Characteristics of Hydrochemistry and Stable Isotopes in a Karst Region in Samcheok, Republic of Korea
Abstract
1. Introduction
2. Study Site and Methods
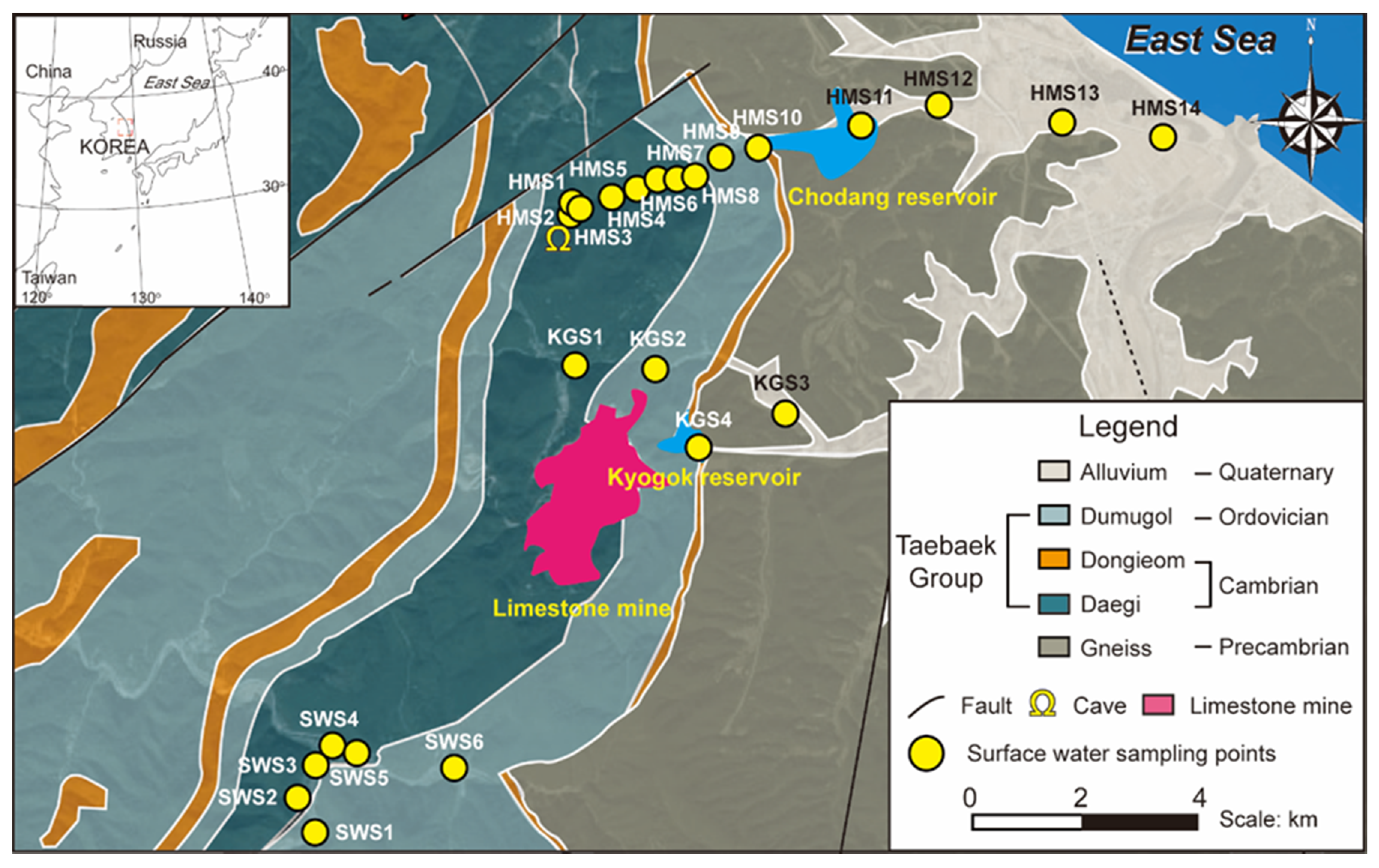
2.1. Study Site
2.1.1. Geologic Setting
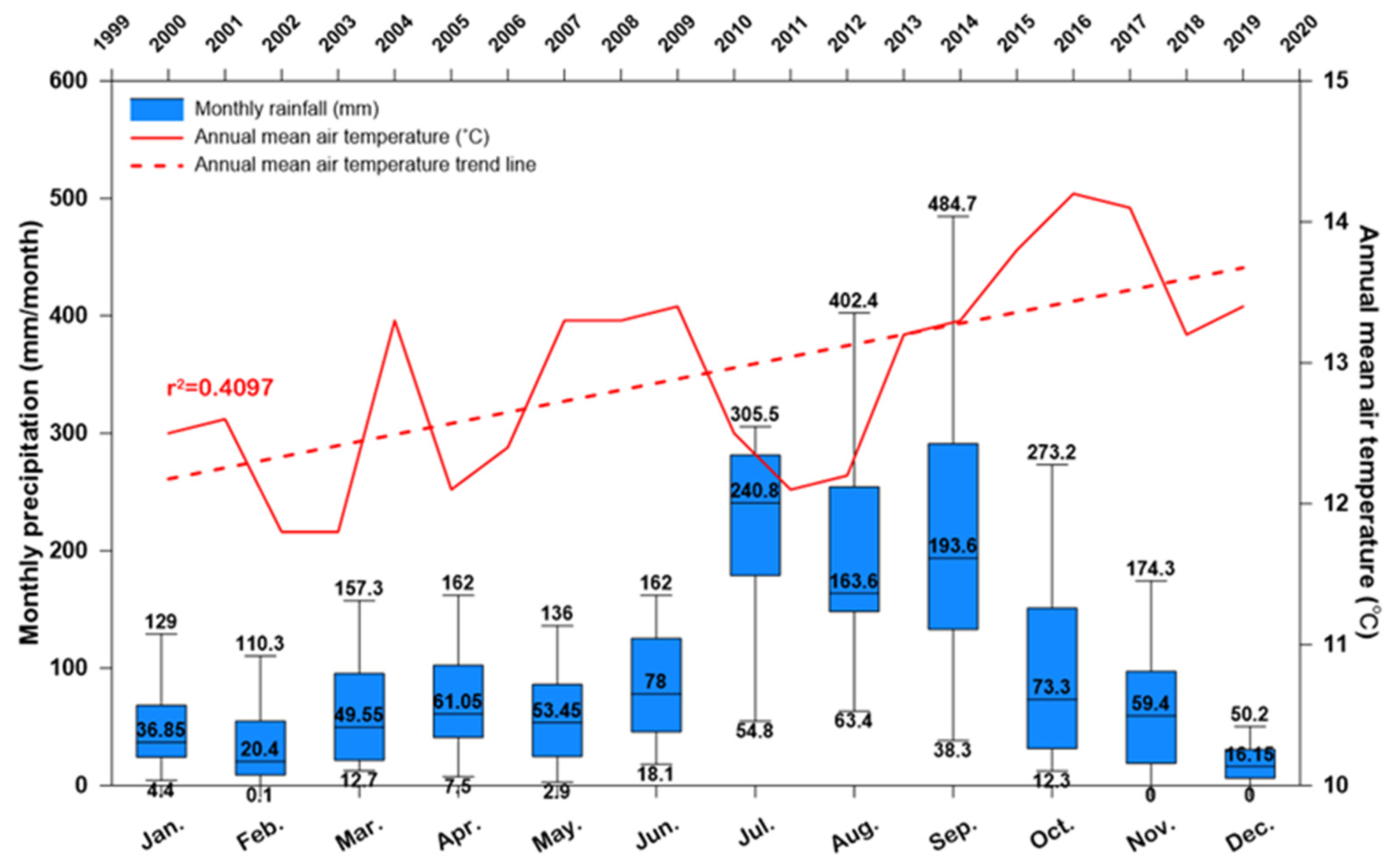
2.1.2. Climate
2.2. Methods
3. Results and Discussion
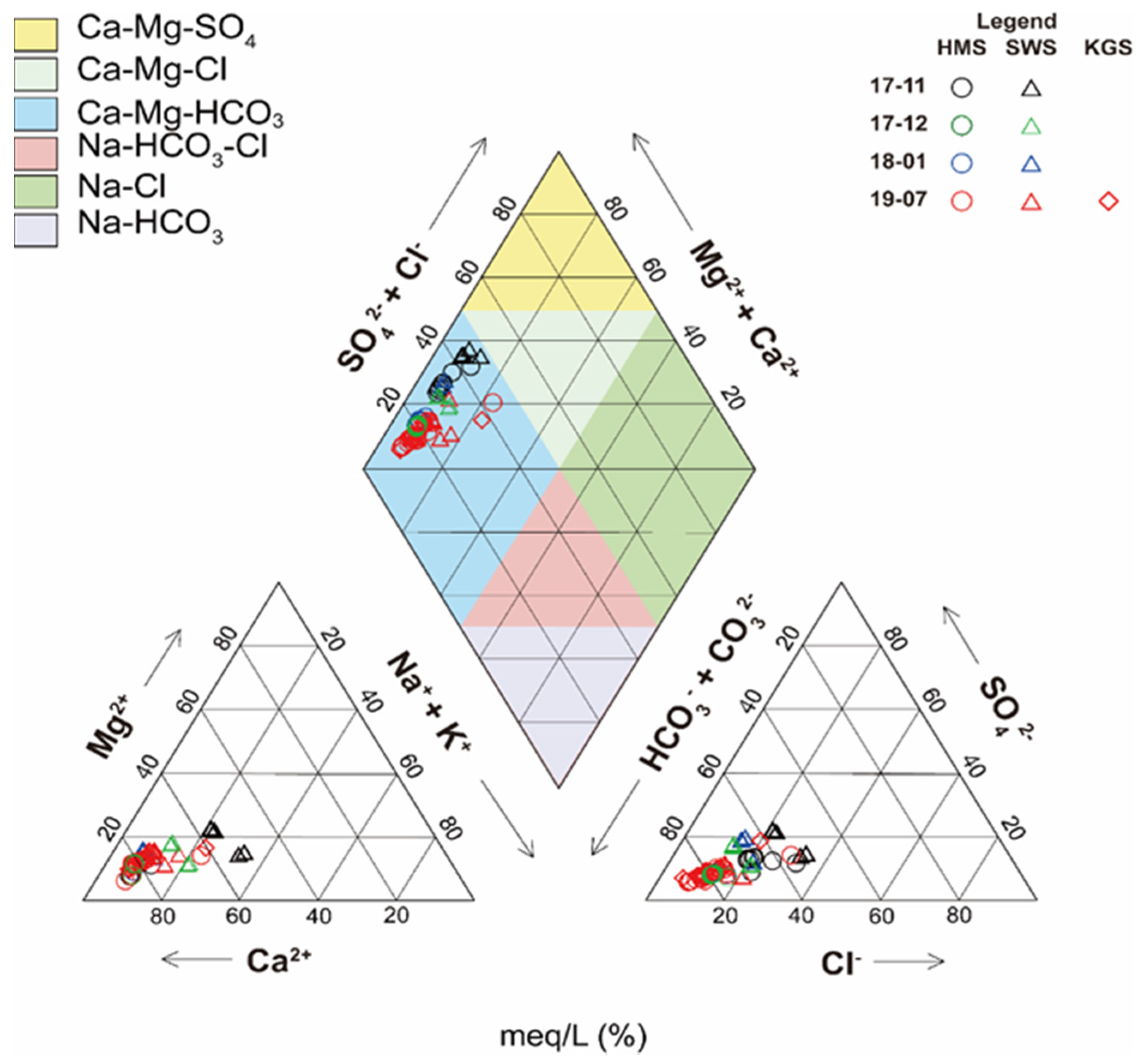
3.1. Hydrochemistry
3.2. Correlation Analysis
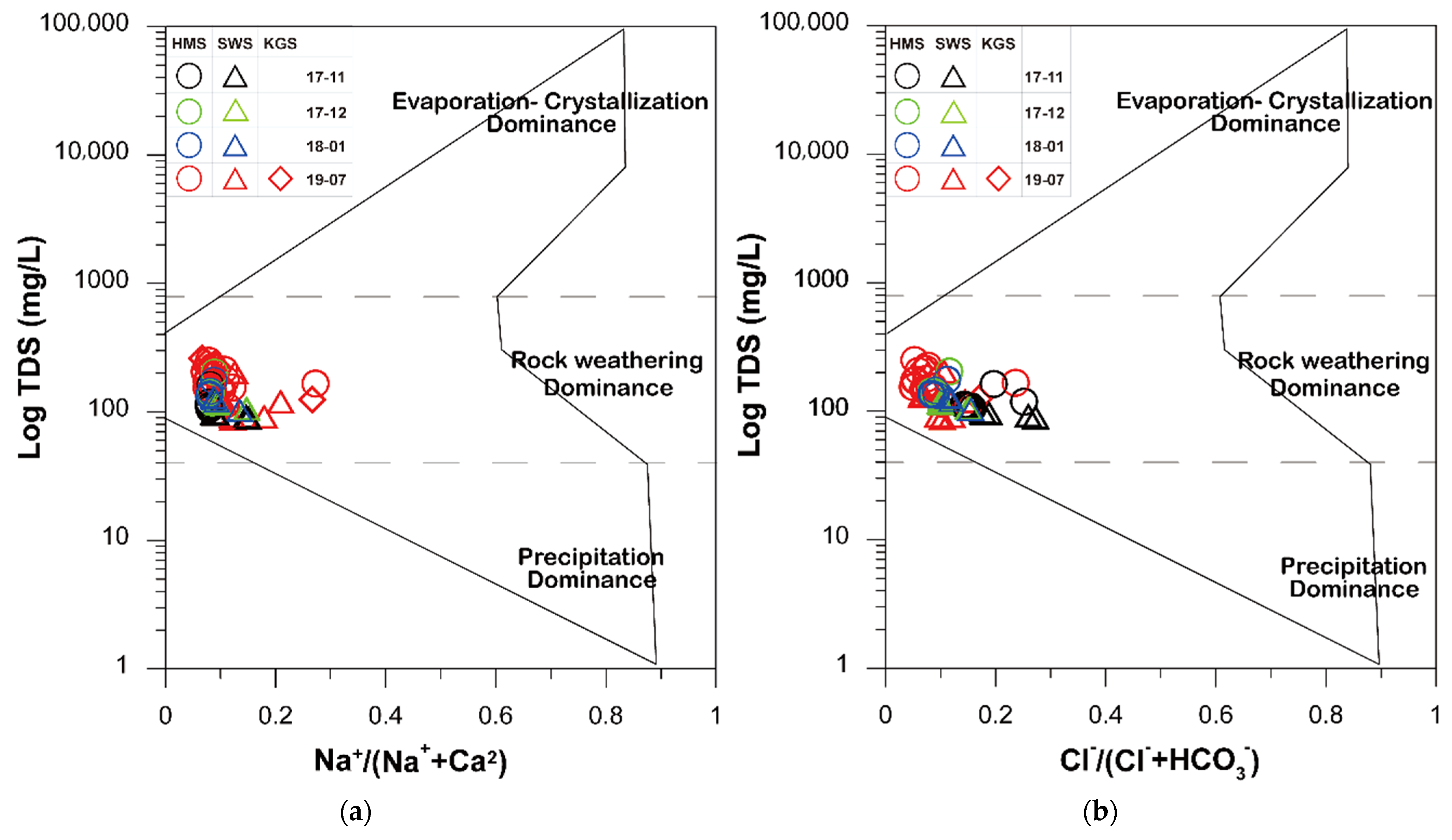
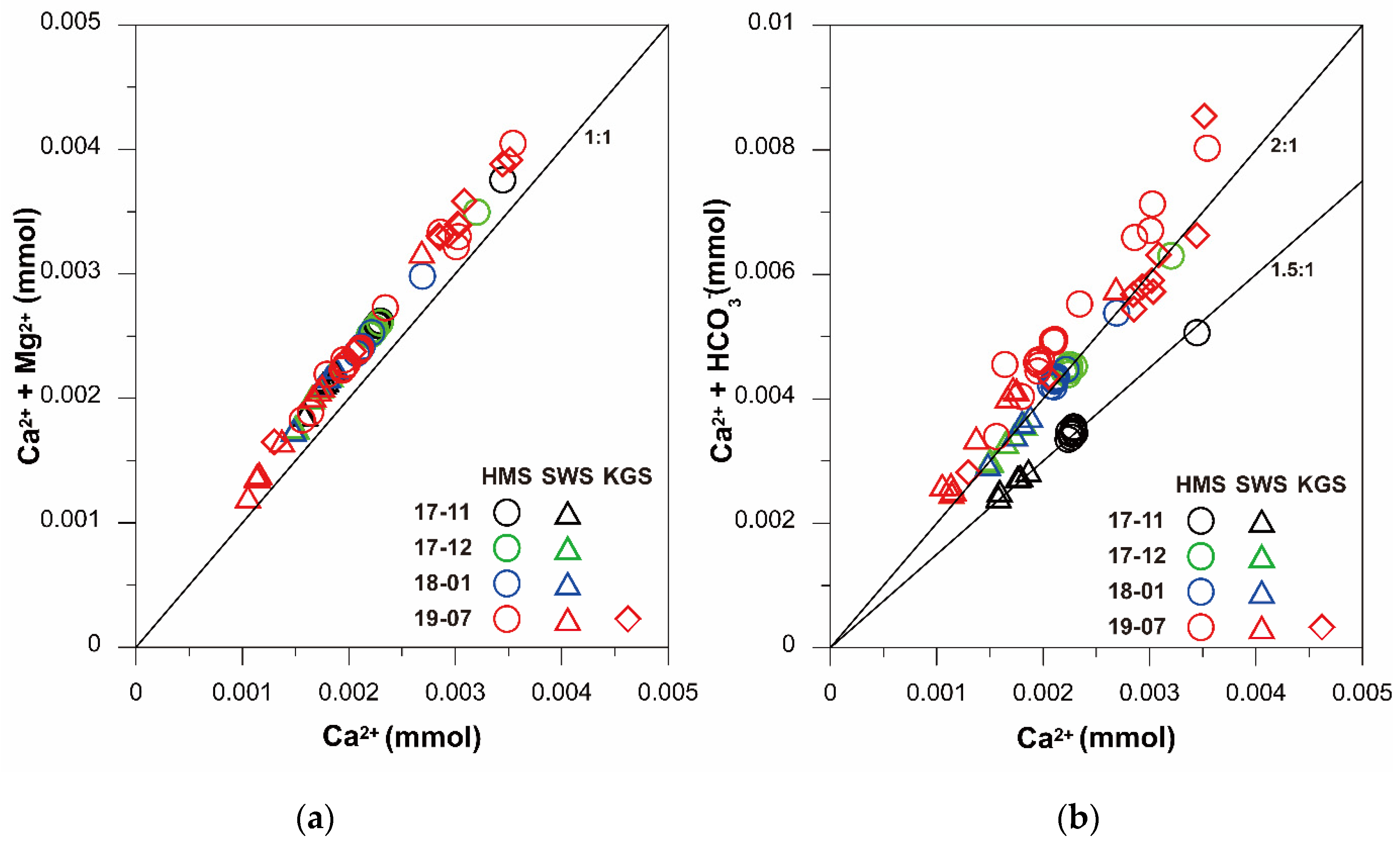
3.3. Ionic Ratios
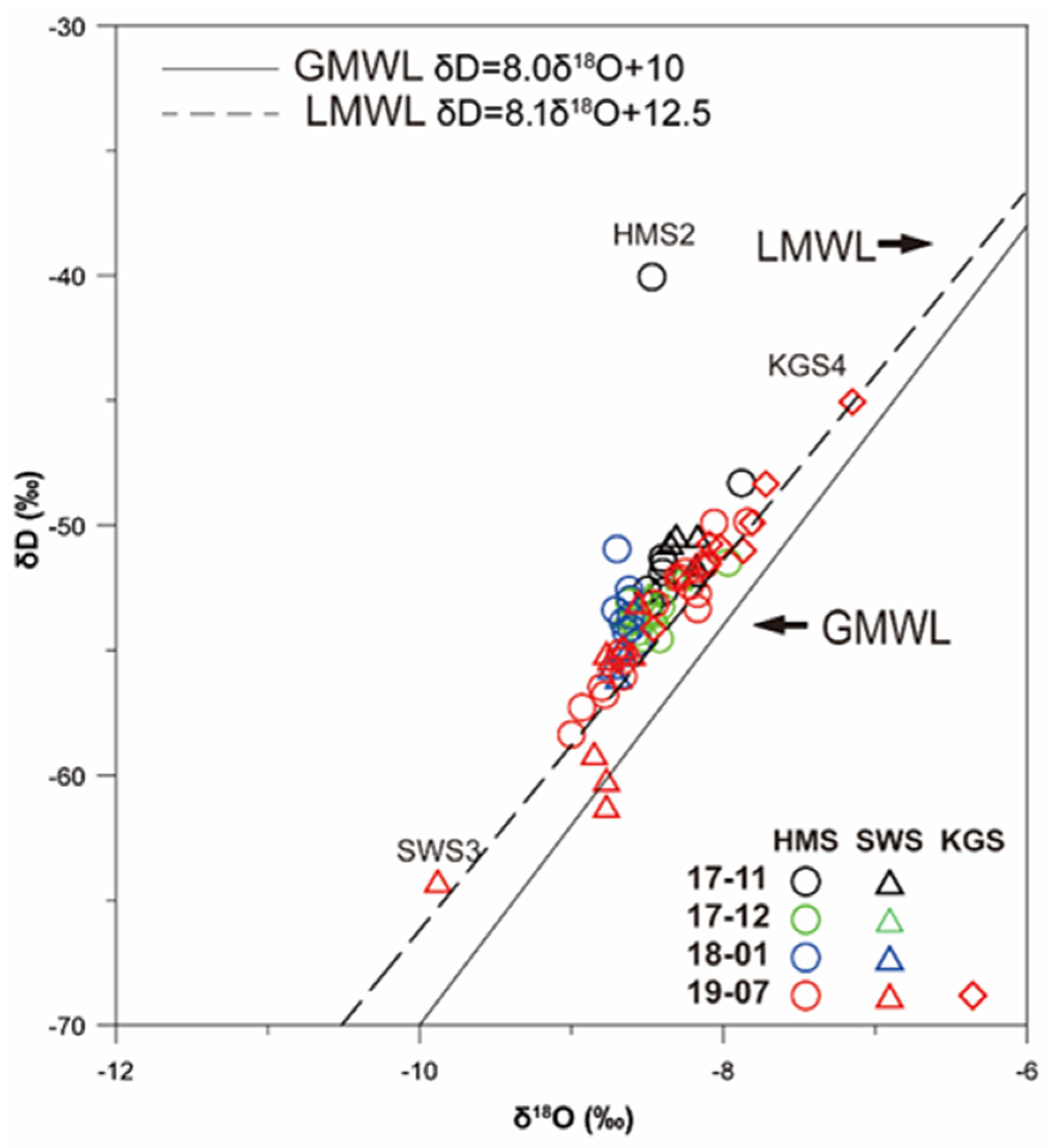
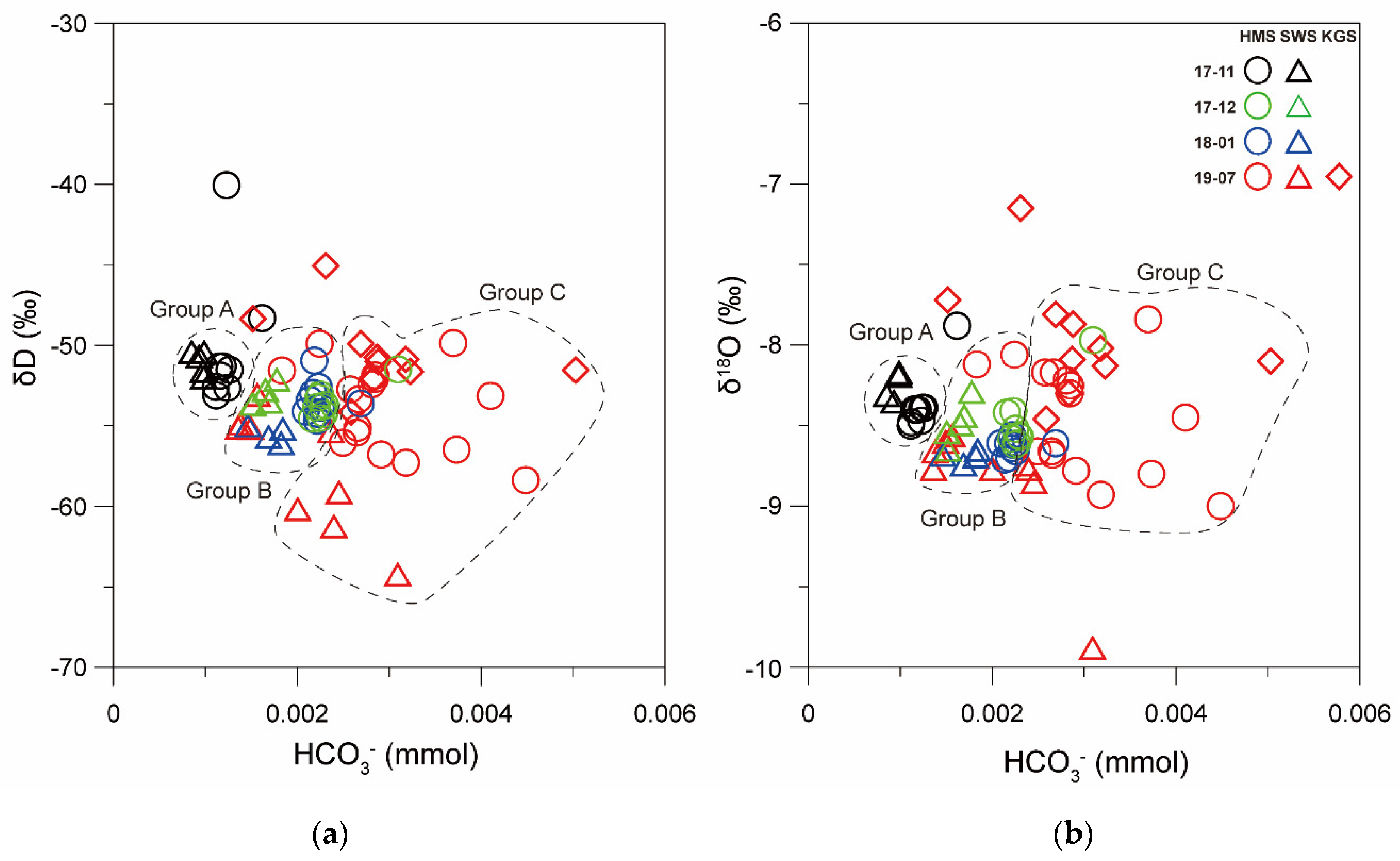
3.4. Stable Isotope Characteristics
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burdon, D.J.; Papakis, N. Handbook of Karst Hydrogeology. United Nations Special Fund; Institute for Geology and Subsurface Research: Athens, Greece, 1963. [Google Scholar]
- Lee, E.S. Review of principle and applications of karst hydrogeology. J. Geol. Soc. Korea. 2004, 40, 349–359, (abstract in Korean). [Google Scholar]
- Tak, H.M.; Son, I. The drainage and irrigation system developed on the karstic uvala: The case of Kwangduk-ri, Nam-myeon, Jungseon-gun, Kangwon-do. J. Korea. Geomorphol. Assoc. 2014, 49, 639–655. (In Korean) [Google Scholar]
- White, W.B. Karst hydrology: Recent developments and open questions. Eng. Geol. 2002, 65, 85–105. [Google Scholar] [CrossRef]
- Hartmann, A.; Goldcheider, N.; Wangener, T.; Lange, J.; Weiler, M. Karst water resources in a changing world: Review of hydrological modeling approaches. Rev. Geophys. 2014, 52, 218–242. [Google Scholar] [CrossRef]
- Pu, J.; Cao, M.; Zhang, Y.; Yuan, D.; Zhao, H. Hydrochemical indications of human impact on karst groundwater in a subtropical karst area, Chongqing, China. Environ. Earth Sci. 2015, 72, 1683–1695. [Google Scholar] [CrossRef]
- Einsiedl, F.; Maloszewski, P.; Stichler, W. Multiple isotope approach to the determination of the natural attenuation potential of a high-alpine karst system. J. Hydrolo. 2009, 365, 113–121. [Google Scholar] [CrossRef]
- Wang, Y.; Guo, Q.; Su, C.; Ma, T. Strontium isotope characterization and major ion geochemistry of karst water flow, Shentou, northern China. J. Hydrolo. 2006, 328, 592–603. [Google Scholar] [CrossRef]
- Raza, M.; Lee, J.Y. Factors affecting spatial pattern of groundwater hydrochemical variables and nitrate in agricultural region of Korea. Episodes 2019, 42, 135–148. [Google Scholar] [CrossRef]
- Barbieri, M.; Boschetti, T.; Petitta, M.; Tallini, M. Stable isotope (2H, 18O and 87Sr/86Sr) and hydrochemistry monitoring for groundwater hydrodynamics analysis in a karst aquifer (Gran Sasso, Central Italy). Appl. Geochem. 2005, 20, 2063–2081. [Google Scholar] [CrossRef]
- Ryu, H.-S.; Lee, J.Y.; Lim, C.W.; Kim, K.T. Hydrochemical characteristics of groundwater and stream water in a karst area of Samcheok, Korea. J. Geol. Soc. Korea. 2019, 55, 117–129. [Google Scholar] [CrossRef]
- Han, G.; Lui, C.-Q. Water geochemistry controlled by carbonate dissolution: A study of the river waters draining karst0dominated terrain, Huizhou Province, China. Chem. Geol. 2004, 204, 1–21. [Google Scholar] [CrossRef]
- Chen, L.; Wang, G.; Hu, F.; Wang, Y.; Liu, L. Groundwater hydrochemistry and isotope geochemistry in the Turpan Basin, northwestern China. J. Arid Land. 2014, 6, 378–388. [Google Scholar] [CrossRef]
- Florea, L.; Forray, F.L.; Banks, S.M. Eurokarst 2018, Catherine Bertrand: Besançon, France; Marc Steinmann: Besançon, France, 2020; pp. 31–46. [Google Scholar]
- Bugna, G.C.; Grace, J.M.; Hsieh, Y.-P. Sensitivity of using stable isotopic tracers to study the hydrology of isolated wetlands in North Florida. J. Hydrolo. 2020, 580, 124321. [Google Scholar] [CrossRef]
- Crag, H. Isotopic Variation in Meteoric Waters. Science 1961, 133, 1702–1703. [Google Scholar] [CrossRef] [PubMed]
- Subyani, A. Use of chloride-mass balance and environmental isotopes for evaluation of groundwater recharge in the alluvial aquifer, Wadi Tharad, western Saudi Arabia. Environ. Geol. 2004, 46, 741–749. [Google Scholar] [CrossRef]
- O’Driscoll, M.A.; DeWalle, D.R. Stream-air temperature relations to classify stream-ground water interactions in a karst setting, central Pennsylvania, USA. J. Hydrolo. 2006, 329, 140–153. [Google Scholar] [CrossRef]
- Dar, F.A.; Perrin, J.; Ahmed, S.; Narayana, A.C. Review: Carbonate aquifers and future perspectives of karst hydrogeology in India. Hydrogeol. J. 2014, 22, 1493–1506. [Google Scholar] [CrossRef]
- Li, G.; Rubinato, M.; Wan, L.; Wu, B.; Luo, J.; Fang, J.; Zhou, J. Preliminary Characterization of Underground Hydrological Processes under Multiple Rainfall Conditions and Rocky Desertification Degrees in Karst Regions of Southwest China. Water 2020, 12, 594. [Google Scholar] [CrossRef]
- Bicalho, C.C.; Guilhe, C.B.; Seidel, J.L.; Exter, S.V.; Jourde, H. Hydrodynamical changes and their consequences on groundwater hydrochemistry induced by three decades of intense exploitation in a Mediterranean Karst system. Environ. Earth Sci. 2012, 65, 2311–2319. [Google Scholar] [CrossRef]
- Cave Research Institute of Korea. Gangwon-do Cave Basic Survey Report; Cave Research Institute of Korea: Chuncheon, Korea, 2018; pp. 52–53. [Google Scholar]
- Woo, K.S.; Won, J.K.; Lee, G.C.; Namkoong, C.; Choi, Y.K. Comprehensive Investigation Report of Chodang Cave; Samcheok City: Samcheok, Korea, 2000; 202p. (In Korean) [Google Scholar]
- Sampyo, K.N.U. Groundwater Impact Investigation Report (1st Stage) on High. Quality Limestone Development of Dongyang 2nd Mine; Sampyo: Samcheok, Korea, 2017; pp. 42–47. [Google Scholar]
- Won, J.K.; Park, B.K.; Lee, S.H. Geologic Report on Samcheok-Gosari Sheet; KIGAM: Daejeon, Korea, 1994; pp. 1–36. [Google Scholar]
- Lee, Y.L.; Choi, D.K. Sedimentology of the Dumugol formation (Lower Ordovician) in the vicinity of the Dongjeom station, Taebaeg City: A preliminary study. J. Geol. Soc. Korea 1987, 23, 331–337. [Google Scholar]
- Choi, D.K.; Chough, S.K.; Kwon, Y.K.; Lee, S.; Woo, J.; Kang, I.; Lee, H.S.; Lee, S.M.; Sohn, J.W.; Shinn, Y.J.; et al. Taebaek Group (Cambrian-Ordovician) in the Seokgaejae section, Taebaeksan Basin: A refined lower Paleozoic stratigraphy in Korea. Geosci. J. 2004, 8, 125–152. [Google Scholar] [CrossRef]
- Kobayashi, T. Stratigraphy of the Chosen Group in Korea and south Manchuria and its relation to the Cambro-Ordovician formations of other areas. Section, B. The Chosen Group of north Korea and northeast China. J. Fac. Sci. 1966, 2, 209–311. [Google Scholar]
- Noh, J.H.; Oh, S.J. Hydrothermal alteration of the Pungchon limestone and the formation of high-Ca limestone. J. Geol. Soc. Korea. 2005, 41, 175–197. [Google Scholar]
- Kim, Y.H.; Kim, G.B.; Choi, S.G.; Kim, C.S. SWIR application for the identification of high-grade limestones from the Upper Pungchon formation. Econ. Environ. Geol. 2016, 49, 335–347. [Google Scholar] [CrossRef]
- Ryu, H.-S.; Kim, H.J.; Lee, J.Y.; Jang, J.W.; Park, S.W. Spatio-temporal variations of hydrochemical and microbial characteristics in karst water in Samcheok, South Korea. Water 2020, 12, 3407. [Google Scholar] [CrossRef]
- Cheong, C.H. Stratigraphy and paleontology of the Samcheog Coalfield, Gangwondo, Korea. J. Geol. Soc. Korea. 1969, 5, 13–55. [Google Scholar]
- Son, C.M.; Cheong, C.H. Sedimentary environment and geologic structure or Taebaegsan district. Seoul National Univ. Sci. Tech. Ser. (A). 1965, 15, 741–749. [Google Scholar]
- Park, S.I.; Lee, H.K.; Lee, S.H. Characteristics and distribution pattern of carbonate rock resources in Kangwon Area: The Gabsan Formation around the Mt. Gachang Area, Chungbuk, Korea. J. Geol. Soc. Korea 2000, 21, 437–448. [Google Scholar]
- Lee, J.Y.; Raza, M.; Kwon, K.D. Land use and land cover changes in the Haean Basin of Korea: Impacts on soil erosion. Episodes. 2019, 42, 17–32. [Google Scholar] [CrossRef]
- Lee, J.Y.; Hahn, J.S. Characterization of groundwater temperature obtained from the Korean national groundwater monitoring stations: Implications for heat pumps. J. Hydrolo. 2006, 329, 514–526. [Google Scholar] [CrossRef]
- Sasowsky, I.D.; Dalton, C.T. Measurement of pH for field studies in karst areas. J. Caves. Karst. Stud. 2005, 67, 127–132. [Google Scholar]
- Lang, Y.-C.; Liu, C.-Q.; Zhao, Z.-Q.; Li, S.-L.; Han, G.L. Geochemistry of surface and ground water in Giuyang, China: Water/rock interaction and pollution in a karst hydrological system. Appl. Geochem. 2006, 21, 887–903. [Google Scholar] [CrossRef]
- Sappa, G.; Ergul, S.; Ferranti, F. Water quality assessment of carbonate aquifers in southern Latium region, Central Italy: A case study for irrigation and drinking purposes. Appl. Water Sci. 2014, 4, 115–128. [Google Scholar] [CrossRef]
- Yun, S.W.; Jeon, W.H.; Lee, J.Y. Evaluation of hydrochemical characteristics of groundwater and stream water in a heavy agricultural region of the Haean basin, Korea. J. Geol. Soc. Korea. 2017, 53, 727–742. [Google Scholar] [CrossRef]
- Yun, S.W.; Lee, J.Y.; Lee, H.G. Variation of stream water quality and baseflow contribution from groundwater during rainfall event in the Haean basin. J. Geol. Soc. Korea. 2015, 51, 611–621. [Google Scholar] [CrossRef]
- Kim, H.; Jang, J.; Park, S.; Kim, C.S.; Ryu, H.S.; Lee, J.Y. Spatial variations in microbial compositions in a karst critical zone in Samcheok, Republic of Korea. Appl. Sci. 2020, 10, 4714. [Google Scholar] [CrossRef]
- Hwang, J.H.; Song, M.H.; Cho, H.L.; Woo, N.C. Influence of groundwater on the origin of Oseepchun in Dogye area, Korea. Econ. Environ. Geol. 2016, 49, 161–179, (In Korean with English abstract). [Google Scholar]
- Nwankwoala, H.O.; Amadi, A.N.; Oborie, E.; Ushie, F.A. Hydrochemical factors and correlation analysis in groundwater quality in Yenagoa, Bayelsa State, Nigeria. Appl. Ecol. Env. Sci. 2014, 2, 100–105. [Google Scholar] [CrossRef][Green Version]
- Voudouris, K.; Panagopoulos, A.; Koumantakis, J. Multivariate statistical analysis in the assessment of hydrochemistry of the Northern Korinthia prefecture alluvial aquifer system (Peloponnese, Greece). Nat. Resour. Res. 2000, 9, 135–146. [Google Scholar] [CrossRef]
- Gaillardet, J.; Calmels, D.; Romero-Mujalli, G.; Zakharova, E.; Hartmann, J. Global climate control on carbonate weathering intensity. Chem. Geol. 2019, 527, 118762. [Google Scholar] [CrossRef]
| Parameter | Hamaengbang Area | Sangwolsan Area | Kyogok Area | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Max. | Min. | Avg. | SD * | n | Max. | Min. | Avg. | SD * | n | Max. | Min. | Avg. | SD * | |
| Temp. (°C) | 45 | 27.8 | 2.1 | 12.3 | 6.3 | 24 | 28.3 | 0.6 | 10.5 | 9.7 | 10 | 29.8 | 17.4 | 20.9 | 3.8 |
| pH | 45 | 8.9 | 7.8 | 8.3 | 0.2 | 24 | 8.9 | 8.0 | 8.4 | 0.2 | 10 | 8.7 | 8.0 | 8.3 | 0.3 |
| EC (μS/cm) | 45 | 321 | 138 | 192 | 36.7 | 24 | 233 | 102 | 139 | 36.2 | 10 | 358 | 170 | 298 | 51.7 |
| DO (mg/L) | 45 | 11.2 | 4.9 | 7.6 | 1.4 | 24 | 10.6 | 5.3 | 7.4 | 1.3 | 10 | 8.0 | 5.1 | 6.4 | 1.0 |
| Eh (mV) | 45 | 530 | 229 | 435 | 47 | 24 | 453 | 286 | 384 | 51 | 10 | 498 | 311 | 426 | 50 |
| Turb. (NTU) | 46 | 74.2 | 0.8 | 15.1 | 23.4 | 24 | 5.1 | 0.5 | 2.6 | 1.3 | 10 | 892 | 6.7 | 169 | 300 |
| Ca2+ (mg/L) | 45 | 70.9 | 31.2 | 45.8 | 8.3 | 22 | 53.7 | 21.0 | 32.9 | 6.7 | 9 | 70.3 | 26.0 | 55.7 | 14.0 |
| Mg2+ (mg/L) | 45 | 6.1 | 2.5 | 3.8 | 0.6 | 22 | 5.9 | 1.9 | 3.9 | 0.8 | 9 | 6.0 | 3.9 | 4.9 | 0.7 |
| Na+ (mg/L) | 45 | 13.5 | 3.4 | 4.5 | 1.6 | 22 | 7.8 | 3.1 | 4.4 | 1.3 | 9 | 9.5 | 4.9 | 5.9 | 1.4 |
| K+ (mg/L) | 45 | 6.6 | 0.5 | 1.1 | 0.9 | 22 | 1.7 | 0.0 | 0.8 | 0.3 | 9 | 3.0 | 0.8 | 1.7 | 0.7 |
| HCO3− (mg/L) | 45 | 139 | 34.5 | 73.2 | 24.4 | 22 | 95.9 | 26.4 | 51.3 | 17.5 | 9 | 155 | 47.0 | 90.4 | 29.3 |
| SO42− (mg/L) | 45 | 14.1 | 3.6 | 6.3 | 1.6 | 22 | 13.2 | 4.3 | 8.3 | 2.8 | 9 | 12.5 | 6.2 | 8.5 | 1.9 |
| Cl− (mg/L) | 45 | 11.4 | 3.6 | 5.5 | 2.1 | 22 | 16.6 | 2.6 | 4.4 | 2.9 | 9 | 70.2 | 6.5 | 36.8 | 21.2 |
| NO3− (mg/L) | 45 | 21.6 | 4.7 | 7.4 | 2.8 | 22 | 11.6 | 4.9 | 7.3 | 1.9 | 9 | 9.7 | 5.5 | 7.6 | 1.5 |
| δ2H (‰) | 45 | −40.1 | −57.3 | −52.9 | 2.8 | 22 | −50.4 | −64.2 | −54.8 | 3.6 | 9 | −45.1 | −54.1 | −50.8 | 2.4 |
| δ13C (‰) | 45 | −8.9 | −14.3 | −11.7 | 1.1 | 22 | −8.5 | −13.0 | −10.7 | 1.7 | 9 | −9.4 | −14.2 | −11.5 | 1.3 |
| δ18O (‰) | 45 | −7.8 | −8.9 | −8.5 | 0.3 | 22 | −8.2 | −9.9 | −8.6 | 0.4 | 9 | −7.2 | −8.5 | −8.0 | 0.4 |
| n = 79 | Temp. | pH | EC | DO | Eh | Turb. | Ca2+ | K+ | Mg2+ | Na+ | Cl− | NO3− | SO42− | HCO3− | δD | δ18O | δ13C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temp. | – | 0.1 | 0.0 | −0.5 | −0.5 | 0.2 | 0.0 | −0.1 | −0.1 | −0.1 | −0.1 | 0.3 | −0.2 | 0.4 | −0.2 | 0.2 | −0.5 |
| pH | 0.1 | – | −0.3 | 0.2 | −0.3 | 0.0 | −0.2 | −0.4 | −0.3 | −0.3 | −0.3 | 0.0 | −0.4 | −0.1 | −0.2 | −0.1 | 0.4 |
| EC | 0.0 | −0.3 | – | 0.0 | −0.1 | 0.1 | 0.2 | 1.0 | 1.0 | 1.0 | 1.0 | 0.3 | 0.9 | 0.0 | 0.2 | 0.2 | 0.0 |
| DO | −0.5 | 0.2 | 0.0 | – | 0.3 | −0.1 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 | −0.1 | 0.0 | −0.2 | 0.2 | 0.0 | 0.5 |
| Eh | −0.5 | −0.3 | −0.1 | 0.3 | – | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | −0.3 | 0.0 | −0.2 | 0.2 | 0.0 | −0.1 |
| Turb. | 0.2 | 0.0 | 0.1 | −0.1 | 0.0 | – | 0.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.6 | 0.0 | 0.2 | 0.1 | 0.3 | −0.2 |
| Ca2+ | 0.0 | −0.2 | 0.2 | 0.1 | 0.0 | 0.3 | – | 0.1 | 0.1 | 0.0 | 0.0 | 0.5 | 0.0 | 0.6 | 0.2 | 0.3 | −0.1 |
| K+ | −0.1 | −0.4 | 1.0 | 0.0 | 0.0 | 0.0 | 0.1 | – | 1.0 | 1.0 | 1.0 | 0.1 | 1.0 | −0.1 | 0.2 | 0.2 | 0.0 |
| Mg2+ | −0.1 | −0.3 | 1.0 | 0.0 | 0.0 | 0.0 | 0.1 | 1.0 | – | 1.0 | 1.0 | 0.2 | 1.0 | −0.1 | 0.1 | 0.1 | 0.1 |
| Na+ | −0.1 | −0.3 | 1.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.0 | 1.0 | – | 1.0 | 0.1 | 1.0 | −0.1 | 0.1 | 0.1 | 0.1 |
| Cl− | −0.1 | −0.3 | 1.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.0 | 1.0 | 1.0 | – | 0.1 | 1.0 | −0.1 | 0.1 | 0.1 | 0.1 |
| NO3− | 0.3 | 0.0 | 0.3 | −0.1 | −0.3 | 0.6 | 0.5 | 0.1 | 0.2 | 0.1 | 0.1 | – | 0.1 | 0.3 | 0.1 | 0.2 | 0.1 |
| SO42− | −0.2 | −0.4 | 0.9 | 0.0 | 0.0 | 0.0 | 0.0 | 1.0 | 1.0 | 1.0 | 1.0 | 0.1 | – | −0.1 | 0.1 | 0.2 | −0.1 |
| HCO3− | 0.4 | −0.1 | 0.0 | −0.2 | −0.3 | 0.2 | 0.6 | −0.1 | −0.1 | −0.1 | −0.1 | 0.3 | −0.1 | – | −0.3 | −0.1 | −0.4 |
| δD | −0.2 | −0.2 | 0.2 | 0.2 | 0.2 | 0.1 | 0.2 | 0.2 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | −0.3 | - | 0.8 | 0.1 |
| δ 18O | 0.2 | −0.1 | 0.2 | 0.0 | 0.0 | 0.3 | 0.3 | 0.2 | 0.1 | 0.1 | 0.1 | 0.2 | 0.2 | −0.1 | 0.8 | - | 0.0 |
| δ13C | −0.5 | 0.4 | 0.0 | 0.5 | −0.1 | −0.2 | −0.1 | 0.0 | 0.1 | 0.1 | 0.1 | 0.1 | −0.1 | −0.4 | 0.1 | 0.0 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, J.; Lee, J.-Y.; Kim, H. Characteristics of Hydrochemistry and Stable Isotopes in a Karst Region in Samcheok, Republic of Korea. Water 2021, 13, 213. https://doi.org/10.3390/w13020213
Jang J, Lee J-Y, Kim H. Characteristics of Hydrochemistry and Stable Isotopes in a Karst Region in Samcheok, Republic of Korea. Water. 2021; 13(2):213. https://doi.org/10.3390/w13020213
Chicago/Turabian StyleJang, Jiwook, Jin-Yong Lee, and Heejung Kim. 2021. "Characteristics of Hydrochemistry and Stable Isotopes in a Karst Region in Samcheok, Republic of Korea" Water 13, no. 2: 213. https://doi.org/10.3390/w13020213
APA StyleJang, J., Lee, J.-Y., & Kim, H. (2021). Characteristics of Hydrochemistry and Stable Isotopes in a Karst Region in Samcheok, Republic of Korea. Water, 13(2), 213. https://doi.org/10.3390/w13020213








