Relationship between Phase Composition and Mechanical Properties of Peat Soils Stabilized Using Oil Shale Ash and Pozzolanic Additive
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Composition of Oil Shale Ashes
3.2. Stress–Strain Tests
3.3. XRD-XRF Analyses of Crystalline Phases
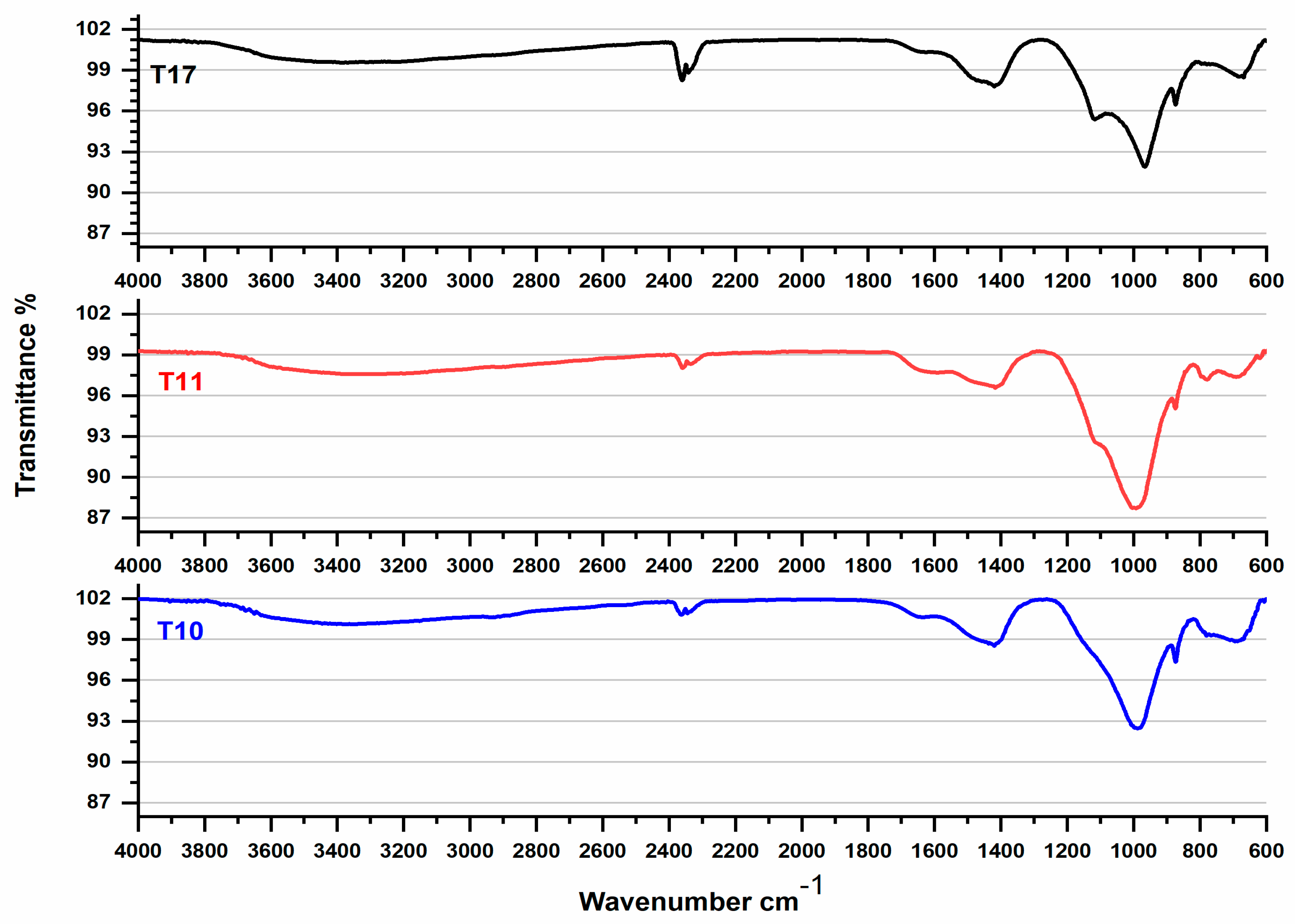
3.4. FTIR Spectra Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liiv, J.; Zekker, I.; Tämm, K.; Rikmann, E. Greenhouse gases emissions and climate change—Beyond mainstream. Moj Bioorg. Org. Chem. 2020, 4, 10–16. [Google Scholar] [CrossRef]
- Orru, M.; Orru, H. Sustainable use of Estonian peat reserves and environmental challenges. Est. J. Earth Sci. 2008, 57, 87. [Google Scholar] [CrossRef]
- Axelsson, K.; Johansson, S.-E.; Andersson, R.; Djupstabilisering, S. Stabilization of Organic Soils by Cement and Puzzolanic Reactions-FEASIBILITY STUDY English Translation. 2002. Available online: http://www.swedgeo.se/sd.htm (accessed on 12 January 2020).
- Vasander, H.; Tuittila, E.-S.; Lode, E.; Lundin, L.; Ilomets, M.; Sallantaus, T.; Heikkilä, R.; Pitkänen, M.-L.; Laine, J. Status and restoration of peatlands in northern Europe. Wetl. Ecol. Manag. 2003, 11, 51–63. [Google Scholar] [CrossRef]
- Gorham, E. Northern Peatlands: Role in the Carbon Cycle and Probable Responses to Climatic Warming. Ecol. Appl. 1991, 1, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Limpens, J.; Berendse, F.; Blodau, C.; Canadell, J.G. Peatlands and the carbon cycle: From local processes to global implications—A synthesis. Biogeosciences 2008, 5, 1475–1491. [Google Scholar] [CrossRef]
- Zulkifley, M.T.M.; Ng, T.F.; Raj, J.K.; Hashim, R.; Bakar, A.F.A.; Paramanthan, S.; Ashraf, M.A. A review of the stabilization of tropical lowland peats. Bull. Eng. Geol. Environ. 2014, 73, 733–746. [Google Scholar] [CrossRef]
- Munro, R. Dealing with Poor Bearing Capacity on Low Volume Roads on Peat in the Northern Periphery. 2005. Available online: www.peatlandsni.gov.uk (accessed on 4 March 2005).
- Huat, B.B.K.; Kazemian, S.; Kuang, W.L. Effect of cement-sodium silicate grout and kaolinite on undrained shear strength of reinforced peat. Electron. J. Geotech. Eng. 2020, 16, 1221–1228. [Google Scholar]
- Moayedi, H.; Huat, B.B.K.; Kazemian, S.; Daneshmand, S. Stabilization of organic soil using sodium silicate system grout. Int. J. Phys. Sci. 2012, 7, 1395–1402. [Google Scholar] [CrossRef]
- Reinik, J.; Irha, N.; Koroljova, A.; Meriste, T. Use of oil shale ash in road construction: Results of follow-up environmental monitoring. Environ. Monit. Assess. 2018, 190. [Google Scholar] [CrossRef]
- Consoli, N.C.; Carretta, M.d.; Leon, H.B.; Filho, H.C.S.; Tomasi, L.F. Strength and Stiffness of Ground Waste Glass–Carbide Lime Blends. J. Mater. Civ. Eng. 2019, 31, 06019010. [Google Scholar] [CrossRef]
- Wong, L.S. Formulation of an optimal mix design of stabilized peat columns with fly ash as a Pozzolan. Arab. J. Sci. Eng. 2015, 40, 1015–1025. [Google Scholar] [CrossRef]
- Wong, J.K.H.; Kok, S.T.; Wong, S.Y. Cementitious, Pozzolanic And Filler Materials For DSM Binders. Civ. Eng. J. 2020, 6, 402–417. [Google Scholar] [CrossRef]
- Dehghanbanadaki, A.; Arefnia, A.; Keshtkarbanaeemoghadam, A.; Ahmad, K.; Motamedi, S.; Hashim, R. Evaluating the compression index of fibrous peat treated with different binders. Bull. Eng. Geol. Environ. 2017, 76, 575–586. [Google Scholar] [CrossRef]
- Liiv, J.; Teppand, T.; Rikmann, E.; Tenno, T. Novel ecosustainable peat and oil shale ash-based 3D-printable composite material. Sustain. Mater. Technol. 2018, 17, e00067. [Google Scholar] [CrossRef]
- Tremblay, H.; Duchesne, J.; Locat, J.; Leroueil, S. Influence of the nature of organic compounds on fine soil stabilization with cement. Can. Geotech. J. 2002, 39, 535–546. [Google Scholar] [CrossRef]
- Ma, C.; Chen, B.; Chen, L. Effect of organic matter on strength development of self-compacting earth-based construction stabilized with cement-based composites. Constr. Build. Mater. 2016, 123, 414–423. [Google Scholar] [CrossRef]
- Tenno, T.; Rikmann, E.; Zekker, I.; Tenno, T. Modelling the solubility of sparingly soluble compounds depending on their particles size. Proc. Est. Acad. of Sci. 2018, 67, 300–302. [Google Scholar] [CrossRef]
- Zekker, I.; Tenno, T.; Selberg, A.; Uiga, K. Dissolution Modeling and Experimental Measurement of CaS-H2O Binary System. Chin. J. Chem. 2011, 29, 2327–2336. [Google Scholar] [CrossRef]
- Tenno, T.; Rikmann, E.; Uiga, K.; Zekker, I.; Mashirin, A.; Tenno, T. A novel proton transfer model of the closed equilibrium system H2O–CO2–CaCO3–NHX. Proc. Est. Acad. of Sci. 2018, 4017, 2. [Google Scholar] [CrossRef]

| Test Piece | Peat Soil, g | CFB Ash, g | PF Ash, g | Silica Fume SiO2, g | Na2SiO3 20% Solution, g | KOH, g |
|---|---|---|---|---|---|---|
| T10 | 200 | 0 | 100 | 100 | 100 | 0 |
| T11 | 200 | 100 | 0 | 50 | 30 | 30 |
| T17 | 200 | 0 | 200 | 50 | 30 | 30 |
| Mineral | PF Cyclone Ash Mass Concentration (%) | CFB I Filter Ash Mass Concentration (%) |
|---|---|---|
| Quartz, SiO2 | 3.3 | 16.8 |
| Orthoclase, KAlSi3O8 | 1.7 | 12.5 |
| Illite+Illite-Smectite, Na,Kx(Al,Mg)2Si4O10(OH)2×H2O | 6.1 | 13.8 |
| Belite, Ca2SiO4 | 15.9 | 5.3 |
| Merwinite, Ca3Mg(SiO4)2 | 13.2 | 3.7 |
| C3A, 3CaO·Al2O3 | 2.2 | 2.3 |
| Pseudowollastonite, CaSiO3 | 1.6 | 3.6 |
| Periclase, MgO | 8.7 | 2.7 |
| Melilite, (Ca,Na)2(Mg,Al)(Si,Al)3O7 | 5.8 | 1.2 |
| Anhydrite, CaSO4 | 5.4 | 9.5 |
| Lime, CaO | 29.3 | 10.8 |
| Calcite, CaCO3 | 2.5 | 13.5 |
| Portlandite, Ca(OH)2 | 3.1 | nd |
| Hematite, Fe2O3 | 1.1 | 4.3 |
| Sample | 1st Parallel, N mm−2 | 2nd Parallel, N mm−2 | 3rd Parallel, N mm−2 | 4th Parallel, N mm−2 | 5th Parallel, N mm−2 |
|---|---|---|---|---|---|
| T10 | 1.62 | 1.55 | 1.60 | 1.65 | 1.58 |
| T11 | 0.99 | 0.95 | 1.00 | 0.99 | 1.02 |
| T17 | 0.20 | 0.17 | 0.16 | 0.15 | 0.17 |
| Specimen | Thickness (mm) | Diameter (mm) | Specific Weight (kN m−3) | Compressive Displacement at Maximum Force (mm) | Maximum Force (kN) | Time at Maximum Force (s) | Compressive Strength (N mm−2) |
|---|---|---|---|---|---|---|---|
| T10 | 41.29 | 97.71 | 132.90 | 16.04 | 12.01 | 481.26 | 1.60 |
| T11 | 34.57 | 97.56 | 142.80 | 16.02 | 7.38 | 480.56 | 0.99 |
| T17 | 49.89 | 95.41 | 145.16 | 16.01 | 1.25 | 480.26 | 0.17 |
| Chemical Element | T10 | T11 | T17 |
|---|---|---|---|
| Al | 1.607 ± 0.046 | 1.252 ± 0.057 | 0.552 ± 0.036 |
| C | 0.808 ± 0.018 | 1.272 ± 0.043 | 0.819 ± 0.020 |
| Ca | 6.912 ± 0.198 | 15.960 ± 0.227 | 19.666 ± 0.110 |
| Cl | 0.108 ± 0.021 | 0.349 ± 0.039 | 0.085 ± 0.026 |
| Fe | 0.319 ± 0.038 | 0.849 ± 0.036 | 0.343 ± 0.048 |
| K | 3.655 ± 0.065 | 4.309 ± 0.128 | 3.296 ± 0.060 |
| Mg | 0.071 ± 0.017 | 1.187 ± 0.055 | 1.153 ± 0.035 |
| Na | 0.340 ± 0.016 | 0.092 ± 0.014 | 0.069 ± 0.012 |
| O | 50.562 ± 0.036 | 48.945 ± 0.083 | 49.409 ± 0.046 |
| S | 0.782 ± 0.010 | 1.089 ± 0.030 | 1.035 ± 0.014 |
| Si | 34.836 ± 0.128 | 24.696 ± 0.072 | 23.572 ± 0.070 |
| Name of Phase | T10 Rwp = 2.9%; Rwp’ = 9.2% | T11 Rwp = 2.4%; Rwp’ = 12.4% | T17 Rwp = 2.7%; Rwp’ = 15.7% |
|---|---|---|---|
| Mass Concentrations, % | |||
| Quartz, SiO2 | 33.6(1) | 10.3(1) | 6.8(1) |
| Calcite, CaCO3 | 4.0(1) | 5.0(1) | 3.7(1) |
| Feldspar, K(AlSi3O8) | 1.4(2) | 1.4(2) | 1.1(1) |
| Orthoclase, K(AlSi3O8) | 2.3(1) | 3.8(2) | 1.1(1) |
| Microcline, (K,Na)(AlSi3O8) | 4.1(2) | 1.3(2) | 0.7(1) |
| Albite, Na(AlSi3O8) | 2.2(1) | 0.5(1) | 0.4(1) |
| Arcanite, K2(SO4) | 2.6(1) | 3.2(1) | 3.3(1) |
| Dolomite, CaMg(CO3)2 | – | 0.7(1) | – |
| Hematite, Fe2O3 | – | 0.7(1) | 0.3(1) |
| Akermanite, Ca2MgSi2O7 | – | 2.2(1) | 0.4(1) |
| Periclase, MgO | – | 0.6(1) | 1.0(1) |
| Belite (larnite), Ca2(SiO4) | – | – | 2.4(1) |
| CSH (calcium silicate hydrate), semi-amorphous | 10(1) | 24(2) | 37(1) |
| Amorphous phase (external standard method) | 40(1) | 46(2) | 42(1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rikmann, E.; Zekker, I.; Teppand, T.; Pallav, V.; Shanskiy, M.; Mäeorg, U.; Tenno, T.; Burlakovs, J.; Liiv, J. Relationship between Phase Composition and Mechanical Properties of Peat Soils Stabilized Using Oil Shale Ash and Pozzolanic Additive. Water 2021, 13, 942. https://doi.org/10.3390/w13070942
Rikmann E, Zekker I, Teppand T, Pallav V, Shanskiy M, Mäeorg U, Tenno T, Burlakovs J, Liiv J. Relationship between Phase Composition and Mechanical Properties of Peat Soils Stabilized Using Oil Shale Ash and Pozzolanic Additive. Water. 2021; 13(7):942. https://doi.org/10.3390/w13070942
Chicago/Turabian StyleRikmann, Ergo, Ivar Zekker, Tõnis Teppand, Vello Pallav, Merrit Shanskiy, Uno Mäeorg, Toomas Tenno, Juris Burlakovs, and Jüri Liiv. 2021. "Relationship between Phase Composition and Mechanical Properties of Peat Soils Stabilized Using Oil Shale Ash and Pozzolanic Additive" Water 13, no. 7: 942. https://doi.org/10.3390/w13070942
APA StyleRikmann, E., Zekker, I., Teppand, T., Pallav, V., Shanskiy, M., Mäeorg, U., Tenno, T., Burlakovs, J., & Liiv, J. (2021). Relationship between Phase Composition and Mechanical Properties of Peat Soils Stabilized Using Oil Shale Ash and Pozzolanic Additive. Water, 13(7), 942. https://doi.org/10.3390/w13070942








