Suppression of Alkalization in Rainwater Regulating Reservoir by Shading on a Pilot Scale
Abstract
:1. Introduction
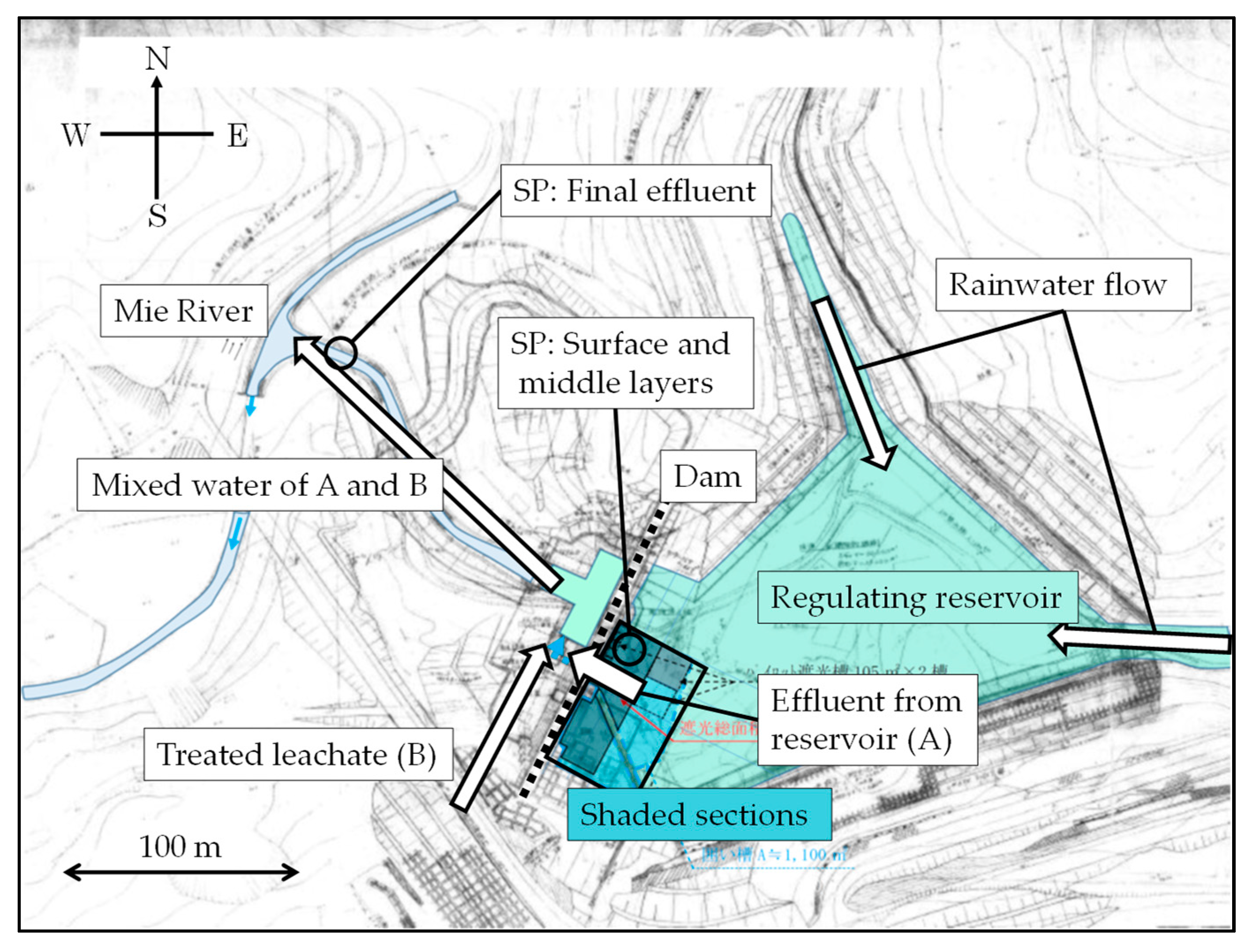
2. Water Alkalization Problem in Rainwater Regulating Reservoir at Sankyo Landfill Site
3. Materials and Methods
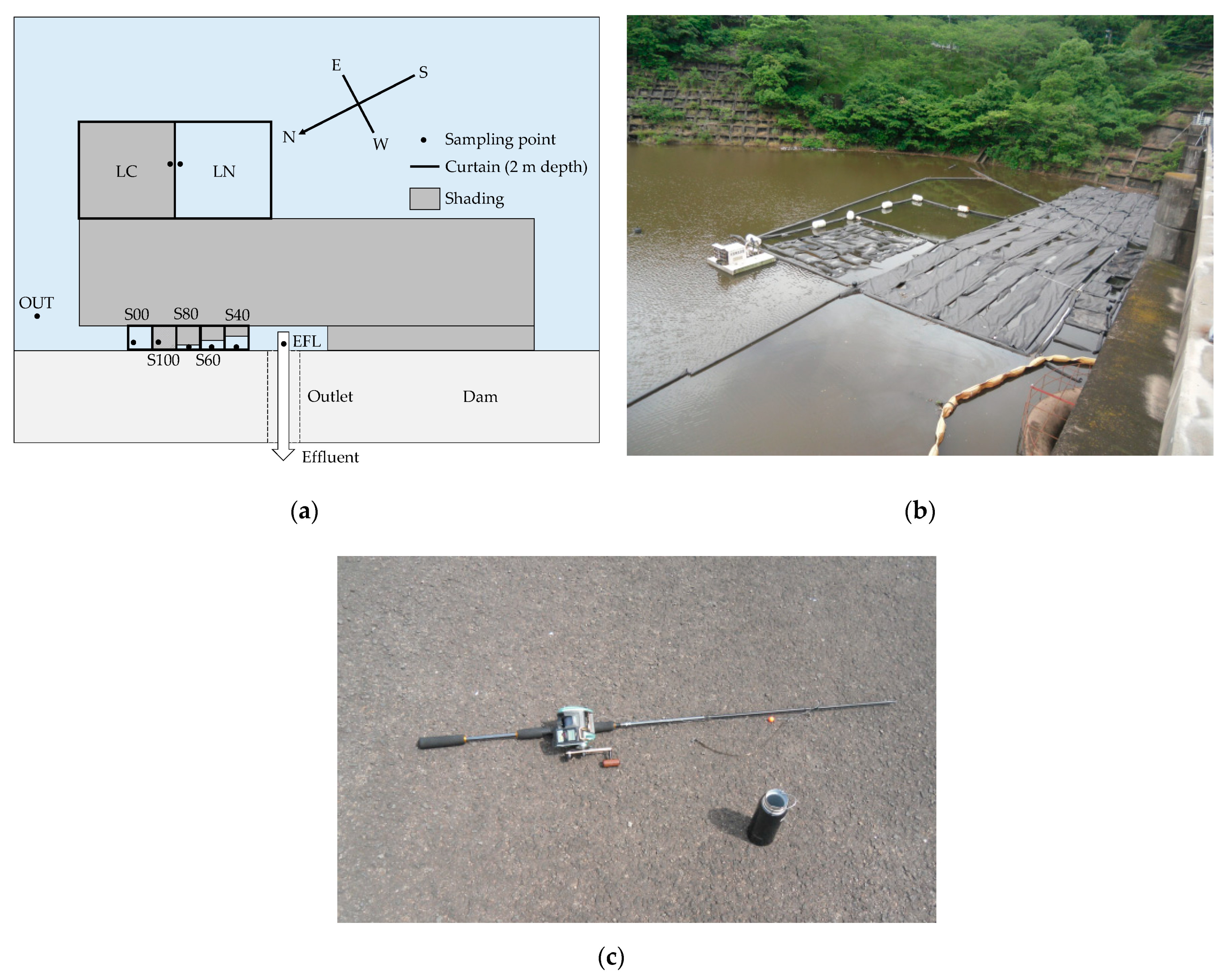
3.1. Outline
3.2. Methods
3.3. Measurement Equipment
4. Results
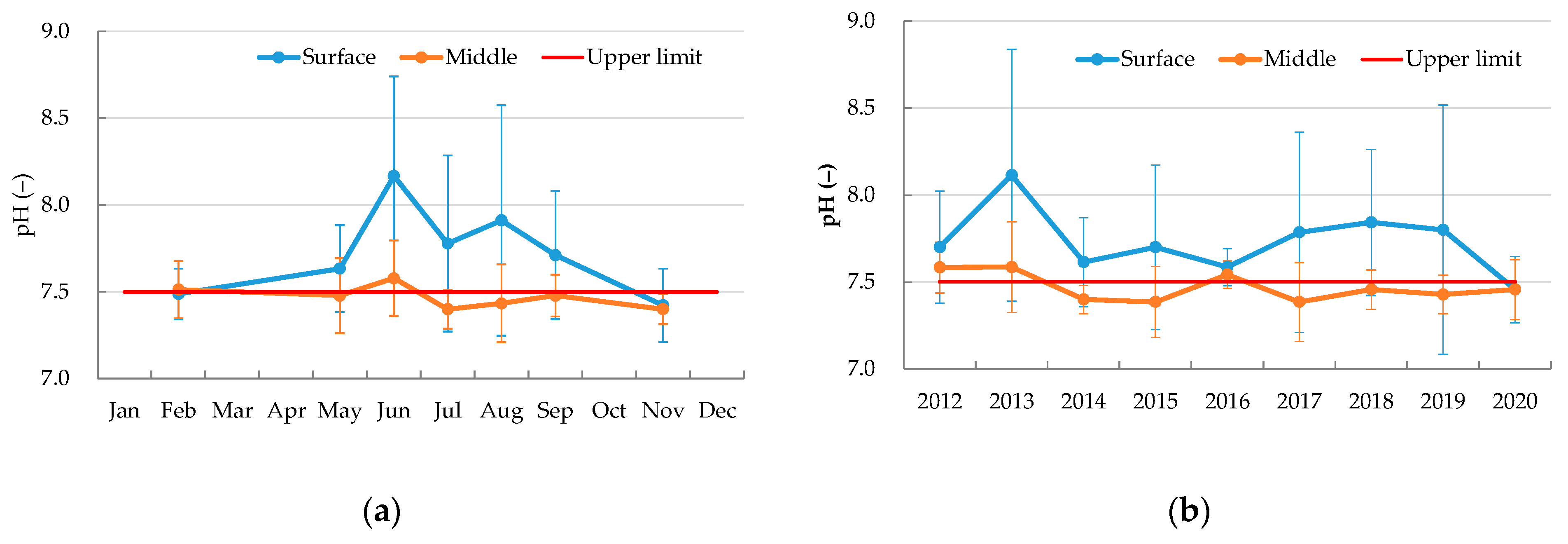
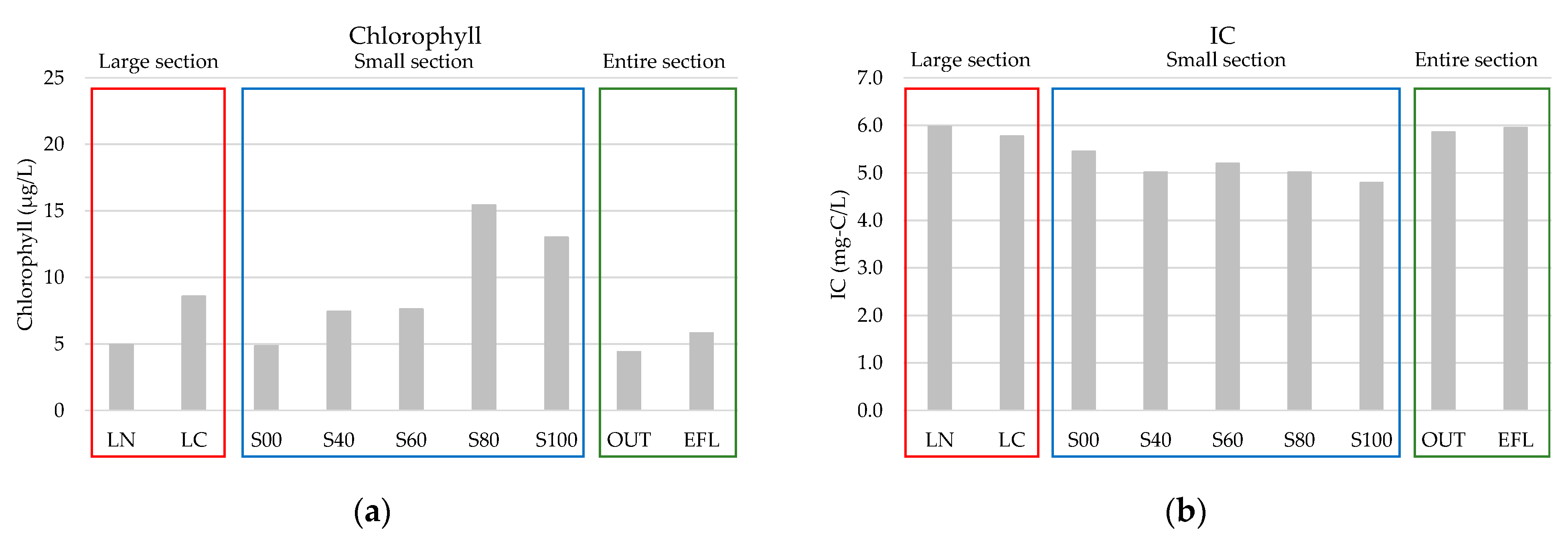
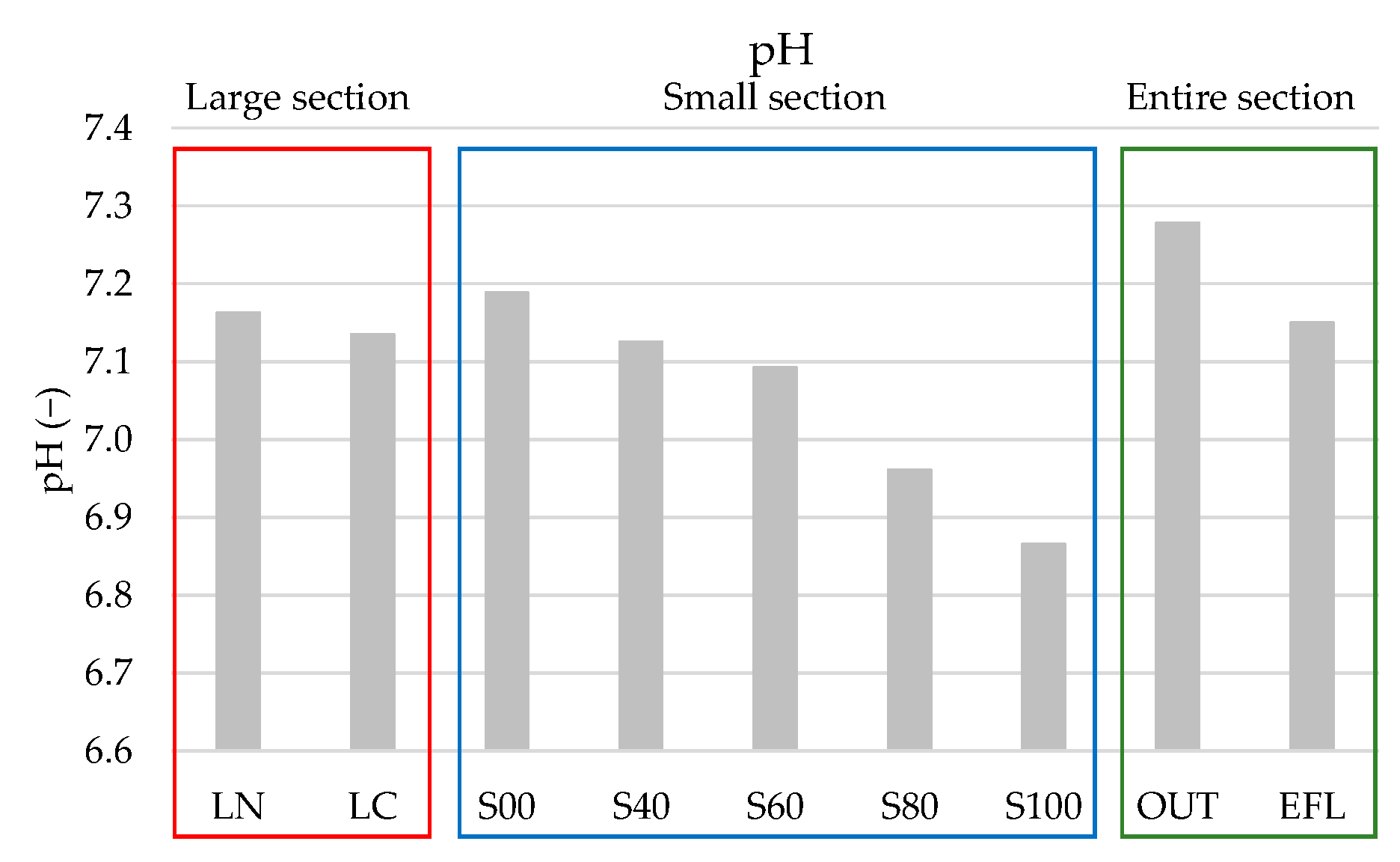
4.1. Average Values of Water Quality Parameters
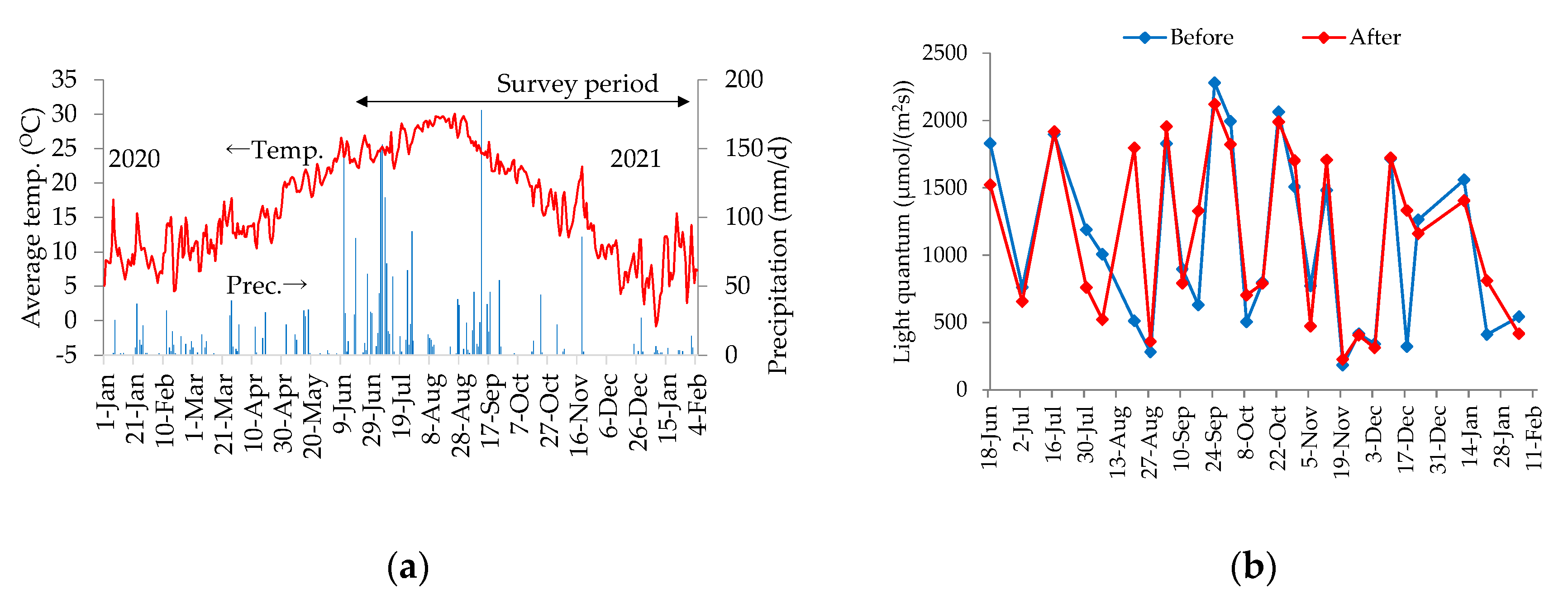
4.2. Weather
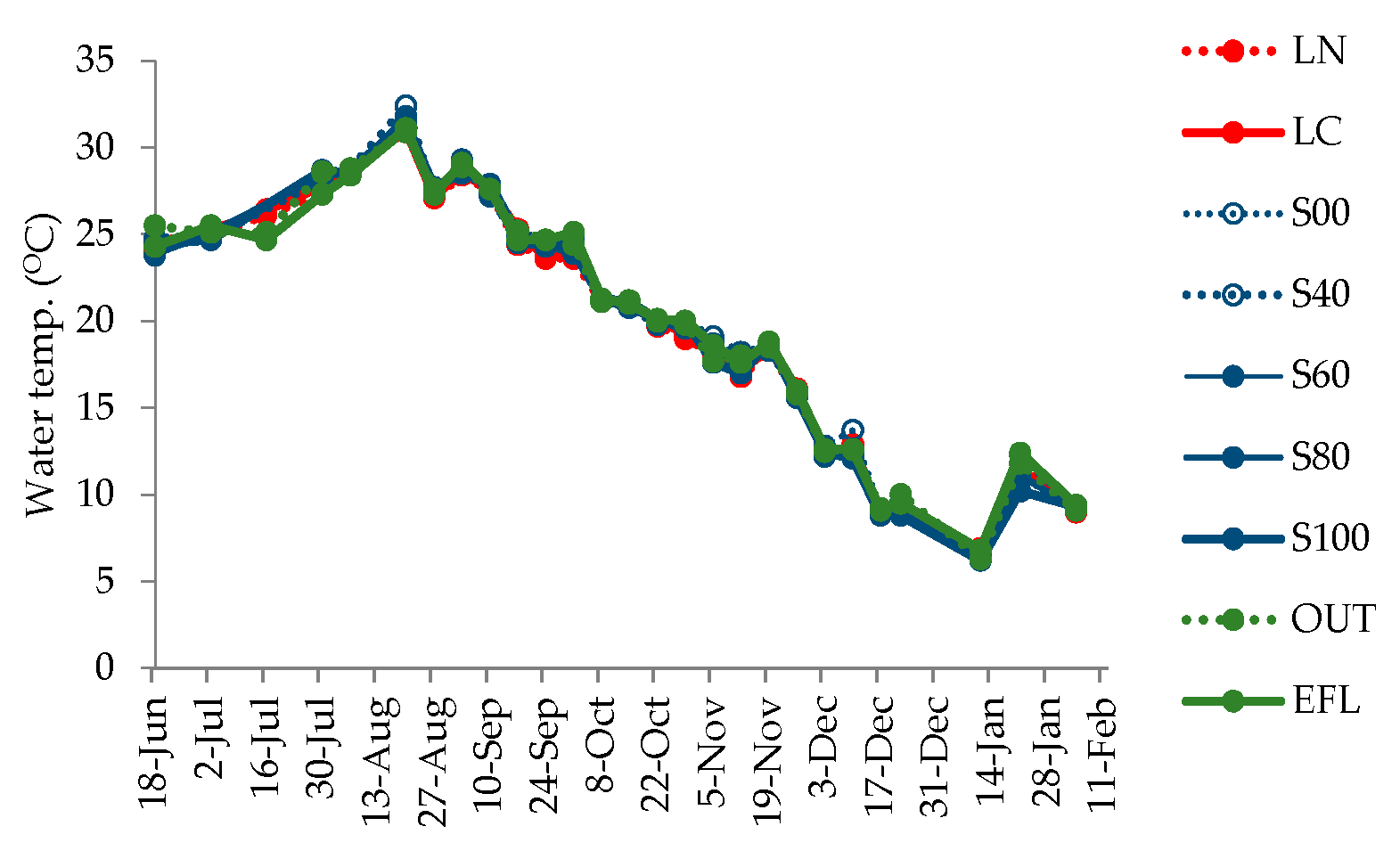
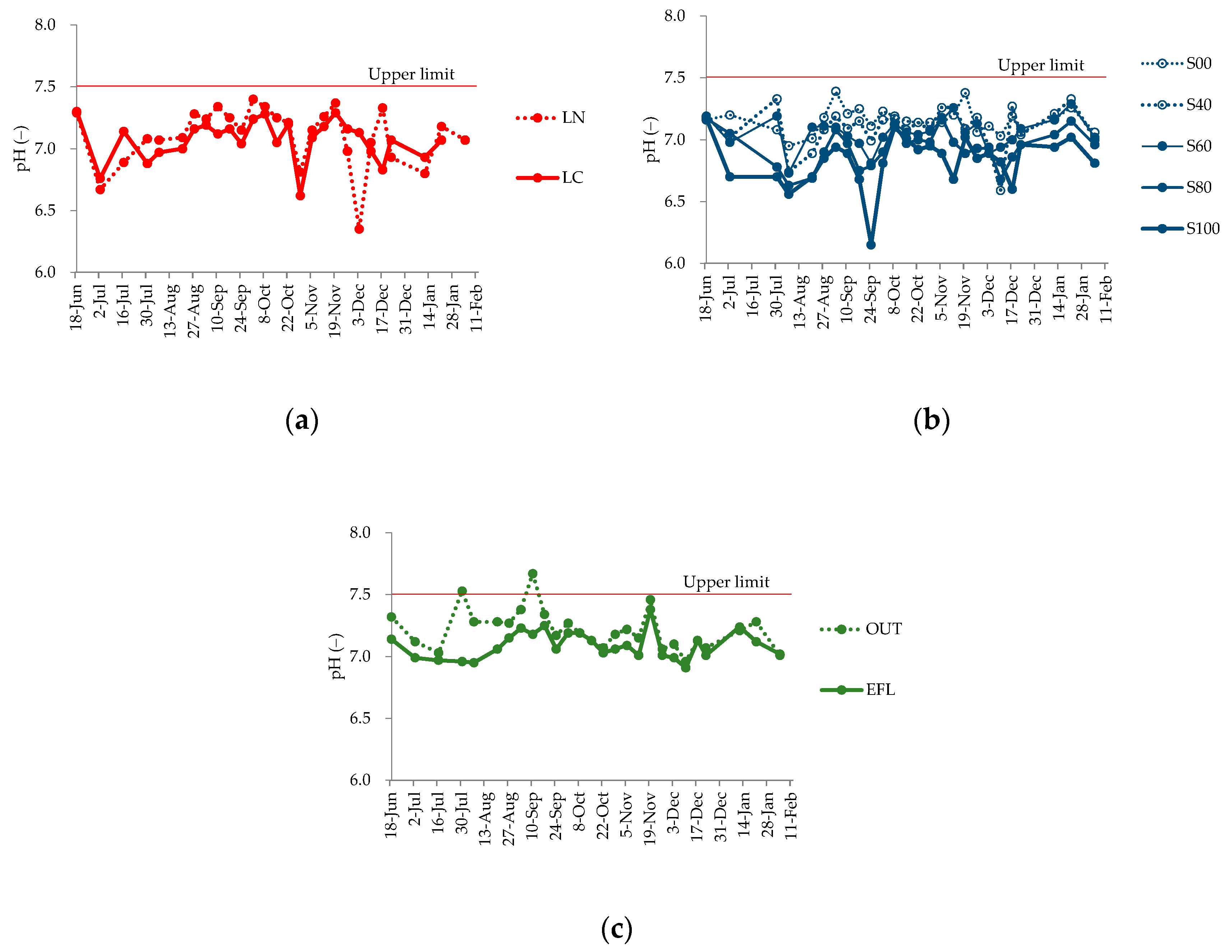
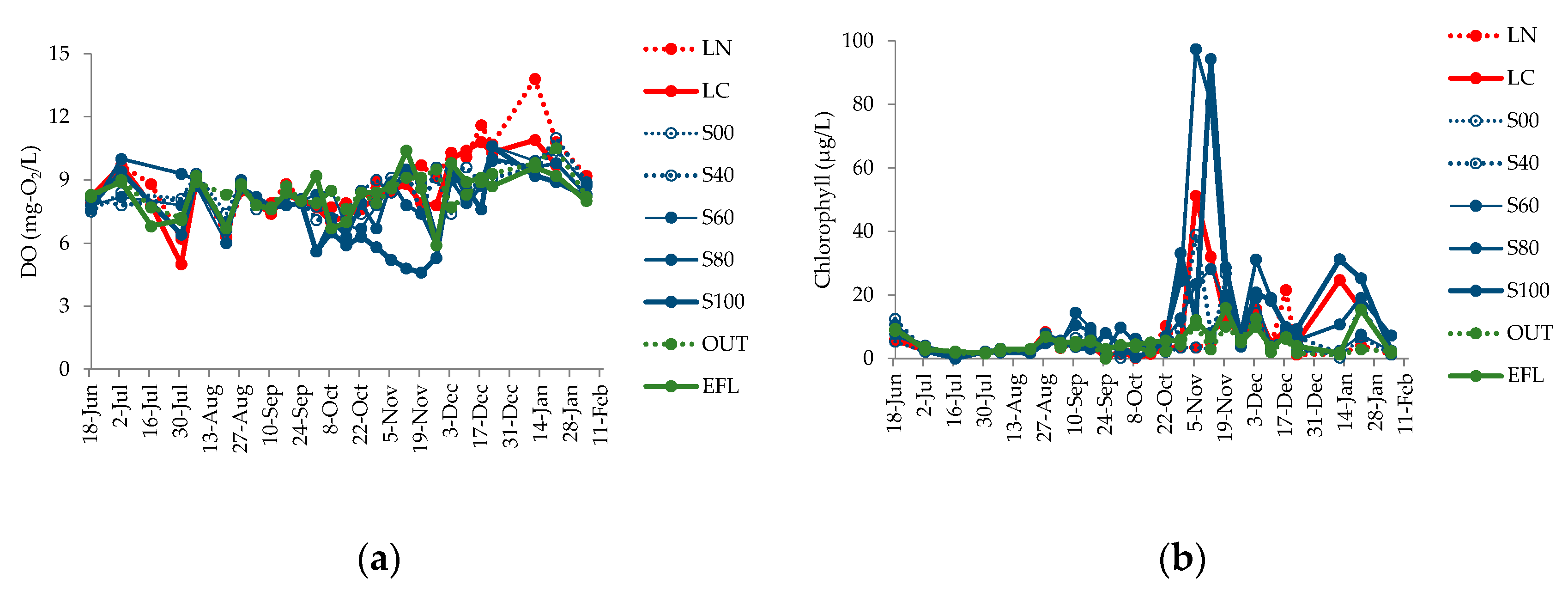
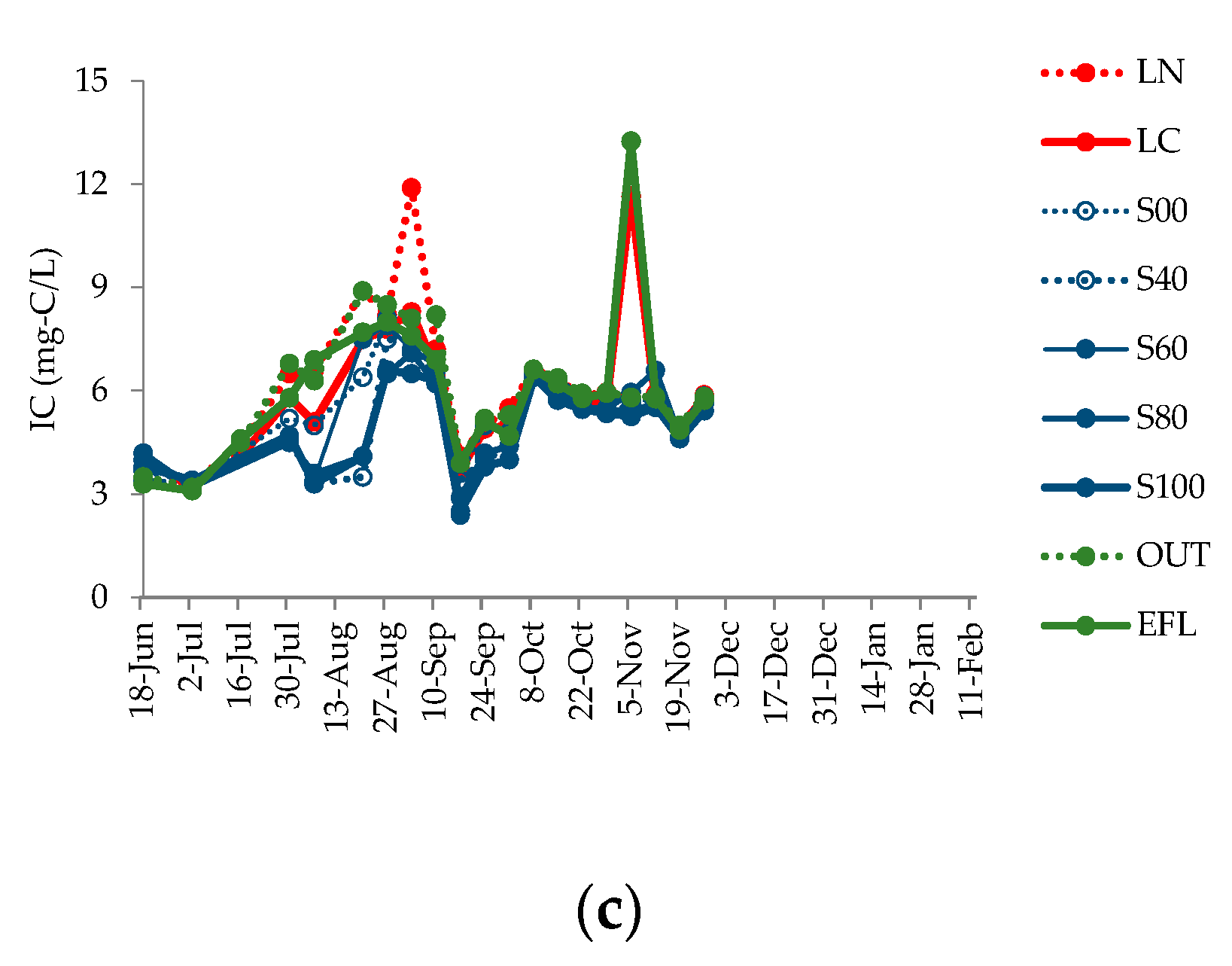
4.3. Changes in Water Quality Parameters
5. Discussion
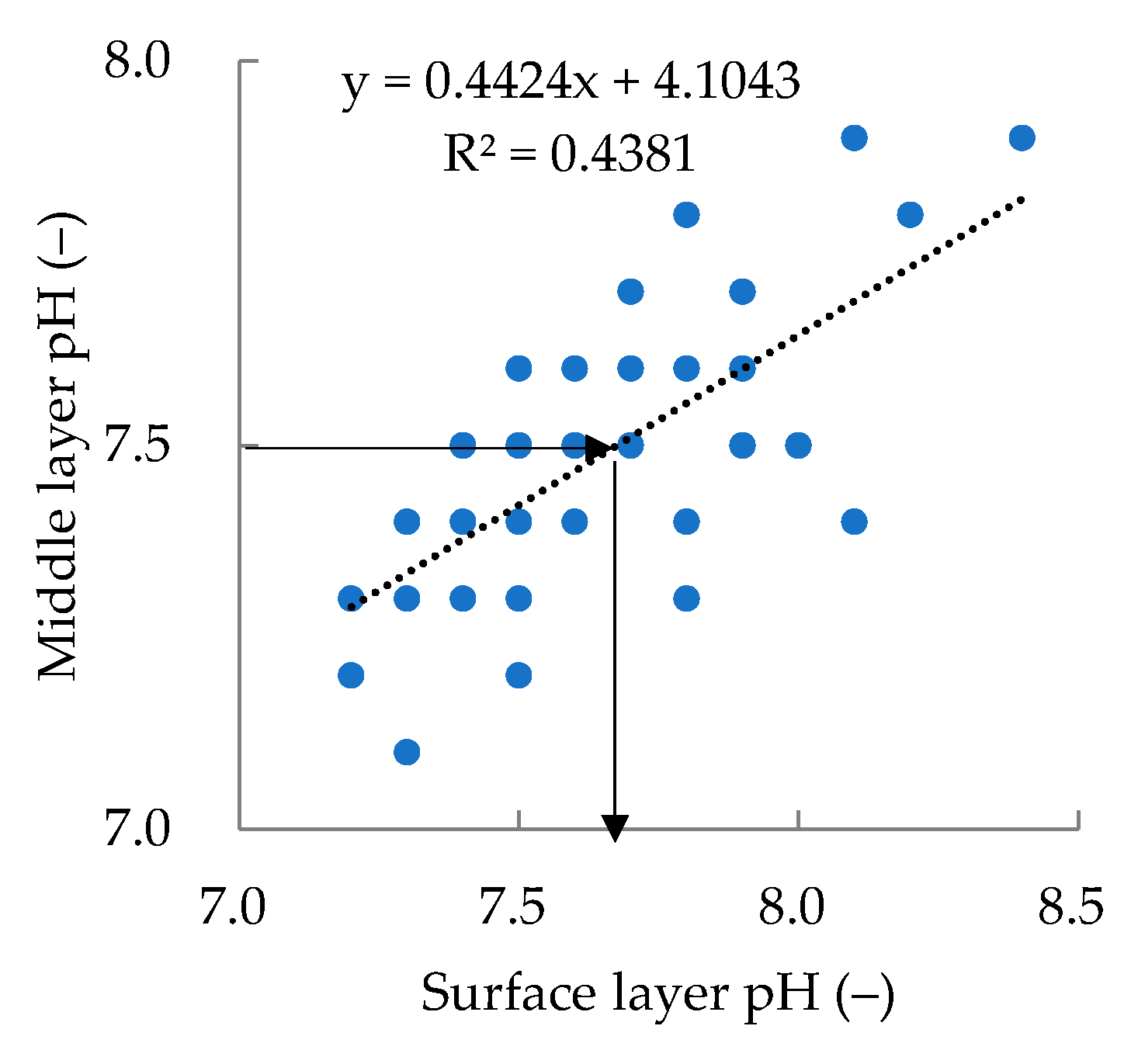
5.1. Relationship between Shading Effect and Alkalization Suppression Effect
5.2. Problems, Prospects, Relevance of This Study
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cook, C.M.; Lanaras, T.; Roubelakis-Angelakis, K.A. Bicarbonate transport and alkalization of the medium by four species of Rhodophyta. J. Exp. Bot. 1988, 39, 1185–1198. [Google Scholar] [CrossRef]
- Matsuyama, M.; Oshio, T.; Kuwana, T.; Kishigami, M. Water quality of farm-use water at main irrigation spots in Hyogo prefecture from 1996 through 1998. Bull. Hyogo Prefect. Agric. Institute. Agric. Sect. 2001, 49, 25–34. (In Japanese) [Google Scholar]
- Yajima, K.; Urabe, A.; Yamada, A.; Fujinami, H.; Yoshida, T.; Mogi, S.; Urano, E. Water environment survey in Kiryu river. Annu. Rep. Gunma Prefect. Inst. Public Health Environ. Sci. 2001, 33, 202–206. (In Japanese) [Google Scholar]
- Nakamura, C.; Yasuda, I.; Azuma, C. The cause of the pH rise downstream in small irrigation streams in Toyama prefecture. Environ. Sci. 2006, 19, 113–122. (In Japanese) [Google Scholar]
- Isagai, K. Effect of the Egeria densal on dissolved oxgen and pH in river water. Bull. Fac. Child. Stud. Nishikyushu Univ. 2010, 1, 63–68. (In Japanese) [Google Scholar]
- Manaka, T.; Ushie, H.; Araoka, D.; Inamura, A.; Suzuki, A.; Kawahata, H. Rapid alkalization in lake Inawashiro, Fukushima, Japan: Implications for future changes in the carbonate system of terrestrial waters. Aquat. Geochem. 2013, 19, 281–302. [Google Scholar] [CrossRef]
- Zerveas, S.; Mente, M.S.; Tsakiri, D.; Kotzabasis, K. Microalgal photosynthesis induces alkalization of aquatic environment as a result of H+ uptake independently from CO2 concentration–New perspectives for environmental applications. J. Environ. Manag. 2021, 289, 112546. [Google Scholar] [CrossRef] [PubMed]
- Lucas, W.J. Photosynthetic assimilation of exogenous HCO3- by aquatic plants. Annu. Rev. Plant. Physiol. 1983, 34, 71–104. [Google Scholar] [CrossRef]
- Axelsson, L. Changes in pH as a measure of photosynthesis by marine macroalgae. Mar. Biol. 1988, 97, 287–294. [Google Scholar] [CrossRef]
- Suzuki, T.; Nishimura, K.; Ohtaguchi, K.; Koide, K. Chemometrics approach to the analysis of a synergism of temperature, light intensity and carbon dioxide concentration on the growth of anacystis nidulans. J. Chem. Eng. Jpn. 1991, 24, 563–568. [Google Scholar] [CrossRef] [Green Version]
- Kunugi, M.; Matsumoto, M.; Kondo, K. Effect of culture conditions on production of useful materials by Haematococcus pluvialis. Bull. Soc. Sea Water Sci. Jpn. 2010, 64, 297–304. (In Japanese) [Google Scholar]
- Iseri, Y.; Mori, M.; Matsuoka, Y. Control technology of abnormal algal blooms in closed water bodies. Environ. Meas. Resour. 2002, 9, 1–14. (In Japanese) [Google Scholar]
- Kirk, J.T.O. Light and Photosynthesis in Aquatic Ecosystem, 2nd ed.; Cambridge University Press: Melbourne, Australia, 1994. [Google Scholar]
- Asakura, H.; Matsuse, U.; Nakagawa, K. Prevention of alkalization of regulating pond for rainwater by shading. J. Jpn. Soc. Civ. Eng. Ser. G Environ. Res. 2019, 75, I_393–I_401. (In Japanese) [Google Scholar] [CrossRef]
- Yoshinaga, I.; Hamada, K.; Miura, A.; Hitomi, T.; Takaki, K. Algae control and water quality change in a regulating reservoir using shading floats. Agric. Upl. 2007, 580, 2–14. (In Japanese) [Google Scholar]
- Kojima, S.; Iida, K.; Namekawa, A. Demonstration of algae (blue-green algae) control by localized shading of lake surface. J. Jpn. Biol. Soc. Water Waste 2001, 21, 101. (In Japanese) [Google Scholar]
- Haghighi, E.; Madani, K.; Hoekstra, A.Y. The water footprint of water conservation using shade balls in California. Nat. Sustain. 2018, 1, 358–360. [Google Scholar] [CrossRef]
- Furusato, E.; Asaeda, T.; Manatunge, J. Tolerance for prolonged darkness of three phytoplankton species, Microcystis aeruginosa (Cyanophyceae), Scenedesmus quadricauda (Chlorophyceae), and Melosira ambigua (Bacillariophyceae). Hydrobiologia 2004, 527, 153–162. [Google Scholar] [CrossRef]
- Chen, X.; He, S.; Huang, Y.; Kong, H.; Lin, Y.; Li, C.; Zeng, G. Laboratory investigation of reducing two algae from eutrophic water treated with light-shading plus aeration. Chemosphere 2009, 76, 1303–1307. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.C.; Kong, H.N.; He, S.B.; Wu, D.Y.; Li, C.J.; Huang, X.C. Reducing harmful algae in raw water by light-shading. Process Biochem. 2009, 44, 357–360. [Google Scholar] [CrossRef]
- Li, W.; Guo, Y.; Fu, K. Enclosure experiment for influence on algae growth by shading light. Procedia Environ. Sci. 2011, 10, 1823–1828. [Google Scholar] [CrossRef] [Green Version]
- Visser, P.M.; Passarge, J.; Mur, L.R. Modelling vertical migration of the cyanobacterium Microcystis. Hydrobiologia 1997, 349, 99–109. [Google Scholar] [CrossRef]
- Yoshida, Y.; Uemura, Y.; Kawahara, O. Study on the effect of mixing conditions associated with flow on photosynthesis of phytoplankton. In Proceedings of the 59th Annual Conference of the Japan Society of Civil Engineers, Aichi Institute of Technology, Toyota, Japan, 8 September 2004; pp. 271–272. (In Japanese). [Google Scholar]
- Umeda, M.; Tomioka, S. Data analysis on relations between water quality and phytoplankton growth in reservoirs. Annu. J. Hydraul. Eng. JSCE 2007, 51, 1373–1378. (In Japanese) [Google Scholar] [CrossRef]
- Okada, M. Vertical migration of blue-green algae (Microcystis aeruginosa) and its growth. J. Environ. Conserv. Eng. 1980, 9, 627–629. (In Japanese) [Google Scholar] [CrossRef]
- Rijn, J.; Shilo, M. Carbohydrate fluctuations, gas vacuolation, and vertical migration of scum-forming cyanobacteria in fishponds. Limnol. Oceanogr. 1985, 30, 1219–1228. [Google Scholar] [CrossRef]
| Category | Abbreviation | Description |
| Large section | LN | non-shaded |
| LC | shaded | |
| Small section | S00 | 0% shaded |
| S40 | 40% shaded | |
| S60 | 60% shaded | |
| S80 | 80% shaded | |
| S100 | 100% shaded | |
| Entire section | OUT | Outside shaded section |
| EFL | Effluent from entire shaded section |
| Large section | Small section | Entire section | ||||||||
| LN | LC | S00 | S40 | S60 | S80 | S100 | OUT | EFL | ||
| Temp. | °C | 20.1 | 20.7 | 20.3 | 20.2 | 20.1 | 20.1 | 20.1 | 20.4 | 20.4 |
| pH | − | 7.16 | 7.13 | 7.19 | 7.13 | 7.09 | 6.96 | 6.87 | 7.28 | 7.15 |
| EC | µS/cm | 585 | 584 | 577 | 564 | 575 | 571 | 567 | 583 | 574 |
| ORP | mV | 210 | 227 | 262 | 260 | 257 | 254 | 245 | 266 | 267 |
| DO | mg-O2/L | 9.0 | 8.5 | 8.5 | 8.5 | 8.4 | 8.1 | 7.5 | 8.6 | 8.3 |
| Chlorophyll | µg/L | 4.9 | 8.6 | 4.9 | 7.4 | 7.6 | 15.5 | 13.0 | 4.4 | 5.8 |
| TOC | mg-C/L | 5.5 | 4.9 | 5.1 | 5.0 | 4.8 | 4.7 | 4.7 | 5.2 | 5.1 |
| IC | mg-C/L | 6.0 | 5.8 | 5.5 | 5.0 | 5.2 | 5.0 | 4.8 | 5.9 | 6.0 |
| T-N | mg-N/L | 2.9 | 3.5 | 3.7 | 4.1 | 3.9 | 3.9 | 3.8 | 4.3 | 3.7 |
| T-P | mg-P/L | 0.17 | 0.14 | 0.16 | 0.13 | 0.13 | 0.12 | 0.17 | 0.13 | 0.12 |
| S00 | S40 | S60 | S80 | S100 | |
| S00 | − | − | 0.70 | 0.96 | 0.96 |
| S40 | 0.238 | − | − | 0.93 | 0.96 |
| S60 | 0.012 | 0.256 | − | 0.89 | 0.96 |
| S80 | <0.001 | <0.001 | <0.001 | − | 0.74 |
| S100 | <0.001 | <0.001 | <0.001 | 0.029 | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asakura, H.; Matsuse, U.; Nakagawa, K. Suppression of Alkalization in Rainwater Regulating Reservoir by Shading on a Pilot Scale. Water 2021, 13, 2557. https://doi.org/10.3390/w13182557
Asakura H, Matsuse U, Nakagawa K. Suppression of Alkalization in Rainwater Regulating Reservoir by Shading on a Pilot Scale. Water. 2021; 13(18):2557. https://doi.org/10.3390/w13182557
Chicago/Turabian StyleAsakura, Hiroshi, Umio Matsuse, and Kei Nakagawa. 2021. "Suppression of Alkalization in Rainwater Regulating Reservoir by Shading on a Pilot Scale" Water 13, no. 18: 2557. https://doi.org/10.3390/w13182557
APA StyleAsakura, H., Matsuse, U., & Nakagawa, K. (2021). Suppression of Alkalization in Rainwater Regulating Reservoir by Shading on a Pilot Scale. Water, 13(18), 2557. https://doi.org/10.3390/w13182557








