Enhanced Single and Simultaneous As(III) Adsorption in Pearl River Delta Water by Hexylamine Functionalized Vermiculite
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Chemicals
2.2. Preparation of Functionalized Materials
2.3. Characterization and Equipment
2.4. Potentiometric Titration Experiment
2.5. Adsorption and Desorption Experiments
3. Results and Discussion
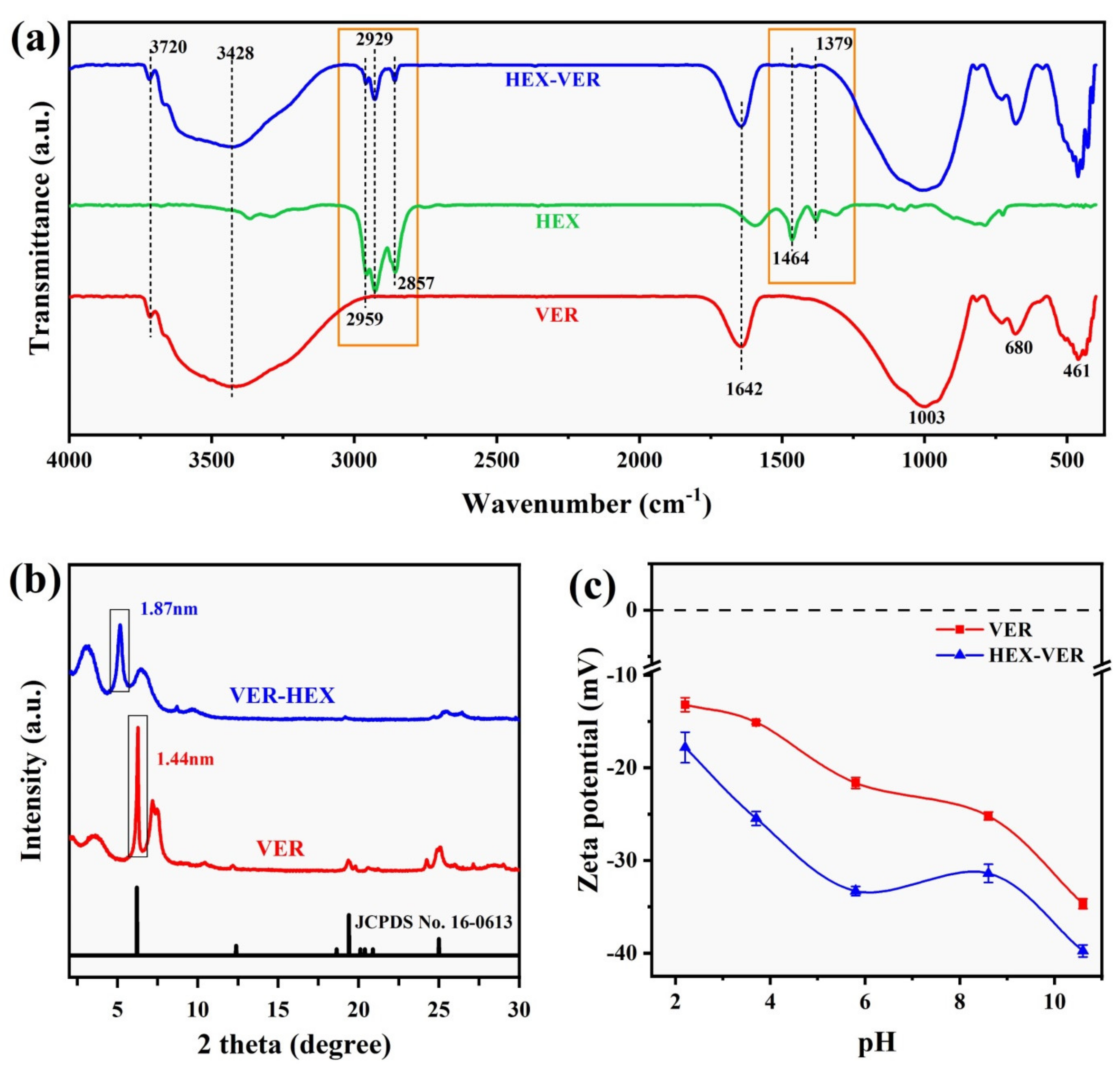
3.1. Characterization Results
3.1.1. Spectroscopy
3.1.2. BET Analysis and Surface Morphology
3.1.3. Thermogravimetric Analysis (TGA)
3.1.4. Potentiometric Analysis
3.2. Adsorption Results
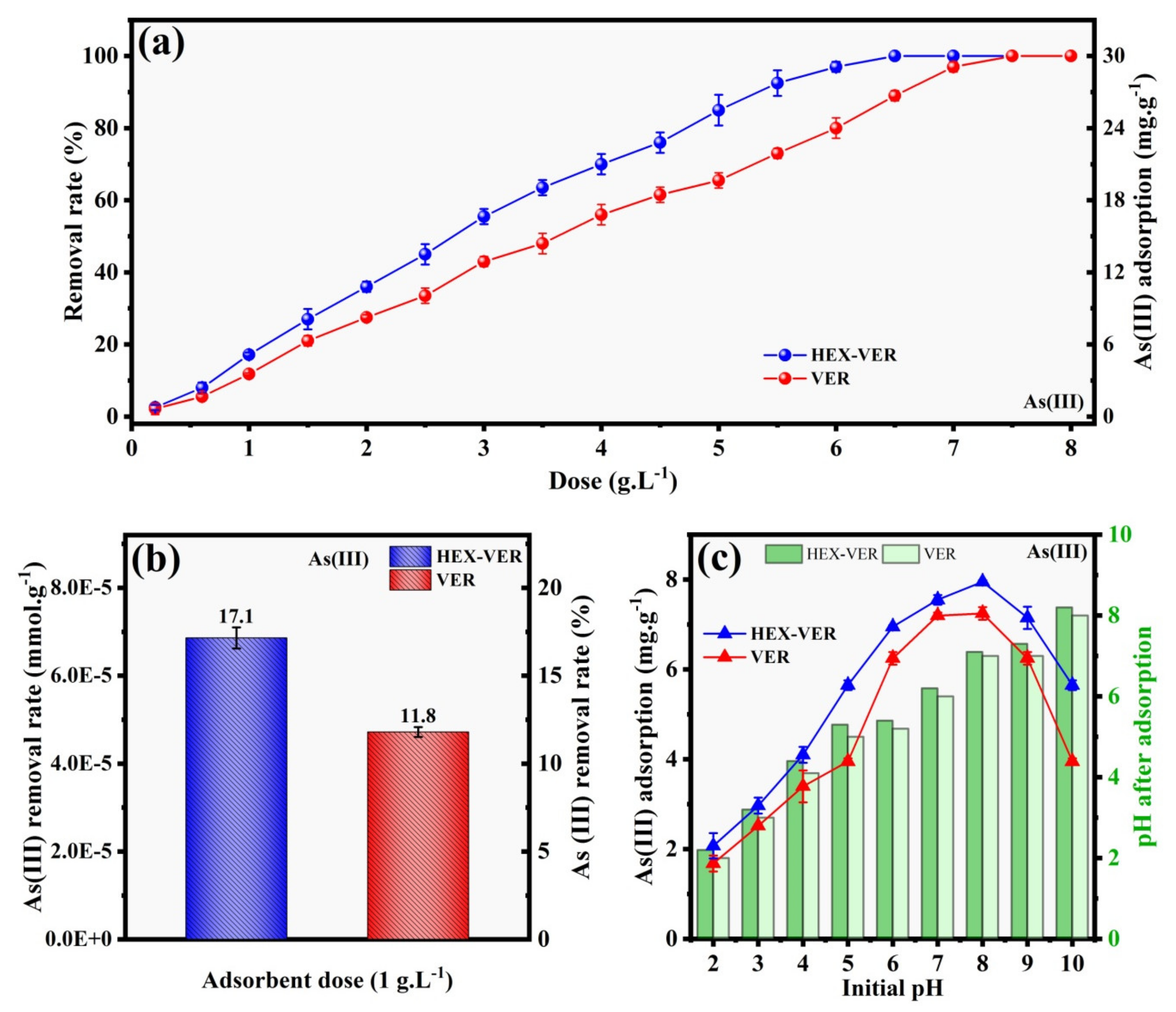
3.2.1. The Effect of Dose and pH
- As(III)mmol.g-1: (HEX-VER, 6.84 × 10−5) > (VER, 4.72 × 10−5)
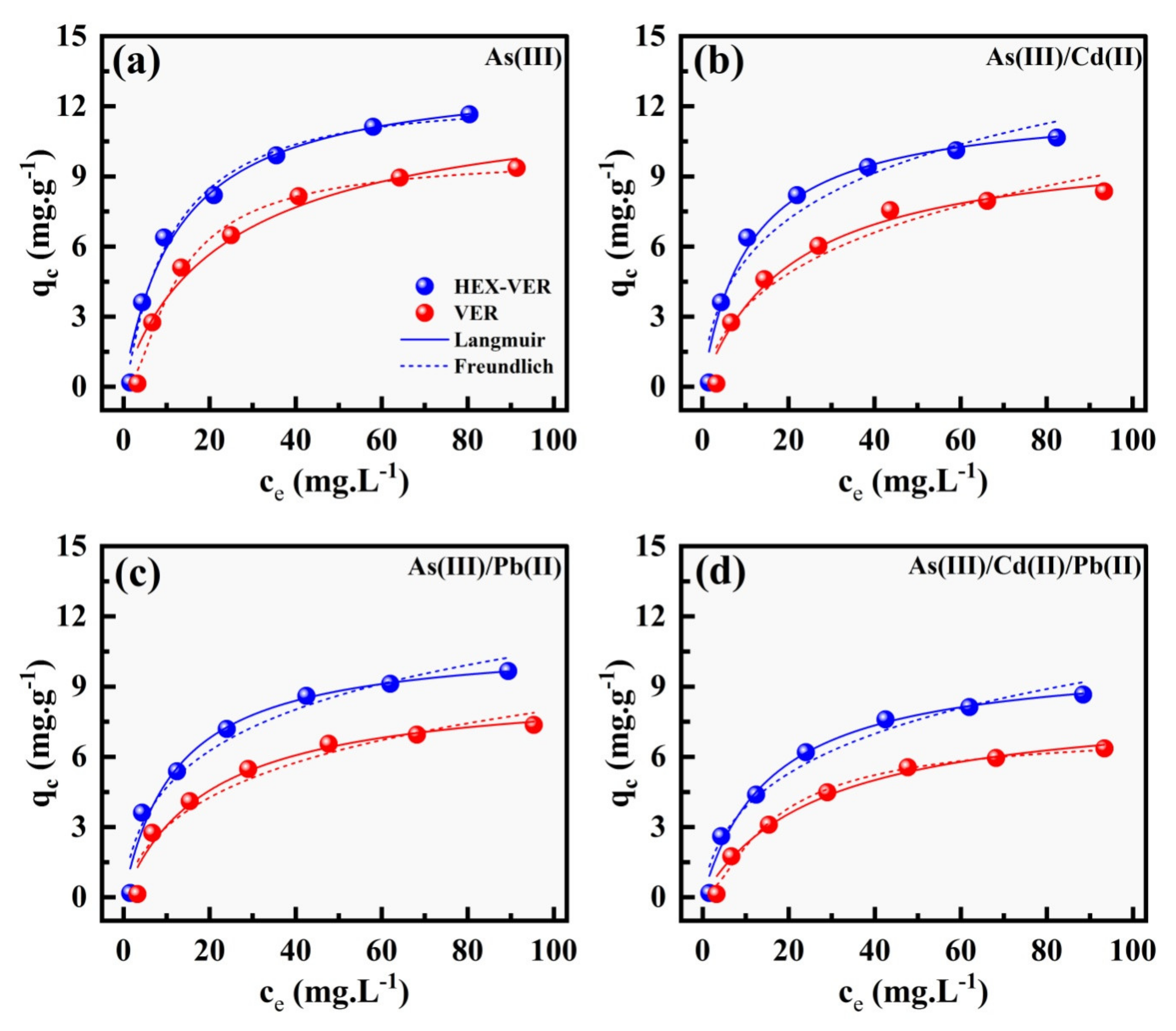
3.2.2. Adsorption Isotherm Experiments
3.2.3. Adsorption Kinetics
3.2.4. Adsorption Thermodynamics
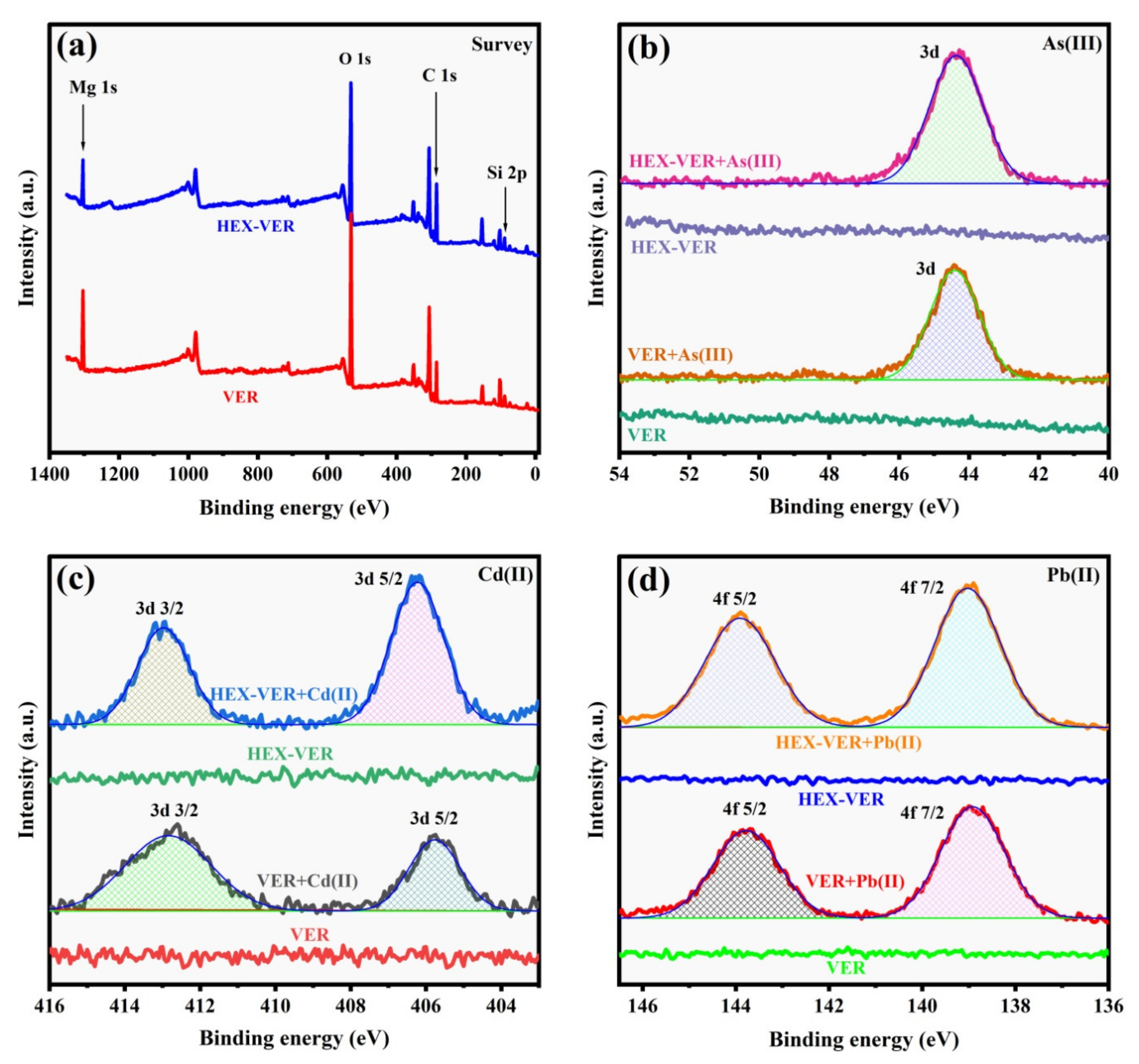
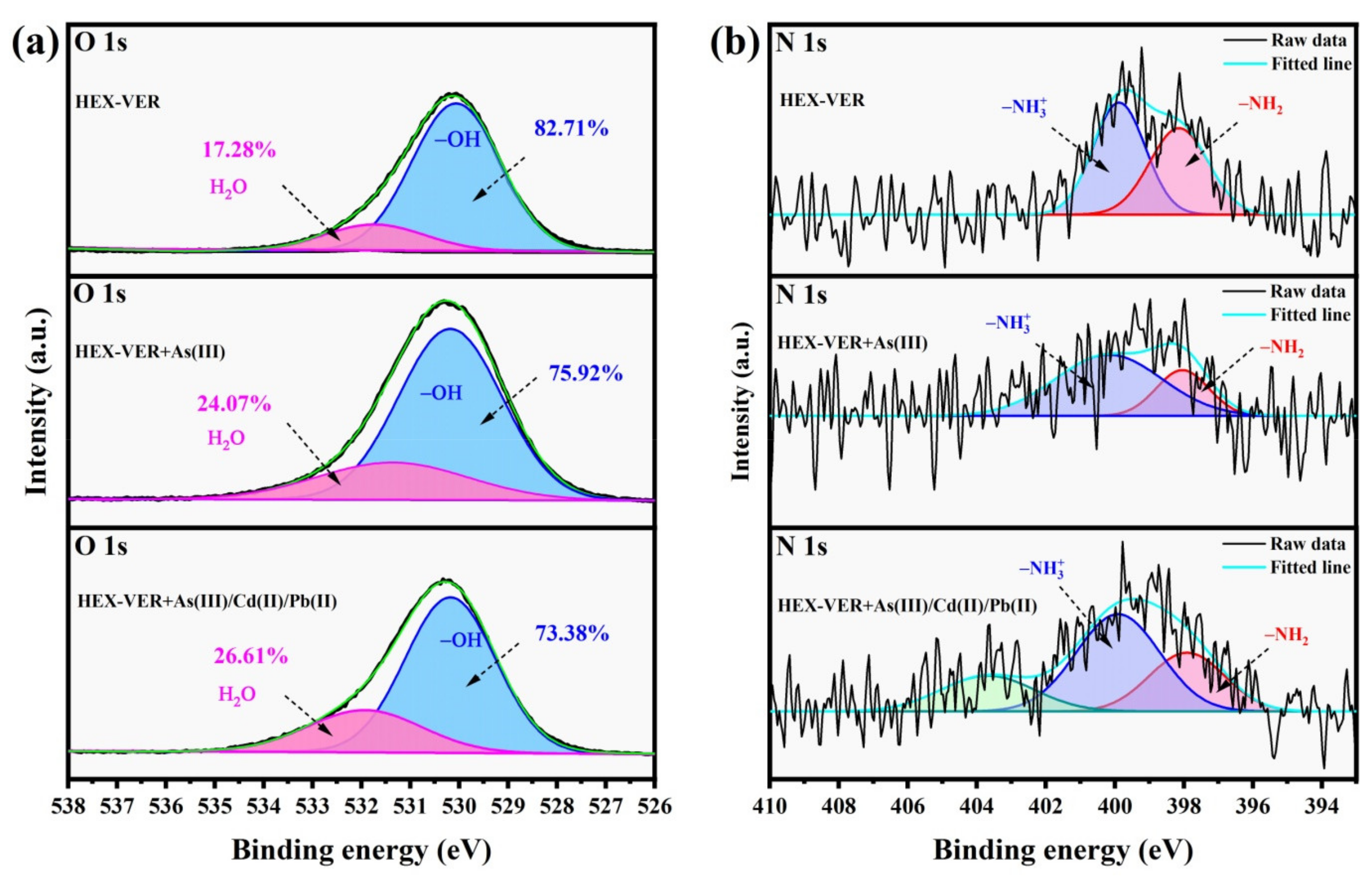
3.3. XPS Analysis
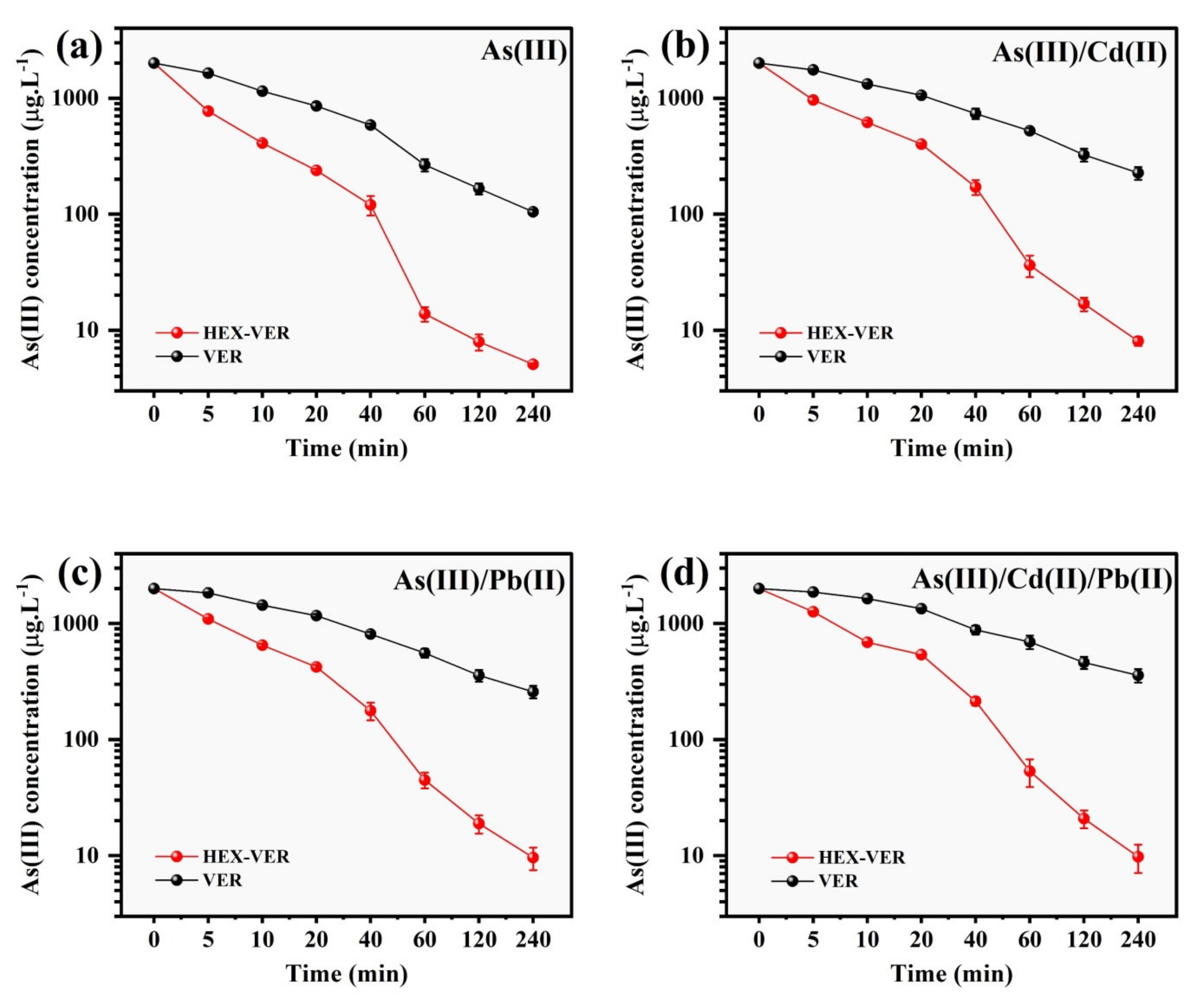
3.4. Effect of Co-Existing Ions
3.5. Pearl River (ZhuJiang) Water Adsorption Test
3.6. Desorption
3.7. Adsorption Mechanism Overview
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Song, C.; He, J.; Wu, L.; Jin, T.; Chen, X.; Li, R.; Ren, P.; Zhang, L.; Mao, H. Health burden attributable to ambient pm2.5 in china. Environ. Pollut. 2017, 223, 575–586. [Google Scholar] [CrossRef]
- Ye, F.; Huang, X.; Zhang, D.; Tian, L.; Zeng, Y. Distribution of heavy metals in sediments of the pearl river estuary, southern china: Implications for sources and historical changes. J. Environ. Sci. 2012, 24, 579–588. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J. Arsenite oxidation-enhanced photocatalytic degradation of phenolic pollutants on platinized tio2. Environ. Sci. Technol. 2014, 48, 13384–13391. [Google Scholar] [CrossRef]
- Liu, Q.; Huang, C.; Chris Le, X. Arsenic species in electronic cigarettes: Determination and potential health risk. J. Environ. Sci. 2020, 91, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, P.; Li, S.; Chen, M.; Cai, W.; Zou, D.; Zhu, N.; Dang, Z. Synergistic deep removal of as(iii) and cd(ii) by a calcined multifunctional mgznfe-co3 layered double hydroxide: Photooxidation, precipitation and adsorption. Chemosphere 2019, 225, 115–125. [Google Scholar] [CrossRef]
- Wang, S.; Cao, X.; Lin, C.; Chen, X. Arsenic content and fractionation in the surface sediments of the guangzhou section of the pearl river in southern china. J. Hazard. Mater. 2010, 183, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.H.; Yuan, H.X.; Hu, Y.G.; Wu, Z.F.; Zhu, L.A.; Zhu, L.; Li, F.B.; Li, D.Q. Spatial distribution and vertical variation of arsenic in guangdong soil profiles, china. Environ. Pollut. 2006, 144, 492–499. [Google Scholar] [CrossRef]
- Luo, C.; Routh, J.; Luo, D.; Wei, L.; Liu, Y. Arsenic in the pearl river delta and its related waterbody, south china: Occurrence and sources, a review. Geosci. Lett. 2021, 8, 12. [Google Scholar] [CrossRef]
- Choong, C.E.; Wong, K.T.; Jang, S.B.; Saravanan, P.; Park, C.; Kim, S.-H.; Jeon, B.-H.; Choi, J.; Yoon, Y.; Jang, M. Granular mg-fe layered double hydroxide prepared using dual polymers: Insights into synergistic removal of as(iii) and as(v). J. Hazard. Mater. 2021, 403, 123883. [Google Scholar] [CrossRef]
- Duker, A.A.; Carranza, E.J.M.; Hale, M. Arsenic geochemistry and health. Environ. Int. 2005, 31, 631–641. [Google Scholar] [CrossRef]
- Straif, K.; Benbrahim-Tallaa, L.; Baan, R.; Grosse, Y.; Secretan, B.; El Ghissassi, F.; Bouvard, V.; Guha, N.; Freeman, C.; Galichet, L.; et al. A review of human carcinogens—Part c: Metals, arsenic, dusts, and fibres. Lancet Oncol. 2009, 10, 453–454. [Google Scholar] [CrossRef]
- Gomez-Caminero, A.; Howe, P.D.; Hughes, M.; Kenyon, E.; Lewis, D.R.; Moore, M.; Aitio, A.; Becking, G.C.; Ng, J.; International Programme on Chemical Safety; et al. Arsenic and Arsenic Compounds; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Cui, J.-L.; Zhao, Y.-P.; Li, J.-S.; Beiyuan, J.-Z.; Tsang, D.C.W.; Poon, C.-S.; Chan, T.-S.; Wang, W.-X.; Li, X.-D. Speciation, mobilization, and bioaccessibility of arsenic in geogenic soil profile from hong kong. Environ. Pollut. 2018, 232, 375–384. [Google Scholar] [CrossRef]
- Zeng, J.; Han, G.; Wu, Q.; Tang, Y. Heavy metals in suspended particulate matter of the zhujiang river, southwest china: Contents, sources, and health risks. Int. J. Environ. Res. Public Health 2019, 16, 1843. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Guo, S.; Wu, B. The source, spatial distribution and risk assessment of heavy metals in soil from the pearl river delta based on the national multi-purpose regional geochemical survey. PLoS ONE 2015, 10, e0132040. [Google Scholar] [CrossRef] [PubMed]
- McConnell, J.R.; Wilson, A.I.; Stohl, A.; Arienzo, M.M.; Chellman, N.J.; Eckhardt, S.; Thompson, E.M.; Pollard, A.M.; Steffensen, J.P. Lead pollution recorded in greenland ice indicates european emissions tracked plagues, wars, and imperial expansion during antiquity. Proc. Natl. Acad. Sci. USA 2018, 115, 5726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, Q.; Huang, Y.; Huang, L.; Hu, L.; Sun, W.; Zhong, H.; He, Z. High adsorption capacity and super selectivity for pb(ⅱ) by a novel adsorbent: Nano humboldtine/almandine composite prepared from natural almandine. Chemosphere 2020, 253, 126650. [Google Scholar] [CrossRef]
- Deng, X.; Lü, L.; Li, H.; Luo, F. The adsorption properties of pb(ii) and cd(ii) on functionalized graphene prepared by electrolysis method. J. Hazard. Mater. 2010, 183, 923–930. [Google Scholar] [CrossRef]
- Thabede, P.M.; Shooto, N.D.; Xaba, T.; Naidoo, E.B. Adsorption studies of toxic cadmium(ii) and chromium(vi) ions from aqueous solution by activated black cumin (nigella sativa) seeds. J. Environ. Chem. Eng. 2020, 8, 104045. [Google Scholar] [CrossRef]
- McManamon, C.; Burke, A.M.; Holmes, J.D.; Morris, M.A. Amine-functionalised sba-15 of tailored pore size for heavy metal adsorption. J. Colloid Interface Sci. 2012, 369, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Katsumata, H.; Kaneco, S.; Inomata, K.; Itoh, K.; Funasaka, K.; Masuyama, K.; Suzuki, T.; Ohta, K. Removal of heavy metals in rinsing wastewater from plating factory by adsorption with economical viable materials. J. Environ. Manag. 2003, 69, 187–191. [Google Scholar] [CrossRef]
- Pouya, E.S.; Abolghasemi, H.; Esmaieli, M.; Fatoorehchi, H.; Hashemi, S.J.; Salehpour, A. Batch adsorptive removal of benzoic acid from aqueous solution onto modified natural vermiculite: Kinetic, isotherm and thermodynamic studies. J. Ind. Eng. Chem. 2015, 31, 199–215. [Google Scholar] [CrossRef]
- Jayanthi, G.; Sumathi, S.; Andal, V. Synthesis and applications of perovskite in heavy metal ions removal-a brief perspective. Mater. Today Proc. 2021, (in press). [Google Scholar] [CrossRef]
- Hamid, Y.; Tang, L.; Hussain, B.; Usman, M.; Liu, L.; Ulhassan, Z.; He, Z.; Yang, X. Sepiolite clay: A review of its applications to immobilize toxic metals in contaminated soils and its implications in soil–plant system. Environ. Technol. Innov. 2021, 23, 101598. [Google Scholar] [CrossRef]
- Barraqué, F.; Montes, M.L.; Fernández, M.A.; Candal, R.; Torres Sánchez, R.M.; Marco-Brown, J.L. Arsenate removal from aqueous solution by montmorillonite and organo-montmorillonite magnetic materials. Environ. Res. 2021, 192, 110247. [Google Scholar] [CrossRef] [PubMed]
- Roshanfekr Rad, L.; Anbia, M. Zeolite-based composites for the adsorption of toxic matters from water: A review. J. Environ. Chem. Eng. 2021, 9, 106088. [Google Scholar] [CrossRef]
- Uddin, M.K. A review on the adsorption of heavy metals by clay minerals, with special focus on the past decade. Chem. Eng. J. 2017, 308, 438–462. [Google Scholar] [CrossRef]
- Crini, G.; Badot, P.-M. Sorption Processes and Pollution: Conventional and Non-Conventional Sorbents for Pollutant Removal from Wastewaters; Presses Universitaires Franche-Comté: Besançon, France, 2010. [Google Scholar]
- Schulze, D.G. Clay minerals. In Encyclopedia of Soils in the Environment; Hillel, D., Ed.; Elsevier: Oxford, UK, 2005; pp. 246–254. [Google Scholar]
- Yang, S.; Huang, Z.; Li, C.; Li, W.; Yang, L.; Wu, P. Individual and simultaneous adsorption of tetracycline and cadmium by dodecyl dimethyl betaine modified vermiculite. Colloids Surf. A: Physicochem. Eng. Asp. 2020, 602, 125171. [Google Scholar] [CrossRef]
- Yu, M.; Gao, M.; Shen, T.; Zeng, H. Single and simultaneous adsorption of rhodamine b and congo red from aqueous solution by organo-vermiculites. J. Mol. Liq. 2019, 292, 111408. [Google Scholar] [CrossRef]
- Chen, L.; Wu, P.; Chen, M.; Lai, X.; Ahmed, Z.; Zhu, N.; Dang, Z.; Bi, Y.; Liu, T. Preparation and characterization of the eco-friendly chitosan/vermiculite biocomposite with excellent removal capacity for cadmium and lead. Appl. Clay Sci. 2018, 159, 74–82. [Google Scholar] [CrossRef]
- Długosz, O.; Banach, M. Sorption of ag+ and cu2+ by vermiculite in a fixed-bed column: Design, process optimization and dynamics investigations. Appl. Sci. 2018, 8, 2221. [Google Scholar] [CrossRef] [Green Version]
- Freitas, E.D.d.; Almeida, H.J.d.; Vieira, M.G.A. Binary adsorption of zinc and copper on expanded vermiculite using a fixed bed column. Appl. Clay Sci. 2017, 146, 503–509. [Google Scholar] [CrossRef]
- Li, F.; Wan, Y.; Chen, J.; Hu, X.; Tsang, D.C.W.; Wang, H.; Gao, B. Novel ball-milled biochar-vermiculite nanocomposites effectively adsorb aqueous as(ⅴ). Chemosphere 2020, 260, 127566. [Google Scholar] [CrossRef] [PubMed]
- Sayari, A.; Hamoudi, S. Periodic mesoporous silica-based organic−inorganic nanocomposite materials. Chem. Mater. 2001, 13, 3151–3168. [Google Scholar] [CrossRef]
- Aguado, J.; Arsuaga, J.M.; Arencibia, A.; Lindo, M.; Gascón, V. Aqueous heavy metals removal by adsorption on amine-functionalized mesoporous silica. J. Hazard. Mater. 2009, 163, 213–221. [Google Scholar] [CrossRef]
- Ahmed, Z.; Wu, P.; Jiang, L.; Liu, J.; Ye, Q.; Yang, Q.; Zhu, N. Enhanced simultaneous adsorption of cd(ii) and pb(ii) on octylamine functionalized vermiculite. Colloids Surf. A: Physicochem. Eng. Asp. 2020, 604, 125285. [Google Scholar] [CrossRef]
- Nkuigue Fotsing, P.; Bouazizi, N.; Djoufac Woumfo, E.; Mofaddel, N.; Le Derf, F.; Vieillard, J. Investigation of chromate and nitrate removal by adsorption at the surface of an amine-modified cocoa shell adsorbent. J. Environ. Chem. Eng. 2021, 9, 104618. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, C.; Zhao, W.; Hua, Z.; Chen, H.; Li, L.; Shi, J. Preparation of multi-amine-grafted mesoporous silicas and their application to heavy metal ions adsorption. J. Non-Cryst. Solids 2007, 353, 4055–4061. [Google Scholar] [CrossRef]
- National Centre for Biotechnology Information. Hexylamine. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Hexylamine (accessed on 19 July 2021).
- Moraes, D.S.; Rodrigues, E.M.S.; Lamarão, C.N.; Marques, G.T.; Rente, A.F.S. New sodium activated vermiculite process. Testing on cu2+ removal from tailing dam waters. J. Hazard. Mater. 2019, 366, 34–38. [Google Scholar] [CrossRef]
- Santos, S.S.G.; Silva, H.R.M.; de Souza, A.G.; Alves, A.P.M.; da Silva Filho, E.C.; Fonseca, M.G. Acid-leached mixed vermiculites obtained by treatment with nitric acid. Appl. Clay Sci. 2015, 104, 286–294. [Google Scholar] [CrossRef]
- Kaur, S.; Singh, S.; Singh, L. Opto-electric and physio-chemical changes in oxygen ion irradiated natural vermiculite mineral. Appl. Radiat. Isot. 2019, 148, 7–12. [Google Scholar] [CrossRef]
- Anandhan, K.; Thilak Kumar, R. Synthesis, ftir, uv–vis and photoluminescence characterizations of triethanolamine passivated cdo nanostructures. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2015, 149, 476–480. [Google Scholar] [CrossRef]
- Yang, X.; Mei, T.; Yang, J.; Zhang, C.; Lv, M.; Wang, X. Synthesis and characterization of alkylamine-functionalized graphene for polyolefin-based nanocomposites. Appl. Surf. Sci. 2014, 305, 725–731. [Google Scholar] [CrossRef]
- Xu, Y.; Khan, M.A.; Wang, F.; Xia, M.; Lei, W. Novel multi amine-containing gemini surfactant modified montmorillonite as adsorbents for removal of phenols. Appl. Clay Sci. 2018, 162, 204–213. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, S.; Sun, D.; Xu, J. Aqueous foams stabilized by hexylamine-modified laponite particles. Colloids Surf. A: Physicochem. Eng. Asp. 2009, 338, 40–46. [Google Scholar] [CrossRef]
- Liu, M.; Feng, J.; Mo, W.; Su, X.; Fu, L. A novel quantitative analysis method of microwave energy absorption during vermiculite expansion process. Thermochim. Acta 2020, 691, 178718. [Google Scholar] [CrossRef]
- Liu, S.; Wu, P.; Yu, L.; Li, L.; Gong, B.; Zhu, N.; Dang, Z.; Yang, C. Preparation and characterization of organo-vermiculite based on phosphatidylcholine and adsorption of two typical antibiotics. Appl. Clay Sci. 2017, 137, 160–167. [Google Scholar] [CrossRef]
- Bourliva, A.; Sikalidis, A.K.; Papadopoulou, L.; Betsiou, M.; Michailidis, K.; Sikalidis, C.; Filippidis, A. Removal of cu2+ and ni2+ ions from aqueous solutions by adsorption onto natural palygorskite and vermiculite. Clay Miner. 2018, 53, 1–15. [Google Scholar] [CrossRef]
- Matusik, J.; Gaweł, A.; Bahranowski, K. Grafting of methanol in dickite and intercalation of hexylamine. Appl. Clay Sci. 2012, 56, 63–67. [Google Scholar] [CrossRef]
- Sim, J.-H.; Seo, H.-J.; Kim, C.-G. Physicochemical characteristics for adsorption of mtbe and cadmium on clay minerals. Environ. Earth Sci. 2009, 59, 537–545. [Google Scholar] [CrossRef]
- Liu, S.; Wu, P.; Chen, M.; Yu, L.; Kang, C.; Zhu, N.; Dang, Z. Amphoteric modified vermiculites as adsorbents for enhancing removal of organic pollutants: Bisphenol A and tetrabromobisphenol A. Environ. Pollut. 2017, 228, 277–286. [Google Scholar] [CrossRef]
- Heimenz, P.C. Principles of Colloid and Surface Chemistry; Marcel Dekker: New York, NY, USA, 1986. [Google Scholar]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Inglezakis, V.J.; Poulopoulos, S.G.; Kazemian, H. Insights into the s-shaped sorption isotherms and their dimensionless forms. Microporous Mesoporous Mater. 2018, 272, 166–176. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (recommendations 1984). Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Vilarrasa-García, E.; Cecilia, J.A.; Azevedo, D.C.S.; Cavalcante, C.L.; Rodríguez-Castellón, E. Evaluation of porous clay heterostructures modified with amine species as adsorbent for the co2 capture. Microporous Mesoporous Mater. 2017, 249, 25–33. [Google Scholar] [CrossRef]
- Zhao, X.; Hu, X.; Hu, G.; Bai, R.; Dai, W.; Fan, M.; Luo, M. Enhancement of co2 adsorption and amine efficiency of titania modified by moderate loading of diethylenetriamine. J. Mater. Chem. A 2013, 1, 6208–6215. [Google Scholar] [CrossRef]
- Long, H.; Wu, P.; Yang, L.; Huang, Z.; Zhu, N.; Hu, Z. Efficient removal of cesium from aqueous solution with vermiculite of enhanced adsorption property through surface modification by ethylamine. J. Colloid Interface Sci. 2014, 428, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Huggett, J.M. Clay minerals☆. In Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Tombácz, E.; Szekeres, M. Surface charge heterogeneity of kaolinite in aqueous suspension in comparison with montmorillonite. Appl. Clay Sci. 2006, 34, 105–124. [Google Scholar] [CrossRef]
- Avena, M.J.; De Pauli, C.P. Proton adsorption and electrokinetics of an argentinean montmorillonite. J. Colloid Interface Sci. 1998, 202, 195–204. [Google Scholar] [CrossRef]
- Helmy, A.K.; Ferreiro, E.A.; de Bussetti, S.G. Cation exchange capacity and condition of zero charge of hydroxy-al montmorillonite. Clays Clay Miner. 1994, 42, 444–450. [Google Scholar] [CrossRef]
- Ijagbemi, C.O.; Baek, M.-H.; Kim, D.-S. Montmorillonite surface properties and sorption characteristics for heavy metal removal from aqueous solutions. J. Hazard. Mater. 2009, 166, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Msadok, I.; Hamdi, N.; Gammoudi, S.; Rodríguez, M.A.; Srasra, E. Effect of cationic surfactant hdpy+ on the acidity and hydrophilicity of tunisian clay. Mater. Chem. Phys. 2019, 225, 279–283. [Google Scholar] [CrossRef]
- Yao, J.; Chen, Y.; Yu, H.; Liu, T.; Yan, L.; Du, B.; Cui, Y. Efficient and fast removal of pb(ii) by facile prepared magnetic vermiculite from aqueous solution. RSC Adv. 2016, 6, 101353–101360. [Google Scholar] [CrossRef]
- Lee, S.M.; Lalhmunsiama; Thanhmingliana; Tiwari, D. Porous hybrid materials in the remediation of water contaminated with as(iii) and as(v). Chem. Eng. J. 2015, 270, 496–507. [Google Scholar] [CrossRef]
- Bhowmick, S.; Chakraborty, S.; Mondal, P.; Van Renterghem, W.; Van den Berghe, S.; Roman-Ross, G.; Chatterjee, D.; Iglesias, M. Montmorillonite-supported nanoscale zero-valent iron for removal of arsenic from aqueous solution: Kinetics and mechanism. Chem. Eng. J. 2014, 243, 14–23. [Google Scholar] [CrossRef]
- Mudzielwana, R.; Gitari, M.W.; Ndungu, P. Performance evaluation of surfactant modified kaolin clay in as(iii) and as(v) adsorption from groundwater: Adsorption kinetics, isotherms and thermodynamics. Heliyon 2019, 5, e02756. [Google Scholar] [CrossRef] [Green Version]
- Zhao, M.; Tang, Z.; Liu, P. Removal of methylene blue from aqueous solution with silica nano-sheets derived from vermiculite. J. Hazard. Mater. 2008, 158, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, Y.; Cao, S.; Liu, S.; Chen, Z.; Chen, J.; Chen, Y.; Fu, J. Fabrication of core@shell structural fe-fe2o3@phcp nanochains with high saturation magnetization and abundant amino groups for hexavalent chromium adsorption and reduction. J. Hazard. Mater. 2020, 384, 121483. [Google Scholar] [CrossRef]
- Zhao, S.; Meng, Z.; Fan, X.; Jing, R.; Yang, J.; Shao, Y.; Liu, X.; Wu, M.; Zhang, Q.; Liu, A. Removal of heavy metals from soil by vermiculite supported layered double hydroxides with three-dimensional hierarchical structure. Chem. Eng. J. 2020, 390, 124554. [Google Scholar] [CrossRef]
- Jian, M.; Liu, B.; Zhang, G.; Liu, R.; Zhang, X. Adsorptive removal of arsenic from aqueous solution by zeolitic imidazolate framework-8 (zif-8) nanoparticles. Colloids Surf. A: Physicochem. Eng. Asp. 2015, 465, 67–76. [Google Scholar] [CrossRef]
- Wu, J.; Wang, Y.; Wu, Z.; Gao, Y.; Li, X. Adsorption properties and mechanism of sepiolite modified by anionic and cationic surfactants on oxytetracycline from aqueous solutions. Sci. Total Environ. 2020, 708, 134409. [Google Scholar] [CrossRef]
- Yan, L.; Hu, S.; Jing, C. Recent progress of arsenic adsorption on tio2 in the presence of coexisting ions: A review. J. Environ. Sci. 2016, 49, 74–85. [Google Scholar] [CrossRef]
- Qi, J.; Zhang, G.; Li, H. Efficient removal of arsenic from water using a granular adsorbent: Fe–mn binary oxide impregnated chitosan bead. Bioresour. Technol. 2015, 193, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Lata, S.; Singh, P.K.; Samadder, S.R. Regeneration of adsorbents and recovery of heavy metals: A review. Int. J. Environ. Sci. Technol. 2015, 12, 1461–1478. [Google Scholar] [CrossRef] [Green Version]
- Hua, J. Synthesis and characterization of bentonite based inorgano–organo-composites and their performances for removing arsenic from water. Appl. Clay Sci. 2015, 114, 239–246. [Google Scholar] [CrossRef]
- Malandrino, M.; Abollino, O.; Giacomino, A.; Aceto, M.; Mentasti, E. Adsorption of heavy metals on vermiculite: Influence of ph and organic ligands. J. Colloid Interface Sci. 2006, 299, 537–546. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, Z.; Wu, P.; Xu, Y.; Keerio, H.A.; Wu, J.; Ye, Q.; Niu, W.; Chen, M.; Dang, Z. Enhanced Single and Simultaneous As(III) Adsorption in Pearl River Delta Water by Hexylamine Functionalized Vermiculite. Water 2021, 13, 2412. https://doi.org/10.3390/w13172412
Ahmed Z, Wu P, Xu Y, Keerio HA, Wu J, Ye Q, Niu W, Chen M, Dang Z. Enhanced Single and Simultaneous As(III) Adsorption in Pearl River Delta Water by Hexylamine Functionalized Vermiculite. Water. 2021; 13(17):2412. https://doi.org/10.3390/w13172412
Chicago/Turabian StyleAhmed, Zubair, Pingxiao Wu, Yijing Xu, Hareef Ahmed Keerio, Jiayan Wu, Quanyun Ye, Wenchao Niu, Meiqing Chen, and Zhi Dang. 2021. "Enhanced Single and Simultaneous As(III) Adsorption in Pearl River Delta Water by Hexylamine Functionalized Vermiculite" Water 13, no. 17: 2412. https://doi.org/10.3390/w13172412






