Treatment and Recycle of Greenhouse Nutrient Feed Water Applying Hydrochar and Activated Carbon Followed by Reverse Osmosis
Abstract
Highlights
- Treatment by self-produced hydrochar (HC) and activated carbon (AC) from tomato plant biomass resolved the issue of toxicity by GNF.
- Other than phytotoxicity from excess ions, no impacts of the pathogens were observed from GNF.
- Higher than the limit concentrations of phytotoxic metal ions in GNF cause impaired plant growth.
- Imbalances of micronutrients in GNFs requires continuous analyses and adjustment.
Abstract
1. Introduction
2. Methods and Materials
2.1. Methods
2.2. Analyses
2.3. Materials
3. Results and Discussions
3.1. Microbial Plant Pathogens Analysis
3.2. Analyses of Leached GNFs including Source Well Water) and Treated GNFs
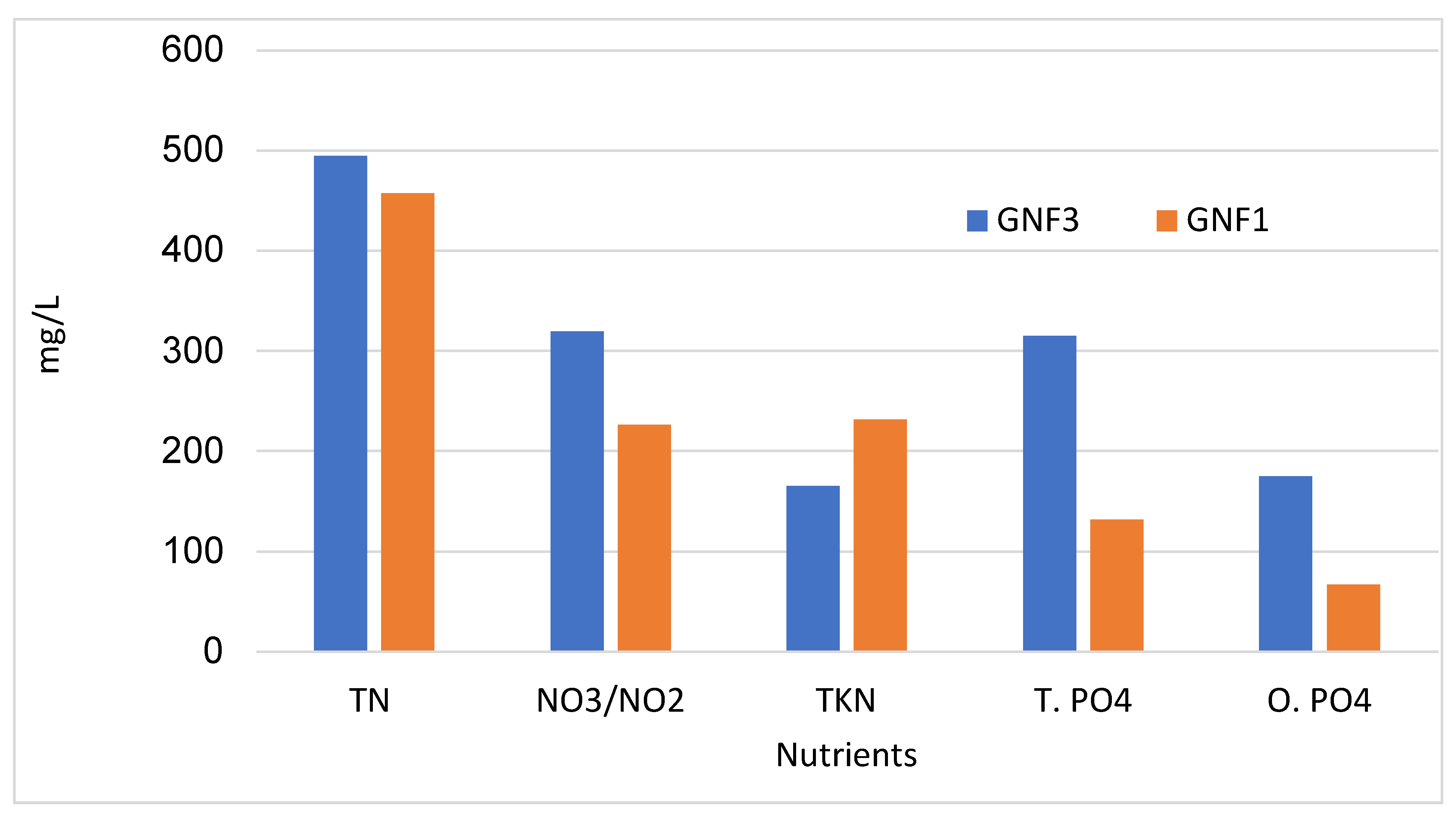
3.3. Evaluation of Nutrient Concentrations in all the GNFs
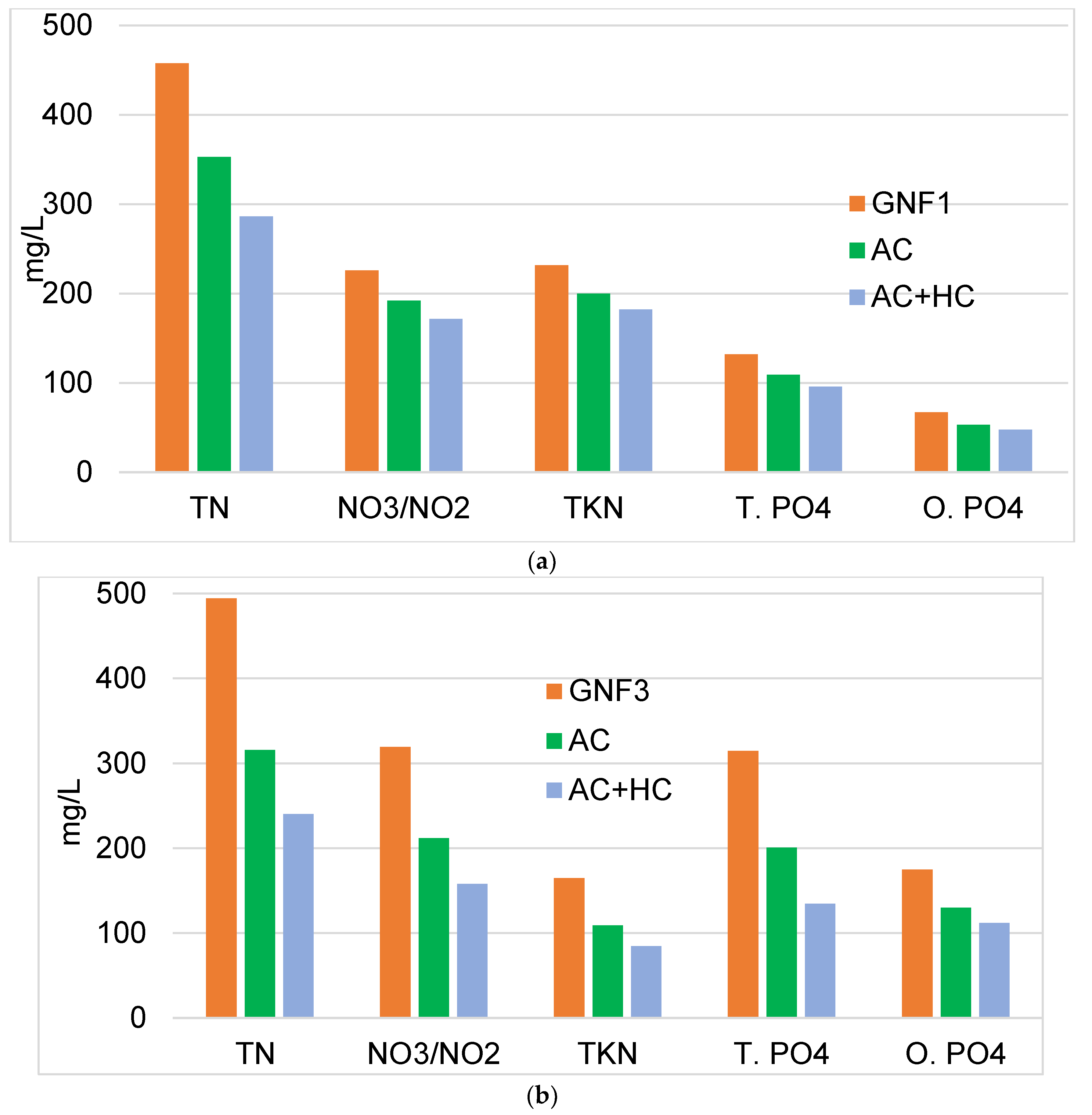
3.4. Treatments of GNFs by Produced Activated Carbon and Hydrochar
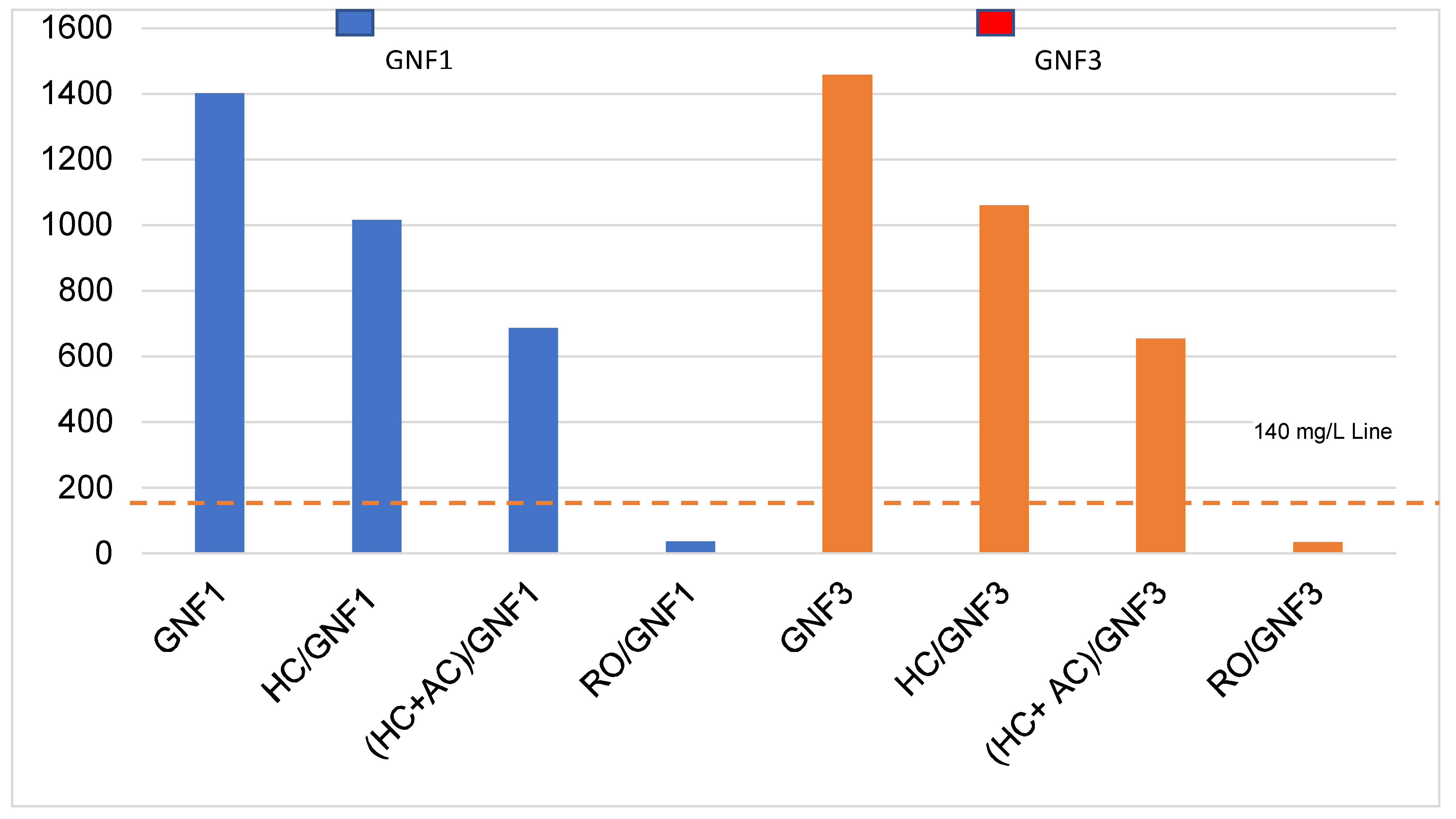
4. Application of Findings
Novelty
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations and Terms
| AC | Activated carbon |
| CF | Cartridge filter |
| Clear leach | Freshly prepared GNF |
| GH | Greenhouse |
| GH1 | 1st GH |
| GH2 | 2nd GH |
| GNF | Greenhouse nutrient feed |
| GNF1 | Clear leach from GH1 |
| GNF2 | Leached dirty GNF from GH2 |
| GNF3 | Leached dirty GNF from GH2 |
| HC | Hydrochar |
| HTC | Hydrothermal carbonization |
| Leached GNF | Circulated nutrients unbalanced dirty GNF |
| LMH | Liters per square meter hour |
| Micronutrients | Metallic nutrients (salts) |
| OMAFRA | Ontario Ministry of Agriculture, Food and Rural Affairs |
| PACL | Poly aluminum chloride |
| PWQA | Provincial Water Quality Objectives |
| RO | Reverse osmosis |
| TGNF | Treated GNF |
| TPB | Tomato plant biomass |
| WW | Well water |
References
- Bunch, K. Binational Plans Call for 40 Percent Reduction to Algal-Fueling Nutrients. 2018. Available online: https://www.ijc.org/en/binational-plans-call-40-percent-reduction-algal-fueling-nutrients (accessed on 15 May 2022).
- Grimm, A. Irrigation water quality challenges. GREENHOUSE CANADA. 2008. Available online: https://www.greenhousecanada.com/irrigation-water-quality-challenges-887 (accessed on 15 May 2022).
- WHO (World Health Organization). Guidelines for the Safe Use of Wastewater, Excreta and Greywater. V2: Wastewater Use in Agriculture. Geneva: World Health Organization (WHO); Food and Agriculture Organization of the United Nations (FAO): Rome, Italy; United Nations Environment Program (UNEP): Nairobi, Kenya, 2006; Available online: https://www.who.int/publications/i/item/9241546832 (accessed on 15 May 2022).
- Jiménez, B.; Mara, D.; Carr, R.; Brissaud, F. Wastewater treatment for pathogen removal and nutrient conservation: Suitable systems for use in developing countries. In Wastewater Irrigation and Health. Assessing and Mitigating Risk in Low-Income Countries; Drechsel, P., Scott, C.A., Raschid-Sally, L., Redwood, M., Bahri, A., Eds.; IWMI: Colombo, Sri Lanka, 2010. [Google Scholar]
- Xu, X.; Du, F.; Wang, J.; Sha, Q.; Chen, G.; Tian, Z.; Zhu, S.; Ge, S.; Jiang, Y. Effects of Potassium Levels on Plant Growth, Accumulation and Distribution of Carbon, and Nitrate Metabolism in Apple Dwarf Rootstock Seedlings. Front. Plant Sci. 2020, 11, 904. [Google Scholar] [CrossRef] [PubMed]
- van Os, E.; Blok Voogt, W.; Waked, L. Water Quality and Salinity Aspects in Hydroponic Cultivation; WUR Glastuinbouw: Wageningen, The Netherlands, 2016; Available online: https://edepot.wur.nl/403810 (accessed on 15 May 2022).
- Elliott, M.; Pernezny, K.; Palmateer, A.; Havranek, N. Guidelines to Identification and Management of Plant Disease Problems: Part I. and 2 Eliminating Insect Damage and Abiotic Disorders; EDIS: Gainesville, FL, USA, 2008; p. 248. Available online: https://edis.ifas.ufl.edu/pdf%5Carchived%5CMG%5CMG441%5CMG441-5806128.pdf (accessed on 16 May 2022).
- De Kreij, C.; Voogt, W.; Baas, R. Nutrient Solutions and Water Quality for Soilless Cultures; Applied Plant Research, Division Glasshouse: Naaldwijk, Holland, 2003. [Google Scholar]
- Richard, S.; Zheng, Y.; Dixon, M. Greenhouse Canada—To Recycle or Not to Recycle? 2008. Available online: https://www.greenhousecanada.com/to-recycle-or-not-to-recycle-926 (accessed on 16 May 2022).
- Makoto, K.; Kim, Y.; Koike, T. Buried charcoal layer and ectomycorrhizae cooperatively promote the growth of Larix gmelinii seedlings. Plant Soil 2010, 327, 143–152. [Google Scholar] [CrossRef]
- Sanogo, S.; Moorman, G. Transmission and Control of Pythium aphanidermatum in an Ebb-and-Flow Subirrigation System. Plant Dis. 1993, 77, 287. [Google Scholar] [CrossRef]
- Scarlett, K.; Tesoriero, L.; Daniel, R.; Guest, D. Detection and Quantification of Fusarium Oxysporum f. Sp. Cucumerinum in Environmental Samples using a Specific Quantitative PCR Assay. Eur. J. Plant Pathol. 2013, 137, 315–324. [Google Scholar] [CrossRef]
- Salm, H.; Gelder, K. Real-time PCR for detection and quantification of Erwinia amylovora, the causal agent of fireblight. Plant Pathol. 2004, 53, 602–610. [Google Scholar] [CrossRef]
- Weller, S.A.; Elphinstone, J.G.; Smith, N.C.; Boonham, N.; Stead, D.E. Detection of Ralstonia solanacearum Strains with a Quantitative, Multiplex, Real-Time, Fluorogenic PCR (TaqMan) Assay. Appl. Environ. Microbiol. 2000, 66, 2853–2858. [Google Scholar] [CrossRef]
- Scholthof, K.-B.G.; Adkins, S.; Czosnek, H.; Palukaitis, P.; Jacquot, E.; Hohn, T.; Hohn, B.; Saunders, K.; Candresse, T.; Ahlquist, P.; et al. Top 10 plant viruses in molecular plant pathology. Mol. Plant Pathol. 2011, 12, 938–954. [Google Scholar] [CrossRef]
- Hamad, S.; Al-ani, R.; Adhab, M.; Diwan, S. Tomato Yellow Leaf Curl Virus, Identification, Virus Vector Relationship, Strains Characterization and a Suggestion for Its Control with Plant Extracts in Iraq. Afr. J. Agric. Res. 2011, 6, 5149–5155. [Google Scholar] [CrossRef]
- Gu, Q.; Sheng, L.; Zhang, T.; Lu, Y.; Zhang, Z.; Zheng, K.; Hu, H.; Zhou, H. Early detection of tomato spotted wilt virus infection in tobacco using the hyperspectral imaging technique and machine learning algorithms. Comput. Electron. Agric. 2019, 167, 105066. [Google Scholar] [CrossRef]
- Kulshrestha, S.; Sharma, A.; Seth, C. Molecular biology of Tomato spotted wilt virus: An update. J. Appl. Hortic. 2013, 15, 71–80. [Google Scholar] [CrossRef]
- Roberts, C.A.; Dietzgen, R.G.; Heelan, L.A.; Maclean, D.J. Real-time RT-PCR fluorescent detection of tomato spotted wilt virus. J. Virol. Methods 2000, 88, 1–8. [Google Scholar] [CrossRef]
- OMAFRA. 2020. Available online: http://www.omafra.gov.on.ca/english/nm/regs/gnfpro/gnfreg.htm (accessed on 12 April 2020).
- Graham, T. New Perspectives on the Maintenance of Aqueous Ozone Residuals in Greenhouse and Nursery Irrigation Solutions. Ph.D. Thesis, University of Guelph, Guelph, ON, Canada, 2012. [Google Scholar]
- Ristvey, A.G.; Belayneh, B.E.; Lea-Cox, J.D. A Comparison of Irrigation-Water Containment Methods and Management Strategies Between Two Ornamental Production Systems to Minimize Water Security Threats. Water 2019, 11, 2558. [Google Scholar] [CrossRef]
- Jamal-Uddin, A.; Zytner, P.; Zytner, R. Hybrid treatment system to remove micromolecular SMPs from fruit wastewater treated with an MBR. Can. J. Civ. Eng. 2022, 49, 548–557. [Google Scholar] [CrossRef]
- Jamal-Uddin, A. Treatments and Recycle of Food Processor Wastewater Effluent. Master’s Theses, University of Guelph, Guelph, ON, Canada, 2019. [Google Scholar]
- Metcalf & Eddy. Wastewater Engineering Treatment and Resource Recovery, 5th ed.; Mc Graw Hill education: New York, NY, USA, 2014. [Google Scholar]
- USEPA. Guidelines for Water Reuse; Environmental Protection Agency (USEPA): Washington, DC, USA, 2012. Available online: https://www.epa.gov/sites/default/files/2019-08/documents/2012-guidelines-water-reuse.pdf (accessed on 20 April 2020).
- Stevik, T.K.; Aa, K.; Ausland, G.; Hanssen, J.F. Retention and removal of pathogenic bacteria in wastewater percolating through porous media: A review. Water Res. 2004, 38, 1355–1367. [Google Scholar] [CrossRef] [PubMed]
- Keraita, B.; Drechsel, P.; Klutse, A.; Cofie, O. On-Farm Treatment Options for Wastewater, Greywater and Fecal Sludge with Special Reference to West Africa; IWMI: Colombo, Sri Lanka, 2014; Volume 1, p. 32. Available online: https://hdl.handle.net/10568/35209 (accessed on 20 April 2020).
- Peters, T. Review: Membrane technology for water treatment. Chem. Eng. Technol. 2010, 33, 1233–1240. [Google Scholar] [CrossRef]
- Huyben, D.C. Evaluation of Membrane Filtration and UV Irradiation for The Control of Flavobacterium Psychrophilum in Recirculation Aquaculture Systems. Master’s Theses, University of Guelph, Guelph, ON, Canada, 2012. [Google Scholar]
- Suárez, A.; Fidalgo, T.; Riera, F.A. Recovery of dairy industry wastewaters by reverse osmosis. Production of boiler water. Sep. Purif. Technol. 2014, 133, 204–211. [Google Scholar] [CrossRef]
- De Jager, D.; Sheldon, M.S.; Edwards, W. Colour removal from textile wastewater using a pilot-scale dual-stage MBR and subsequent RO system. Sep. Purif. Technol. 2014, 135, 135–144. [Google Scholar] [CrossRef]
- Tomczak, W.; Gryta, M. The Application of Cellulose Acetate Membranes for Separation of Fermentation Broths by the Reverse Osmosis: A Feasibility Study. Int. J. Mol. Sci. 2022, 23, 11738. [Google Scholar] [CrossRef]
- Gunathilake, K.D.P.P.; Yu, L.J.; Rupasinghe, H.P.V. Reverse Osmosis as A Potential Technique to Improve Antioxidant Properties of Fruit Juices Used for Functional Beverages. Food Chem. 2014, 148, 335–341. [Google Scholar] [CrossRef]
- Öner, Ş.G.; Kabay, N.; Güler, E.; Kitiş, M.; Yüksel, M. A comparative study for the removal of boron and silica from geothermal water by crossflow flat sheet reverse osmosis method. Desalination 2011, 283, 10–15. [Google Scholar] [CrossRef]
- Sahar, E.; David, I.; Gelman, Y.; Chikurel, H.; Aharoni, A.; Messalem, R.; Brenner, A. The use of RO to remove emerging micropollutants following CAS/UF or MBR treatment of municipal wastewater. Desalination 2011, 273, 142–147. [Google Scholar] [CrossRef]
- Guo, X.; Zhang, Z.; Fang, L.; Su, L. Study on ultrafiltration for surface water by a polyvinylchloride hollow fiber membrane. Desalination 2009, 238, 183–191. [Google Scholar] [CrossRef]
- Liu, M.; Lau, A.K.; Lo, K. Ultrafiltration tests for the reutilization of greenhouse wastewater. J. Environ. Sci. Health Part B 1999, 34, 1101–1110. [Google Scholar] [CrossRef]
- Pernezny, K.; Elliott, M.; Palmateer, A.; Havranek, N. Guidelines for Identification and Management of Plant Disease Problems: Part II. Diagnosing Plant Diseases Caused by Fungi, Bacteria and Viruses; Florida EDIS: Charleston, WV, USA, 2008; p. 249. Available online: https://edis.ifas.ufl.edu/pdf%5Carchived%5CMG%5CMG442%5CMG442-15333041.pdf (accessed on 16 May 2022).
- OMAFRA. Self-Assessment and Best Management Practices for Water and Fertilizer Use in Greenhouse Vegetable Production. 2019. Available online: http://www.omafra.gov.on.ca/english/environment/bmp/greenhouse.pdf (accessed on 12 April 2020).
- O. Reg. 300/14. Greenhouse Nutrient Feedwater Regulation, under Nutrient Management Act. 2014. Available online: http://www.omafra.gov.on.ca/english/nm/regs/gnfpro/gnfreg.htm (accessed on 21 February 2020).
- Jamal-Uuddin, A.; Hassan, A.M.; Al-Reweli, A.; Al-Rubaian, A.; Hauge, L. An Efficient Energy Recovery Prospect In SWRO Process. In Proceedings of the 6th Saudi Engineering Conference, Dhahran, Saudi Arabia, 14–17 December 2002. [Google Scholar]
- Schnitzler, W.H. Pest and disease management of soilless culture. Acta Hortic. 2004, 648, 191–203. [Google Scholar] [CrossRef]
- McPherson, G.M.; Harriman, M.R.; Pattisson, D. The potential for spread of root diseases in recirculating hydroponic systems and their control with disinfection. Meded. Fac. Landbouwkd. Toegep. Biol. Wet. Univ. Gent 1995, 60, 371–379. Available online: https://agris.fao.org/agris-search/search.do?recordID=BE9600917 (accessed on 12 March 2020).
- Neitsch, S.L.; Arnold, J.G.; Kiniry, J.R.; Williams, J.R. Soil and Water Assessment Tool, Theoretical Documentation Version 2009. Texas Water Resources Institute Technical Report No. 406. 2011. Available online: https://swat.tamu.edu/media/99192/swat2009-theory.pdf (accessed on 12 January 2020).
- Dagnew, A.; Scavia, D.; Wang, Y.; Muenich, R.; Kalcic, M. Modeling phosphorus reduction strategies from the international St. Clair-Detroit River system watershed. J. Great Lakes Res. 2019, 45, 742–751. [Google Scholar] [CrossRef]





| Bacteria | Virus | Fungi | Micron Filter | K Ion (K+) | RO Pore Size |
|---|---|---|---|---|---|
| 200–1000 nm | 20–400 nm | 2000–10,000 nm | 200 nm | 0.15 nm | 0.1–1.5 nm |
| 2–10 µ | 0.20 µ | 152 pm | 1–15 Å | ||
| 174–871 Da | 17–348 Da | 1741–8707 Da | 174 Da | 0.13 Da | 0.09–1.3 Da |
| (a) | ||||
|---|---|---|---|---|
| GNF1 | Alum coagulation | PACL treatment | RO filtration | |
| pH | 7.05 | 6.63 | 6.55 | 6.8 |
| Conductivity (µS/cm) | 1262 | 1300 | 1440 | 24 |
| TDS (mg/L) | 770 | 788 | 798 | 13 |
| Turbidity (NTU) | 2.2 | 0.24 | 0.07 | Nil |
| (b) | ||||
| Samples Type | Sodium-mg/L (mmol/L) | Potassium–mg/L (mmol/L) | Magnesium–mg/L (mmol/L) | Calcium–mg/L (mmol/L) |
| Raw GNF1 water | 61 (2.65) | 670 (17.18) | 81 (3.33) | 291 (14.55) |
| CF (11 µ) | 25 (1.09 | 444 (11.38) | 45 (1.85) | 107 (5.35) |
| RO filtrate | 1 (0.04) | 20 (0.5) | <1 (0.00) | 1 (0.05) |
| Alum treatment | ||||
| 35 mg/L dosage | 21 (0.91) | 458 (11.74) | 47 (1.93) | 109 (5.45) |
| 40 mg/L dosage | 21 (0.91) | 456 (11.69) | 46 (1.89) | 106 (5.30) |
| 50 mg/L dosage | 22 (0.96) | 775 (19.87) | 32 (1.32) | 57 (2.85) |
| PACL treatments | ||||
| 150 mg/L dosage | 22 (0.96) | 475 (12.18) | 48 (1.98) | 108 (5.40) |
| 100 mg/L dose | 23 (1.00) | 644 (16.51) | 47 (1.93) | 121 (6.05) |
| Components | Target for Closed System | Target for Open System | Root Zone Target Values | Unit Conversion Factors mmol/L–mg/L | |
|---|---|---|---|---|---|
| EC mS/cm | 1.60 | 2.60 | 3.70 | ||
| pH | 5.5 | ||||
| K+ | 6.50 | 9.50 | 8.00 | 1 | 39.1 |
| Ca++ | 2.75 | 5.40 | <8.00 | 1 | 20.1 |
| Na+ | <8.00 | 1 | 23.0 | ||
| Mg+ | 1.00 | 2.40 | 4.50 | 1 | 24.3 |
| NO3− | 10.75 | 16.00 | 23.00 | 1 | 62.0 |
| Cl− | <12.00 | 1 | 35.5 | ||
| HCO3− | <1.000 | 1 | 61.0 | ||
| H2PO4− | 1.25 | 1.50 | 1.00 | 1 | 91.0 |
| PO4−3 | 1 | 26.3 | |||
| (a) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Leached GNF2 | Alum Coagulation (57 mg/L Dosage) | PACL Treatment (100 mg/L Dosage) | RO Filtrate ** | |||||
| Adjusted pH | Adjusted pH | Adjusted pH | ||||||
| pH * | 3.77 | 6.34 | 4.51 | 5.90 | 3.82 | 3.79 | 3.49 | 5.65 |
| Conductivity (µS/cm) | 2430 | 2038 | 2360 | 1913 | 2450 | 2012 | 439 | 71.90 |
| TDS (mg/L) | 1334 | 1260 | 1281 | 1048 | 1324 | 1151 | 225 | 35.70 |
| Turbidity (NTU) | 1.40 | 2.28 | 0.17 | 0.42 | 0.10 | 0.15 | 0.10 | 0.12 |
| (b) | ||||||||
| Samples Type | Sodium mg/L (mmol/L) | Potassium mg/L (mmol/L) | Magnesium mg/L (mmol/L) | Calcium mg/L (mmol/L) | ||||
| Leached GNF2 | 85 (3.70) | 599 (15.36) | 72 (2.96) | 255 (12.75) | ||||
| RO filtrate | 3 (0.13) | 25 (0.64) | 4 (0.16) | 6 (0.30) | ||||
| (a) | ||||
|---|---|---|---|---|
| GNF3 | Alum Dose (59 mg/L) | PACL Dose (200 µL/L) | RO Filtrate | |
| pH | 4.34 | 6.00 | 3.68 | 3.88 |
| Conductivity (µS/cm | 2490 | 2670 | 2770 | 170 |
| TDS (mg/L) | 1467 | 1495 | 1544 | 89 |
| Turbidity (NTU) | 0.28 | 0.12 | 0.42 | 0.002 |
| (b) | ||||
| Samples Type | Sodium mg/L (mmol/L) | Potassium mg/L (mmol/L) | Magnesium mg/L (mmol/L) | Calcium mg/L (mmol/L) |
| GNF3 | 70 (3.04) | 614 (15.74) | 182 (7.49) | 329 (16.45) |
| RO filtration | 2 (0.09) | 17 (0.44) | 3 (0.12) | 6 (0.30) |
| Alum treatments | ||||
| 52 mg/L dosage | 101 (4.39) | 623 (15.97) | 185 (7.61) | 336 (16.80) |
| 60 mg/L dosage | 102 (4.43) | 633 (16.23) | 183 (7.53) | 341 (17.05) |
| PACL treatments | ||||
| 200 mg/L dosage | 71 (3.09) | 619 (15.87) | 187 (7.70) | 338 16.90) |
| 150 mg/L dosage | 73 (3.17) | 627 (16.08) | 189 (7.78) | 342 (17.10) |
| Nutrients–mg/L | GNF1 | GNF2 | GNF3 | WW | PWQO |
|---|---|---|---|---|---|
| NO3, & NO2-N (mmol/L) | 226.00 (3.65) | 184.00 (2.97) | 319.50 (5.15) | 0.50 (0.01) | 20 µg/L (Unionized NH3) |
| TKN | 231.50 | 175.00 | 165.00 | 151.00 | |
| TN (mmol/L) | 457.50 (32.68) | 358.50(25.61) | 494.50(35.32) | 151.50(10.61) | |
| Phosphate total PO4−3 (mmol/L) | 132.00 (4.17) | 154.2 (4.87) | 315.00 (9.95) | 41.9 (1.32) | 20 µg/L |
| Phosphate reactive (mmol/L) | 67.00(2.12) | 70.50(2.23) | 175.00(5.53) | 0.50(0.02) | |
| Free chlorine | 0.10 | 0.20 | 0.02 | 0.00 | |
| Chlorine total | 0.23 | 0.22 | 0.10 | 0.02 | |
| Aluminum | UMR | Negative | 0.051 | Negative | |
| pH | 7.05 | 3.77 | 4.34 | 7.75 |
| Class 1 Source Water for Hydroponic (OMAFRA) | Leached GNF Safe Land Disposal (O. Reg. 300/14) | ||
|---|---|---|---|
| Electric conductivity (EC) | <500 µS/cm | NH3/NH4 | 0.10 mg/L |
| Na (mg/L) | <30 | Nitrite, Nitrate | 0.04 mg/L |
| Cl (mg/L) | <50 | TKN | 0.05 mg/L |
| SO4 (mg/L) | <100 | Phosphorous | 0.10 mg/L |
| Raw Well Water | RO Filtrate | Leached GNF2 | RO Filtrate | |
|---|---|---|---|---|
| pH | 7.75 | 5.00 | 3.82 (adj. to 6.4) | 5.23 |
| Conductivity (µS/cm) | 565 | 37 | 2038 | 59 |
| TDS (mg/L) | 297 | 18.34 | 1201 | 37 |
| Turbidity (NTU) | 11.43 | 0.06 | 0.18 | 0.10 |
| Na–mg/L (mmol/L) | 49 (2.13) | 2 (0.05) | 54 (2.22) | 2 (0.05) |
| K–mg/L (mmol/L) | 3 (0.13) | 0.00 | 583 (23.99) | 22 (0.55) |
| Mg–mg/L (mmol/L) | 25 (1.09) | 4 (0.10) | 87 (3.58) | 4 (0.10) |
| Ca–mg/L (mmol/L) | 96 (4.17) | 5 (0.13) | 291 (11.98) | 5 (0.13) |
| (a) | |||||||
|---|---|---|---|---|---|---|---|
| Raw GNF | Treated GNF | ||||||
| PO43− (mg/L) | RO | AC1 | AC2 | HC1 | HC2 | HC3 | |
| GNF1 | 0.61 | 0.0096 | 3.3 | 1.14 | 1.15 | 0.78 | |
| GNF3 | 1.85 | 0.0221 | 2.5 | 0.80 | 1.95 | 2.04 | 3.46 |
| Removal from GNF1 | 99% | ||||||
| Removal from GNF3 | 98% | ||||||
| (b) | |||||||
| Untreated GNF Water (mg/L) | AC400 treated water (mg/L) | ||||||
| GNF1 | GNF3 | GNF1 | GNF3 | ||||
| Total nitrogen (TN) | 457.00 | 494.50 | 188.00 | 199.80 | |||
| NO3 and NO2-N | 226.00 | 319.50 | 188.00 | 188.20 | |||
| TKN | 231.00 | 165.00 | 0 | 11.48 | |||
| Reactive-ortho PO4 | 67.00 | 70.50 | 13.60 | 63.20 | |||
| Total phosphate (TP) | 132.00 | 154.20 | 25.20 | 125.40 | |||
| Total nitrogen (TN)% removal | 59 | 60 | |||||
| NO3 & NO2−% removal | 17 | 41 | |||||
| TKN% removal | - | 93 | |||||
| Ortho PO4% removal | 80 | 64 | |||||
| Total phosphate (TP)% removal | 81 | 19 | |||||
| TKN, TN, Ortho PO4 and TP removal% by RO | 98–99 | 97–99 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jamal-Uddin, A.-T.; Matsuura, T.; Al-Daoud, F.; Zytner, R.G. Treatment and Recycle of Greenhouse Nutrient Feed Water Applying Hydrochar and Activated Carbon Followed by Reverse Osmosis. Water 2022, 14, 3573. https://doi.org/10.3390/w14213573
Jamal-Uddin A-T, Matsuura T, Al-Daoud F, Zytner RG. Treatment and Recycle of Greenhouse Nutrient Feed Water Applying Hydrochar and Activated Carbon Followed by Reverse Osmosis. Water. 2022; 14(21):3573. https://doi.org/10.3390/w14213573
Chicago/Turabian StyleJamal-Uddin, Abu-Taher, Takashi Matsuura, Fadi Al-Daoud, and Richard G. Zytner. 2022. "Treatment and Recycle of Greenhouse Nutrient Feed Water Applying Hydrochar and Activated Carbon Followed by Reverse Osmosis" Water 14, no. 21: 3573. https://doi.org/10.3390/w14213573
APA StyleJamal-Uddin, A.-T., Matsuura, T., Al-Daoud, F., & Zytner, R. G. (2022). Treatment and Recycle of Greenhouse Nutrient Feed Water Applying Hydrochar and Activated Carbon Followed by Reverse Osmosis. Water, 14(21), 3573. https://doi.org/10.3390/w14213573






