A Biological Method of Treating Surface Water Contaminated with Industrial Waste Leachate
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Material
2.2. Methodology Biotechnological Research
- 1.
- “DBC Plus Type R5” Biopreparation (BioArcus Company, Warsaw, Poland)This biopreparation was used in two forms:
- As granule—lyophilised bacteria;
- As bacteria previously multiplied on nutrient broth (the bacterial population was determined by the culture method and amounted to 82 × 107).
- 2.
- “ACS ODO-1” Biopreparation (ACS Company, Chęciny, Poland).
2.3. Methodolody for Determining Physicochemical and Microbiological Properties of Water
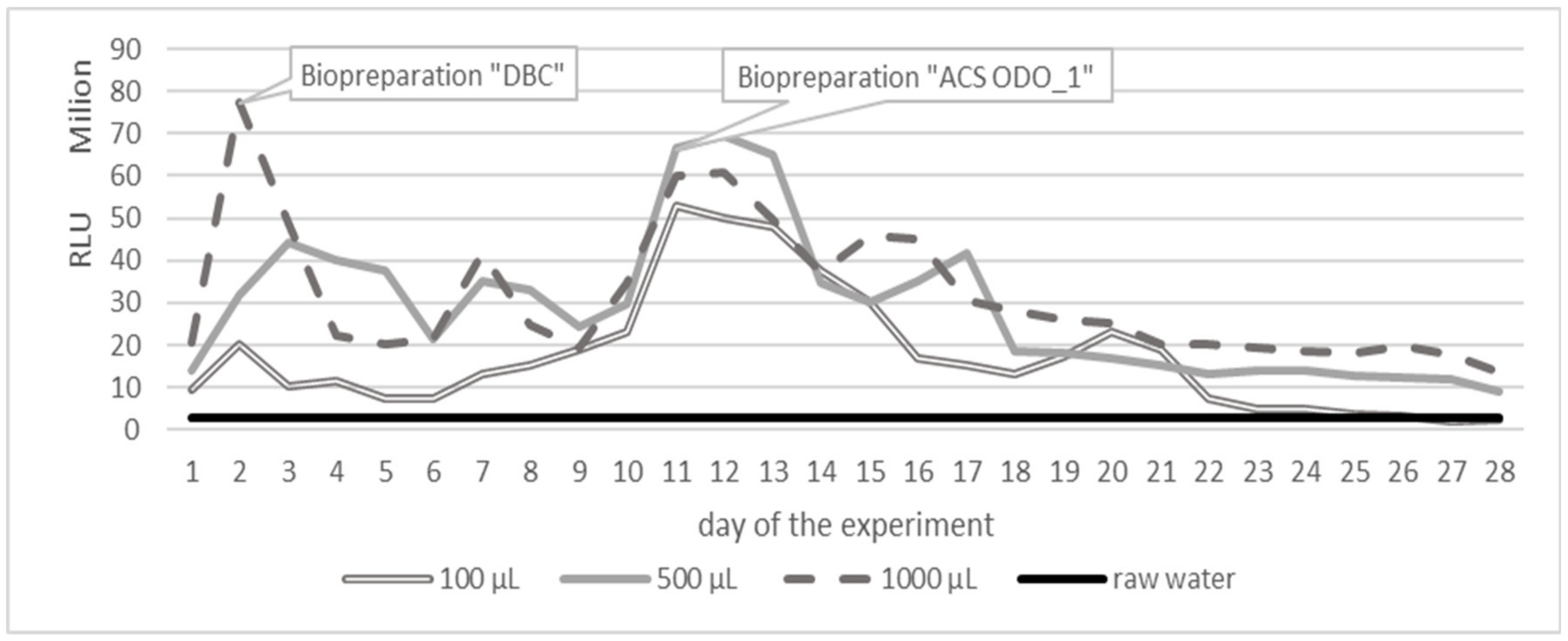
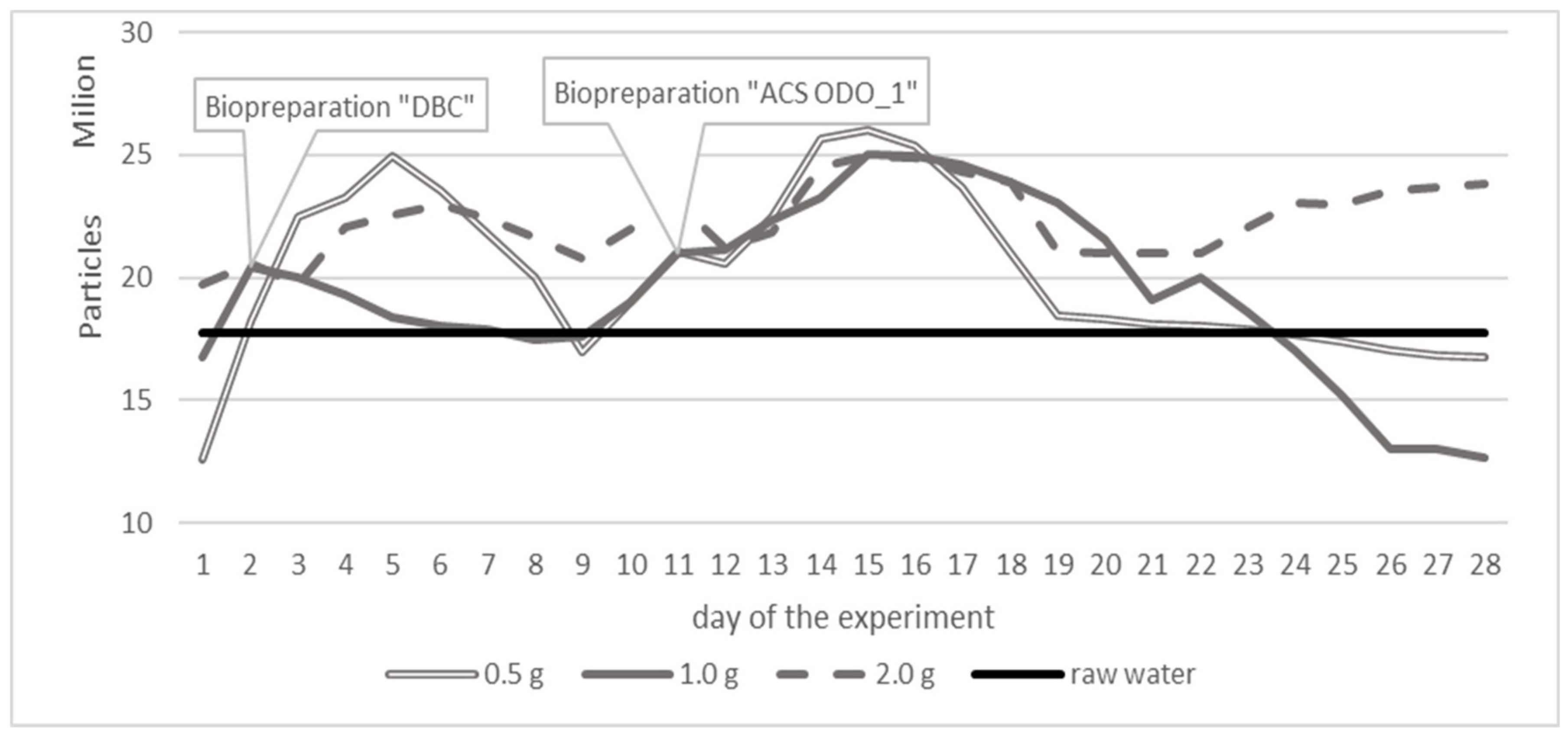
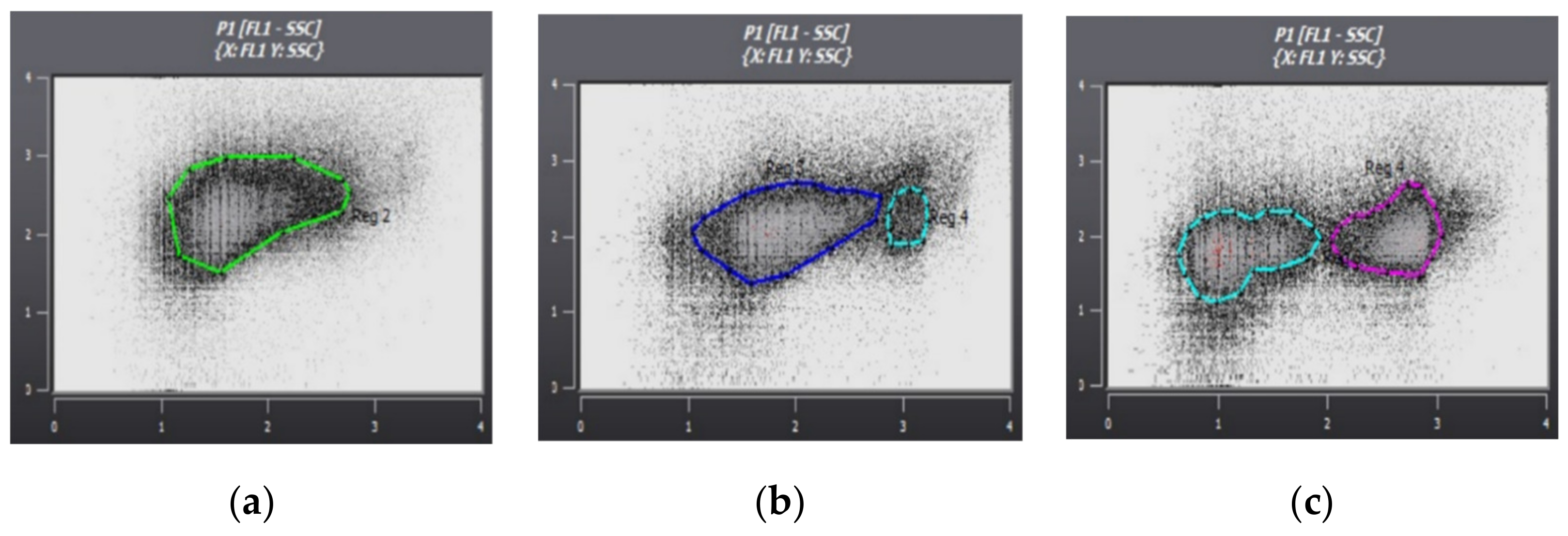
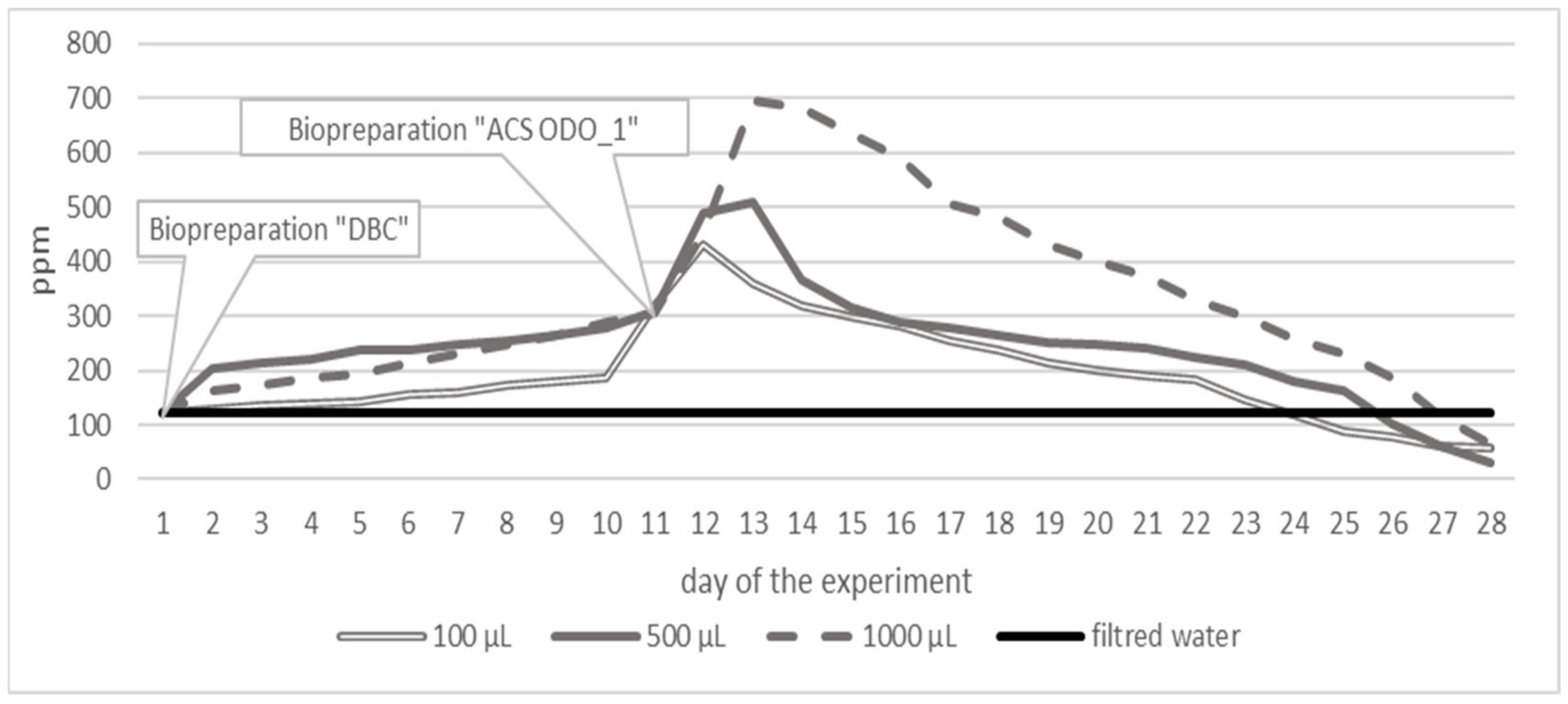
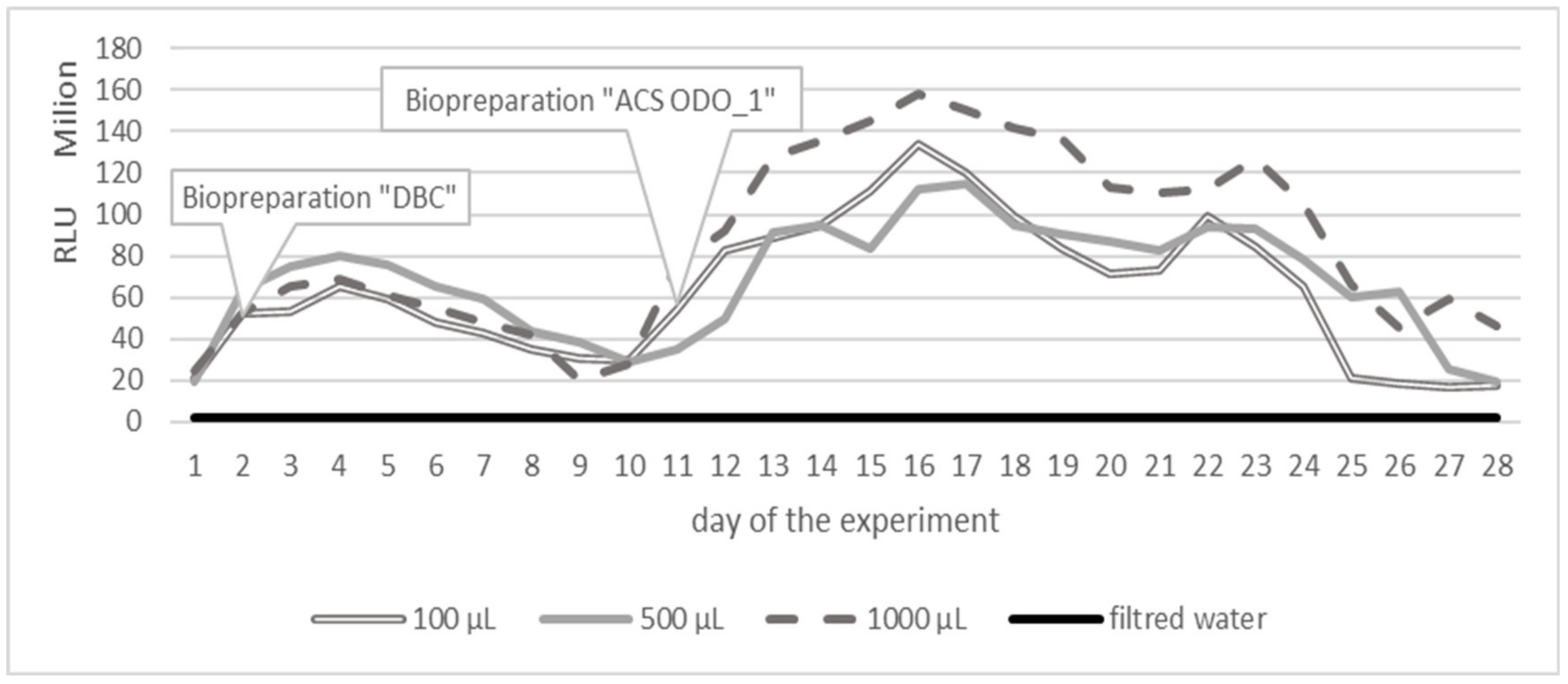
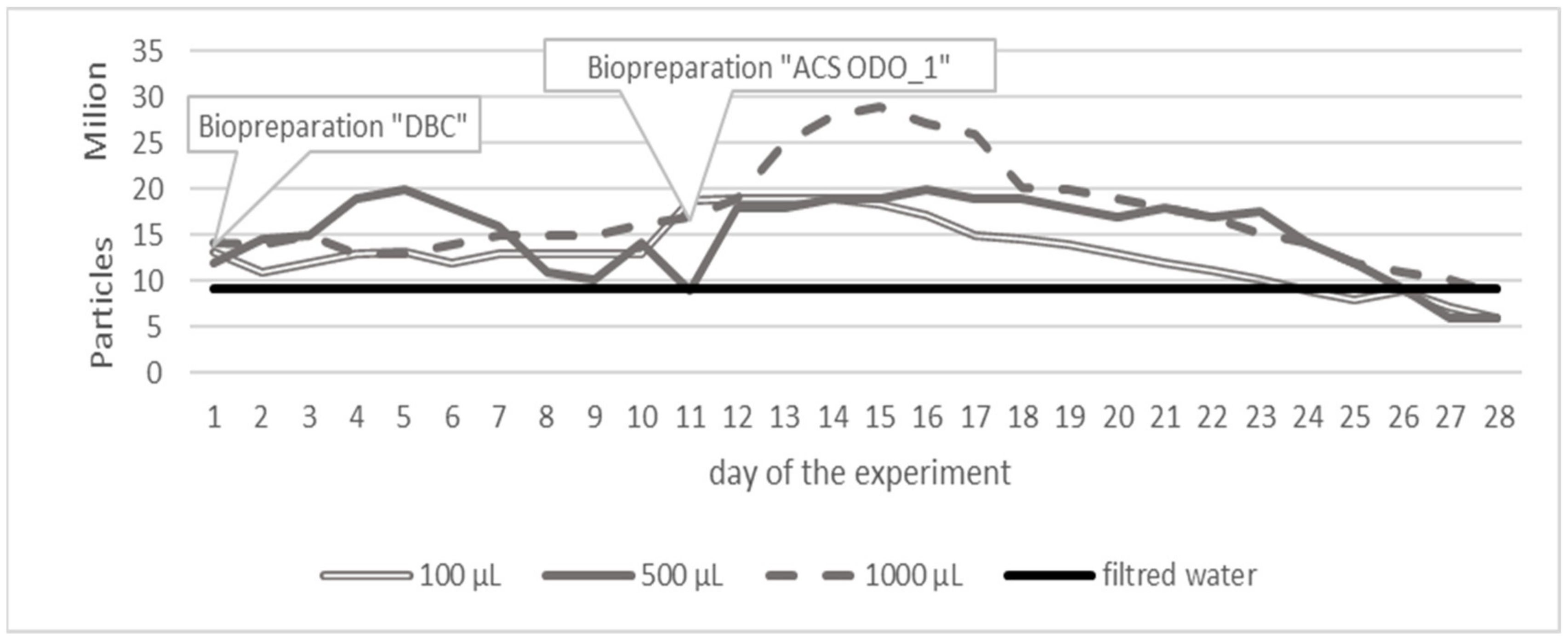
3. Results
3.1. Variant 1
3.2. Variant 2
3.3. Variant 3
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schulte, P.A.; Hauser, J.E. The use of biomarkers in occupational health research, practice, and policy. Toxicol. Lett. 2012, 213, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Shimp, R.J.; Pfaender, F.K. Effect of adaptation to phenol on biodegradation of monosubstituted phenols by aquatic microbial communities. Appl. Environ. Microbiol. 1987, 53, 1496–1499. [Google Scholar] [CrossRef] [PubMed]
- Aburas, M.M.A. Removal of Phenol Using Spore Forming Bacillus ABO11 Isolated from Waste Water Treatment Plant. Adv. Microbiol. 2016, 6, 898–908. [Google Scholar] [CrossRef][Green Version]
- Pop, C.-E.; Draga, S.; Măciucă, R.; Niță, R.; Crăciun, N.; Wolff, R. Bisphenol A Effects in Aqueous Environment on Lemna minor. Processes 2021, 9, 1512. [Google Scholar] [CrossRef]
- Weber, M.; Weber, M. Phenols. In Phenolic Resins: A Century of Progress; Pilato, L., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 9–22. [Google Scholar] [CrossRef]
- Hamdi, A.M.; Sillanpaa, M.; Dutta, J. Intermediate formation during photodegradation of phenol using lanthanum doped tin dioxide nanoparticles. Res. Chem. Intermed. 2016, 42, 3055–3069. [Google Scholar] [CrossRef]
- Commissioned by the General Directorate for Environmental Protection 2019. Expert Study: Analysis of the Information Necessary to Assess the Occurrence of A Significant Threat to Human Health or the State of the Environment in the Event of Exceeding the Permissible Content of Substances Causing Risk in Soil, Ground or Groundwater. Available online: https://sdr.gdos.gov.pl/Documents/GO/Ekspertyzy/Analiza%20informacji%20-%20gleby.pdf (accessed on 26 October 2020).
- Ghafari, S.; Baboli, Z.; Neisi, A.; Mirzaee, S.A.; Darvishi Cheshmeh Soltani, R.; Saeedi, R.; Abtahi, M.; Jorfi, S. Surfactant-enhanced Bioremediation of n-Hexadecane-contaminated Soil Using Halo-tolerant Bacteria Paenibacillus glucanolyticus sp. Strain T7-AHV Isolated from Marine Environment. Chem. Biochem. Eng. Q. 2019, 33, 111–123. [Google Scholar] [CrossRef]
- Integrated Risk Information System. Available online: https://www.epa.gov/iris (accessed on 26 October 2020).
- Das, N.; Chandran, P. Microbial Degradation of Petroleum Hydrocarbon Contaminants: An Overview. Biotechnol. Res. Int. 2011, 2011, 941810. [Google Scholar] [CrossRef]
- Jiang, J.C.; Stumpferl, S.W.; Tiwari, A.; Qin, Q.; Rodriguez-Quinones, J.F.; Jazwinski, M. Identification of the Target of the Retrograde Response that Mediates Replicative Lifespan Extension in Saccharomyces cerevisiae. Genetics 2016, 204, 659–673. [Google Scholar] [CrossRef] [PubMed]
- Kebria, D.Y.; Khodadadi, A.; Ganjidoust, H.; Badkoubi, A.; Amoozegar, M.A. Isolation and characterization of a novel native Bacillus strain capable of degrading diesel fuel. Int. J. Environ. Sci. Technol. 2009, 6, 435–442. [Google Scholar] [CrossRef]
- Shibulal, B.; Al-Bahry, S.N.; Al-Wahaibi, Y.M.; Elshafie, A.E.; Al-Bemani, A.S.; Joshi, S.J. The potential of indigenous Paenibacillus ehimensis BS1 for recovering heavy crude oil by biotransformation to light fractions. PLoS ONE 2017, 12, 2. [Google Scholar] [CrossRef]
- Kang, H.I.; Shin, H.S. Ultra-sensitive determination of cyanide in surface water by gas chromatography-tandem mass spectrometry after derivatization with 2-(dimethylamino) ethanethiol. Anal. Chim. Acta 2014, 828, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Frizzarin, R.M.; Rocha, F.R.P. A multi-pumping flow-based procedure with improved sensitivity for the spectrophotometric determination of acid-dissociable cyanide in natural waters. Anal. Chim. Acta 2013, 758, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Oluwole, O.; Onabolu, A.; Cotgreave, I.; Rosling, H.; Persson, A.; Link, H. Incidence of endemic ataxia polyneuropathy and its relation to exposure to cyanide in a Nigerian community. J. Neurol. Neurosurg. Psychiatry 2003, 74, 1417–1422. [Google Scholar] [CrossRef]
- Baumann, H.A.; Morrison, L.; Stengel, D.B. Metal accumulation and toxicity measured by PAM-chlorophyll fluorescence in seven species of marine macroalgae. Ecotoxicol. Environ. Saf. 2009, 72, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Ociepa-Kubicka, A.; Ociepa, E. Toksyczne oddziaływanie metali ciężkich na rośliny, zwierzęta i ludzi. Inżynieria I Ochr. Sr. 2012, 15, 169–180. [Google Scholar]
- Bartkiewicz, B.; Umiejewska, K. Oczyszczanie Ścieków Przemysłowych, 2nd ed.; Wydawnictwo Naukowe PWN Warszawa: Warszawa, Poland, 2010; ISBN 978-83-01-16267-2. [Google Scholar]
- Hassani, A.; Faraji, M.; Eghbali, P. Faciale fabrication of mpg-C3N4/Ag/ZnO nanovires/Zn photocatalyst plates for photodegradation of dye pollutant. J. Photochem. Photobiol. A Chem. 2020, 400, 112665. [Google Scholar] [CrossRef]
- Motlagh, P.Y.; Alireza Khataee, A.; Hassani, A.; Rad, T.S. ZnFe-LDH/GO nanocomposite coated on the glass support as a highly efficient catalyst for visible light photodegradation of an emerging pollutant. J. Mol. Liq. 2020, 302, 112532. [Google Scholar] [CrossRef]
- Brzychczyk, B.; Famielec, S.; Malinowski, M.; Salamon, J. Zastosowanie klinoptylolitu z procesie samooczyszczania wód małych zbiorników wodnych. Probl. Agric. Eng. 2015, 3, 71–82. [Google Scholar]
- Łopata, M.; Augustyniak, R.; Grochowska, J.; Parszuto, K.; Tandyrak, R. Selected Aspects of Lake Restorations in Poland. In Polish River Basins and Lakes—Part II; Korzeniewska, E., Harnisz, M., Eds.; The Handbook of Environmental Chemistry Springer: Cham, Switzerland, 2019; pp. 119–137. [Google Scholar]
- Nazir, R.; Rehman, S.; Nisa, N.; ali Baba, U. Freshwater Microbiology, Chapter 7—Exploring Bacterial Diversity: From Cell to Sequence; Bandh, S.A., Shafi, S., Shameem, N., Eds.; Academic Press: London, UK, 2019; pp. 263–306. [Google Scholar]
- Ni, Z.; Wu, X.; Li, L.; Lv, Z.; Zhang, Z.; Hao, A.; Iseri, Y.; Kuba, T.; Zhang, X.; Wu, W.M.; et al. Pollution control and in situ bioremediation for lake aquaculture using an ecological dam. J. Clean. Prod. 2017, 172, 2256–2265. [Google Scholar] [CrossRef]
- Shan, M.; Wang, Y.; Xue, S. Study on bioremediation of eutrophic lake. J. Environ. Sci. 2009, 21, 16–18. [Google Scholar] [CrossRef]
- Sitarek, M.; Napiórkowska-Krzebietke, A.; Mazur, R.; Czarnecki, B.; Pyka, J.P.; Stawecki, K.; Olech, M.; Sołtysiak, S.; Kapusta, A. Application of Effective Microorganisms Technology as a lake restoration tool-A case study of Muchawka Reservoir. J. Elem. 2017, 22, 529–543. [Google Scholar] [CrossRef]
- Gillespie, I.M.M.; Philp, J.C. Bioremediation, an environmental remediation technology for the bioeconomy. Trends Biotechnol. 2016, 31, 329–332. [Google Scholar] [CrossRef]
- Brito, S.F.; Pinheiro, C.L.; Nogueira, F.C.B.; Filho, S.M.; da Silva Matos, D.M. Influence of light on the initial growth of invasive Cryptostegia madagascariensis Bojer in the Brazilian semiarid region. In Project: Ecology of Cryptostegia Madagascariensis in the Semi-Arid Region Northeast Brazil; Acta Scientiarum Biological Sciences: Maringá, Brazil, 2015; pp. 385–392. [Google Scholar] [CrossRef]
- Mazurkiewicz, J.; Mazur, A.; Mazur, R.; Chmielowski, K.; Czekała, W.; Janczak, D. The Process of Microbiological Remediation of the Polluted Słoneczko Reservoir in Poland: For Reduction of Water Pollution and Nutrients Management. Water 2020, 12, 3002. [Google Scholar] [CrossRef]
- Mazur, A.; Wagner, A.; Mazur, R.; Bedla, D. Microbiological bioremediation of polluted waters as an alternative for chemical water treatment method. Przem. Chem. 2020, 99, 1344–1347. [Google Scholar] [CrossRef]
- Mazur, R. The application of microbiological biopreparations in the process of water remediation of the dam reservoir in Głuchów. Acta Sci. Pol. Form. Circumiectus 2020, 19, 81–95. [Google Scholar] [CrossRef]
- Kołodziej, G.; Tapper, J. Uporządkowanie gospodarki wodno-ściekowej w rejonie stawu “Kalina w Świętochłowicach”. Koncepcja TB-Proj. 2016, 19, 1–16. [Google Scholar]
- Kwinta, W. Świętochłowice: Rozpoczęła Się Remediacja Toksycznego Stawu Kalina. Available online: https://inzynieria.com/geoinzynieria/wiadomosci/60852,swietochlowice-rozpoczela-sie-remediacja-toksycznego-stawu-kalina (accessed on 26 April 2021).
- Michalska, J.; Greń, I.; Żur, J.; Wasilkowski, D.; Mrozik, A. Impact of the Biological Cotreatment of the Kalina Pond Leachate on Laboratory Sequencing Batch Reactor Operation and Activated Sludge Quality. Water 2019, 11, 1539. [Google Scholar] [CrossRef]
- Tkaczyk, A.M.; Pietrzak, M.; Kołak, A. Case of the environment reclamation in the region of Kalina pond in Świętochłowice. Pol. Geol. Inst. Spec. Pap. 2005, 17, 77–83. [Google Scholar]
- Wrześniak, A.; Kopyczok, J. Ocena stanu środowiska w rejonie obiektów objętych monitoringiem lokalnym, na terenie województwa śląskiego. In Biblioteka Monitoringu Środowiska; Environmental Monitoring Library: Katowice, Poland, 2012; ISBN 978-83-931871-8-8. [Google Scholar]
- Hammes, F.A.; Egli, T. New Method for Assimilable Organic Carbon Determination Using Flow-Cytometric Enumeration and a Natural Microbial Consortium as Inoculum. Environ. Sci. Technol. 2005, 39, 3289–3294. [Google Scholar] [CrossRef] [PubMed]
- Hammes, F.; Berney, M.; Wang, Y.; Vital, M.; Koster, O.; Egli, T. Flow-cytometric total bacterial cell counts as a descriptive microbiological parameter for drinking water treatment processes. Water Res. 2008, 42, 269–277. [Google Scholar] [CrossRef]
- Berney, M.; Vital, M.; Hulshoff, I.; Weilenmann, H.U.; Egli, T.; Hammes, F. Rapid, cultivation-independent assessment of microbial viability in drinking water. Water Res. 2008, 42, 4010–4018. [Google Scholar] [CrossRef] [PubMed]
- Brito, E.M.S.; De la Cruz Barron, M.; Caretta, C.A.; Goni-Urriza, M.; Andrade, L.H.; Cuevas-Rodriguez, G.; Malm, O.; Torres, J.P.M.; Simon, M.; Guyoneaud, R. Impact of hydrocarbons, PCBs and heavy metals on bacterial communities in Lerma River, Salamanca, Mexico: Investigation of hydrocarbon degradation potential. Sci. Total. Environ. 2015, 521, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Cirja, M.; Hommes, G.; Ivashechkin, P.; Prell, J.; Schaffer, A.; Corvini, P.F.X.; Lenz, M. Impact of bio-augmentation with Sphingomonas sp. strain TTNP3 in membrane bioreactors degrading nonylphenol. Appl. Microbiol. Biotechnol. 2009, 84, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Gao, X. Heavy metal pollution status in surface sediments of the coastal Bohai Bay. Water Res. 2012, 46, 1901–1911. [Google Scholar] [CrossRef]
- Nwanyanwu, C.E.; Abu, G.O. Influence of growth media on hydrophobicity of phenol-utilizing bacteria found in petroleum refinery effluent. Int. Res. J. Biol. Sci. 2013, 2, 6–11. [Google Scholar]
- Nwanyanwu, C.E.; Abu, G.O. Biodegradation of phenol at low and high doses by bacterial strains indigenous to Okrika River in the Niger Delta of Nigeria. J. Biol. Res. 2013, 3, 911–921. [Google Scholar]
- Singh, U.; Arora, N.K.; Sachan, P. Simultaneous biodegradation of phenol and cyanide present in coke-oven effluent using immobilized Pseudomonas putida and Pseudomonas stutzeri. Environ. Microbiol. Res. Pap. Braz. J. Microbiol. 2018, 49, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.A.; Jabeen, S. Degradation kinetics and pathway of phenol by Pseudomonas and Bacillus species. Biotechnol. Biotechnol. Equip. 2015, 29, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Rucká, L.; Nesvera, J.; Patek, M. Biodegradation of phenol and its derivatives by engineered bacteria: Current knowledge and perspectives. World J. Microbiol. Biotechnol. 2017, 33, 174. [Google Scholar] [CrossRef]
- Mingming, G.; Diao, M.H.; Yuan, S.; Wang, Y.K.; Xu, H.; Wang, X.H. Effects of phenol on physicochemical properties and treatment performances of partial nitrifying granules in sequencing batch reactors. Biotechnol. Rep. 2017, 13, 13–18. [Google Scholar] [CrossRef]
- Nweke, C.O.; Okpokwasili, G.C. Influence of exposure time on phenol toxicity to refinery wastewater bacteria. J. Environ. Chem. Ecotoxicol. 2010, 2, 20–27. [Google Scholar]
- Gullotto, A.; Branciamore, S.; Duchi, I.; Cano, M.F.P.; Randazzo, D.; Tilli, S.; Giardina, P.; Sannia, G.; Scozzafava, A.; Briganti, F. Combined action of a bacterial monooxygenase and a fungal laccase for the biodegradation of mono- and poly-aromatic hydrocarbons. Bioresour. Technol. 2008, 99, 8353–8359. [Google Scholar] [CrossRef] [PubMed]
- Haritash, A.K.; Kaushik, C.P. Biodegradation aspects of Polycyclic Aromatic Hydrocarbons (PAHs): A review. J. Hazard. Mater. 2009, 169, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Luque-Almagro, V.M.; Blasco, R.; Martinez-Luque, M.; Mareno-Vivian, C.; Castillo, F.; Roldan, M.D. Bacterial cyanide degradation is under review: Pseudomonas pseudoalcaligenes CECT5344, a case of an alkaliphilic cyanotroph. Biochem. Soc. Trans. 2011, 39, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Luque-Almagro, V.M.; Cabello, P.; Saez, L.P.; Olaya-Abril, A.; Moreno-Vivian, C.; Roldan, M.D. Exploring anaerobic environments for cyanide and cyano-derivatives microbial degradation. Appl. Microbiol. Biotechnol. 2018, 102, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Mirizadeh, S.; Yaghamaei, S.; Nejad, Z.G. Biodegradation of cyanide by a new isolated strain under alkaline conditions and optimization by response surface methodology (RSM). J. Environ. Health Sci. Eng. 2014, 12, 85. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Balomalumder, C.; Agarwal, V.K. Enzymatic mechanism and biochemistry for cyanide degradation: A review. J. Hazard. Mater. 2010, 15, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Potivichayanon, S.; Supromin, N.; Toensakes, R. Development of a mixed microbial culture for thiocyanate and metal cyanide degradation. 3 Biotech 2017, 7, 191. [Google Scholar] [CrossRef] [PubMed]
- Karamba, K.I.; Ahmad, S.A.; Zulkharnain, A.; Yasid, N.A.; Ibrahim, S.; Shukor, M.Y. Batch growth kinetic studies of locally isolated cyanide-degrading Serratia marcescens strain AQ07. 3 Biotech 2018, 8, 11. [Google Scholar] [CrossRef]
- Olszewska, M.; Łaniewska-Trokenheim, Ł. Odpowiedź bakterii fermentacji mlekowej na stres—stadium VBNC. Żywn. Nauk. Technol. Jakosc/Food. Sci. Technol. Qual. 2013, 5, 29–41. [Google Scholar]
- Piaskowski, K.; Anielak, A. Zeolity naturalne i ich zastosowanie w oczyszczaniu wody i ścieków. Ekol. I Tech. 2000, 8, 31–41. [Google Scholar]
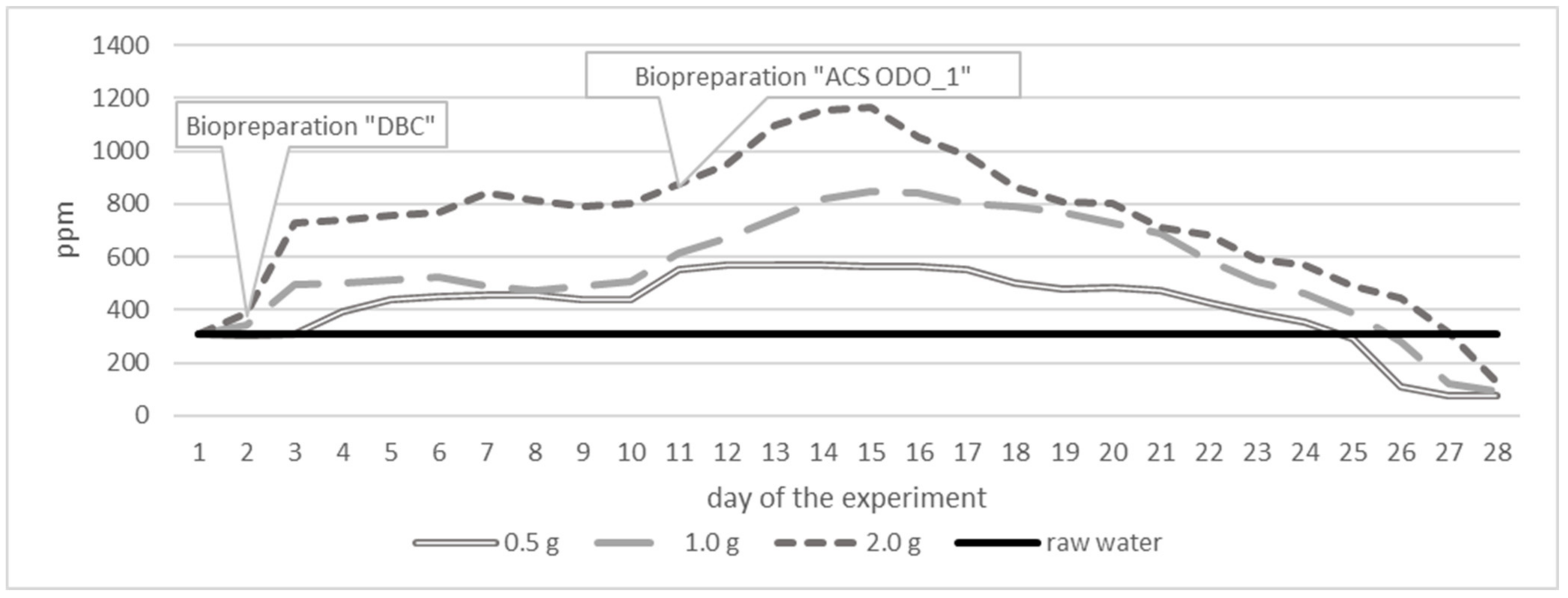
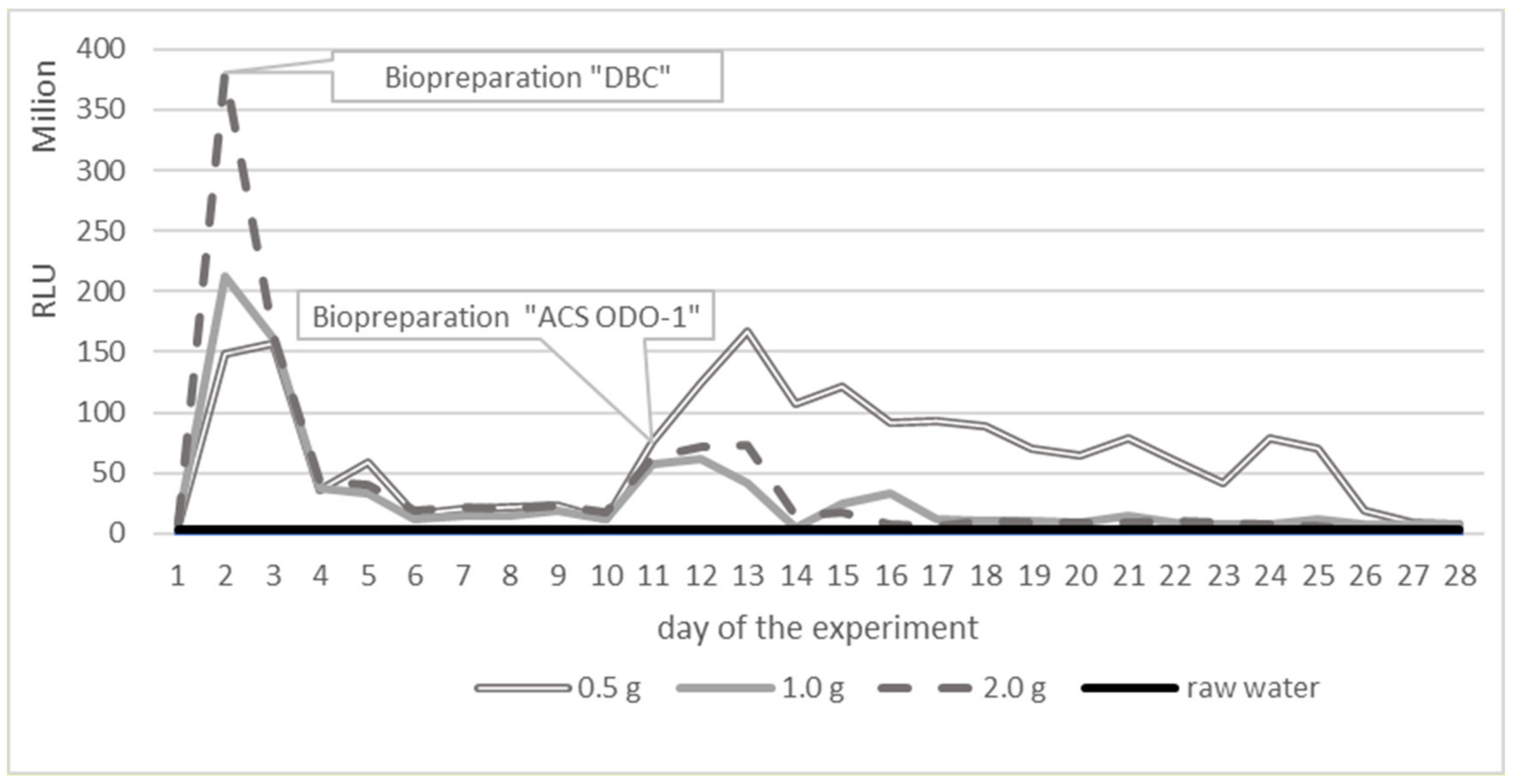
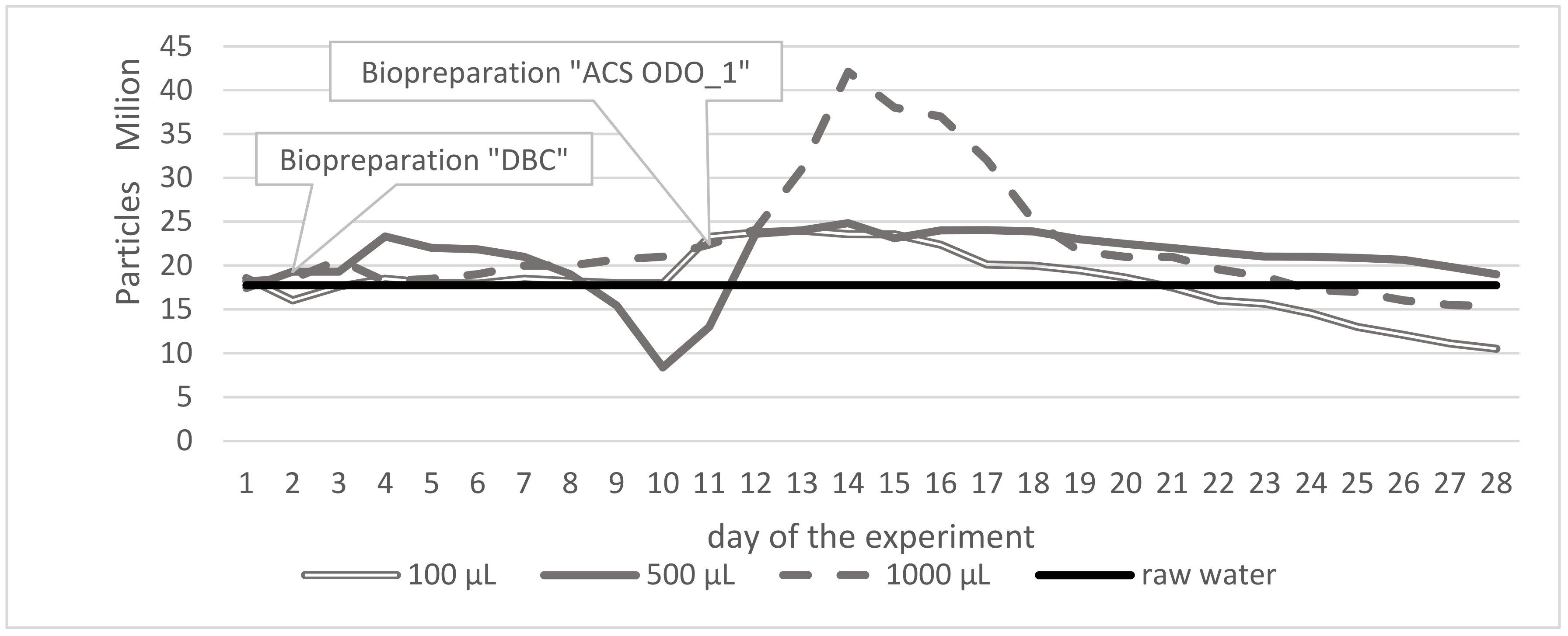

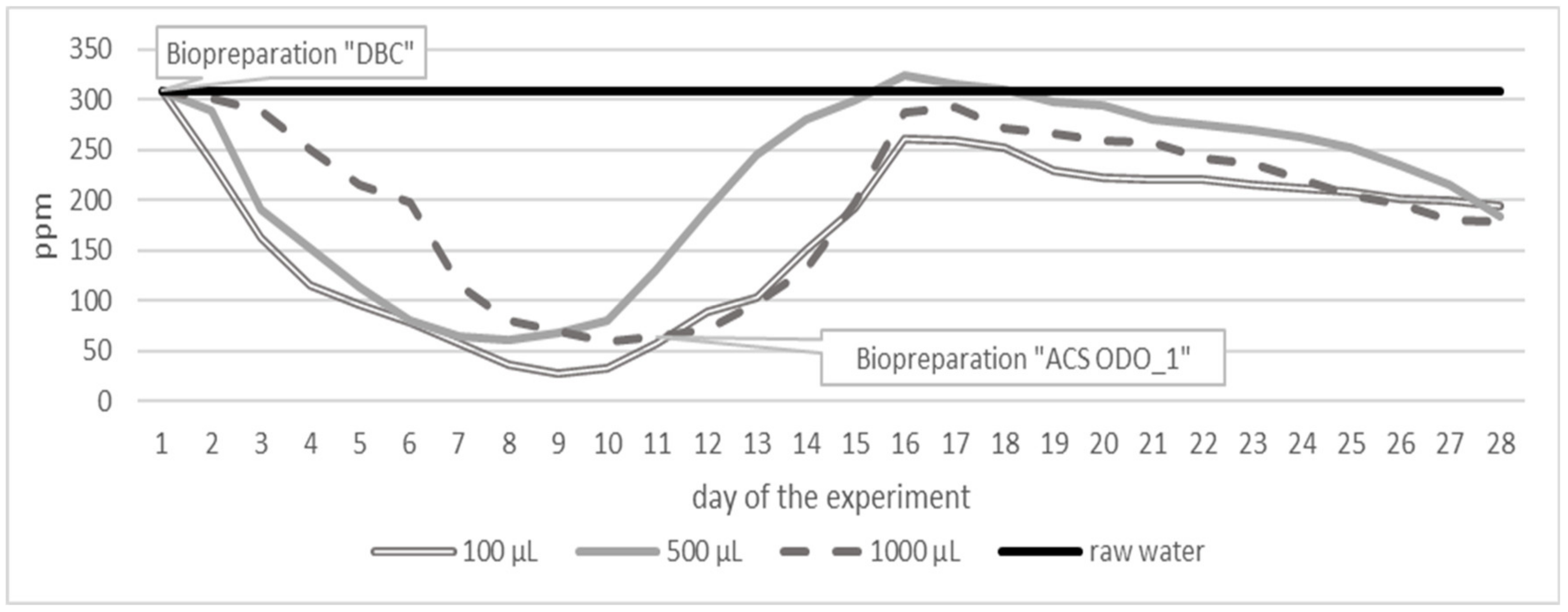
| Parameter | Unit | Surface Water Taken from 30 Different Places of Different Depth | ||
|---|---|---|---|---|
| Minimum | Maximum | Average | ||
| TOC | ppm | 308 | 310 | 309 |
| ATP | RLU | 3,018,500 | 3,018,905 | 3,018,703 |
| Turbidity | NTU | 175 | 185 | 180 |
| Arsenic (As) | µg/L | 16.10 | 19.90 | 17.64 |
| Barium (Ba) | µg/L | 53.60 | 86.20 | 70.39 |
| Chromium (Cr) | µg/L | 13.80 | 20.70 | 16.39 |
| Zinc (Zn) | µg/L | 7.10 | 21.20 | 9.96 |
| Nickel (Ni) | µg/L | 8.20 | 11.50 | 9.55 |
| Lead (Pb) | µg/L | 3.10 | 10.70 | 4.58 |
| Free cyanides | µg/L | 9.90 | 18.00 | 11.99 |
| Concentration of thirates | µg/L | 3900 | 4600 | 4190 |
| Cyanides total | µg/L | 240 | 260 | 247 |
| Phenol index | µg/L | 33,000 | 68,000 | 51,900 |
| O-xylene | µg/L | 7.24 | 49.70 | 16.90 |
| Benzene | µg/L | 2.34 | 97.70 | 44.76 |
| Toluene | µg/L | 1.05 | 64.00 | 26.85 |
| Etylobenzene | µg/L | 1.84 | 22.90 | 7.15 |
| (m + p)-xylene | µg/L | 18.10 | 117 | 43.61 |
| Acenaphthene | µg/L | 0.49 | 21.20 | 5.13 |
| Phenanthrene | µg/L | 1.02 | 41.40 | 8.59 |
| Fluoranthene | µg/L | 0.29 | 15.20 | 2.86 |
| Fluoren | µg/L | 0.19 | 12.80 | 2.06 |
| Naphthalene | µg/L | 3.22 | 350 | 99.01 |
| Pyrene | µg/L | 0.07 | 3.85 | 0.93 |
| PAH sum | µg/L | 8.73 | 453 | 120.36 |
| Index mineral oil/petroleum hydrocarbons | mg/L | 1.20 | 8.90 | 2.73 |
| pH | pH | 7.15 | 7.19 | 7.17 |
| Tested Parameter | Method/Standard |
|---|---|
| Turbidity | Nephelometric method; PN-EN ISO 7027: 2003 |
| Total Organic Carbon (TOC) | Sievers TOC analyzer 5310 C; PN-EN 1484: 1999 |
| ATP (Adenosine triphosphate) concentration | Luminometric determination; www.promega.com/protocols (accessed on 10 June 2021) |
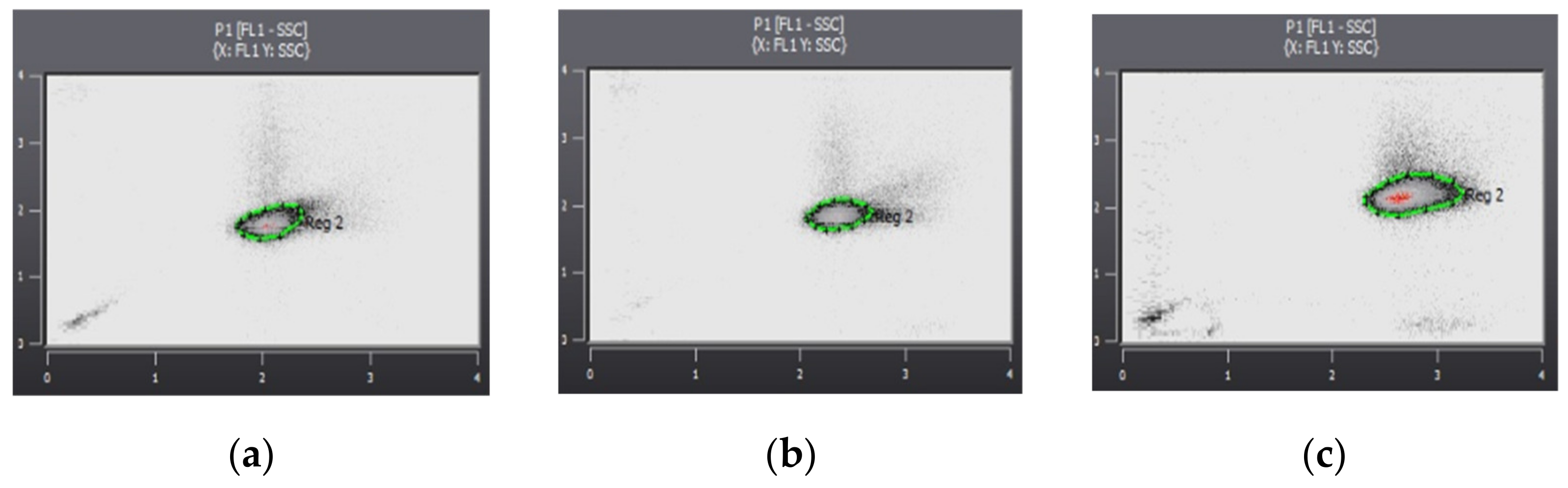
| Enumeration of microorganisms | Partec Cube 6 flow cytometer (FCM) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zamorska, J.; Kiełb-Sotkiewicz, I. A Biological Method of Treating Surface Water Contaminated with Industrial Waste Leachate. Water 2021, 13, 3644. https://doi.org/10.3390/w13243644
Zamorska J, Kiełb-Sotkiewicz I. A Biological Method of Treating Surface Water Contaminated with Industrial Waste Leachate. Water. 2021; 13(24):3644. https://doi.org/10.3390/w13243644
Chicago/Turabian StyleZamorska, Justyna, and Izabela Kiełb-Sotkiewicz. 2021. "A Biological Method of Treating Surface Water Contaminated with Industrial Waste Leachate" Water 13, no. 24: 3644. https://doi.org/10.3390/w13243644
APA StyleZamorska, J., & Kiełb-Sotkiewicz, I. (2021). A Biological Method of Treating Surface Water Contaminated with Industrial Waste Leachate. Water, 13(24), 3644. https://doi.org/10.3390/w13243644






