Diversity of Silica-Scaled Chrysophytes in Central Vietnam
Abstract
1. Introduction
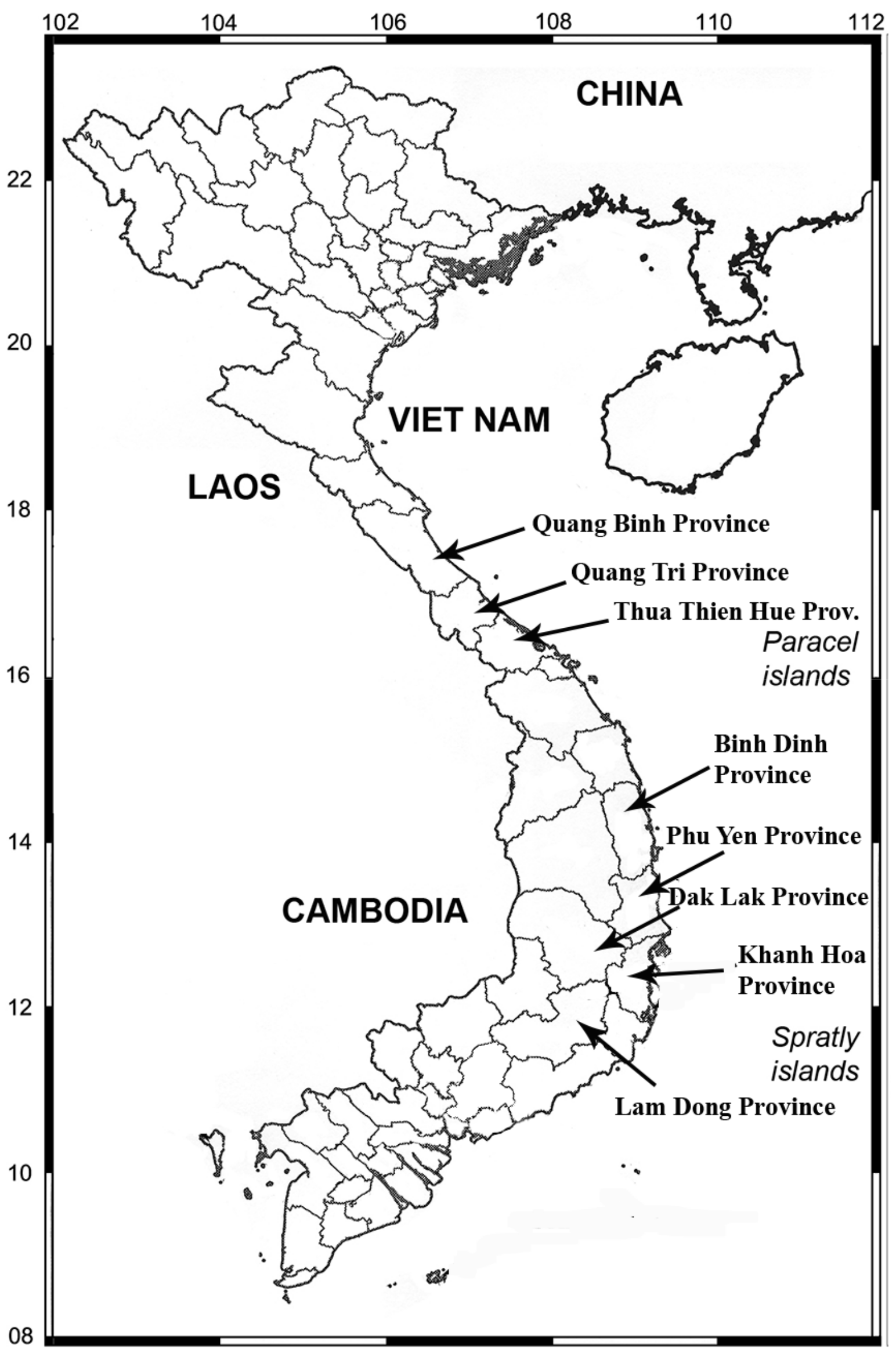
2. Materials and Methods
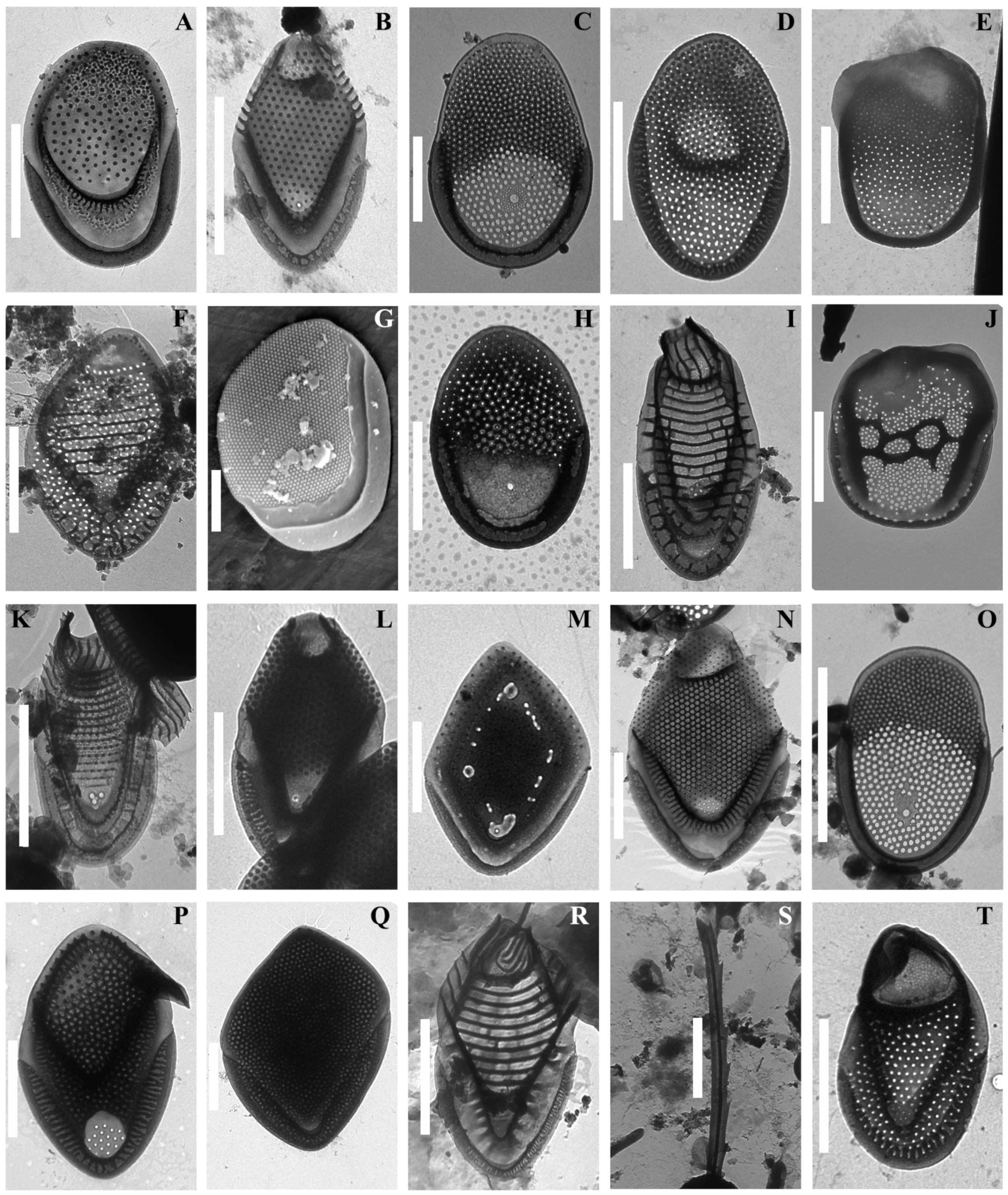
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dudgeon, D. The Ecology of Tropical Asian Rivers and Streams in Relation to Biodiversity Conservation. Annu. Rev. Ecol. Syst. 2000, 31, 239–263. [Google Scholar] [CrossRef]
- Santos, L.; Santos, F.; Gil, C.; Pereira, M. Diversity of Freshwater Algae. In Aquatic Ecology of the Mondego River Basin Global Importance of Local Experience; Graca, M.A., Pardal, M.A., Marques, J.C., Eds.; Imprensa da Universidade de Coimbra: Coimbra, Portugal, 2002; pp. 63–103. [Google Scholar]
- Andersen, R.A. Diversity of Eukaryotic Algae. Biodivers. Conserv. 1992, 1, 267–292. [Google Scholar] [CrossRef]
- Beaudrot, L.; Ahumada, J.A.; O’Brien, T.; Alvarez-Loayza, P.; Boekee, K.; Campos-Arceiz, A.; Eichberg, D.; Espinosa, S.; Fegraus, E.; Fletcher, C.; et al. Standardized Assessment Of Biodiversity Trends In Tropical Forest Protected Areas: The End Is Not in Sight. PLoS Biol. 2016, 14, e1002357. [Google Scholar] [CrossRef]
- Mittermeier, R.A.; Turner, W.R.; Larsen, F.W.; Brooks, T.M.; Gascon, C. Global Biodiversity Conservation: The Critical Role of Hotspots. In Biodiversity Hotspots: Distribution and Protection of Conservation Priority Areas; Zachos, F.E., Habel, J.C., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 3–22. [Google Scholar] [CrossRef]
- Van Dijk, P.P.; Tordoff, A.W.; Fellowes, J.; Lau, M.; Ma, J.S. Indo-Burma. In Hotspots Revisited: Earth’s Biologically Richest and Most Endangered Terrestrial Ecoregions; Mittermeier, R.A., Robles-Gil, P., Hoffmann, M., Pilgrim, J., Brooks, T., Mittermeier, C.G., Lamoreux, J., da Fonseca, G.A.B., Eds.; CEMEX, Monterrey Conservation International: Washington, DC, USA; Mexico City, Mexico, 2004; pp. 323–330. ISBN 968-639-77-79. [Google Scholar]
- Vyverman, W. The Indo-Malaysian North-Australian Phycogeographical Region Revised. Hydrobiologia 1996, 336, 107–120. [Google Scholar] [CrossRef]
- WEPA. Water Environment Partnership in Asia Under the Initiative of the Ministry of the Environment of Japan. 2013. Available online: http://www.wepa-db.net/index.html (accessed on 18 November 2021).
- Allen, D.J.; Smith, K.G.; Darwall, W.R.T. The Status and Distribution of Freshwater Biodiversity in Indo-Burma; IUCN: Cambridge, UK; Gland, Switzerland, 2012; p. 160. ISBN 978-2-8317-1424-0. [Google Scholar]
- Pavlov, D.S.; Zvorykin, D.D. Inland Water Ecology of Vietnam; KMK Press: Moscow, Russia, 2014; p. 435. (In Russian) [Google Scholar] [CrossRef]
- Zagumyonnyi, D.G.; Gusev, E.S.; Mylnikov, A.A.; Mylnikov, A.P. Novel Species of Centrohelid Heliozoans (Centroplasthelida) for Freshwaters of Central and Southern Vietnam. Inland Water Biol. 2020, 13, 358–371. [Google Scholar] [CrossRef]
- Tran, Q.H. The First Data on Testate Amoebae in the Coco River in Quang Nam and Da Nang Provinces, Vietnam. Mosc. Univ. Biol. Sci. Bull. 2019, 74, 207–214. [Google Scholar] [CrossRef]
- Tran, Q.H. First Data on Testate Amoeba Composition in Tropical Karst Wetlands of Northern Vietnam in Relation to Type of Biotope and Season: New Bioindication Potentialities. Inland Water Biol. 2020, 13, 251–261. [Google Scholar] [CrossRef]
- Tran, Q.H.; Tran, V.T.H.; Tikhonenkov, D.V. Freshwater Testate Amoebae from Waterbodies of North Vietnam with the Finding of Indicator Species. Limnology 2021, 22, 151–160. [Google Scholar] [CrossRef]
- Gusev, E.S.; Kulikovskiy, M.S. Centric Diatoms from Vietnam Reservoirs with Description of One New Urosolenia Species. Nova Hedwig. Beih. 2014, 143, 111–126. [Google Scholar] [CrossRef]
- Glushchenko, A.M.; Kuznetsova, I.V.; Kociolek, J.P.; Kulikovskiy, M.S. Karthickia verestigmata gen. et sp. nov.—An Interesting Diatom with Frustular Morphology Similar to Several Different Cymbelloid Diatom Genera. Phycologia 2019, 58, 605–613. [Google Scholar] [CrossRef]
- Glushchenko, A.M.; Kociolek, J.P.; Kuznetsova, I.V.; Kezlya, E.M.; Kulikovskiy, M.S. Prestauroneis genkalii—A New Diatom Species (Bacillariophyceae: Stauroneidaceae) from Southeast Asia. Phytotaxa 2019, 414, 156–164. [Google Scholar] [CrossRef]
- Glushchenko, A.M.; Kociolek, J.P.; Kuznetsova, I.V.; Kezlya, E.M.; Kulikovskiy, M.S. Neidiomorpha gusevii—A New Diatom Species (Bacillariophyceae: Neidiaceae) from Southeast Asia. Phytotaxa 2019, 415, 279–285. [Google Scholar] [CrossRef]
- Kezlya, E.; Glushchenko, A.; Maltsev, Y.; Gusev, E.; Genkal, S.; Kuznetsov, A.; Kociolek, J.P.; Kulikovskiy, M. Placoneis cattiensis sp. nov.—A New Diatom (Bacillariophyceae: Cymbellales) Soil Species from Cát Tiên National Park (Vietnam). Phytotaxa 2020, 460, 237–248. [Google Scholar] [CrossRef]
- Kezlya, E.; Glushchenko, A.; Maltsev, Y.; Gusev, E.; Genkal, S.; Kociolek, J.P.; Kulikovskiy, M. Three New Species from Diatom Genus Placoneis (Bacillariophyceae: Cymbellales) with Comments on Cryptic Diversity in Placoneis elginensis-Group. Water 2021, 13, 3276. [Google Scholar]
- Kulikovskiy, M.; Maltsev, Y.; Glushchenko, A.; Kuznetsova, I.; Kapustin, D.; Gusev, E.; Lange-Bertalot, H.; Genkal, S.; Kociolek, J.P. Gogorevia, a New Monoraphid Diatom Genus for Achnanthes exigua and Allied Taxa (Achnanthidiaceae) Described on the Basis of an Integrated Molecular and Morphological Approach. J. Phycol. 2020, 56, 1601–1613. [Google Scholar] [CrossRef]
- Martynenko, N.A.; Gusev, E.S.; Kapustin, D.A.; Guseva, E.E.; Kulikovskiy, M.S. Cryptomonas cattiensis sp. nov. (Cryptophyceae: Cryptomonadales), a New Species Described from Vietnam. Phytotaxa 2020, 454, 127–136. [Google Scholar] [CrossRef]
- Gusev, E.; Podunay, Y.; Martynenko, N.; Shkurina, N.; Kulikovskiy, M. Taxonomic Studies of Cryptomonas lundii Clade (Cryptophyta: Cryptophyceae) with Description of a New Species from Vietnam. Fottea 2020, 20, 137–143. [Google Scholar] [CrossRef]
- Bass, D.; Tikhonenkov, D.V.; Foster, R.; Dyal, P.; Janouškovec, J.; Keeling, P.J.; Gardner, M.; Neuhauser, S.; Hartikainen, H.; Mylnikov, A.P.; et al. Rhizarian ‘Novel Clade 10’ Revealed as Abundant and Diverse Planktonic and Terrestrial Flagellates, including Aquavolon n. gen. J. Eukaryot. Microbiol. 2018, 65, 828–842. [Google Scholar] [CrossRef]
- Irwin, N.A.T.; Tikhonenkov, D.V.; Hehenberger, E.; Mylnikov, A.P.; Burki, F.; Keeling, P.J. Phylogenomics Supports the Monophyly of the Cercozoa. Mol. Phylogenet. Evol. 2019, 130, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Prokina, K.I.; Radaykina, L.V.; Mylnikov, A.P. Centrohelid heliozoans (Centroplasthelida Febvre-Chevalier et Febvre 1984) from Vietnam. Zool. Zhurnal 2019, 98, 965–977. [Google Scholar] [CrossRef]
- Tran, Q.H.; Mazei, A.Y. Testate Amoebae from South Vietnam Waterbodies with the Description of New Species Difflugia vietnamica sp. nov. Acta Protozool. 2018, 57, 215–230. [Google Scholar] [CrossRef]
- Del Campo, J.; Massana, R. Emerging Diversity within Chrysophytes, Choanoflagellates and Bicosoecids Based on Molecular Surveys. Protist 2011, 162, 435–448. [Google Scholar] [CrossRef]
- Škaloud, P.; Kristiansen, J.; Škaloudová, M. Developments in The Taxonomy of Silica-Scaled Chrysophytes—From Morphological and Ultrastructural to Molecular Approaches. Nord. J. Bot. 2013, 31, 385–402. [Google Scholar] [CrossRef]
- Kristiansen, J.; Škaloud, P. Chrysophyta. In Handbook of the Protists, 2nd ed.; Archibald, J.M., Simpson, A.G.B., Slamovits, C.H., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 331–366. [Google Scholar] [CrossRef]
- Škaloudová, M.; Škaloud, P. A New Species of Chrysosphaerella (Chrysophyceae: Chromulinales), Chrysosphaerella rotundata, sp. nov., from Finland. Phytotaxa 2013, 130, 34–42. [Google Scholar] [CrossRef]
- Scoble, J.M.; Cavalier-Smith, T. Scale Evolution in Paraphysomonadida (Chrysophyceae): Sequence Phylogeny and Revised Taxonomy of Paraphysomonas, New Genus Clathromonas, and 25 New Species. Eur. J. Protistol. 2014, 50, 551–592. [Google Scholar] [CrossRef] [PubMed]
- Siver, P.A.; Jo, B.Y.; Kim, J.I.; Shin, W.; Lott, A.M.; Wolfe, A.P. Assessing the Evolutionary History of The Class Synurophyceae (Heterokonta) Using Molecular, Morphometric, and Paleobiological Approaches. Am. J. Bot. 2015, 102, 921–941. [Google Scholar] [CrossRef] [PubMed]
- Čertnerová, D.; Čertner, M.; Škaloud, P. Molecular Phylogeny and Evolution of Phenotype in Silica-Scaled Chrysophyte Genus Mallomonas. J. Phycol. 2019, 55, 912–923. [Google Scholar] [CrossRef]
- Škaloud, P.; Škaloudová, M.; Jadrná, I.; Bestová, H.; Pusztai, M.; Kapustin, D.; Siver, P.A. Comparing Morphological and Molecular Estimates of Species Diversity in the Freshwater Genus Synura (Stramenopiles): A Model for Understanding Diversity of Eukaryotic Microorganisms. J. Phycol. 2020, 56, 574–591. [Google Scholar] [CrossRef] [PubMed]
- Manton, I. Observations with the Electron Microscope on Synura caroliniana Whitford. Proc. Leeds Phil. Soc. 1955, 6, 306–316. [Google Scholar]
- Petersen, J.B.; Hansen, J.B. On the Scales of Some Synura Species; Biologiske Meddelelser udgivet af Det Kongelige Danske Videnskabernes Selskab: Copenhagen, Denmark, 1956; Volume 23, pp. 1–27. [Google Scholar]
- Fott, B.; Ludvík, J. Die submikroskopische Struktur der Kieselschuppen bei Synura und ihre Bedeutung für die Taxonomie der Gattung. Preslia 1957, 29, 5–16. [Google Scholar]
- Asmund, B. Electron Microscope Observations on Mallomonas caudata and Some Remarks on Its Occurrence in Four Danish Ponds. Bot. Tidsskr. 1955, 52, 163–168. [Google Scholar]
- Fott, B. Scales of Mallomonas Observed in the Electron Microscope. Preslia 1955, 27, 280–282. [Google Scholar]
- Neustupa, J.; Řezáčová, M. The genus Mallomonas (Mallomonadales, Synurophyceae) in Several Southeast Asian Urban Water Bodies—The Biogeographical Implications. Nova Hedwig. 2007, 84, 249–259. [Google Scholar] [CrossRef]
- Řezáčová, M.; Neustupa, J. Distribution of the Genus Mallomonas (Synurophyceae)—Ubiquitous Dispersal in Microorganisms Evaluated. Protist 2007, 158, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Kristiansen, J. Cosmopolitan chrysophytes. Syst. Geog. Plants 2000, 70, 291–300. [Google Scholar] [CrossRef]
- Kristiansen, J. Dispersal and Biogeography of Silica-Scaled Chrysophytes. Biodivers. Conserv. 2008, 17, 419–426. [Google Scholar] [CrossRef]
- Siver, P.A. The distribution of chrysophytes along environmental gradients: Their use as biological indicators. In Chrysophyte Algae: Ecology, Phylogeny and Development; Sandgren, C., Smol, J., Kristiansen, J., Eds.; Cambridge University Press: Cambridge, UK, 1995; pp. 232–268. [Google Scholar] [CrossRef]
- Smol, J.P. Applications of chrysophytes to problems in paleoecology. In Chrysophyte Algae: Ecology, Phylogeny and Development; Sandgren, C., Smol, J., Kristiansen, J., Eds.; Cambridge University Press: Cambridge, UK, 1995; pp. 303–330. [Google Scholar] [CrossRef]
- Kristiansen, J.; Preisig, H.R. Chrysophyta and Haptophyta Algae, 2nd part: Synurophyceae. In Süsswasserflora Von Mitteleuropa (Freshwater Flora of Central Europe); Büdel, B., Gärtner, G., Krienitz, L., Preisig, H.R., Schagerl, M., Eds.; Spektrum Akademisher Verlag, Springer: Berlin/Heidelberg, Germany, 2007; pp. 1–252. ISBN 978-3-8274-1701-5. [Google Scholar]
- Škaloud, P.; Škaloudová, M.; Pichrtová, M.; Němcová, Y.; Kreidlová, J.; Pusztai, M. www.chrysophytes.eu—A Database on Distribution and Ecology of Silica-Scaled Chrysophytes in Europe. Nova Hedwigia Beiheft. 2013, 142, 141–146. [Google Scholar]
- Siver, P.A. The biology of Mallomonas: Morphology, taxonomy and ecology. In Developments in Hydrobiology; Dumont, H.J., Ed.; Springer: Dordrecht, The Netherlands, 1991; Volume 63, p. 230. [Google Scholar] [CrossRef]
- Siver, P.A. Synurophyte Algae. In Freshwater Algae of North America: Ecology and Classification, 2nd ed.; Wehr, J.D., Sheath, R.G., Kociolek, J.P., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 607–651. [Google Scholar]
- Siver, P.A.; Lott, A.M. Biogeographic Patterns in Scaled Chrysophytes from the East Coast of North America. Freshw. Biol. 2012, 57, 451–466. [Google Scholar] [CrossRef]
- Siver, P.A.; Lott, A.M. The Scaled Chrysophyte Flora in Freshwater Ponds and Lakes from Newfoundland, Canada, and Their Relationship to Environmental Variables. Cryptogam. Algol. 2017, 38, 325–347. [Google Scholar] [CrossRef]
- Nicholls, K.H.; Wujek, D.E. Chrysophycean Algae. In Freshwater Algae of North America: Ecology and Classification, 2nd ed.; Wehr, J.D., Sheath, R.G., Kociolek, J.P., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 471–500. [Google Scholar]
- Cronberg, G. Scaled Chrysophytes from the Tropics. Nova Hedwig. Beih. 1989, 95, 191–232. [Google Scholar]
- Takahashi, E.; Hayakawa, T. The Synuraceae (Chrysophyceae) in Bangladesh. Phykos 1979, 18, 129–147. [Google Scholar]
- Saha, L.C.; Wujek, D.E. Scale-bearing Chrysophytes from Tropical Northeast India. Nord. J. Bot. 1990, 10, 343–355. [Google Scholar] [CrossRef]
- Wujek, D.E.; Saha, L.C. Scale-Bearing Chrysophytes (Chrysophyceae and Synurophyceae) from India. II. Nova Hedwig. 1996, 112, 367–377. [Google Scholar]
- Dürrschmidt, M.; Croome, R. Mallomonadaceae (Chrysophyceae) from Malaysia and Australia. Nord. J. Bot. 1985, 5, 285–298. [Google Scholar] [CrossRef]
- Dürrschmidt, M.; Cronberg, G. Contribution to The Knowledge of Tropical Chrysophytes: Mallomonadaceae and Paraphysomonadaceae from Sri Lanka. Algol. Stud. 1989, 54, 15–37. [Google Scholar]
- Vyverman, W.; Cronberg, G. Scale Bearing Chrysophytes from Papua New Guinea. Nord. J. Bot. 1993, 13, 111–120. [Google Scholar] [CrossRef]
- Croome, R.L.; Tyler, P.A. Mallomonas morrisonensis (Chrysophyceae) a New Species from Australia. Br. Phycol. J. 1983, 18, 383–389. [Google Scholar] [CrossRef]
- Croome, R.L.; Tyler, P.A. Mallomonas sabulosa (Chrysophyceae), a New Species from Australia. Br. Phycol. J. 1986, 21, 93–96. [Google Scholar] [CrossRef]
- Croome, R.; Diirrschmidt, M.; Tyler, P.A. Light and Electron Microscopical Investigation of Mallomonas splendens (G.S. West) Playfair (Mallomonadaceae, Chrysophyceae). Nova Hedwig. 1985, 41, 463–470. [Google Scholar]
- Croome, R.; Kristiansen, J.; Qler, P.A. A Description of Mallomonas marsupialis nov. sp. (Synurophyceae), a New Chrysophyte from Australia, with Comments on the Endemicity of Australian Freshwater Algae. Nord. J. Bot. 1998, 18, 633–639. [Google Scholar] [CrossRef]
- Cronberg, G.; Hickel, B. Mallomonas fenestrata sp. nov. and M. perforata sp. nov. (Chrysophyceae, Mallomonadaceae) from Tropical Lakes. Nord. J. Bot. 1985, 5, 105–110. [Google Scholar] [CrossRef]
- Kapustin, D.A.; Gusev, E.S. Silica-Scaled Chrysophytes from West Java (Indonesia) Including Description of a New Chrysosphaerella Species. Nova Hedwig. Beih. 2019, 148, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Kapustin, D.A.; Gusev, E.S.; Kulikovskiy, M.S. Mallomonas papuensis sp. nov. (Chrysophyceae, Synurales), a New Species from The High Mountain Bog Pool in Papua Province, Indonesia. Phytotaxa 2019, 402, 281–287. [Google Scholar] [CrossRef]
- Wei, Y.X.; Yuan, X.P. Studies on Silica-Scaled Chrysophytes from the Tropics and Subtropics of China. Nova Hedwig. Beih. 2001, 122, 169–187. [Google Scholar]
- Wei, Y.X.; Yuan, X.P.; Kristiansen, J. Silica-Scaled Chrysophytes from Hainan, Guangdong Provinces and Hong Kong Special Administrative Region, China. Nord. J. Bot. 2014, 32, 881–896. [Google Scholar] [CrossRef]
- Gusev, E.S.; Nguyen, T.H.T. Silica-Scaled Chrysophytes (Chrysophyceae and Synurophyceae) from Vietnam (Khanh Hoa and Quang Nam provinces). Nova Hedwig. 2011, 93, 191–199. [Google Scholar] [CrossRef]
- Gusev, E.S. Studies on Synurophycean Algae from Mangrove Wetlands (Vietnam). Nova Hedwig. Beih. 2013, 142, 87–95. [Google Scholar]
- Gusev, E.S.; Doan-Nhu, H.; Nguyen-Ngoc, L.; Guseva, E.E.; Phan Tan, L. Silica-Scaled Chrysophytes from Cam Ranh Peninsula (Khanh Hoa Province, Vietnam). Nova Hedwig. Beih. 2019, 148, 63–76. [Google Scholar] [CrossRef]
- Gusev, E.S.; Doan-Nhu, H.; Nguyen-Ngoc, L. Silica-Scaled Chrysophytes from Cat Tien National Park (Dong Nai Province, Vietnam). Nova Hedwig. 2017, 105, 347–364. [Google Scholar] [CrossRef]
- Gusev, E.S.; Gusakov, V.A.; Guseva, E.E.; Kulikovskiy, M.S.; Tsvetkov, A.I.; Định, C.N. Flora of Silica-Scaled Chrysophytes (Chrysophyceae: Synurales, Paraphysomonadales) of the Mekong Delta. Inland Water Biol. 2020, 13, 349–357. [Google Scholar] [CrossRef]
- Doan-Nhu, H.; Thi-Tinh, T.; Gusev, E.; Kulikovskiy, M.; Phan-Tan, L.; Nguyen-Ngoc, L. Taxonomic Composition of Silica-Scaled Chrysophytes in a Tropical Mountain Reservoir. Inland Water Biol. 2021, 14, 490–499. [Google Scholar] [CrossRef]
- Gusev, E.; Kezlya, E.; Tran, H.; Kulikovskiy, M. Mallomonas vietnamica sp. nov. (Synurales, Chrysophyceae), a New Species, That Shares Some Features with Fossil Taxa. Cryptogam. Algol. 2021, 42, 39–46. [Google Scholar] [CrossRef]
- Gusev, E.; Shkurina, N.; Kulikovskiy, M. Mallomonas loricata sp. nov. (Synurales, Chrysophyceae), a New Tropical Species from Section Plantae. Phytotaxa 2021, 500, 225–233. [Google Scholar] [CrossRef]
- Gusev, E.; Kulikovskiy, M. Mallomonas siderea sp. nov. (Synurales, Chrysophyceae), a New Tropical Species from the Section Torquatae. Nova Hedwig. 2021, 113, 291–301. [Google Scholar] [CrossRef]
- Gusev, E.S. A New Species of the Genus Mallomonas (Synurophyceae), Mallomonas spinosa sp. nov., from Vietnam. Phytotaxa 2012, 66, 1–5. [Google Scholar] [CrossRef]
- Gusev, E.S. A New Species in Genus Mallomonas Perty (Synurales, Chrysophyceae) from Vietnam. Int. J. Algae 2015, 17, 351–362. [Google Scholar] [CrossRef]
- Gusev, E.S. A New Species of the Genus Mallomonas (Synurales, Chrysophyceae), Mallomonas fimbriata sp. nov. Phytotaxa 2015, 195, 291–296. [Google Scholar] [CrossRef]
- Gusev, E.; Siver, P.A. Mallomonas neoampla sp. nov. from Vietnam, a New Species That Bridges the Gap Between Fossil and Modern Taxa. Nova Hedwig. 2017, 104, 521–528. [Google Scholar] [CrossRef]
- Gusev, E.S.; Kulikovskiy, M.S. Two New Species of Genus Mallomonas from Swamp Localities in Vietnam. Phytotaxa 2020, 468, 121–129. [Google Scholar] [CrossRef]
- Gusev, E.S.; Doan-Nhu, H.; Nguyen-Ngoc, L. Mallomonas cattiensis sp. nov. (Synurales, Chrysophyceae), a New Species from Viet Nam. Phytotaxa 2015, 221, 188–192. [Google Scholar] [CrossRef]
- Gusev, E.S.; Doan-Nhu, H.; Nguyen-Ngoc, L.; Kapustin, D.A. Two New Species of the Genus Mallomonas from the Cat Tien National Park (Viet Nam): Mallomonas distinguenda and Mallomonas skvortsovii. Phytotaxa 2016, 273, 59–64. [Google Scholar] [CrossRef]
- Gusev, E.S.; Siver, P.A.; Shin, W. Mallomonas bronchartiana Compère Revisited: Two New Species Described from Asia. Cryptogam. Algol. 2017, 38, 3–16. [Google Scholar] [CrossRef]
- Gusev, E.S.; Čertnerová, D.; Škaloudová, M.; Škaloud, P. Exploring Cryptic Diversity and Distribution Patterns in the Mallomonas kalinae/rasilis Species Complex with a Description of a New Taxon—Mallomonas furtiva sp. nov. J. Eukaryot. Microbiol. 2018, 65, 38–47. [Google Scholar] [CrossRef]
- Gusev, E.; Guseva, E.; Kezlya, E.; Kulikovskiy, M. Mallomonas minuscula sp. nov. (Synurales, Chrysophyceae), a New Member in the Section Torquatae from Vietnam. Fottea 2019, 19, 132–137. [Google Scholar] [CrossRef]
- Gusev, E.S.; Kapustin, D.A.; Martynenko, N.A.; Guseva, E.E.; Kulikovskiy, M.S. Mallomonas gusakovii sp. nov. (Chrysophyceae, Synurales), a New Species from Phu Quoc Island, Vietnam. Phytotaxa 2019, 406, 199–205. [Google Scholar] [CrossRef]
- Gusev, E.; Kulizin, P.; Guseva, E.; Shkurina, N.; Kulikovskiy, M. Mallomonas lamii sp. nov. (Synurales, Chrysophyceae), a New Species Bearing Large Scales Described from the Tropics. Phytotaxa 2019, 423, 266–272. [Google Scholar] [CrossRef]
- Siver, P.A.; Kapustin, D.; Gusev, E. Investigations of Two-Celled Colonies of Synura Formerly Described as Chrysodidymus with Descriptions of Two New Species. Eur. J. Phycol. 2018, 53, 245–255. [Google Scholar] [CrossRef]
- Schmidt-Thomé, P.; Nguyen, H.; Pham, L.; Jarva, J.; Nuottimäki, K. Climate Change Adaptation Measures in Vietnam; Springer International Publishing: Cham, Switzerland, 2015; p. 95. [Google Scholar] [CrossRef]
- Clescerl, L.S.; Greenberg, A.E.; Eaton, A.D. Standard Methods for The Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1999; pp. 1–1325. [Google Scholar]
- Rowan, K.S. Photosynthetic Pigments of Algae; Cambridge University Press: Cambridge, UK, 1989; pp. 1–334. ISBN 9780521105941. [Google Scholar]
- Nürnberg, G.K.; Shaw, M. Productivity of clear and humic lakes: Nutrients, phytoplankton, bacteria. Hydrobiologia 1999, 382, 97–112. [Google Scholar] [CrossRef]
- Ngo, V.S.; Le, L.T. Status of reservoir Fisheries in Vietnam. In Reservoir and Culture-Based Fisheries: Biology and Management; De Silva, S.S., Ed.; ACIAR Proceeding: Canberra, Australia, 2001; Volume 98, pp. 29–35. [Google Scholar]
- Gusev, E.; Martynenko, N.; Tran, H. Studies on Algae from the Order Synurales (Chrysophyceae) in Northern Vietnam. Diversity 2021, 13, 602. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M.; AlgaeBase. World-Wide Electronic Publication, National University of Ireland, Galway. Available online: https://www.algaebase.org (accessed on 5 October 2021).
- Gusev, E.S.; Kapustin, D.A.; Martynenko, N.A.; Kulikovskiy, M.S. Diversity and Biogeography of Silica-Scaled Chrysophytes (Stramenopiles: Chrysophyceae) from Indonesian Papua. Cryptogam. Algol. 2022, in press. [Google Scholar]
- Hansen, P. Silica-scaled Chrysophyceae and Synurophyceae from Madagascar. Arch. Protistenkd. 1996, 147, 145–172. [Google Scholar] [CrossRef]
- Franceschini, I.M.; Kristiansen, J.M. New records of scale-bearing chrysophytes for Florianópolis, Santa Catarina Island, Southern Brazil. Algol. Stud. Arch. Hydrobiol. 2004, 111, 63–77. [Google Scholar] [CrossRef]
- Wujek, D.E.; Dziedic, R.M. Silica-scaled Chrysophytes from Ecuador. Gayana Bot. 2005, 62, 1–8. [Google Scholar] [CrossRef][Green Version]
- Asmund, B.; Kristiansen, J. The Genus Mallomonas (Chrysophyceae). A Taxonomic Survey Based on The Ultrastructure of Silica Scales and Bristles. Opera Bot. 1986, 85, 1–128. [Google Scholar]
- Couté, A.; FranceschinI, I.M. Scale-Bearing Chrysophytes from Acid Water of Florianopolis, Santa Catarina Island, South Brazil. Algol. Stud. 1998, 88, 37–61. [Google Scholar] [CrossRef]
- Jo, B.Y.; Shin, W.; Boo, S.M.; Kim, H.S.; Siver, P.A. Studies on Ultrastructure and Three-Gene Phylogeny of the Genus Mallomonas (Synurophyceae). J. Phycol. 2011, 47, 415–425. [Google Scholar] [CrossRef]
- Takahashi, E. Studies on Genera Mallomonas and Synura, and Other Plankton in Fresh Water with the Electron Microscope. IX. Mallomonas harrisae sp. nov. (Chrysophyceae). Phycologia 1975, 14, 41–44. [Google Scholar] [CrossRef]
- Wei, Y.-X.; Yuan, X.-P. Studies on Silica-Scaled Chrysophytes from Zhejiang, Jiangsu and Jiangxi Provinces, China. Beih. Nova Hedw. 2013, 142, 163–179. [Google Scholar]
- Dürrschmidt, M. A Taxonomic Study of the Mallomonas mangofera Group (Mallomonadaceae, Chrysophyceae), Including the Description of four New Taxa. Plant Syst. Evol. 1983, 143, 175–196. [Google Scholar] [CrossRef]
- Hansen, A.F.; Johansen, J.E.; Skovgard, A. Floristik undersøgelse af skælbærende gulalger i Dyrehaven. Urt 1993, 17, 13–18. [Google Scholar]
- Hällfors, G.; Hällfors, S. Records of Chrysophytes with Siliceous Scales (Mallomonadaceae and Paraphysomonadaceae) from Finnish Inland Waters. In Flagellates in Freshwater Ecosystems; Jones, R., Ilmavirta, V., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands; Boston, MA, USA; London, UK, 1988; pp. 1–29. [Google Scholar]
- Škaloud, P.; Kynčlová, A.; Benada, O.; Kofroňová, O.; Škaloudová, M. Toward a Revision of the Genus Synura, Section Petersenianae (Synurophyceae, Heterokontophyta): Morphological Characterization of Six Pseudo-Cryptic Species. Phycologia 2012, 51, 303–329. [Google Scholar] [CrossRef]
- Škaloud, P.; Škaloudová, M.; Procházková, A.; Němcová, Y. Morphological Delineation and Distribution Patterns of Four Newly Described Species within the Synura petersenii Species Complex (Chrysophyceae, Stramenopiles). Eur. J. Phycol. 2014, 49, 213–229. [Google Scholar] [CrossRef]
- Jo, B.Y.; Shin, W.; Kim, H.S.; Siver, P.A.; Andersen, R.A. Phylogeny of the Genus Mallomonas (Synurophyceae) and Descriptions of Five New Species on the Basis of Morphological Evidence. Phycologia 2013, 52, 266–278. [Google Scholar] [CrossRef]
- Jo, B.Y.; Kim, J.I.; Škaloud, P.; Siver, P.A.; Shin, W. Multigene Phylogeny of Synura (Synurophyceae) and Descriptions of Four New Species Based on Morphological and DNA Evidence. Eur. J. Phycol. 2016, 51, 413–430. [Google Scholar] [CrossRef]
- Nemcova, Y.; Rott, E. Diversity of Silica-Scaled Chrysophytes in High-Altitude Alpine Sites (North Tyrol, Austria) Including a Description of Mallomonas pechlaneri sp. nov. Cryptogam. Algol. 2018, 39, 63–83. [Google Scholar] [CrossRef]
- Gusev, E.; Kezlya, E. Mallomonas lusca sp. nov—A Rare Species from Southeast Asia. Phytotaxa 2021, 529, 105–112. [Google Scholar] [CrossRef]
- Dürrschmidt, M. New Species of the Genus Mallomonas (Mallomonadaceae, Chrysophyceae) from New Zealand. In Chrysophytes. Aspects and Problems; Kristiansen, J., Andersen, R.A., Eds.; Cambridge University Press: Cambridge, UK, 1986; pp. 87–106. [Google Scholar]
- Kim, J.; Hee Park, Y.; Jung Kim, H. Silica-Scaled Chrysophytes (Synurophyceae) from Jeju Island, Korea. Nova Hedwig. 2009, 89, 201–218. [Google Scholar] [CrossRef]
- Asmund, B.; Hilliard, D.K. Studies on Chrysophyceae from Some Ponds and Lakes in Alaska. I. Mallomonas species examined with the electron microscope. Hydrobiologia 1961, 17, 237–258. [Google Scholar] [CrossRef]
- Dürrschmidt, M. Mallomonas cristata sp. nov. (Chrysophyceae, Synuraceae) from South Chilean Inland Waters. Phycologia 1981, 20, 298–302. [Google Scholar] [CrossRef]
- Harris, K.; Bradley, D.E. Some Unusual Chrysophyceae Studied in the Electron Microscope. Microbiology 1958, 18, 71–83. [Google Scholar] [CrossRef]
- Siver, P.A. Two New Species of Mallomonas (Synurophyceae) from the Ocala National Forest, Florida, U.S.A. Nord. J. Bot. 2002, 22, 115–121. [Google Scholar] [CrossRef]
- Siver, P.A. Lott, A.M. The Scaled Chrysophyte Flora from the Pinelands National Preserve of Southern New Jersey, U.S.A. Nova Hedwig. Beih. 2010, 136, 167–180. [Google Scholar] [CrossRef]
- Gusev, E.S.; Guseva, E.E.; Dien, T.D.; Kulikovskiy, M.S. Flora of Silica-Scaled Chrysophytes (Chrysophyceae) of Two Provinces in Southern Viet Nam. Inland Water Biol. 2022, 15. in press. [Google Scholar]
- Wujek, D.E.; Kadiri, M.O.; Dziedzic, R.M. Silica-Scaled Chrysophyceae and Synurophyceae from Nigeria. III. Chrysophytes from Rivers of Edo State. Fottea 2010, 10, 93–98. [Google Scholar] [CrossRef]
- Kristiansen, J.; Tong, D. Chrysosphaerella annulata n. sp., a New Scale-Bearing Chrysophyte. Nord. J. Bot. 1989, 9, 329–333. [Google Scholar] [CrossRef]
- Finlay, B.J. Global Dispersal of Free-Living Microbial Eukaryote Species. Science 2002, 296, 1061–1063. [Google Scholar] [CrossRef]
- Finlay, B.J.; Fenchel, T. Cosmopolitan Metapopulations of Free-Living Microbial Eukaryotes. Protist 2004, 155, 237–244. [Google Scholar] [CrossRef]
- Finlay, B.J.; Monaghan, E.B.; Maberly, S.C. Hypothesis: The Rate and Scale of Dispersal of Freshwater Diatom Species Is a Function of Their Global Abundance. Protist 2002, 153, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Foissner, W. Biogeography and Dispersal of Micro-Organisms: A Review Emphasizing Protists. Acta Protozool. 2006, 45, 111–136. [Google Scholar]
- Kristiansen, J.; Lind, J.F. Endemicity in Silica-Scaled Chrysophytes. Nova Hedwig. Beih. 2005, 128, 65–83. [Google Scholar]



| Taxon | Provinces | |||||||
|---|---|---|---|---|---|---|---|---|
| Lam Dong | Dak Lak | Khanh Hoa | Phu Yen | Binh Dinh | Thua Thien Hue | Quang Tri | Quang Binh | |
| Mallomonas acaroides Perty emend. Iwanoff | + | |||||||
| M. akrokomos Ruttner | + | + | + | |||||
| M. adamas Harris & Bradley | + | + | + | + | + | |||
| M. bangladeshica (E. Takahashi & T. Hayakawa) Siver & A.P. Wolfe | + | + | + | + | ||||
| M. binocularis Siver | + | + | ||||||
| M. bronchartiana Compère | + | + | + | + | + | |||
| M. cattiensis Gusev, Doan-Nhu & Nguyen-Ngoc | + | + | + | + | + | |||
| M. caudata Iwanoff | + | + | + | + | + | + | + | + |
| M. ceylanica Dürrschmidt & Cronberg | + | + | + | + | + | + | ||
| M. crassisquama (Asmund) Fott | + | + | + | + | + | + | + | + |
| M. crassisquama var. papillosa Siver & Skogstad | + | + | ||||||
| M. cf. cristata Dürrschmidt | + | + | + | + | ||||
| M. distinguenda Gusev, Doan-Nhu, Nguyen-Ngoc & Kapustin | + | |||||||
| M. elevata Han Soon Kim | + | + | + | + | + | |||
| M. elongata Reverdin | + | + | ||||||
| M. favosa f. favosa K.H. Nicholls | + | + | + | + | + | + | ||
| M. favosa f. gemina Dürrschmidt & Croome | + | + | + | + | + | + | ||
| M. fimbriata Gusev | + | |||||||
| M. furtiva Gusev, Certnerová, Škaloudová & Škaloud | + | + | + | + | + | |||
| M. harrisiae E. Takahashi | + | |||||||
| M. hexareticulata Jo, Shin, Kim, Siver & Andersen | + | + | + | + | + | |||
| M. grata E. Takahashi | + | + | + | + | ||||
| M. guttata var. guttata Wujek | + | + | + | + | + | + | + | + |
| M. guttata var. simplex Nicholls | + | + | + | + | + | + | + | |
| M. kenyana (Wujek & Asmund) Kapustin & Gusev | + | + | + | + | + | + | + | + |
| M. korshikovii Gusev | + | + | + | |||||
| M. lamii Gusev, Kulizin, Guseva, Shkurina & Kulikovskiy | + | + | + | + | + | + | ||
| M. loricata Gusev, Shkurina & Kulikovskiy | + | + | + | + | + | + | ||
| M. lusca Gusev & Kezlya | + | |||||||
| M. mangofera Harris & Bradley var. mangofera apud Dürrschmidt (1983) | + | + | + | + | ||||
| M. mangofera var. foveata (Dürrschmidt) Kristiansen | + | + | + | + | + | + | + | |
| M. cf. mangofera var. reticulata (Cronberg) Kristiansen | + | + | + | + | + | + | ||
| M. mangofera var. sulcata Dürrschmidt | + | + | + | + | ||||
| M. mangofera var. gracilis (Dürrschmidt) Kristiansen | + | |||||||
| M. minuscula Gusev, Guseva, Kezlya & Kulikovskiy | + | + | + | + | + | |||
| M. morrisonensis Croome & P.A. Tyler | + | + | + | + | + | + | + | |
| M. multisetigera Dürrschmidt | + | + | + | |||||
| M. neoampla Gusev & Siver | + | + | ||||||
| M. ocellata Dürrschmidt & Croome | + | + | + | + | + | + | + | |
| Mallomonas cf. ouradion Harris & Bradley | + | + | ||||||
| M. papillosa Harris & Bradley | + | |||||||
| M. paragrandis Gusev | + | + | + | + | + | + | ||
| M. peronoides (K. Harris) Momeu & Péterfi | + | + | + | + | + | + | ||
| M. plumosa Croome & Tyler | + | + | + | + | + | + | ||
| M. portae-ferreae Péterfi & Asmund | + | + | + | |||||
| M. pseudobronchartiana Gusev, Siver & Shin | + | |||||||
| M. pseudomatvienkoae Jo, Shin, Kim, Siver & Andersen | + | + | + | + | + | + | + | |
| M. pseudocratis Dürrschmidt | + | |||||||
| M. punctifera Korshikov | + | |||||||
| M. punctostriata Gusev & Kulikovskiy | + | |||||||
| M. rasilis Dürrschmidt | + | + | + | + | + | + | ||
| M. siderea Gusev & Kulikovskiy | + | + | + | + | + | |||
| M. cf. skvortsovii Gusev, Doan-Nhu, Nguyen-Ngoc & Kapustin | + | + | ||||||
| M. sorohexareticulata Jo, Shin, Kim, Siver & Andersen | + | + | + | + | + | + | + | |
| M. spinosa Gusev emend. Wei & Kristiansen | + | + | + | + | + | + | ||
| M. splendens (G.S. West) Playfair | + | + | + | + | + | |||
| M. striata var. serrata Harris & Bradley | + | + | ||||||
| M. tonsurata Teiling | + | + | + | + | + | + | + | + |
| M. tropica Dürrschmidt & Croome | + | + | + | |||||
| Mallomonas sp. 1 | + | + | + | + | + | + | + | |
| Mallomonas sp. 2 | + | + | + | + | + | + | + | |
| Mallomonas sp. 3 | + | |||||||
| Mallomonas sp. 4 | + | + | + | + | ||||
| Mallomonas sp. 5 | + | + | + | + | + | |||
| Mallomonas sp. 6 | + | |||||||
| Mallomonas sp. 7 | + | |||||||
| Synura australiensis Playfair | + | + | + | + | ||||
| S. echinulata Korshikov | + | + | + | + | + | |||
| S. mammillosa E. Takahashi | + | + | + | + | + | |||
| S. papillosa Kapustin, Gusev & Siver | + | + | + | + | + | + | ||
| S. sphagnicola (Korshikov) Korshikov | + | + | + | |||||
| Synura sp. 1 (S. longitubularis/ S. curtispina group) | + | + | + | + | + | + | + | + |
| Synura sp. 2 (S. petersenii sensu lato group) | + | + | ||||||
| Spiniferomonas trioralis E. Takahashi | + | + | + | + | + | + | ||
| Chrysosphaerella annulata Kristiansen & Tong | + | + | + | |||||
| Ch. nichollsii Kapustin & Gusev | + | + | + | + | + | + | + | + |
| Total | 36 | 41 | 53 | 47 | 45 | 56 | 31 | 20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gusev, E.; Martynenko, N. Diversity of Silica-Scaled Chrysophytes in Central Vietnam. Water 2022, 14, 65. https://doi.org/10.3390/w14010065
Gusev E, Martynenko N. Diversity of Silica-Scaled Chrysophytes in Central Vietnam. Water. 2022; 14(1):65. https://doi.org/10.3390/w14010065
Chicago/Turabian StyleGusev, Evgeniy, and Nikita Martynenko. 2022. "Diversity of Silica-Scaled Chrysophytes in Central Vietnam" Water 14, no. 1: 65. https://doi.org/10.3390/w14010065
APA StyleGusev, E., & Martynenko, N. (2022). Diversity of Silica-Scaled Chrysophytes in Central Vietnam. Water, 14(1), 65. https://doi.org/10.3390/w14010065






