Sewage Markers as Determinants to Differentiate Origins of Emerging Organic Pollutants in an Urban Sri Lankan Water Drainage Network
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pre-Survey
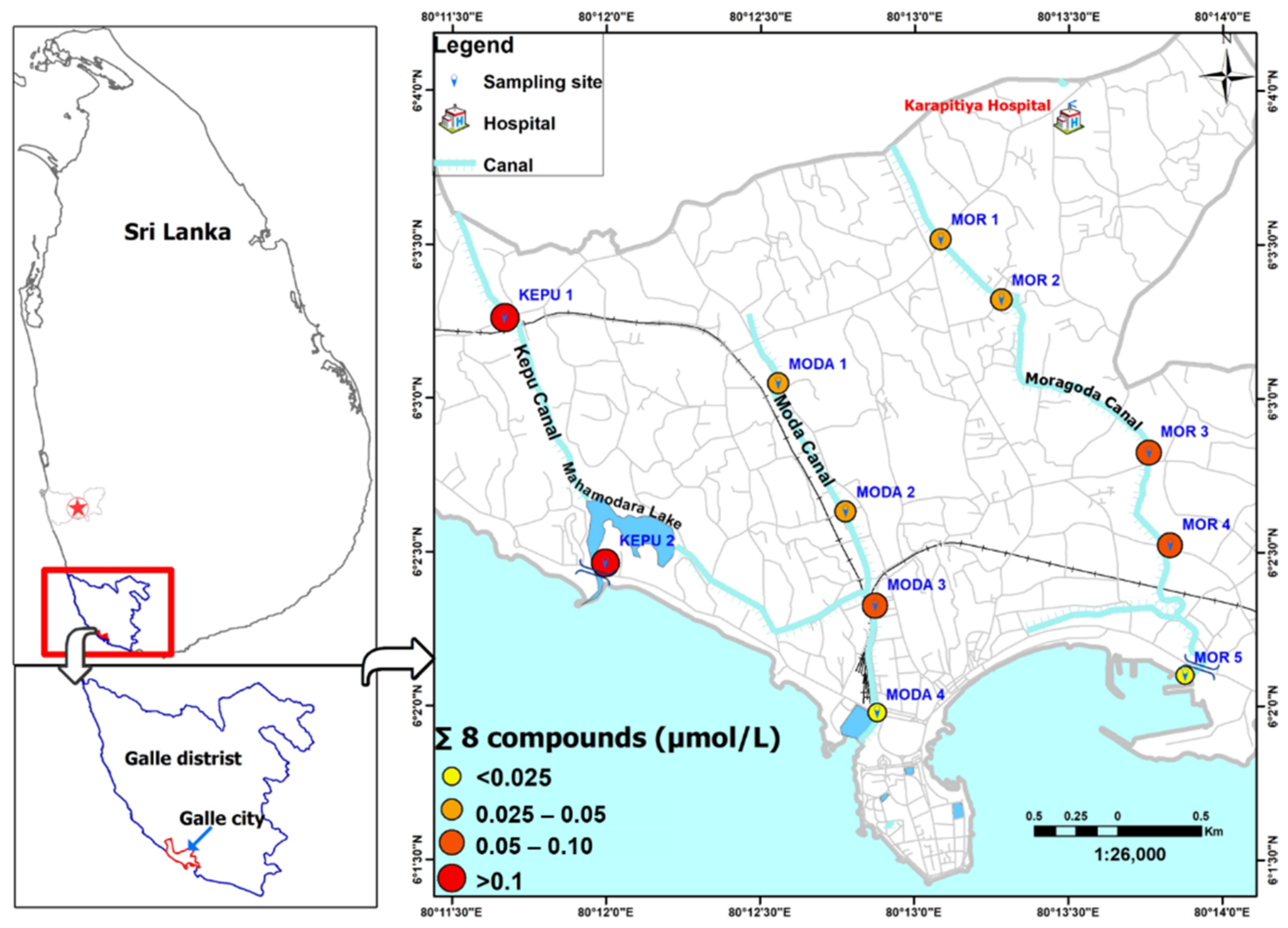
2.2. Sample Collection
2.3. Chemicals and Standards
2.4. LC-MS/MS Analysis
2.5. Proposed Method for Evaluating Pollutant Sources in Surface Water
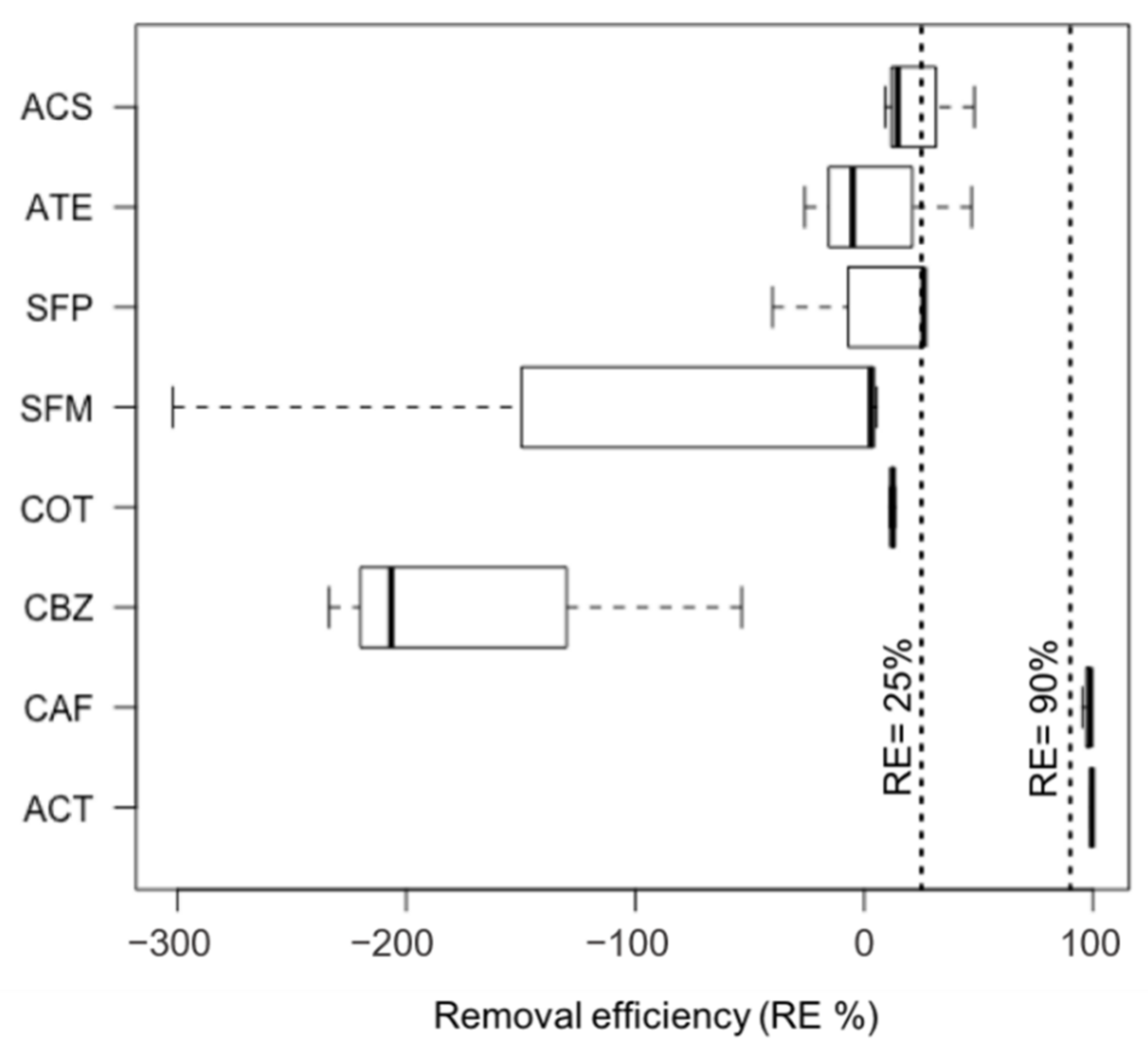
2.5.1. Removal Efficiency (RE) in the STP
2.5.2. Concentration Magnitude (CM) of Target Compounds in Raw Wastewater
2.5.3. Excretion Rate
2.5.4. Wastewater Burden (WB) in Surface Water
3. Results and Discussion
3.1. Study Area Sanitation
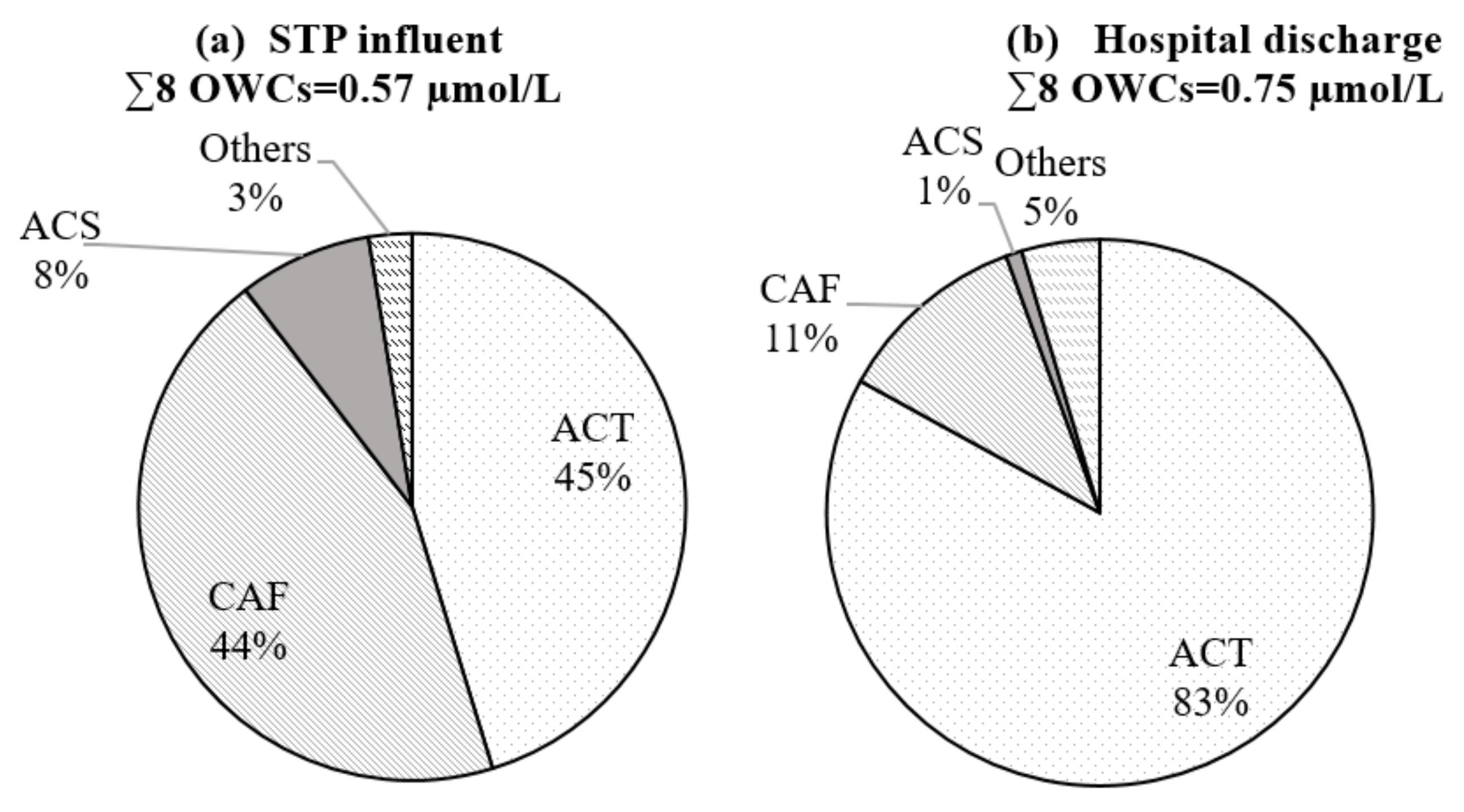
3.2. Occurrence and Removal Efficiency of Targeted Compounds in Raw Wastewater
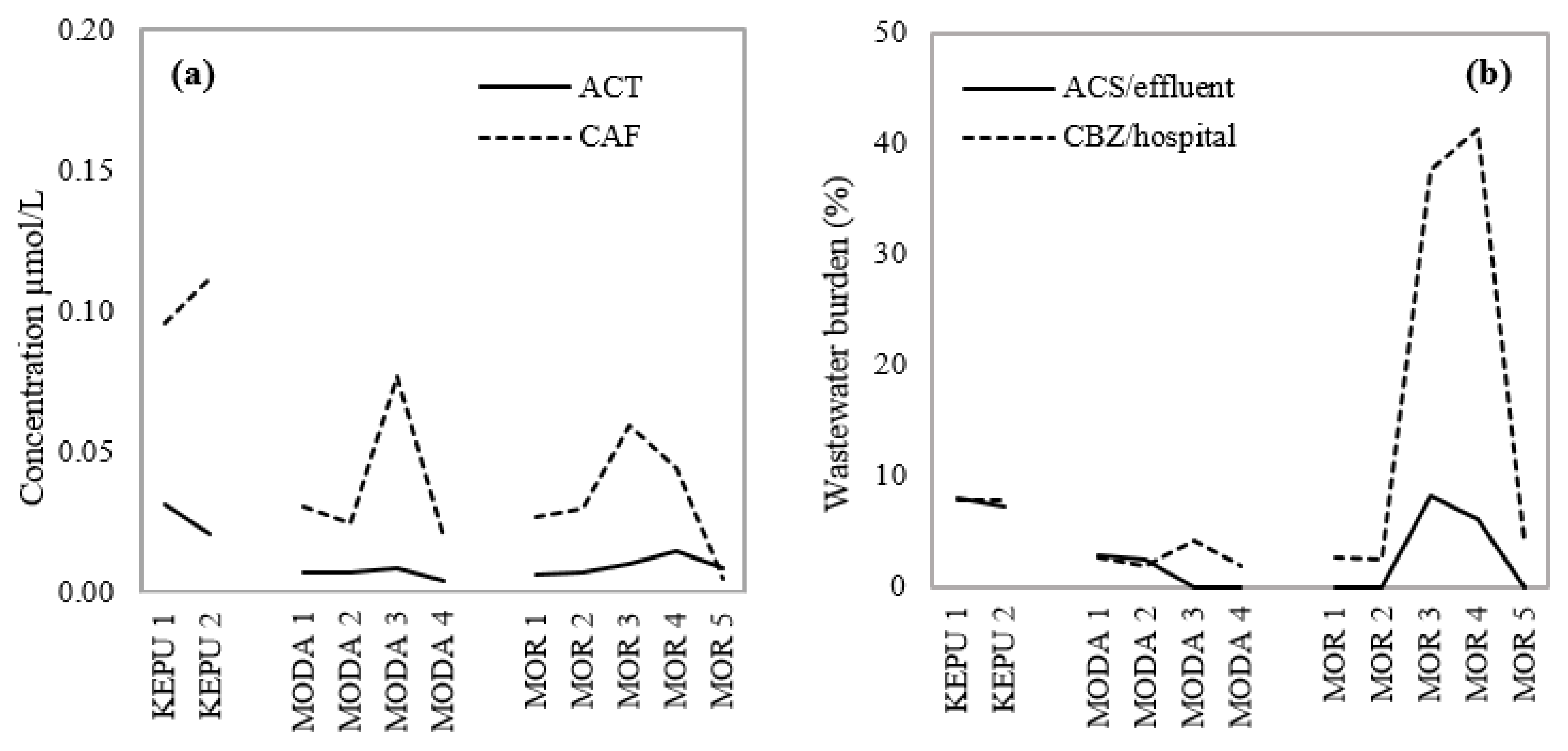
3.3. Selecting Wastewater Indicators to Differentiate Pollutant Sources
- -
- CAF indicates the magnitude of graywater loading at each sampling site.
- -
- ACT indicates the impact of untreated human excretion over short distances.
- -
- ACS indicates the general impact of domestic wastewater (both black and gray) on surface water.
- -
- CBZ indicates the presence of hospital residues over long distances.
| Marker | Category | Persistency | Excretion Rate (%) | Potential Pathway | Hospital Discharge CM ** | Urban Sewage CM | Hospital/Urban Ratio |
|---|---|---|---|---|---|---|---|
| ACS | Artificial sweetener | Y | >99 [32] | B, G | <10 | 60 | 0.2 |
| ACT | Antipyretic | N | <5 [33] | B | 625 | 260 | 2.4 |
| ATE | Beta-blocker | Y | 50 [34] | B | <10 | <10 | 2.6 |
| CAF | Stimulant | N | <5 [35] | G | 111 | 327 | 0.3 |
| CBZ | Neuroactive drugs | Y | <1 [36] | B | 167 | 43 | 3.9 |
| COT | Tobacco metabolite | Y | 13 * [37] | B | 21 | 21 | 1.0 |
| SFM | Antibiotic | Y | <20 [38] | B | 29 | <10 | 15.5 |
| SFP | Antibiotic | Y | <2 [39] | B | 24 | <10 | 5.0 |
3.4. Implications for Pollutants in Galle City Canal Drainage
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nation, Department of Economic and Social Affairs, Population Division. World Urbanization Prospects; The 2018 Revision (ST/ESA/SER.A/420); United Nations: New York, NY, USA, 2019; p. 9. [Google Scholar]
- WHO; UNICEF. Progress on Drinking Water, Sanitation and Hygiene: 2017 Update and SDG Baseline; WHO: Geneva, Switzerland, 2017; p. 66. [Google Scholar]
- Wicken, J.; Tep, M.; Seth, K.; Etherington, A. Water, Sanitation and Hygiene (WASH) Situation and Issues for Urban Poor People and Vulnerable Groups, Cambodia; WaterAid Cambodia: Phnom Penh, Cambodia, 2015; p. 101. [Google Scholar]
- Freeman, M.C.; Garn, J.V.; Sclar, G.D.; Boisson, S.; Medlicott, K.; Alexander, K.T.; Penakalapati, G.; Anderson, D.; Mahtani, A.G.; Grimes, J.; et al. The impact of sanitation on infectious disease and nutritional status: A systematic review and meta-analysis. Int. J. Hyg. Environ. Health 2017, 220, 928–949. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, P.; Blackett, I.; Heymans, C. Poor-Inclusive Urban Sanitation: An Overview; World Bank, Water and Sanitation Program (WSP); World Bank: Washington, DC, USA, 2013. [Google Scholar]
- Lim, F.Y.; Ong, S.L.; Hu, J. Recent Advances in the Use of Chemical Markers for Tracing Wastewater Contamination in Aquatic Environment: A Review. Water 2017, 9, 143. [Google Scholar] [CrossRef]
- Kümmerer, K. The presence of pharmaceuticals in the environment due to human use—Present knowledge and future challenges. J. Environ. Manag. 2009, 90, 2354–2366. [Google Scholar] [CrossRef] [PubMed]
- Boxall, A.B.; Rudd, M.; Brooks, B.; Caldwell, D.J.; Choi, K.; Hickmann, S.; Innes, E.; Ostapyk, K.; Staveley, J.P.; Verslycke, T.; et al. Pharmaceuticals and Personal Care Products in the Environment: What Are the Big Questions? Environ. Health Perspect. 2012, 120, 1221–1229. [Google Scholar] [CrossRef]
- Simazaki, D.; Kubota, R.; Suzuki, T.; Akiba, M.; Nishimura, T.; Kunikane, S. Occurrence of selected pharmaceuticals at drinking water purification plants in Japan and implications for human health. Water Res. 2015, 76, 187–200. [Google Scholar] [CrossRef]
- Schwab, B.W.; Hayes, E.P.; Fiori, J.M.; Mastrocco, F.J.; Roden, N.M.; Cragin, D.; Meyerhoff, R.D.; D’Aco, V.J.; Anderson, P.D. Human pharmaceuticals in US surface waters: A human health risk assessment. Regul. Toxicol. Pharmacol. 2005, 42, 296–312. [Google Scholar] [CrossRef]
- Gaffney, V.D.J.; Almeida, C.M.; Rodrigues, A.; Ferreira, E.; Benoliel, M.J.; Cardoso, V.V. Occurrence of pharmaceuticals in a water supply system and related human health risk assessment. Water Res. 2015, 72, 199–208. [Google Scholar] [CrossRef]
- Tran, N.H.; Reinhard, M.; Gin, K.Y.-H. Occurrence and fate of emerging contaminants in municipal wastewater treatment plants from different geographical regions-a review. Water Res. 2018, 133, 182–207. [Google Scholar] [CrossRef]
- Gaston, L.; Lapworth, D.J.; Stuart, M.; Arnscheidt, J. Prioritization Approaches for Substances of Emerging Concern in Groundwater: A Critical Review. Environ. Sci. Technol. 2019, 53, 6107–6122. [Google Scholar] [CrossRef]
- Kumar, M.; Ram, B.; Honda, R.; Poopipattana, C.; Canh, V.D.; Chaminda, T.; Furumai, H. Concurrence of antibiotic resistant bacteria (ARB), viruses, pharmaceuticals and personal care products (PPCPs) in ambient waters of Guwahati, India: Urban vulnerability and resilience perspective. Sci. Total. Environ. 2019, 693, 133640. [Google Scholar] [CrossRef]
- Yang, Y.-Y.; Zhao, J.-L.; Liu, Y.-S.; Liu, W.-R.; Zhang, Q.-Q.; Yao, L.; Hu, L.-X.; Zhang, J.-N.; Jiang, Y.-X.; Ying, G.-G. Pharmaceuticals and personal care products (PPCPs) and artificial sweeteners (ASs) in surface and ground waters and their application as indication of wastewater contamination. Sci. Total. Environ. 2018, 616–617, 816–823. [Google Scholar] [CrossRef]
- Vumazonke, S.; Khamanga, S.M.; Ngqwala, N. Detection of Pharmaceutical Residues in Surface Waters of the Eastern Cape Province. Int. J. Environ. Res. Public Health 2020, 17, 4067. [Google Scholar] [CrossRef] [PubMed]
- Do, T.T.Q.; Otaki, M.; Otaki, Y.; Chaminda, T.; Wijesinghe, I.S. Pharmaceutical contaminants in shallow groundwater and their implication to poor sanitation facilities in low-income countries. Environ. Toxicol. Chem. 2021, 1–9. [Google Scholar]
- Valdez-Carrillo, M.; Abrell, L.; Ramírez-Hernández, J.; Reyes-López, J.A.; Carreón-Diazconti, C. Pharmaceuticals as emerging contaminants in the aquatic environment of Latin America: A review. Environ. Sci. Pollut. Res. 2020, 27, 44863–44891. [Google Scholar] [CrossRef]
- ADB/JICA/World Bank. Toward Sustainable Water and Sanitation Services in Sri Lanka: Beyond Sustainable Development Goals to Supporting the National Economic Vision; World Bank Publications: Washington, DC, USA, 2016; p. 13. [Google Scholar]
- Sri Lanka Department of Census and Statistics, Ministry of National Policies and Economic Affairs. Sri Lanka Demographic and Health Survey 2016; S&S Printers: Colombo, Sri Lanka, 2017.
- Morel, A.; Diener, S. Greywater Management in Low and Middle-Income Countries, Review of Different Treatment Systems for Households or Neighbourhoods; Swiss Federal Institute of Aquatic Science and Technology (Eawag): Dubendorf, Switzerland, 2006. [Google Scholar]
- Wiest, L.; Chonova, T.; Bergé, A.; Baudot, R.; Bessueille-Barbier, F.; Ayouni-Derouiche, L.; Vulliet, E. Two-year survey of specific hospital wastewater treatment and its impact on pharmaceutical discharges. Environ. Sci. Pollut. Res. 2017, 25, 9207–9218. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.H.; Reinhard, M.; Khan, E.; Chen, H.; Nguyen, V.T.; Li, Y.; Goh, S.G.; Nguyen, Q.; Saeidi, N.; Gin, K.Y.-H. Emerging contaminants in wastewater, stormwater runoff, and surface water: Application as chemical markers for diffuse sources. Sci. Total. Environ. 2019, 676, 252–267. [Google Scholar] [CrossRef] [PubMed]
- Kosma, C.I.; Lambropoulou, D.A.; Albanis, T.A. Investigation of PPCPs in wastewater treatment plants in Greece: Occurrence, removal and environmental risk assessment. Sci. Total Environ. 2014, 466–467, 421–438. [Google Scholar] [CrossRef]
- Paulus, G.K.; Hornstra, L.M.; Alygizakis, N.; Slobodnik, J.; Thomaidis, N.; Medema, G. The impact of on-site hospital wastewater treatment on the downstream communal wastewater system in terms of antibiotics and antibiotic resistance genes. Int. J. Hyg. Environ. Health 2019, 222, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Thai, P.K.; Kaserzon, S.L.; O’Brien, J.W.; Eaglesham, G.; Mueller, J.F. Assessment of drugs and personal care products biomarkers in the influent and effluent of two wastewater treatment plants in Ho Chi Minh City, Vietnam. Sci. Total Environ. 2018, 631–632, 469–475. [Google Scholar] [CrossRef]
- Palli, L.; Spina, F.; Varese, G.C.; Vincenzi, M.; Aragno, M.; Arcangeli, G.; Mucci, N.; Santianni, D.; Caffaz, S.; Gori, R. Occurrence of selected pharmaceuticals in wastewater treatment plants of Tuscany: An effect-based approach to evaluate the potential environmental impact. Int. J. Hyg. Environ. Health 2019, 222, 717–725. [Google Scholar] [CrossRef]
- O’Brien, J.W.; Banks, A.P.W.; Novic, A.J.; Mueller, J.F.; Jiang, G.; Ort, C.; Eaglesham, G.; Yuan, Z.; Thai, P.K. Impact of in-Sewer Degradation of Pharmaceutical and Personal Care Products (PPCPs) Population Markers on a Population Model. Environ. Sci. Technol. 2017, 51, 3816–3823. [Google Scholar] [CrossRef]
- Nakada, N.; Tanishima, T.; Shinohara, H.; Kiri, K.; Takada, H. Pharmaceutical chemicals and endocrine disrupters in municipal wastewater in Tokyo and their removal during activated sludge treatment. Water Res. 2006, 40, 3297–3303. [Google Scholar] [CrossRef]
- Tran, N.H.; Hoang, L.; Nghiem, L.; Nguyen, N.M.H.; Ngo, H.H.; Guo, W.; Trinh, Q.T.; Mai, N.H.; Chen, H.; Nguyen, D.D.; et al. Occurrence and risk assessment of multiple classes of antibiotics in urban canals and lakes in Hanoi, Vietnam. Sci. Total. Environ. 2019, 692, 157–174. [Google Scholar] [CrossRef]
- Oppenheimer, J.; Eaton, A.; Badruzzaman, M.; Haghani, A.W.; Jacangelo, J.G. Occurrence and suitability of sucralose as an indicator compound of wastewater loading to surface waters in urbanized regions. Water Res. 2011, 45, 4019–4027. [Google Scholar] [CrossRef]
- Magnuson, B.A.; Carakostas, M.C.; Moore, N.H.; Poulos, S.; Renwick, A.G. Biological fate of low-calorie sweeteners. Nutr. Rev. 2016, 74, 670–689. [Google Scholar] [CrossRef] [Green Version]
- Cummings, A.J.; King, M.L.; Martin, B.K. A kinetic study of drug elimination: The excretion of paracetamol and its metabolites in manmetabolites in man. Br. J. Pharmacol. Chemother. 1967, 29, 150–157. [Google Scholar] [CrossRef]
- Kirch, W.; Görg, K.G. Clinical pharmacokinetics of atenolol—A review. Eur. J. Drug Metab. Pharmacokinet. 1982, 7, 81–91. [Google Scholar] [CrossRef]
- Magkos, F.; Kavouras, S.A. Caffeine use in sports, pharmacokinetics in man, and cellular mechanisms of action. Crit. Rev. Food Sci. Nutr. 2005, 45, 535–562. [Google Scholar] [CrossRef] [PubMed]
- Richter, W.J.; Kriemler, P.; Faigle, J.W. Newer Aspects of the Biotransformation of Carbamazepine: Structural Characterization of Highly Polar Metabolites; Springer: Boston, MA, USA, 1978; pp. 1–14. [Google Scholar] [CrossRef]
- Heinrich-Ramm, R.; Wegner, R.; Garde, A.H.; Baur, X. Cotinine excretion (tobacco smoke biomarker) of smokers and non-smokers: Comparison of GC/MS and RIA results. Int. J. Hyg. Environ. Health 2002, 205, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Vree, T.B.; van der Ven, A.J.A.M.; Koopmans, P.P.; Va Kolmer, E.W.J.; Van Wissen, C.P.W.G.M. Pharmacokinetics of Sulfamethoxazole with its Hydroxy Metabolites and N4-Acetyl-, N1-Glucuronide Conjugates in Healthy Human Volunteers. Clin. Drug Investig. 1995, 9, 43–53. [Google Scholar] [CrossRef]
- Schröder, H.; Campbell, D.E.S. Absorption, metabolism, and excretion of salicylazosulfapyridine in man. Clin. Pharmacol. Ther. 1972, 13, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Takagi, K.; Otaki, M.; Otaki, Y.; Chaminda, T. Availability and public acceptability of residential rainwater use in Sri Lanka. J. Clean. Prod. 2019, 234, 467–476. [Google Scholar] [CrossRef]
| Wastewater | Facility Type | National (%) | Galle City (%) |
|---|---|---|---|
| Blackwater | Improved sewerage | 12.3 | 0 |
| Improved septic | 5.1 | 19.2 | |
| Improved pit latrine | 80.3 | 80.8 | |
| Open defecation | 0.4 | 0 | |
| Graywater (laundry, kitchen, bathroom) | Direct discharge | n.a. | 61.3 |
| Septic/pit | n.a. | 38.7 |
| Target Compounds | IQL a µg/L | STP Influent | Hospital Wastewater | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| This Study | Previous Studies | This Study | Previous Studies | |||||||
| Effluent | Influent | Effluent | ||||||||
| Range | Mean | CM | Influent | Range | Mean | CM * | Mean/Median Value | |||
| ACT | 0.15 | 29.7–44.3 | 39.0 | 260 | 116.0 a 77.7 b | 70.2–123.6 | 92.8 | 625 | 813.5 a | 0.04 a |
| CAF | 0.15 | 16.2–68.7 | 49.1 | 327 | 35.2 b | 12.5–23.7 | 16.6 | 110 | 28.2 c | 0.35 c |
| CBZ | 0.003 | 0.07–0.24 | 0.13 | 43 | 0.53 a 0.37 b | 0.44–0.60 | 0.50 | 167 | 0.3 a 0.08 c | 0.63 a 0.015 c |
| COT | 0.1 | 1.9–2.2 | 1.9 | 21 | n.a. | 1.36–2.35 | 1.86 | 21 | n.a. | n.a. |
| SFM | 0.1 | 0.1–0.2 | 0.17 | 2 | 0.5 a 0.01–0.1 d | 2.20–2.96 | 2.58 | 29 | 3.9 a 1.4 c 0.1–1 d | 0.81 a 0.21 c 0.001–0.01 d |
| SFP | 0.1 | 0.38–0.47 | 0.44 | 5 | 0.072–1 d | 2.05–2.32 | 2.18 | 24 | 0.01–0.1 d | bql d |
| ATE | 0.15 | 0.19–0.48 | 0.30 | 2 | 2.4 a | 0.54–1.11 | 0.79 | 4 | 3.2 a | 0.019 a |
| ACS | 0.15 | 8.2–10.6 | 9.0 | 60 | 11.5 b | 0.98–2.0 | 1.43 | 9 | n.a. | n.a. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quyen, D.T.T.; Masahiro, O.; Otaki, Y.; Chaminda, T. Sewage Markers as Determinants to Differentiate Origins of Emerging Organic Pollutants in an Urban Sri Lankan Water Drainage Network. Water 2021, 13, 2898. https://doi.org/10.3390/w13202898
Quyen DTT, Masahiro O, Otaki Y, Chaminda T. Sewage Markers as Determinants to Differentiate Origins of Emerging Organic Pollutants in an Urban Sri Lankan Water Drainage Network. Water. 2021; 13(20):2898. https://doi.org/10.3390/w13202898
Chicago/Turabian StyleQuyen, Do Thi Thuy, Otaki Masahiro, Yurina Otaki, and Tushara Chaminda. 2021. "Sewage Markers as Determinants to Differentiate Origins of Emerging Organic Pollutants in an Urban Sri Lankan Water Drainage Network" Water 13, no. 20: 2898. https://doi.org/10.3390/w13202898
APA StyleQuyen, D. T. T., Masahiro, O., Otaki, Y., & Chaminda, T. (2021). Sewage Markers as Determinants to Differentiate Origins of Emerging Organic Pollutants in an Urban Sri Lankan Water Drainage Network. Water, 13(20), 2898. https://doi.org/10.3390/w13202898







