Butyltin Contamination in Fishing Port Sediments after the Ban of Tributyltin Antifouling Paint: A Case of Qianzhen Fishing Port in Taiwan
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Sediment Sampling
2.2. Sediment Sample Analysis
2.3. Data Analysis
3. Results and Discussion
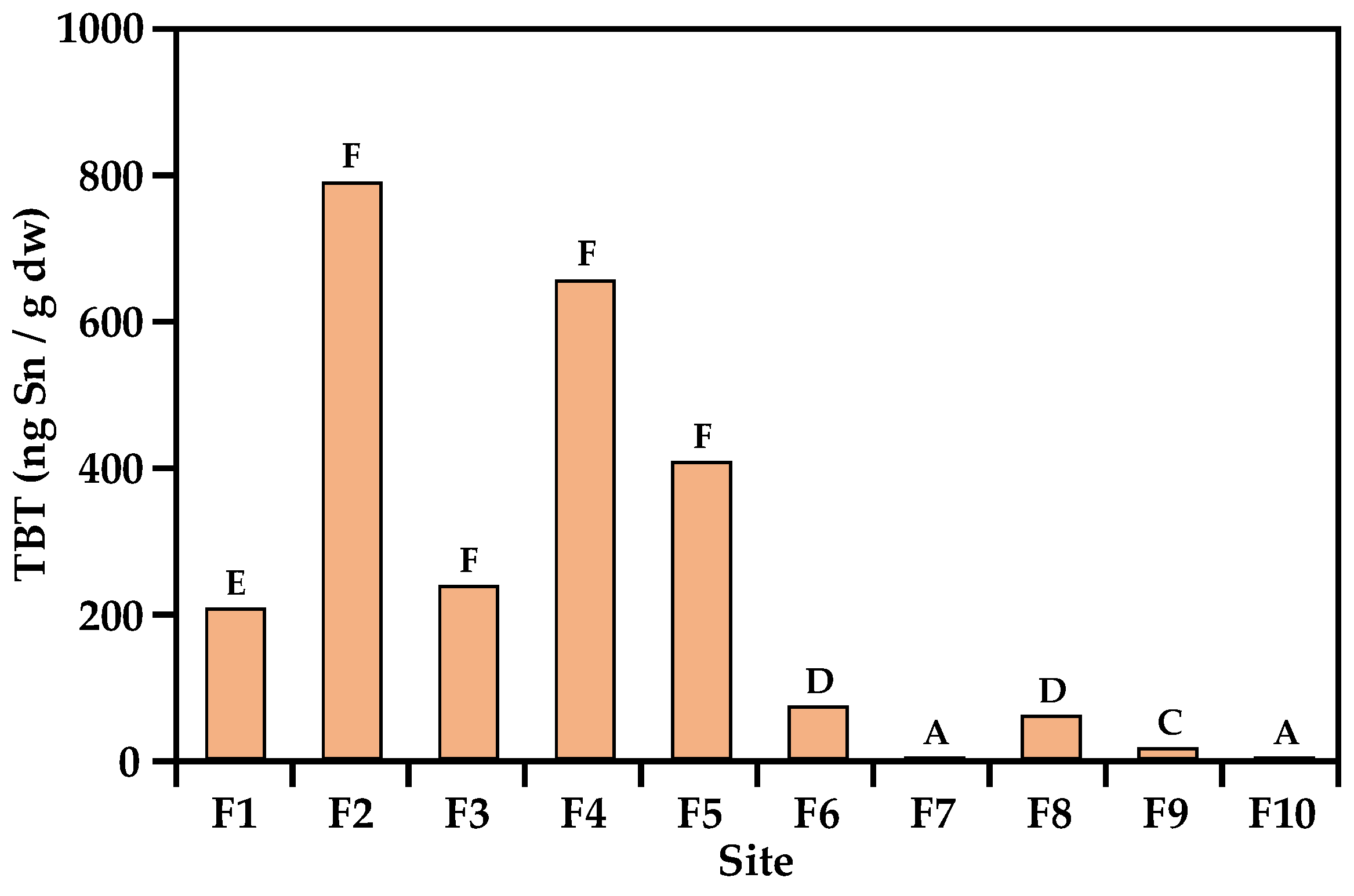
3.1. Concentrations of Butyltins in Surface Sediments
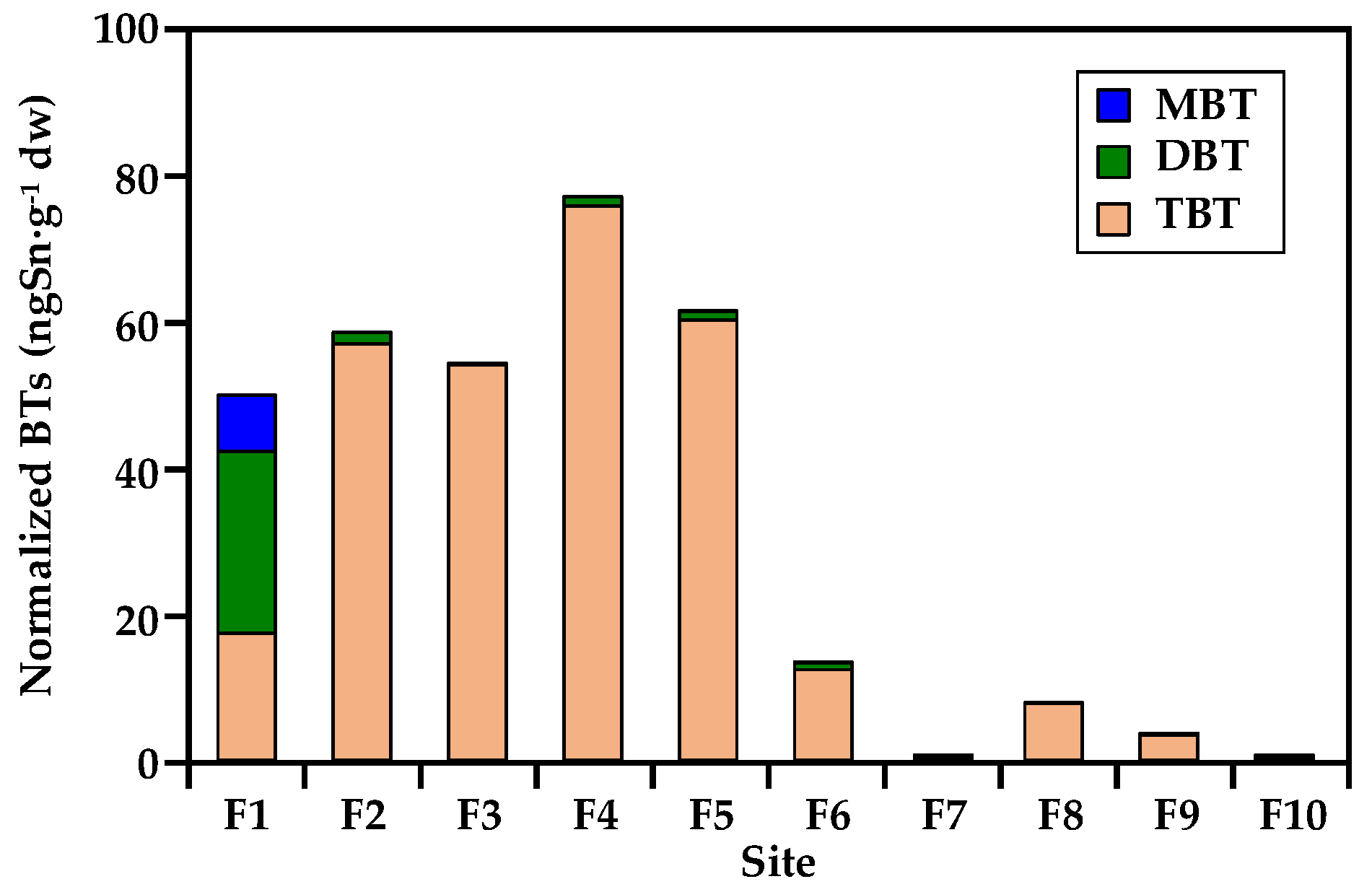
3.2. Spatial Distribution and Composition of Butyltins
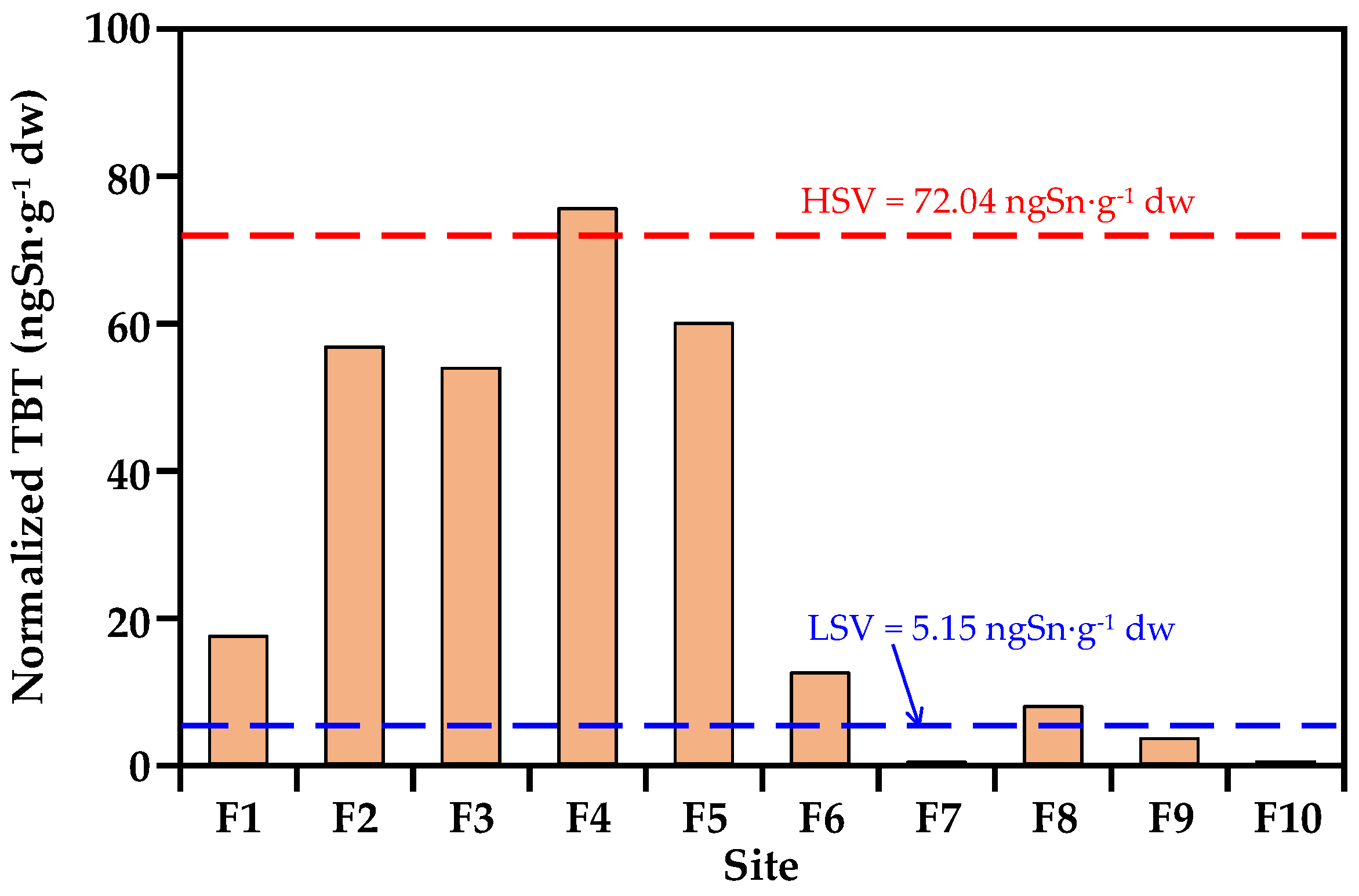
3.3. Evaluation of the Contamination Levels and Ecological Toxicity of TBT
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hoch, M. Organotin compounds in environmentean overview. Appl. Geochem. 2001, 16, 719–743. [Google Scholar] [CrossRef]
- Omae, I. Review organotin antifouling paint and their alternatives. Appl. Organomet. Chem. 2003, 17, 81–105. [Google Scholar] [CrossRef]
- Berg, M.; Arnold, C.G.; Müller, S.R.; Mühlemann, J.; Schwarzenbach, R.P. Sorption and desorption behavior of organotin compounds in sediment-pore water systems. Environ. Sci. Technol. 2001, 35, 3151–3157. [Google Scholar] [CrossRef] [PubMed]
- Dowson, P.H.; Bubb, J.M.; Williams, T.P.; Lester, J.N. Degradation of tributyltin in freshwater and estuarine marina sediments. Water Sci. Technol. 1993, 28, 133–137. [Google Scholar] [CrossRef]
- Dowson, P.H.; Bubb, J.M.; Lester, J.N. Persistence and degradation pathways of tributyltin in freshwater and estuarine sediments. Estuar. Coast. Shelf Sci. 1996, 42, 551–562. [Google Scholar] [CrossRef]
- Rodríguez-Grimon, R.; Campos, N.H.; Castro, Í.B. Imposex incidence in gastropod species from Santa Marta coastal zone, Colombian Caribbean Sea. Bull. Environ. Contam. Toxicol. 2020, 105, 728–735. [Google Scholar] [CrossRef]
- Uc-Peraza, R.G.; Delgado-Blas, V.H.; Rendón-von Osten, J.; Castro, Í.B.; Proietti, M.C.; Fillmann, G. Mexican paradise under threat: The impact of antifouling biocides along the Yucatán Peninsula. J. Hazard. Mater. 2022, 427, 128162. [Google Scholar] [CrossRef]
- Titley-O’Neal, C.P.; Munkittrick, K.R.; Macdonald, B.A. The effects of organotin on female gastropods. J. Environ. Monit. 2011, 13, 2360–2388. [Google Scholar] [CrossRef]
- Tanabe, S.; Prudente, M.S.; Kan-Atireklap, S.; Subramanian, A. Mussel watch: Marine pollution monitoring of butyltins and organochlorines in coastal waters of Thailand, Philippines and India. Ocean. Coast. Manag. 2000, 43, 819–839. [Google Scholar] [CrossRef]
- Zhou, Q.F.; Li, Z.Y.; Jiang, G.B.; Yang, R.Q. Preliminary investigation of a sensitive biomarker of organotin pollution in Chinese coastal aquatic environment and marine organisms. Environ. Pollut. 2003, 125, 301–304. [Google Scholar] [CrossRef]
- Meador, J.P. Organotins in aquatic biota: Occurrence in tissue and toxicological significance. In Environmental Contaminants in Biota: Interpreting Tissue Concentrations; Beyer, W.N., Meador, J.P., Eds.; Taylor and Francis: Boca Raton, FL, USA, 2011. [Google Scholar]
- IMO (International Maritime Organization). International Convention on the Control of Harmful Anti-Fouling Systems on Ships. 2001. Available online: https://www.imo.org/en/About/Conventions/Pages/International-Convention-on-the-Control-of-Harmful-Anti-fouling-Systems-on-Ships-(AFS).aspx (accessed on 5 February 2022).
- Morton, B. Recovery from imposex by a population of the dogwhelk, Nucella lapillus (Gastropoda: Caenogastropoda), on the southeastern coast of England since May 2004: A 52-month study. Mar. Pollut. Bull. 2009, 58, 1530–1538. [Google Scholar] [CrossRef] [PubMed]
- Castro, I.B.; Arroyo, M.; Costa, P.; Fillmann, G. Butyltin compounds and imposex levels in Ecuador. Arch. Environ. Contam. Toxicol. 2012, 62, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Castro, I.B.; Iannacone, J.; Santos, S.; Fillmann, G. TBT is still a matter of concern in Peru. Chemosphere 2018, 205, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.R.L.; Ross, P.M. Recovery of the New Zealand muricid dogwhelk Haustrum scobina from TBT-induced imposex. Mar. Pollut. Bull. 2018, 126, 396–401. [Google Scholar] [CrossRef]
- Ruiz, J.M.; Carro, B.; Albaina, N.; Barreiro, R.; Rial, D.; Bellas, J. Extended imposex monitoring in N Atlantic Spain confirms punctual attainment of European environmental objectives for TBT. Mar. Pollut. Bull. 2018, 126, 462–466. [Google Scholar] [CrossRef]
- Wang, X.; Kong, L.; Cheng, J.; Zhao, D.; Chen, H.; Sun, R.; Yang, W.; Han, J. Distribution of butyltins at dredged material dumping sites around the coast of China and the potential ecological risk. Mar. Pollut. Bull. 2019, 138, 491–500. [Google Scholar] [CrossRef]
- El Ayari, T.; El Menif, N.T. First record of imposex in euthria cornea (Neogastropoda: Buccinidae) collected from the complex lagoon-channel of Bizerta. Thalassas 2019, 35, 87–93. [Google Scholar] [CrossRef]
- Rossato, M.; Costa, M.B.; de Castro, Í.B.; Pinho, G.L.L. Size, season and origin of gastropods matter in imposex assessments. Ecotoxicol. Environ. Saf. 2018, 159, 324–331. [Google Scholar] [CrossRef]
- FACOA (Fisheries Agency, Council of Agriculture). Taiwan Fisheries Yearbook. 2020. Available online: https://www.fa.gov.tw/cht/PublicationsFishYear/content.aspx?id=35&chk=6c7f7242-297c-47d1-8bb1-51e7c8a5961c (accessed on 5 February 2022).
- KCMB (Marine Bureau Kaohsiung City Government). Fishing Ports under the Jurisdiction of Kaohsiung City. 2022. Available online: https://kcmb.kcg.gov.tw/cp.aspx?n=39CE512C6CBF067A (accessed on 5 February 2022).
- Chen, C.F.; Lim, Y.C.; Ju, Y.R.; Albarico, F.P.J.B.; Cheng, J.W.; Chen, C.W.; Dong, C.D. Method development for low-concentration PAHs analysis in seawater to evaluate the impact of ship scrubber washwater effluents. Water 2022, 14, 287. [Google Scholar] [CrossRef]
- TCSB (Toxic and Chemical Substances Bureau). The Handbook of Investigation Achievements on the Toxic Substances Distribution in Taiwan; Version 2009. 9/2009; Environmental Protection Administration: Taipei, Taiwan, 2009.
- Dowson, P.H.; Bubb, J.M.; Lester, J.N. Temporal distribution of organotins in the aquatic environment: Five years after the 1987 UK retail began on TBT-base antifouling paints. Mar. Pollut. Bull. 1993, 26, 487–494. [Google Scholar] [CrossRef]
- Chen, C.W.; Chen, C.F.; Dong, C.D.; Tu, Y.T. Composition and source apportionment of PAHs in sediments at river mouths and channel in Kaohsiung Harbor, Taiwan. J. Environ. Monit. 2012, 14, 105–115. [Google Scholar] [CrossRef]
- Dong, C.D.; Chen, C.F.; Chen, C.W. Composition and source of butyltins in sediments of Kaohsiung Harbor, Taiwan. Estuar. Coast. Shelf. Sci. 2015, 156, 134–143. [Google Scholar] [CrossRef]
- Michaud, M.H.; Pelletier, E. Sources and fate of butyltins in the St. Lawrence Estuary ecosystem. Chemosphere 2006, 64, 1074–1082. [Google Scholar] [CrossRef] [PubMed]
- US EPA (US Environmental Protection Agency). Recommendation for Screening Values for Tributyltin in Sediments at Superfund Sites in Puget Sound; DCN 4000-09-013-ADDS, 13-01-AADK, and 33-01-AAAV; Roy F. Weston, Inc.: Seattle, WA, USA, 1996; Prepared for US Environmental Protection Agency Superfund Program. Region X.
- OSPAR (OsIo and Paris Commission). Co-ordinated Environmental Monitoring Programme (CEMP) Assessment Report: 2008/2009. In Assessment of Trends and Concentrations of Selected Hazardous Substances in Sediments and Biota; OSPAR Publication: London, UK, 2009. [Google Scholar]
- Chen, C.F.; Chen, C.W.; Dong, C.D.; Kao, C.M. Assessment of toxicity of polycyclic aromatic hydrocarbons in sediments of Kaohsiung Harbor, Taiwan. Sci. Total Environ. 2013, 463–464, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Filipkowska, A.; Kowalewska, G.; Pavoni, B.; Łęczyński, L. Organotin compounds in surface sediments from seaports on the Gulf of Gdańsk (southern Baltic coast). Environ. Monit. Assess. 2011, 182, 455–466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radke, B.; Wasik, A.; Jewell, L.L.; Piketh, S.; Paczek, U.; Gałuszka, A.; Namieśnik, J. Seasonal changes in organotin compounds in water and sediment samples from the semi-closed Port of Gdynia. Sci. Total Environ. 2012, 441, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Filipkowska, A.; Kovalewska, G. Butyltins in sediments from the Southern Baltic coastal zone: Is it still a matter of concern, 10 years after implementation of the total ban? Mar. Pollut. Bull. 2019, 146, 343–348. [Google Scholar] [CrossRef]
- Kim, N.S.; Hong, S.H.; An, J.G.; Shin, K.H.; Shim, W.J. Distribution of butyltins and alternative antifouling biocides in sediments from shipping and shipbuilding areas in South Korea. Mar. Pollut. Bull. 2015, 95, 484–490. [Google Scholar] [CrossRef]
- Lam, N.H.; Jeong, H.H.; Kang, S.D.; Kim, D.J.; Ju, M.J.; Horiguchi, T.; Cho, H.S. Organotins and new antifouling biocides in water and sediments from three Korean Special Management Sea Areas following ten years of tributyltin regulation: Contamination profiles and risk assessment. Mar. Pollut. Bull. 2017, 121, 302–312. [Google Scholar] [CrossRef]
- Champ, M.A. A review of organotin regulatory strategies, pending actions, related costs and benefits. Sci. Total Environ. 2000, 258, 21–71. [Google Scholar] [CrossRef]
- Chen, C.F.; Kao, C.M.; Dong, C.D.; Chen, C.W. Butyltin contamination in sediments and seawater from Kaohsiung Harbor, Taiwan. Environ. Monit. Assess. 2010, 169, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Finnegan, C.; Ryan, D.; Enright, A.M.; Garcia-Cabellos, G. Microbes a Tool for the Remediation of Organotin Pollution Determined by Static Headspace Gas Chromatography-Mass Spectrometry. Molecules 2018, 23, 627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filipkowska, A.; Kowalewska, G.; Pavoni, B. Organotin compounds in surface sediments of the Southern Baltic coastal zone: A study on the main factors for their accumulation and degradation. Environ. Sci. Pollut. Res. 2014, 21, 2077–2087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filipkowska, A.; Lubecki, L.; Szymczak-Żyła, M.; Ciesielski, T.M.; Jenssen, B.M.; Ardelan, M.V.; Mazur-Marzec, H.; Breedveld, G.D.; Oen, A.M.P.; Zamojska, A.; et al. Anthropogenic impact on marine ecosystem health: A comparative multi-proxy investigation of recent sediments in coastal waters. Mar. Pollut. Bull. 2018, 133, 328–335. [Google Scholar] [CrossRef] [Green Version]
- Sousa, A.C.A.; Oliveira, I.B.; Laranjeiro, F.; Takahashi, S.; Tanabe, S.; Cunha, M.R.; Barroso, C.M. Organotin levels in Nazaré canyon (west Iberian Margin, NE Atlantic) and adjacent coastal area. Mar. Pollut. Bull. 2012, 64, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Anastasiou, T.I.; Chatzinikolaou, E.; Mandalakis, M.; Arvanitidis, C. Imposex and organotin compounds in ports of the Mediterranean and the Atlantic: Is the story over? Sci. Total Environ. 2016, 569–570, 1315–1329. [Google Scholar] [CrossRef]
- Quintas, P.Y.; Arias, A.H.; Alvarez, M.B.; Domini, C.E.; Garrido, M.; Marcovecchio, J.E. Distribution of butyltin compounds in the coastal environment of the Bahía Blanca estuary, Argentina. Arch. Environ. Contam. Toxicol. 2021, 81, 307–323. [Google Scholar] [CrossRef]
- Okoro, H.K.; Fatoki, O.S.; Adekola, F.A.; Ximba, B.J.; Snyman, R.G. Spatio-temporal variation of organotin compounds in seawater and sediments from Cape Town harbor, South Africa using gas chromatography with flame photometric detector (GC-FPD). Arab. J. Chem. 2016, 9, 95–104. [Google Scholar] [CrossRef] [Green Version]
- Artifon, V.; Castro, Í.B.; Fillmann, G. Spatiotemporal appraisal of TBT contamination and imposex along a tropical bay (Todos os Santos Bay, Brazil). Environ. Sci. Pollut. Res. 2016, 23, 16047–16055. [Google Scholar] [CrossRef]
- Abreu, F.E.L.; Lima da Silva, J.N.; Castro, Í.B.; Fillmann, G. Are antifouling residues a matter of concern in the largest South American port? J. Hazard. Mater. 2020, 398, 122937. [Google Scholar] [CrossRef]
- Mukhtar, A.; Mohamat-Yusuff, F.; Zulkifli, S.Z.; Harino, H.; Ismail, A.; Inoue, K. Concentration of organotin and booster biocides in sediments of seagrass area from Sungai Pulai estuary, south of Johor, Malaysia. Environments 2019, 6, 26. [Google Scholar] [CrossRef] [Green Version]
- Zhang, K.; Shi, J.; He, B.; Xu, W.; Li, X.; Jiang, G. Organotin compounds in surface sediments from selected fishing ports along the Chinese coast. Chin. Sci. Bull. 2013, 58, 231–237. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Chen, L.; Li, F.; Huang, Q.; Wu, L.; Zhang, W. Urgent caution to trace organometal pollution: Occurrence, distribution and sources of methyltins, butyltins and phenyltins in sediments from South Hangzhou Bay, China. Environ. Pollut. 2019, 246, 571–577. [Google Scholar] [CrossRef]
- Abraham, M.; Westphal, L.; Hand, I.; Lerz, A.; Jeschek, J.; Bunke, D.; Leipe, T.; Schulz-Bull, D. TBT and its metabolites in sediments: Survey at a German coastal site and the central Baltic Sea. Mar. Pollut. Bull. 2017, 121, 404–410. [Google Scholar] [CrossRef]
- Filipkowska, A.; Złoch, I.; Wawrzyniak-Wydrowska, B.; Kowalewska, G. Organotins in fish muscle and liver from the Polish coast of the Baltic Sea: Is the total ban successful? Mar. Pollut. Bull. 2016, 111, 493–499. [Google Scholar] [CrossRef]
- Egardt, J.; Nilsson, P.; Dahllöf, I. Sediments indicate the continued use of banned antifouling compounds. Mar. Pollut. Bull. 2017, 125, 282–288. [Google Scholar] [CrossRef] [Green Version]
- Kucharski, D.; Drzewicz, P.; Nałęcz-Jawecki, G.; Mianowicz, K.; Skowronek, A.; Giebułtowicz, J. Development and application of a novel QuEChERS method for monitoring of tributyltin and triphenyltin in bottom sediments of the Odra River estuary, north westernmost part of Poland. Molecules 2020, 25, 591. [Google Scholar] [CrossRef] [Green Version]
- Díez, S.; Ábalos, M.; Bayona, J.M. Organotin contamination in sediments from the Western Mediterranean enclosures following 10 years of TBT regulation. Water Res. 2002, 36, 905–918. [Google Scholar] [CrossRef]
- Bartolomé, L.; Etxebarria, N.; Martínez-Arkarazo, I.; Raposo, J.C.; Usobiaga, A.; Zuloaga, O.; Raingeard, D.; Cajaraville, M.P. Distribution of organic microcontaminants, butyltins, and metals in mussels from the estuary of Bilbao. Arch. Environ. Contam. Toxicol. 2010, 59, 244–254. [Google Scholar] [CrossRef]



| Item | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | F10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Latitude (22° N) | 34′11.56″ | 34′09.18″ | 34′06.52″ | 34′02.94″ | 34′07.97″ | 34′10.83″ | 34′19.96″ | 34′16.91″ | 34′14.36″ | 34′11.72″ |
| Longitude (120° E) | 19′06.86″ | 19′02.28″ | 18′57.70″ | 18′53.37″ | 18′49.57″ | 18′54.00″ | 18′52.73″ | 18′49.12″ | 18′45.26″ | 18′40.78″ |
| Mud (<63 μm, %) | 66.3 | 73.3 | 87.9 | 86.6 | 88.9 | 89.0 | 73.1 | 82.7 | 98.2 | 70.2 |
| Sand (>63 μm, %) | 33.7 | 26.7 | 12.1 | 13.4 | 11.1 | 11.0 | 26.9 | 17.3 | 1.8 | 29.8 |
| TOC (%) | 11.8 | 13.9 | 4.38 | 8.66 | 6.76 | 5.7 | 8.42 | 7.48 | 3.89 | 2.11 |
| MBT (ngSn·g−1 dw) | 89.8 | <0.5 a | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 |
| DBT (ngSn·g−1 dw) | 291 | 20.7 | <0.6 | 9.9 | 7.9 | 5.4 | <0.6 | <0.6 | <0.6 | <0.6 |
| TBT (ngSn·g−1 dw) | 204 | 786 | 235 | 652 | 404 | 70.1 | <0.4 | 57.7 | 13.1 | <0.4 |
| ∑BTs (ngSn·g−1 dw) b | 585 | 807 | 236 | 662 | 412 | 75.8 | – | 57.7 | 13.7 | – |
| TBT contamination class c | high | severe | severe | severe | severe | high | non | high | moderate | non |
| Locations | Sampling Time | MBT | DBT | TBT | Reference |
|---|---|---|---|---|---|
| Port of Gdynia (Poland) | 2008 | 4–165 | 5–391 | 8–1910 | [32] |
| Port of Gdynia (Poland) | 2009 | 134–968 | 250–2716 | 1143–6408 | [33] |
| Port of Gdynia (Poland) | 2018 | 20.9–112 | 32.2–225 | 111–1120 | [34] |
| Port of Gdańsk (Poland) | 2008 | 7–684 | 9–2060 | 13–15780 | [32] |
| Port of Gdańsk (Poland) | 2018 | 40.4–490 | 80.1–889 | 122–1942 | [34] |
| Gulf of Gdańsk (Poland) | 2008 | 0.54–10.9 | 0.51–11.1 | 0.2–19.60 | [40] |
| Gulf of Gdańsk (Poland) | 2014 | 1.2–15.2 | 0.5–18.9 | 0.9–28.5 | [41] |
| Vistula Lagoon (Poland) | 2008 | 5.10–24.00 | <4.5–6.03 | 2.02–24.51 | [40] |
| Szczecin Lagoon (Poland) | 2008 | <1.0–33.97 | <1.2–38.66 | 2.39–97.98 | [40] |
| Oslofjord/Drammensfjord (Norway) | 2014 | 0.9–78.2 | 1.0–64.3 | n.d. a–145.0 | [41] |
| Nazaré Canyon (Portugal) | 2005 | <5.1–14 | <0.4–2.7 | <0.1–0.8 | [42] |
| Olhão (Portugal) | 2012 | 3.9 | 2.0 | 2.6 | [43] |
| Cagliari (Italy) | 2012 | 43.8 | 79.9 | 74.5 | [43] |
| El Kantaoui (Tunisi) | 2012 | 7.2 | 8.4 | 6.1 | [43] |
| Bahía Blanca Estuary (Argentina) | 2014 | <3.5–831 | <1.08–782 | <0.78–259 | [44] |
| Cape Town Harbor (South Africa) | 2011–2012 | n.a. b | n.a. | 10–829 | [45] |
| Todos os Santos Bay (Brazil) | 2010–2011 | <3–4.5 | 4–21 | <2–262 | [46] |
| Todos os Santos Bay (Brazil) | 2012 | <3–108 | <2–72 | <2–77 | [46] |
| Santos-São Vicente Estuarine (Brazil) | 2015 | <0.5–809 | <0.5–304 | <0.5–688 | [47] |
| Gulf of Mexico (Mexico) | 2018 | <0.3–195.8 | <0.3–79.0 | <0.3–90.6 | [7] |
| Sungai Pulai Estuary (Malaysia) | - | 6.5–12.2 | <0.1–6.1 | 8.1–10.6 | [48] |
| Masan, Haengam, and Gohyun Bay (Korea) | 2009 | 24–1048 | 22–455 | 13–1018 | [35] |
| Fishing port (Korea) | 2010 | 40–281 | 62–200 | 44–116 | [35] |
| Harbors (Korea) | 2010 | 15–6212 | n.d.–8747 | 3–55264 | [35] |
| Ulsan, Busan, and Gwangyang Bay (Korea) | 2014–2015 | <0.1–56.9 | <0.1–160 | <0.1–2304 | [36] |
| Fishing ports (China) | 2007 | <3.6–194 | <2.3–41.5 | <0.7–86 | [49] |
| Dumping sites along coastal (China) | 2012–2013 | n.d.–294.71 | 0.40–263.90 | 1.09–74.20 | [18] |
| South Hangzhou Bay (China) | 2013–2014 | n.d.–51.8 | n.d.–16.2 | n.d.–28.0 | [50] |
| Kaohsiung Harbor (Taiwan) | 2006 | n.d.–7.3 | n.d.–18.4 | 1.7–125 | [38] |
| Kaohsiung Harbor (Taiwan) | 2009 | 0.5–83.4 | 0.5–31.6 | 1.2–112 | [27] |
| Qianzhen Fishing Port (Taiwan) | 2003 | n.a. | n.a. | <2.4–7400 | [24] |
| Qianzhen Fishing Port (Taiwan) | 2020 | <0.5–89.8 | <0.6–291 | <0.4–786 | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.-H.; Chen, Y.-S.; Chen, C.-F.; Albarico, F.P.J.B.; Lim, Y.C.; Wang, M.-H.; Chen, C.-W.; Dong, C.-D. Butyltin Contamination in Fishing Port Sediments after the Ban of Tributyltin Antifouling Paint: A Case of Qianzhen Fishing Port in Taiwan. Water 2022, 14, 813. https://doi.org/10.3390/w14050813
Lee S-H, Chen Y-S, Chen C-F, Albarico FPJB, Lim YC, Wang M-H, Chen C-W, Dong C-D. Butyltin Contamination in Fishing Port Sediments after the Ban of Tributyltin Antifouling Paint: A Case of Qianzhen Fishing Port in Taiwan. Water. 2022; 14(5):813. https://doi.org/10.3390/w14050813
Chicago/Turabian StyleLee, Shu-Hui, Yung-Sheng Chen, Chih-Feng Chen, Frank Paolo Jay B. Albarico, Yee Cheng Lim, Ming-Huang Wang, Chiu-Wen Chen, and Cheng-Di Dong. 2022. "Butyltin Contamination in Fishing Port Sediments after the Ban of Tributyltin Antifouling Paint: A Case of Qianzhen Fishing Port in Taiwan" Water 14, no. 5: 813. https://doi.org/10.3390/w14050813
APA StyleLee, S.-H., Chen, Y.-S., Chen, C.-F., Albarico, F. P. J. B., Lim, Y. C., Wang, M.-H., Chen, C.-W., & Dong, C.-D. (2022). Butyltin Contamination in Fishing Port Sediments after the Ban of Tributyltin Antifouling Paint: A Case of Qianzhen Fishing Port in Taiwan. Water, 14(5), 813. https://doi.org/10.3390/w14050813







