands and Their Services to People: A Compilation of Recent Analyses; Ramsar Briefing Note No. 7; Ramsar Convention Secretariat: Gland, Switzerland, 2015. [Google Scholar]
- Iveša, N.; Piria, M.; Gelli, M.; Trnski, T.; Špelić, I.; Radočaj, T.; Kljak, K.; Jug-Dujaković, J.; Gavrilović, A. Feeding Habits of Predatory Thermophilic Fish Species and Species with Subtropical Affinity from Recently Extended Distributional Range in Northeast Adriatic Sea, Croatia. Diversity 2021, 13, 357. [Google Scholar] [CrossRef]
- Rehage, J.S.; Lopez, L.K.; Sih, A. A comparison of the establishment success, response to competition, and community impact of invasive and non-invasive Gambusia species. Biol. Invasions 2019, 22, 509–522. [Google Scholar] [CrossRef]
- Cabral, J.A.; Marques, J.C. Life history, population dynamics and production of eastern mosquitofish, Gambusia holbrooki (Pisces, Poeciliidae), in rice fields of the lower Mondego River Valley, western Portugal. Acta Oecologica 1999, 20, 607–620. [Google Scholar] [CrossRef] [Green Version]
- Monti, F.; Marcelli, M.; Fastelli, P.; Fattorini, N. Pushed to the edge: Environmental factors drive ecological responses of Aphanius fasciatus when in sympatry with invasive Gambusia holbrooki. Aquat. Conserv. Mar. Freshw. Ecosyst. 2021, 31, 2547–2559. [Google Scholar] [CrossRef]
- Koral, S.; Kose, S.; Tufan, B. Investigating the Quality Changes of Raw and Hot Smoked Garfish (Belone belone euxini, Günther, 1866) at Ambient and Refrigerated Temperatures. Turk. J. Fish. Aquat. Sci. 2009, 9, 53–58. [Google Scholar]
- Zerunian, S. Pesci Delle Acque Interne d’Italia; Ministero dell’Ambiente e della Tutela del Territorio, Direzione Conservazione della Natura: Rome, Italy, 2004. [Google Scholar]
- Morais, S.; Aragão, C.; Cabrita, E.; Conceição, L.E.; Constenla, M.; Costas, B.; Dinis, M.T. New developments and biological insights into the farming of Solea senegalensis reinforcing its aquaculture potential. Aquaculture 2016, 8, 227–263. [Google Scholar] [CrossRef] [Green Version]
- Taktak, W.; Nasri, R.; Lopez-Rubio, A.; Hamdi, M.; Gomez-Mascaraque, L.G.; Ben Amor, N.; Kabadou, A.; Li, S.; Nasri, M.; Karra-Chaâbouni, M. Improved antioxidant activity and oxidative stability of spray dried European eel (Anguilla anguilla) oil microcapsules: Effect of emulsification process and eel protein isolate concentration. Mater. Sci. Eng. C 2019, 104, 109867. [Google Scholar] [CrossRef]
- Bosco, A.D.; Mattioli, S.; Mancini, S.; Mancinelli, A.C.; Cotozzolo, E.; Castellini, C. Nutritional composition of raw and fried big-scale sand smelt (Atherina boyeri) from trasimeno lake. Ital. J. Anim. Sci. 2019, 18, 608–614. [Google Scholar] [CrossRef] [Green Version]
- Reale, A.; Ziino, M.; Ottolenghi, F.; Pelusi, P.; Romeo, V.; Condurso, C.; Sanfilippo, M. Chemical composition and nutritional value of some marine species from the Egadi Islands. Chem. Ecol. 2006, 22, S173–S179. [Google Scholar] [CrossRef]
- Rossi, R.; Carrieri, A.; Rizzo, M.G.; Lucchini, M. Use of intensive rearing systems as back-up for coastal lagoon aquaculture: An experience with eels, Anguilla anguilla L., in the Comacchio lagoons. Aquac. Res. 1988, 19, 355–361. [Google Scholar] [CrossRef]
- Zogaris, S. Conservation study of the Mediterranean Killifish Aphanius fasciatus in Akrotiri Marsh, (Akrotiri SBA, Cyprus)—Final Report. Darwin Project DPLUS034 “Akrotiri Marsh Restoration: A flagship wetland in the Cyprus SBAs BirdLife Cyprus.” Nicosia Cyprus. Unpublished Final Report, 2017; pp. 1–64.
- Molinari, A.; Bonel, N. Indagine Visuale delle Specie Ittiche e Bentoniche Presenti Presso la Barriera Artificiale di Ripopolamento del Comune di Alassio; RSTA: Genoa, Italy, 2010; pp. 1–6. [Google Scholar]
- Cautadella, S.; Bronzi, P. dei Prodotti Ittici, C.D.P. Aquacoltura Responsabile—Verso le Produzioni Acquatiche del Terzo Millennio; Unimar-Uniprom: Rome, Italy, 2007; pp. 1–683. [Google Scholar]
- Santos, N.; Lino, P.G.; Pousão-Ferreira, P.; Monteiro, C.C. Preliminary results of hatchery-reared seabreams released at artificial reefs off the algarve coast (southern portugal): A pilot study. Bull. Mar. Sci. 2006, 78, 177–184. [Google Scholar]
- Pereira, J.C.; Lino, P.G.; Leitão, A.; Joaquim, S.; Chaves, R.; Pousãao-Ferreira, P.; Guedes-Pinto, H.; dos Santos, M.N. Genetic differences between wild and hatchery populations of Diplodus sargus and D. vulgaris inferred from RAPD markers: Implications for production and restocking programs design. J. Appl. Genet. 2010, 51, 67–72. [Google Scholar] [CrossRef]
- AIIAD (Associazione Italiana Ittologi Acque Dolci). Principi Guida Riguardanti le Immissioni di Fauna Ittica Nelle Acque Interne Italiane; AIIAD: Rome, Italy, 2021; pp. 1–31. [Google Scholar]
- Zgüla, A.; Löka, A.; Tanrıkulb, T.T.; Alós, J. Home range and residency of Scorpaena porcus and Scorpaena scrofa in artificial reefs revealed by fine-scale acoustic tracking. Fish. Res. 2019, 210, 22–30. [Google Scholar]
- Abate, F.S.; Abella, A.; Accadia, P.; Addis, P.; Andaloro, F. L’acquacoltura. Lo Stato della Pesca e dell’Acquacoltura nei Mari Italiani; Ministero delle Politiche Agricole Alimentari e Forestali: Rome, Italy, 2012. [Google Scholar]
- Rodríguez, A.S.; Grau, A.; Castro-Fernández, J.; Castejón, I.; Terrados, J.; Morales-Nin, B.; Arechavala-Lopez, P. Reproductive Biology of Pipefish Syngnathus typhle and S. abaster (Syngnathidae) from Western Mediterranean Sea. J. Ichthyol. 2021, 61, 608–615. [Google Scholar] [CrossRef]
- Lovatelli, A.; Holthus, P.F. Capture-Based Aquaculture. Global Overview; FAO Fisheries Technical Paper; No. 508; FAO: Rome, Italy, 2008; p. 298. [Google Scholar]
- Rosenthal, H.; Fonds, M. Biological observations during rearing experiments with the garfish Belone belone. Mar. Biol. 1973, 21, 203–218. [Google Scholar] [CrossRef]
- Iandoli, C.; Trincanato, A. Quadro Generale dell’Acquacoltura Italiana; ICRAM and API: Ahmedabad, India, 2020; p. 56. [Google Scholar]
- Novák, J.; Kalous, L.; Patoka, J. Modern ornamental aquaculture in Europe: Early history of freshwater fish imports. Rev. Aquac. 2020, 12, 2042–2060. [Google Scholar] [CrossRef]
- Sila, A.; Martinez-Alvarez, O.; Krichen, F.; Gómez-Guillén, M.C.; Bougates, A. Gelatin prepared from European eel (Anguilla anguilla) skin: Physicochemical, textural, viscoelastic and surface properties. Colloids Surf. A Physicochem. Eng. Asp. 2017, 529, 643–650. [Google Scholar] [CrossRef] [Green Version]
- Gümüş, E. Effect of Replacement of Fishmeal with Sand Smelt (Atherina boyeri) Meal on Growth, Feed Utilization and Body Composition of Mirror Carp Fry (Cyprinus carpio). Kafkas Univ. Vet. Fak. Derg. 2011, 17, 363–369. [Google Scholar]
- Estefanell, J.; Roo, J.; Guirao, R.; Izquierdo, M.; Socorro, J. Benthic cages versus floating cages in Octopus vulgaris: Biological performance and biochemical composition feeding on Boops boops discarded from fish farms. Aquac. Eng. 2012, 49, 46–52. [Google Scholar] [CrossRef]
- Lassoued, I.; Trigui, M.; Ghlissi, Z.; Nasri, R.; Jamoussi, K.; Kessis, M.; Sahnoun, Z.; Rebai, T.; Boualga, A.; Lamri-Senhadji, M.; et al. Evaluation of hypocholesterolemic effect and antioxidant activity of Boops boops proteins in cholesterol-fed rats. Food Funct. 2014, 5, 1224–1231. [Google Scholar] [CrossRef]
- Rosa, A.; Scano, P.; Atzeri, A.; Deiana, M.; Falchi, A.M. Potential anti-tumor effects of Mugil cephalus processed roe extracts on colon cancer cells. Food Chem. Toxicol. 2013, 60, 471–478. [Google Scholar] [CrossRef]
- Ho, S.S.; Bond, N.R.; Thompson, R.M. Does seasonal flooding give a native species an edge over a global invader? Freshw. Biol. 2013, 58, 159–170. [Google Scholar] [CrossRef]
- Barbato, M.; Barría, C.; Bellodi, A.; Bonanomi, S.; Borme, D.; Ćetković, I.; Colloca, F.; Colmenero, A.I.; Crocetta, F.; De Carlo, F.; et al. The use of fishers’ Local Ecological Knowledge to reconstruct fish behavioural traits and fishers’ perception of conservation relevance of elasmobranchs in the Mediterranean Sea. Mediterr. Mar. Sci. 2021, 22, 603–622. [Google Scholar] [CrossRef]
- Heymans, J.J.; Coll, M.; Libralato, S.; Morissette, L.; Christensen, V. Global Patterns in Ecological Indicators of Marine Food Webs: A Modelling Approach. PLoS ONE 2014, 9, e95845. [Google Scholar] [CrossRef] [Green Version]
- Moullec, F.; Barrier, N.; Drira, S.; Guilhaumon, F.; Marsaleix, P.; Somot, S.; Ulses, C.; Velez, L.; Shin, Y.-J. An End-to-End Model Reveals Losers and Winners in a Warming Mediterranean Sea. Front. Mar. Sci. 2019, 6, 345. [Google Scholar] [CrossRef]
- Voldoire, A.; Sanchezgomez, E.; Mélia, D.S.Y.; Decharme, B.; Cassou, C.; Senesi, S.; Valcke, S.; Beau, I.; Alias, A.; Chevallier, M.; et al. The CNRM-CM5.1 global climate model: Description and basic evaluation. Clim. Dyn. 2013, 40, 2091–2121. [Google Scholar] [CrossRef] [Green Version]
- Sevault, F.; Somot, S.; Alias, A.; Dubois, C.; Lebeaupin-Brossier, C.; Nabat, P.; Adloff, F.; Déqué, M.; Decharme, B. A fully coupled Mediterranean regional climate system model: Design and evaluation of the ocean component for the 1980–2012 period. Tellus A: Dyn. Meteorol. Oceanogr. 2014, 66, 23967. [Google Scholar] [CrossRef] [Green Version]
- Auger, P.A.; Diaz, F.; Ulses, C.; Estournel, C.; Neveux, J.; Joux, F.; Pujo-Pay, M.; Naudin, J.J. Functioning of the planktonic ecosystem on the Gulf of Lions shelf (NW Mediterranean) during spring and its impact on the carbon deposition: A field data and 3-D modelling combined approach. Biogeosciences 2011, 8, 3231–3261. [Google Scholar] [CrossRef] [Green Version]
| Common Name (Arabic and English) | Identified Species Taxonomy (Genus, Species, Family) | COI Genbank Accession Number | ||
|---|---|---|---|---|
| Thoaban elbahr eloroby | ثعبان البحر الأوروبي | European eel | Anguilla anguilla Anguillidae | KX870840.1 |
| Samaket cardinal elbahr elabyad elmotawasit | سمك كاردينال البحر الأبيض المتوسط | Mediterranean cardinalfish | Apogon imberbis Apogonidae | KY176390.1 |
| Samak elkharman (elhaqul) | سمك الخرمان (الحاقول) | Garfish | Belone belone Belonidae | KJ768216.1 |
| Hasaas elbahr elabyad elmotawasit alramlia | حُساس متوسّطيّ (حساس البحر الأبيض المتوسط الرملي) | Big-scale sand smelt | Atherina boyeri Atherinidae | KM538220.1 |
| Sarsaran | صرصران | Leerfish | Lichia amia Carangidae | JF493771.1 |
| Blenny halqy elraqaba | حلقي الرقبةبليني | Ringneck blenny | Parablennius pilicornis Blenniidae | KY176597.1 |
| Kanjir thoaban elbahr | كونجر ثعبان البحر | Conger eel | Conger conger Congridae | KJ709742.1 |
| kilifish elbahr elmotawasit | كيليفيش البحر المتوسط | Mediterranean killifish | Aphanius fasciatus Cyprinodontidae | MH410032.1 |
| Jubi eimlaq | جوبي عملاق | Giant goby | Gobius cobitis Gobiidae | JF935262.1 |
| Jubi bwkish | بوكيش جوبي | Bucchich’s goby | Gobius bucchichi Gobiidae | MT884415.1 |
| Jubi aswad | جوبي أسود | Black goby | Gobius niger Gobiidae | MT670248.1 |
| Jubi elsakhr | الصخر جوبي | Rock goby | Gobius paganellus Gobiidae | MT670258.1 |
| Quris muqazah motawasity (Arisa) | قورس مقزح متوسطي (عريسة) | Mediterranean rainbow wrasse | Coris julis Labridae | KJ709510.1 |
| Ellibrus eltaawws | اللبروس الطاووس | Peacock wrasse | Simphodus tinca Labridae | KJ768310.1 |
| Ellibrus elmuzakhrafa | اللبروس المزخرفة | Ornate wrasse | Thalassoma pavo Labridae | MT216188.1 |
| Elqarus | سمك القاروص | Sea bass | Dicentrarchus labrax Moronidae | KC5000512.1 |
| Samak elburi samik elshafa | سمك البوري سميك الشفة | Thichlip mullet | Chelon labrosus Mugilidae | EU715474.1 |
| Elburi elzahabi | البوري الذهبي | Golden mullet | Chelon aurata Mugilidae | KC500832.1 |
| Elburi raqiq elshafa | البوري رقيق الشفة | Mowel | Chelon ramada Mugilidae | KY683175.1 |
| Elburi elsaghir | البوري الصغير | Small mullet | Liza saliens Mugilidae | Y683176.1 |
| Samak elburi elmuntashir | السمك البوري المنتشر | Common mullet | Mugil cephalus Mugilidae | KJ205081.1 |
| Elburi elmukhatat ahmar | البوري المخطط أحمر | Surmullet | Mullus surmuletus Mullidae | KJ768264.1 |
| Elshiq elmotawasity (thoban elbahr) | الشيق المتوسطي (ثعبان البحر) | Mediterranean moray | Muraena helena Muraenidae | LC163889.1 |
| Samakat elbaeud | سمكة البعوض | Eastern mosquitofish | Gambusia holbrookii Poeciliidae | MT456153.1 |
| Aqrab elbahr elbirazilii | عقرب البحر البرازيلي | Orange scorpionfish | Scorpaena scrofa Scorpaenidae | JN312316.1 |
| Samak elshaykh | الشيخ | Comber | Serranus hepatus Serranidae | KM538559.1 |
| Samak mousa | سمك موسى | Senegalese sole | Solea senegalensis Soleidae | KC501568.1 |
| Samak muza | سمك موزة | Bogue | Boops boops Sparidae | KM538238.1 |
| Eldunis shibshid | الدنيس شيبشيد | Sheephead bream | Diplodus puntazzo Sparidae | KJ012350.1 |
| Eldunis elabyad | الدنيس الأبيض | White seabream | Diplodus sargus Sparidae | MT943711.1 |
| Om jiwa | أم جواء | Twoband bream | Diplodus vulgaris Sparidae | KJ012360.1 |
| Eldunis ehmukhatat | الدنيس المخطط | Sand steenbras | Lithognathus mormyrus Sparidae | JF493795.1 |
| Shalba | شلبة | Goldline | Sarpa salpa Sparidae | JF494424.1 |
| Eldunis (eldunis mozhab elraasa) | دنيس (الدنيس مذهب الرأس) | Gilthead bream | Sparus aurata Sparidae | KF280323.1 |
| Eldunis Elmusraj (minuraa) | المسرج الدنيس (منورى) | Saddle bream | Oblada melanura Sparidae | HQ945848.1 |
| Hosan elbahr | حصان البحر | Seahorse | Hippocampus ramulosus Syngnathidae | GQ502143.1 |
| Elsamakat elanbobia zat elkhutut elsawda | السمكة الأنوبية ذات الخطوط السوداء | Blackstripe pipefish | Syngnathus abaster Syngnathidae | KY176665.1 |
| Country | Thermophilic Species | Tropical Species | |||
|---|---|---|---|---|---|
| Coris julis | Thalassoma pavo | Aphanius fasciatus | Gambusia holbrookii | Parablennius pilicornis | |
| Algeria | Chaoui et al. [22]; Embarek et al. [23]; present study | Chaoui et al. [22]; Embarek et al. [23]; present study | Chaoui et al. [22]; Embarek et al. [23]; present study | Chaoui et al. [22]; Embarek et al. [23]; present study | Chaoui et al. [22]; Embarek et al. [23]; present study |
| Tunisia | * Milazzo et al. [53]; Zarrad et al. [44] | * Milazzo et al. [53] | Kraïem et al. [59]; Sellami et al. [60]; Cataudella et al. [9] | Kraïem et al. [59] | Otero et al. [77] |
| Egypt | * Akel [45]; Akel and Karachle [46] | * Akel and Karachle [46] | Lotan and Ben-Tuvia [61] | Kraïem et al. [59] | * Akel and Karachle [46] |
| Turkey | Ayaz et al. [41] | * Karachle and Stergiou [54] | Acarli et al. [62]; Innal [63] | Innal [63] | n.d. |
| Greece | Costa et al. [42] | *Papadakis et al. [55] | Nicolaidou et al. [64]; Kottelat et al. [65]; Cataudella et al. [9]; Facca et al. [66] | Kalogianni et al. [71] | n.d. |
| Albania | * Soldo and Bakiu [47] | * Soldo and Bakiu [47] | Peja et al. [67]; Cataudella et al. [9] | Kalogianni et al. [71] | n.d. |
| Montenegro | n.d. | n.d. | Cataudella et al. [9] | n.d. | n.d. |
| Italy | * Guidetti et al. [48] | * Azzurro et al. [56]; * Encarnação et al. [57] | Franco et al. [68]; Cataudella et al. [9]; Valdesalici et al. [69]; Facca et al. [66]; Zucchetta et al. [70] | Franco et al. [68]; Locatello et al. [72] | * Pastor and Francour [77]; * Madonna et al. [28] |
| France | * Lejeune [49]; Bray and Bartoli [50] | * Sala [58] | Facca et al. [66] | Le Direach et al. [73] | * Pastor and Francour [77] |
| Spain | * Crec’hriou et al. [51] | Pérez-Ruzafa et al. [52] | Franco et al. [68] | Srean et al. [74] | Pérez-Ruzafa et al. [52] |
| Morocco | Selfati et al. [43] | Selfati et al. [43] | Kraïem et al. [59] | Kraïem et al. [59]; Taybi et al. [75] | Embarek et al. [22] |
| Fish Species | Socio-Economic Use | ||||
|---|---|---|---|---|---|
| Syngnathus abaster | Restocking | ||||
| Lithognathus mormyrus | Human food | ||||
| Thalassoma pavo | Aquariology | ||||
| Gobius cobitis | Human food | ||||
| Parablennius pilicornis | Aquariology | ||||
| Sarpa salpa | Human food | Restocking | |||
| Diplodus vulgaris | Human food | Restocking | |||
| Diplodus puntazzo | Human food | Culture | |||
| Serranus hepatus | Human food | Aquariology | |||
| Gambusia holbrookii | Aquariology | Restocking | |||
| Muraena helena | Human food | Restocking | |||
| Gobius paganellus | Human food | Aquariology | |||
| Gobius niger | Human food | Aquariology | |||
| Gobius bucchichi | Aquariology | Animal Feeding | |||
| Aphanius fasciatus | Aquariology | Restocking | |||
| Conger conger | Human food | Restocking | |||
| Lichia amia | Human food | Medical | |||
| Apogon imberbis | Aquariology | Restocking | |||
| Solea senegalensis | Human food | Restocking | Culture | ||
| Oblada melanura | Human food | Restocking | Medical | ||
| Diplodus sargus | Human food | Restocking | Culture | ||
| Boops boops | Human food | Animal Feeding | Medical | ||
| Scorpaena scrofa | Human food | Restocking | Medical | ||
| Mullus surmuletus | Human food | Restocking | Medical | ||
| Liza saliens | Human food | Culture | Animal Feeding | ||
| Liza ramada | Human food | Culture | Animal Feeding | ||
| Liza aurata | Human food | Culture | Animal Feeding | ||
| Symphodus tinca | Human food | Restocking | Aquariology | ||
| Coris julis | Human food | Restocking | Aquariology | ||
| Chelon labrosus | Human food | Restocking | Culture | Animal Feeding | |
| Dicentrarchus labrax | Human food | Restocking | Culture | Medical | |
| Belone belone | Human food | Culture | Animal Feeding | Medical | |
| Atherina boyeri | Human food | Restocking | Culture | Animal Feeding | |
| Anguilla anguilla | Human food | Restocking | Culture | Animal Feeding | |
| Sparus aurata | Human food | Restocking | Culture | Medical | |
| Mugil cephalus | Human food | Restocking | Culture | Animal Feeding | Medical |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parisi, C.; De Marco, G.; Labar, S.; Hasnaoui, M.; Grieco, G.; Caserta, L.; Inglese, S.; Vangone, R.; Madonna, A.; Alwany, M.; et al. Biodiversity Studies for Sustainable Lagoon: Thermophilic and Tropical Fish Species vs. Endemic Commercial Species at Mellah Lagoon (Mediterranean, Algeria). Water 2022, 14, 635. https://doi.org/10.3390/w14040635
Parisi C, De Marco G, Labar S, Hasnaoui M, Grieco G, Caserta L, Inglese S, Vangone R, Madonna A, Alwany M, et al. Biodiversity Studies for Sustainable Lagoon: Thermophilic and Tropical Fish Species vs. Endemic Commercial Species at Mellah Lagoon (Mediterranean, Algeria). Water. 2022; 14(4):635. https://doi.org/10.3390/w14040635
Chicago/Turabian StyleParisi, Costantino, Giuseppe De Marco, Sofiane Labar, Mustapha Hasnaoui, Gaetano Grieco, Lidia Caserta, Sara Inglese, Rubina Vangone, Adriano Madonna, Magdy Alwany, and et al. 2022. "Biodiversity Studies for Sustainable Lagoon: Thermophilic and Tropical Fish Species vs. Endemic Commercial Species at Mellah Lagoon (Mediterranean, Algeria)" Water 14, no. 4: 635. https://doi.org/10.3390/w14040635










