Groundwater Quality Issues and Challenges for Drinking and Irrigation Uses in Central Ganga Basin Dominated with Rice-Wheat Cropping System
Abstract
:1. Introduction
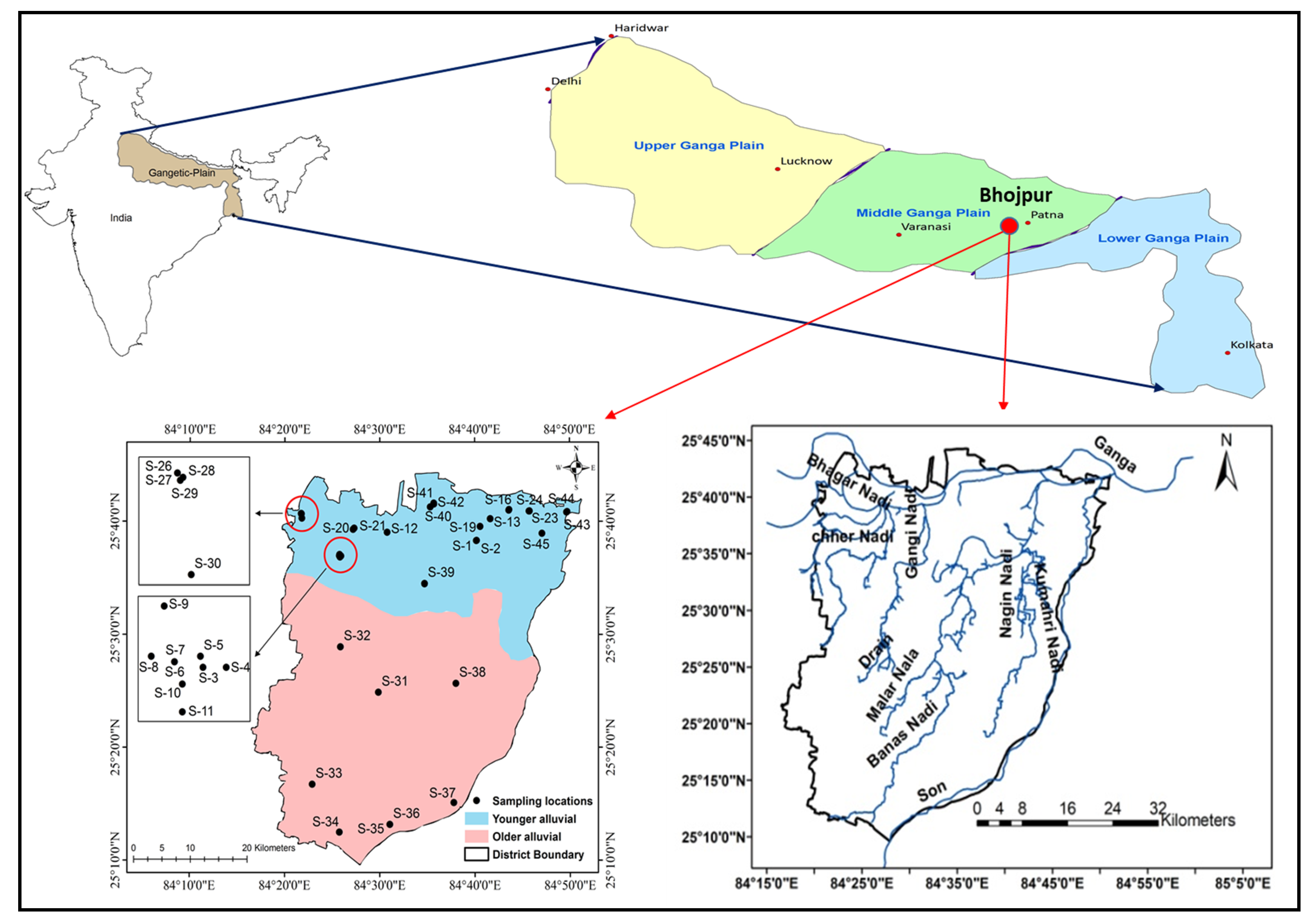
2. Study Area
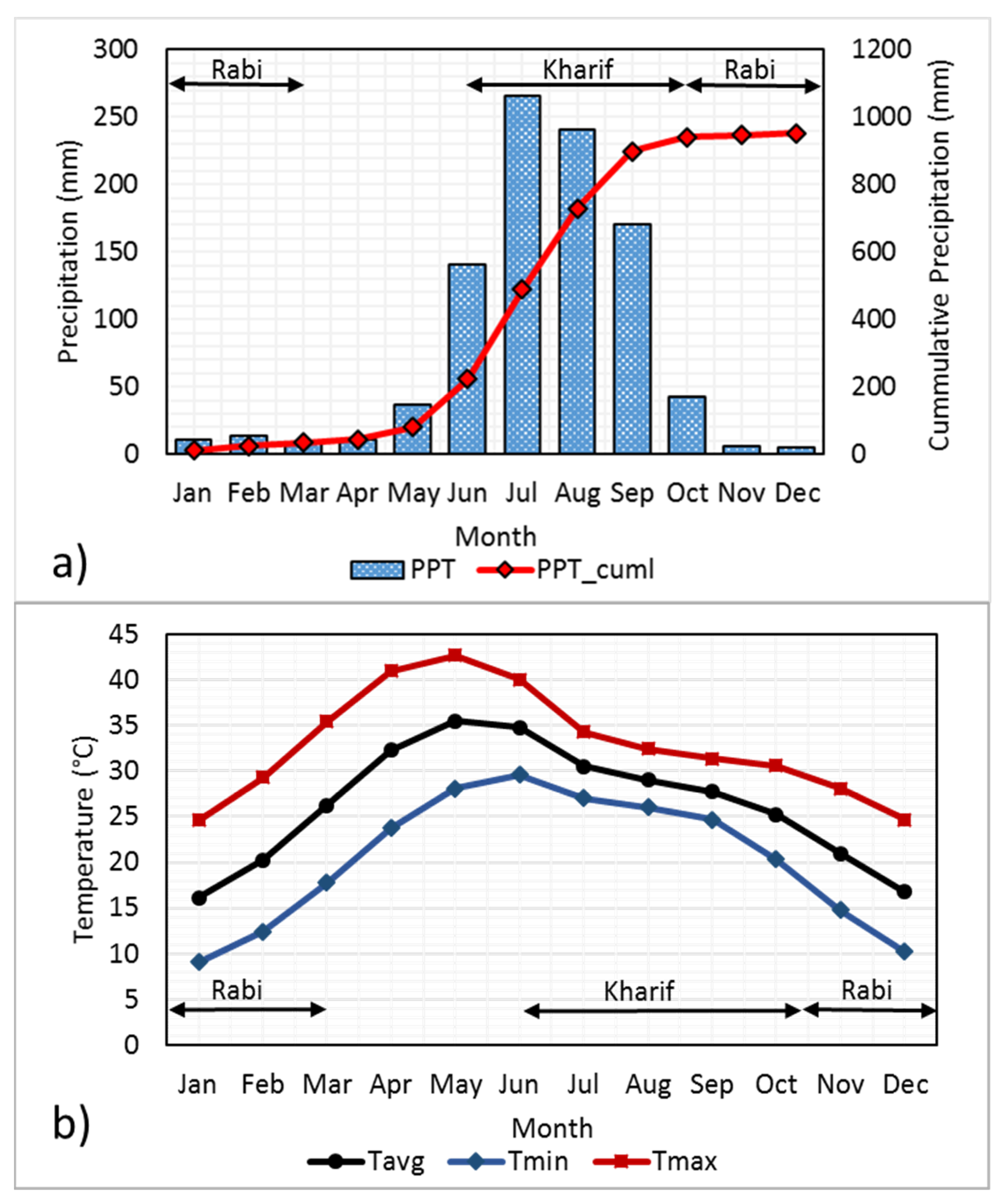
2.1. Hydroclimatology
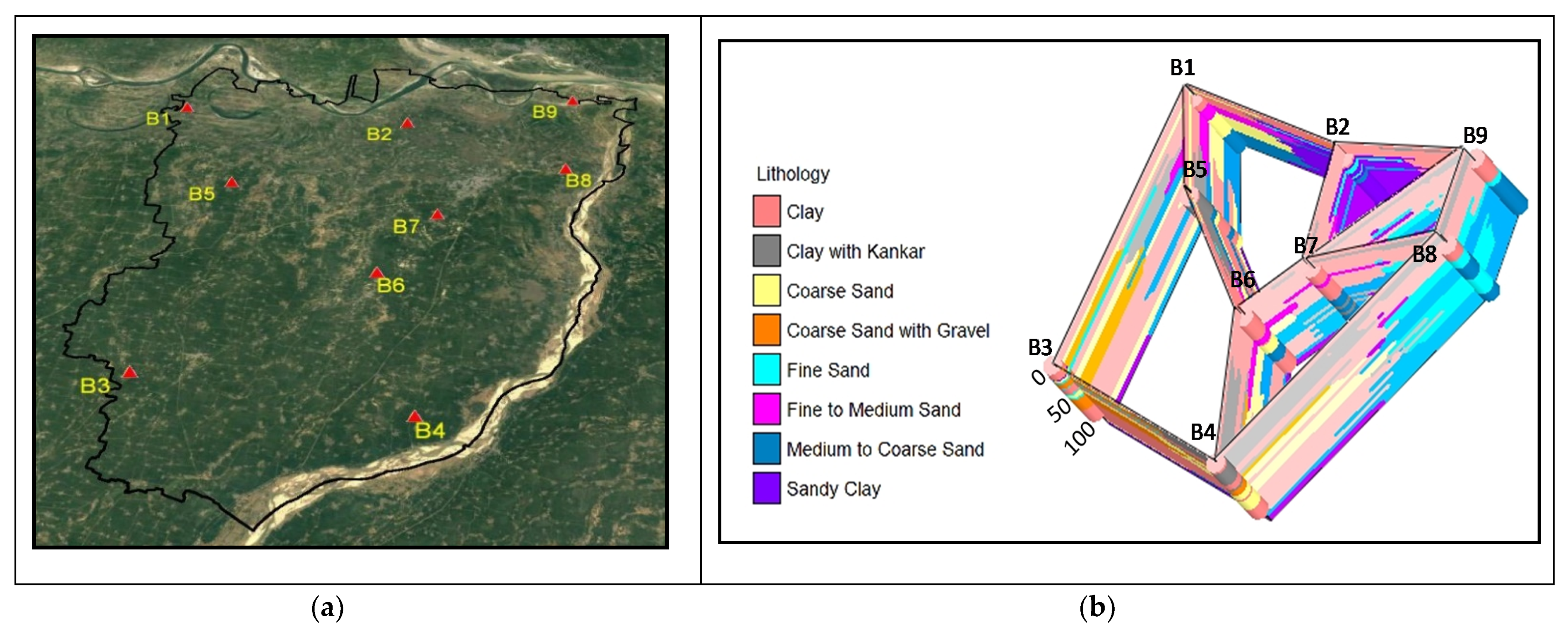
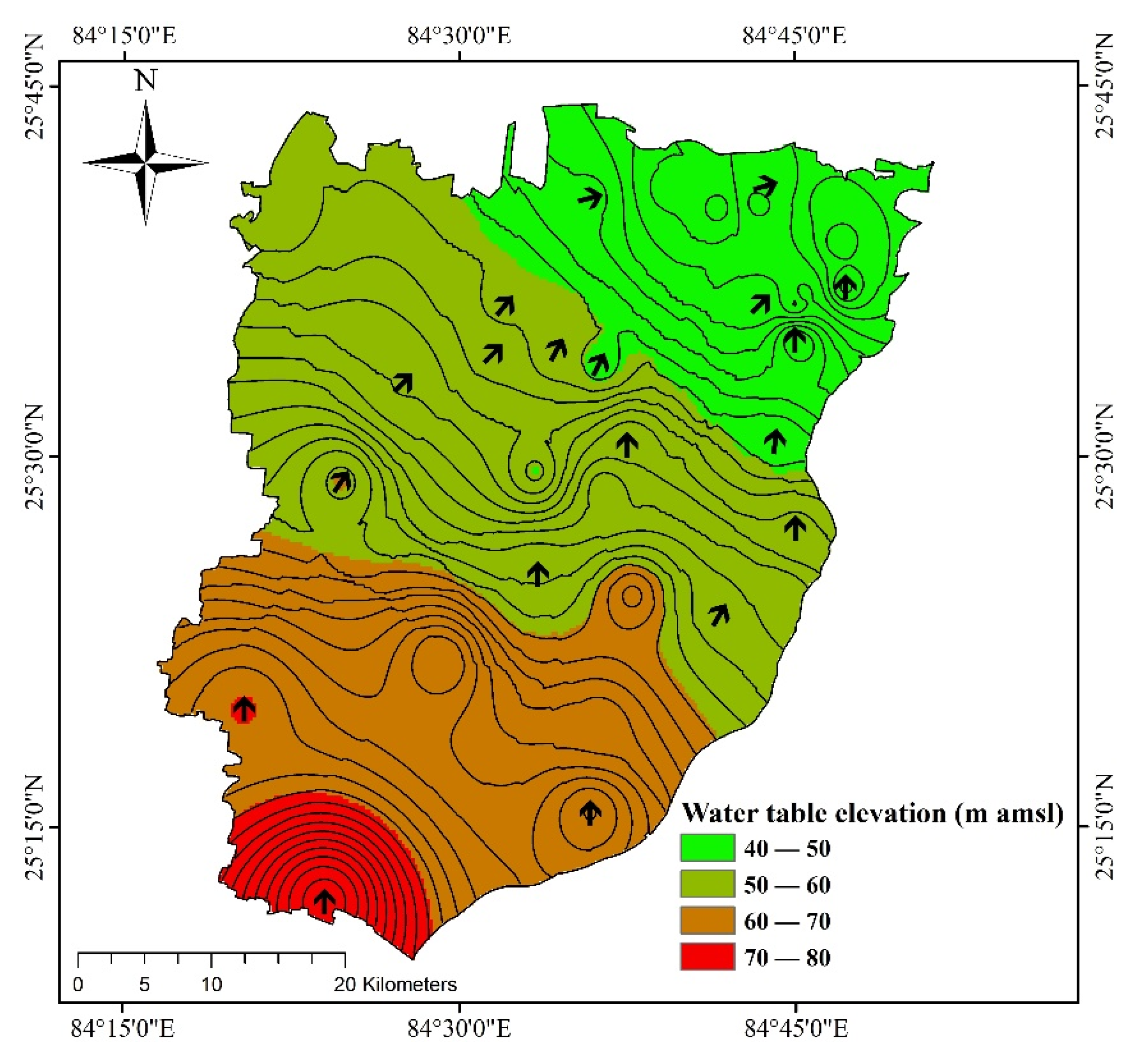
2.2. Hydrogeology
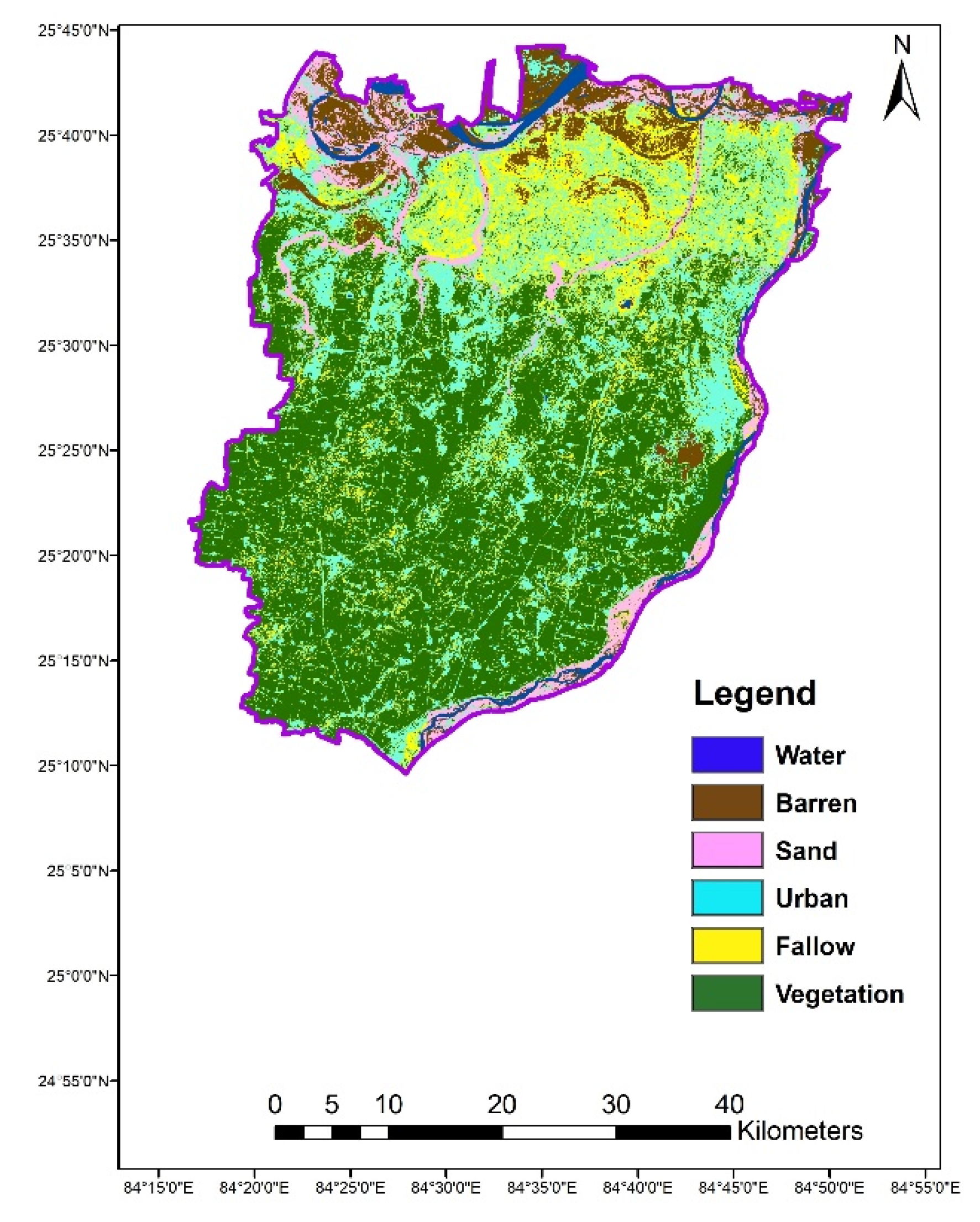
2.3. Land Use Land Cover
2.4. Agricultural Practices
3. Methodology
3.1. Data Collection and Instrumentation
3.2. Electrical Conductivity (EC) and Sodium Percentage (Na%)
3.3. Alkali and Salinity Hazard (SAR)
3.4. Residual Sodium Carbonate (RSC)
3.5. Chloroalkaline Indices (CAI-I and CAI-II)
3.6. Permeability Index (PI)
4. Results and Discussion
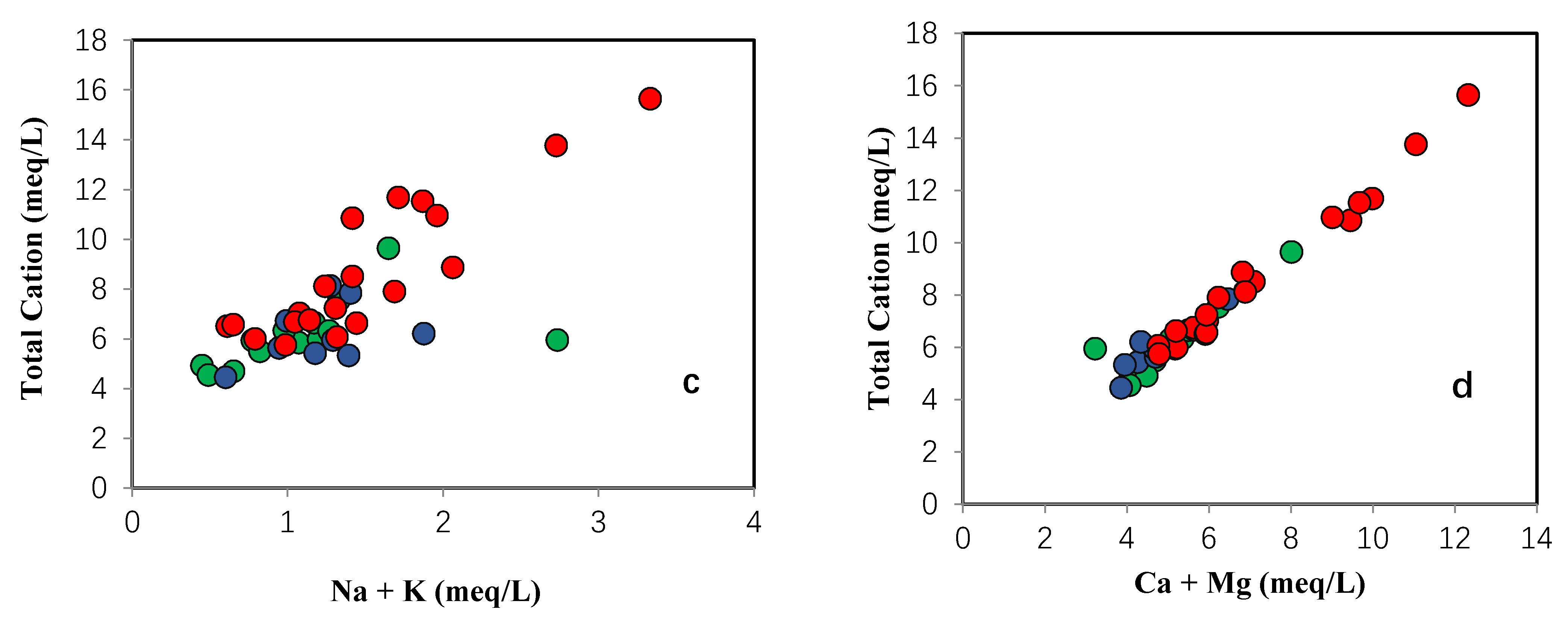
4.1. Hydrochemistry and Groundwater Quality Assessment for Drinking Water
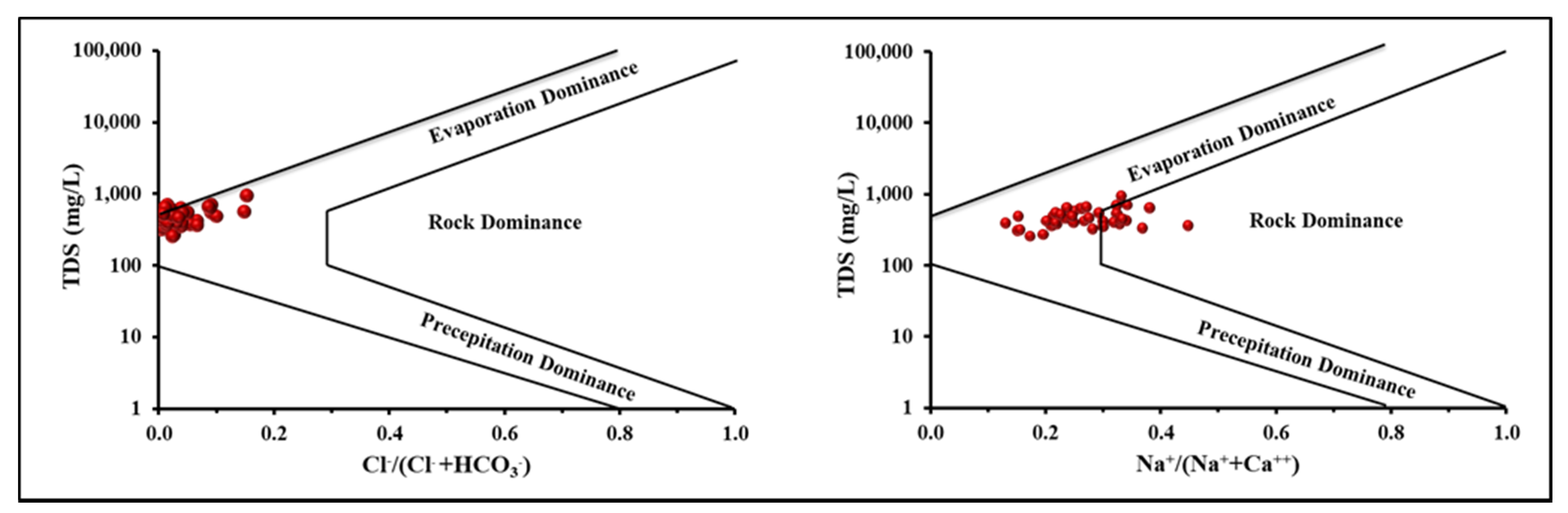
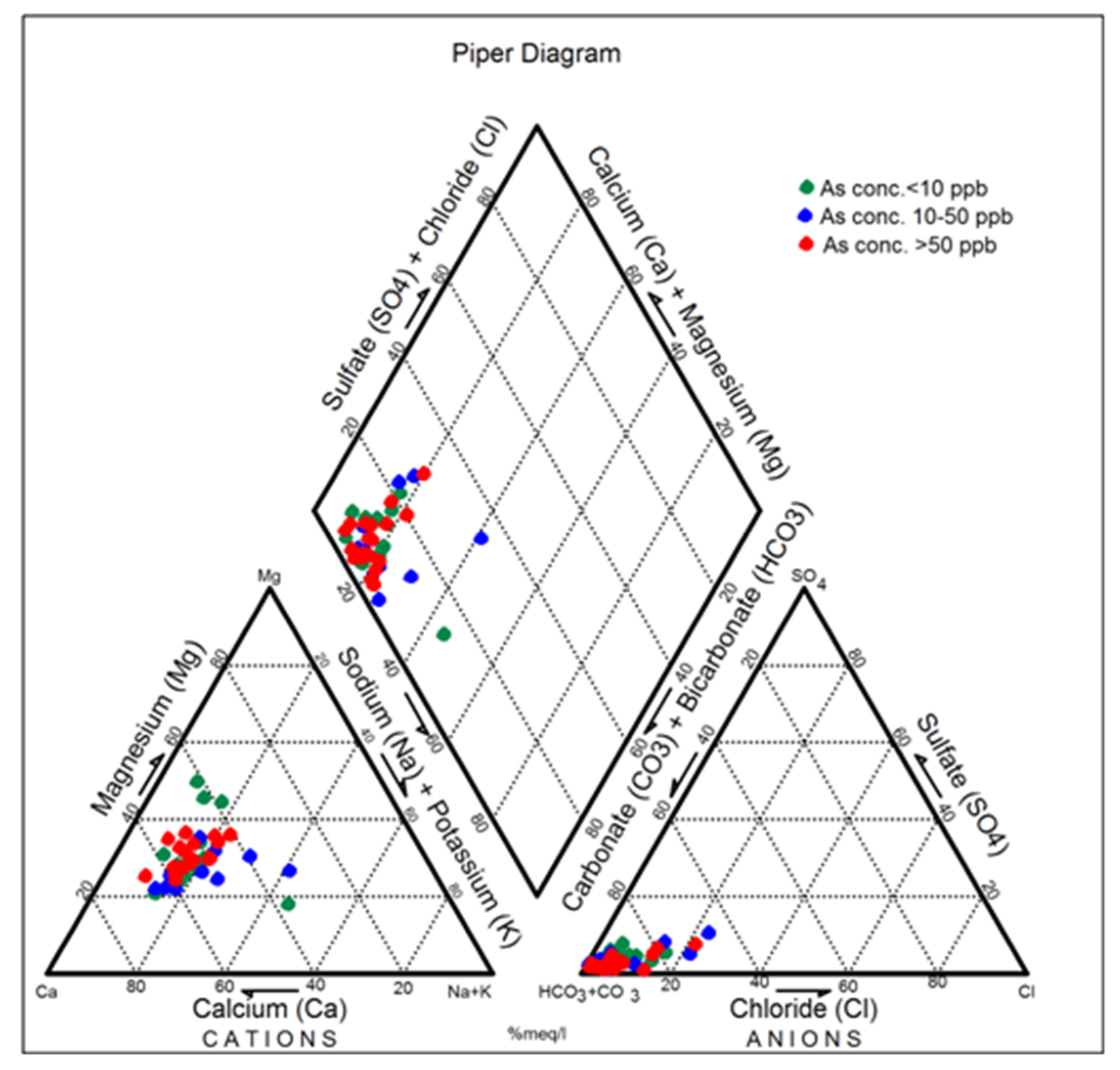
4.2. Groundwater Facies Classification
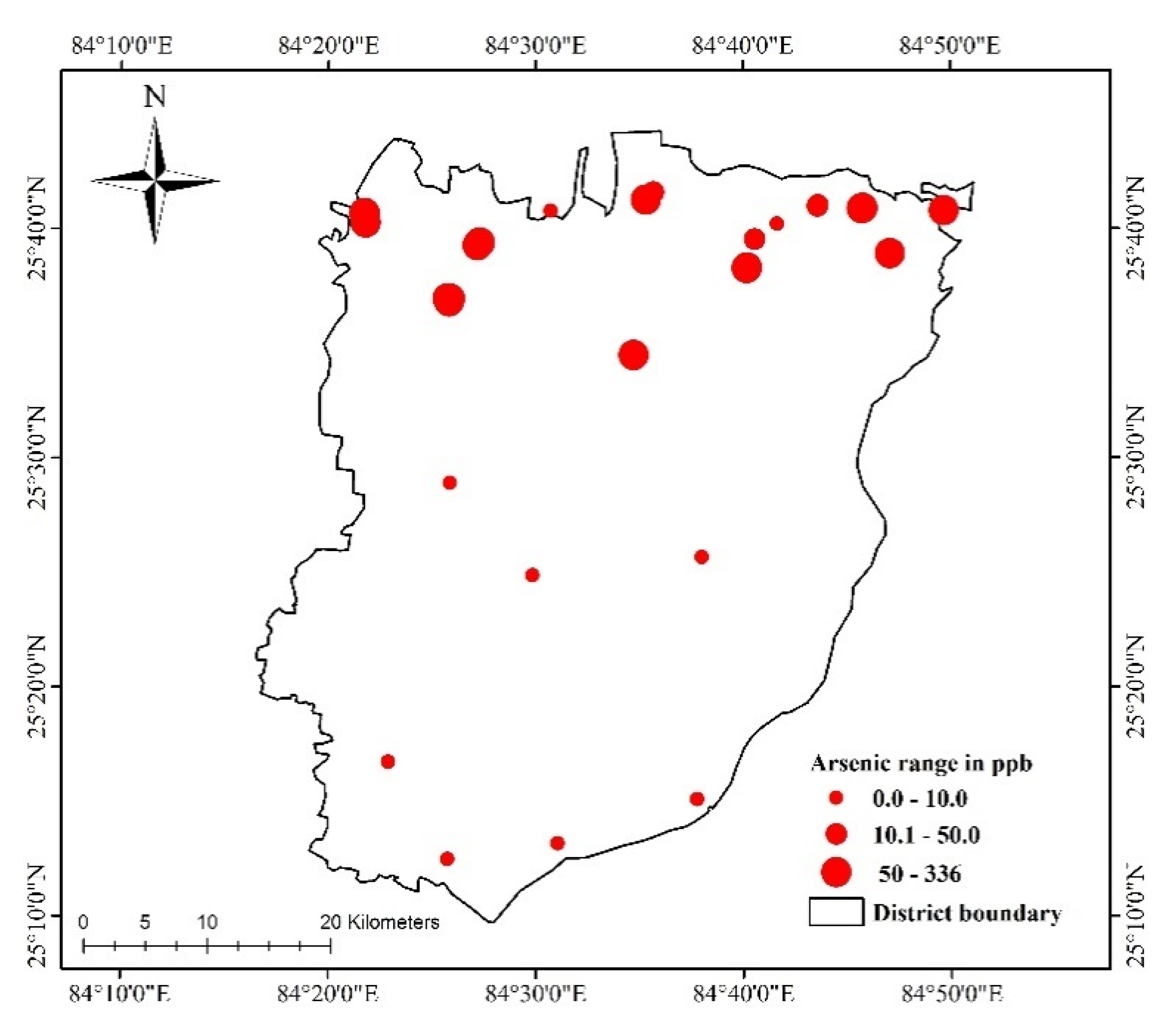
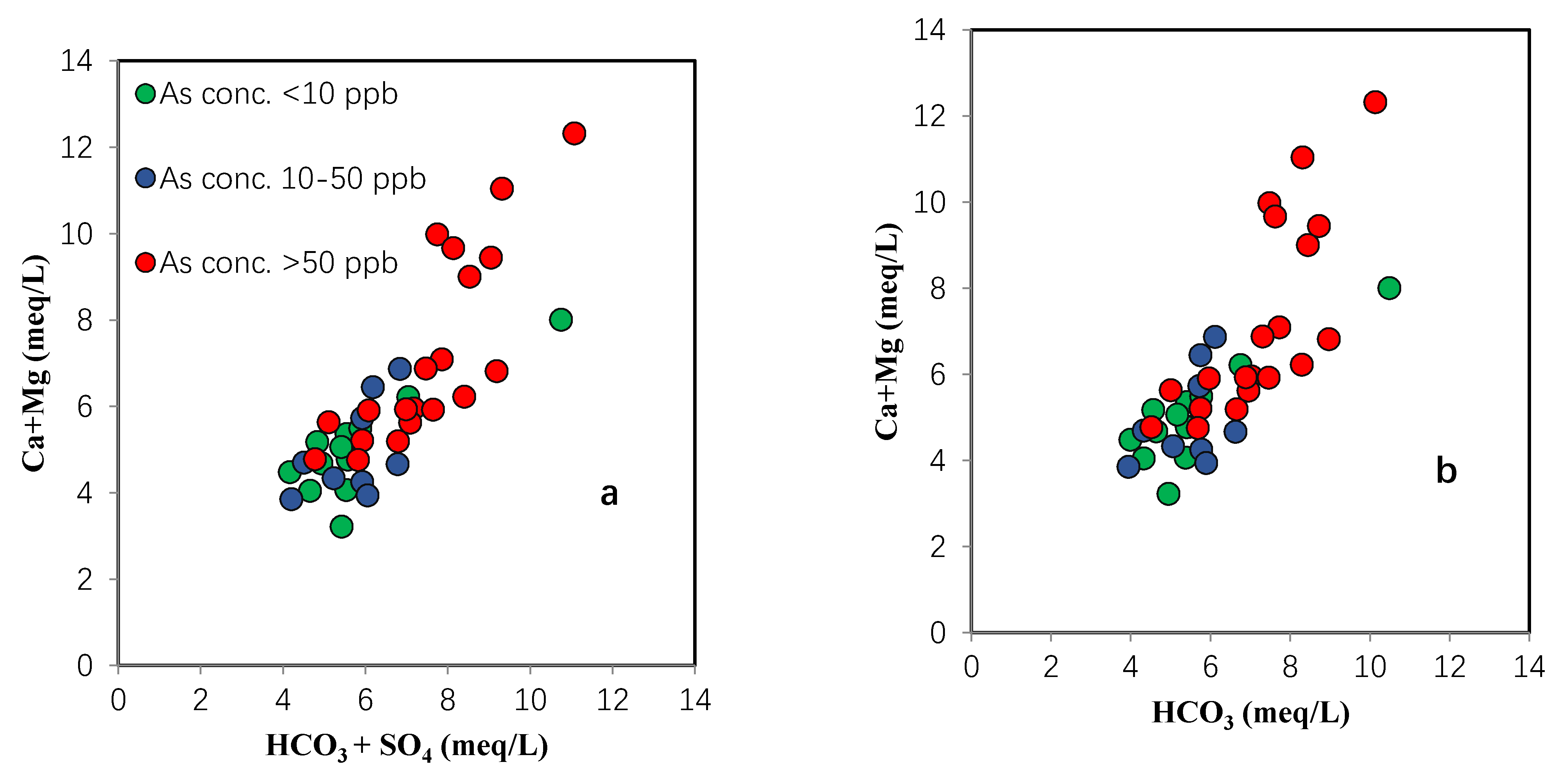
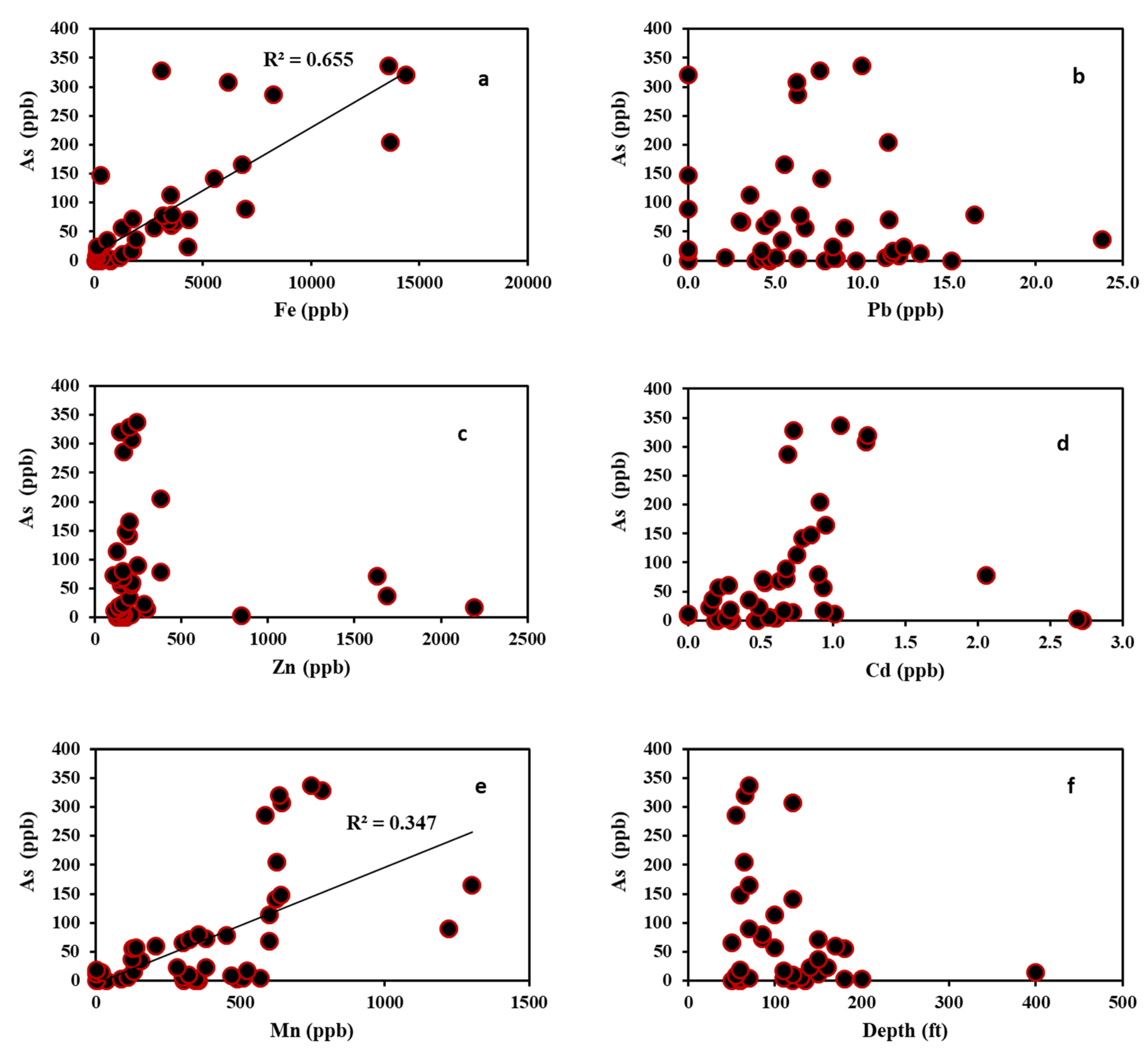
4.3. Trace Metals
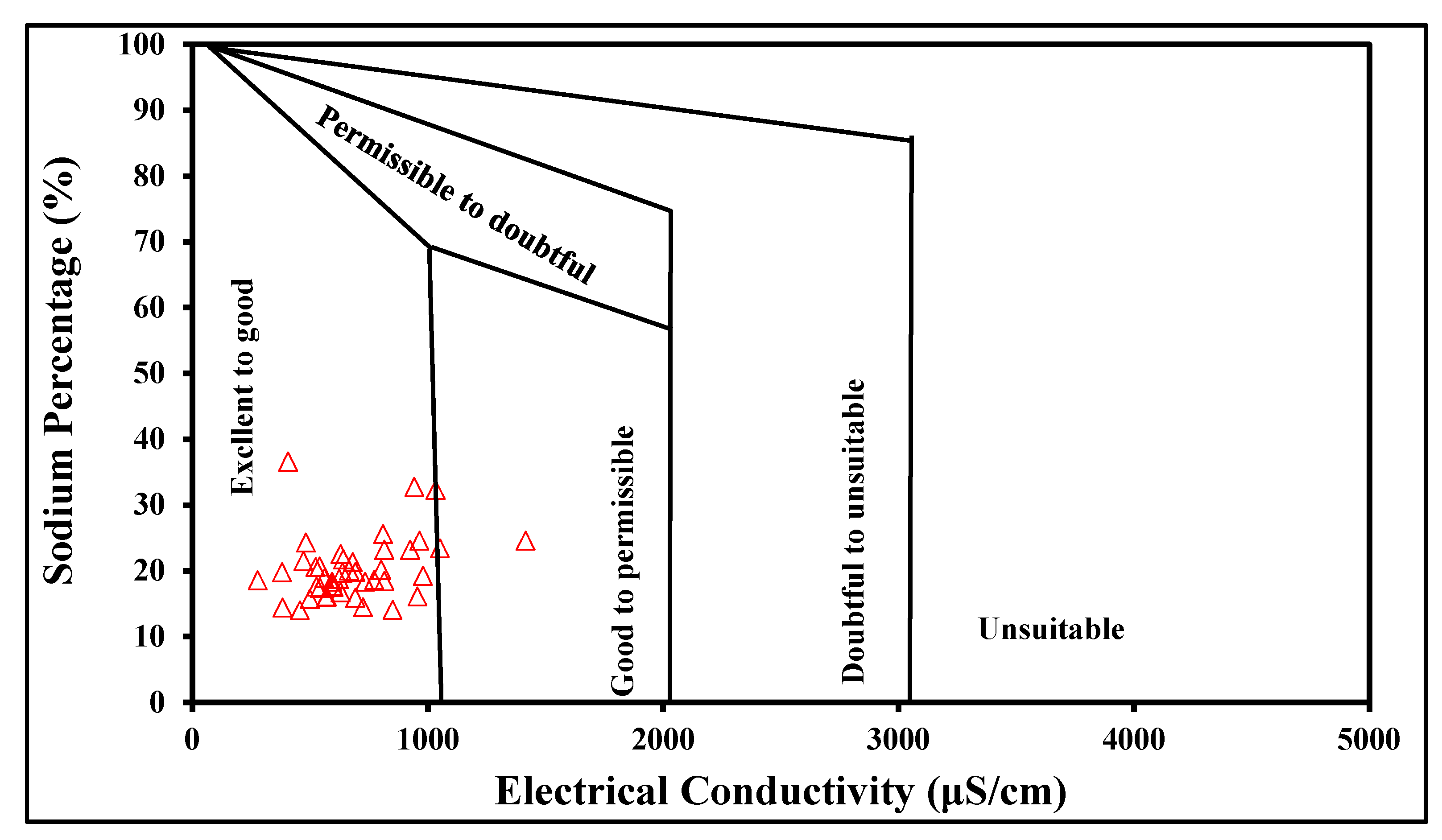
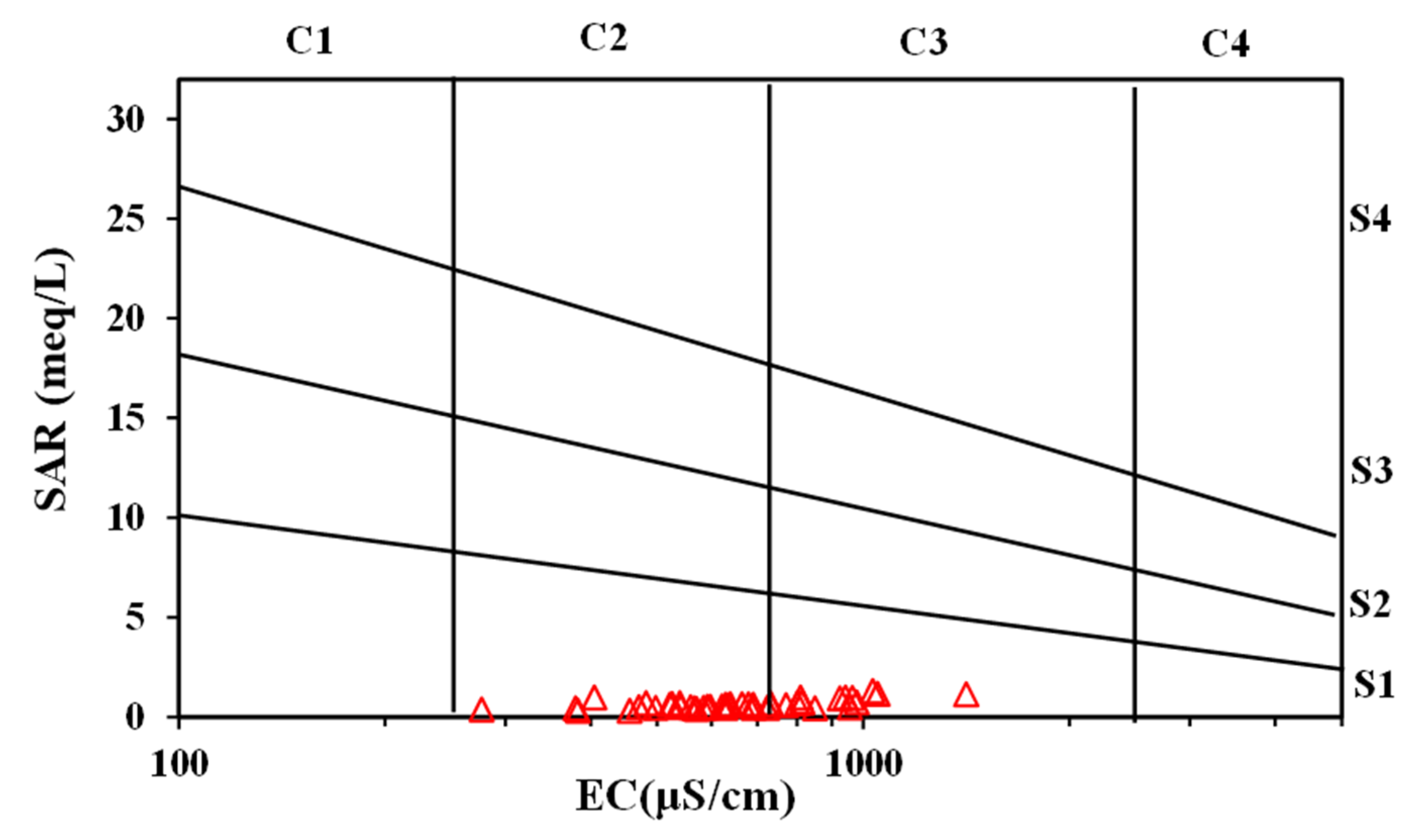
4.4. Groundwater Quality Assessment for Irrigation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| S.No. | Sample Code | Location | Latitude | Longitude | Aquifer Type | Screening Depth of Hand Pumps (Meter) | Arsenic (ppb) |
|---|---|---|---|---|---|---|---|
| 1 | S-1 | Paiga | 25.6387 | 84.6699 | Unconfined | 24.4 | 68 |
| 2 | S-2 | Paiga | 25.6384 | 84.6696 | ” | 21.3 | 78 |
| 3 | S-3 | Semaria ojhapati | 25.6158 | 84.4311 | ” | 36.6 | 11 |
| 4 | S-4 | Semaria ojhapati | 25.6158 | 84.4320 | ” | 61.0 | 4 |
| 5 | S-5 | Semaria ojhapati | 25.6162 | 84.4310 | ” | 15.2 | 65 |
| 6 | S-6 | Semaria ojhapati | 25.6160 | 84.4300 | ” | 36.6 | 308 |
| 7 | S-7 | Semaria ojhapati | 25.6160 | 84.4300 | ” | 48.8 | 23 |
| 8 | S-8 | Semaria ojhapati | 25.6162 | 84.4291 | ” | 20.1 | 320 |
| 9 | S-9 | Semaria ojhapati | 25.6180 | 84.4296 | ” | 121.9 | 14 |
| 10 | S-10 | Semaria ojhapati | 25.6152 | 84.4303 | ” | 54.9 | 3 |
| 11 | S-11 | Sahana/mangla | 25.6142 | 84.4303 | ” | 25.9 | 80 |
| 12 | S-12 | Sudarpur barja | 25.6801 | 84.5121 | ” | 33.5 | 4 |
| 13 | S-13 | Balaharpur | 25.6707 | 84.6940 | ” | 36.6 | BDL |
| 14 | S-14 | Balaharpur | 25.6707 | 84.6940 | ” | 41.1 | BDL |
| 15 | S-15 | Barahara | 25.6842 | 84.7269 | ” | 30.5 | 35 |
| 16 | S-16 | Barahara | 25.6834 | 84.7265 | ” | 45.7 | 37 |
| 17 | S-17 | Sirsiyan | 25.6820 | 84.7628 | ” | 21.3 | 337 |
| 18 | S-18 | Sirsiyan | 25.6819 | 84.7623 | ” | 51.8 | 60 |
| 19 | S-19 | Hazipur | 25.6592 | 84.6761 | ” | 42.7 | 23 |
| 20 | S-20 | Chamarpur | 25.6550 | 84.4532 | ” | 36.6 | 141 |
| 21 | S-21 | chamarpur | 25.6569 | 84.4550 | ” | 30.5 | 56 |
| 22 | S-22 | Sirhiya | 25.6821 | 84.7627 | ” | 21.3 | 4 |
| 23 | S-23 | Sirhiya | 25.6819 | 84.7622 | ” | 33.5 | 17 |
| 24 | S-24 | Sirsiya | 25.6819 | 84.7622 | ” | 18.3 | 3 |
| 25 | S-25 | Ishwarpura | 25.6784 | 84.3622 | ” | 18.3 | 148 |
| 26 | S-26 | Ishwarpura | 25.6784 | 84.3622 | ” | 54.9 | 56 |
| 27 | S-27 | Ishwarpura | 25.6784 | 84.3622 | ” | 21.3 | 165 |
| 28 | S-28 | Ishwarpura | 25.6781 | 84.3626 | ” | 16.8 | 286 |
| 29 | S-29 | Ishwarpura | 25.6779 | 84.3624 | ” | 19.8 | 205 |
| 30 | S-30 | Ishwarpura | 25.6715 | 84.3632 | ” | 27.4 | 328 |
| 31 | S-31 | Baligaon | 25.4149 | 84.4975 | ” | 16.8 | 8 |
| 32 | S-32 | Naika tola | 25.4821 | 84.4311 | ” | 39.6 | 3 |
| 33 | S-33 | Jaithwar | 25.2791 | 84.3818 | ” | 18.3 | 19 |
| 34 | S-34 | Bahnuwa | 25.2083 | 84.4293 | ” | 15.2 | BDL |
| 35 | S-35 | Anhari | 25.2198 | 84.5176 | ” | 42.7 | BDL |
| 36 | S-36 | Anhari Surya temple | 25.2198 | 84.5176 | ” | 45.7 | 11 |
| 37 | S-37 | Sahar | 25.2520 | 84.6297 | ” | 18.3 | BDL |
| 38 | S-38 | Bhagawanpur | 25.4281 | 84.6334 | ” | 18.3 | BDL |
| 39 | S-39 | Bibiganj | 25.5752 | 84.5786 | ” | 25.9 | 72 |
| 40 | S-40 | maulighat | 25.6937 | 84.5948 | ” | 38.1 | 16 |
| 41 | S-41 | maulighat | 25.6925 | 84.5951 | ” | 39.6 | 5 |
| 42 | S-42 | mauzampur | 25.6882 | 84.5886 | ” | 45.7 | 71 |
| 43 | S-43 | Bindgaon | 25.6809 | 84.8289 | ” | 27.4 | 6 |
| 44 | S-44 | Bindgaon | 25.6803 | 84.8281 | ” | 30.5 | 113 |
| 45 | S-45 | Manikpur | 25.6490 | 84.7849 | ” | 21.3 | 89 |
References
- Asadi, E.; Isazadeh, M.; Samadianfard, S.; Ramli, M.F.; Mosavi, A.; Nabipour, N.; Chau, K.W. Groundwater quality assessment for sustainable drinking and irrigation. Sustainability 2020, 12, 177. [Google Scholar] [CrossRef] [Green Version]
- MacDonald, A.; Bonsor, H.C.; Ahmed, K.M.; Burgess, W.G.; Basharat, M.; Calow, R.C.; Dixit, A.; Foster, S.S.D.; Gopal, K.; Lapworth, D.J.; et al. Groundwater quality and depletion in the Indo-Gangetic Basin mapped from in situ observations. Nat. Geosci. 2016, 9, 762–766. [Google Scholar] [CrossRef]
- Kumari, N.; Srivastava, A.; Sahoo, B.; Raghuwanshi, N.S.; Bretreger, D. Identification of Suitable Hydrological Models for Streamflow Assessment in the Kangsabati River Basin, India, by Using Different Model Selection Scores. Nat. Resour. Res. 2021, 1–19. [Google Scholar] [CrossRef]
- Srivastava, A.; Sahoo, B.; Raghuwanshi, N.S.; Singh, R. Evaluation of variable-infiltration capacity model and MODIS-terra satellite-derived grid-scale evapotranspiration estimates in a River Basin with Tropical Monsoon-Type climatology. J. Irrig. Drain. Eng. 2017, 143, 04017028. [Google Scholar] [CrossRef] [Green Version]
- Chaudhary, V.; Satheeshkumar, S. Assessment of groundwater quality for drinking and irrigation purposes in arid areas of Rajasthan, India. Appl. Water Sci. 2018, 8, 218. [Google Scholar] [CrossRef] [Green Version]
- Gautam, S.K.; Maharana, C.; Sharma, D.; Singh, A.K.; Tripathi, J.K.; Singh, S.K. Evaluation of groundwater quality in the Chotanagpur plateau region of the Subarnarekha river basin, Jharkhand State, India. Sustain. Water Qual. Ecol. 2015, 6, 57–74. [Google Scholar] [CrossRef]
- Rawat, K.S.; Singh, S.K. Water Quality Indices and GIS-based evaluation of a decadal groundwater quality. Geol. Ecol. Landsc. 2018, 2, 240–255. [Google Scholar] [CrossRef] [Green Version]
- Breida, M.; Younssi, S.A.; Ouammou, M.; Bouhria, M.; Hafsi, M. Pollution of Water Sources from Agricultural and Industrial Effluents: Special Attention to NO3−, Cr (VI), and Cu (II). In Water Chemistry; IntechOpen: London, UK, 2019. [Google Scholar]
- Lwimbo, Z.D.; Komakech, H.C.; Muzuka, A.N. Impacts of Emerging Agricultural Practices on Groundwater Quality in Kahe Catchment, Tanzania. Water 2019, 11, 2263. [Google Scholar] [CrossRef] [Green Version]
- Fianko, J.R.; Nartey, V.K.; Donkor, A. The hydrochemistry of groundwater in rural communities within the Tema District, Ghana. Environ. Monit. Assess. 2009, 168, 441–449. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, V.; Saini, R.K.; Pant, N.; Singh, R.; Singh, A.; Kumar, S.; Singh, S.; Yadav, B.K.; Krishan, G.; et al. Floodplains landforms, clay deposition and irrigation return flow govern arsenic occurrence, prevalence and mobilization: A geochemical and isotopic study of the mid-Gangetic floodplains. Environ. Res. 2021, 21, 111516. [Google Scholar] [CrossRef]
- Biswas, A.; Bhattacharya, P.; Mukherjee, A.; Nath, B.; Alexanderson, H.; Kundu, A.K.; Chatterjee, D.; Jacks, G. Shallow hydrostratigraphy in an arsenic affected region of Bengal Basin: Implication for targeting safe aquifers for drinking water supply. Sci. Total Environ. 2014, 485, 12–22. [Google Scholar] [CrossRef]
- Kumar, S.; Ghosh, N.C.; Singh, R.P.; Singh, R.; Singh, S. Impact of canal recharge on groundwater quality of Kolayat area, district Bikaner, India. In Geostatistical and Geospatial Approaches for the Characterization of Natural Resources in the Environment; Springer: Cham, Switzerland, 2016; pp. 341–347. [Google Scholar]
- Saha, D.; Sahu, S. A decade of investigations on groundwater arsenic contamination in Middle Ganga Plain, India. Environ. Geochem. Health 2015, 38, 315–337. [Google Scholar] [CrossRef]
- Dillon, P.; Fernandez, E.E.; Tuinhof, A. Management of aquifer recharge and discharge processes and aquifer storage equilibrium. In IAH Contribution to GEF-FAO Groundwater Governance Thematic Paper 4; GEF-FAO: Rome, Italy, 2012; Available online: https://recharge.iah.org/files/2016/11/Management-of-aquifer-recharge-and-discharge-processes-and-aquifer-storage-equilibrium.pdf (accessed on 1 May 2021).
- Singh, S.; Ghosh, N.C.; Krishan, G.; Kumar, S.; Gurjar, S.; Sharma, M.K. Development of indices for surface and ground water quality assessment and characterization for Indian conditions. Environ. Monit. Assess. 2019, 191, 182. [Google Scholar] [CrossRef]
- Gupta, P.; Vishwakarma, M.; Rawtani, P.M. Assessment of water quality parameters of Kerwa Dam for drinking suitability. Int. J. Theor. Appl. Sci. 2009, 1, 53–55. [Google Scholar]
- Ramesh, K.; Elango, L. Groundwater quality and its suitability for domestic and agricultural use in Tondiar river basin, Tamil Nadu, India. Environ. Monit. Assess. 2011, 184, 3887–3899. [Google Scholar] [CrossRef]
- Jacintha, T.G.A.; Rawat, K.S.; Mishra, A.; Singh, S.K. Hydrogeochemical characterization of groundwater of peninsular Indian region using multivariate statistical techniques. Appl. Water Sci. 2017, 7, 3001–3013. [Google Scholar] [CrossRef] [Green Version]
- CGWB Groundwater Information Booklet: Bhojpur District, Bihar; Central Ground Water Board, Ministry of Water Resources, RD & GR. Government of India: New Delhi, India, 2013. Available online: http://cgwb.gov.in/District_Profile/Bihar/Bhojpur.pdf (accessed on 10 April 2021).
- APHA American Public Health Association Standard Methods for the Examination of Water & Wastewaters, 22nd ed.; APHA: Washington, DC, USA, 2012; Available online: http://srjcstaff.santarosa.edu/~oraola/Assets/APHA_SM_20.pdf (accessed on 13 April 2021).
- Jain, C.K.; Bhatia, K.K.S. Physico-Chemical Analysis of Water and Wastewater, User’s Manual; National Institute of Hydrology: Roorkee, India, 1998; Available online: http://117.252.14.250:8080/xmlui/handle/123456789/490 (accessed on 2 April 2021).
- Kumar, S.; Ghosh, N.C.; Singh, R.P.; Sonkusare, M.M.; Singh, S.; Mittal, S. Assessment of water quality of lakes for drinking and irrigation purposes in Raipur City, Chhattisgarh, India. Int. J. Eng. Res. Appl. 2015, 5, 42–49. [Google Scholar]
- Raghunath, H.M. Ground Water, 2nd ed.; Wiley: New Delhi, India, 1987; p. 353. [Google Scholar]
- Schoeller, H. Geochemistry of groundwater. In Groundwater Studies—An International Guide for Research and Practice; UNESCO: Paris, France, 1977; Volume 15, pp. 1–18. [Google Scholar]
- Doneen, L.D. Notes on Water Quality in Agriculture; Published as a water science and engineering paper 4001; Department of Water Science and Engineering, University of California: Davis, CA, USA, 1964. [Google Scholar]
- BIS. Indian Standard—Drinking Water Specification (2nd Revision); IS 10500; BIS: New Delhi, India, 2012. Available online: http://cgwb.gov.in/Documents/WQ-standards.pdf (accessed on 13 May 2021).
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water; Department of the Interior, US Geological Survey: Reston, Virginia, 1985. [Google Scholar]
- Chakrapani, G.J.; Saini, R.K.; Yadav, S.K. Chemical weathering rates in the Alaknanda–Bhagirathi river basins in Himalayas, India. J. Asian Earth Sci. 2009, 34, 347–362. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, W.; Létolle, R.; Jusserand, C. Major element chemistry of the Huanghe (Yellow River), China—Weathering processes and chemical fluxes. J. Hydrol. 1995, 168, 173–203. [Google Scholar] [CrossRef]
- Pant, N.; Rai, S.P.; Singh, R.; Kumar, S.; Saini, R.K.; Purushothaman, P.; Nijesh, P.; Rawat, Y.S.; Sharma, M.; Pratap, K. Impact of geology and anthropogenic activities over the water quality with emphasis on fluoride in water scarce Lalitpur district of Bundelkhand region, India. Chemosphere 2021, 279, 130496. [Google Scholar] [CrossRef] [PubMed]
- Piper, A.M. A graphic procedure in the chemical interpretation of water analysis. Am. Geophys. Union Transcr. 1944, 25, 914–923. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, V.; Saini, R.K.; Kumar, C.P.; Raju, M.; Singh, S.; Singh, O.; Chakravorty, B. Detection of arsenic in groundwater of Laksar area, Haridwar District, Uttarakhand. J. Indian Water Res. Soc. 2019, 39, 42–48. [Google Scholar]
- Saha, D. Arsenic groundwater contamination in parts of middle Ganga plain, Bihar. Curr. Sci. 2009, 97, 753–755. [Google Scholar]
- Kunar, S.; Jain, S.K.; Shekhar, S.; Sharma, V. Arsenic in groundwater in India: An overview. Bhujal News 2009, 24, 6–14. [Google Scholar]
- Mukherjee, A.; Scalon, B.R.; Chaudhary, S. Regional hydrogeochemical study of groundwater arsenic contamination along transects from the Himalayan alluvial deposits to the India Shield, Central Gangetic Basin, India. Geol. Soc. Am. 2007, 39, 519. [Google Scholar]
- Singh, S.K.; Ghosh, A. Entry of arsenic into food material—A case study. World Appl. Sci. J. 2011, 13, 385–390. [Google Scholar]
- Kumar, S.; Joshi, S.K.; Pant, N.; Singh, S.; Chakraborty, B.; Saini, R.K.; Kumar, V.; Singh, A.; Ghosh, N.C.; Mukherjee, A.; et al. Hydrogeochemical evolution and groundwater recharge processes in arsenic enriched area in central Gangetic plain, India. Appl. Geochem. 2021, 131, 105044. [Google Scholar] [CrossRef]
- Saha, D.; Sinha, U.; Dwivedi, S. Characterization of recharge processes in shallow and deeper aquifers using isotopic signatures and geochemical behavior of groundwater in an arsenic-enriched part of the Ganga Plain. Appl. Geochem. 2011, 26, 432–443. [Google Scholar] [CrossRef]
- Saha, D.; Sarangam, S.S.; Dwivedi, S.N.; Bhartariya, K.G. Evaluation of hydrogeochemical processes in arsenic-contaminated alluvial aquifers in parts of Mid-Ganga Basin, Bihar, Eastern India. Environ. Earth Sci. 2009, 61, 799–811. [Google Scholar] [CrossRef]
- Wilcox, L.V. The Quality of Water for Irrigation Use; US Department of Agricultural Technical Bulletin: Washington, WA, USA, 1948. [Google Scholar]
- IS-11624: Indian Standard—Guidelines for the Quality of Irrigation Water; Bureau of Indian Standards: New Delhi, India, 1986; Available online: https://law.resource.org/pub/in/bis/S06/is.11624.1986.pdf (accessed on 12 April 2021).
| Parameters | Minimum | Maximum | Average | Std. Dev. | BIS (2012) Acceptable Limit | BIS (2012) Permissible Limit |
|---|---|---|---|---|---|---|
| pH | 6.9 | 8.5 | 7.6 | 0.4 | 6.5–8.5 | - |
| EC | 277 | 1415 | 683 | 213 | - | - |
| TDS (mg/L) | 237 | 1034 | 538 | 161 | 500 | 2000 |
| TH (mg/L) | 122 | 616 | 292 | 105 | 200 | 600 |
| Ca (mg/L) | 32 | 140 | 74 | 25.1 | 75 | 200 |
| Mg (mg/L) | 10 | 65 | 26 | 12.2 | 30 | 100 |
| Na (mg/L) | 10 | 73 | 28 | 13.0 | - | - |
| K (mg/L) | 0.42 | 70.37 | 3.93 | 10.1 | - | - |
| F (mg/L) | BDL | 0.58 | 0.22 | 0.13 | 1 | 1.5 |
| Cl (mg/L) | 1 | 91 | 18 | 19.0 | 250 | 1000 |
| HCO3 (mg/L) | 166 | 639 | 373 | 373 | - | - |
| SO4 (mg/L) | 4 | 43 | 11 | 11 | 200 | 400 |
| NO3 (mg/L) | BDL | 29.4 | 2.3 | 6.7 | 45 | NR |
| As (ppb) | BDL | 336 | 73 | 99.7 | 10 | 50 |
| Fe (ppm) | 0.06 | 14.39 | 2.8 | 3.69 | 0.3 | NR |
| Mn (ppb) | 2.1 | 1303 | 390 | 291 | 100 | 300 |
| Pb (ppb) | BDL | 24 | 7.1 | 4.97 | 10 | NR |
| Zn (ppb) | 110 | 2190 | 310 | 432 | 5000 | 15,000 |
| Cd (ppb) | BDL | 2.7 | 0.7 | 0.58 | 3 | NR |
| Cu (ppb) | BDL | 10 | 4 | 1.86 | 50 | 1500 |
| pH | EC | TDS | F | Cl | HCO3 | SO4 | NO3 | Ca | Mg | Na | K | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | 1.0 | |||||||||||
| EC | −0.3 | 1.0 | ||||||||||
| TDS | −0.6 | 0.5 | 1.0 | |||||||||
| F | 0.2 | 0.1 | −0.3 | 1.0 | ||||||||
| Cl | 0.0 | 0.6 | 0.3 | 0.0 | 1.0 | |||||||
| HCO3 | −0.3 | 0.8 | 0.3 | 0.1 | 0.3 | 1.0 | ||||||
| SO4 | 0.1 | 0.5 | 0.2 | 0.1 | 0.8 | 0.2 | 1.0 | |||||
| NO3 | 0.1 | 0.3 | 0.2 | 0.1 | 0.5 | 0.2 | 0.5 | 1.0 | ||||
| Ca | −0.4 | 0.7 | 0.4 | 0.0 | 0.6 | 0.7 | 0.4 | 0.3 | 1.0 | |||
| Mg | −0.1 | 0.8 | 0.3 | 0.1 | 0.6 | 0.8 | 0.5 | 0.4 | 0.7 | 1.0 | ||
| Na | −0.1 | 0.7 | 0.4 | 0.1 | 0.7 | 0.6 | 0.6 | 0.4 | 0.6 | 0.7 | 1.0 | |
| K | 0.1 | −0.1 | 0.0 | 0.0 | 0.0 | −0.1 | 0.2 | 0.0 | −0.1 | −0.1 | 0.0 | 1.0 |
| Parameters | Minimum | Maximum | Average | IS 11,624 (1986): Classification of Irrigation Water |
|---|---|---|---|---|
| EC (µS/cm) | 277 | 1415 | 683 | Class: Low (Below 1500), Medium (1500–3000), High (3000–6000) Very high (Above 6000) |
| Na (%) | 12 | 62 | 24 | - |
| SAR | 0.3 | 1.7 | 0.7 | Class: Low (Below 10), Medium (10–18), High (18–26), Very high (Above 26) |
| RSC | −2.7 | 2.5 | 0.3 | Class-I RSC < 1.25 (low-Safe), Class-II RSC 1.25–2.5 (med. marginal) Class III > 2.5 (high-unsafe) |
| PI | 72.4 | 41.3 | 52.1 | Suitable (Class I, >75%), Good (Class II, 25–75%), Unsuitable (Class III, <25%). |
| CAI-I | −11.7 | −0.93 | −1.6 | Class I Positive (+) CAI, Class II Negative (−) |
| CAI-II | −0.14 | −0.20 | −0.13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, S.; Kumar, M.; Chandola, V.K.; Kumar, V.; Saini, R.K.; Pant, N.; Kumari, N.; Srivastava, A.; Singh, S.; Singh, R.; et al. Groundwater Quality Issues and Challenges for Drinking and Irrigation Uses in Central Ganga Basin Dominated with Rice-Wheat Cropping System. Water 2021, 13, 2344. https://doi.org/10.3390/w13172344
Kumar S, Kumar M, Chandola VK, Kumar V, Saini RK, Pant N, Kumari N, Srivastava A, Singh S, Singh R, et al. Groundwater Quality Issues and Challenges for Drinking and Irrigation Uses in Central Ganga Basin Dominated with Rice-Wheat Cropping System. Water. 2021; 13(17):2344. https://doi.org/10.3390/w13172344
Chicago/Turabian StyleKumar, Sumant, Manish Kumar, Veerendra Kumar Chandola, Vinod Kumar, Ravi K. Saini, Neeraj Pant, Nikul Kumari, Ankur Srivastava, Surjeet Singh, Rajesh Singh, and et al. 2021. "Groundwater Quality Issues and Challenges for Drinking and Irrigation Uses in Central Ganga Basin Dominated with Rice-Wheat Cropping System" Water 13, no. 17: 2344. https://doi.org/10.3390/w13172344
APA StyleKumar, S., Kumar, M., Chandola, V. K., Kumar, V., Saini, R. K., Pant, N., Kumari, N., Srivastava, A., Singh, S., Singh, R., Krishan, G., Induwar, S. P., Kumar, S., Yadav, B. K., Maurya, N. S., & Chaudhary, A. (2021). Groundwater Quality Issues and Challenges for Drinking and Irrigation Uses in Central Ganga Basin Dominated with Rice-Wheat Cropping System. Water, 13(17), 2344. https://doi.org/10.3390/w13172344










