Road Runoff Characterization: Ecotoxicological Assessment Combined with (Non-)Target Screenings of Micropollutants for the Identification of Relevant Toxicants in the Dissolved Phase
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites and Sampling
2.2. Global Parameters, 3D Fluorescence, and Targeted Screening
2.3. Non-Target Screening with HRMS
2.4. Biological Approaches
2.4.1. Animals
2.4.2. General Fish Breeding
2.4.3. Embryos Exposure
2.4.4. Teratology Screening and Behavioral Testing
2.4.5. RR Samples and Chemicals Preparation
2.4.6. Assay of Mitochondrial NADH-Ubiquinone Reductase (Complex I) Activity
3. Results
3.1. Overview of the Two Sites: Global Characterization
3.2. (Eco)Toxicity Assessment of RR Samples on Zebrafish Larvae Development and Behaviour
3.2.1. Toxicity of RR Samples
3.2.2. Light–Dark Stress Test
3.2.3. Mitochondrial Respiratory Chain Complex I
3.3. Non-Target Screening and Identification of Specific Micropollutants by HRMS
3.4. (Eco)Toxicity of 1,3-Diphenylguanidine & Benzotriazole
3.4.1. Light–Dark Stress Test
3.4.2. Mitochondrial Respiratory Chain Complex I
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gasperi, J.; Sebastian, C.; Ruban, V.; Delamain, M.; Percot, S.; Wiest, L.; Mirande, C.; Caupos, E.; Demare, D.; Kessoo, M.D.K.; et al. Micropollutants in Urban Stormwater: Occurrence, Concentrations, and Atmospheric Contributions for a Wide Range of Contaminants in Three French Catchments. Environ. Sci. Pollut. Res. Int. 2014, 21, 5267–5281. [Google Scholar] [CrossRef] [Green Version]
- Revitt, D.M.; Lundy, L.; Coulon, F.; Fairley, M. The Sources, Impact and Management of Car Park Runoff Pollution: A Review. J. Environ. Manag. 2014, 146, 552–567. [Google Scholar] [CrossRef] [Green Version]
- Markiewicz, A.; Björklund, K.; Eriksson, E.; Kalmykova, Y.; Strömvall, A.-M.; Siopi, A. Emissions of Organic Pollutants from Traffic and Roads: Priority Pollutants Selection and Substance Flow Analysis. Sci. Total Environ. 2017, 580, 1162–1174. [Google Scholar] [CrossRef] [PubMed]
- Amorello, D.; Barreca, S.; Orecchio, S.; Ferro, S. Platinum in Indoor Settled Dust Matter (Homes and Cars). Microchem. J. 2015, 123, 76–83. [Google Scholar] [CrossRef]
- Orecchio, S.; Amorello, D.; Barreca, S.; Valenti, A. Wood Pellets for Home Heating Can Be Considered Environmentally Friendly Fuels? Polycyclic Aromatic Hydrocarbons (PAHs) in Their Ashes. Microchem. J. 2016, 124, 267–271. [Google Scholar] [CrossRef]
- Orecchio, S.; Amorello, D.; Barreca, S. (II) Wood Pellets for Home Heating Can Be Considered Environmentally Friendly Fuels? Heavy Metals Determination by Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES) in Their Ashes and the Health Risk Assessment for the Operators. Microchem. J. 2016, 127, 178–183. [Google Scholar] [CrossRef]
- Eriksson, E.; Baun, A.; Scholes, L.; Ledin, A.; Ahlman, S.; Revitt, M.; Noutsopoulos, C.; Mikkelsen, P.S. Selected Stormwater Priority Pollutants—A European Perspective. Sci. Total Environ. 2007, 383, 41–51. [Google Scholar] [CrossRef]
- Wagner, S.; Hüffer, T.; Klöckner, P.; Wehrhahn, M.; Hofmann, T.; Reemtsma, T. Tire Wear Particles in the Aquatic Environment—A Review on Generation, Analysis, Occurrence, Fate and Effects. Water Res. 2018, 139, 83–100. [Google Scholar] [CrossRef]
- Gasperi, J.; Le Roux, J.; Deshayes, S.; Ayrault, S.; Bordier, L.; Boudahmane, L.; Budzinski, H.; Caupos, E.; Caubrière, N.; Flanagan, K.; et al. Micropollutants in the Urban Runoff from Traffic Areas: Target and Non-Target Screening on Four Contrasted Sites. Water 2022, 14, 394. [Google Scholar] [CrossRef]
- Bressy, A.; Gromaire, M.-C.; Lorgeoux, C.; Chebbo, G. Alkylphenols in Atmospheric Depositions and Urban Runoff. Water Sci. Technol. 2011, 63, 671–679. [Google Scholar] [CrossRef]
- Bressy, A.; Gromaire, M.-C.; Lorgeoux, C.; Saad, M.; Leroy, F.; Chebbo, G. Towards the Determination of an Optimal Scale for Stormwater Quality Management: Micropollutants in a Small Residential Catchment. Water Res. 2012, 46, 6799–6810. [Google Scholar] [CrossRef] [PubMed]
- Krauss, M.; Singer, H.; Hollender, J. LC–High Resolution MS in Environmental Analysis: From Target Screening to the Identification of Unknowns. Anal. Bioanal. Chem. 2010, 397, 943–951. [Google Scholar] [CrossRef] [Green Version]
- Schymanski, E.L.; Singer, H.P.; Slobodnik, J.; Ipolyi, I.M.; Oswald, P.; Krauss, M.; Schulze, T.; Haglund, P.; Letzel, T.; Grosse, S.; et al. Non-Target Screening with High-Resolution Mass Spectrometry: Critical Review Using a Collaborative Trial on Water Analysis. Anal. Bioanal. Chem. 2015, 407, 6237–6255. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Tian, Z.; Peter, K.T.; Kolodziej, E.P.; Wong, C.S. Developing Unique Nontarget High-Resolution Mass Spectrometry Signatures to Track Contaminant Sources in Urban Waters. Environ. Sci. Technol. Lett. 2020, 7, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Seiwert, B.; Klöckner, P.; Wagner, S.; Reemtsma, T. Source-Related Smart Suspect Screening in the Aqueous Environment: Search for Tire-Derived Persistent and Mobile Trace Organic Contaminants in Surface Waters. Anal. Bioanal. Chem. 2020, 412, 4909–4919. [Google Scholar] [CrossRef]
- Du, B.; Lofton, J.M.; Peter, K.T.; Gipe, A.D.; James, C.A.; McIntyre, J.K.; Scholz, N.L.; Baker, J.E.; Kolodziej, E.P. Development of Suspect and Non-Target Screening Methods for Detection of Organic Contaminants in Highway Runoff and Fish Tissue with High-Resolution Time-of-Flight Mass Spectrometry. Environ. Sci.: Process. Impacts 2017, 19, 1185–1196. [Google Scholar] [CrossRef]
- Robinson, P.D. Behavioural Toxicity of Organic Chemical Contaminants in Fish: Application to Ecological Risk Assessments (ERAs). Can. J. Fish. Aquat. Sci. 2009, 66, 1179–1188. [Google Scholar] [CrossRef]
- Linbo, T.L.; Baldwin, D.H.; McIntyre, J.K.; Scholz, N.L. Effects of Water Hardness, Alkalinity, and Dissolved Organic Carbon on the Toxicity of Copper to the Lateral Line of Developing Fish. Environ. Toxicol. Chem. 2009, 28, 1455–1461. [Google Scholar] [CrossRef] [Green Version]
- Wernersson, A.-S.; Carere, M.; Maggi, C.; Tusil, P.; Soldan, P.; James, A.; Sanchez, W.; Dulio, V.; Broeg, K.; Reifferscheid, G.; et al. The European Technical Report on Aquatic Effect-Based Monitoring Tools under the Water Framework Directive. Environ. Sci. Eur. 2015, 27, 7. [Google Scholar] [CrossRef] [Green Version]
- Ginebreda, A.; Kuzmanovic, M.; Guasch, H.; de Alda, M.L.; López-Doval, J.C.; Muñoz, I.; Ricart, M.; Romaní, A.M.; Sabater, S.; Barceló, D. Assessment of Multi-Chemical Pollution in Aquatic Ecosystems Using Toxic Units: Compound Prioritization, Mixture Characterization and Relationships with Biological Descriptors. Sci. Total Environ. 2014, 468–469, 715–723. [Google Scholar] [CrossRef]
- Prasse, C.; Stalter, D.; Schulte-Oehlmann, U.; Oehlmann, J.; Ternes, T.A. Spoilt for Choice: A Critical Review on the Chemical and Biological Assessment of Current Wastewater Treatment Technologies. Water Res. 2015, 87, 237–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neale, P.A.; Altenburger, R.; Aït-Aïssa, S.; Brion, F.; Busch, W.; de Aragão Umbuzeiro, G.; Denison, M.S.; Du Pasquier, D.; Hilscherová, K.; Hollert, H.; et al. Development of a Bioanalytical Test Battery for Water Quality Monitoring: Fingerprinting Identified Micropollutants and Their Contribution to Effects in Surface Water. Water Res. 2017, 123, 734–750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tousova, Z.; Oswald, P.; Slobodnik, J.; Blaha, L.; Muz, M.; Hu, M.; Brack, W.; Krauss, M.; Di Paolo, C.; Tarcai, Z.; et al. European Demonstration Program on the Effect-Based and Chemical Identification and Monitoring of Organic Pollutants in European Surface Waters. Sci. Total Environ. 2017, 601–602, 1849–1868. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.J.; Teraoka, H.; Heideman, W.; Peterson, R.E. Zebrafish as a Model Vertebrate for Investigating Chemical Toxicity. Toxicol. Sci. 2005, 86, 6–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, J.-H.; Gao, J.-M.; Huang, C.-J.; Li, C.-Q. Zebrafish Models for Assessing Developmental and Reproductive Toxicity. Neurotoxicol. Teratol. 2014, 42, 35–42. [Google Scholar] [CrossRef]
- McCollum, C.W.; Ducharme, N.A.; Bondesson, M.; Gustafsson, J.-A. Developmental Toxicity Screening in Zebrafish. Birth Defects Res. Part C Embryo Today Rev. 2011, 93, 67–114. [Google Scholar] [CrossRef]
- Mione, M.C.; Trede, N.S. The Zebrafish as a Model for Cancer. Dis. Model. Mech. 2010, 3, 517–523. [Google Scholar] [CrossRef] [Green Version]
- Bai, Q.; Burton, E.A. Zebrafish Models of Tauopathy. Biochim. Biophys. Acta 2011, 1812, 353–363. [Google Scholar] [CrossRef] [Green Version]
- Goldsmith, J.R.; Jobin, C. Think Small: Zebrafish as a Model System of Human Pathology. J. Biomed. Biotechnol. 2012, 2012, e817341. [Google Scholar] [CrossRef]
- Chávez, M.N.; Aedo, G.; Fierro, F.A.; Allende, M.L.; Egaña, J.T. Zebrafish as an Emerging Model Organism to Study Angiogenesis in Development and Regeneration. Front. Physiol. 2016, 7, 56. [Google Scholar] [CrossRef] [Green Version]
- Scott, G.R.; Sloman, K.A. The Effects of Environmental Pollutants on Complex Fish Behaviour: Integrating Behavioural and Physiological Indicators of Toxicity. Aquat. Toxicol. 2004, 68, 369–392. [Google Scholar] [CrossRef] [PubMed]
- Selderslaghs, I.W.T.; Hooyberghs, J.; Blust, R.; Witters, H.E. Assessment of the Developmental Neurotoxicity of Compounds by Measuring Locomotor Activity in Zebrafish Embryos and Larvae. Neurotoxicol. Teratol. 2013, 37, 44–56. [Google Scholar] [CrossRef]
- Legradi, J.B.; Di Paolo, C.; Kraak, M.H.S.; van der Geest, H.G.; Schymanski, E.L.; Williams, A.J.; Dingemans, M.M.L.; Massei, R.; Brack, W.; Cousin, X.; et al. An Ecotoxicological View on Neurotoxicity Assessment. Environ. Sci. Eur. 2018, 30, 46. [Google Scholar] [CrossRef] [PubMed]
- Ogungbemi, A.; Leuthold, D.; Scholz, S.; Küster, E. Hypo- or Hyperactivity of Zebrafish Embryos Provoked by Neuroactive Substances: A Review on How Experimental Parameters Impact the Predictability of Behavior Changes. Environ. Sci. Eur. 2019, 31, 88. [Google Scholar] [CrossRef]
- Young, A.; Kochenkov, V.; McIntyre, J.K.; Stark, J.D.; Coffin, A.B. Urban Stormwater Runoff Negatively Impacts Lateral Line Development in Larval Zebrafish and Salmon Embryos. Sci. Rep. 2018, 8, 2830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McIntyre, J.K.; Edmunds, R.C.; Redig, M.G.; Mudrock, E.M.; Davis, J.W.; Incardona, J.P.; Stark, J.D.; Scholz, N.L. Confirmation of Stormwater Bioretention Treatment Effectiveness Using Molecular Indicators of Cardiovascular Toxicity in Developing Fish. Environ. Sci. Technol. 2016, 50, 1561–1569. [Google Scholar] [CrossRef]
- Wu, L.; Jiang, Y.; Zhang, L.; Chen, L.; Zhang, H. Toxicity of Urban Highway Runoff in Shanghai to Zebrafish (Danio Rerio) Embryos and Luminous Bacteria (Vibrio Qinghaiensis.Q67). Environ. Sci. Pollut. Res. Int. 2014, 21, 2663–2676. [Google Scholar] [CrossRef]
- Chen, X.D.; Culbert, E.; Hebert, V.; Stark, J.D. Mixture Effects of the Nonylphenyl Polyethoxylate, R-11 and the Insecticide, Imidacloprid on Population Growth Rate and Other Parameters of the Crustacean, Ceriodaphnia Dubia. Ecotoxicol. Environ. Saf. 2010, 73, 132–137. [Google Scholar] [CrossRef]
- Chen, H.; Yao, J.; Wang, F.; Zhou, Y.; Chen, K.; Zhuang, R.; Choi, M.M.F.; Zaray, G. Toxicity of Three Phenolic Compounds and Their Mixtures on the Gram-Positive Bacteria Bacillus Subtilis in the Aquatic Environment. Sci. Total Environ. 2010, 408, 1043–1049. [Google Scholar] [CrossRef]
- Shao, Y.; Chen, Z.; Hollert, H.; Zhou, S.; Deutschmann, B.; Seiler, T.-B. Toxicity of 10 Organic Micropollutants and Their Mixture: Implications for Aquatic Risk Assessment. Sci. Total Environ. 2019, 666, 1273–1282. [Google Scholar] [CrossRef]
- Peng, X.; Lin, J.; Zhu, Y.; Liu, X.; Zhang, Y.; Ji, Y.; Yang, X.; Zhang, Y.; Guo, N.; Li, Q. Anxiety-Related Behavioral Responses of Pentylenetetrazole-Treated Zebrafish Larvae to Light-Dark Transitions. Pharm. Biochem. Behav. 2016, 145, 55–65. [Google Scholar] [CrossRef]
- Leuthold, D.; Klüver, N.; Altenburger, R.; Busch, W. Can Environmentally Relevant Neuroactive Chemicals Specifically Be Detected with the Locomotor Response Test in Zebrafish Embryos? Environ. Sci. Technol. 2019, 53, 482–493. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, K.; Branchu, P.; Boudahmane, L.; Caupos, E.; Demare, D.; Deshayes, S.; Dubois, P.; Meffray, L.; Partibane, C.; Saad, M.; et al. Field Performance of Two Biofiltration Systems Treating Micropollutants from Road Runoff. Water Res. 2018, 145, 562–578. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, K.; Branchu, P.; Boudahmane, L.; Caupos, E.; Demare, D.; Deshayes, S.; Dubois, P.; Meffray, L.; Partibane, C.; Saad, M.; et al. Retention and Transport Processes of Particulate and Dissolved Micropollutants in Stormwater Biofilters Treating Road Runoff. Sci. Total Environ. 2019, 656, 1178–1190. [Google Scholar] [CrossRef] [PubMed]
- Alberts, J.J.; Takács, M. Total Luminescence Spectra of IHSS Standard and Reference Fulvic Acids, Humic Acids and Natural Organic Matter: Comparison of Aquatic and Terrestrial Source Terms. Org. Geochem. 2004, 35, 243–256. [Google Scholar] [CrossRef]
- Lawaetz, A.J.; Stedmon, C.A. Fluorescence Intensity Calibration Using the Raman Scatter Peak of Water. Appl. Spectrosc. 2009, 63, 936–940. [Google Scholar] [CrossRef]
- Parlanti, E. Dissolved Organic Matter Fluorescence Spectroscopy as a Tool to Estimate Biological Activity in a Coastal Zone Submitted to Anthropogenic Inputs. Org. Geochem. 2000, 31, 1765–1781. [Google Scholar] [CrossRef]
- Zsolnay, A.; Baigar, E.; Jimenez, M.; Steinweg, B.; Saccomandi, F. Differentiating with Fluorescence Spectroscopy the Sources of Dissolved Organic Matter in Soils Subjected to Drying. Chemosphere 1999, 38, 45–50. [Google Scholar] [CrossRef]
- Huguet, A.; Vacher, L.; Relexans, S.; Saubusse, S.; Froidefond, J.M.; Parlanti, E. Properties of Fluorescent Dissolved Organic Matter in the Gironde Estuary. Org. Geochem. 2009, 40, 706–719. [Google Scholar] [CrossRef]
- Lamprea, K.; Bressy, A.; Mirande-Bret, C.; Caupos, E.; Gromaire, M.-C. Alkylphenol and Bisphenol A Contamination of Urban Runoff: An Evaluation of the Emission Potentials of Various Construction Materials and Automotive Supplies. Environ. Sci. Pollut. Res. 2018, 25, 21887–21900. [Google Scholar] [CrossRef]
- Van De Voorde, A. Incidence of Roof Maintenance Practices on the Quality of Runoff Water. Case of Biocidal Treatment Products. Ph.D. Thesis, Université Paris Est, Paris, France, June 2012. [Google Scholar]
- Huynh, N.; Caupos, E.; Soares Peirera, C.; Le Roux, J.; Bressy, A.; Moilleron, R. Evaluation of Sample Preparation Methods for Non-Target Screening of Organic Micropollutants in Urban Waters Using High-Resolution Mass Spectrometry. Molecules 2021, 26, 7064. [Google Scholar] [CrossRef] [PubMed]
- Rosnack, K.J.; Reid, M.J.; Ladak, A.; Cleland, G. Screening Solution Using the Software Platform UNIFI: An Integrated Workflow by Waters. In Assessing Transformation Products of Chemicals by Non-Target and Suspect Screening—Strategies and Workflows Volume 2; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2016; Volume 1242, pp. 155–172. ISBN 978-0-8412-3195-5. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing, v.3.6.2; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Schlager, S. Morpho and Rvcg—Shape Analysis in R. In Statistical Shape and Deformation Analysis; Zheng, G., Li, S., Szekely, G., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 217–256. ISBN 978-0-12-810493-4. [Google Scholar]
- Kassambara, A.; Mundt, F. Factoextra: Extract and Visualize the Results of Multivariate Data Analyses; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Sievert, C. Interactive Web-Based Data Visualization with R, Plotly, and Shiny; Chapman and Hall/CRC: Boca Raton, FL, USA, 2020; ISBN 978-1-138-33145-7. [Google Scholar]
- Council of the European Union. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the Protection of Animals Used for Scientific Purposes. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32010L0063 (accessed on 26 December 2021).
- Hernandez, R.E.; Galitan, L.; Cameron, J.; Goodwin, N.; Ramakrishnan, L. Delay of Initial Feeding of Zebrafish Larvae Until 8 Days Postfertilization Has No Impact on Survival or Growth Through the Juvenile Stage. Zebrafish 2018, 15, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Strähle, U.; Scholz, S.; Geisler, R.; Greiner, P.; Hollert, H.; Rastegar, S.; Schumacher, A.; Selderslaghs, I.; Weiss, C.; Witters, H.; et al. Zebrafish Embryos as an Alternative to Animal Experiments—A Commentary on the Definition of the Onset of Protected Life Stages in Animal Welfare Regulations. Reprod. Toxicol. 2012, 33, 128–132. [Google Scholar] [CrossRef]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of Embryonic Development of the Zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
- Cormier, A.; Morin, C.; Zini, R.; Tillement, J.-P.; Lagrue, G. Nicotine Protects Rat Brain Mitochondria against Experimental Injuries. Neuropharmacology 2003, 44, 642–652. [Google Scholar] [CrossRef]
- Zhou, S.; Chen, Q.; Di Paolo, C.; Shao, Y.; Hollert, H.; Seiler, T.-B. Behavioral Profile Alterations in Zebrafish Larvae Exposed to Environmentally Relevant Concentrations of Eight Priority Pharmaceuticals. Sci. Total Environ. 2019, 664, 89–98. [Google Scholar] [CrossRef]
- Guo, N.; Lin, J.; Peng, X.; Chen, H.; Zhang, Y.; Liu, X.; Li, Q. Influences of Acute Ethanol Exposure on Locomotor Activities of Zebrafish Larvae under Different Illumination. Alcohol 2015, 49, 727–737. [Google Scholar] [CrossRef]
- Degli Esposti, M. Inhibitors of NADH-Ubiquinone Reductase: An Overview. Biochim. Biophys. Acta 1998, 1364, 222–235. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.N.; Leung, M.C.K.; Rooney, J.P.; Sendoel, A.; Hengartner, M.O.; Kisby, G.E.; Bess, A.S. Mitochondria as a Target of Environmental Toxicants. Toxicol. Sci. 2013, 134, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Hughey, C.A.; Hendrickson, C.L.; Rodgers, R.P.; Marshall, A.G.; Qian, K. Kendrick Mass Defect Spectrum: A Compact Visual Analysis for Ultrahigh-Resolution Broadband Mass Spectra. Anal. Chem. 2001, 73, 4676–4681. [Google Scholar] [CrossRef]
- Thurman, E.M.; Ferrer, I.; Blotevogel, J.; Borch, T. Analysis of Hydraulic Fracturing Flowback and Produced Waters Using Accurate Mass: Identification of Ethoxylated Surfactants. Anal. Chem. 2014, 86, 9653–9661. [Google Scholar] [CrossRef]
- Hinnenkamp, V.; Balsaa, P.; Schmidt, T.C. Target, Suspect and Non-Target Screening Analysis from Wastewater Treatment Plant Effluents to Drinking Water Using Collision Cross Section Values as Additional Identification Criterion. Anal. Bioanal. Chem. 2021, 414, 425–438. [Google Scholar] [CrossRef]
- Schulze, S.; Zahn, D.; Montes, R.; Rodil, R.; Quintana, J.B.; Knepper, T.P.; Reemtsma, T.; Berger, U. Occurrence of Emerging Persistent and Mobile Organic Contaminants in European Water Samples. Water Res. 2019, 153, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Challis, J.K.; Popick, H.; Prajapati, S.; Harder, P.; Giesy, J.P.; McPhedran, K.; Brinkmann, M. Occurrences of Tire Rubber-Derived Contaminants in Cold-Climate Urban Runoff. Environ. Sci. Technol. Lett. 2021, 8, 961–967. [Google Scholar] [CrossRef]
- Tian, Z.; Zhao, H.; Peter, K.T.; Gonzalez, M.; Wetzel, J.; Wu, C.; Hu, X.; Prat, J.; Mudrock, E.; Hettinger, R.; et al. A Ubiquitous Tire Rubber–Derived Chemical Induces Acute Mortality in Coho Salmon. Science 2021, 371, 185–189. [Google Scholar] [CrossRef]
- Farré, M.L.; Pérez, S.; Kantiani, L.; Barceló, D. Fate and Toxicity of Emerging Pollutants, Their Metabolites and Transformation Products in the Aquatic Environment. TrAC Trends Anal. Chem. 2008, 27, 991–1007. [Google Scholar] [CrossRef]
- Tran, C.M.; Do, T.N.; Kim, K.-T. Comparative Analysis of Neurotoxicity of Six Phthalates in Zebrafish Embryos. Toxics 2021, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Liu, J.; Lin, Z.; Chen, X.; Yuan, L.; Shen, G.; Yang, W.; Wang, D.; Huang, Y.; Pang, S.; et al. Evaluation of the Spinal Effects of Phthalates in a Zebrafish Embryo Assay. Chemosphere 2020, 249, 126144. [Google Scholar] [CrossRef] [PubMed]
- Zahn, D.; Mucha, P.; Zilles, V.; Touffet, A.; Gallard, H.; Knepper, T.P.; Frömel, T. Identification of Potentially Mobile and Persistent Transformation Products of REACH-Registered Chemicals and Their Occurrence in Surface Waters. Water Res. 2019, 150, 86–96. [Google Scholar] [CrossRef]
- OECD. Screening Information Dataset—Initial Assessment Report—1,3-Diphenylguanidine; OECD: Paris, France, 2002. [Google Scholar]
- Damalas, D.E.; Bletsou, A.A.; Agalou, A.; Beis, D.; Thomaidis, N.S. Assessment of the Acute Toxicity, Uptake and Biotransformation Potential of Benzotriazoles in Zebrafish (Danio Rerio) Larvae Combining HILIC- with RPLC-HRMS for High-Throughput Identification. Environ. Sci. Technol. 2018, 52, 6023–6031. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.-Q.; Liu, Y.-S.; Xiong, Q.; Cai, W.-W.; Ying, G.-G. Occurrence, Toxicity and Transformation of Six Typical Benzotriazoles in the Environment: A Review. Sci. Total Environ. 2019, 661, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Lagarde, F.; Beausoleil, C.; Belcher, S.M.; Belzunces, L.P.; Emond, C.; Guerbet, M.; Rousselle, C. Non-Monotonic Dose-Response Relationships and Endocrine Disruptors: A Qualitative Method of Assessment. Environ. Health 2015, 14, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tangtian, H.; Bo, L.; Wenhua, L.; Shin, P.K.S.; Wu, R.S.S. Estrogenic Potential of Benzotriazole on Marine Medaka (Oryzias Melastigma). Ecotoxicol. Environ. Saf. 2012, 80, 327–332. [Google Scholar] [CrossRef]
- Duan, Z.; Xing, Y.; Feng, Z.; Zhang, H.; Li, C.; Gong, Z.; Wang, L.; Sun, H. Hepatotoxicity of Benzotriazole and Its Effect on the Cadmium Induced Toxicity in Zebrafish Danio Rerio. Environ. Pollut. 2017, 224, 706–713. [Google Scholar] [CrossRef] [PubMed]
- Porseryd, T.; Kellner, M.; Reyhanian Caspillo, N.; Volkova, K.; Elabbas, L.; Ullah, S.; Olsén, H.; Dinnétz, P.; Porsch Hällström, I. Combinatory Effects of Low Concentrations of 17α-Etinylestradiol and Citalopram on Non-Reproductive Behavior in Adult Zebrafish (Danio Rerio). Aquat. Toxicol. 2017, 193, 9–17. [Google Scholar] [CrossRef]
- Kinch, C.D.; Kurrasch, D.M.; Habibi, H.R. Adverse Morphological Development in Embryonic Zebrafish Exposed to Environmental Concentrations of Contaminants Individually and in Mixture. Aquat. Toxicol. 2016, 175, 286–298. [Google Scholar] [CrossRef]
- Sharma, L.K.; Lu, J.; Bai, Y. Mitochondrial Respiratory Complex I: Structure, Function and Implication in Human Diseases. Curr. Med. Chem. 2009, 16, 1266–1277. [Google Scholar] [CrossRef] [Green Version]
- Satoh, T.; Miyoshi, H.; Sakamoto, K.; Iwamura, H. Comparison of the Inhibitory Action of Synthetic Capsaicin Analogues with Various NADH-Ubiquinone Oxidoreductases. Biochim. Biophys. Acta 1996, 1273, 21–30. [Google Scholar] [CrossRef] [Green Version]
- OECD/UNEP Global PFC Group. Synthesis Paper on Per- and Polyfluorinated Chemicals (PFCs); OECD: Paris, France, 2013. [Google Scholar]
- Savoca, D.; Melfi, R.; Palumbo Piccionello, A.; Barreca, S.; Buscemi, S.; Arizza, V.; Arculeo, M.; Pace, A. Presence and Biodistribution of Perfluorooctanoic Acid (PFOA) in Paracentrotus Lividus Highlight Its Potential Application for Environmental Biomonitoring. Sci. Rep. 2021, 11, 18763. [Google Scholar] [CrossRef]
- Mashayekhi, V.; Tehrani, K.H.M.E.; Hashemzaei, M.; Tabrizian, K.; Shahraki, J.; Hosseini, M.-J. Mechanistic Approach for the Toxic Effects of Perfluorooctanoic Acid on Isolated Rat Liver and Brain Mitochondria. Hum. Exp. Toxicol. 2015, 34, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Steenland, K.; Fletcher, T.; Savitz, D.A. Epidemiologic Evidence on the Health Effects of Perfluorooctanoic Acid (PFOA). Environ. Health Perspect. 2010, 118, 1100–1108. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, C.D.; Di Giulio, R.T. Polycyclic Aromatic Hydrocarbon and Hypoxia Exposures Result in Mitochondrial Dysfunction in Zebrafish. Aquat. Toxicol. 2019, 216, 105298. [Google Scholar] [CrossRef] [PubMed]
- Müller, A.; Österlund, H.; Marsalek, J.; Viklander, M. The Pollution Conveyed by Urban Runoff: A Review of Sources. Sci. Total Environ. 2020, 709, 136125. [Google Scholar] [CrossRef]
- Khan, S.; Beigh, S.; Chaudhari, B.P.; Sharma, S.; Aliul Hasan Abdi, S.; Ahmad, S.; Ahmad, F.; Parvez, S.; Raisuddin, S. Mitochondrial Dysfunction Induced by Bisphenol A Is a Factor of Its Hepatotoxicity in Rats. Environ. Toxicol 2016, 31, 1922–1934. [Google Scholar] [CrossRef]
- Tener, R.F.; Holt, W.L. Effect of Antioxidants on the Natural and the Accelerated Aging of Rubber; National Bureau of Standards: Gaithersburg, MD, USA, 1935. [Google Scholar]
- Wong, H.-S.; Dighe, P.A.; Mezera, V.; Monternier, P.-A.; Brand, M.D. Production of Superoxide and Hydrogen Peroxide from Specific Mitochondrial Sites under Different Bioenergetic Conditions. J. Biol. Chem. 2017, 292, 16804–16809. [Google Scholar] [CrossRef] [Green Version]
- Joseph, A.M.; George, B.; Madhusoodanan, K.N.; Alex, R. Current Status of Sulphur Vulcanization and Devulcanization Chemistry: Process of Vulcanization. Rubber Sci. 2015, 28, 82–121. [Google Scholar]
- Brack, W.; Ait-Aissa, S.; Burgess, R.M.; Busch, W.; Creusot, N.; Di Paolo, C.; Escher, B.I.; Mark Hewitt, L.; Hilscherova, K.; Hollender, J.; et al. Effect-Directed Analysis Supporting Monitoring of Aquatic Environment—An in-Depth Overview. Sci. Total Environ. 2016, 544, 1073–1118. [Google Scholar] [CrossRef]
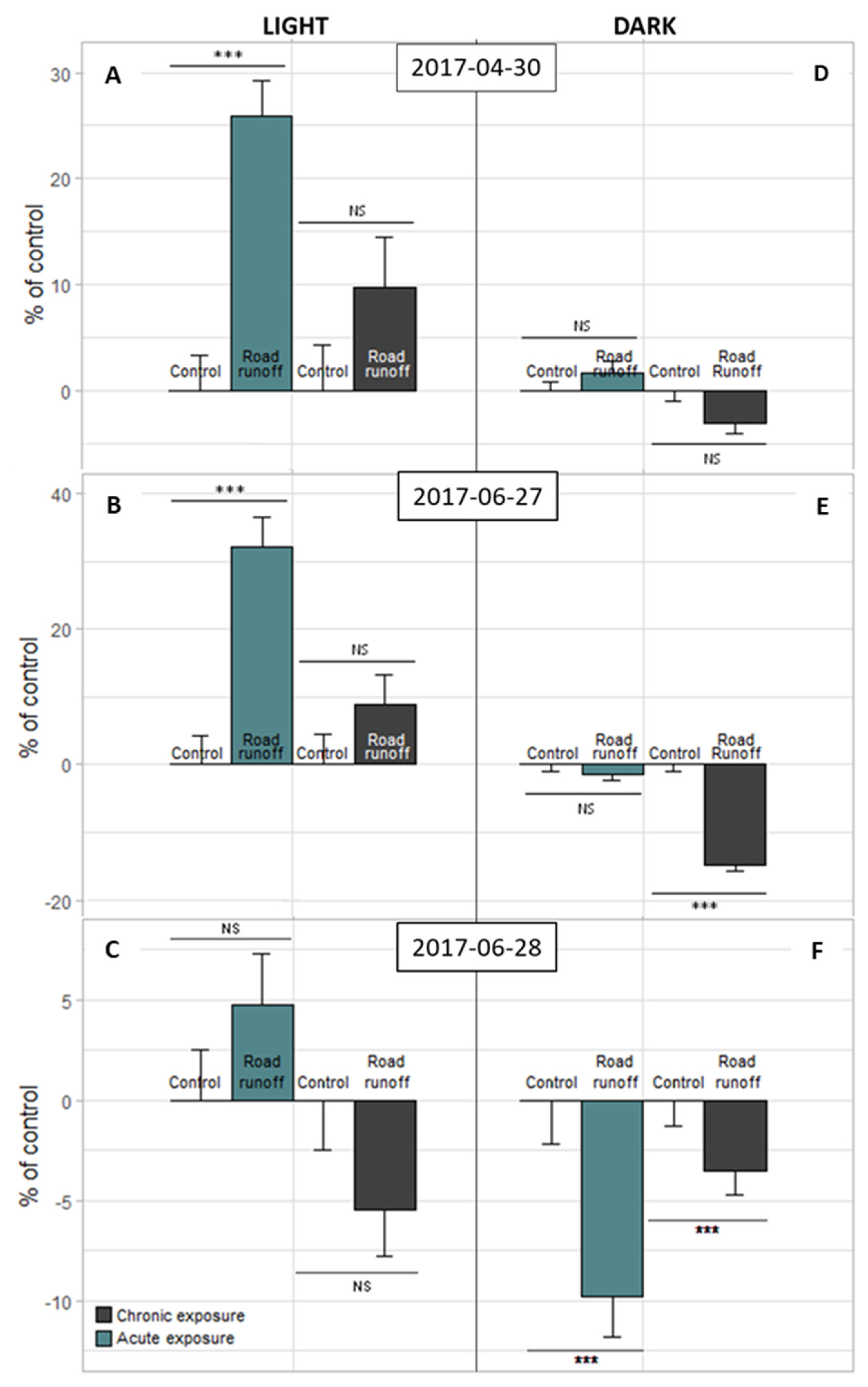
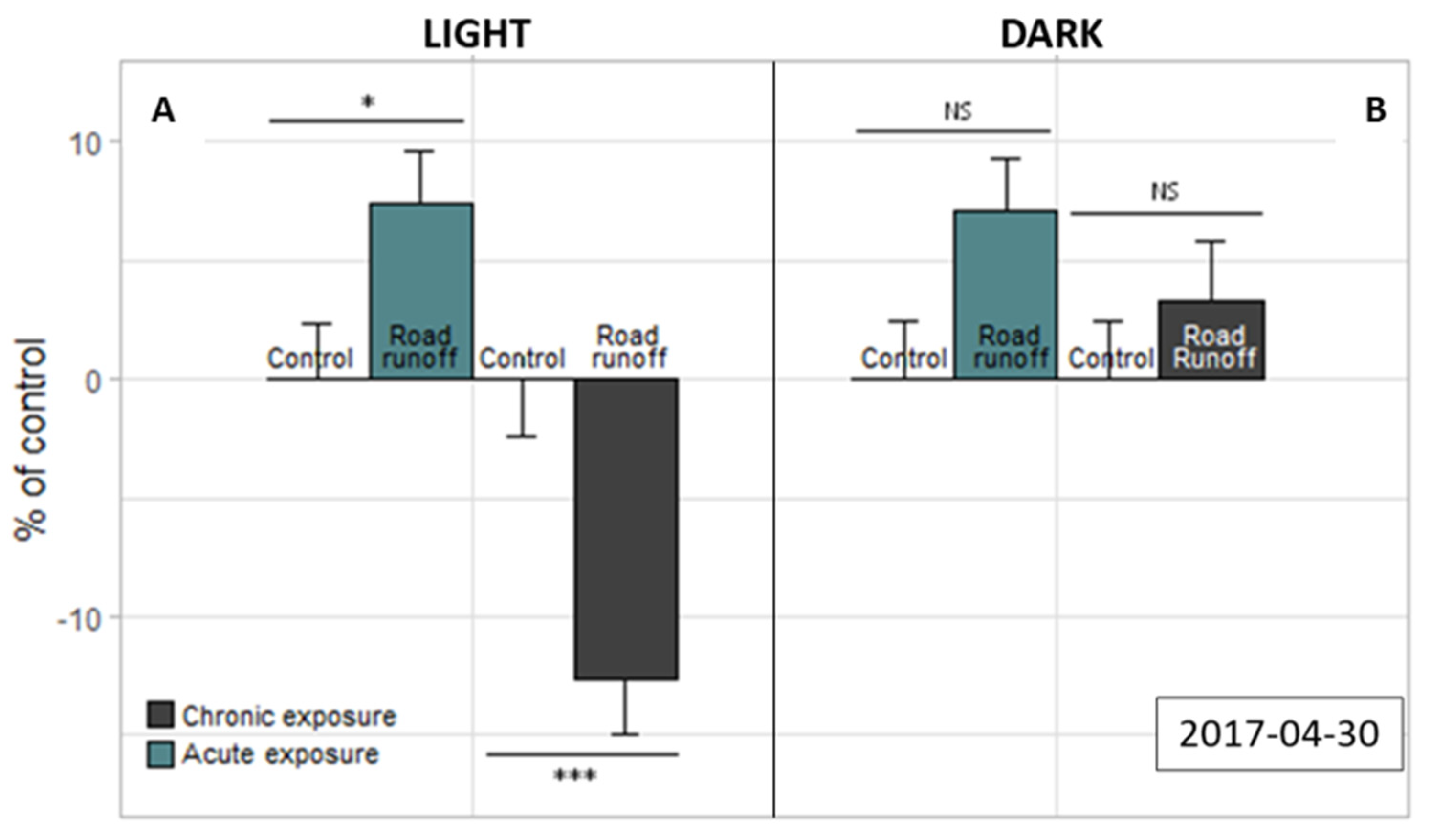
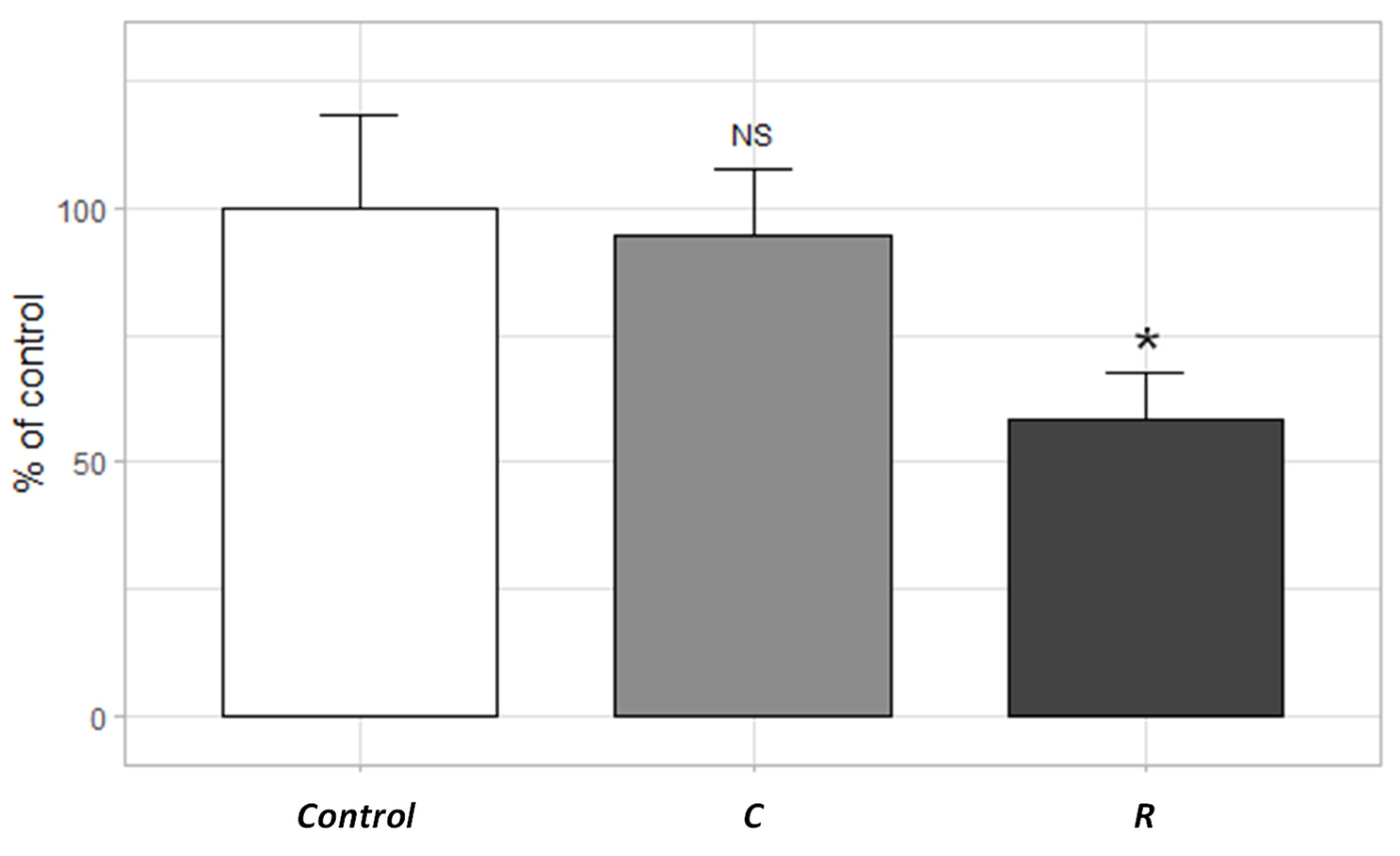
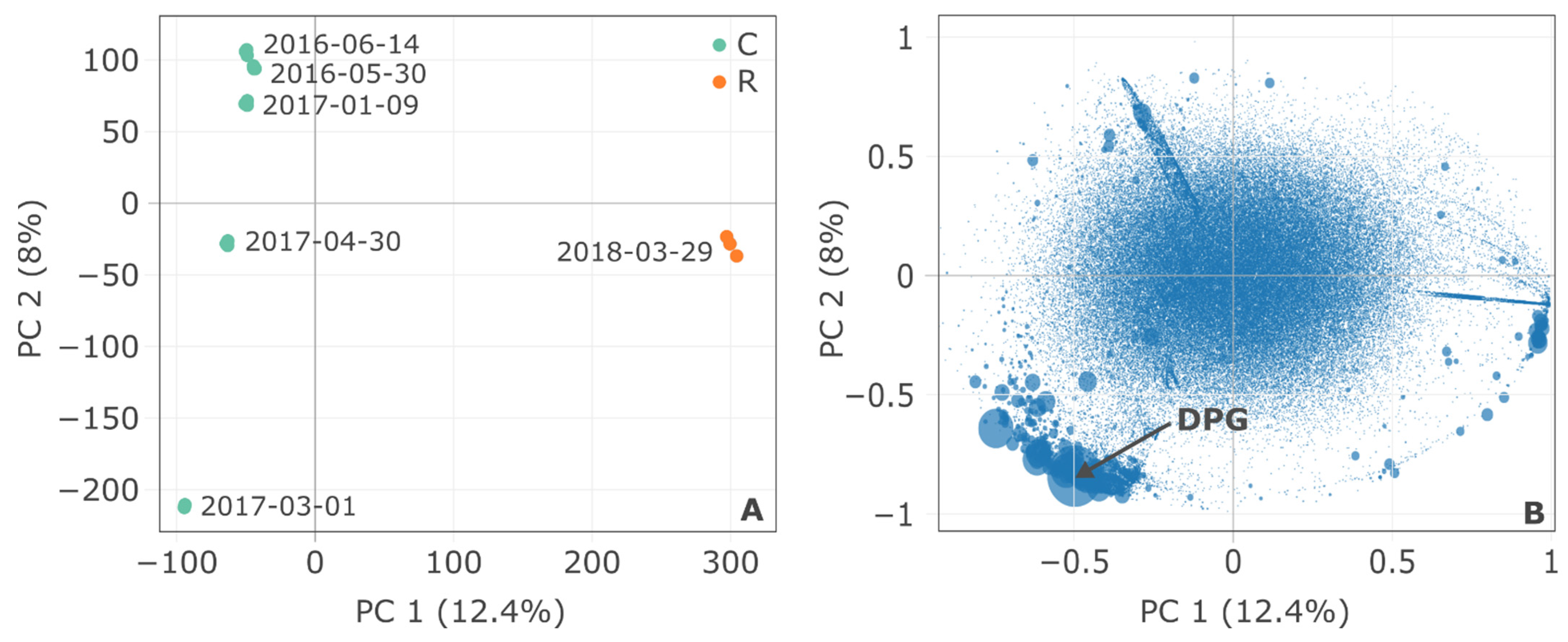
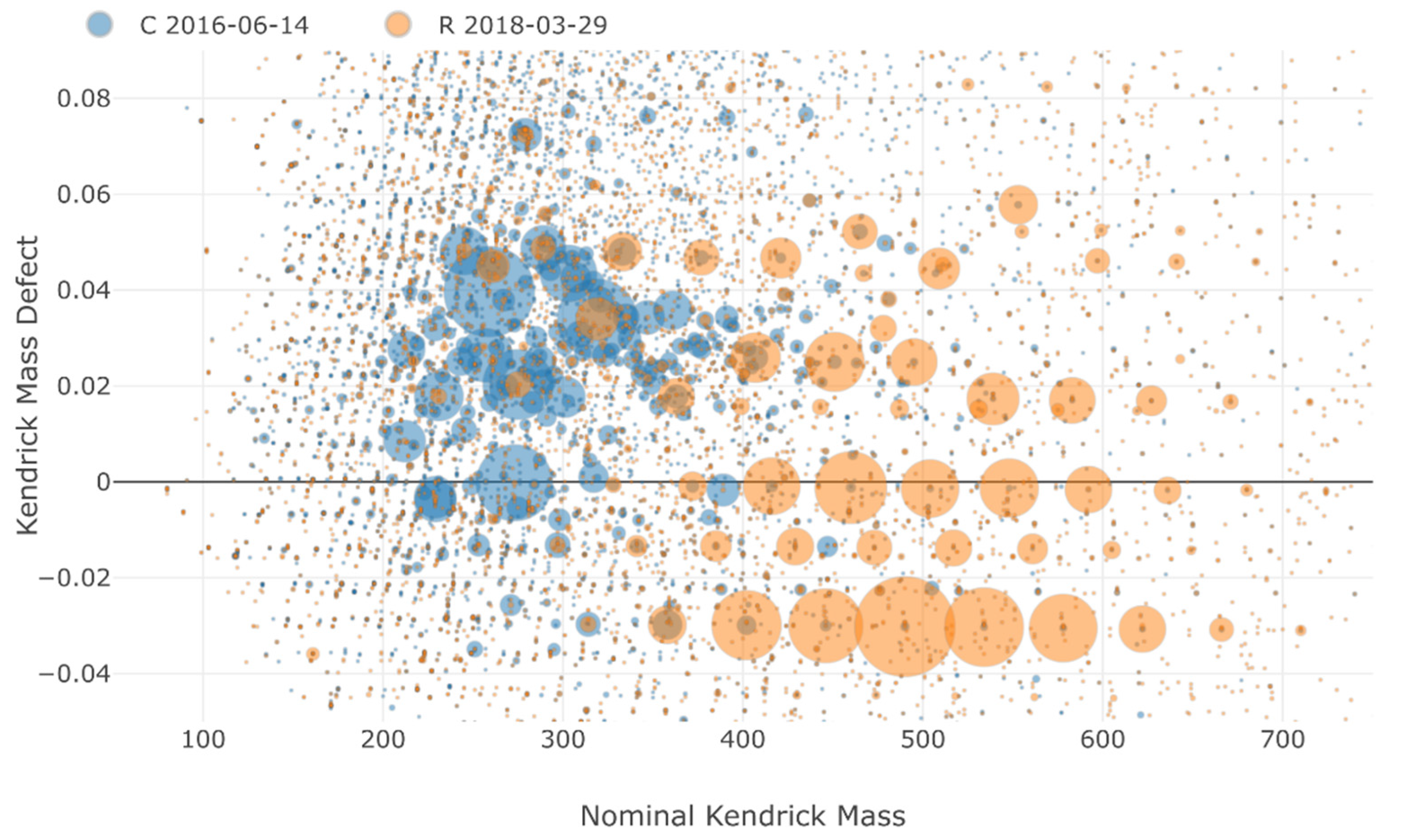
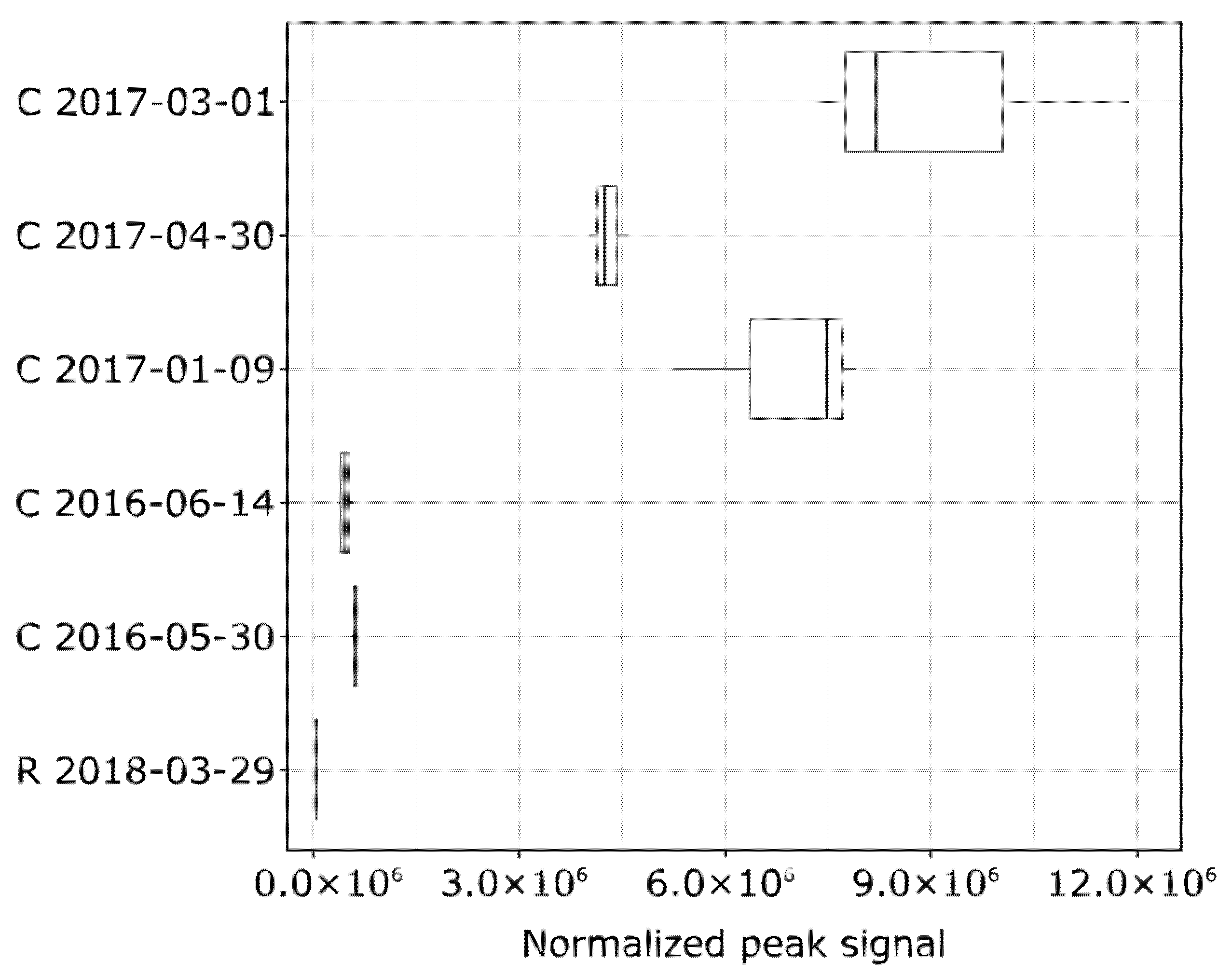
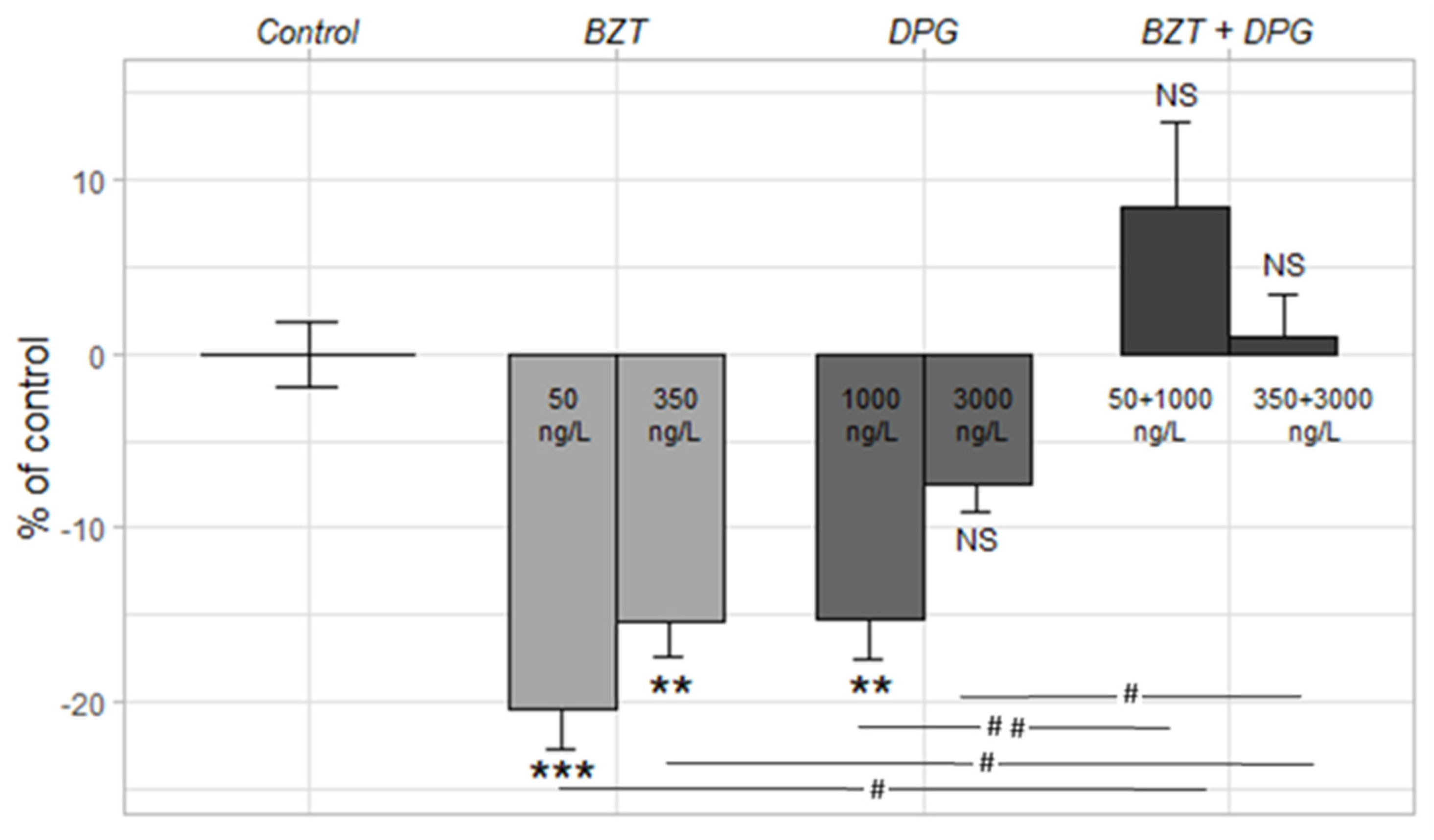
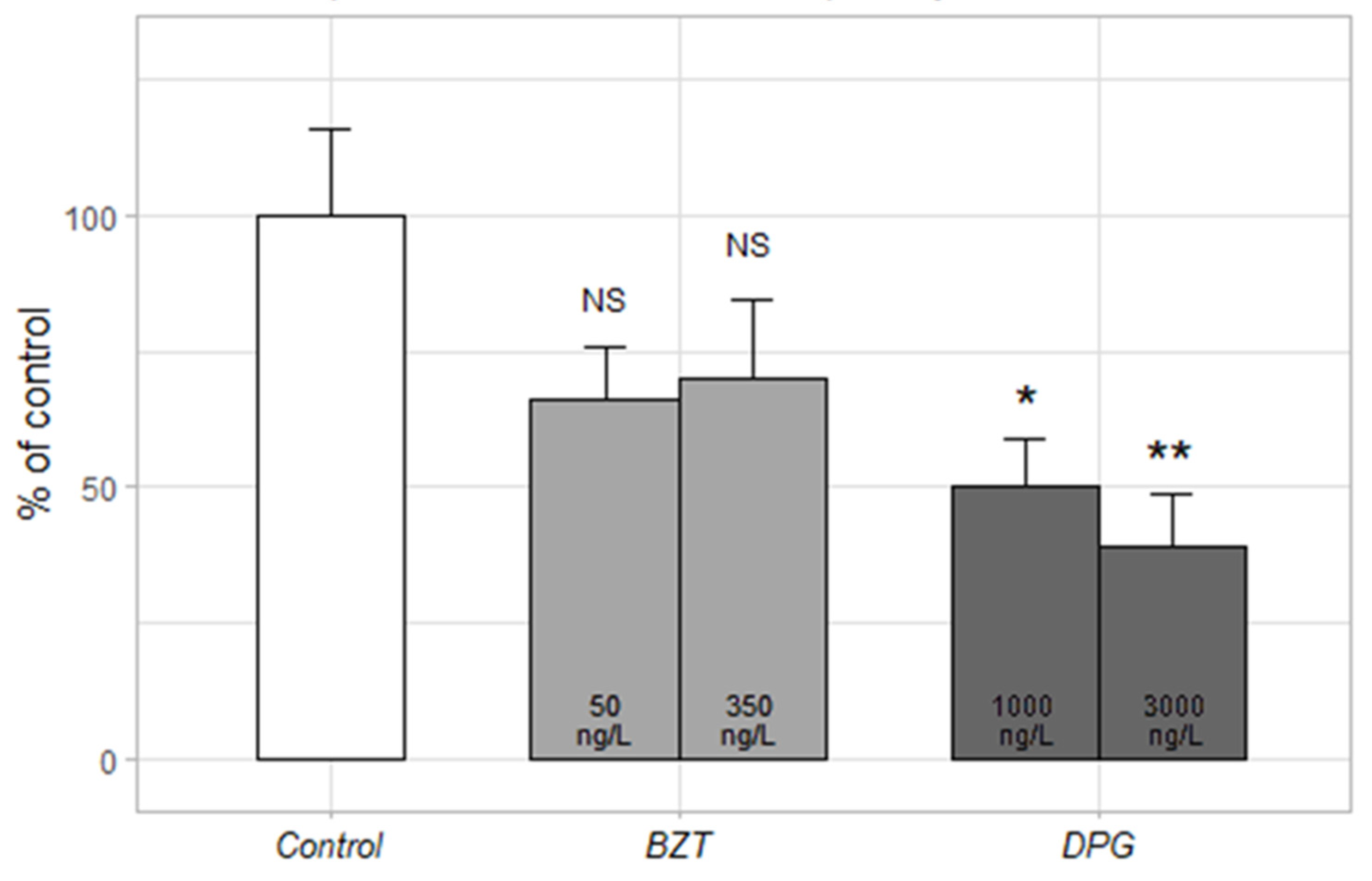
| Site | Sampling Date | TSS (mg/L) | pH | Cond. (µS/cm) | DOC (mg C/L) |
|---|---|---|---|---|---|
| C | all samples | 249.0 | 8.0 | 200.3 | 5.9 |
| (99.8–597.4) | (7.6–8.4) | (93.2–1852.6) | (3.6–11.5) | ||
| 2016-05-30 | 71.7 | 8.0 | 87.9 | 1.7 | |
| 2016-06-14 | 172.2 | 8.0 | 156.7 | 6.2 | |
| 2017-01-09 | 582.3 | 8.4 | 1949.5 | 5.7 | |
| 2017-03-01 | 932.8 | 8.4 | 530.5 | 5.6 | |
| 2017-04-30 | 231.9 | 7.8 | 304 | 10.7 | |
| 2017-06-27 | 733.6 | 7.8 | 93.8 | 8.5 | |
| 2017-06-28 | 532.5 | 8.1 | 265.2 | 14.2 | |
| R | all samples | 44.4 | 7.9 | 152.8 | 8.5 |
| (17.3–442.8) | (7.6–9.4) | (113.9–450.4) | (4.7–16.8) | ||
| 2017-09-14 | 51 | 7.9 | 122.9 | 8.5 | |
| 2018-03-29 | 738 | 9.0 | 465.0 | 13.7 |
| Concentrations (µg/L)—Median (Min–Max) | ||||||
|---|---|---|---|---|---|---|
| C | R | |||||
| Substances | All Campaigns | Samples for HRMS | Samples for Ecotoxicity | All Campaigns | Sample for HRMS | Sample for Ecotoxicity |
| Cu | 26 (14–42) | 27 (22–38) | 30 (23–38) | 11.1 (4.6–19.3) | 4.6 | NA |
| Zn | 32 (16–71) | 32 (22–66) | 32 (22–43) | 18.9 (2.4–53.5) | 2.4 | NA |
| Ni | 1.1 (06–4.5) | 1.3 (0.7–4.5) | 1.1 (0.8–1.4) | 1.5 (1.1–10.0) | 10.0 | NA |
| Pb | 0.4 (0.2–0.9) | 0.3 (0.2–0.5) | 0.3 (0.2–0.4) | 0.6 (0.3–1.4) | ND | NA |
| Ti | 4 (2–76) | 2 (2–76) | 3 (ND–3) | 28 (1–81) | 4 | NA |
| DEHP | 3.4 (0.9–12.1) | 2.5 (1.6–3.6) | 3.6 (0.9–4.7) | 1.2 (1.0–1.9) | 1.9 | ND |
| NP | 386 (99–752) | 307 (189–752) | 386 (99–752) | 477 (311–774) | 578 | 376 |
| OP | 126 (33–224) | 166 (117–194) | 114 (72–166) | 97 (32–223) | 141 | 52 |
| BPA | 280 (66–696) | 280 (225–499) | 265 (225–609) | 535 (306–716) | 397 | 716 |
| ∑PAHs | 61 (40–212) | 132 (60–212) | 61 (50–68) | 86 (29–115) | 86 | ND |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sandré, F.; Huynh, N.; Gromaire, M.-C.; Varrault, G.; Morin, C.; Moilleron, R.; Le Roux, J.; Garrigue-Antar, L. Road Runoff Characterization: Ecotoxicological Assessment Combined with (Non-)Target Screenings of Micropollutants for the Identification of Relevant Toxicants in the Dissolved Phase. Water 2022, 14, 511. https://doi.org/10.3390/w14040511
Sandré F, Huynh N, Gromaire M-C, Varrault G, Morin C, Moilleron R, Le Roux J, Garrigue-Antar L. Road Runoff Characterization: Ecotoxicological Assessment Combined with (Non-)Target Screenings of Micropollutants for the Identification of Relevant Toxicants in the Dissolved Phase. Water. 2022; 14(4):511. https://doi.org/10.3390/w14040511
Chicago/Turabian StyleSandré, Fidji, Nina Huynh, Marie-Christine Gromaire, Gilles Varrault, Christophe Morin, Régis Moilleron, Julien Le Roux, and Laure Garrigue-Antar. 2022. "Road Runoff Characterization: Ecotoxicological Assessment Combined with (Non-)Target Screenings of Micropollutants for the Identification of Relevant Toxicants in the Dissolved Phase" Water 14, no. 4: 511. https://doi.org/10.3390/w14040511







