Micropollutants in Urban Runoff from Traffic Areas: Target and Non-Target Screening on Four Contrasted Sites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites and Sampling Methods
2.2. Screening Methodology
2.2.1. Targeted Screening Methodology
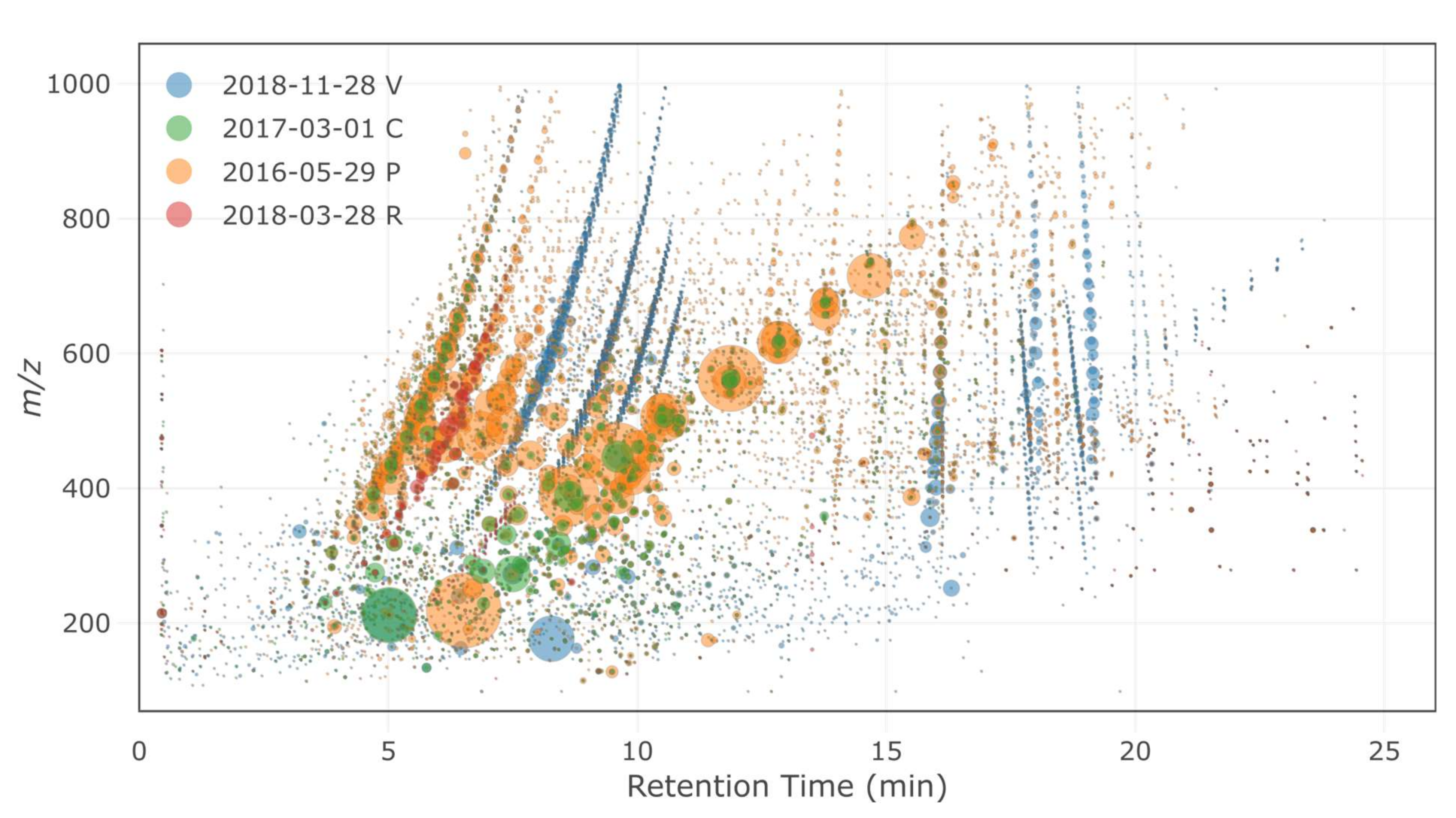
2.2.2. Non-Targeted Screening Methodology
2.3. Sample Characteristics
3. Results
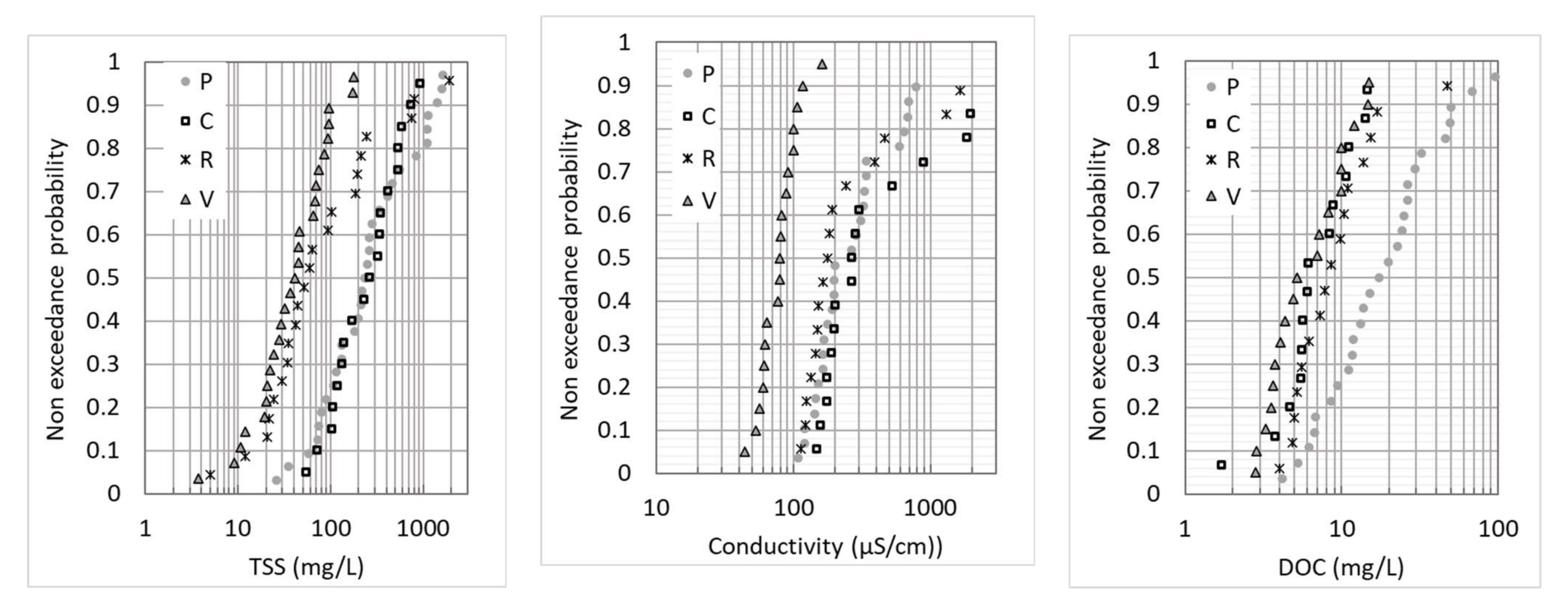
3.1. Physical–Chemical Characteristics
3.2. Micropollutants in the Road and Parking Lot Runoff—Prevalence and Concentration Ranges in the Targeted Screening
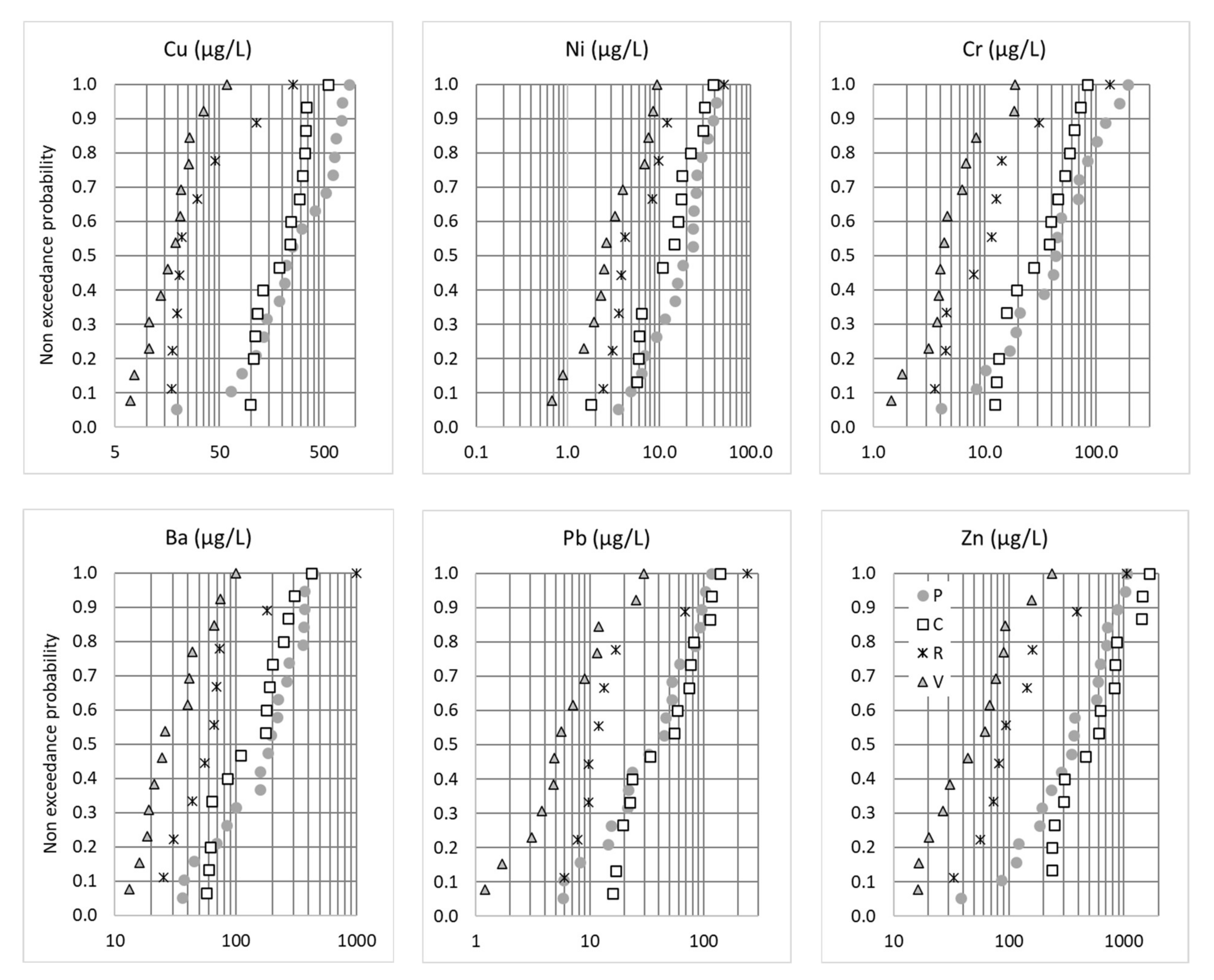
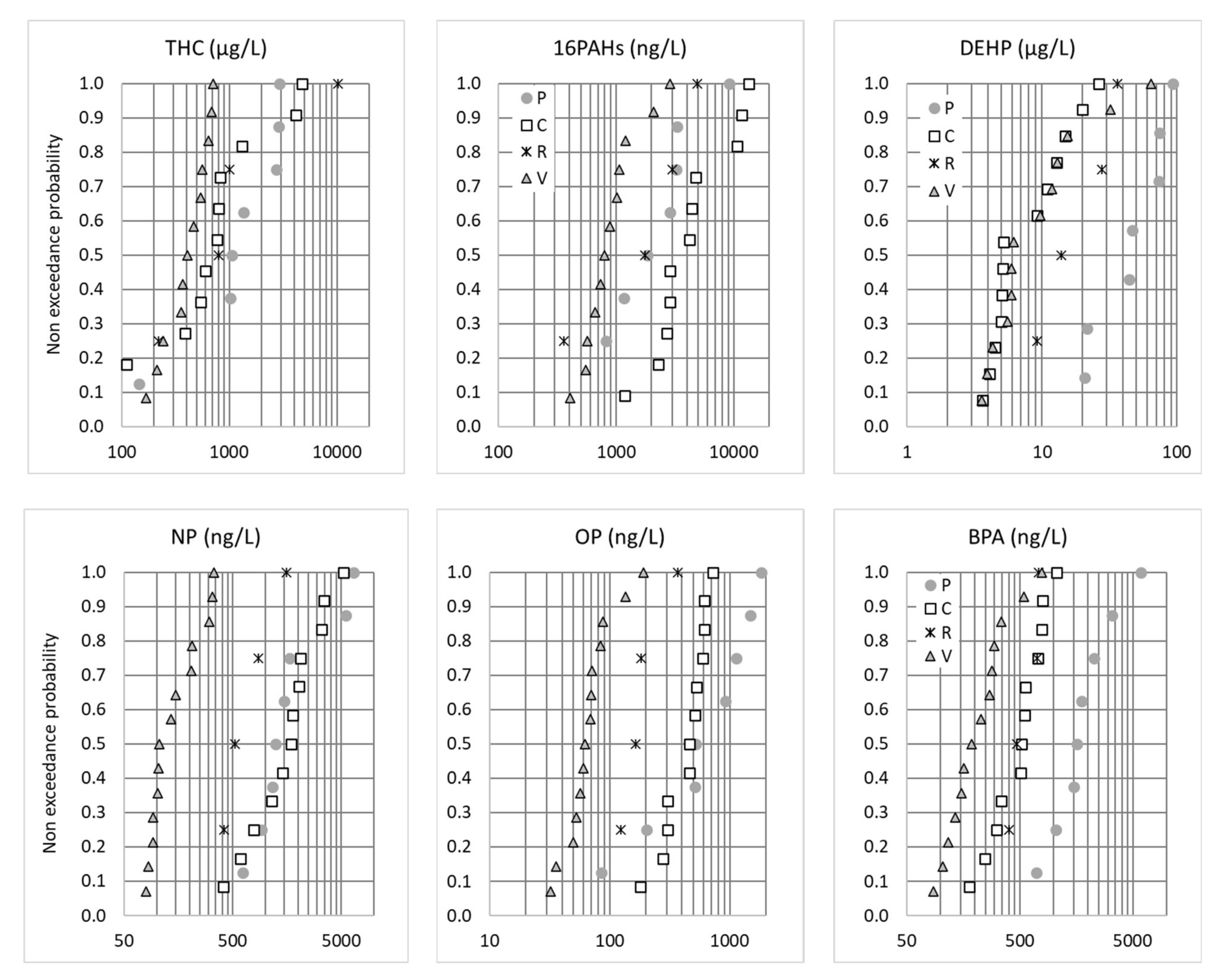
3.3. Inter-Site Variability of Concentrations—Targeted Screening
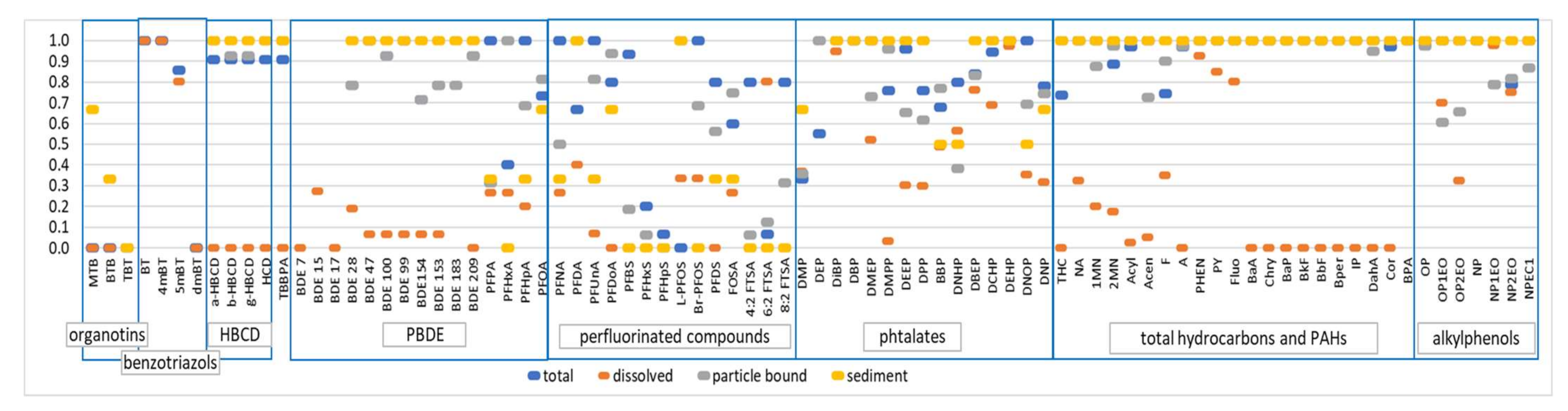
3.4. Non-Target Screening: Comparison of Site Signatures and New Compounds of Interest
4. Discussion
4.1. How Significant Is Road Runoff Contamination?
4.2. Distribution of Pollutants between Dissolved and Particulate Phase
4.3. Should We Promote On-Site Depollution Systems?
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pitt, R.; Clark, S.E. Integrated Storm-Water Management for Watershed Sustainability. J. Irrig. Drain. Eng. 2008, 134, 548–555. [Google Scholar] [CrossRef]
- Clark, S.E.; Burian, S.; Pitt, R.; Field, R. Urban Wet-Weather Flows. Water Environ. Res. 2006, 78, 1133–1192. [Google Scholar] [CrossRef]
- Revitt, D.M.; Lundy, L.; Coulon, F.; Fairley, M. The Sources, Impact and Management of Car Park Runoff Pollution: A Review. J. Environ. Manag. 2014, 146, 552–567. [Google Scholar] [CrossRef]
- Müller, A.; Österlund, H.; Marsalek, J.; Viklander, M. The Pollution Conveyed by Urban Runoff: A Review of Sources. Sci. Total Environ. 2020, 709, 136125. [Google Scholar] [CrossRef] [PubMed]
- Kayhanian, M.; Fruchtman, B.D.; Gulliver, J.S.; Montanaro, C.; Ranieri, E.; Wuertz, S. Review of Highway Runoff Characteristics: Comparative Analysis and Universal Implications. Water Res. 2012, 46, 6609–6624. [Google Scholar] [CrossRef]
- Bjorklund, K. Substance Flow Analyses of Phthalates and Nonylphenols in Stormwater. Water Sci. Technol. 2010, 62, 1154–1160. [Google Scholar] [CrossRef]
- Gasperi, J.; Sebastian, C.; Ruban, V.; Delamain, M.; Percot, S.; Wiest, L.; Mirande, C.; Caupos, E.; Demare, D.; Kessoo, M.D.; et al. Micropollutants in Urban Stormwater: Occurrence, Concentrations, and Atmospheric Contributions for a Wide Range of Contaminants in Three French Catchments. Environ. Sci. Pollut. Res. 2014, 21, 5267–5281. [Google Scholar] [CrossRef]
- Zgheib, S.; Moilleron, R.; Chebbo, G. Priority Pollutants in Urban Stormwater: Part 1 — Case of Separate Storm Sewers. Water Res. 2012, 46, 6683–6692. [Google Scholar] [CrossRef]
- Markiewicz, A.; Björklund, K.; Eriksson, E.; Kalmykova, Y.; Strömvall, A.-M.; Siopi, A. Emissions of Organic Pollutants from Traffic and Roads: Priority Pollutants Selection and Substance Flow Analysis. Sci. Total Environ. 2017, 580, 1162–1174. [Google Scholar] [CrossRef]
- Flanagan, K.; Branchu, P.; Boudahmane, L.; Caupos, E.; Demare, D.; Deshayes, S.; Dubois, P.; Meffray, L.; Partibane, C.; Saad, M.; et al. Field Performance of Two Biofiltration Systems Treating Micropollutants from Road Runoff. Water Res. 2018, 145, 562–578. [Google Scholar] [CrossRef]
- Froger, C.; Ayrault, S.; Evrard, O.; Monvoisin, G.; Bordier, L.; Lefèvre, I.; Quantin, C. Tracing the Sources of Suspended Sediment and Particle-Bound Trace Metal Elements in an Urban Catchment Coupling Elemental and Isotopic Geochemistry, and Fallout Radionuclides. Environ. Sci. Pollut. Res. 2018, 25, 28667–28681. [Google Scholar] [CrossRef] [PubMed]
- Tlili, K.; Labadie, P.; Alliot, F.; Bourges, C.; Desportes, A.; Chevreuil, M. Influence of Hydrological Parameters on Organohalogenated Micropollutant (Polybrominated Diphenyl Ethers and Polychlorinated Biphenyls) Behaviour in the Seine (France). Arch. Environ. Contam. Toxicol. 2012, 62, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Munoz, G.; Giraudel, J.-L.; Botta, F.; Lestremau, F.; Devier, M.-H.; Budzinski, H.; Labadie, P. Spatial Distribution and Partitioning Behavior of Selected Poly- and Perfluoroalkyl Substances in Freshwater Ecosystems: A French Nationwide Survey. Sci. Total Environ. 2015, 517, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Oturan, N.; van Hullebusch, E.D.; Zhang, H.; Mazeas, L.; Budzinski, H.; Le Menach, K.; Oturan, M.A. Occurrence and Removal of Organic Micropollutants in Landfill Leachates Treated by Electrochemical Advanced Oxidation Processes. Environ. Sci. Technol. 2015, 49, 12187–12196. [Google Scholar] [CrossRef] [PubMed]
- Munoz, G.; Fechner, L.C.; Geneste, E.; Pardon, P.; Budzinski, H.; Labadie, P. Spatio-Temporal Dynamics of per and Polyfluoroalkyl Substances (PFASs) and Transfer to Periphytic Biofilm in an Urban River: Case-Study on the River Seine. Environ. Sci. Pollut. Res. 2018, 25, 23574–23582. [Google Scholar] [CrossRef]
- Sandré, F.; Huynh, N.; Gromaire, M.-C.; Varrault, G.; Morin, C.; Moilleron, R.; Le Roux, J.; Garrigue-Antar, L. Road Runoff Characterization: Ecotoxicological Assessment Combined with (Non-)Target Screenings of Micropollutants for the Identification of Relevant Toxicants in the Dissolved Phase. Water 2022. special issue on “Research on Urban Runoff”. [Google Scholar]
- Schulze, S.; Zahn, D.; Montes, R.; Rodil, R.; Quintana, J.B.; Knepper, T.P.; Reemtsma, T.; Berger, U. Occurrence of Emerging Persistent and Mobile Organic Contaminants in European Water Samples. Water Res. 2019, 153, 80–90. [Google Scholar] [CrossRef]
- Challis, J.K.; Popick, H.; Prajapati, S.; Harder, P.; Giesy, J.P.; McPhedran, K.; Brinkmann, M. Occurrences of Tire Rubber-Derived Contaminants in Cold-Climate Urban Runoff. Environ. Sci. Technol. Lett. 2021, 8, 961–967. [Google Scholar] [CrossRef]
- Tian, Z.; Zhao, H.; Peter, K.T.; Gonzalez, M.; Wetzel, J.; Wu, C.; Hu, X.; Prat, J.; Mudrock, E.; Hettinger, R.; et al. A Ubiquitous Tire Rubber–Derived Chemical Induces Acute Mortality in Coho Salmon. Science 2021, 371, 185–189. [Google Scholar] [CrossRef]
- Wicke, D.; Matzinger, A.; Sonnenberg, H.; Caradot, N.; Schubert, R.-L.; Dick, R.; Heinzmann, B.; Dünnbier, U.; von Seggern, D.; Rouault, P. Micropollutants in Urban Stormwater Runoff of Different Land Uses. Water 2021, 13, 1312. [Google Scholar] [CrossRef]
- Göbel, P.; Dierkes, C.; Coldewey, W.G. Storm Water Runoff Concentration Matrix for Urban Areas. J. Contam. Hydrol. 2007, 91, 26–42. [Google Scholar] [CrossRef] [PubMed]
- Lundy, L.; Ellis, J.B.; Revitt, D.M. Risk Prioritisation of Stormwater Pollutant Sources. Water Res. 2012, 46, 6589–6600. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.; Welker, A.; Helmreich, B. Critical Review of Heavy Metal Pollution of Traffic Area Runoff: Occurrence, Influencing Factors, and Partitioning. Sci. Total Environ. 2016, 541, 895–919. [Google Scholar] [CrossRef] [PubMed]
- Peikertova, P.; Filip, P. Influence of the Automotive Brake Wear Debris on the Environment—A Review of Recent Research. SAE Int. J. Mater. Manuf. 2016, 9, 133–146. [Google Scholar] [CrossRef]
- Gietl, J.K.; Lawrence, R.; Thorpe, A.J.; Harrison, R.M. Identification of Brake Wear Particles and Derivation of a Quantitative Tracer for Brake Dust at a Major Road. Atmos. Environ. 2010, 44, 141–146. [Google Scholar] [CrossRef]
- Achten, C.; Kolb, A.; Püttmann, W. Methyl Tert-Butyl Ether (MTBE) in Urban and Rural Precipitation in Germany. Atmos. Environ. 2001, 35, 6337–6345. [Google Scholar] [CrossRef]
- Borden, R.C.; Black, D.C.; McBlief, K.V. MTBE and Aromatic Hydrocarbons in North Carolina Stormwater Runoff. Environ. Pollut. 2002, 118, 141–152. [Google Scholar] [CrossRef]
- Sablayrolles, C.; Breton, A.; Vialle, C.; Vignoles, C.; Montréjaud-Vignoles, M. Priority Organic Pollutants in the Urban Water Cycle (Toulouse, France). Water Sci. Technol. 2011, 64, 541. [Google Scholar] [CrossRef]
- Stachel, B.; Holthuis, J.-U.; Schulz, W.; Seitz, W.; Weber, W.H.; Tegge, K.-T.; Dobner, I. Treatment Techniques and Analysis of Stormwater Run-off from Roads in Hamburg, Germany. In Xenobiotics in the Urban Water Cycle; Fatta-Kassinos, D., Bester, K., Kümmerer, K., Eds.; Springer: Dordrecht, The Netherlands, 2010; Volume 16, pp. 445–461. ISBN 978-90-481-3508-0. [Google Scholar]
- INERIS. Synthèse Des Valeurs Réglementaires Pour Les Substances Chimiques, En Vigueur Dans l’eau, Les Denrées Alimentaires et Dans l’air En France Au 30 Juin 2020; INERIS: Paris, France, 2021; p. 97. [Google Scholar]
- Clara, M.; Windhofer, G.; Hartl, W.; Braun, K.; Simon, M.; Gans, O.; Scheffknecht, C.; Chovanec, A. Occurrence of Phthalates in Surface Runoff, Untreated and Treated Wastewater and Fate during Wastewater Treatment. Chemosphere 2010, 78, 1078–1084. [Google Scholar] [CrossRef]
- Yunker, M.B.; Macdonald, R.W.; Vingarzan, R.; Mitchell, R.H.; Goyette, D.; Sylvestre, S. PAHs in the Fraser River Basin: A Critical Appraisal of PAH Ratios as Indicators of PAH Source and Composition. Org. Geochem. 2002, 33, 489–515. [Google Scholar] [CrossRef]
- Tomy, G.T.; Budakowski, W.; Halldorson, T.; Whittle, D.M.; Keir, M.J.; Marvin, C.; MacInnis, G.; Alaee, M. Biomagnification of α- and γ-Hexabromocyclododecane Isomers in a Lake Ontario Food Web. Environ. Sci. Technol. 2004, 38, 2298–2303. [Google Scholar] [CrossRef] [PubMed]
- Ahmadireskety, A.; Da Silva, B.F.; Robey, N.M.; Douglas, T.E.; Aufmuth, J.; Solo-Gabriele, H.M.; Yost, R.A.; Townsend, T.G.; Bowden, J.A. Per- and Polyfluoroalkyl Substances (PFAS) in Street Sweepings. Environ. Sci. Technol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-K.; Kannan, K. Perfluorinated Acids in Air, Rain, Snow, Surface Runoff, and Lakes: Relative Importance of Pathways to Contamination of Urban Lakes. Environ. Sci. Technol. 2007, 41, 8328–8334. [Google Scholar] [CrossRef] [PubMed]
- Sternbeck, J.; Sjodin, A.; Andreasson, K. Metal Emissions from Road Traffic and the Influence of Resuspension - Results from Two Tunnel Studies. Atmos. Environ. 2002, 36, 4735–4744. [Google Scholar] [CrossRef]
- INERIS. Identification Des Sources Résiduelles d’alkylphénols; INERIS: Paris, France, 2015; p. 57. [Google Scholar]
- Lamprea, K.; Bressy, A.; Mirande-Bret, C.; Caupos, E.; Gromaire, M.-C. Alkylphenol and Bisphenol A Contamination of Urban Runoff: An Evaluation of the Emission Potentials of Various Construction Materials and Automotive Supplies. Environ. Sci. Pollut. Res. 2018, 25, 21887–21900. [Google Scholar] [CrossRef]
- Deshayes, S.; Gasperi, J.; Caupos, E.; Gromaire, M.-C. Evaluation du Potentiel d’Emission d’Alkylphénols, Bisphénol A et Phtalates par les Matériaux et Consommables Automobiles; Roulépur—Livrable 2.4; HAL: Ojhar, India, 2020; p. 49. [Google Scholar]
- Alves, C.A.; Vicente, A.M.P.; Calvo, A.I.; Baumgardner, D.; Amato, F.; Querol, X.; Pio, C.; Gustafsson, M. Physical and Chemical Properties of Non-Exhaust Particles Generated from Wear between Pavements and Tyres. Atmos. Environ. 2020, 224, 117252. [Google Scholar] [CrossRef]
- Staples, C.A.; Guinn, R.; Kramarz, K.; Lampi, M. Assessing the Chronic Aquatic Toxicity of Phthalate Ester Plasticizers. Hum. Ecol. Risk Assess. 2011, 17, 1057–1076. [Google Scholar] [CrossRef]
- Zhao, L.; Zhou, M.; Zhang, T.; Sun, H. Polyfluorinated and Perfluorinated Chemicals in Precipitation and Runoff from Cities Across Eastern and Central China. Arch. Environ. Contam. Toxicol. 2013, 64, 198–207. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, X.; Wu, L.; Wang, T.; Zhao, J.; Zhang, Y.; Men, Z.; Mao, H. Occurrence of Benzothiazole and Its Derivates in Tire Wear, Road Dust, and Roadside Soil. Chemosphere 2018, 201, 310–317. [Google Scholar] [CrossRef]
- Brudler, S.; Rygaard, M.; Arnbjerg-Nielsen, K.; Hauschild, M.Z.; Ammitsøe, C.; Vezzaro, L. Pollution Levels of Stormwater Discharges and Resulting Environmental Impacts. Sci. Total Environ. 2019, 663, 754–763. [Google Scholar] [CrossRef]
- Petrucci, G.; Gromaire, M.-C.; Shorshani, M.F.; Chebbo, G. Nonpoint Source Pollution of Urban Stormwater Runoff: A Methodology for Source Analysis. Environ. Sci. Pollut. Res. 2014, 21, 10225–10242. [Google Scholar] [CrossRef] [PubMed]
- Chèvre, N.; Gremaud, D.; Guignard, C.; Rossi, L.; De Alencastro, L.; Bader, H.-P.; Scheidegger, R. Substance Flow Analysis: A Management Tool for Heavy Metals in Urban Water Systems. In Proceedings of the International Conference NOVATECH 2010, Lyon, France, 28 June–1 July 2010. [Google Scholar]
- Le Pape, P.; Ayrault, S.; Quantin, C. Trace Element Behavior and Partition versus Urbanization Gradient in an Urban River (Orge River, France). J. Hydrol. 2012, 472–473, 99–110. [Google Scholar] [CrossRef]
- Meybeck, M.; Lestel, L.; Bonté, P.; Moilleron, R.; Colin, J.L.; Rousselot, O.; Hervé, D.; de Pontevès, C.; Grosbois, C.; Thévenot, D.R. Historical Perspective of Heavy Metals Contamination (Cd, Cr, Cu, Hg, Pb, Zn) in the Seine River Basin (France) Following a DPSIR Approach (1950-2005). Sci. Total Environ. 2007, 375, 204–231. [Google Scholar] [CrossRef] [PubMed]
- Zoller, W.H.; Gladney, E.S.; Duce, R.A. Atmospheric Concentrations and Sources of Trace Metals at the South Pole. Science 1974, 183, 198–200. [Google Scholar] [CrossRef]
- Ayrault, S.; Priadi, C.R.; Pape, P.L.; Bonté, P. Occurrence, Sources and Pathways of Antimony and Silver in an Urban Catchment. In Proceedings of the Urban Environment; Rauch, S., Morrison, G., Norra, S., Schleicher, N., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2013; pp. 425–435. [Google Scholar]
- Ayrault, S.; Priadi, C.R.; Evrard, O.; Lefèvre, I.; Bonté, P. Silver and Thallium Historical Trends in the Seine River Basin. J. Environ. Monit. 2010, 12, 2177–2185. [Google Scholar] [CrossRef]
- Le Cloarec, M.-F.; Bonte, P.H.; Lestel, L.; Lefèvre, I.; Ayrault, S. Sedimentary Record of Metal Contamination in the Seine River during the Last Century. Phys. Chem. Earth Parts ABC 2011, 36, 515–529. [Google Scholar] [CrossRef]
- Gromaire-Mertz, M.C.; Garnaud, S.; Gonzalez, A.; Chebbo, G. Characterisation of Urban Runoff Pollution in Paris. Water Sci. Technol. 1999, 39, 1–8. [Google Scholar] [CrossRef]
- Filella, M.; Belzile, N.; Chen, Y.-W. Antimony in the Environment: A Review Focused on Natural Waters: I. Occurrence. Earth-Sci. Rev. 2002, 57, 125–176. [Google Scholar] [CrossRef]
- Flanagan, K.; Branchu, P.; Boudahmane, L.; Caupos, E.; Demare, D.; Deshayes, S.; Dubois, P.; Meffray, L.; Partibane, C.; Saad, M.; et al. Retention and Transport Processes of Particulate and Dissolved Micropollutants in Stormwater Biofilters Treating Road Runoff. Sci. Total Environ. 2019, 656, 1178–1190. [Google Scholar] [CrossRef]
- Hong, Y.; Bonhomme, C.; Le, M.-H.; Chebbo, G. A New Approach of Monitoring and Physically-Based Modelling to Investigate Urban Wash-off Process on a Road Catchment near Paris. Water Res. 2016, 102, 96–108. [Google Scholar] [CrossRef]
- Li, H.; Davis, A.P. Urban Particle Capture in Bioretention Media. I: Laboratory and Field Studies. J. Environ. Eng.-Asce 2008, 134, 409–418. [Google Scholar] [CrossRef]
- LeFevre, G.H.; Paus, K.H.; Natarajan, P.; Gulliver, J.S.; Novak, P.J.; Hozalski, R.M. Review of Dissolved Pollutants in Urban Storm Water and Their Removal and Fate in Bioretention Cells. J. Environ. Eng. 2015, 141, 04014050. [Google Scholar] [CrossRef]
- Lange, K.; Österlund, H.; Viklander, M.; Blecken, G.-T. Metal Speciation in Stormwater Bioretention: Removal of Particulate, Colloidal and Truly Dissolved Metals. Sci. Total Environ. 2020, 724, 138121. [Google Scholar] [CrossRef]
- Søberg, L.C.; Viklander, M.; Blecken, G.-T. Do Salt and Low Temperature Impair Metal Treatment in Stormwater Bioretention Cells with or without a Submerged Zone? Sci. Total Environ. 2017, 579, 1588–1599. [Google Scholar] [CrossRef]
- GeoSyntec Consultants; Urban Water Resources Research Council of ASCE. Urban Stormwater BMP Performance Monitoring; GeoSyntec Consultants; Washington, DC, USA; Urban Water Resources Research Council of ASCE: Washington, DC, USA, 2002; p. 248. Available online: https://www3.epa.gov/npdes/pubs/montcomplete.pdf (accessed on 16 December 2021).
- Larm, T.; Wahlsten, A. Applying the Parameter “Irreducible Concentration” in Modelling of Stormwater Treatment Facilities. J. Civ. Eng. Archit. 2019, 13, 469–476. [Google Scholar] [CrossRef]
- Tedoldi, D.; Chebbo, G.; Pierlot, D.; Kovacs, Y.; Gromaire, M.-C. Assessment of Metal and PAH Profiles in SUDS Soil Based on an Improved Experimental Procedure. J. Environ. Manag. 2017, 202, 151–166. [Google Scholar] [CrossRef]
- LeFevre, G.H.; Hozalski, R.M.; Novak, P.J. The Role of Biodegradation in Limiting the Accumulation of Petroleum Hydrocarbons in Raingarden Soils. Water Res. 2012, 46, 6753–6762. [Google Scholar] [CrossRef]
- Costello, D.M.; Hartung, E.W.; Stoll, J.T.; Jefferson, A.J. Bioretention Cell Age and Construction Style Influence Stormwater Pollutant Dynamics. Sci. Total Environ. 2020, 712, 135597. [Google Scholar] [CrossRef]

| Site Name | Paris (P) | Compans (C) | Rosny-Sous-Bois (R) | Villeneuve-le-Roi (V) |
|---|---|---|---|---|
| Location | Central Paris | Suburban area, close to Roissy airport | Town center, suburbs | Residential area, suburbs, close to Orly airport |
| Characteristics of the traffic area | One-way urban boulevard on the Seine riverbank 2 lanes 50 km/h | Departmental road Smooth traffic flow 2 × 2 lanes 90 km/h | Town centre street Traffic light 2 × 1 lane + side parking 50 km/h | Residential parking lot |
| Daily traffic load | 40,000 veh/d | 2 × 11,000 veh/d High HGV * traffic | Low and variable traffic (<3000 veh/d) | Very low traffic 1 to 3 rotations per day |
| Catchment area | 1045 m2 | 945 m2 | 3410 m2 | 730 m2 |
| Inorganic Elements | Organic Micropollutants | ||||
|---|---|---|---|---|---|
| Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | |
| Paris (P) | 11–19 | 4 | 7–8 | 3 | 0 |
| Compans (C) | 11–15 | 4 | 11–12 | 4 | 3 |
| Rosny-sous-Bois (R) | 9–13 | 4 | 4 | 2–4 | 2 |
| Villeneuve-le-Roi (V) | 11–18 | 3–4 | 6–11 | 2–4 | 2 |
| Rain Depth (mm) | Duration (h) | Imax6min (mm/h) | ADW (day) | Runoff Volume (m3) | |
|---|---|---|---|---|---|
| Paris (P) | 4.2 (0.2–62) | 4 (0.7–38) | 6.0 (0.8–67) | 0.7 (0.03–13) | 2.0 (0.2–29) |
| Compans (C) | 9.9 (4.3–47) | 19 (2–40) | 12 (4–43) | 1 (0.1–18) | 12 (3.2–36) |
| Rosny-sous-Bois (R) | 13.8 (0.6–50) | 17 (0.8–54) | 10 (1.2–124) | 0.3 (0.0–4) | NA |
| Villeneuve-le-Roi (V) | 11.4 (2.8–29) | 12 (1–124) | 6.8 (2.3–45) | 1.6 (0.2–21) | 5.5 (1.4–19) |
| Site | Group | pH | Electrical Cond. (µS/cm) | TSS (mg/L) | DOC (mg/L) | POC (%) |
|---|---|---|---|---|---|---|
| P | 2,4,5 | 7.2 (7.0–8.0) | 152 (88–642) | 191 (26–1237) | 13.7 (4.2–68.,4) | 24.4 (10.8–37.8) |
| 1,3 | 7.6 (6.9–9.8) | 198 (88–786) | 235 (28–1620) | 16.9 (4.2–95.2) | 24.1 (7.8–38.0) | |
| C | 2,4,5 | 7.9 (7.4–8.4) | 182 (88–1950) | 123 (54–535) | 5.6 (1.7–7.6) | 16.0 (11.4–18.0) |
| 1,3 | 8.0 (7.6–8.4) | 234 (88–1950) | 296 (72–933) | 6.1 (1.7–14.7) | 14.2 (8.4–18.0) | |
| R | 2,4,5 | 7.6 (7.5–9.4) | 135 (105–192) | 34 (5–214) | 9.9 (4.3–16.9) | 16.0 (4.6–33.0) |
| 1,3 | 7.9 (7.6–11) | 165 (105–1643) | 58 (5–1901) | 8.2 (4.0–47.6) | 12.8 (4.6–33.0) | |
| V | 2,4,5 | 7.6 (6.6–8.1) | 68 (44–90) | 45 (4–177) | 9.9 (4.1–12.4) | 17.9 (14.6–33.0) |
| 1,3 | 7.7 (6.9–8.3) | 81 (61–162) | 26 (9–174) | 4.1 (2.8–15.0) | 24.1 (14.2–29.6) |
| Family | Substance | Abbv. | AA-EQS 1 | N | Quantif. | Q10 | Median | Q90 | Literature 2 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Group 1 | Majors (mg/L) | Aluminum | Al | 57 | 96% | 0.81 | 5.0 | 19 | 0.15–4.9 | |
| Calcium | Ca | 57 | 100% | 15.0 | 32 | 74 | ||||
| Iron | Fe | 56 | 100% | 0.91 | 5.5 | 23 | 0.33–3.7 | |||
| Magnesium | Mg | 0.1 | 57 | 100% | 0.87 | 2.4 | 7.1 | |||
| Phosphor | P | 56 | 95% | 0.15 | 0.51 | 1.5 | ||||
| Potassium | K | 57 | 100% | 1.5 | 4.8 | 12 | ||||
| Sodium | Na | 5 | 57 | 100% | 1.7 | 9.3 | 90 | |||
| Major (µg/L) | Barium | Ba | 57 | 100% | 25 | 74 | 361 | |||
| Manganese | Mn | 58 | 100% | 21 | 90 | 358 | 11–659 | |||
| Strontium | Sr | 57 | 100% | 44 | 120 | 246 | ||||
| Titanium | Ti | 2 | 58 | 98% | 75 | 405 | 1594 | 13–179 | ||
| Trace metals (µg/L) | Arsenate | As | 0.83 | 57 | 100% | 0.6 | 2.3 | 6.8 | 0.7–12 | |
| Cadmium | Cd | 0.09 | 54 | 85% | 0.1 | 0.1 | 1.0 | 0.05–37 | ||
| Chromium | Cr | 3.4 | 56 | 100% | 3.9 | 17 | 84 | 1–105 | ||
| Cobalt | Co | 0.3 | 55 | 89% | 0.3 | 1.6 | 6.5 | 1.5–13 | ||
| Copper | Cu | 1 | 57 | 100% | 15 | 111 | 570 | 6–430 | ||
| Molybden. | Mo | 56 | 95% | 0.6 | 4.1 | 22 | ||||
| Nickel | Ni | 4 | 57 | 100% | 2.4 | 8.9 | 32 | 2–145 | ||
| Lead | Pb | 1.2 | 57 | 100% | 5.3 | 21 | 98 | 1.4–380 | ||
| Vanadium | V | 2.5 | 57 | 100% | 3.6 | 12 | 36 | 16 | ||
| Zinc | Zn | 7.8 | 57 | 100% | 36 | 236 | 932 | 21–2234 | ||
| Group 2 | Antimony | Sb | 0.6 | 15 | 100% | 1.3 | 7.7 | 39.8 | 0.8–10.7 | |
| Silver | Ag | 15 | 100% | 0.04 | 0.1 | 0.3 | ||||
| Rare earths (µg/L) | Cesium | Cs | 15 | 100% | 0.3 | 1.1 | 2.9 | |||
| Rubidium | Rb | 15 | 100% | 6.1 | 14.1 | 33.6 | ||||
| Yttrium | Y | 16 | 100% | 33 | 49 | 80 | ||||
| Lanthanum | La | 15 | 100% | 19 | 37 | 63 | ||||
| Cerium | Ce | 16 | 100% | 32 | 54 | 127 | ||||
| Praseodym. | Pr | 16 | 100% | 5 | 9 | 16 | ||||
| Neodymium | Nd | 16 | 100% | 24 | 41 | 78 | ||||
| Samarium | Sm | 16 | 100% | 10 | 16 | 23 | ||||
| Europium | Eu | 16 | 100% | 3.4 | 5.7 | 9.4 | ||||
| Gadolinium | Ga | 16 | 100% | 0.9 | 1.5 | 3.0 | ||||
| Terbium | Tb | 16 | 100% | 5.5 | 9.3 | 15.8 | ||||
| Dysprosium | Dy | 16 | 100% | 4.8 | 8.0 | 13.6 | ||||
| Holmium | Ho | 16 | 100% | 1.2 | 1.7 | 3.4 | ||||
| Thulium | Tm | 16 | 100% | 3.2 | 5.1 | 8.0 | ||||
| Erbium | Er | 16 | 100% | 0.5 | 0.7 | 1.0 | ||||
| Ytterbium | Yb | 16 | 100% | 2.8 | 4.2 | 7.5 | ||||
| Lutecium | Lu | 16 | 100% | 0.4 | 0.7 | 1.1 | ||||
| Substance | Abbv. | EQS 1 | n | Quantif | Q10 | Median | Q90 | Literature 2 | |
|---|---|---|---|---|---|---|---|---|---|
| Phtalates (µg/L) | dimethyl phthalate | DMP | 38 | 55% | 0.1 | 0.3 | 0.8 | <0.01–0.08 | |
| diisobutyl phthalate | DiBP | 36 | 100% | 1.1 | 3.3 | 8.3 | |||
| dibutyl phthalate | DBP | 10 | 38 | 100% | 0.6 | 1.3 | 2.8 | <0.05–0.96 | |
| dimethoxyethyl phthalate | DMEP | 25 | 76% | 0.1 | 0.8 | 3.7 | |||
| di-4-methyl-2-pentyl phthalate | DMPP | 25 | 96% | 0.2 | 0.5 | 4.4 | |||
| diethoxyethyl phthalate | DEEP | 25 | 76% | 0.04 | 0.3 | 1.4 | |||
| di-n-pentyl phthalate | DPP | 25 | 68% | 0.02 | 0.2 | 2.6 | |||
| butylbenzyl phthalate | BBP | 7.5 | 25 | 80% | 0.1 | 0.3 | 0.7 | <0.01–0.08 | |
| di-n-hexyl phthalate | DNHP | 25 | 84% | 0.1 | 1.3 | 2.1 | |||
| di-2-butoxyethyl phthalate | DBEP | 18 | 94% | 0.1 | 0.7 | 3.5 | |||
| dicyclohexyl phthalate | DCHP | 25 | 100% | 0.6 | 1.8 | 6.4 | |||
| di-2-ethylhexyl phthalate | DEHP | 1.3 | 37 | 100% | 4.2 | 11.9 | 53.5 | 0.38–78 | |
| di-n-octyl-phthalate | DNOP | 23 | 78% | 0.2 | 1.5 | 5.1 | <0.01–1.1 | ||
| dinonyl phtalate | DNP | 38 | 74% | 0.1 | 0.5 | 2.4 | |||
| Total hydrocarbons (ng/L) | THC | 35 | 100% | 0.2 × 106 | 0.7 × 106 | 2.9 × 106 | |||
| PAHs (ng/L) | Naphthalene | Nap | 2000 | 35 | 100% | 10 | 34 | 152 | |
| 1-methylnaphtalene | 1MN | 35 | 89% | 2 | 9 | 54 | |||
| 2-methylnaphtalene | 2MN | 35 | 97% | 5 | 18 | 78 | |||
| Acenaphthylene | Acyl | 35 | 100% | 10 | 32 | 140 | |||
| Acenaphthene | Acen | 35 | 74% | 4 | 11 | 37 | |||
| Fluorene | F | 35 | 97% | 5 | 17 | 59 | |||
| Anthracene | A | 100 | 35 | 100% | 12 | 45 | 143 | ||
| Phenanthrene | PHEN | 35 | 100% | 65 | 162 | 522 | |||
| Pyrene | PY | 35 | 100% | 128 | 404 | 1573 | |||
| Fluoranthene | Fluo | 6.3 | 35 | 100% | 75 | 302 | 1368 | ||
| Benzo(a)anthracene | BaA | 35 | 100% | 24 | 127 | 390 | |||
| Chrysene | Chry | 35 | 100% | 39 | 159 | 690 | |||
| Benzo(a)pyrene | BaP | 0.17 | 35 | 100% | 23 | 91 | 282 | ||
| Benzo(k)fluoranthene | BkF | 35 | 100% | 19 | 68 | 219 | |||
| Benzo(b)fluoranthene | BbF | 35 | 100% | 56 | 213 | 837 | |||
| Benzo(ghi)perylene | Bper | 35 | 100% | 37 | 166 | 665 | |||
| Indeno(1,2,3-cd)pyrene | IP | 35 | 100% | 25 | 113 | 329 | |||
| Dibenzo(ah)anthracene | DahA | 35 | 97% | 8 | 47 | 115 | |||
| Coronene | Cor | 35 | 100% | 15 | 51 | 429 | |||
| ∑16 PAH | ∑16 PAH | 35 | 100% | 604 | 2068 | 7481 | 300–6000 | ||
| Alkylphenols (ng/L) | Bisphenol A | BPA | 1600 | 38 | 100% | 148 | 513 | 1657 | <200–2500 |
| 4-tert-octylphenol | OP | 100 | 38 | 100% | 56 | 197 | 780 | 110–1900 | |
| Octylphenol-mono-ethoxylate | OP1EO | 38 | 79% | 12 | 31 | 159 | |||
| Octylphenol-di-ethoxylate | OP2EO | 38 | 79% | 3 | 8 | 70 | |||
| Nonylphenol | NP | 300 | 38 | 100% | 99 | 699 | 3375 | 170–3600 | |
| Nonylphenol-mono-ethoxylate | NP1EO | 38 | 100% | 34 | 125 | 668 | |||
| Nonylphenol-di-ethoxylate | NP2EO | 38 | 89% | 40 | 141 | 719 | |||
| Nonylphenoxy acetic acid | NP1EC | 38 | 100% | 25 | 82 | 447 | |||
| Family | Substance | Acronym | EQS 1 | N | Quantif. | C10 | Median | C90 |
|---|---|---|---|---|---|---|---|---|
| Hexabromocyclododecane (ng/L) | a-hexabromocyclododecane | a-HBCDD | 11 | 91% | 0.9 | 1.4 | 11 | |
| b-hexabromocyclododecane | b-HBCDD | 11 | 91% | 0.1 | 0.3 | 1.6 | ||
| g-hexabromocyclododecane | g-HBCDD | 11 | 91% | 0.3 | 1.1 | 2.4 | ||
| Sum | HBCDD | 1.6 | 11 | 91% | 1.5 | 2.9 | 14 | |
| Tetrabromobisphenol A (ng/L) | TBBPA | 260 | 11 | 91% | 0.1 | 0.5 | 1.7 | |
| Polybrominated biphenyl ethers (ng/L) | BDE 28 | BDE 28 | Σ6BDE 0.5 | 14 | 79% | 0.02 | 0.05 | 0.1 |
| BDE 47 | BDE 47 | 14 | 100% | 0.05 | 0.2 | 0.4 | ||
| BDE 100 | BDE 100 | 14 | 93% | 0.02 | 0.1 | 0.1 | ||
| BDE 99 | BDE 99 | 14 | 100% | 0.08 | 0.3 | 0.7 | ||
| BDE154 | BDE154 | 14 | 71% | 0.02 | 0.1 | 0.1 | ||
| BDE 153 | BDE 153 | 14 | 79% | 0.03 | 0.1 | 0.3 | ||
| BDE 209 | BDE 209 | 14 | 93% | 3.0 | 20 | 72 | ||
| Perfluoroalkyl carboxylic acids (ng/L) | perfluoropentanoic acid | PFPeA | 15 | 40% | 0.40 | 17 | 81 | |
| perfluorohexanoic acid | PFHxA | - | 15 | 100% | 0.03 | 0.5 | 22 | |
| perfluoroheptanoic acid | PFHpA | 15 | 73% | 0.002 | 0.04 | 15 | ||
| perfluorooctanoc acid | PFOA | 15 | 100% | 0.20 | 1.9 | 34 | ||
| perfluoronoanoic acid | PFNA | 15 | 67% | 0.05 | 0.2 | 5.4 | ||
| perfluorodecanoique acid | PFDA | 15 | 100% | 0.18 | 0.6 | 8.4 | ||
| perfluoroundecanoic acid | PFUnA | 15 | 80% | 0.02 | 0.2 | 1.0 | ||
| perfluorododecanoic acid | PFDoA | 15 | 93% | 0.10 | 0.4 | 1.4 | ||
| Perfluoroalkylsulfonic acids (ng/L) | perfluorobutane sulfonic acid | PFBS | 15 | 20% | 0.01 | 0.05 | 0.41 | |
| perfluorohexane sulfonic acid | PFHxS | 15 | 7% | 0.02 | 0.02 | 0.02 | ||
| perfluorooctane sulfonic acid, linear isomer | L-PFOS | 0.65 | 15 | 100% | 0.15 | 0.55 | 2.73 | |
| perfluorooctane sulfonic acid, sum of branched isomer | Br-PFOS | 15 | 80% | 0.06 | 0.28 | 0.93 | ||
| perfluorodecane sulfonic acid | PFDS | 15 | 60% | 0.02 | 0.05 | 0.09 | ||
| Perfluorooctane sulfonamide (ng/L) | FOSA | 15 | 80% | 0.02 | 0.1 | 0.6 | ||
| Fluorotelomere sulfonates (ng/L) | 4:2 fluorotelomere sulfonate | 4:2 FTSA | 15 | 7% | 0.1 | 0.1 | 0.1 | |
| 6:2 fluorotelomere sulfonate | 6:2 FTSA | 15 | 80% | 0.6 | 1.8 | 4.0 | ||
| 8:2 fluorotelomere sulfonate | 8:2 FTSA | 15 | 33% | 0.0 | 0.1 | 0.3 | ||
| Benzotriazoles (µg/L) | 1H-Benzotriazole | BT | 7 | 100% | 0.5 | 1.4 | 2.2 | |
| 4-Methyl- 1H-benzotriazole | 4mBT | 7 | 100% | 0.2 | 1.0 | 1.4 | ||
| 5-Methyl- 1H-benzotriazole | 5mBT | 7 | 86% | 0.2 | 0.8 | 1.2 | ||
| Substance | CAS | Raw Formula | Observed Ion (m/z) | Mass Error (ppm) | Observed RT (min) | Observed CCS (Å2) | Confidence Level * |
|---|---|---|---|---|---|---|---|
| 1,3-Diphenylguanidine | 102-06-7 | C13H13N3 | 212.1177 | −2.4 | 5.02 | 143.5 | 1 |
| 1,3-Di-o-tolylguanidine | 97-39-2 | C15H17N3 | 240.1487 | −3.4 | 6.43 | 151.0 | 2 |
| 6PPD-quinone | - | C18H22N2O2 | 299.1744 | −3.1 | 14.71 | 174.3 | 2 |
| Benzotriazole | 95-14-7 | C6H5N3 | 120.0551 | −4.1 | 4.14 | 113.0 | 1 |
| 2-Hydroxybenzothiazole | 934-34-9 | C7H5NOS | 152.0158 | −4.1 | 6.76 | 118.3 | 2 |
| Aminobenzothiazole | 136-95-8 | C7H6N2S | 151.0320 | −3.0 | 3.33 | 118.8 | 2 |
| tert-Butylhydroquinone | 1948-33-0 | C10H14O2 | 167.1061 | −3.3 | 5.52 | 126.7 | 3 |
| Pollutants with AA-EQS but Not Detected | <1 | 1–10 | 10–100 | >100 |
|---|---|---|---|---|
| Organotins Chloroalkanes | Cd, Cr, Ni, Pb benzotriazole, Nap | As, Sb, Zn BPA, OP, NP, PBDE *, PFOS, HBCDD, A | Cu DEHP | BaP, Fluo |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gasperi, J.; Le Roux, J.; Deshayes, S.; Ayrault, S.; Bordier, L.; Boudahmane, L.; Budzinski, H.; Caupos, E.; Caubrière, N.; Flanagan, K.; et al. Micropollutants in Urban Runoff from Traffic Areas: Target and Non-Target Screening on Four Contrasted Sites. Water 2022, 14, 394. https://doi.org/10.3390/w14030394
Gasperi J, Le Roux J, Deshayes S, Ayrault S, Bordier L, Boudahmane L, Budzinski H, Caupos E, Caubrière N, Flanagan K, et al. Micropollutants in Urban Runoff from Traffic Areas: Target and Non-Target Screening on Four Contrasted Sites. Water. 2022; 14(3):394. https://doi.org/10.3390/w14030394
Chicago/Turabian StyleGasperi, Johnny, Julien Le Roux, Steven Deshayes, Sophie Ayrault, Louise Bordier, Lila Boudahmane, Hélène Budzinski, Emilie Caupos, Nadège Caubrière, Kelsey Flanagan, and et al. 2022. "Micropollutants in Urban Runoff from Traffic Areas: Target and Non-Target Screening on Four Contrasted Sites" Water 14, no. 3: 394. https://doi.org/10.3390/w14030394
APA StyleGasperi, J., Le Roux, J., Deshayes, S., Ayrault, S., Bordier, L., Boudahmane, L., Budzinski, H., Caupos, E., Caubrière, N., Flanagan, K., Guillon, M., Huynh, N., Labadie, P., Meffray, L., Neveu, P., Partibane, C., Paupardin, J., Saad, M., Varnede, L., & Gromaire, M.-C. (2022). Micropollutants in Urban Runoff from Traffic Areas: Target and Non-Target Screening on Four Contrasted Sites. Water, 14(3), 394. https://doi.org/10.3390/w14030394








