Making Waves: Zero Liquid Discharge for Sustainable Industrial Effluent Management
Abstract
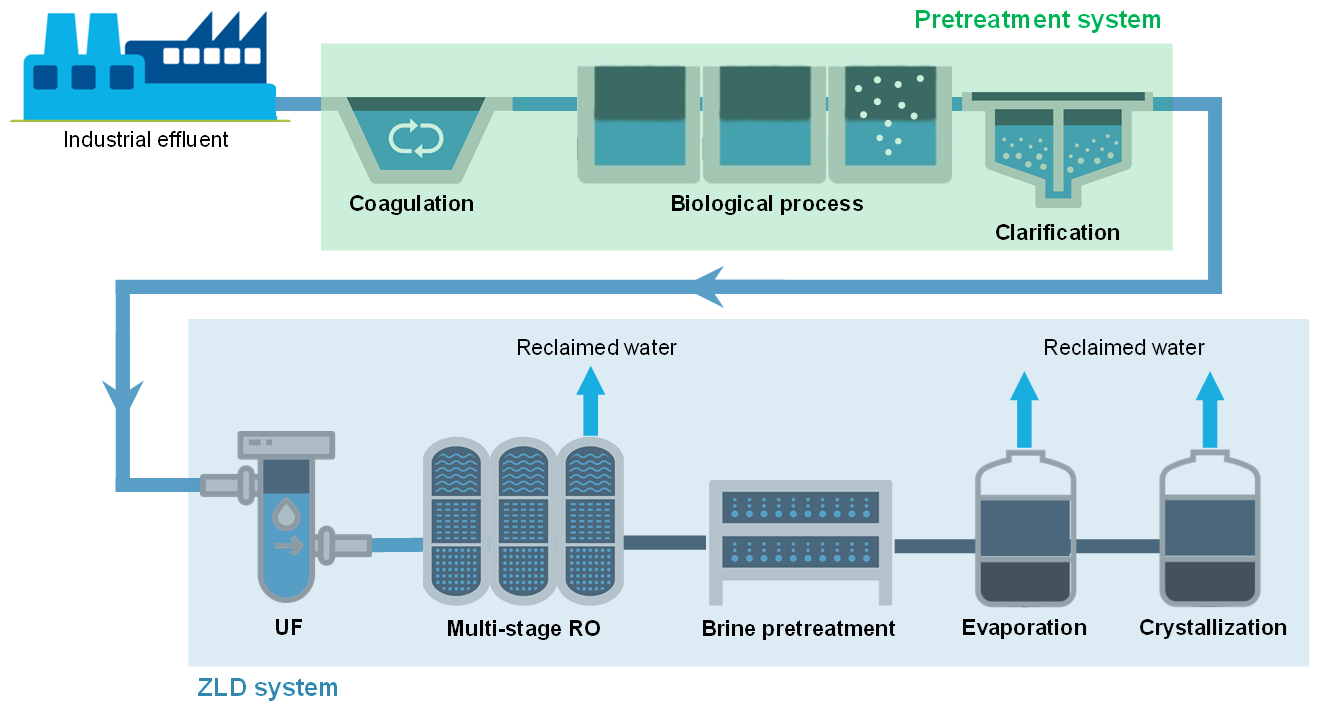
:1. Introduction
2. Abatement of Organic Pollutants in ZLD
3. Desalination and Water Recovery in ZLD
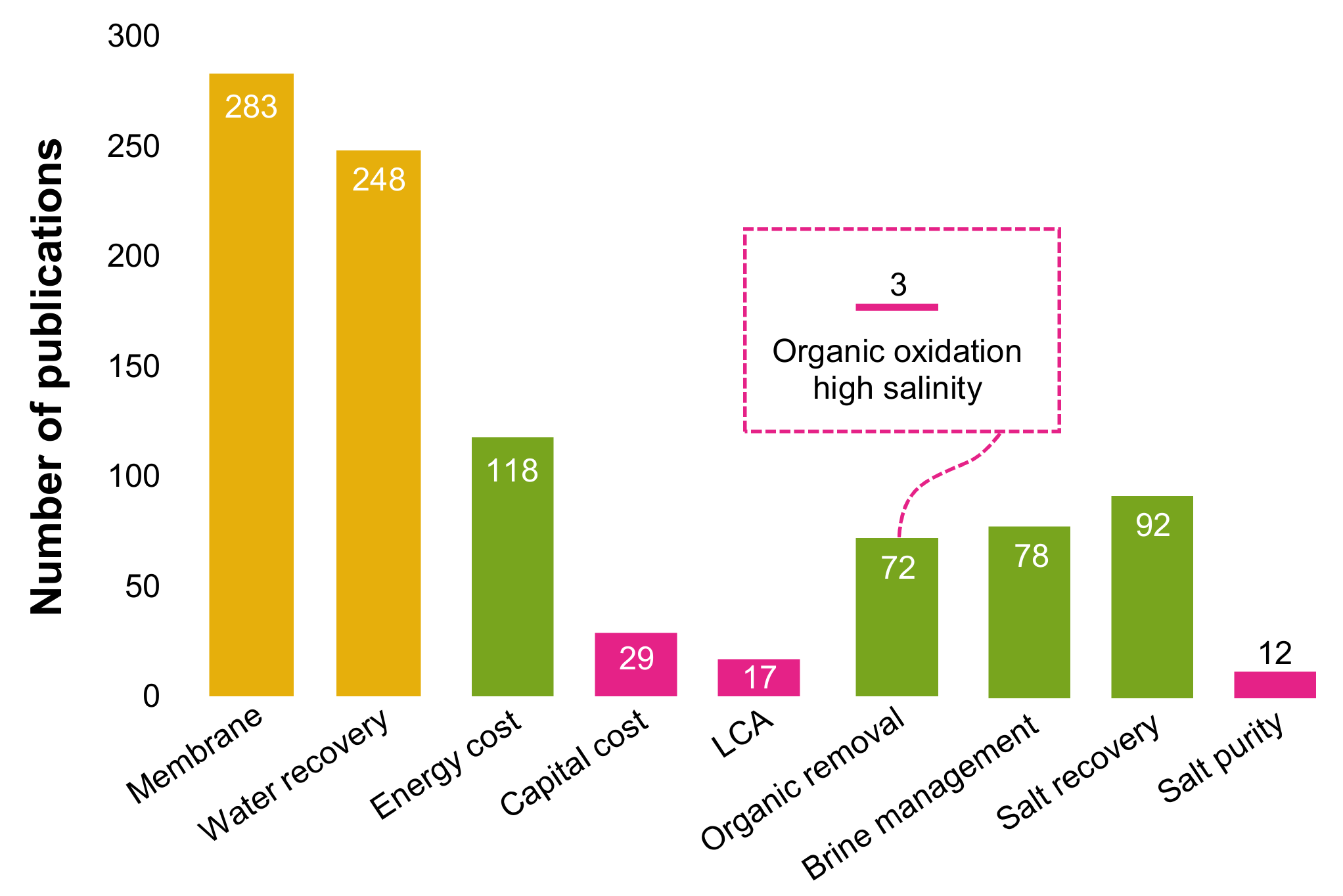
4. Challenges, Research Needs and Future Opportunities in ZLD
4.1. Are the Capital Expenditures (CAPEX) too High?
4.2. How to Improve the Removal of Organics from the RO Brine?
4.3. Salt/Ammonia Recovery: Resource or Waste?
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, J.; Ma, J.; Dai, R.; Wang, X.; Chen, M.; Waite, T.D.; Wang, Z. Self-enhanced decomplexation of Cu-organic complexes and Cu recovery from wastewaters using an electrochemical membrane filtration system. Environ. Sci. Technol. 2021, 55, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, H.; Roser, M. Water use and stress, Our World in Data. 2017. Published online at OurWorldInData.org. Available online: https://ourworldindata.org/water-use-stress (accessed on 4 September 2021).
- Tong, T.; Elimelech, M. The global rise of zero liquid discharge for wastewater management: Drivers, technologies, and future directions. Environ. Sci. Technol. 2016, 50, 6846–6855. [Google Scholar] [CrossRef]
- Yaqub, M.; Lee, W. Zero-liquid discharge (ZLD) technology for resource recovery from wastewater: A review. Sci. Total. Environ. 2019, 681, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, M.; Jensen, O. Making water reuse safe: A comparative analysis of the development of regulation and technology uptake in the US and Australia. Saf. Sci. 2020, 121, 5–14. [Google Scholar] [CrossRef]
- Panagopoulos, A.; Haralambous, K.-J. Minimal liquid discharge (MLD) and zero liquid discharge (ZLD) strategies for wastewater management and resource recovery—Analysis, challenges and prospects. J. Environ. Chem. Eng. 2020, 8, 104418. [Google Scholar] [CrossRef]
- Zhang, C.; Ma, J.; Wu, L.; Sun, J.; Wang, L.; Li, T.; Waite, T.D. Flow electrode capacitive deionization (FCDI): Recent develop-ments, environmental applications, and future perspectives. Environ. Sci. Technol. 2021, 55, 4243–4267. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, F.; Ding, N.; Hu, X.; Shen, C.; Li, F.; Huang, M.; Wang, Z.; Sand, W.; Wang, C.-C. Recent advances on electroactive CNT-based membranes for environmental applications: The perfect match of electrochemistry and membrane separation. Chin. Chem. Lett. 2020, 31, 2539–2548. [Google Scholar] [CrossRef]
- Research and Markets, Zero Liquid Discharge Systems Market Size, Share & Analysis, by System, by Technology, and by End-Use, and by Region, Forecast 2018–2028. Available online: ResearchAndMarkets.com (accessed on 4 September 2021).
- Xiong, R.; Wei, C. Current status and technology trends of zero liquid discharge at coal chemical industry in China. J. Water Process. Eng. 2017, 19, 346–351. [Google Scholar] [CrossRef]
- Mohammadtabar, F.; Khorshidi, B.; Hayatbakhsh, A.; Sadrzadeh, M. Integrated coagulation-membrane processes with zero liquid discharge (ZLD) configuration for the treatment of oil sands produced water. Water 2019, 11, 1348. [Google Scholar] [CrossRef] [Green Version]
- Semblante, G.U.; Lee, J.Z.; Lee, L.Y.; Ong, S.L.; Ng, H.Y. Brine pre-treatment technologies for zero liquid discharge systems. Desalination 2018, 441, 96–111. [Google Scholar] [CrossRef]
- Wu, Q.; Li, W.-T.; Yu, W.-H.; Li, Y.; Li, A.-M. Removal of fluorescent dissolved organic matter in biologically treated textile wastewater by ozonation-biological aerated filter. J. Taiwan Inst. Chem. Eng. 2016, 59, 359–364. [Google Scholar] [CrossRef]
- Gupta, S.K.; Gupta, S. Closed loop value chain to achieve sustainable solution for tannery effluent. J. Clean. Prod. 2018, 213, 845–846. [Google Scholar] [CrossRef]
- Lin, H.; Gao, W.; Meng, F.; Liao, B.-Q.; Leung, K.-T.; Zhao, L.; Chen, J.; Hong, H. Membrane bioreactors for industrial wastewater Treatment: A critical review. Crit. Rev. Environ. Sci. Technol. 2012, 42, 677–740. [Google Scholar] [CrossRef]
- Haberkamp, J.; Ruhl, A.S.; Ernst, M.; Jekel, M. Impact of coagulation and adsorption on DOC fractions of secondary effluent and resulting fouling behaviour in ultrafiltration. Water Res. 2007, 41, 3794–3802. [Google Scholar] [CrossRef] [PubMed]
- Breitner, L.N.; Howe, K.J.; Minakata, D. Effect of functional chemistry on the rejection of low-molecular weight neutral organics through reverse osmosis membranes for potable reuse. Environ. Sci. Technol. 2019, 53, 11401–11409. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, J.; Chen, Z.; Cheng, F. Factors controlling adsorption of recalcitrant organic contaminant from bio-treated coking wastewater using lignite activated coke and coal tar-derived activated carbon. J. Chem. Technol. Biotechnol. 2017, 93, 112–120. [Google Scholar] [CrossRef]
- Yuan, Y.; Xing, G.; Garg, S.; Ma, J.; Kong, X.; Dai, P.; Waite, T.D. Mechanistic insights into the catalytic ozonation process using iron oxide-impregnated activated carbon. Water Res. 2020, 177, 115785. [Google Scholar] [CrossRef]
- Liu, Z.-Q.; Huang, C.; Li, J.-Y.; Yang, J.; Qu, B.; Yang, S.-Q.; Cui, Y.-H.; Yan, Y.; Sun, S.; Wu, X. Activated carbon catalytic ozona-tion of reverse osmosis concentrate after coagulation pretreatment from coal gasification wastewater reclamation for zero liq-uid discharge. J. Clean. Prod. 2021, 286, 124951. [Google Scholar] [CrossRef]
- Mukhopadhyay, D. Method and Apparatus for High Efficiency Reverse Osmosis Operation, 1997. U.S. Patent No. 09/242,249. U.S. Patent and Trademark Office. Available online: https://patft.uspto.gov/netacgi/nph-Parser?Sect1=PTO1&Sect2=HITOFF&p=1&u=/netahtml/PTO/srchnum.html&r=1&f=G&l=50&d=PALL&s1=6537456.PN (accessed on 8 September 2021).
- Mickley, M. Survey of High-Recovery and Zero Liquid Discharge Technologies for Water Utilities, WateReuse Foundation 2008. Available online: https://watereuse.org/watereuse-research/02-06a-survey-of-high-recovery-and-zero-liquid-discharge-technologies-for-water-utilities/ (accessed on 8 September 2021).
- Loganathan, K.; Chelme-Ayala, P.; El-Din, M.G. Treatment of basal water using a hybrid electrodialysis reversal–reverse osmosis system combined with a low-temperature crystallizer for near-zero liquid discharge. Desalination 2015, 363, 92–98. [Google Scholar] [CrossRef]
- Oasys Water Inc. Changxing Power Plant Debuts the World’s First Forward Osmosis-Based Zero Liquid Discharge Applica-tion. Available online: https://www.wateronline.com/doc/changxing-power-plant-debuts-the-world-s-first-forward-osmosis-based-zero-liquid-discharge-application-0001 (accessed on 9 September 2021).
- Wang, Z.; Feng, D.; Chen, Y.; He, D.; Elimelech, M. Comparison of energy consumption of osmotically assisted reverse osmosis and low-salt-rejection reverse osmosis for brine management. Environ. Sci. Technol. 2021, 55, 10714–10723. [Google Scholar] [CrossRef]
- SAMCO. How much Will a Zero Liquid Discharge System Cost Your Facility. Available online: https://www.samcotech.com/how-much-will-a-zero-liquid-discharge-system-cost-your-facility/ (accessed on 9 September 2021).
- Othman, Z.A.; Linke, P.; Elhalwagi, M.M. A Systematic Approach for Targeting Zero Liquid Discharge in Industrial Parks. In Computer Aided Chemical Engineering; Elsevier BV: Amsterdam, The Netherlands, 2015; pp. 887–892. [Google Scholar]
- Mark, P. Improved Resource Recovery from Zero Liquid Discharge (ZLD) Processes Using Novel Forward Osmosis (FO) Membranes, Smart Water & Waste World, Shailesh Ramaswamy Iyer. 2019, pp. 32–33. Available online: https://www.forwardosmosistech.com/improved-resource-recovery-from-zero-liquid-discharge-zld-processes-using-novel-forward-osmosis-fo-membranes/ (accessed on 10 September 2021).
- Cui, P.; Qian, Y.; Yang, S. New water treatment index system toward zero liquid discharge for sustainable coal chemical processes. ACS Sustain. Chem. Eng. 2018, 6, 1370–1378. [Google Scholar] [CrossRef]
- Garcia-Herrero, I.; Margallo, M.; Onandía, R.; Aldaco, R.; Irabien, A. Connecting wastes to resources for clean technologies in the chlor-alkali industry: A life cycle approach. Clean Technol. Environ. Policy. 2018, 20, 229–242. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Ma, J.; Waite, T.D. The impact of absorbents on ammonia recovery in a capacitive membrane stripping system. Chem. Eng. J. 2020, 382, 122851. [Google Scholar] [CrossRef]
- Zhang, C.; Ma, J.; He, D.; Waite, T.D. Capacitive membrane stripping for ammonia recovery (CapAmm) from dilute wastewaters. Environ. Sci. Technol. Lett. 2017, 5, 43–49. [Google Scholar] [CrossRef]
- Zhang, C.; Ma, J.; Song, J.; He, C.; Waite, T.D. Continuous ammonia recovery from wastewaters using an integrated capacitive flow electrode membrane stripping system. Environ. Sci. Technol. 2018, 52, 14275–14285. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.C.; Xie, Y.; Hou, J.; Cheetham, A.K.; Chen, V.; Darling, S.B. Janus membranes: Creating asymmetry for energy efficiency. Adv. Mater. 2018, 30, 1801495. [Google Scholar] [CrossRef]
- Dongare, P.; Alabastri, A.; Pedersen, S.; Zodrow, K.R.; Hogan, N.J.; Neumann, O.; Wu, J.; Wang, T.; Deshmukh, A.; Elimelech, M.; et al. Nanophotonics-enabled solar membrane distillation for off-grid water purification. Proc. Natl. Acad. Sci. USA 2017, 114, 6936–6941. [Google Scholar] [CrossRef] [Green Version]
- Ma, J.; Ma, J.; Zhang, C.; Song, J.; Dong, W.; Waite, T.D. Flow-electrode capacitive deionization (FCDI) scale-up using a membrane stack configuration. Water Res. 2020, 168, 115186. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, G.; Vecitis, C.D. Prospects of an Electroactive Carbon Nanotube Membrane toward Environmental Applications. Accounts Chem. Res. 2020, 53, 2892–2902. [Google Scholar] [CrossRef]
- Zheng, J.; Wang, Z.; Ma, J.; Xu, S.; Wu, Z. Development of an electrochemical ceramic membrane filtration system for efficient contaminant removal from waters. Environ. Sci. Technol. 2018, 52, 4117–4126. [Google Scholar] [CrossRef]
- Zheng, J.; Ma, J.; Wang, Z.; Xu, S.; Waite, T.D.; Wu, Z. Contaminant removal from source waters using cathodic electrochemical membrane filtration: Mechanisms and implications. Environ. Sci. Technol. 2017, 51, 2757–2765. [Google Scholar] [CrossRef] [PubMed]
- Chaplin, B.P. The prospect of electrochemical technologies advancing worldwide water treatment. Accounts Chem. Res. 2019, 52, 596–604. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Y.; Lin, X.; Kong, X.; Duan, Q.; Wang, P.; Mei, X.; Ma, J. Making Waves: Zero Liquid Discharge for Sustainable Industrial Effluent Management. Water 2021, 13, 2852. https://doi.org/10.3390/w13202852
Liang Y, Lin X, Kong X, Duan Q, Wang P, Mei X, Ma J. Making Waves: Zero Liquid Discharge for Sustainable Industrial Effluent Management. Water. 2021; 13(20):2852. https://doi.org/10.3390/w13202852
Chicago/Turabian StyleLiang, Yinglin, Xin Lin, Xiangtong Kong, Qiushi Duan, Pan Wang, Xiaojie Mei, and Jinxing Ma. 2021. "Making Waves: Zero Liquid Discharge for Sustainable Industrial Effluent Management" Water 13, no. 20: 2852. https://doi.org/10.3390/w13202852
APA StyleLiang, Y., Lin, X., Kong, X., Duan, Q., Wang, P., Mei, X., & Ma, J. (2021). Making Waves: Zero Liquid Discharge for Sustainable Industrial Effluent Management. Water, 13(20), 2852. https://doi.org/10.3390/w13202852






