Effect of Biochar Amendment in Woodchip Denitrifying Bioreactors for Nitrate and Phosphate Removal in Tile Drainage Flow
Abstract
:1. Introduction
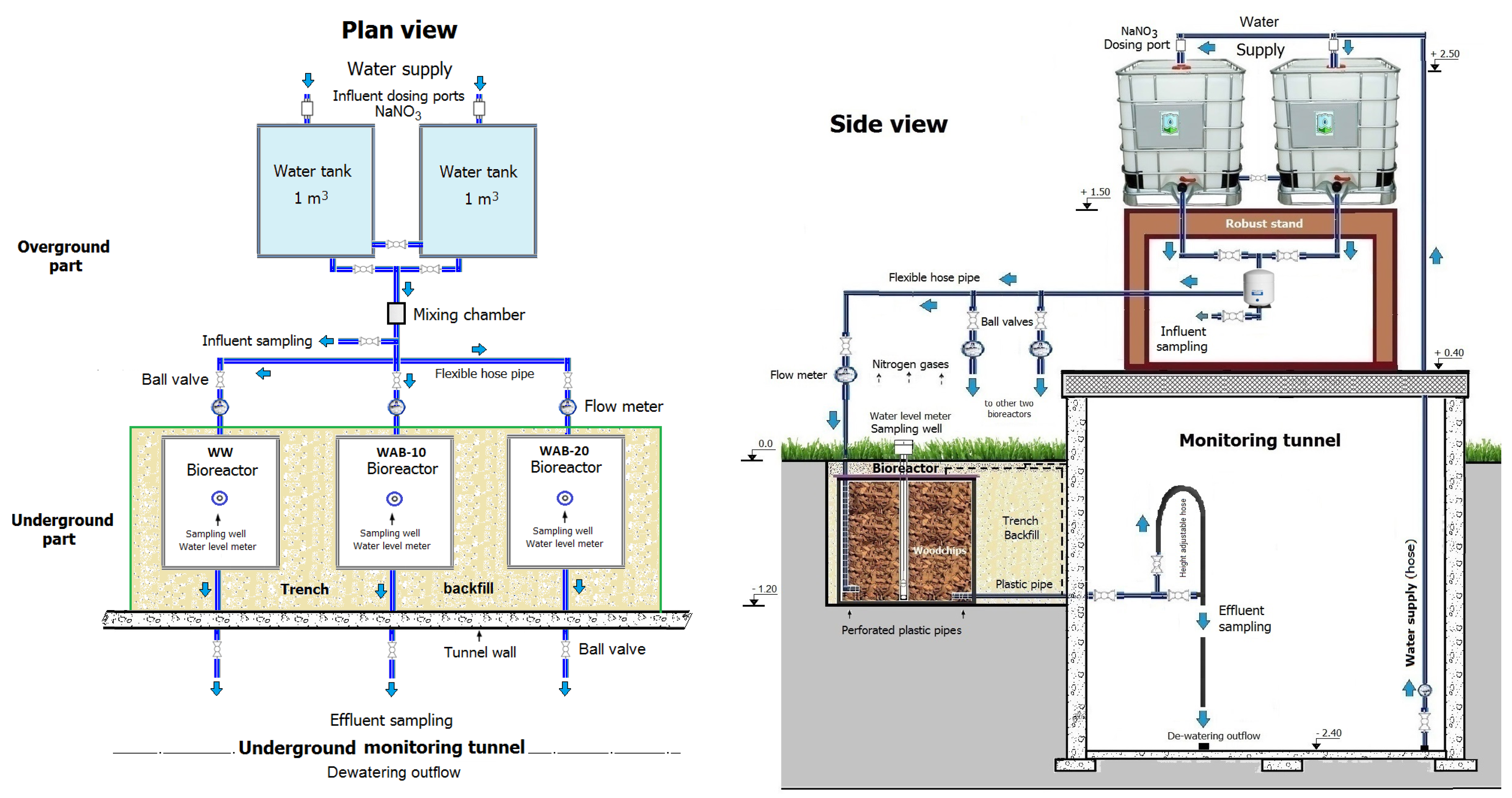
2. Materials and Methods
3. Results
3.1. Inflow Parameters Change
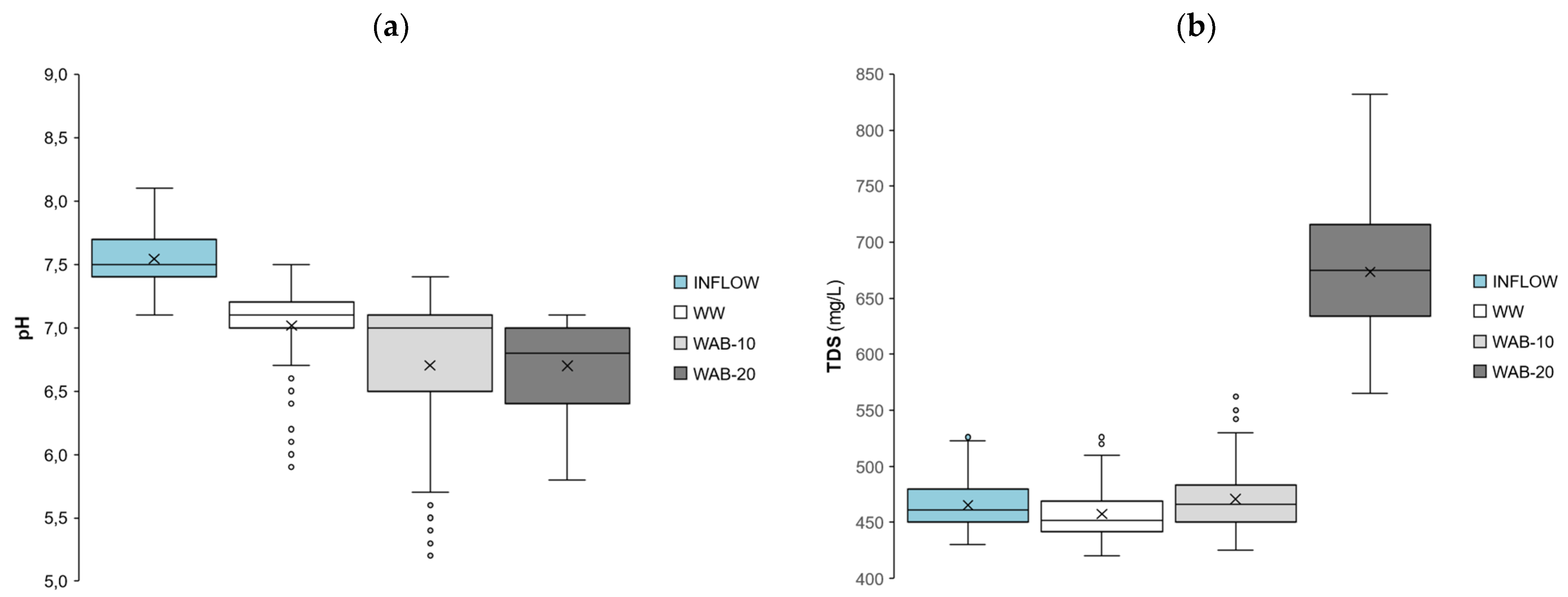
3.2. The pH and Total Dissolved Solids
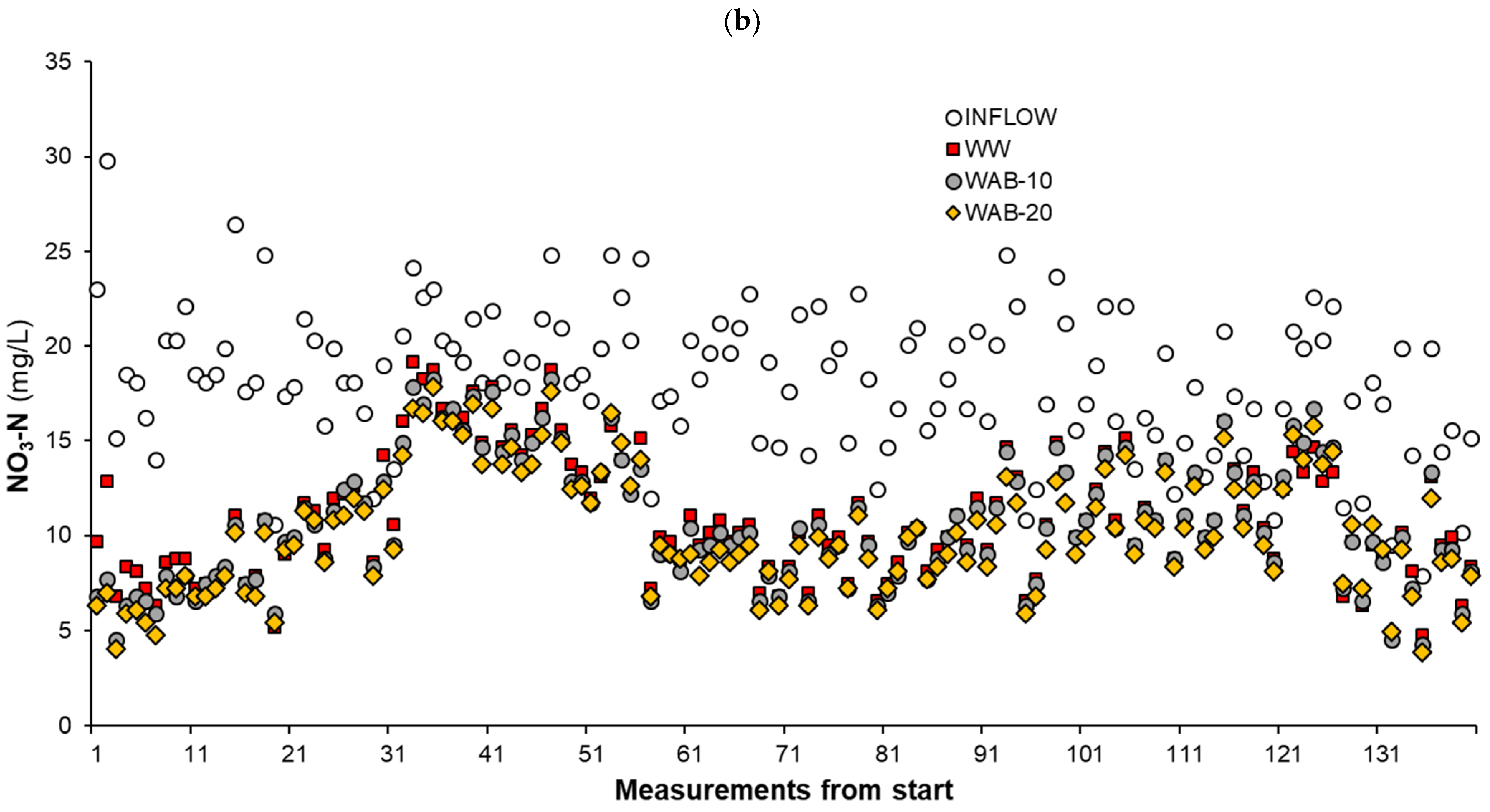
3.3. The NO3-N Removal
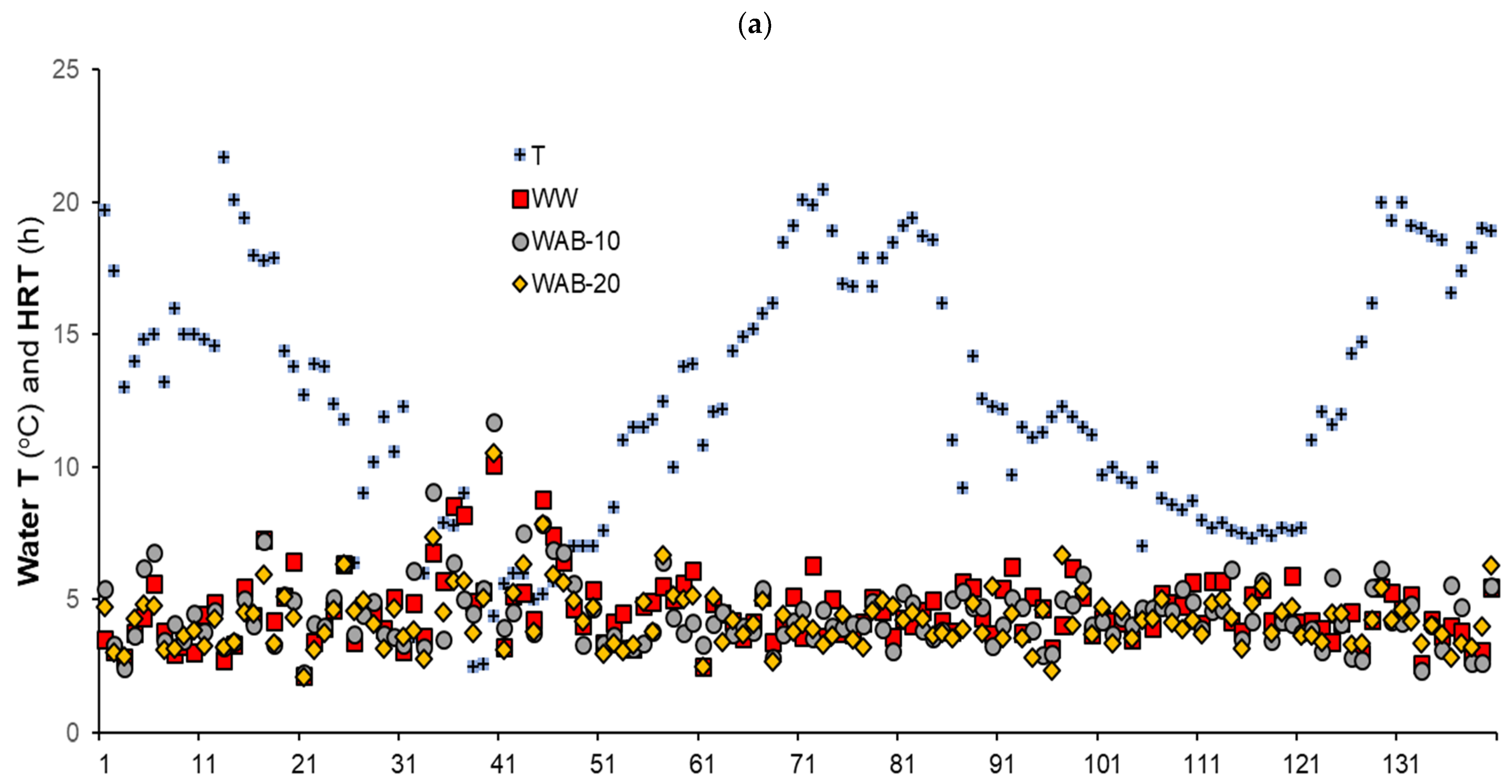
3.4. NO3-N Removal vs. Water Temperature
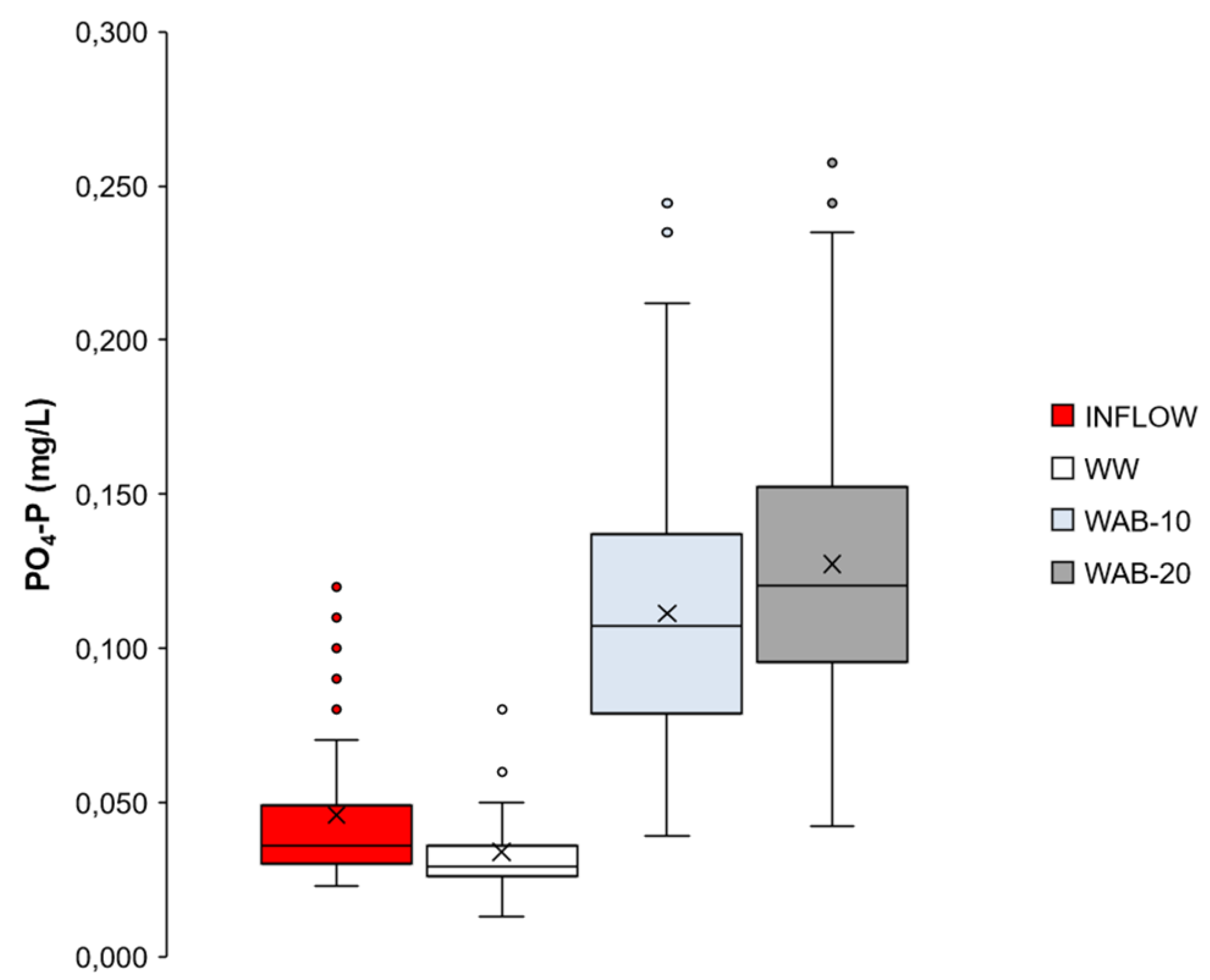
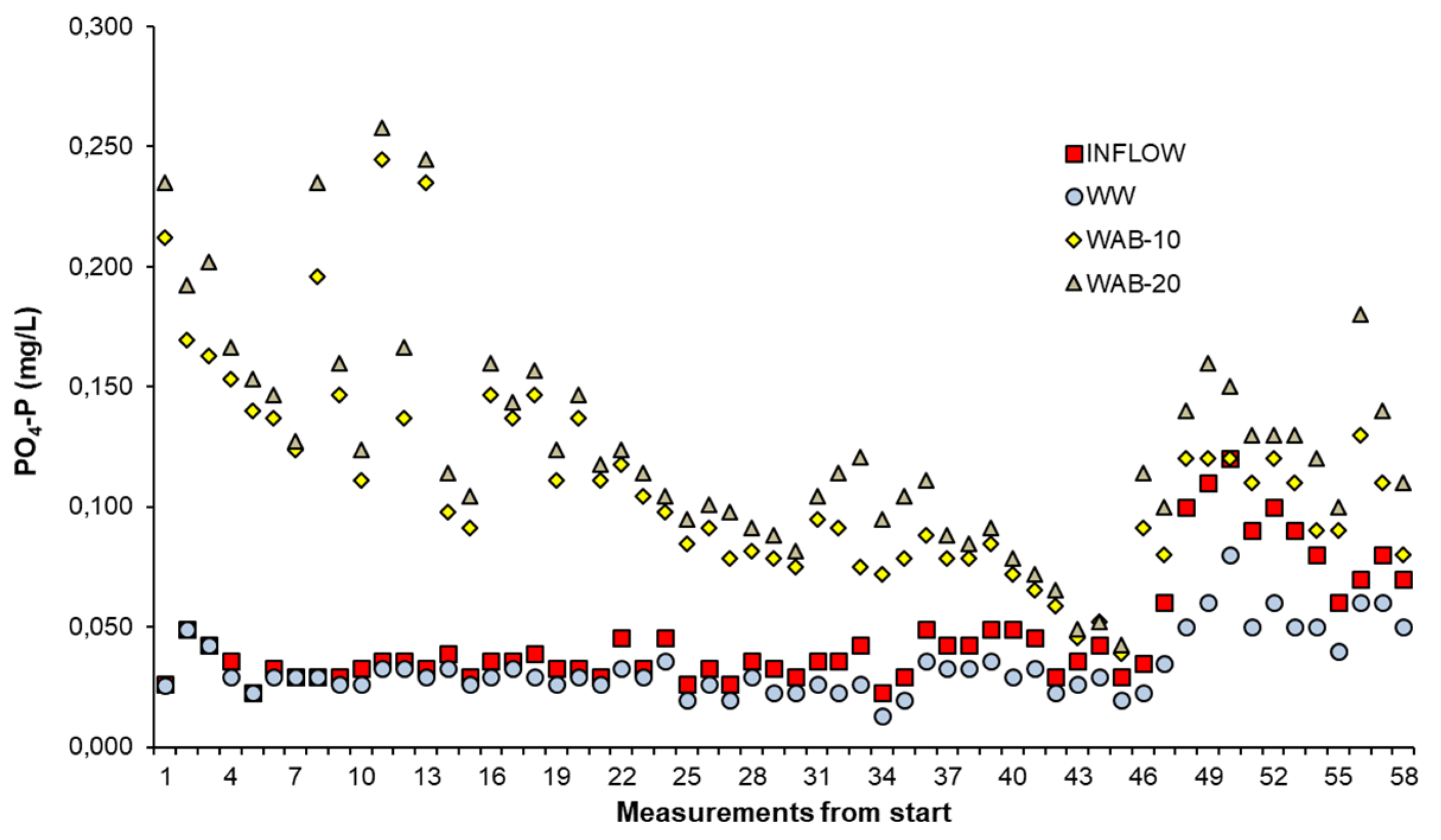
3.5. The Behavior of PO4-P
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Qi, H.; Qi, Z. Simulating phosphorus loss to subsurface tile drainage flow: A review. Environ. Rev. 2017, 25, 150–162. [Google Scholar] [CrossRef] [Green Version]
- Randall, G.W.; Goss, M.J. Nitrate losses to surface water through subsurface, tile drainage. In Nitrogen in the Environment; Academic Press: Cambridge, MA, USA, 2008; pp. 145–175. [Google Scholar]
- Povilaitis, A. Hydrological effect of artificial drainage in lowland river catchments in Lithuania. Environ. Eng. Manag. J. 2015, 14, 2243–2253. [Google Scholar] [CrossRef]
- Deelstra, J.; Iital, A.; Povilaitis, A.; Kyllmar, K.; Greipsland, I.; Blicher-Mathiesen, G.; Jansons, V.; Koskiaho, J.; Lagzdins, A. Hydrological pathways and nitrogen runoff in agricultural dominated catchments. Agric. Ecosyst. Environ. 2014, 198, 65–73. [Google Scholar] [CrossRef]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Controlling eutrophication: Nitrogen and phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Wua, Q.; Yu, P.; Zhang, L.; Ye, L.; Zhang, X.-X.; Ren, H. Denitrification using excess activated sludge as carbon source: Performance and the microbial community dynamics. Bioresour. Technol. 2017, 238, 624–632. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. More People, More Food, Worse Water? A Global Review of Water Pollution from Agriculture; FAO; IWMI: Rome, Italy, 2018. [Google Scholar]
- Addy, K.; Gold, A.J.; Christianson, L.E.; David, M.B.; Schipper, L.; Ratigan, N.A. Denitrifying Bioreactors for Nitrate Removal: A Meta-Analysis. J. Environ. Qual. 2016, 45, 873–881. [Google Scholar] [CrossRef]
- Christianson, L.E.; Cooke, R.A.; Hay, C.H.; Helmers, M.J.; Feyereisen, G.W.; Ranaivoson, A.Z.; McMaine, J.T.; McDaniel, R.; Rosen, T.R.; Pluer, W.T.; et al. Effectiveness of denitrifying bioreactors on water pollutant reduction from agricultural areas. Trans. ASABE 2021, 64, 641–658. [Google Scholar] [CrossRef]
- Rivas, A.; Barkle, G.; Stenger, R.; Moorhead, B.; Clague, J. Nitrate removal and secondary effects of a woodchip bioreactor for the treatment of subsurface drainage with dynamic flows under pastoral agriculture. Ecol. Eng. 2020, 148, 105786. [Google Scholar] [CrossRef]
- Schipper, L.A.; Robertson, W.D.; Gold, A.J.; Jaynes, D.B.; Cameron, S.C. Denitrifying bioreactors: An approach for reducing nitrate loads to receiving waters. Ecol. Eng. 2010, 36, 1532–1543. [Google Scholar] [CrossRef]
- Shah, F.A.; Mahmood, Q.; Shah, M.M.; Pervez, A.; Asad, S.A. Microbial ecology of anaerobic digesters: The key players of anaerobiosis. Sci. World J. Hindawi 2014, 2014, 183752. [Google Scholar]
- Zhang, Y.; Wanga, L.; Han, W.; Wang, X.; Guo, Z.; Peng, F.; Yang, F.; Kong, M.; Gao, Y.; Chao, J.; et al. Nitrate removal, spatiotemporal communities of denitrifiers and the importance of their genetic potential for denitrification in novel denitrifying bioreactors. Bioresour. Technol. 2017, 241, 552–562. [Google Scholar] [CrossRef]
- Jaynes, D.B.; Kaspar, T.C.; Moorman, T.B.; Parkin, T.B. In Situ Bioreactors and Deep Drain-Pipe Installation to Reduce Nitrate Losses in Artificially Drained Fields. J. Environ. Qual. 2008, 37, 429–436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lepine, C.; Christianson, L.; Sharrer, K.; Summerfelt, S. Optimizing hydraulic retention times in denitrifying woodchip bioreactors treating recirculating aquaculture system wastewater. J. Environ. Qual. 2016, 45, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Warneke, S.; Schipper, L.A.; Matiasek, M.G.; Scow, K.M.; Cameron, S.; Bruesewitz, D.A.; McDonald, I.R. Nitrate removal, communities of denitrifiers, and adverse effects in different carbon substrates for use in denitrification beds. Water Res. 2011, 45, 5463–5475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dodds, W.K.; Smith, V.H. Nitrogen, phosphorus, and eutrophication in streams. Inland Waters 2016, 6, 155–164. [Google Scholar] [CrossRef]
- Gentry, L.E.; David, M.B.; Royer, T.V.; Mitchell, C.A.; Starks, K.M. Phosphorus Transport Pathways to Streams in Tile-Drained Agricultural Watersheds. J. Environ. Qual. 2007, 36, 408–415. [Google Scholar] [CrossRef] [Green Version]
- Almanassra, I.W.; Kochkodan, V.; Mckay, G.; Atieh, M.A.; Al-Ansari, T. Review of phosphate removal from water by carbonaceous sorbents. J. Environ. Manag. 2021, 287, 112245. [Google Scholar] [CrossRef]
- Kumar, P.S.; Korving, L.; van Loosdrecht, M.C.; Witkamp, G.J. Adsorption as a technology to achieve ultra-low concentrations of phosphate: Research gaps and economic analysis. Water Res. 2019, 4, 100029. [Google Scholar] [CrossRef]
- Halaburka, B.J.; Lefevre, G.H.; Luthy, R.G. Quantifying the temperature dependence of nitrate reduction in woodchip bioreactors: Experimental and modeled results with applied case-study. Environ. Sci. Water Res. Technol. 2019, 5, 782–797. [Google Scholar] [CrossRef]
- Hassanpour, B.; Giri, S.; Pluer, W.T.; Steenhuis, T.S.; Geohring, L.D. Seasonal performance of denitrifying bioreactors in the Northeastern United States: Field trials. J. Environ. Manag. 2017, 202, 242–253. [Google Scholar] [CrossRef]
- Hoover, N.L.; Bhandari, A.; Soupir, M.L.; Moorman, T.B. Woodchip Denitrification Bioreactors: Impact of Temperature and Hydraulic Retention Time on Nitrate Removal. J. Environ. Qual. 2016, 45, 803–812. [Google Scholar] [CrossRef] [Green Version]
- Feyereisen, G.W.; Moorman, T.B.; Christianson, L.E.; Venterea, R.T.; Coulter, J.A.; Tschirner, U.W. Performance of agricultural residue media in laboratory denitrifying bioreactors at low temperatures. J. Environ. Qual. 2016, 45, 779–787. [Google Scholar] [CrossRef]
- Mazarji, M.; Aminzadeh, B.; Baghdadi, M.; Bhatnagar, A. Removal of nitrate from aqueous solution using modified granular activated carbon. J. Mol. Liq. 2017, 233, 139–148. [Google Scholar] [CrossRef] [Green Version]
- Povilaitis, A.; Matikienė, J. Nitrate removal from tile drainage water: The performance of denitrifying woodchip bioreactors amended with activated carbon and flaxseed cake. Agric. Water Manag. 2020, 229, 105937. [Google Scholar] [CrossRef]
- Weber, K.; Quicker, P. Properties of biochar. Fuel 2018, 217, 240–261. [Google Scholar] [CrossRef]
- Godlewska, P.; Schmidt, H.P.; Ok, Y.S.; Oleszczuk, P. Biochar for composting improvement and contaminants reduction. A review. Bioresour. Technol. 2017, 246, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Hassanpour, B.; Riazi, S.F.; Pluer, E.M.; Geohring, L.D.; Guzman, C.D.; Steenhuis, T.S. Biochar acting as an electron acceptor reduces nitrate removal in woodchip denitrifying bioreactors. Ecol. Eng. 2020, 149, 105724. [Google Scholar] [CrossRef]
- Povilaitis, A.; Matikienė, J.; Vismontienė, R. Effects of three types of amendments in woodchip-denitrifying bioreactors for tile drainage water treatment. Ecol. Eng. 2020, 158, 106054. [Google Scholar] [CrossRef]
- Christianson, L.; Castelló, A.L.; Christianson, L.; Helmers, M.; Bhandari, A. Hydraulic property determination of denitrifying bioreactor fill media. Appl. Eng. Agric. 2010, 26, 849–854. [Google Scholar] [CrossRef]
- Povilaitis, A.; Rudzianskaitė, A.; Misevičienė, S.; Gasiūnas, V.; Miseckaitė, O.; Živatkauskienė, I. Efficiency of drainage practices for improving water quality in Lithuania. Trans. ASABE 2018, 61, 179–196. [Google Scholar] [CrossRef] [Green Version]
- Christianson, L.E.; Lepine, C.; Sibrell, P.L.; Penn, C.; Summerfelt, S.T. Denitrifying woodchip bioreactor and phosphorus filter pairing to minimize pollution swapping. Water Res. 2017, 121, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Van Rijn, J.; Tal, Y.; Schreier, H.J. Denitrification in recirculating systems: Theory and applications. Aquac. Eng. 2006, 34, 364–376. [Google Scholar] [CrossRef]
- Oehmen, A.; Lopez-Vazquez, C.M.; Carvalho, G.; Reis, M.A.M.; Van Loosdrecht, M.C.M. Modelling the population dynamics and metabolic diversity of organisms relevant in anaerobic/anoxic/aerobic enhanced biological phosphorus removal processes. Water Res. 2010, 44, 4473–4486. [Google Scholar] [CrossRef] [PubMed]
- Azam, H.M.; Finneran, K.T. Fe (III) reduction-mediated phosphate removal as vivianite (Fe3 (PO4)2⋅ 8H2O) in septic system wastewater. Chemosphere 2014, 97, 1–9. [Google Scholar] [CrossRef]
- Wu, H.; Che, X.; Ding, Z.; Hu, X.; Creamer, A.E.; Chen, H.; Gao, B. Release of soluble elements from biochars derived from various biomass feedstocks. Environ. Sci. Pollut. Res. 2016, 23, 1905–1915. [Google Scholar] [CrossRef]
- Oliveira, F.R.; Patel, A.K.; Jaisi, D.P.; Adhikari, S.; Lu, H.; Khanal, S.K. Environmental application of biochar: Current status and perspectives. Bioresour. Technol. 2017, 246, 110–122. [Google Scholar] [CrossRef]
- Coleman, B.S.L.; Easton, Z.M.; Bock, E.M. Biochar fails to enhance nutrient removal in woodchip bioreactor columns following saturation. J. Environ. Manag. 2019, 232, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; DeLuca, T.H. Wood biochar impacts soil phosphorus dynamics and microbial communities in organically-managed croplands. Soil Biol. Biochem. 2018, 126, 144–150. [Google Scholar] [CrossRef]
- Cayuela, M.L.; Sanchez-Monedero, M.; Roig, A.; Hanley, K.; Enders, A.; Lehmann, J. Biochar and denitrification in soils: When, how much and why does biochar reduce N2O emissions? Sci. Rep. 2013, 3, 1732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weldon, S.; Rasse, D.P.; Budai, A.; Tomic, O.; Dörsch, P. The effect of a biochar temperature series on denitrification: Which biochar properties matter? Soil Biol. Biochem. 2019, 135, 173–183. [Google Scholar] [CrossRef]
- Ashoori, N.; Teixido, M.; Spahr, S.; LeFevre, G.H.; Sedlak, D.L.; Luthy, R.G. Evaluation of pilot-scale biochar-amended woodchip bioreactors to remove nitrate, metals, and trace organic contaminants from urban stormwater runoff. Water Res. 2019, 154, 1–11. [Google Scholar] [CrossRef]
- Domingues, R.R.; Trugilho, P.F.; Silva, C.A.; Melo, I.C.N.D.; Melo, L.C.; Magriotis, Z.M.; Sanchez-Monedero, M.A. Properties of biochar derived from wood and high-nutrient biomasses with the aim of agronomic and environmental benefits. PLoS ONE 2017, 12, e0176884. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosales, E.; Meijide, J.; Pazos, M.; Sanromán, M.A. Challenges and recent advances in biochar as low-cost biosorbent: From batch assays to continuous-flow systems. Bioresour. Technol. 2017, 246, 176–192. [Google Scholar] [CrossRef]
- Heaney, N.; Ukpong, E.; Lin, C. Low-molecular-weight organic acids enable biochar to immobilize nitrate. Chemosphere 2020, 240, 124872. [Google Scholar] [CrossRef] [PubMed]
- Mehrabinia, P.; Ghanbari-Adivi, E. Examining nitrate surface absorption method from polluted water using activated carbon of agricultural wastes. Modeling Earth Syst. Environ. 2021, 2121, 1–9. [Google Scholar]
- Schreiber, B.; Schmalz, V.; Brinkmann, T.; Worch, E. The effect of water temperature on the adsorption equilibrium of dissolved organic matter and atrazine on granular activated carbon. Environ. Sci. Technol. 2007, 41, 6448–6453. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.; Joseph, S. (Eds.) Biochar for Environmental Management: Science, Technology and Implementation; Routledge: London, UK, 2015; 448p. [Google Scholar] [CrossRef]
- Choi, Y.; Park, H.; Lee, M.; Lee, G.-S.; Choi, Y.-J. Seasonal variation of assimilable organic carbon and its impact to the biostability of drinking water. Environ. Eng. Res. 2018, 24, 501–512. [Google Scholar] [CrossRef]
- Porter, M.; Andrus, J.; Bartolerio, N.; Rodriguez, L.; Zhang, Y.; Zilles, J.; Kent, A. Seasonal Patterns in Microbial Community Composition in Denitrifying Bioreactors Treating Subsurface Agricultural Drainage. Microb. Ecol. 2015, 70, 710–723. [Google Scholar] [CrossRef]
- Jang, J.; Anderson, E.L.; Venterea, R.T.; Sadowsky, M.; Rosen, C.J.; Feyereisen, G.W.; Ishii, S. Denitrifying Bacteria Active in Woodchip Bioreactors at Low-Temperature Conditions. Front. Microbiol. 2019, 10, 635. [Google Scholar] [CrossRef]
- Gottschall, N.; Edwards, M.; Craiovan, E.; Frey, S.; Sunohara, M.; Ball, B.; Zoski, E.; Topp, E.; Khan, I.; Clark, I.; et al. Amending woodchip bioreactors with water treatment plant residuals to treat nitrogen, phosphorus, and veterinary antibiotic compounds in tile drainage. Ecol. Eng. 2016, 95, 852–864. [Google Scholar] [CrossRef]
- Dougherty, H. Hydraulic Evaluation of a Denitrifying Bioreactor with Baffles. Master’s Thesis, University of Illinois, Urbana, IL, USA, 2018. [Google Scholar]
- Husk, B.; Sanchez, J.; Anderson, B.; Whalen, J.; Wootton, B. Removal of phosphorus from agricultural subsurface drainage water with woodchip and mixed-media bioreactors. J. Soil Water Conserv. 2018, 73, 265–275. [Google Scholar] [CrossRef]
- Zoski, E.D.; Lapen, D.R.; Gottschall, N.; Murrell, R.S.; Schuba, B.R. Nitrogen, phosphorus, and bacteria removal in laboratory-scale woodchip bioreactors amended with drinking water treatment residuals. Trans. ASABE 2013, 56, 1339–1347. [Google Scholar]
- Mino, T.; van Loosdrecht, M.C.M.; Heijnen, J.J. Microbiology and biochemistry of the enhanced biological phosphate removal processes. Water. Res. 1998, 32, 3193–3207. [Google Scholar] [CrossRef]
- Oehmen, A.; Carvalho, G.; Freitas, F.; Reis, M.A. Assessing the abundance and activity of denitrifying polyphosphate accumulating organisms through molecular and chemical techniques. Water Sci. Technol. 2010, 61, 2061–2068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerrn-Jespersen, J.P.; Henze, M. Biological phosphorus uptake under anoxic and aerobic conditions. Water Res. 1993, 27, 617–624. [Google Scholar] [CrossRef]
- Lopez-Vazquez, C.M.; Oehmen, A.; Hooijmans, C.M.; Brdjanovic, D.; Gijzen, H.J.; Yuan, Z.; van Loosdrecht, M.C. Modeling the PAO–GAO competition: Effects of carbon source, pH and temperature. Water Res. 2009, 43, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Erdal, U.G.; Erdal, Z.K.; Randall, C.W. The competition between PAOs (phosphorus accumulating organisms) and GAOs (glycogen accumulating organisms) in EBPR (enhanced biological phosphorus removal) systems at different temperatures and the effects on system performance. Water Sci. Technol. 2003, 47, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Harris, W. Properties of dairy-manure-derived biochar pertinent to its potential use in remediation. Bioresour. Technol. 2010, 101, 5222–5228. [Google Scholar] [CrossRef]
- Qian, T.; Zhang, X.; Hu, J.; Jiang, H. Effects of environmental conditions on the release of phosphorus from biochar. Chemosphere 2013, 93, 2069–2075. [Google Scholar] [CrossRef] [PubMed]
- Silber, A.; Levkovitch, I.; Graber, E.R. pH-dependent mineral release and surface properties of cornstraw biochar: Agronomic implications. Environ. Sci. Technol. 2010, 44, 9318–9323. [Google Scholar] [CrossRef] [PubMed]
- Bock, E.; Smith, N.; Rogers, M.; Coleman, B.; Reiter, M.; Benham, B.; Easton, Z.M. Enhanced nitrate and phosphate removal in a denitrifying bioreactor with biochar. J. Environ. Qual. 2014, 44, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Laird, D.; Fleming, P.; Wang, B.; Horton, R.; Karlen, D. Biochar impact on nutrient leaching from a Midwestern agricultural soil. Geoderma 2010, 158, 436–442. [Google Scholar] [CrossRef] [Green Version]
- Buss, W.; Assavavittayanon, K.; Shepherd, J.G.; Heal, K.V.; Sohi, S. Biochar phosphorus release is limited by high pH and excess calcium. J. Environ. Qual. 2018, 47, 1298–1303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glaser, B.; Lehr, V.-I. Biochar effects on phosphorus availability in agricultural soils: A meta-analysis. Sci. Rep. 2019, 9, 9338. [Google Scholar] [CrossRef] [Green Version]
- Kameyama, K.; Miyamoto, T.; Shiono, T.; Shinogi, Y. Influence of Sugarcane Bagasse-derived Biochar Application on Nitrate Leaching in Calcaric Dark Red Soil. J. Environ. Qual. 2012, 41, 1131–1137. [Google Scholar] [CrossRef]
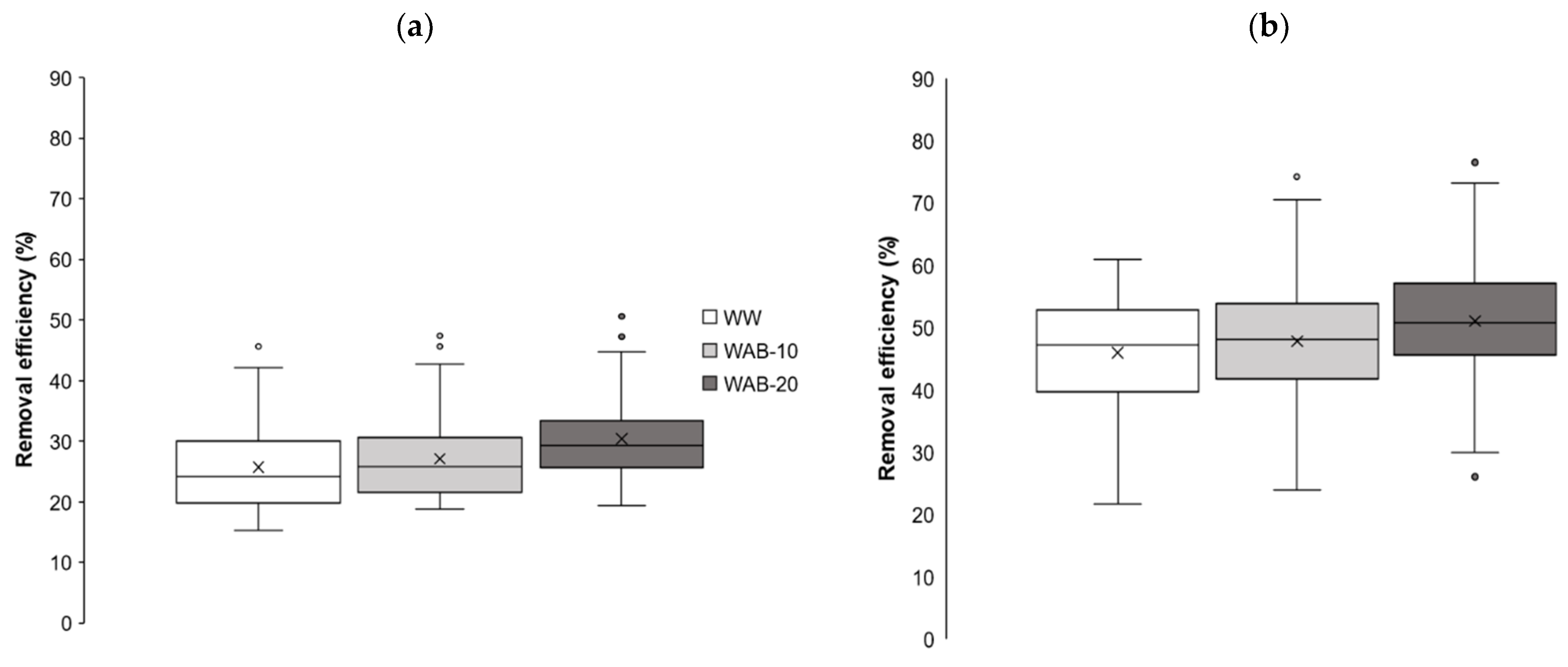
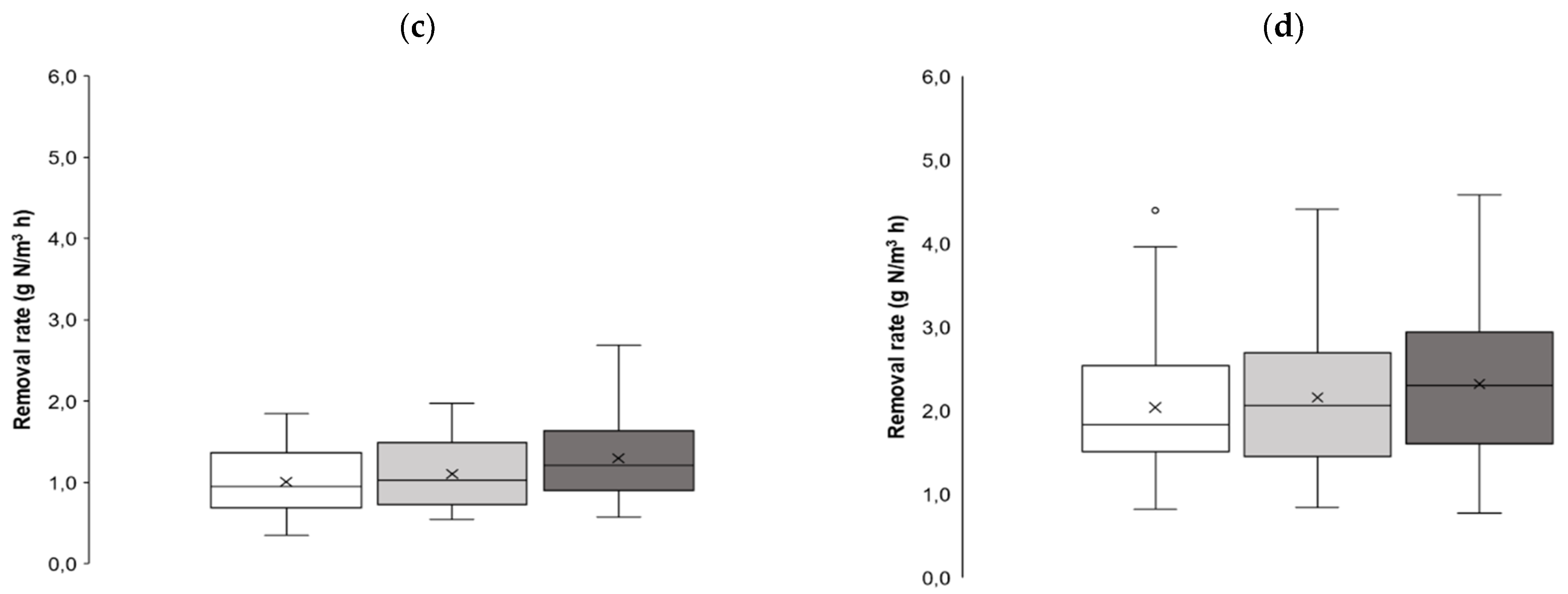
| Abbreviated Name of Bioreactor | Removal Efficiency, ** % | Removal Rates, g N/(m3·h) | Hydraulic Retention Time, h |
|---|---|---|---|
| WW | 15.3/61.0 (39.2 ± 13.2) | 0.35/4.40 (1.70 ± 0.89) | 2.13/10.1 (4.58 ± 1.27) |
| WAB-10 | 18.8/74.3 (41.0 ± 14.1) | 0.54/4.41 (1.81 ± 0.88) | 2.24/11.7 (4.45 ± 1.31) |
| WAB-20 | 19.3/76.6 (44.0 ± 13.7) | 0.58/4.58 (2.00 ± 0.95) | 2.11/10.6 (4.30 ± 1.26) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vismontienė, R.; Povilaitis, A. Effect of Biochar Amendment in Woodchip Denitrifying Bioreactors for Nitrate and Phosphate Removal in Tile Drainage Flow. Water 2021, 13, 2883. https://doi.org/10.3390/w13202883
Vismontienė R, Povilaitis A. Effect of Biochar Amendment in Woodchip Denitrifying Bioreactors for Nitrate and Phosphate Removal in Tile Drainage Flow. Water. 2021; 13(20):2883. https://doi.org/10.3390/w13202883
Chicago/Turabian StyleVismontienė, Rasa, and Arvydas Povilaitis. 2021. "Effect of Biochar Amendment in Woodchip Denitrifying Bioreactors for Nitrate and Phosphate Removal in Tile Drainage Flow" Water 13, no. 20: 2883. https://doi.org/10.3390/w13202883
APA StyleVismontienė, R., & Povilaitis, A. (2021). Effect of Biochar Amendment in Woodchip Denitrifying Bioreactors for Nitrate and Phosphate Removal in Tile Drainage Flow. Water, 13(20), 2883. https://doi.org/10.3390/w13202883






