Evaluating Permeable Clay Brick Pavement for Pollutant Removal from Varying Strength Stormwaters in Arid Regions
Abstract
:1. Introduction
2. Materials and Methods
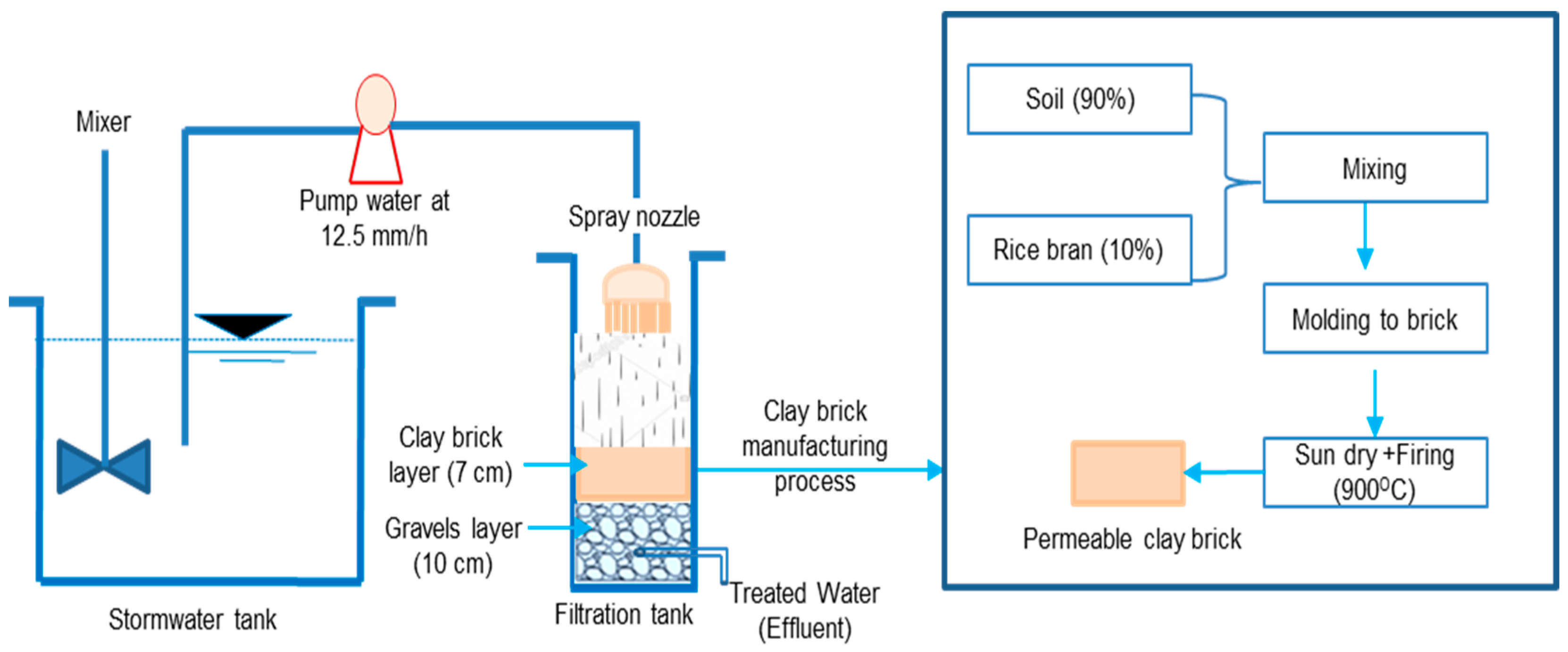
2.1. Permeable Clay Bricks
2.2. Stormwater Sampling
2.3. Experimental Setup
2.4. Runoff Infiltration Experiments
3. Results
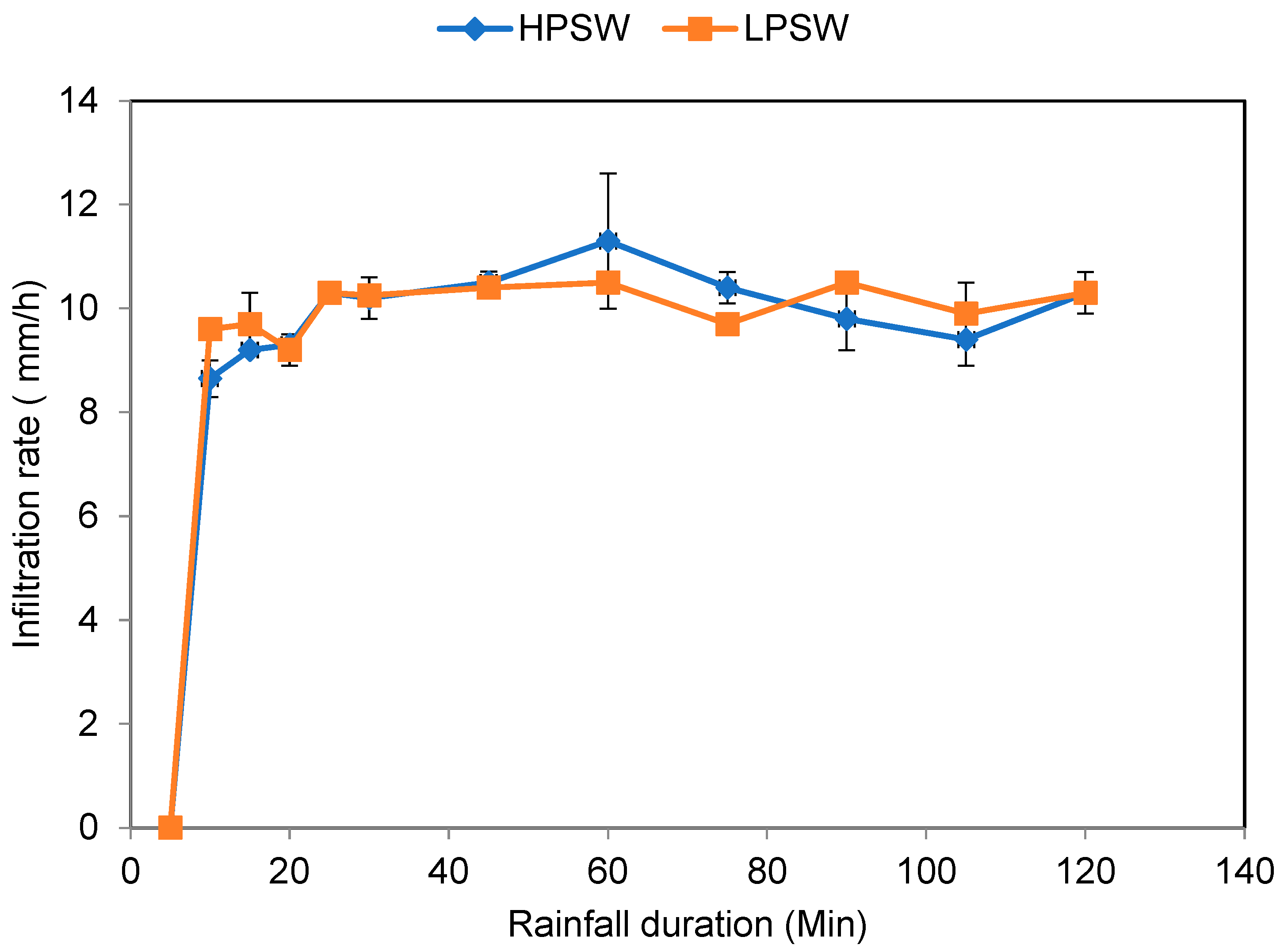
3.1. Infiltration Rate of the Permeable Clay Brick Layer
3.2. Stormwater Quality
3.3. Stormwater Treatment Performance of the Clay Bricks
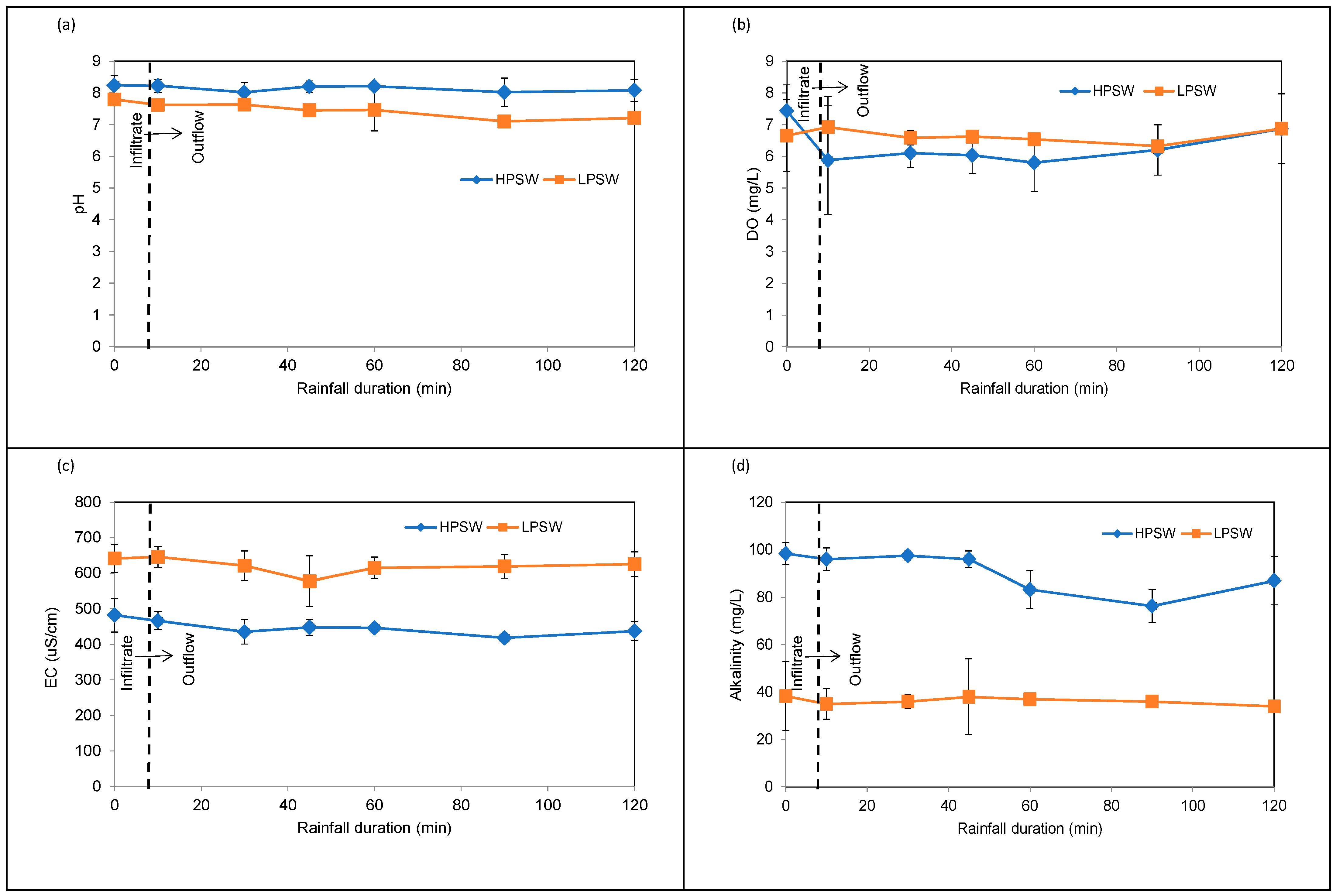
3.3.1. Variation of pH, DO, Alkalinity, and EC
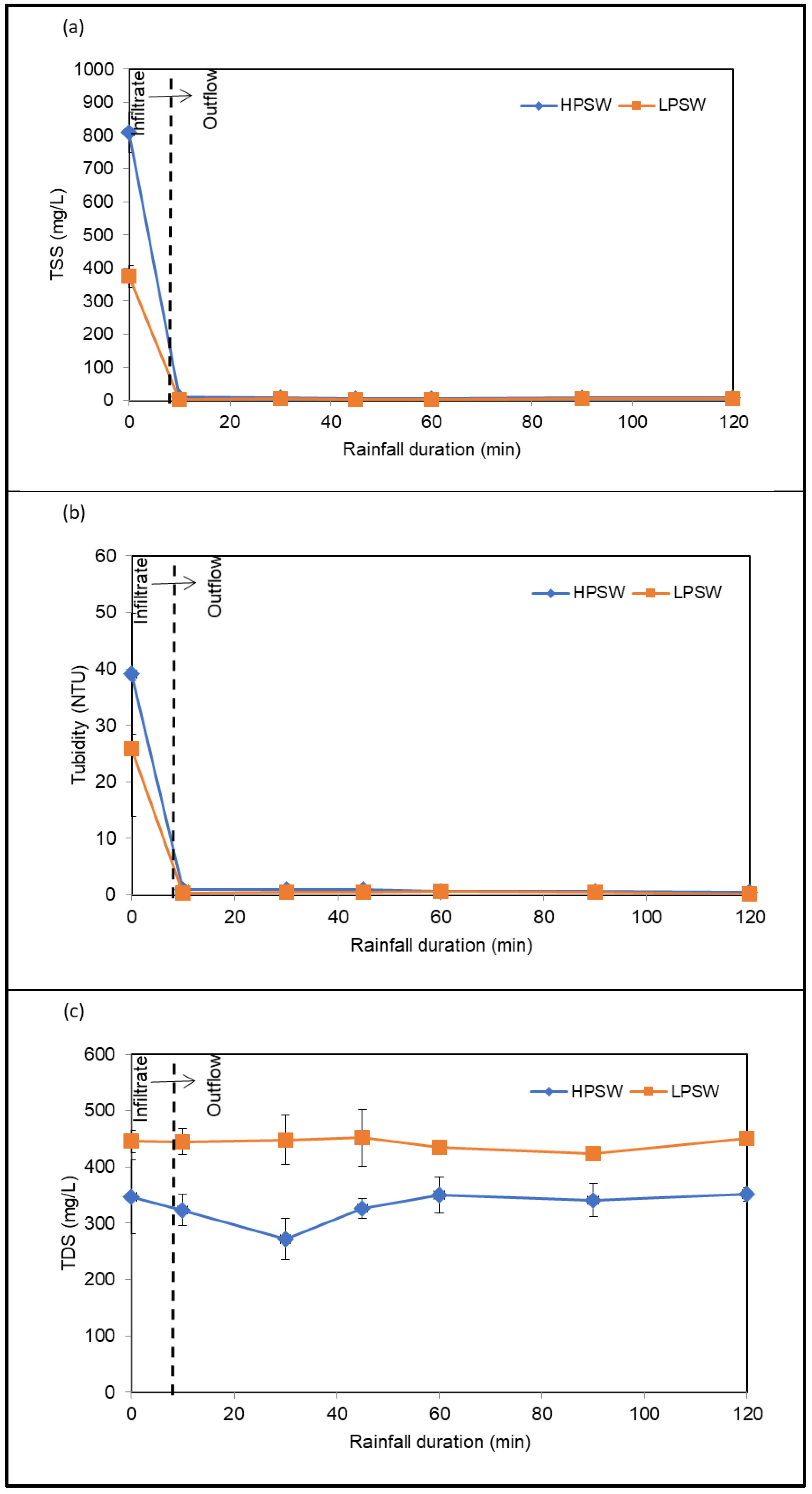
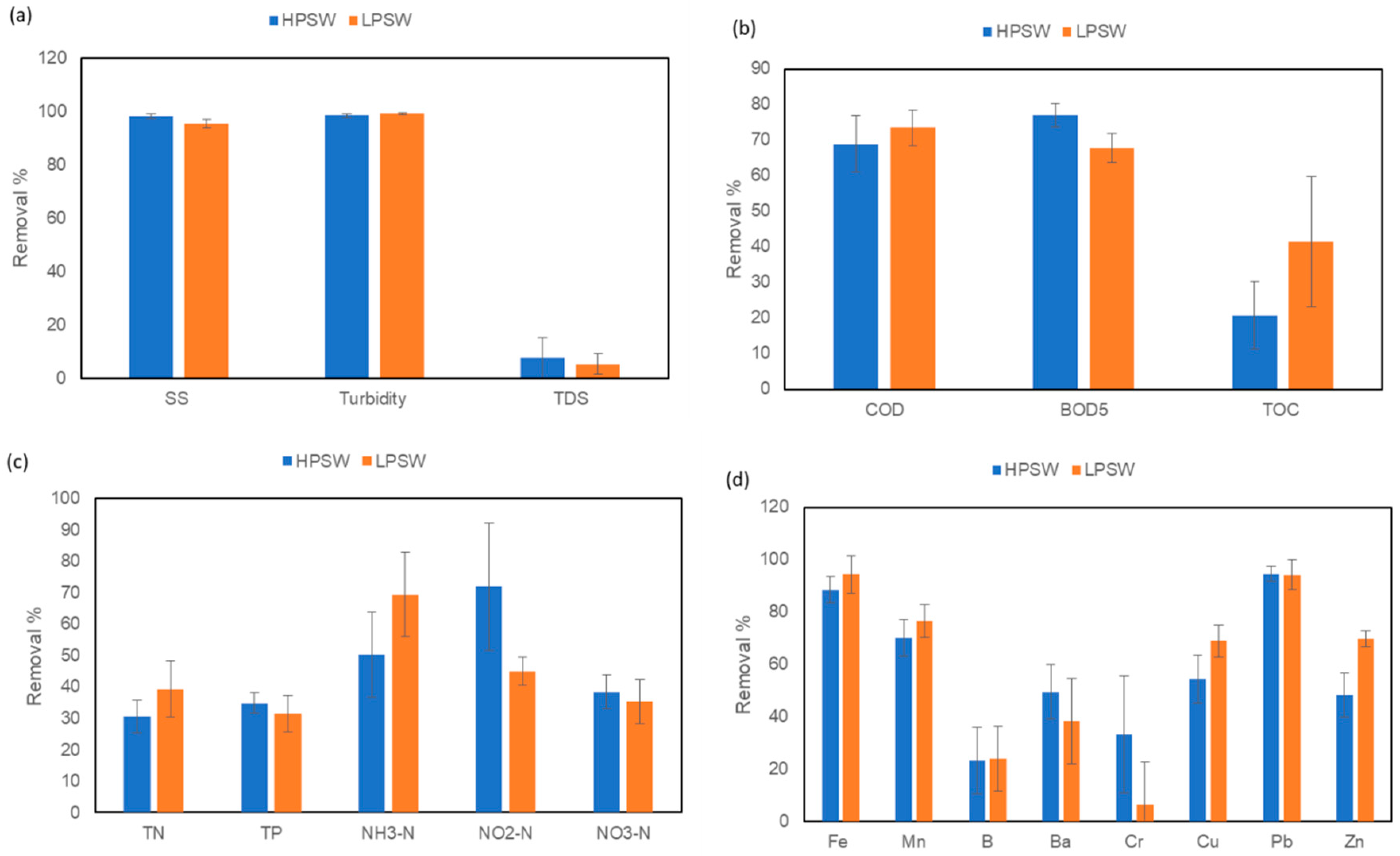
3.3.2. TSS, Turbidity and TDS Removal
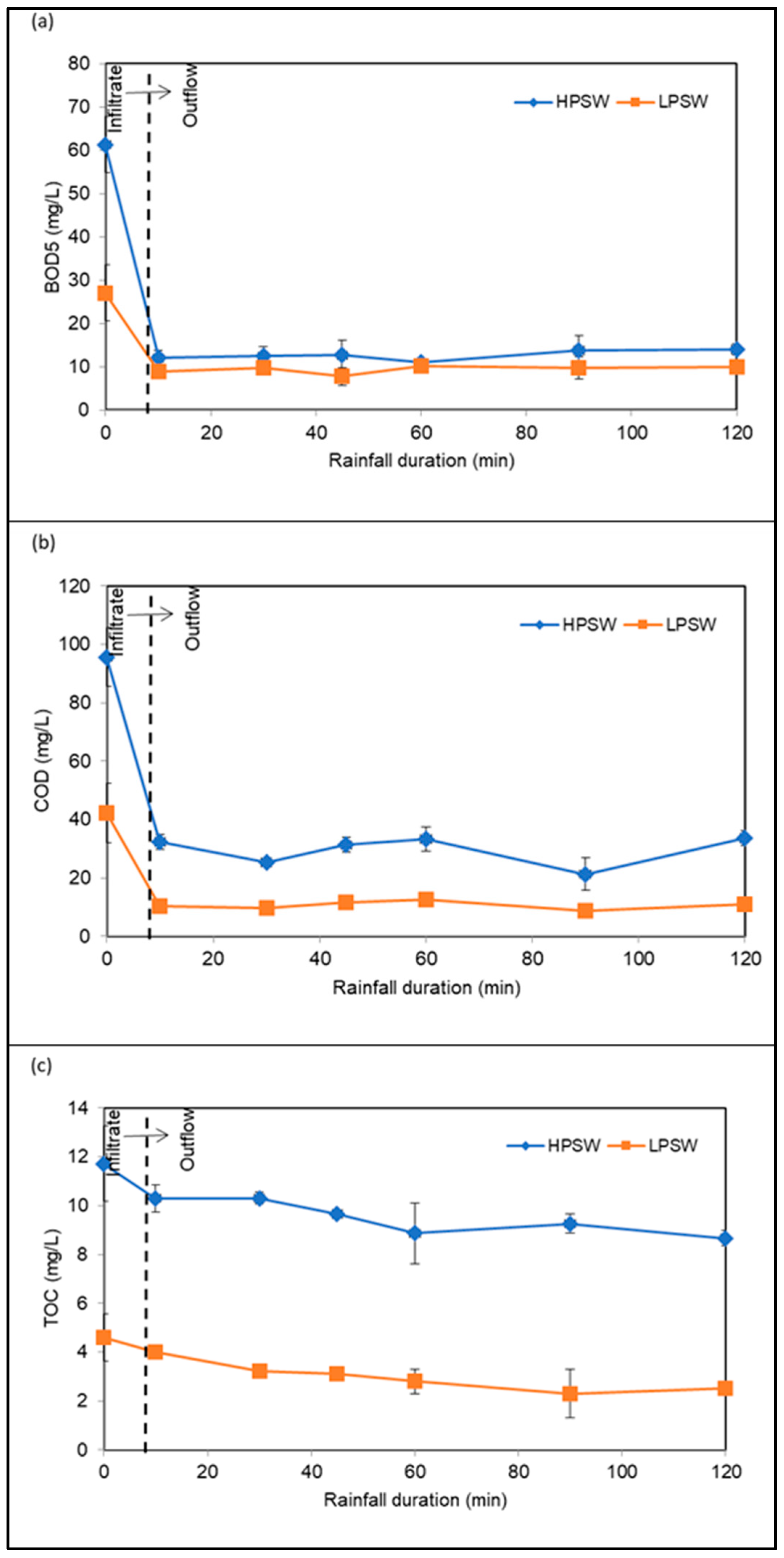
3.3.3. Removal of Organics
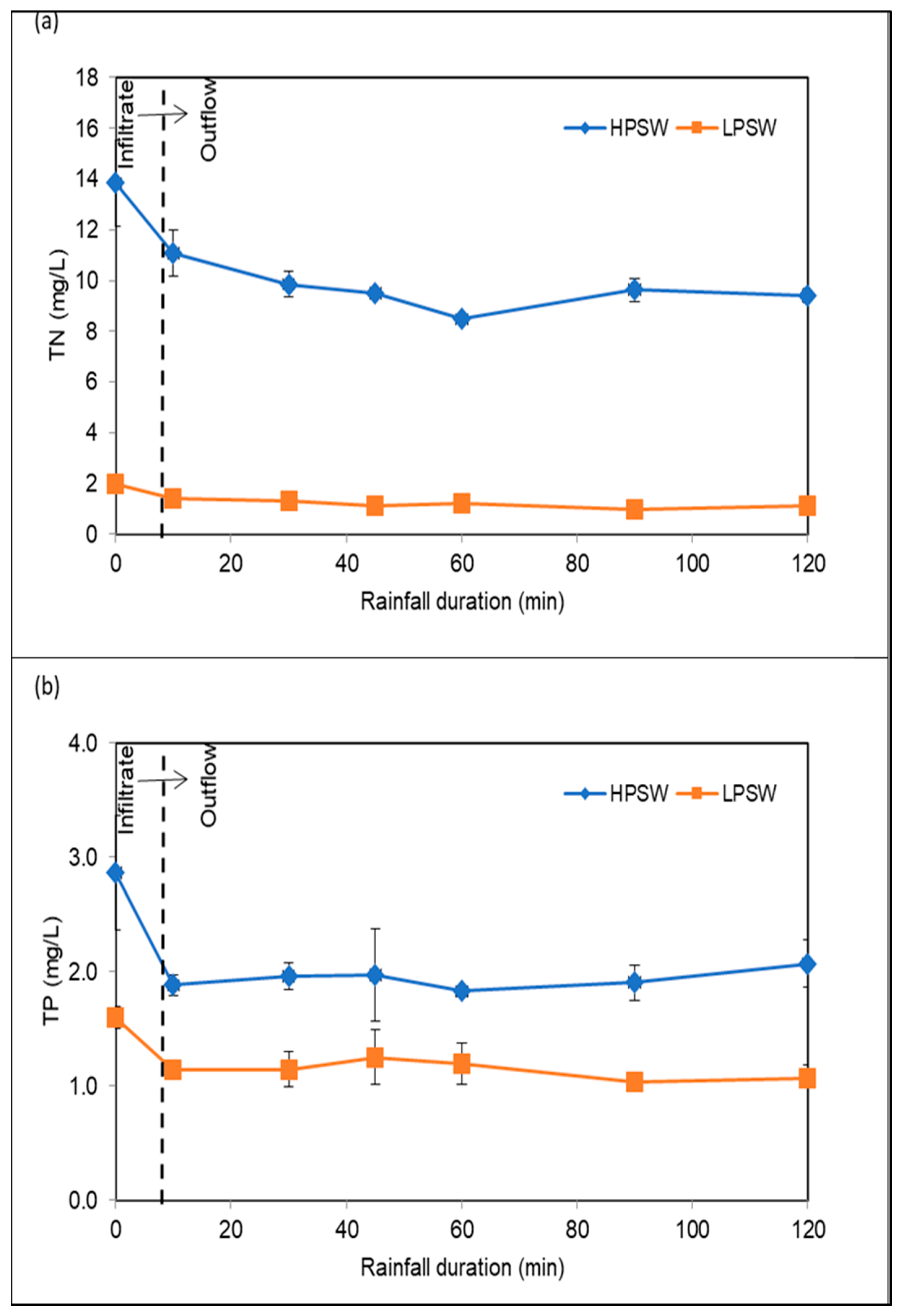
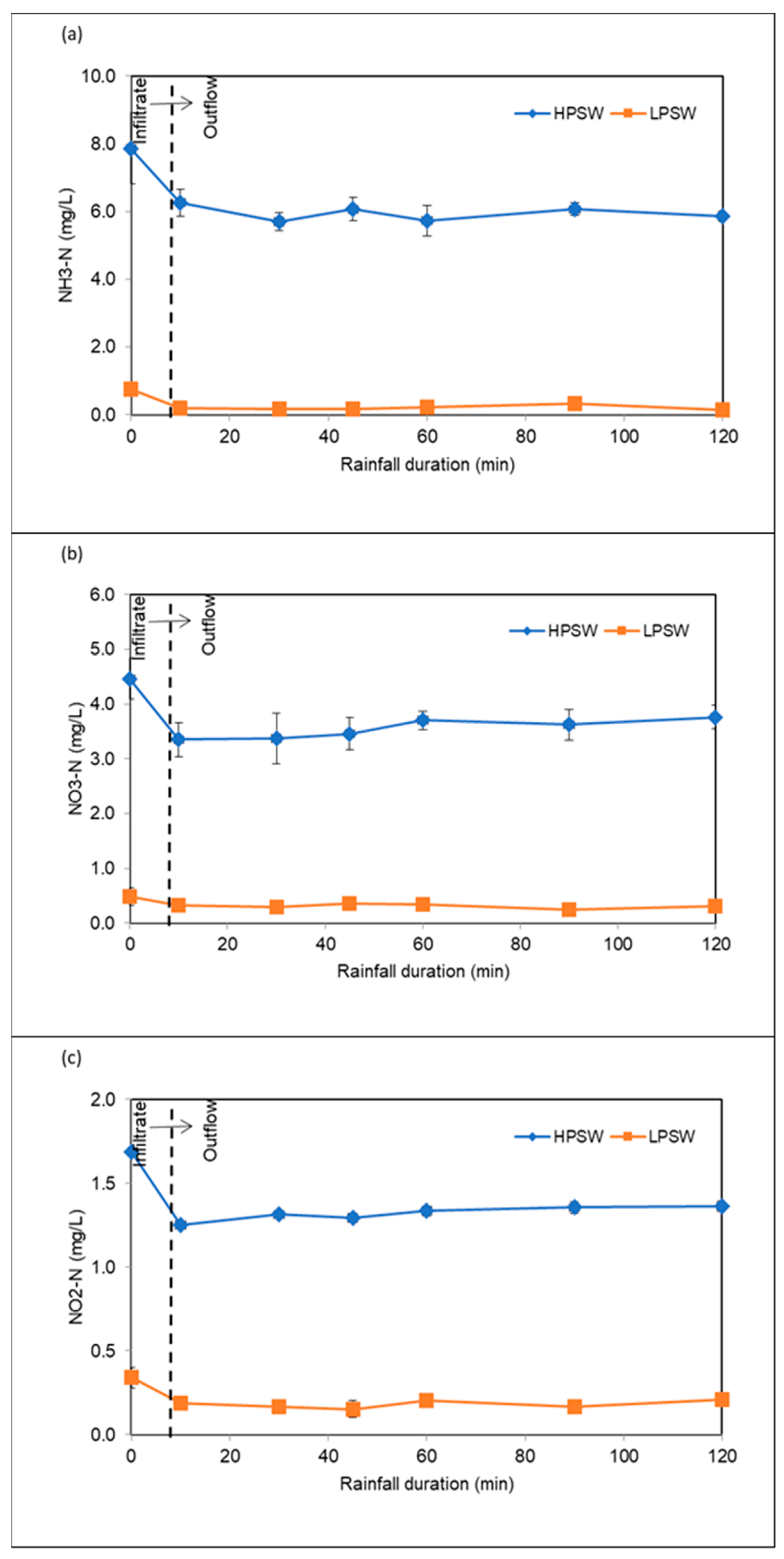
3.3.4. Removal of Nutrients
3.3.5. Removal of Heavy Metals
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Davis, A.P.; Shokouhian, M.; Sharma, H.; Minami, C. Water quality improvement through bioretention media: Nitrogen and phosphorus removal. Water Environ. Res. 2006, 78, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Grebel, J.E.; Mohanty, S.K.; Torkelson, A.A.; Boehm, A.B.; Higgins, C.P.; Maxwell, R.M.; Nelson, K.L.; Sedlak, D.L. Engineered infiltration systems for urban stormwater reclamation. Environ. Eng. Sci. 2013, 30, 437–454. [Google Scholar] [CrossRef]
- LeFevre, G.H.; Paus, K.H.; Natarajan, P.; Gulliver, J.S.; Novak, P.J.; Hozalski, R.M. Review of dissolved pollutants in urban storm water and their removal and fate in bioretention cells. J. Environ. Eng. 2015, 141, 4014050. [Google Scholar] [CrossRef]
- Deblonde, T.; Cossu-Leguille, C.; Hartemann, P. Emerging pollutants in wastewater: A review of the literature. Int. J. Hyg. Environ. Health 2011, 214, 442–448. [Google Scholar] [CrossRef]
- Yu, J.; Yu, H.; Xu, L. Performance evaluation of various stormwater best management practices. Environ. Sci. Pollut. Res. 2013, 20, 6160–6171. [Google Scholar] [CrossRef]
- Reddy, K.R.; Xie, T.; Dastgheibi, S. Mixed-media filter system for removal of multiple contaminants from urban storm water: Large-scale laboratory testing. J. Hazard. Toxic. Radioact. Waste 2014, 18, 1–8. [Google Scholar] [CrossRef]
- Anderson, B.S.; Phillips, B.M.; Voorhees, J.P.; Siegler, K.; Tjeerdema, R. Bioswales reduce contaminants associated with toxicity in urban storm water. Environ. Toxicol. Chem. 2016, 35, 3124–3134. [Google Scholar] [CrossRef] [PubMed]
- LeFevre, G.H.; Hozalski, R.M.; Novak, P.J. The role of biodegradation in limiting the accumulation of petroleum hydrocarbons in raingarden soils. Water Res. 2012, 46, 6753–6762. [Google Scholar] [CrossRef]
- Zhang, K.; Randelovic, A.; Page, D.; McCarthy, D.T.; Deletic, A. The validation of stormwater biofilters for micropollutant removal using in situ challenge tests. Ecol. Eng. 2014, 67, 1–10. [Google Scholar] [CrossRef]
- USEPA, Green Infrastructure in the Semi-Arid West, Low-Impact Development and Green Infrastructure in the Semi-Arid West. Available online: https://www.epa.gov/green-infrastructure/green-infrastructure-semi-arid-west#6 (accessed on 20 November 2020).
- Sambito, M.; Severino, A.; Freni, G.; Neduzha, L. A Systematic Review of the Hydrological, Environmental and Durability Performance of Permeable Pavement Systems. Sustainability 2021, 13, 4509. [Google Scholar] [CrossRef]
- Weiss, P.T.; Kayhanian, M.; Gulliver, J.S.; Khazanovich, L. Permeable pavement in northern North American urban areas: Research review and knowledge gaps. Int. J. Pavement Eng. 2019, 20, 143–162. [Google Scholar] [CrossRef]
- Legret, M.; Colandini, V. Effects of a porous pavement with reservoir structure on runoff water: Water quality and fate of heavy metals. Water Sci. Technol. 1999, 39, 111–117. [Google Scholar] [CrossRef]
- Roseen, R.M.; Ballestero, T.P.; Houle, J.J.; Briggs, J.F.; Houle, K.M. Water quality and hydrologic performance of a porous asphalt pavement as a storm-water treatment strategy in a cold climate. J. Environ. Eng. 2012, 138, 81–89. [Google Scholar] [CrossRef]
- Yuan, X.; Tang, Y.; Li, Y.; Wang, Q.; Zuo, J.; Song, Z. Environmental and economic impacts assessment of concrete pavement brick and permeable brick production process- a case study in China. J. Clean. Prod. 2018, 171, 198–208. [Google Scholar] [CrossRef]
- Xie, N.; Akin, M.; Shi, X. Permeable concrete pavements: A review of environmental benefits and durability. J. Clean. Prod. 2019, 210, 1605–1621. [Google Scholar] [CrossRef]
- Jiang, W.; Sha, A.; Xiao, J.; Li, Y.; Huang, Y. Experimental study on filtration effect and mechanism of pavement runoff in permeable asphalt pavement. Constr. Build. Mater. 2015, 100, 102–110. [Google Scholar] [CrossRef]
- Niu, Z.G.; Lv, Z.W.; Zhang, Y.; Cui, Z.Z. Stormwater infiltration and surface runoff pollution reduction performance of permeable pavement layers. Environ. Sci. Pollut. Res. 2016, 23, 2576–2587. [Google Scholar] [CrossRef]
- Li, H.; Li, Z.; Zhang, X.; Li, Z.; Liu, D.; Li, T.; Zhang, Z. The effect of different surface materials on runoff quality in permeable pavement systems. Environ. Sci. Pollut. Res. 2017, 24, 21103–21110. [Google Scholar] [CrossRef]
- Zhang, K.; Yong, F.; McCarthy, D.T.; Deletic, A. Predicting long term removal of heavy metals from porous pavements for stormwater treatment. Water Res. 2018, 142, 236–245. [Google Scholar] [CrossRef]
- Drake, J.A.P.; Bradford, A.; Marsalek, J. Review of environmental performance of permeable pavement systems: State of the knowledge. Water Qual. Res. J. 2013, 48, 203–222. [Google Scholar] [CrossRef]
- Brown, C.; Chu, A.; Duin, B.; Valeo, C. Characteristics of sediment removal in two types of permeable pavement. Water Qual. Res. J. Can. 2009, 44, 59–70. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Yan, H.; Liao, Z.; Zhang, K.; Schmidt, A.R.; Tao, T. Laboratory analysis on the surface runoff pollution reduction performance of permeable pavements. Sci. Total. Environ. 2019, 691, 1–8. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, Z.; Zhang, X.; Liu, D.; Li, Z.; Li, H. Systematically investigated the influences of permeable pavement materials on the water quality of runoff: Batch and column experiments. Water Air Soil Pollut. 2018, 229, 155. [Google Scholar] [CrossRef]
- Yu, Z.; Gan, H.; Xiao, M.; Huang, B.; Zhu, D.Z.; Zhang, Z.; Wang, H.; Lin, Y.; Hou, Y.; Peng, S.; et al. Performance of permeable pavement systems on stormwater permeability and pollutant removal. Environ. Sci. Pollut. Res. 2021, 28, 28571–28584. [Google Scholar] [CrossRef]
- Schlea, D.; Martin, J.F.; Ward, A.D.; Brown, L.C.; Suter, S.A. Performanceand water table responses of retrofit rain gardens. J. Hydrol. Eng. 2014, 19, 05014002. [Google Scholar] [CrossRef]
- Lin, Z.; Yang, H.; Chen, H.; Ouyang, X.; Li, Z. Comparison of the decontamination performance of three permeable bricks: Adsorption and filtration experiments. Pol. J. Environ. Stud. 2020, 29, 3225–3233. [Google Scholar] [CrossRef]
- Han, S.; Yang, Y.; Liu, S.; Lu, M. Decontamination performance and cleaning characteristics of three common used paved permeable bricks. Environ. Sci. Pollut. Res. 2020, 28, 15114–15122. [Google Scholar] [CrossRef]
- Alharbi, F.; Almoshaogeh, M.; Shafiquzzaman, M.; Haider, H.; Rafiquzzaman, M.; Alragi, A.; ElKholy, S.; Bayoumi, E.A.; EL-Ghoul, Y. Development of Rice Bran Mixed Porous Clay Bricks for Permeable Pavements: A Sustainable LID Technique for Arid Regions. Sustainability 2021, 13, 1443. [Google Scholar] [CrossRef]
- Al-Amri, N.; Subyani, A.M. Generation of Rainfall Intensity Duration Frequency (IDF) Curves for Ungauged Sites in Arid Region. Earth Syst. Environ. 2017, 1, 8. [Google Scholar] [CrossRef]
- Alotaibi, K.; Ghumman, A.R.; Haider, H.; Ghazaw, Y.M.; Shafiquzzaman, M. Future Predictions of Rainfall and Temperature Using GCM and ANN for Arid Regions: A Case Study for the Qassim Region, Saudi Arabia. Water 2018, 10, 1260. [Google Scholar] [CrossRef] [Green Version]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- MWE. Technical Guidelines for the Use of Treated Sanitary Wastewater in Irrigation for Landscaping and Agricultural Irrigation; Ministry of Water and Electricity: Riyadh, Saudi Arabia, 2006.
- World Health Organization (WHO). Guidelines for the Safe Use of Wastewater, Excreta and Greywater, Volume 4: Excreta and Greywater Use in Agriculture; WHO: Geneva, Switzerland, 2006.
- World Health Organization (WHO). Guidelines for Drinking Water Quality, 2nd ed.; World Health Organization: Geneva, Switzerland, 2004.
- Saudi Arabian Standards Organization (SASO). Un-Bottled Drinking Water. SASO 701 and mkg 149 (In Arabic); SASO: Riyadh, Saudi Arabia, 2000.
- Ogunmodede, O.T.; Adebayo, O.L.; Ojo, A.A. Enhancing adsorption capacity of clay and application in dye removal from wastewater. Int. Lett. Chem. Phys. Astron. 2014, 39, 35–51. [Google Scholar] [CrossRef]
- Song, R.; Gong, Y.; Li, J.; Li, X. Progress in controlling urban nonpoint source pollution bypermeable pavement. Environ. Sci. Technol. 2014, 37, 57–63. [Google Scholar]
- Soetardji, J.P.; Claudia, J.C.; Ju, Y.H.; Hriljac, J.A.; Chen, T.Y.; Soetaredjo, F.E.; Ismadji, S. Ammonia removal from water using sodium hydroxide modified zeolite mordenite. RSC Adv. 2014, 5, 83689–83699. [Google Scholar] [CrossRef]
- Uddin, M.K. A review on the adsorption of heavy metals by clay minerals, with special focus on the past decade. Chem. Eng. J. 2017, 308, 438–462. [Google Scholar] [CrossRef]
- Veli, S.; Alyüz, B. Adsorption of copper and zinc from aqueous solutions by using natural clay. J. Hazard. Mater. 2007, 149, 226–233. [Google Scholar] [CrossRef]
- Ahiablame, L.M.; Engel, B.A.; Chaubey, I. Effectiveness of low impact development practices: Literature review and suggestions for future research. Water Air Soil Pollut. 2012, 223, 4253–4273. [Google Scholar] [CrossRef]
- Zarezadeh, V.; Lung, T.; Dorman, T.; Shipley, H.T.; Giacomoni, M. Assessing the performance of sand filter basins in treating urban stormwater runoff. Environ. Monit. Assess. 2018, 190, 697. [Google Scholar] [CrossRef]

| Parameters | Values |
|---|---|
| Compressive strength (MPa) | 18.8 |
| Flexural strength (MPa) | 0.75 |
| Permeability coefficient (mm/s) | 2.38 × 10−3 |
| Porosity (%) | 29.2 |
| Water adsorption (%) | 22.3 |
| Bulk density (kg/m3) | 1.61 |
| Parameter | Method | Manufacturer |
|---|---|---|
| pH, DO, and EC | HACH portable pH, EC, and DO meter | HQ11D53000000, HACH, Loveland, CO, USA |
| Turbidity | Hach 2100Q turbidity meter | 2100Q, HACH, Loveland, CO, USA |
| TDS | Hach HQ411d TDS meter | HACH, Loveland, CO, USA |
| Alkalinity | Titration Method 2320 | - |
| BOD | 5-d BOD Method 5210: 2000 | - |
| TOC | TOC analyzer | Shimadzu, Japan |
| COD | Closed Reflux, Colorimetric Method 5220 D: 2000 | - |
| Total Phosphorous (TP) | 4500-P E. ascorbic acid method | - |
| Total nitrogen (TN) | Kjeldahl method (4500-Norg B: 2000) | - |
| Nitrate (NO3–N) | Nitrate electrode method (4500-NO3 D: 2000) | - |
| Nitrite (NO2–N) | Colorimetric method (4500-NO2 B: 2000) | - |
| Ammonia nitrogen (NH3–N) | Titrimetric method (4500-NH3 C: 2000) | - |
| Fe, Mn, B, Ba, Pb, Zn, Ni, Cu, Cd, Se, Ba and Cr | Inductive coupled Plasma Mass Spectrometer | PerkinElmer, NexION™ 300 ICP-MS |
| Parameter | Unit | HPSW | LPSW | ||
|---|---|---|---|---|---|
| MEAN | SD | MEAN | SD | ||
| pH | - | 8.24 | 0.30 | 7.79 | 0.12 |
| TSS | mg/L | 810.00 | 61.49 | 374.33 | 32.96 |
| Alkalinity | mg/L | 98.33 | 4.73 | 38.33 | 14.57 |
| EC | μS/cm | 482.67 | 47.88 | 641.33 | 39.80 |
| DO | mg/L | 7.43 | 0.81 | 6.65 | 1.14 |
| Turbidity | NTU | 39.10 | 10.69 | 25.93 | 12.10 |
| TDS | mg/L | 347.00 | 65.64 | 446.00 | 19.92 |
| BOD5 | mg/L | 61.33 | 6.43 | 27.08 | 6.51 |
| COD | mg/L | 95.68 | 10.03 | 42.24 | 10.15 |
| TOC | mg/L | 11.72 | 1.54 | 4.59 | 0.97 |
| TN | mg/L | 13.84 | 1.70 | 1.96 | 0.15 |
| NH3–N | mg/L | 7.87 | 1.05 | 0.76 | 0.14 |
| NO2–N | mg/L | 0.07 | 0.02 | 0.26 | 0.06 |
| NO3–N | mg/L | 4.47 | 0.38 | 0.48 | 0.17 |
| TP | mg/L | 2.87 | 0.50 | 1.60 | 0.10 |
| Fe | μg/L | 210.00 | 20.00 | 26.67 | 20.82 |
| Mn | μg/L | 66.67 | 11.55 | 49.67 | 10.50 |
| B | μg/L | 175.73 | 52.10 | 63.50 | 24.20 |
| Ba | μg/L | 46.23 | 9.54 | 26.80 | 6.58 |
| Cd | μg/L | ND | ND | ND | ND |
| Cr | μg/L | 20.99 | 6.37 | 20.02 | 4.89 |
| Cu | μg/L | 98.75 | 4.12 | 84.90 | 8.40 |
| Ni | μg/L | ND | ND | ND | ND |
| Pb | μg/L | 38.51 | 17.15 | 9.65 | 0.57 |
| Se | μg/L | 32.31 | 14.27 | ND | ND |
| Zn | μg/L | 372.30 | 75.32 | 366.83 | 107.92 |
| Parameters | Contaminants Removal by Clay Bricks (This Study) | Contaminants Removal % by Other LID Technologies | National and International Standard for Drinking Water and Wastewater Reuse | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HPSW Effluent | LPSW Effluent | Removal (%) HPSW | Removal (%) LPSW | Bio-Retention Systems [42] | Permeable Pavements [42] | Grass Swales [42] | Sand Filter [43] | KSA Reuse Standard | WHO Reuse Standard | KSA Drinking Water Standard | WHO Drinking Water Standard | |
| pH | 8.0 ± 0.34 | 7.33 ± 0.31 | - | - | - | - | - | - | 6–8.4 | 6.0–9.0 | 6.5–8.5 | 6.5–8 |
| DO (mg/L) | 6.5 ± 0.88 | 6.9 ± 0.52 | - | - | - | - | - | - | - | - | - | - |
| Alkalinity (mg/L) | 87.8 ± 6.5 | 33.7 ± 6.5 | - | - | - | - | - | - | - | - | - | - |
| EC (us/cm) | 441 ± 31 | 585 ± 44 | 8.6 | 8.7 | - | - | - | −0.92 | - | - | - | - |
| Turbidity (NTU) | 0.62 ± 0.28 | 0.24 ± 0.12 | 98.4 | 99 | 5 | - | <5 | - | ||||
| TSS (mg/L) | 8.2 ± 1.6 | 6.1 ± 2.2 | 98.9 | 98.3 | 47–99 | 58–94 | 46–97 | 32–93.5 | ||||
| TDS (mg/L) | 320 ± 28 | 422 ± 18 | 7.7 | 5.2 | - | - | - | - | 2500 | - | 700 | - |
| BOD5 (mg/L) | 14.1 ± 2.1 | 8.7 ± 2.7 | 77 | 67.8 | - | - | - | - | 10 | 10 | - | |
| COD (mg/L) | 29.6 ± 8.3 | 11.1 ± 2.3 | 68.9 | 73.5 | - | - | - | - | 50 | - | - | - |
| TOC (mg/L) | 9.3 ± 1.2 | 2.6 ± 0.9 | 20.8 | 41.5 | - | - | 23–64 | - | - | - | - | - |
| TN (mg/L) | 9.6 ± 0.79 | 1.19 ± 0.19 | 30.5 | 39.1 | 32–99 | >75 | 14–61 | −107.6 | - | - | - | - |
| NH3–N (mg/L) | 6.1 ± 0.4 | 0.24 ± 0.11 | 22.7 | 69.2 | 5 | - | - | - | ||||
| NO2–N (mg/L) | 0.02 ± 0.02 | 0.14 ± 0.01 | 71.8 | 44.8 | - | - | - | - | - | - | - | - |
| NO3–N (mg/L) | 3.9 ± 0.11 | 0.31 ± 0.04 | 13.2 | 35.2 | 1–83 | - | - | - | 10 | - | - | - |
| TP (mg/L) | 1.87 ± 0.1 | 1.10 ± 0.1 | 34.7 | 31.3 | - | - | - | - | - | - | - | - |
| Fe (µg/L) | 24 ± 11 | 1.5 ± 2.5 | 88.5 | 94.3 | 5000 | 5000 | 300 | 300 | ||||
| Mn (µg/L) | 20 ± 5 | 11 ± 4 | 70.2 | 76.5 | 200 | 200 | 100 | 100 | ||||
| B (µg/L) | 135 ± 24 | 48.1± 8.5 | 23.2 | 24.1 | - | - | - | - | - | - | - | 24,000 |
| Ba (µg/L) | 23.3 ± 5.3 | 16.5 ± 4.7 | 49.5 | 38.3 | - | - | - | - | - | - | - | 1300 |
| Cr(µg/L) | 14.0 ± 5.1 | 111 ± 12.1 | 33.2 | 6.7 | - | - | - | - | 100 | 100 | 50 | 50 |
| Cu (µg/L) | 44.9 ± 34 | 26.3 ± 5.6 | 54.5 | 68.9 | 43–97 | 20–99 | 14–67 | −83 | 300 | 200 | 1000 | 2000 |
| Zn(µg/L) | 191.8 ± 9.9 | 26.3 ± 5.7 | 48.7 | 69.7 | 62–97 | 73–99 | 47–81 | 0–93 | 4000 | 2000 | 5000 | 3000 |
| Pb (µg/L) | 2.1 ± 1.2 | 0.55 ± 0.61 | 94.5 | 94.2 | 31–98 | 75–99 | 18–94 | −122 | 100 | 5000 | 50 | 10 |
| Se (µg/L) | 1.97 ± 0.77 | ND | 93.9 | - | - | - | - | - | 20 | 20 | 10 | 40 |
| Cd (µg/L) | ND | ND | - | - | - | - | 12–98 | - | 10 | 100 | 5 | 3 |
| Ni (µg/L) | ND | ND | - | - | - | - | - | - | 200 | 200 | - | 70 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shafiquzzaman, M.; Alqarawi, S.M.A.; Haider, H.; Rafiquzzaman, M.; Almoshaogeh, M.; Alharbi, F.; El-Ghoul, Y. Evaluating Permeable Clay Brick Pavement for Pollutant Removal from Varying Strength Stormwaters in Arid Regions. Water 2022, 14, 491. https://doi.org/10.3390/w14030491
Shafiquzzaman M, Alqarawi SMA, Haider H, Rafiquzzaman M, Almoshaogeh M, Alharbi F, El-Ghoul Y. Evaluating Permeable Clay Brick Pavement for Pollutant Removal from Varying Strength Stormwaters in Arid Regions. Water. 2022; 14(3):491. https://doi.org/10.3390/w14030491
Chicago/Turabian StyleShafiquzzaman, Md., Saad Mohammed A. Alqarawi, Husnain Haider, Md. Rafiquzzaman, Meshal Almoshaogeh, Fawaz Alharbi, and Yassine El-Ghoul. 2022. "Evaluating Permeable Clay Brick Pavement for Pollutant Removal from Varying Strength Stormwaters in Arid Regions" Water 14, no. 3: 491. https://doi.org/10.3390/w14030491
APA StyleShafiquzzaman, M., Alqarawi, S. M. A., Haider, H., Rafiquzzaman, M., Almoshaogeh, M., Alharbi, F., & El-Ghoul, Y. (2022). Evaluating Permeable Clay Brick Pavement for Pollutant Removal from Varying Strength Stormwaters in Arid Regions. Water, 14(3), 491. https://doi.org/10.3390/w14030491









