The Status of Arsenic Pollution in the Greek and Cyprus Environment: An Overview
Abstract
1. Introduction
2. Methodology
3. Health Effects
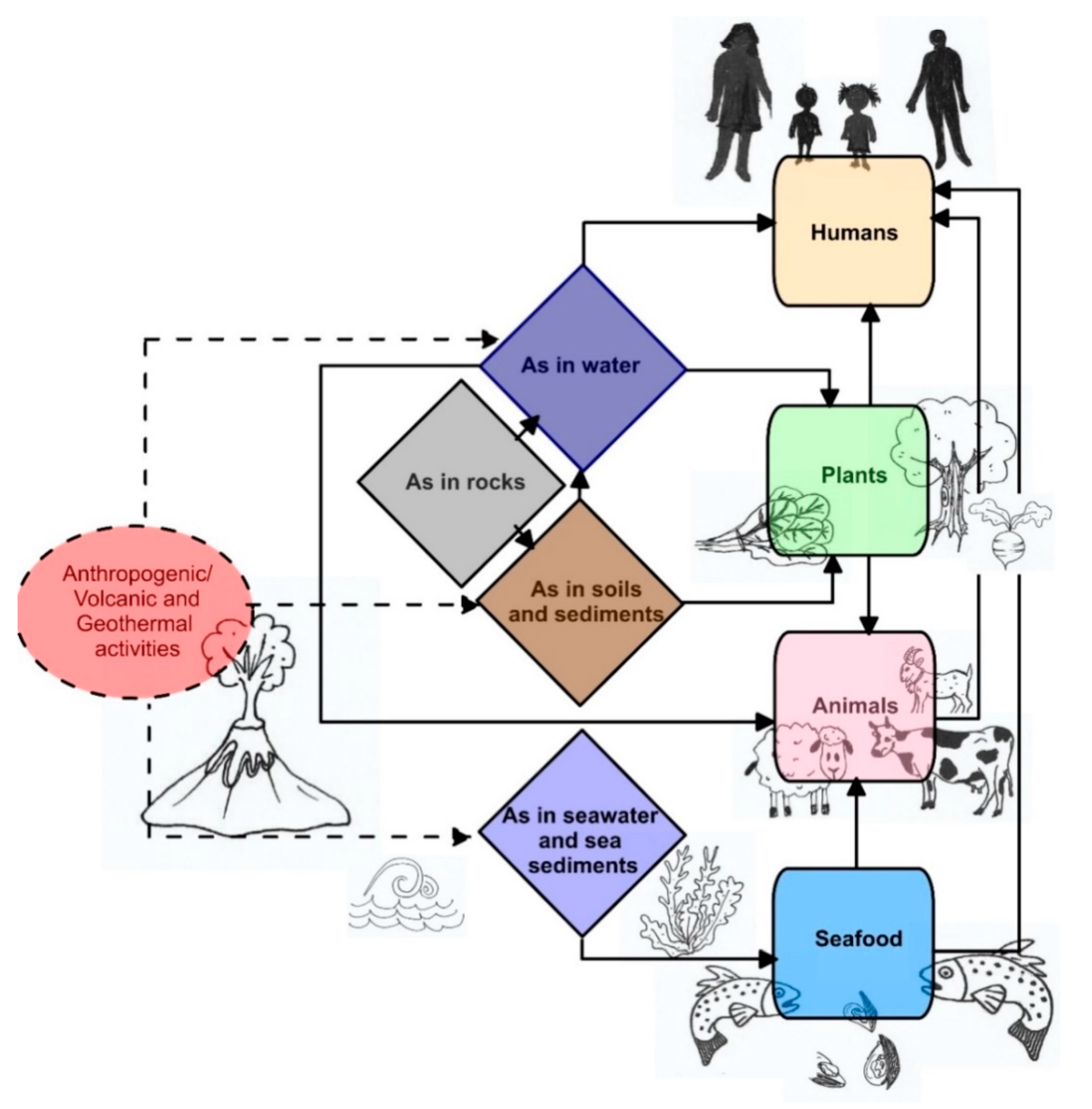
4. Accumulation
5. Transportation
6. Sources
7. Factors
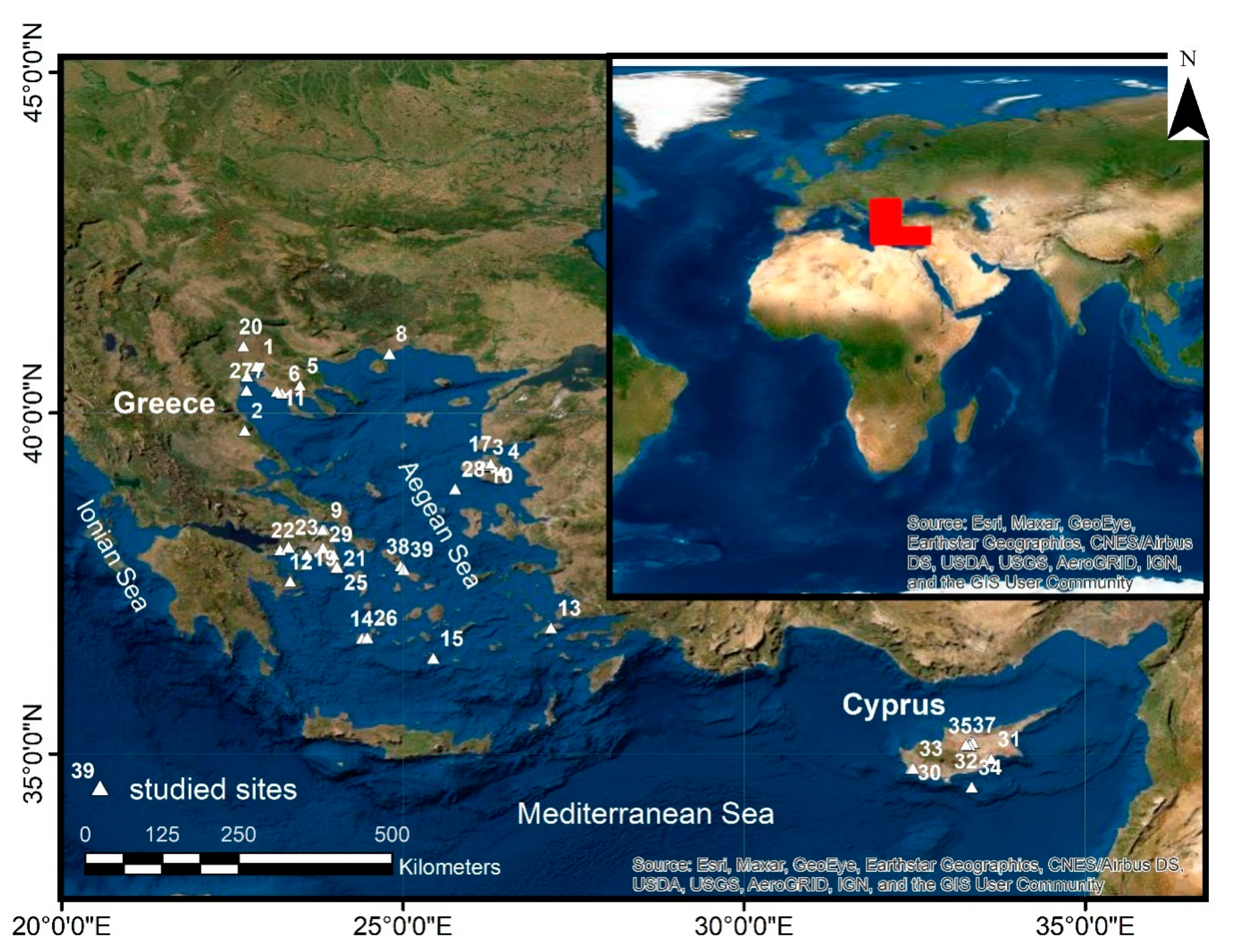
8. The Occurrence of As in Greece and Cyprus
8.1. Water
8.2. Geothermal Activities
8.3. Marine Environment
8.3.1. Marine Sediments
8.3.2. Marine Waters
8.3.3. Factors Controlling the Spatial Variability of As in Greek Submarine Hydrothermal Sediments and Waters
8.3.4. Effect of As on Bacteria Biomass
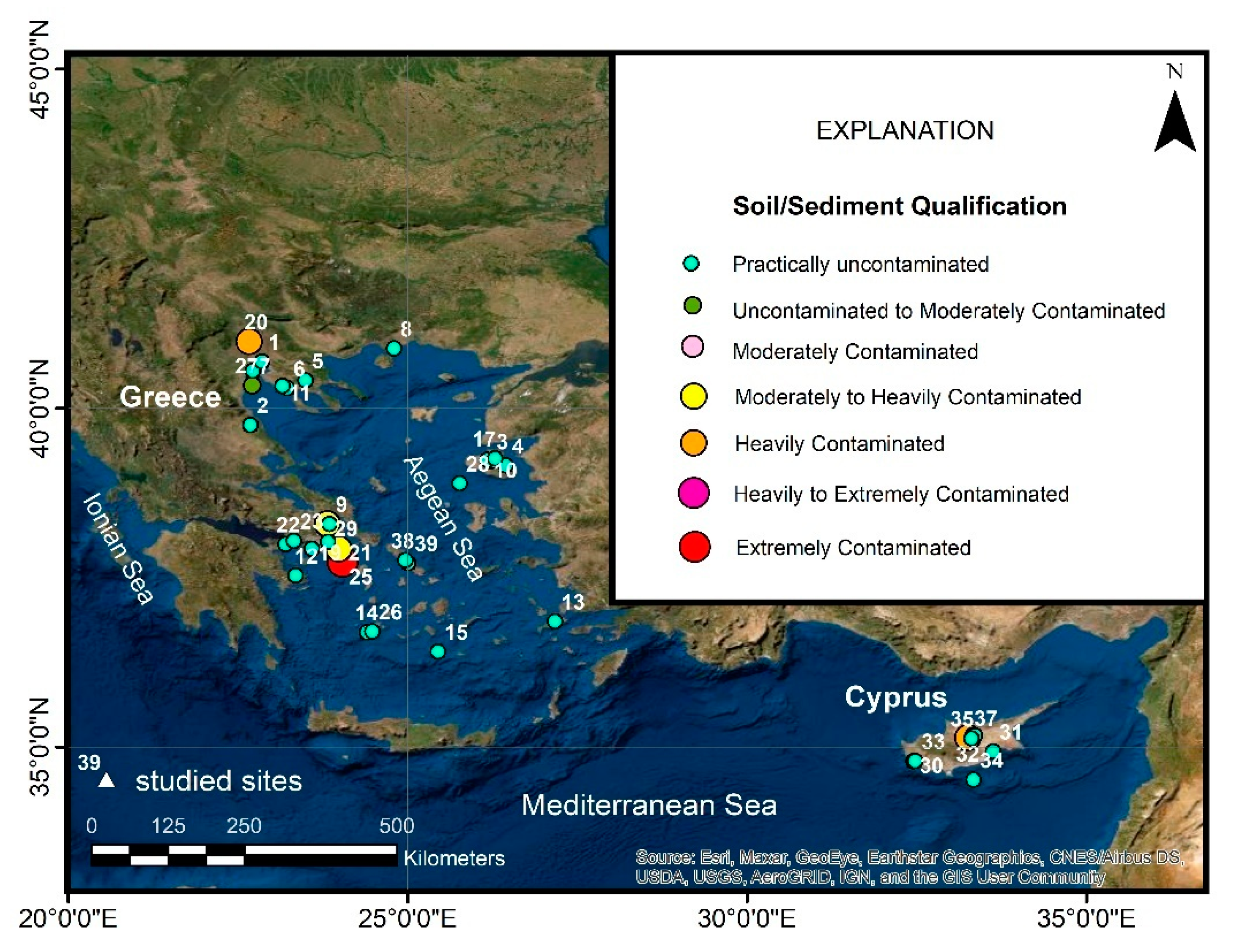
8.4. Soil
8.5. Mines
8.6. Seafood
8.7. Bottled Water
9. Igeo Assessment
10. Removal of As from Water
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kampouroglou, E.E.; Economou-Eliopoulos, M. Assessment of arsenic and associated metals in the soil-plant-water system in neogene basins of Attica, Greece. Catena 2017, 150, 206–222. [Google Scholar] [CrossRef]
- Mandal, B.K.; Suzuki, K.T. Arsenic round the world: A review. Talanta 2002, 58, 201–235. [Google Scholar] [CrossRef]
- Sanchez-Rodas, D.; de la Campa, A.M.S.; Alsioufi, L. Analytical approaches for arsenic determination in air: A critical review. Anal. Chim. Acta 2015, 898, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Arslan, B.; Djamgoz, M.B.A.; Akun, E. Arsenic: A review on exposure pathways, accumulation, mobility and transmission into the human food chain. Rev. Environ. Contam. Toxicol. 2017, 243, 27–51. [Google Scholar] [PubMed]
- Varnavas, S.P.; Salomons, W.; Förstner, U.; Kersten, M.; Brill, J.; Loizides, L.; Golik, A. Environmental contamination processes in the Limni mine area Cyprus. In Proceedings of the 6th International Conference on Contamination, Edinburgh, UK, 10–12 October 1994; Varnavas, S.P., Ed.; CEP Consultants Ltd.: Blackpool, UK, 1994; pp. 28–34. [Google Scholar]
- Varnavas, S.P.; Forstner, U.; Calmano, W. Environmental assessment and human health in a highly metal polluted coastal zone associated with toxic solid waste. The need of immediate action. In Proceedings of the 7th International Conference on Environmental Science and Technology, Ermoupolis, Greece, 3–6 September 2001; pp. 903–908. [Google Scholar]
- Christou, A.; Theologides, C.P.; Costa, C.; Kalavrouziotis, I.K.; Varnavas, S.P. Assessment of toxic heavy metals concentrations in soils and wild and cultivated plant species in Limni abandoned copper mining site, Cyprus. J. Geochem. Explor. 2017, 178, 16–22. [Google Scholar] [CrossRef]
- Hadjipanagiotou, C.; Christou, A.; Zissimos, A.M.; Hatzitheodoridis, E.; Varnavas, S.P. Contamination of stream waters, sediments, and agricultural soil in the surroundings of an abandoned copper mine by potentially toxic elements and associated and environmental and potential human health derived risks. A case study from Agrokipia, Cyprus. Environ. Sci. Pollut. Res. 2020. [Google Scholar] [CrossRef]
- Varnavas, S.P.; Panagos, A.G. Mesozoic metalliferous sediments from the ophiolites of Ermioni (Greece); analogue to recent mid-ocean ridge ferromanganese deposits. Chem. Geol. 1984, 42, 227–242. [Google Scholar] [CrossRef]
- Varnavas, S.P.; Panagos, A.G.; Philippakis, S. On the metallogenesis of the Hermioni area, Greece. Mesozonic mid-ocean ridge deposits. Geol. Carpathica 1985, 36, 219–233. [Google Scholar]
- Robertson, A.F.; Varnavas, S.P.; Panagos, A.G. Ocean ridge origin and tectonic setting of Mesozoic sulphide and oxide deposits of the Argolis Peninsula of the Peloponnesus, Greece. Sed. Geol. 1987, 53, 1–32. [Google Scholar] [CrossRef]
- Varnavas, S.P.; Panagos, A.G.; Kritsotakis, K.G. Environmental impact of mining activities on the Hermioni area Greece. In Environmental Contamination; Vernet, J.P., Ed.; Elsevier: Amsterdam, The Netherlands, 1993; pp. 119–146. [Google Scholar]
- Duman, M.; Kucuksezgin, F.; Atalar, M.; Akcali, B. Geochemistry of the northern Cyprus (NE Mediterranean) shelf sediments: Implications for anthropogenic and lithogenic impact. Mar. Pollut. Bull. 2012, 64, 2245–2250. [Google Scholar] [CrossRef]
- Muller, G. Schwermetalle in den Sedimenten des Rheins-Veranderungenseit 1971. Umschau 1979, 79, 778–783. [Google Scholar]
- Turekian, K.K.; Wedepohl, K.H. Distribution of the elements in some major units of the Earth’s crust. Geol. Soc. Am. Bull. 1961, 72, 175–192. [Google Scholar] [CrossRef]
- Kelepertsis, A.; Alexakis, D.; Skordas, K. Arsenic, antimony and other toxic elements in the drinking water of Eastern Thessaly in Greece and its possible effects on human health. Environ. Geol. 2006, 50, 76–84. [Google Scholar] [CrossRef]
- Zaspalis, V.; Pagana, A.; Sklari, S. Arsenic removal from contaminated water by iron oxide sorbents and porous ceramic membranes. Desalination 2007, 217, 167–180. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Mitrakas, M.; Zouboulis, A.I. Arsenic occurrence in Europe: Emphasis in Greece and description of the applied full-scale treatment plants. Desalin. Water Treat. 2015, 54, 2100–2107. [Google Scholar] [CrossRef]
- EC (European Community) Council. Directive 98/83/EC Directive of the European Parliament on the quality of water intended for human consumption. The European Parliament and the Council of the European Union. Off. J. 1998, L330, 32–54. [Google Scholar]
- Gamaletsos, P.; Godelitsas, A.; Dotsika, E.; Tzamos, E.; Göttlicher, J.; Filippidis, A. Geological sources of As in the environment of Greece: A review. In Threats to the Quality of Groundwater Resources: Prevention and Control; Scozzari, A., Dotsika, E., Eds.; The Handbook of Environmental Chemistry; Springer: Berlin/Heidelberg, Germany, 2013; Volume 40, pp. 77–113. [Google Scholar]
- Hudson-Edwards, K.A.; Edwards, S. Mineralogical controls on storage of As, Cu, Pb and Zn at the abandoned Mathiatis massive sulphide mine, Cyprus. Mineral. Mag. 2005, 69, 695–706. [Google Scholar] [CrossRef]
- Matschullat, J. Arsenic in the geosphere—A review. Sci. Total Environ. 2002, 249, 297–312. [Google Scholar] [CrossRef]
- Vaxevanidou, K.; Giannikou, S.; Papassiopi, N. Microbial arsenic reduction in polluted and unpolluted soils from Attica, Greece. J. Hazard. Mater. 2012, 241–242, 307–315. [Google Scholar] [CrossRef]
- Gamaletsos, P.N.; Kalatha, S.; Godelitsas, A.; Economou-Eliopoulos, M.; Göttlicher, J.; Steininger, R. Arsenic distribution and speciation in the bauxitic Fe-Ni-laterite ore deposit of the Patitira mine, Lokris area (Greece). J. Geochem. Explor. 2018, 194, 189–197. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Hug, S.J.; Ammann, A.; Zikoudi, A.; Hatziliontos, C. Arsenic speciation and uranium concentrations in drinking water supply wells in Northern Greece: Correlations with redox indicative parameters and implications for groundwater treatment. Sci. Total Environ. 2007, 383, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Zkeri, E.; Aloupi, M.; Gaganis, P. Seasonal and spatial variation of arsenic in groundwater in a rhyolithic volcanic area of Lesvos Island, Greece. Environ. Monit. Assess. 2018, 190, 44. [Google Scholar] [CrossRef] [PubMed]
- Zevenhoven, R.; Mukherjee, A.B.; Bhattacharya, P. Arsenic flows in the environment of the European Union: A synoptic review. In Arsenic in Soil and Groundwater Environment: Trace Metals and Other Contaminants in the Environment; Bhattacharya, P., Mukherjee, A.B., Bundschuh, J., Zevenhoven, R., Loeppert, R.H., Eds.; Elsevier: Amsterdan, The Netherlands, 2007; Volume 9, pp. 527–547. [Google Scholar]
- Devesa, V.; Velez, D.; Montoro, R. Effect of thermal treatments on arsenic species contents in food. Food Chem. Toxicol. 2008, 46, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Abdul, K.S.; Jayasinghe, S.S.; Chandana, E.P.; Jayasumana, C.; De Silva, P.M. Arsenic and human health effects: A Review. Environ. Toxicol. Pharmacol. 2015, 40, 828–846. [Google Scholar] [CrossRef]
- Chung, J.Y.; Yu, S.D.; Hong, Y.S. Environmental source of arsenic exposure. J. Prev. Med. Public Health 2014, 47, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Anawar, H.M.; Akai, J.; Mihaljevič, M.; Sikder, A.M.; Ahmed, G.; Tareq, S.M.; Rahman, M.M. Arsenic contamination in groundwater of Bangladesh: Perspectives on geochemical, microbial and anthropogenic issues. Water 2011, 3, 1050–1076. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Mukherjee, A.B.; Bundschuh, J.; Zevenhoven, R.; Loeppert, R.H. (Eds.) Arsenic in Soil and Groundwater Environment. Biogeochemical Interactions, Health Effects and Remediation, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Chakraborti, D.; Singh, S.K.; Rahman, M.M.; Dutta, R.N.; Mukherjee, S.C.; Pati, S.; Kar, P.B. Groundwater arsenic contamination in the Ganga River basin: A future health danger. J. Environ. Res. Public Health 2018, 15, 180. [Google Scholar] [CrossRef] [PubMed]
- Medunić, G.; Fiket, Ž.; Ivanić, M. Arsenic contamination status in Europe, Australia, and other parts of the World. In Arsenic in Drinking Water and Food, 1st ed.; Srivastava, S., Ed.; Springer: Singapore, 2020. [Google Scholar]
- Nicomel, N.R.; Leus, K.; Folens, K.; Van Der Voort, P.; Du Laing, G. Technologies for arsenic removal from water: Current status and future perspectives. J. Environ. Res. Public Health 2016, 13, 62. [Google Scholar] [CrossRef]
- Handley, K.M.; Boothman, C.; Mills, R.A.; Pancost, R.D.; Lloyd, J.R. Functional diversity of bacteria in a ferruginous hydrothermal sediment. ISME J. 2010, 4, 1193–1205. [Google Scholar] [CrossRef]
- Casentini, B.; Hug, S.J.; Nikolaidis, N.P. Arsenic accumulation in irrigated agricultural soils in Northern Greece. Sci. Total Environ. 2011, 409, 4802–4810. [Google Scholar] [CrossRef]
- Thakur, J.K.; Thakur, R.K.; Ramanathan, A.; Kumar, M.; Singh, S.K. Arsenic contamination of groundwater in Nepal—An overview. Water 2011, 3, 1–20. [Google Scholar] [CrossRef]
- Cheng, R.C.; Liang, S.; Wang, H.C.; Beuhler, M.D. Enhanced coagulation for arsenic removal. J. AWWA 1994, 86, 79–90. [Google Scholar] [CrossRef]
- Ferguson, J.F.; Gavis, J. A review of the arsenic cycle in natural waters. Water Res. 1972, 6, 1259–1274. [Google Scholar] [CrossRef]
- Hering, J.G.; Chiu, V.Q. Arsenic occurrence and speciation in municipal groundwater based supply system. J. Environ. Eng. 2000, 126, 800–807. [Google Scholar] [CrossRef]
- Korte, N.E.; Fernando, Q. A review of arsenic (III) in groundwater. Crit. Rev. Environ. Sci. Technol. 1991, 21, 1–39. [Google Scholar] [CrossRef]
- Falcó, G.; Llobet, J.M.; Bocio, A.; Domingo, J.L. Daily Intake of Arsenic, Cadmium, Mercury, and Lead by Consumption of Edible Marine Species. J. Agric. Food Chem. 2006, 54, 6106–6112. [Google Scholar] [CrossRef]
- Varol, M.; Kaya, G.K.; Sünbül, M.R. Evaluation of health risks from exposure to arsenic and heavy metals through consumption of ten fish species. Environ. Sci. Pollut. Res. 2019, 26, 33311–33320. [Google Scholar] [CrossRef]
- Storelli, M.M.; Giacominelli-Stuffler, R.; Storelli, A.; Marcotrigiano, G.O. Accumulation of mercury, cadmium, lead and arsenic in swordfish and bluefin tuna from the Mediterranean Sea: A comparative study. Mar. Pollut. Bull. 2005, 50, 993–1018. [Google Scholar] [CrossRef]
- Kampouroglou, E.E.; Economou-Eliopoulos, M. Assessment of the environmental impact by As and heavy metals in lacustrine travertine limestone and soil in Attica, Greece: Mapping of potentially contaminated sites. Catena 2016, 139, 137–166. [Google Scholar] [CrossRef]
- Zkeri, E.; Aloupi, M.; Gaganis, P. Natural occurrence of arsenic in groundwater from Lesvos island, Greece. Water Air Soil Pollut. 2015, 226, 294. [Google Scholar] [CrossRef]
- Price, R.E.; Savon, I.; Planer-Friedrich, B.; Bühring, S.I.; Amend, J.; Pichler, T. Processes influencing extreme As enrichment in shallow-sea hydrothermal fluids of Milos Island, Greece. Chem. Geol. 2013, 348, 15–26. [Google Scholar] [CrossRef]
- Singh, R.; Singh, S.; Parihar, P.; Singh, V.P.; Prasad, S.M. Arsenic contamination, consequences and remediation techniques: A review. Ecotoxicol. Environ. Saf. 2015, 112, 247–270. [Google Scholar] [CrossRef] [PubMed]
- Vimercati, L.; Baldassarre, A.; Gatti, M.F.; Gagliardi, T.; Serinelli, M.; De Maria, L.; Caputi, A.; Dirodi, A.A.; Galise, I.; Cuccaro, F.; et al. Non-occupational exposure to heavy metals of the residents of an industrial area and biomonitoring. Environ. Monit. Assess. 2016, 188, 673. [Google Scholar] [CrossRef] [PubMed]
- Vimercati, L.; Gatti, M.F.; Gagliardi, T.; Cuccaro, F.; De Maria, L.; Caputi, A.; Quarato, M.; Baldassarre, A. Environmental exposure to arsenic and chromium in an industrial area. Environ. Sci. Pollut. Res. Int. 2017, 24, 11528–11535. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, I.; Daponte, A.; Gil, F.; Hernández, A.F.; Godoy, P.; Pla, A.; Ramos, J.L.; DASAHU Group. Urinary levels of arsenic and heavy metals in children and adolescents living in the industrialised area of Ria of Huelva (SW Spain). Environ. Int. 2010, 36, 563–569. [Google Scholar] [CrossRef]
- Khlifi, R.; Olmedo, P.; Gil, F.; Feki-Tounsi, M.; Hammami, B.; Rebai, A.; Hamza-Chaffai, A. Biomonitoring of cadmium, chromium, nickel and arsenic in general population living near mining and active industrial areas in Southern Tunisia. Environ. Monit. Assess. 2014, 186, 761–779. [Google Scholar] [CrossRef]
- Aloupi, M.; Angelidis, M.O.; Gavriil, A.M.; Koulousaris, M.; Varnavas, S. Influence of geology on arsenic concentrations in ground and surface water in central Lesvos, Greece. Environ. Monit. Assess. 2009, 151, 383–396. [Google Scholar] [CrossRef]
- Christodoulidou, M.; Charalambous, C.; Aletrari, M.; Nicolaidou Kanari, P.; Petronda, A.; Ward, N.I. Arsenic concentrations in groundwaters of Cyprus. J. Hydrol. 2012, 468–469, 94–100. [Google Scholar] [CrossRef]
- Google Earth. Greece, Eye Alt 1116.55 m, World Imagery. Available online: https://earth.google.com/web (accessed on 26 May 2020).
- UN (United Nations). Available online: https://www.un.org/sustainabledevelopment/sustainabledevelopment-goals/ (accessed on 7 June 2020).
- Stamatis, G.; Alexakis, D.; Gamvroula, D.; Migiros, G. Groundwater quality assessment in Oropos-Kalamos basin, Attica, Greece. Environ. Earth Sci. 2011, 64, 973–988. [Google Scholar] [CrossRef]
- Varnavas, S.P.; Cronan, D.S. Arsenic, antimony and bismuth in sediments and waters from the Santorini hydrothermal field, Greece. Chem. Geol. 1988, 67, 295–305. [Google Scholar] [CrossRef]
- Alexakis, D. Meta-Evaluation of water quality indices. Application into groundwater resources. Water 2020, 12, 1890. [Google Scholar] [CrossRef]
- Akter, T.; Jhohura, F.T.; Akter, F.; Chowdhury, T.R.; Mistry, S.K.; Dey, D.; Barua, M.K.; Islam, M.A.; Rahman, M. Water Quality Index for measuring drinking water quality in rural Bangladesh: A cross sectional study. J. Health Popul. Nutr. 2016, 35, 4. [Google Scholar] [CrossRef] [PubMed]
- Alexakis, D.; Gamvroula, D. Arsenic, chromium, and other potentially toxic elements in the rocks and sediments of Oropos-Kalamos Basin, Attica, Greece. Appl. Environ. Soil Sci. 2014, 718534. [Google Scholar] [CrossRef]
- Tzoraki, O.; Dokou, Z.; Christodoulou, G.; Gaganis, P.; Karatzas, G. Assessing the efficiency of a coastal Managed Aquifer Recharge (MAR) system in Cyprus. Sci. Total. Environ. 2018, 626, 875–886. [Google Scholar] [CrossRef]
- Tyrovola, K.; Nikolaidis, N.P.; Veranis, N.; Kallithrakas-Kontos, N.; Koulouridakis, P.E. Arsenic removal from geothermal waters with zero-valent iron—Effect of temperature, phosphate and nitrate. Water Res. 2006, 40, 2375–2386. [Google Scholar] [CrossRef]
- Ruiz-Chancho, M.J.; Pichler, T.; Price, R.E. Arsenic occurrence and speciation in Cyclopeneritea, a gastropod inhabiting the arsenic-rich marine shallow-water hydrothermal system off Milos Island, Greece. Chem. Geol. 2013, 348, 56–64. [Google Scholar] [CrossRef]
- Angelidis, M.; Grimanis, A.P. Arsenic geochemistry in sediments near the Athens sewage outfall. Mar. Pollut. Bull. 1987, 18, 247–298. [Google Scholar] [CrossRef]
- Varnavas, S.P.; Cronan, D.S. Hydrothermal metallogenic processes off the islands of Nisiros and Kos in the Hellenic Volcanic Arc. Mar. Geol. 1991, 99, 109–133. [Google Scholar] [CrossRef]
- Varnavas, S.P.; Papavasiliou, C. Submarine hydrothermal mineralization processes and insular mineralization in the Hellenic Volcanic Arc system: A review. Ore Geol. Rev. 2020, 124, 103541. [Google Scholar] [CrossRef]
- Varnavas, S.P.; Cronan, D.S. Submarine hydrothermal activity off Santorini and Milos in the Central Hellenic Volcanic Arc: A synthesis. Chem. Geol. 2005, 224, 40–54. [Google Scholar] [CrossRef]
- Cronan, D.S.; Varnavas, S.P.; Hodkinson, R. Hydrothermal mineralizing processes and associated sedimentation in the Santorini hydrothermal embayments. Mar. Georesour. Geotechnol. 2000, 18, 77–118. [Google Scholar] [CrossRef]
- Hubner, A.; Rahders, E.; Rahner, S.; Halbach, P.; Varnavas, S.P. Geochemistry of hydrothermally influenced sediments of Methana (western Hellenic volcanic arc). Geochemistry 2004, 64, 75–94. [Google Scholar] [CrossRef]
- Cronan, D.S.; Varnavas, S.P. Metalliferous sediments off Milos, Hellenic Volcanic Arc. Explor. Min. Geol. 1999, 8, 289–297. [Google Scholar]
- Onishi, H. Arsenic. In Handbook of Geochemistry; Wedepohl, K.H., Ed.; Springer: Berlin, Germany, 1970; Volume 2, pp. 3301–3305. [Google Scholar]
- Dando, P.R.; Hughes, J.A.; Thiermann, F. Preliminary observations on biological communities at shallow hydrothermal vents in the Aegean Sea. In Hydrothermal Vents and Processes; Parson, L.M., Walker, C.L., Dixon, D.R., Eds.; Geological Society Special Publication: London, UK, 1995; Volume 87, pp. 303–317. [Google Scholar]
- Alexakis, D. Diagnosis of stream sediment quality and assessment of toxic element contamination sources in East Attica, Greece. Environ. Earth Sci. 2011, 63, 1369–1383. [Google Scholar] [CrossRef]
- Akun, M.E.; Yamacı, R.F.; Charalambous, C.; Lechtvich, S.; Djamgoz, M.B. The distribution of carcinogenic heavy metals in Cyprus soil. In Survival and Sustainability. Environmental Earth Sciences; Gökçekus, H., Türker, U., LaMoreaux, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 353–359. [Google Scholar]
- Zissimos, A.M.; Cohen, D.R.; Christoforou, I.C. Land use influences on soil geochemistry in Lefkosia (Nicosia) Cyprus. J. Geochem. Explor. 2018, 187, 6–20. [Google Scholar] [CrossRef]
- Alexakis, D.; Gamvroula, D.; Theofili, E. Environmental availability of potentially toxic elements in an agricultural Mediterranean site. Environ. Eng. Geosci. 2019, 25, 169–178. [Google Scholar] [CrossRef]
- Alexakis, D. Suburban areas in flames: Dispersion of potentially toxic elements from burned vegetation and buildings. Estimation of the associated ecological and human health risk. Environ. Res. 2020, 183, 109153. [Google Scholar] [CrossRef]
- Alexakis, D. Contaminated land by wildfire effect on ultramafic soil and associated human health and ecological risk. Land 2020, 9, 409. [Google Scholar] [CrossRef]
- Kampouroglou, E.; Economou-Eliopoulos, M. Natural contamination by As and heavy metals in soil, their bio-accumulation and potential sources: The case of a travertine limestone quarry, Greece. Cent. Eur. J. Geosci. 2013, 5, 174–188. [Google Scholar] [CrossRef]
- Stylianou, M.; Tsiftes, K.; Gavriel, I.; Kostarelos, K.; Demetriou, C.; Papaioannou, A. Environmental impacts of abandoned sulphide mines—The example of Mathiatis Mine in Cyprus. In Proceedings of the e-Symbiosis International Conference, Athens, Greece, 19–21 June 2014. [Google Scholar]
- Lortzie, K.; Stylianou, M.; Kostarelos, K. Assessment of environmental pollution at the abandoned gold-silver enrichment plant in Mitsero, Cyprus. In Proceedings of the 13th International Conference on Environmental Science and Technology, Athens, Greece, 5–7 September 2013. [Google Scholar]
- Antivachis, D. The geology of the northern part of the Αpliki Cyprus-type ore deposit. Bull. Geol. Soc. Greece 2015, 49, 4–28. [Google Scholar] [CrossRef][Green Version]
- Charalambous, A. A diachronic study of Cypriot copper alloy artefacts. J. Archaeol. Sci. Rep. 2016, 7, 566–573. [Google Scholar] [CrossRef]
- Schaeffer, R.; Soeroes, C.; Ipolyi, I.; Fodor, P.; Thomaidis, N.S. Determination of arsenic species in seafood samples from the Aegean Sea by liquid chromatography–(photo-oxidation)–hydride generation–atomic fluorescence spectrometry. Anal. Chim. Acta 2005, 547, 109–118. [Google Scholar] [CrossRef]
- Pell, Α.; Kokkinis, G.; Malea, P.; Pergantis, S.A.; Rubio, R.; López-Sánchez, J.F. LC–ICP–MS analysis of arsenic compounds in dominant seaweeds from the Thermaikos Gulf (Northern Aegean Sea, Greece). Chemosphere 2013, 93, 2187–2194. [Google Scholar] [CrossRef] [PubMed]
- Sazakli, E.; Zouvelou, S.V.; Kalavrouziotis, I.; Leotsinidis, M. Arsenic and antimony removal from drinking water by adsorption on granular ferric oxide. Water Sci. Technol. 2015, 71, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Figoli, A.; Fuoco, I.; Apollaro, C.; Chabane, M.; Mancuso, R.; Gabriele, B.; De Rosa, R.; Vespasiano, G.; Barca, D.; Criscuoli, A. Arsenic-contaminated groundwaters remediation by nanofiltration. Sep. Purif. Technol. 2020, 238, 116461. [Google Scholar] [CrossRef]
- Figoli, A.; Fuoco, I.; Apollaro, C.; Mancuso, R.; Desiderio, G.; De Rosa, R.; Gabriele, B.; Criscuoli, A. Arsenic removal from natural contaminated groundwaters in Calabria Region (Italy) by Nanofiltration. In Environmental Arsenic in a Changing World (As 2018), Proceedings of the 7th International Congress and Exhibition Arsenic in the Environment, Beijing, China, 1–6 July 2018; Zhu, Y., Guo, H., Bhattacharya, P., Ahmad, A., Bundschuh, J., Naidu, R., Eds.; CRC Press Taylor and Francis Group: London, UK, 2018; pp. 1–5. [Google Scholar]
- Azamat, J.; Khataee, A.; Sadikoglu, F. Computational study on the efficiency of MoS2 membrane for removing arsenic from contaminated water. J. Mol. Liq. 2018, 249, 110–116. [Google Scholar] [CrossRef]
- Elson, C.M.; Davies, D.H.; Hayes, E.R. Removal of arsenic from contaminated drinking water by a chitosan/chitin mixture. Water Res. 1980, 14, 1307–1311. [Google Scholar] [CrossRef]
- Garrido Hoyos, S.E.; Avilés Flores, M.; Ramírez Gonzalez, A.; Grajeda Fajardo, C.; Cardoso Zoloeta, S.; Velásquez Orozco, H. Comparing two operating configurations in a full-scale arsenic removal plant. Case study: Guatemala. Water 2013, 5, 834–851. [Google Scholar] [CrossRef]
- McBean, E.A.; Rajib, M.A.; Rahman, M.M. Improved sustainability of water supply options in areas with arsenic-impacted groundwater. Water 2013, 5, 1941–1951. [Google Scholar] [CrossRef]
- Ojemaye, M.O.; Okoh, O.O.; Okoh, A.I. Uptake of Zn2+ and As3+ from wastewater by adsorption onto imine functionalized magnetic nanoparticles. Water 2018, 10, 36. [Google Scholar] [CrossRef]
- Liang, T.; Li, L.; Zhu, C.; Liu, X.; Li, H.; Su, Q.; Ye, J.; Geng, B.; Tian, Y.; Sardar, M.F.; et al. Adsorption of As(V) by the novel and efficient adsorbent cerium-manganese modified biochar. Water 2020, 12, 2720. [Google Scholar] [CrossRef]
- Larroca, F.P.; Olschewski, E.S.; Quino-Favero, J.; Rosales Huamaní, J.; Castillo Sequera, J.-L. Water treatment plant prototype with pH control modeled on fuzzy logic for removing arsenic using Fe(VI) and Fe(III). Water 2020, 12, 2834. [Google Scholar] [CrossRef]
- Xu, X.; Lin, L.; Papelis, C.; Xu, P. Sorption of arsenic from desalination concentrate onto drinking water treatment solids: Operating conditions and kinetics. Water 2018, 10, 96. [Google Scholar] [CrossRef]
- Khan, S.U.; Farooqi, I.H.; Usman, M.; Basheer, F. Energy efficient rapid removal of arsenic in an electrocoagulation reactor with hybrid Fe/Al electrodes: Process optimization using CCD and kinetic modeling. Water 2020, 12, 2876. [Google Scholar] [CrossRef]
- Bae, J.; Kim, S.; Kim, K.S.; Hwang, H.-K.; Choi, H. Adsorptive removal of arsenic by mesoporous iron oxide in aquatic systems. Water 2020, 12, 3147. [Google Scholar] [CrossRef]
- Nam, G.; Kim, M.-S.; Lee, N.; Choi, Y.-H.; Ahn, J.-W. An environmentally benign approach for As (V) absorption from wastewater using untreated coffee grounds—Preliminary results. Water 2017, 9, 867. [Google Scholar] [CrossRef]
- Serrano, J.; Leiva, E. Removal of arsenic using acid/metal-tolerant sulfate reducing bacteria: A new approach for bioremediation of high-arsenic acid mine waters. Water 2017, 9, 994. [Google Scholar] [CrossRef]
- Alkhudhiri, A.; Hakami, M.; Zacharof, M.-P.; Homod, H.A.; Alsadun, A. Mercury, arsenic and lead removal by air gap membrane distillation: Experimental study. Water 2020, 12, 1574. [Google Scholar] [CrossRef]
- Ogata, F.; Nagai, N.; Toda, M.; Otani, M.; Saenjum, C.; Nakamura, T.; Kawasaki, N. Removal of arsenic (III) ion from aqueous media using complex nickel-aluminum and nickel-aluminum-zirconium hydroxides. Water 2020, 12, 1697. [Google Scholar] [CrossRef]
| Medium | Country | Concentration |
|---|---|---|
| Drinking water | Argentina | 0.3–0.8 μg L−1 |
| Drinking water | Northern Chile (period 1958–1970) | 860 mg L−1 |
| Groundwater | Eastern Croatia | 1.3–491 μg L−1 |
| Groundwater | Argentina | 133–305 μg L−1 |
| Groundwater | China, Huhhot Basin | >1500 mg L−1 |
| Rural wells | Argentina | 25–76 μg L−1 |
| Urban wells | Argentina | 31–357 μg L−1 |
| Wells (48 %) | Vietnam | >50 μg L−1 |
| Wells (45 %) | India (West Bengal) | >50 μg L−1 |
| Wells | Bangladesh | <0.5 to 3200 μg L−1 |
| Surface water | Argentina | 11–133 μg L−1 |
| Medium | Location Number (as Shown in Figure 2) | Location Name | Sampling Period | Detection Method (DL) | Concentration | Ref. |
|---|---|---|---|---|---|---|
| Groundwater | 1 | Thessaloniki | 2016 | HGAFS | >70 μg L−1 | [25] |
| Groundwater | 2 | Eastern Thessaly | 2003–2004 | AAS-MHS | 1–125 μg L−1 | [16] |
| Groundwater | 3 | Lesvos Island | 2005 | HGAAS-GFAAS (0.7 μg L−1 for dissolved As, 0.1 μg L−1 for particulate As) | <0.7–88.3 μg L−1 | [54] |
| Groundwater | 4 | Mandamados Lesvos island, | 2010–2011 | ICP-MS (0.1 μg L−1) | 1.78–54.7 μg L−1 | [26] |
| Groundwater (for irrigation use) | 5 | Chalkidiki, Northern Greece | n.d. | n.d. | >130 μg L−1 | [54] |
| Groundwater (for irrigation use) | 6 | Nea Triglia, Chalkidiki, Northern Greece | 2008–2009 | ICP-MS | >1000 μg L−1 | [37] |
| Groundwater | 7 | Axios basin, Northern Greece | n.d. | n.d. | 20–45 μg L−1 | [18] |
| Groundwater | 8 | Nestos basin, Northern Greece | n.d. | n.d. | 15–20 μg L−1 | [18] |
| Groundwater | 9 | Oropos–Kalamos basin, North Attica | 2008 | ICP-MS (0.5 μg L−1) | 0.5–246.5 μg L−1 | [58] |
| Stream water | 10 | Lesvos Island, Kalloni Gulf | 2005 | HG-AAS (0.7 μg L−1 for dissolved, 0.1 μg L−1 for particulate As) | 0.4–13.2 μg L−1 | [54] |
| Geothermal groundwater | 11 | Chalkidiki | n.d. | n.d. | >2000 μg L−1 | [18] |
| Geothermal water | 12 | Methana island | n.d. | n.d. | >20.5 mg L−1 | [18] |
| Geothermal water | 13 | Kos island | n.d. | n.d. | 56 mg L−1 | [18] |
| Geothermal water | 14 | Milos island | n.d. | n.d. | 108 mg L−1 | [18] |
| Geothermal water | 15 | Santorini island | n.d. | n.d. | 493 mg L−1 | [18] |
| Thermal springs water | 16 | Kalloni Gulf, Lesvos island | 2005 | HG-AAS (0.7 μg L−1 for dissolved, 0.1 μg L−1 for particulate As) | 0.4–13.2 μg L−1 | [54] |
| Stream sediment | 17 | Kalloni Gulf, Lesvos island | 2005 | HG-AAS | 2.0–22 mg Kg−1 | [54] |
| Stream sediment | 18 | East Attica | 2006–2007 | ICP-MS (1 mg Kg−1) | 8–273 mg Kg−1 | [75] |
| Sediment | 19 | Northern Saronicos Gulf | 1981 | INAA | 17–25 mg Kg−1 | [66] |
| Soil | 20 | Northern Greece | 2008–2009 | ICP-MS | 5–520 mg Kg−1 | [37] |
| Garden soil | 21 | Lavrion, Attica | n.d. | n.d. | >14,800 mg Kg−1 | [2] |
| Wildfire ash | 22 | Kineta, West Attica | 2018 | ICP-AES (2 mg Kg−1) | 4–16 mg Kg−1 | [79] |
| Soil | 23 | Megara basin | 2010 | ICP-MS (5 mg Kg−1) | 5–18 mg Kg−1 | [78] |
| Bulk lignite | 24 | Oropos-Kalamos basin, Attica | 2008 | ICP-MS (0.1 mg Kg−1) | 226.2 mg Kg−1 | [62] |
| House dust | 25 | Lavrion, Attica | n.d. | >3800 mg Kg−1 | [2] | |
| Groundwater | 26 | Milos island | 2009–2010 | HR-ICP-MS (8–155 ng L−1) | 2955–5850 μg L−1 | [65] |
| Seaweed species | 27 | Thermaikos Gulf | 2007 | LC–ICP–MS [0.01 mg Kg−1 for As(III), 0.03 mg Kg−1 for As(V)] | 1.39–55.0 mg Kg−1 | [87] |
| Seafood | 28 | Aegean Sea | n.d. | ICP-TOFMS (0.385 mg Kg−1) | 2.8–34 mg Kg−1 | [86] |
| Bottled water | 29 | Greece | n.d. | n.d. | <10 μg L−1 | [18] |
| Groundwater | 30 | Paphos, Cyprus | 2007–2009 | ICP-MS (0.3 μg L−1) | 26 μg L−1 | [55] |
| Groundwater | 31 | Larnaca, Cyprus | 2007–2009 | ICP-MS (0.3 μg L−1) | 12–13 μg L−1 | [55] |
| Groundwater | 32 | Lefkosia, Cyprus | 2007–2009 | ICP-MS (0.3 μg L−1) | 23–41 μg L−1 | [55] |
| Groundwater | 33 | Paphos, Cyprus | 2006–2011 | n.d. | 1.3 μg L−1 | [63] |
| Sea sediment | 34 | Cicilian basin, Cyprus | 2011 | ICP-MS | 3.3–55.4 mg Kg−1 | [13] |
| Soil | 35 | Cyprus | n.d. | n.d. | 0.2–22.5 mg Kg−1 | [76] |
| Soil | 36 | Lefkosia, Cyprus | n.d. | ICP-MS (2 mg Kg−1) | < 2–41.8 mg Kg−1 | [77] |
| Soil | 37 | Mitsero, Lefkosia, Cyprus | 2012 | ICP-OES | 390 mg Kg−1 | [83] |
| Wildfire impacted soil | 38 | Central Evia, Greece | 2019 | ICP-AES (2 mg Kg−1) | 3–10 mg Kg−1 | [80] |
| Wildfire ash | 39 | Central Evia, Greece | 2019 | ICP-AES (2 mg Kg−1) | 2–23 mg Kg−1 | [80] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golfinopoulos, S.K.; Varnavas, S.P.; Alexakis, D.E. The Status of Arsenic Pollution in the Greek and Cyprus Environment: An Overview. Water 2021, 13, 224. https://doi.org/10.3390/w13020224
Golfinopoulos SK, Varnavas SP, Alexakis DE. The Status of Arsenic Pollution in the Greek and Cyprus Environment: An Overview. Water. 2021; 13(2):224. https://doi.org/10.3390/w13020224
Chicago/Turabian StyleGolfinopoulos, Spyros K., Soterios P. Varnavas, and Dimitrios E. Alexakis. 2021. "The Status of Arsenic Pollution in the Greek and Cyprus Environment: An Overview" Water 13, no. 2: 224. https://doi.org/10.3390/w13020224
APA StyleGolfinopoulos, S. K., Varnavas, S. P., & Alexakis, D. E. (2021). The Status of Arsenic Pollution in the Greek and Cyprus Environment: An Overview. Water, 13(2), 224. https://doi.org/10.3390/w13020224







