Method Development for Low-Concentration PAHs Analysis in Seawater to Evaluate the Impact of Ship Scrubber Washwater Effluents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Sample Collection
2.3. Sample Pretreatment and Analysis
2.4. Analytical Characteristics
3. Results and Discussion
3.1. PAHs Concentrations in the Surface Seawater
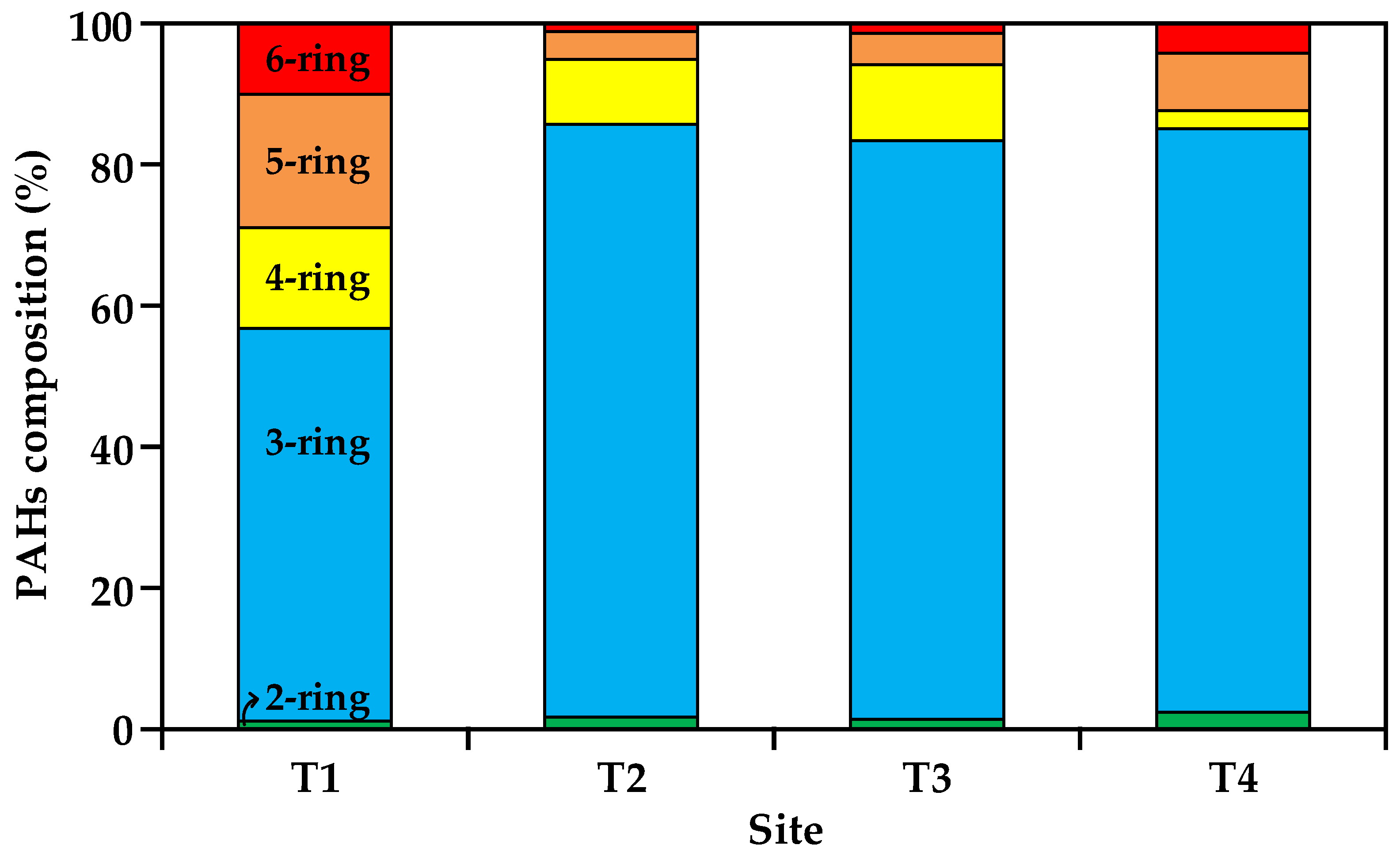
3.2. Composition Patterns of PAHs
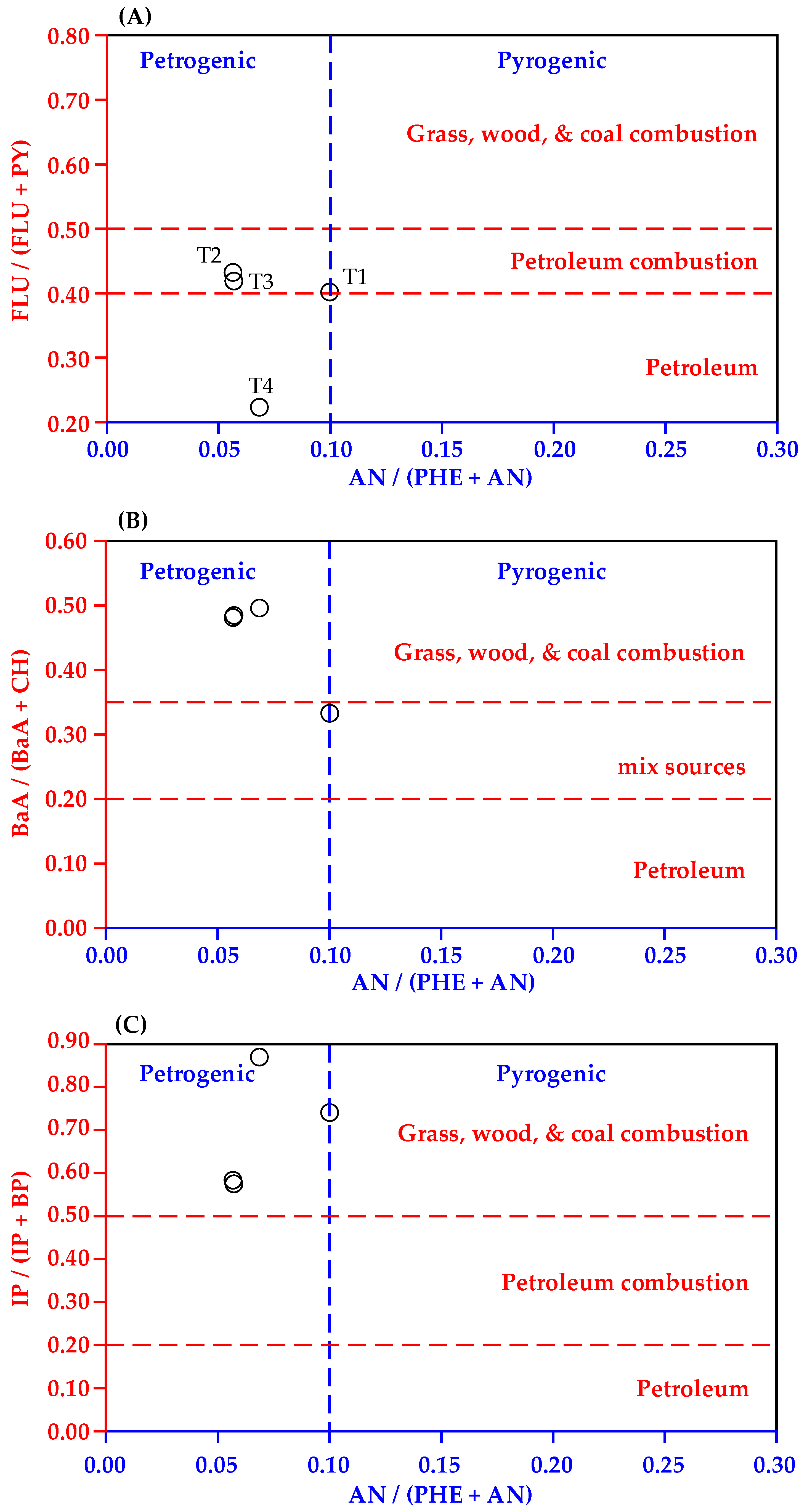
3.3. Identification of PAHs Sources
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Honda, M.; Suzuki, N. Toxicities of Polycyclic Aromatic Hydrocarbons for Aquatic Animals. Int. J. Environ. Res. Public Health 2020, 17, 1363. [Google Scholar] [CrossRef] [Green Version]
- Sakellari, A.; Karavoltsos, S.; Moutafis, I.; Koukoulakis, K.; Dassenakis, M.; Bakeas, E. Occurrence and distribution of polycyclic aromatic hydrocarbons in the marine surface microlayer of an industrialized coastal area in the eastern Mediterranean. Water 2021, 13, 3174. [Google Scholar] [CrossRef]
- Szatyłowicz, E.; Skoczko, I. Evaluation of the PAH content in soot from solid fuels combustion in low power boilers. Energies 2019, 12, 4254. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.F.; Ju, Y.R.; Su, Y.C.; Lim, Y.C.; Kao, C.M.; Dong, C.D.; Chen, C.W. Distribution, sources, and behavior of PAHs in estuarine water systems exemplified by Salt River, Taiwan. Mar. Pollut. Bull. 2020, 154, 111029. [Google Scholar] [CrossRef]
- Szatyłowicz, E.; Walendziuk, W. Analysis of polycyclic aromatic hydrocarbon content in ash from solid fuel combustion in low-power boilers. Energies 2021, 14, 6801. [Google Scholar] [CrossRef]
- Olayinka, O.O.; Adewusi, A.A.; Olujimi, O.O.; Aladesida, A.A. Polycyclic aromatic hydrocarbons in sediment and health risk of fish, crab and shrimp around Atlas Cove, Nigeria. J. Health Pollut. 2019, 9, 191204. [Google Scholar] [CrossRef]
- Pouch, A.; Zaborska, A.; Mazurkiewicz, M.; Winogradow, A.; Pazdro, K. PCBs, HCB and PAHs in the seawater of Arctic fjords—Distribution, sources and risk assessment. Mar. Pollut. Bull. 2021, 164, 111980. [Google Scholar] [CrossRef]
- Zaborska, A.; Carroll, C.; Pazdro, K.; Pempkowiak, J. Spatio-temporal patterns of PAHs, PCBs and HCB in sediments of the western Barents Sea. Oceanologia 2011, 53, 1005–1026. [Google Scholar] [CrossRef] [Green Version]
- Berrojalbiz, N.; Lacorte, S.; Calbet, A.; Saiz, E.; Barata, C.; Dachs, J. Accumulation and cycling of polycyclic aromatic hydrocarbons in zooplankton. Environ. Sci. Technol. 2009, 43, 2295–2301. [Google Scholar] [CrossRef]
- Verbruggen, E.M.J. Environmental Risk Limits for Polycyclic Aromatic Hydrocarbons (PAHs). For Direct Aquatic, Benthic, and Terrestrial Toxicity; RIVM Report 607711007/2012; National Institute for Public Health and the Environment: Bilthoven, The Netherlands, 2012.
- IEA. Oil 2019 by the International Energy Agency; IEA: Paris, France, 2019. [Google Scholar]
- Endres, S.; Maes, F.; Hopkins, F.; Houghton, K.; Mårtensson, E.M.; Oeffner, J.; Quack, B.; Singh, P.; Turner, D. A new perspective at the ship-air-sea-interface: The environmental impacts of exhaust gas scrubber discharge. Front. Mar. Sci. 2018, 5, 139. [Google Scholar] [CrossRef] [Green Version]
- Comer, B.; Georgeff, E.; Osipova, L. Air emissions and water pollution discharges from ships with scrubbers. In International Council on Clean Transportation (ICCT), ICCT Consulting Report; ICCT: Washington, DC, USA, 2020. [Google Scholar]
- Thor, P.; Granberg, M.E.; Winnes, H.; Magnusson, K. Severe toxic effects on pelagic copepods from maritime exhaust gas scrubber effluents. Environ. Sci. Technol. 2021, 55, 5826–5835. [Google Scholar] [CrossRef]
- Lunde Hermansson, A.; Hassellöv, I.; Moldanová, J.; Ytreberg, E. Comparing emissions of polyaromatic hydrocarbons and metals from marine fuels and scrubbers. Transp. Res. Transp. Environ. 2021, 97, 102912. [Google Scholar] [CrossRef]
- Ren, H.F.; Kawagoe, T.; Jia, H.J.; Endo, H.; Kitazawa, A.; Goto, S.; Hayashi, T. Continuous surface seawater surveillance on poly aromatic hydrocarbons (PAHs) and mutagenicity of East and South China Seas. Estuar. Coast. Shelf Sci. 2010, 86, 395–400. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Z.F.; Zhang, X.; Yang, P.F.; Li, Y.F.; Cai, M.; Kallenborn, R. Dissolved polycyclic aromatic hydrocarbons from the Northwestern Pacific to the Southern Ocean: Surface seawater distribution, source apportionment, and air-seawater exchange. Water Res. 2021, 207, 117780. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Wu, X.; Simonich, S.L.M.; Kang, H.; Xie, Z. Volatilization of polycyclic aromatic hydrocarbons (PAHs) over the North Pacific and adjacent Arctic Ocean: The impact of offshore oil drilling. Environ. Pollut. 2021, 268, 115963. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.D.; Tsai, C.L.; Hsu, Y.C.; Chen, Y.W.; Weng, Y.M.; Chang, M.B. PCDD/Fs and dl-PCBs concentrations in water samples of Taiwan. Chemosphere 2017, 173, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Linders, J.; Adams, E.; Behrends, B.; Dock, A.; Hanayama, S.; Luit, R.; Rouleau, C.; Tronczynski, J. Exhaust Gas Cleaning Systems—A Roadmap to Risk Assessment. Report of the GESAMP Task Team on Exhaust Gas Cleaning Systems (PPR 7/INF.23); International Maritime Organization: London, UK, 2019. Available online: http://docs.imo.org/ (accessed on 8 December 2021).
- Kalf, D.F.; Crommentuijn, T.; van de Plassche, E.J. Environmental quality objectives for 10 polycyclic aromatic hydrocarbons (PAHs). Ecotoxicol. Environ. Saf. 1997, 36, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Liu, J.; Luan, Y.; Li, Y.; Ma, M.; Xu, J.; Han, S. Distribution and ecosystem risk assessment of polycyclic aromatic hydrocarbons in the Luan River, China. Ecotoxicology 2010, 19, 827–837. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.G.; Duan, M.S.; Guo, J.Q.; Liu, M.Y.; Qi, A.X.; Lin, Y.; Liang, J.H. PAHs in the Northern South China Sea: Horizontal transport and downward export on the continental shelf. Mar. Chem. 2018, 202, 121–129. [Google Scholar] [CrossRef]
- Huang, Y.J.; Lin, B.S.; Lee, C.L.; Brimblecombe, P. Enrichment behavior of contemporary PAHs and legacy PCBs at the sea-surface microlayer in harbor water. Chemosphere 2020, 245, 125647. [Google Scholar] [CrossRef] [PubMed]
- Teuchies, J.; Cox, T.J.S.; Van Itterbeeck, K.; Meysman, F.J.R.; Blust, R. The impact of scrubber discharge on the water quality in estuaries and ports. Environ. Sci. Eur. 2020, 32, 103. [Google Scholar] [CrossRef]
- Yunker, M.B.; Backus, S.M.; Graf Pannatier, E.; Jeffries, D.S.; Macdonald, R.W. Sources and significance of alkane and PAH hydrocarbons in Canadian arctic rivers. Estuar. Coast. Shelf Sci. 2002, 55, 1–31. [Google Scholar] [CrossRef]
- Hayakawa, K.; Makino, F.; Yasuma, M.; Yoshida, S.; Chondo, Y.; Toriba, A.; Kameda, T.; Tang, N.; Kunugi, M.; Nakase, H.; et al. Polycyclic aromatic hydrocarbons in surface water of the southeastern Japan Sea. Chem. Pharm. Bull. 2016, 64, 625–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adhikari, P.L.; Maiti, K.; Overton, E.B. Vertical fluxes of polycyclic aromatic hydrocarbons in the northern Gulf of Mexico. Mar. Chem. 2015, 168, 60–68. [Google Scholar] [CrossRef]
- Tang, G.W.; Liu, M.Y.; Zhou, Q.; He, H.X.; Chen, K.; Zhang, H.B.; Hu, J.H.; Huang, Q.H.; Luo, Y.M.; Ke, H.W.; et al. Microplastics and polycyclic aromatic hydrocarbons (PAHs) in Xiamen coastal areas: Implications for anthropogenic impacts. Sci. Total Environ. 2018, 634, 811–820. [Google Scholar] [CrossRef]
- Ya, M.; Wu, Y.; Li, Y.; Wang, X. Transport of terrigenous polycyclic aromatic hydrocarbons affected by the coastal upwelling in the northwestern coast of South China Sea. Environ. Pollut. 2017, 229, 60–68. [Google Scholar] [CrossRef]
- Ya, M.; Xu, L.; Wu, Y.; Li, Y.; Zhao, S.; Wang, X. Fossil fuel-derived polycyclic aromatic hydrocarbons in the Taiwan Strait, China, and fluxes across the air-water interface. Environ. Sci. Technol. 2018, 52, 7307–7316. [Google Scholar] [CrossRef]
- Witt, G. Occurrence and transport of polycyclic aromatic hydrocarbons in the water bodies of the Baltic Sea. Mar. Chem. 2002, 79, 49–66. [Google Scholar] [CrossRef]
- Yuan, K.; Wang, X.W.; Lin, L.; Zou, S.C.; Li, Y.; Yang, Q.S.; Luan, T.G. Characterizing the parent and alkyl polycyclic aromatic hydrocarbons in the Pearl River Estuary, Daya Bay and northern South China Sea: Influence of riverine input. Environ. Pollut. 2015, 199, 66–72. [Google Scholar] [CrossRef]
- Gao, Y.; Zheng, H.; Xia, Y.; Chen, M.; Meng, X.Z.; Cai, M. Spatial distributions and seasonal changes of current-use pesticides from the North Pacific to the Arctic Oceans. J. Geophys. Res. Atmos. 2019, 124, 9716–9729. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.; Lin, T.; Li, Z.; Jiang, Y.; Li, Y.; Yao, X.; Gao, H.; Guo, Z. Air-sea exchange and gas-particle partitioning of polycyclic aromatic hydrocarbons over the northwestern Pacific Ocean: Role of East Asian continental outflow. Environ. Pollut. 2017, 230, 444–452. [Google Scholar] [CrossRef]
- Wang, Q.; Peng, F.; Chen, Y.; Jin, L.; Lin, J.; Zhao, X.; Yin, J.; Li, J.Y. Heavy metals and PAHs in an open fishing area of the East China Sea: Multimedia distribution, source diagnosis, and dietary risk assessment. Environ. Sci. Pollut. Res. Int. 2019, 26, 21140–21150. [Google Scholar] [CrossRef]
- Cheng, J.O.; Ko, F.C.; Lee, C.L.; Fang, M.D. Air-water exchange fluxes of polycyclic aromatic hydrocarbons in the tropical coast, Taiwan. Chemosphere 2013, 90, 2614–2622. [Google Scholar] [CrossRef]
- Cao, S.K.; Na, G.S.; Li, R.J.; Ge, L.K.; Gao, H.; Jin, S.C.; Hou, C.; Gao, Y.Z.; Zhang, Z.F. Fate and deposition of polycyclic aromatic hydrocarbons in the Bransfield Strait, Antarctica. Mar. Pollut. Bull. 2018, 137, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, J.; Silva, T.; Filho, J.G.M.; Flores, D. Polycyclic aromatic hydrocarbons (PAHs) in burning and non-burning coal waste piles. J. Hazard. Mater. 2012, 199–200, 105–110. [Google Scholar] [CrossRef] [PubMed]
- González-Pérez, D.M.; Garralón, G.; Plaza, F.; Pérez, J.I.; Moreno, B.; Gómez, M.A. Removal of low concentrations of phenanthrene, fluoranthene and pyrene from urban wastewater by membrane bioreactors technology. J. Environ. Sci. Health Part A Toxic Hazard. Subst. Environ. Eng. 2012, 47, 2190–2197. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Falcon, M.S.; Soto-Gonzalez, B.; Simal-Gandara, J. Evolution of the concentrations of polycyclic aromatic hydrocarbons in burnt woodland soils. Environ. Sci. Technol. 2006, 40, 759–763. [Google Scholar] [CrossRef]
- Chen, C.W.; Chen, C.F.; Dong, C.D.; Tu, Y.T. Composition and source apportionment of PAHs in sediments at river mouths and channel in Kaohsiung Harbor, Taiwan. J. Environ. Monit. 2012, 14, 105–115. [Google Scholar] [CrossRef]
- Budzinski, H.; Jones, I.; Bellocq, J.; Pierard, C.; Garrigues, P. Evaluation of sediment contamination by polycyclic aromatic hydrocarbons in the Gironde Estuary. Mar. Chem. 1997, 58, 85–97. [Google Scholar] [CrossRef]



| Instrument Parameter | Condition Setting |
|---|---|
| Autosampler | Agilent 7693A |
| Injection volume | 1 µL |
| Gas chromatography (GC) | Agilent 7890B |
| Carrier gas | Helium (≥99.999%) |
| Injection mode | Splitless |
| Column | HP-5MS (30 m, 0.25 mm, 0.25 μm) |
| Column flow rate | 1 mL/min |
| Temperature of inlet | 300 °C |
| Oven temperature program | 40 °C (hold for 1 min), 120 °C (35 °C/min), 160 °C (10 °C/min), 300 °C (5 °C/min, hold for 10 min) |
| Mass selective detector (MSD) | Agilent 5977A |
| Temperature of ion source | 230 °C |
| Temperature of quadrupole | 150 °C |
| Temperature of transfer line | 280 °C |
| Electronic energy | 70 eV |
| Ionization mode | Electron ionization (EI) |
| Type of mass scan | Selective ion monitoring (SIM) (see Table 2) |
| Compounds | Ring | Retention Time (min) | Quantitative Ion (m/z) | Time Window (min) | Selected Ions (m/z) |
|---|---|---|---|---|---|
| Naphthalene-ds (IS1) | 2 | 5.653 | 136 | 4.00–8.00 | 127, 128, 129, 136, 172 |
| Naphthalene (NA) | 2 | 5.676 | 128 | ||
| 2-Fluorobiphenyl (SS1) | 2 | 7.372 | 172 | ||
| Acenaphthylene (ACY) | 3 | 8.363 | 152 | 8.00–11.00 | 151, 152, 153, 154, 164, 165, 166, 167 |
| Acenaphthene-d10 (IS2) | 3 | 8.713 | 164 | ||
| Acenaphthene (ACE) | 3 | 8.782 | 154 | ||
| Fluorene (FL) | 3 | 10.077 | 166 | ||
| Phenanthrene-d10 (IS3) | 3 | 12.986 | 188 | 11.00–19.00 | 101, 176, 178, 179, 188, 200, 202, 203 |
| Phenanthrene (PHE) | 3 | 13.062 | 178 | ||
| Anthracene (AN) | 3 | 13.221 | 178 | ||
| Fluoranthene (FLU) | 4 | 17.702 | 202 | ||
| Pyrene (PY) | 4 | 18.582 | 202 | ||
| 4-Terphenyl-d14 (SS2) | 4 | 19.733 | 244 | 19.00–27.00 | 226, 228, 229, 240, 244 |
| Benzo[a]anthracene (BaA) | 4 | 23.978 | 228 | ||
| Chrysene-d12 (IS4) | 4 | 24.043 | 240 | ||
| Chrysene (CH) | 4 | 24.155 | 228 | ||
| Benzo[b]fluoranthene (BbF) | 5 | 28.612 | 252 | 27.00–45.29 | 125, 138, 139, 252, 253, 264, 276, 277 |
| Benzo[k]fluoranthene (BkF) | 5 | 28.723 | 252 | ||
| Benzo[a]pyrene (BaP) | 5 | 29.833 | 252 | ||
| Perylene-d12 (IS5) | 5 | 30.072 | 264 | ||
| Indeno[1,2,3-c,d]pyrene (IP) | 6 | 33.919 | 276 | ||
| Dibenz[a,h]anthracene (DBA) | 5 | 34.094 | 278 | ||
| Benzo[g,h,i]perylene (BP) | 6 | 34.719 | 276 |
| PAHs a | Response Factor (RF) (n = 5) | Check Standard (n = 3) R b (%) | Repeat Analysis (n = 5) RPD b (%) | Duplicate Sample (n = 3) RPD b (%) | Detection Limits (ng/L) | Field Blank (n = 3) (ng/L) | Equipment Blank (n = 5) (ng/L) | |
|---|---|---|---|---|---|---|---|---|
| Average ± SD a | CV b (%) | |||||||
| NA | 1.01 ± 0.16 | 15.7 | 94 ± 4.5 | 5.3 ± 5.1 | 15.5 ± 3.0 | 0.001 | 0.001 ± 0.001 | 0.001 ± 0.000 |
| ACY | 0.75 ± 0.08 | 10.2 | 101 ± 2.9 | 3.7 ± 3.2 | 17.0 ± 5.2 | 0.001 | 0.001 ± 0.001 | 0.001 ± 0.000 |
| ACE | 0.46 ± 0.05 | 12.0 | 106 ± 1.3 | 7.2 ± 5.1 | 15.8 ± 4.2 | 0.006 | 0.003 ± 0.001 | 0.004 ± 0.002 |
| FL | 0.58 ± 0.07 | 11.5 | 100 ± 7.9 | 4.1 ± 8.4 | 11.4 ± 3.2 | 0.002 | 0.003 ± 0.001 | 0.002 ± 0.001 |
| PHE | 0.74 ± 0.08 | 11.0 | 110 ± 3.3 | 2.8 ± 5.8 | 13.5 ± 3.4 | 0.004 | 0.002 ± 0.001 | 0.003 ± 0.001 |
| AN | 0.87 ± 0.12 | 13.3 | 106 ± 4.1 | 4.1 ± 4.2 | 7.5 ± 3.8 | 0.009 | 0.002 ± 0.002 | 0.002 ± 0.003 |
| FLU | 0.97 ± 0.14 | 14.4 | 101 ± 12.0 | 5.4 ± 8.2 | 12.7 ± 5.4 | 0.007 | 0.002 ± 0.002 | 0.003 ± 0.002 |
| PY | 1.01 ± 0.17 | 16.3 | 107 ± 10.0 | 6.1 ± 7.1 | 14.1 ± 10.6 | 0.012 | 0.002 ± 0.001 | 0.005 ± 0.004 |
| BaA | 0.69 ± 0.06 | 8.5 | 108 ± 5.6 | 9.8 ± 5.0 | 9.8 ± 4.5 | 0.013 | 0.004 ± 0.002 | 0.007 ± 0.004 |
| CH | 0.90 ± 0.08 | 9.0 | 102 ± 8.3 | 2.1 ± 7.5 | 14.5 ± 4.6 | 0.013 | 0.002 ± 0.001 | 0.009 ± 0.004 |
| BbF | 0.69 ± 0.07 | 9.9 | 110 ± 5.8 | 6.2 ± 9.0 | 12.5 ± 4.4 | 0.011 | 0.003 ± 0.001 | 0.013 ± 0.004 |
| BkF | 1.01 ± 0.10 | 8.9 | 106 ± 5.8 | 4.4 ± 6.0 | 12.9 ± 3.7 | 0.011 | 0.002 ± 0.001 | 0.004 ± 0.004 |
| BaP | 0.77 ± 0.03 | 4.4 | 117 ± 6.1 | 2.8 ± 7.7 | 16.4 ± 7.8 | 0.015 | 0.004 ± 0.002 | 0.008 ± 0.005 |
| IP | 0.77 ± 0.08 | 10.7 | 103 ± 1.4 | 3.2 ± 3.4 | 14.8 ± 6.7 | 0.011 | 0.004 ± 0.001 | 0.010 ± 0.004 |
| DBA | 0.83 ± 0.12 | 13.9 | 102 ± 0.8 | 6.1 ± 6.1 | 15.8 ± 5.9 | 0.014 | 0.004 ± 0.002 | 0.008 ± 0.005 |
| BP | 0.95 ± 0.11 | 11.6 | 109 ± 5.3 | 7.5 ± 4.6 | 14.5 ± 5.0 | 0.009 | 0.002 ± 0.001 | 0.004 ± 0.003 |
| SS1 | 1.22 ± 0.08 | 1.6 | 103 ± 5.4 | 6.1 ± 3.1 | 8.3 ± 0.5 | - | - | - |
| SS2 | 1.40 ± 0.06 | 9.2 | 103 ± 2.4 | 3.7 ± 6.5 | 9.1 ± 2.5 | - | - | - |
| Item | MDL a | MPCs b | T1 | T2 | T3 | T4 |
|---|---|---|---|---|---|---|
| Water depth (m) | – | – | 683 | 626 | 852 | 587 |
| Water temp (°C) | – | – | 30.4 | 30.5 | 30.6 | 30.5 |
| Salinity (psu) | – | – | 34.0 | 34.0 | 34.0 | 33.9 |
| PAHs (ng/L) | ||||||
| Naphthalene (NA) | 0.001 | 2000 | 0.030 | 0.037 | 0.043 | 0.092 |
| Acenaphthylene (ACY) | 0.001 | 130 | 0.012 | 0.013 | 0.017 | 0.022 |
| Acenaphthene (ACE) | 0.006 | 380 | 0.026 | 0.016 | 0.019 | 0.406 |
| Fluorene (FL) | 0.002 | 300 | 0.137 | 0.179 | 0.235 | 0.552 |
| Phenanthrene (PHE) | 0.004 | 1100 | 1.215 | 1.624 | 2.208 | 2.169 |
| Anthracene (AN) | 0.009 | 100 | 0.135 | 0.098 | 0.134 | 0.160 |
| Fluoranthene (FLU) | 0.007 | 120 | 0.087 | 0.078 | 0.120 | 0.014 |
| Pyrene (PY) | 0.012 | 23 | 0.130 | 0.102 | 0.167 | 0.048 |
| Benzo[a]anthracene (BaA) | 0.013 | 1.2 | 0.058 | 0.016 | 0.028 | 0.021 |
| Chrysene (CH) | 0.013 | 7 | 0.114 | 0.017 | 0.029 | 0.021 |
| Benzo[b]fluoranthene (BbF) | 0.011 | 17 | 0.094 | 0.018 | 0.033 | 0.221 |
| Benzo[k]fluoranthene (BkF) | 0.011 | 17 | 0.078 | 0.012 | 0.014 | 0.033 |
| Benzo[a]pyrene (BaP) | 0.015 | 10 | 0.094 | 0.020 | 0.029 | 0.025 |
| Indeno[1,2,3-c,d]pyrene (IP) | 0.011 | 0.27 | 0.202 | 0.016 | 0.026 | 0.148 |
| Dibenz[a,h]anthracene (DBA) | 0.014 | 0.14 | 0.253 | 0.040 | 0.065 | 0.046 |
| Benzo[g,h,i]perylene (BP) | 0.009 | 0.82 | 0.075 | 0.012 | 0.020 | 0.025 |
| Total PAHs (sum of 16 PAHs) | – | – | 2.740 | 2.297 | 3.187 | 4.001 |
| Locations | No. of PAHs | Sample Volume (L) | Total PAHs (ng/L) | Reference |
|---|---|---|---|---|
| Pacific Ocean | 21 | 200 | 4.0 ± 1.4 | [17] |
| Southern Ocean | 21 | 200 | 2.3 ± 0.52 | |
| Indian Ocean | 21 | 200 | 2.7 ± 1.0 | |
| East Asia | 15 | 45–181 | 3.0–3.7 (3.3) | [18] |
| North Pacific | 15 | 120–166 | 1.0–1.6 (1.3) | |
| Arctic Ocean | 15 | 49–142 | 0.82–2.1 (1.4) | |
| Northwestern Pacific Ocean | 15 | 60–100 | 5.5–15.6 (9.4) | [35] |
| East China Sea | 16 | 3 | 380–4000 | [36] |
| Northern South China Sea | 15 | 4 | 0.98–13 (4.7) | [23] |
| South eastern Japan Sea | 13 | 3 | 3.7–10 (6.0 ± 1.8) | [27] |
| East and South China Seas | 15 | 765–1080 | 30.4–120.3 (66 ± 32) | [16] |
| Taiwan Strait | 15 | 8 | 53–79 (62 ± 8.3) | [31] |
| Gulf of Mexico | 43 | 20 | 24.2–58 (35.5 ± 9.0) | [28] |
| Xiamen coast, China | 13 | 4 | 18–250 (62) | [29] |
| Hainan Island coast, China | 15 | 12–18 | 28–207 (104 ± 38) | [30] |
| Kaohsiung Harbor and adjacent areas, Taiwan | 15 | 1 | 1.25–9.39 (3.85 ± 2.43) | [24] |
| Kenting coast, Taiwan | 22 | 40 | 0.9–8.0 (2.17 ± 1.19) | [37] |
| Offshore southern Taiwan | 16 | 100 | 2.30–4.00 (3.06 ± 0.73) | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-F.; Lim, Y.C.; Ju, Y.-R.; Albarico, F.P.J.B.; Cheng, J.-W.; Chen, C.-W.; Dong, C.-D. Method Development for Low-Concentration PAHs Analysis in Seawater to Evaluate the Impact of Ship Scrubber Washwater Effluents. Water 2022, 14, 287. https://doi.org/10.3390/w14030287
Chen C-F, Lim YC, Ju Y-R, Albarico FPJB, Cheng J-W, Chen C-W, Dong C-D. Method Development for Low-Concentration PAHs Analysis in Seawater to Evaluate the Impact of Ship Scrubber Washwater Effluents. Water. 2022; 14(3):287. https://doi.org/10.3390/w14030287
Chicago/Turabian StyleChen, Chih-Feng, Yee Cheng Lim, Yun-Ru Ju, Frank Paolo Jay B. Albarico, Jia-Wei Cheng, Chiu-Wen Chen, and Cheng-Di Dong. 2022. "Method Development for Low-Concentration PAHs Analysis in Seawater to Evaluate the Impact of Ship Scrubber Washwater Effluents" Water 14, no. 3: 287. https://doi.org/10.3390/w14030287
APA StyleChen, C.-F., Lim, Y. C., Ju, Y.-R., Albarico, F. P. J. B., Cheng, J.-W., Chen, C.-W., & Dong, C.-D. (2022). Method Development for Low-Concentration PAHs Analysis in Seawater to Evaluate the Impact of Ship Scrubber Washwater Effluents. Water, 14(3), 287. https://doi.org/10.3390/w14030287







