Predicting Macroinvertebrate Responses to Water Abstraction in Alpine Streams
Abstract
:1. Introduction
2. Materials and Methods
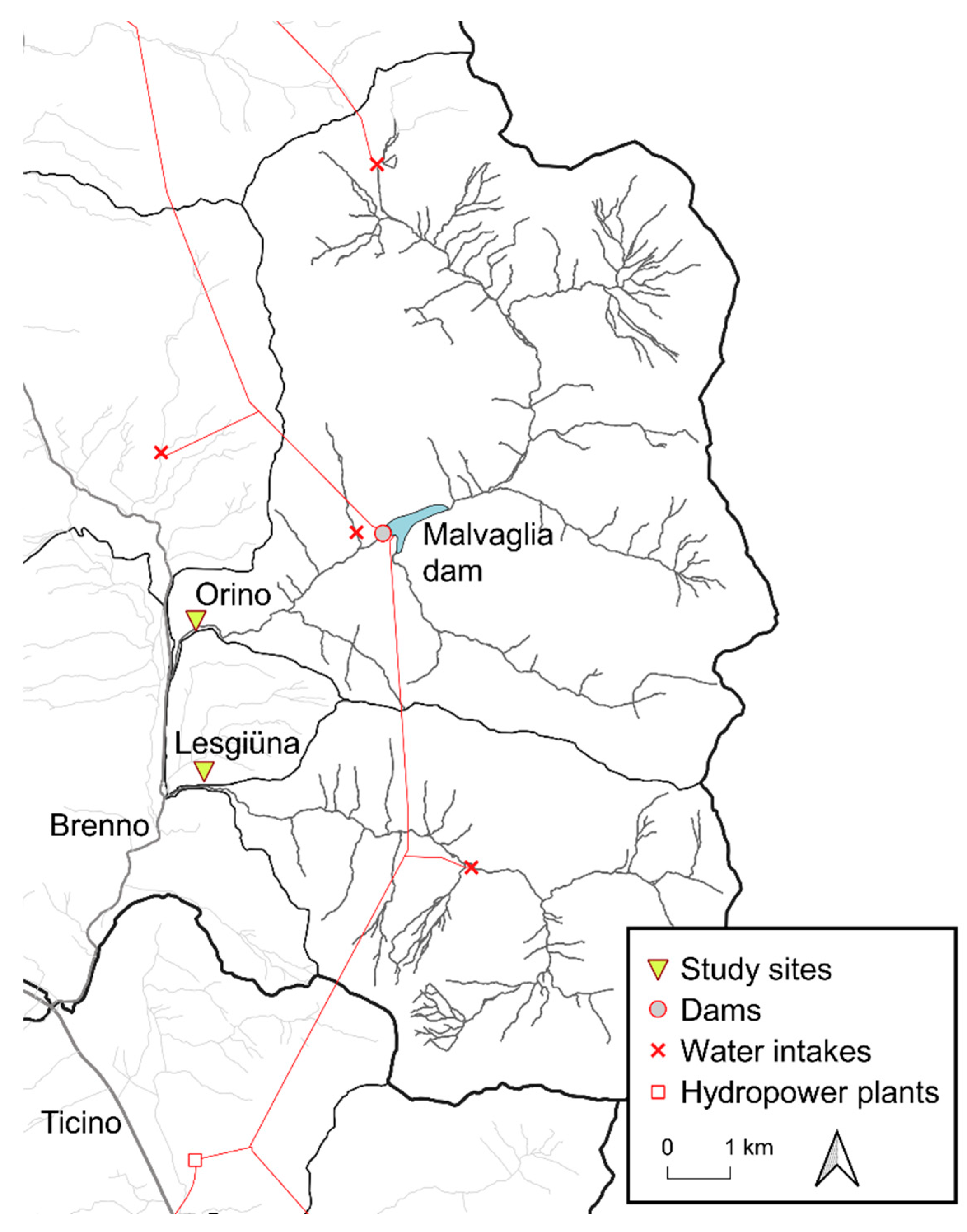
2.1. Study Sites
2.2. Data Collection and Treatment
2.3. Data Analysis
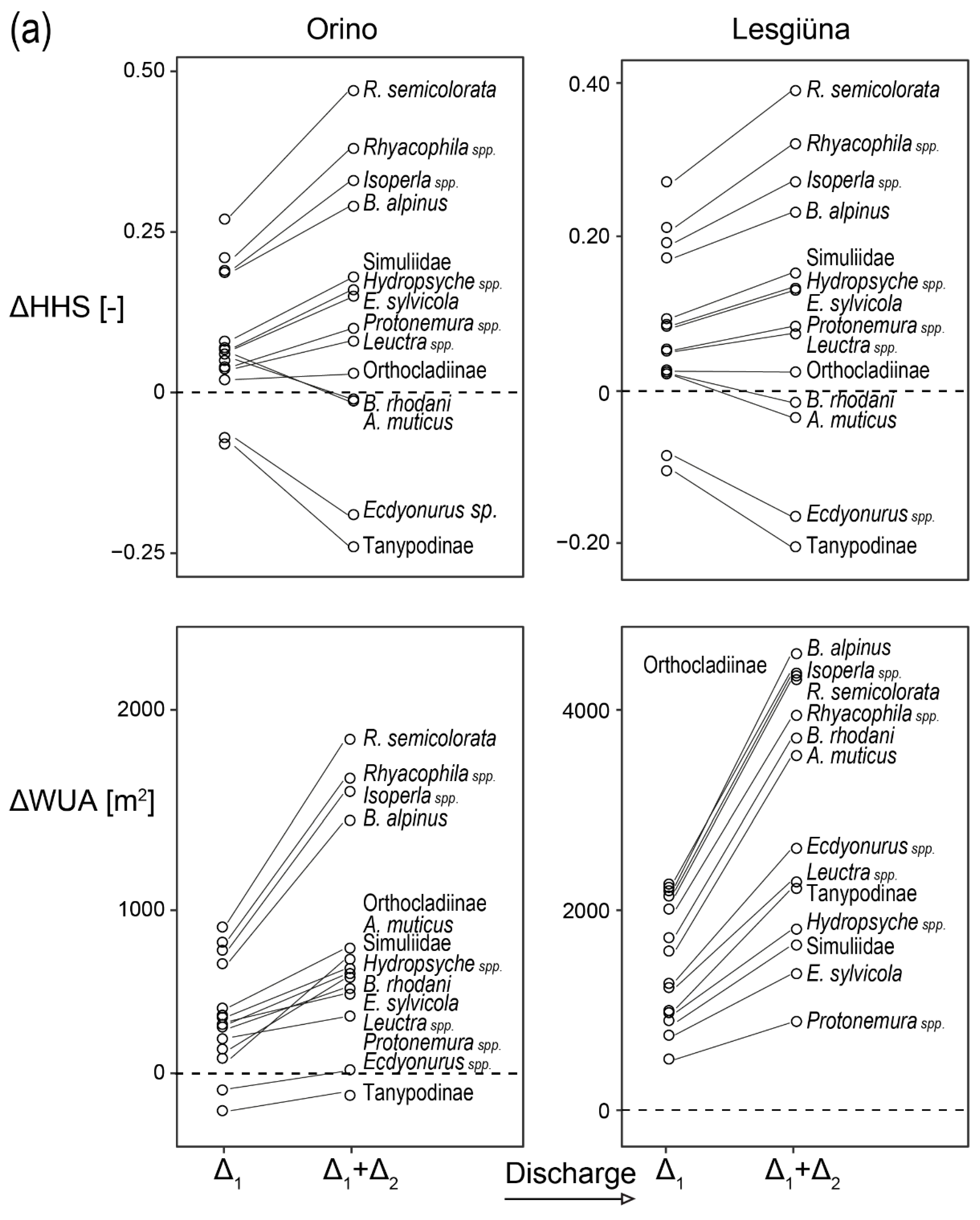
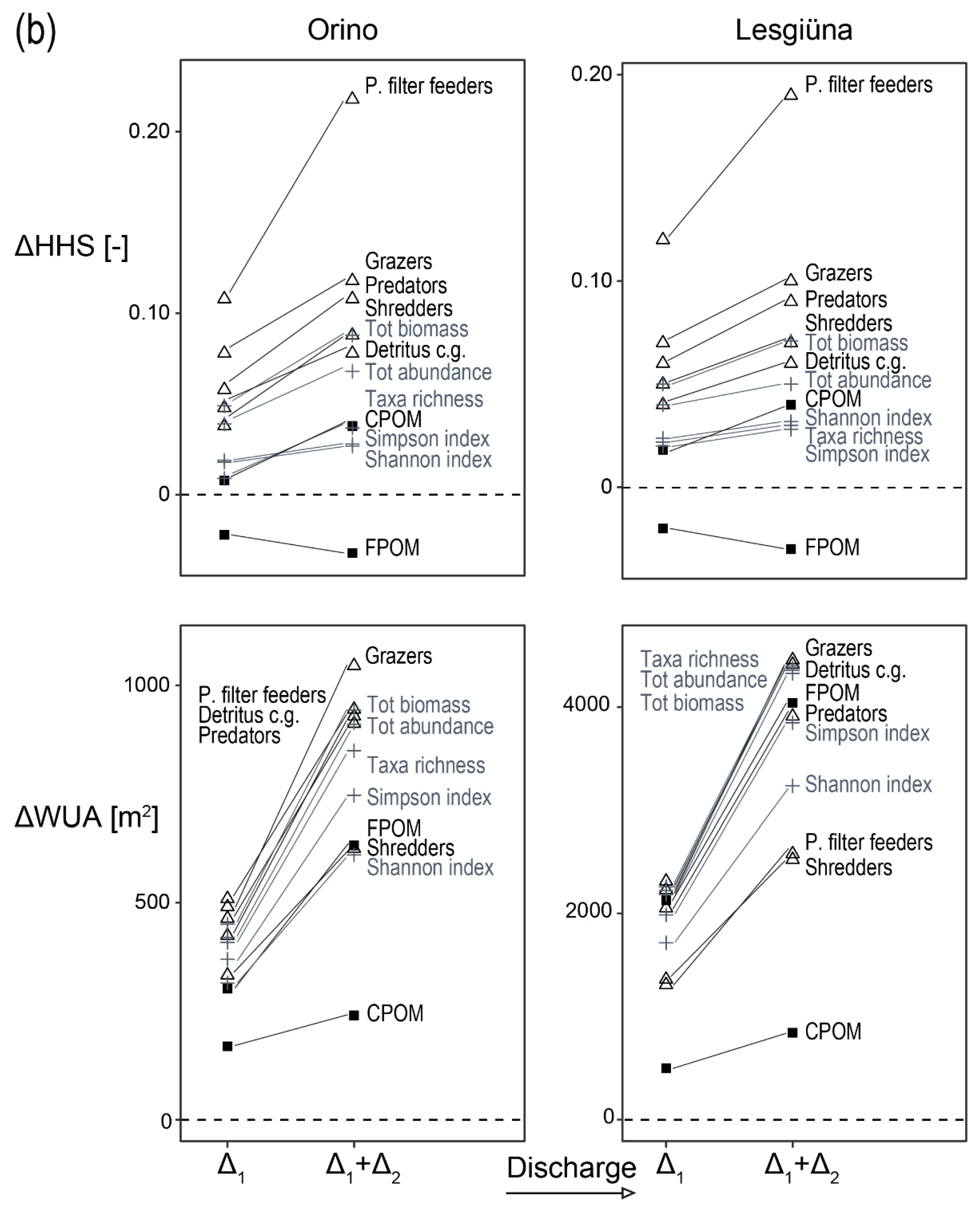
3. Results
3.1. Water Physico-Chemistry and Physical Habitat Assessment
3.2. Macroinvertebrates and Benthic Organic Matter
3.3. Hydraulic Model
3.4. Preference Model
4. Discussion
4.1. Hydraulic Models
4.2. Preference Models
4.3. Changes in Habitat Suitability in Response to Discharge
4.4. Differences between Streams
4.5. Management Implications and Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

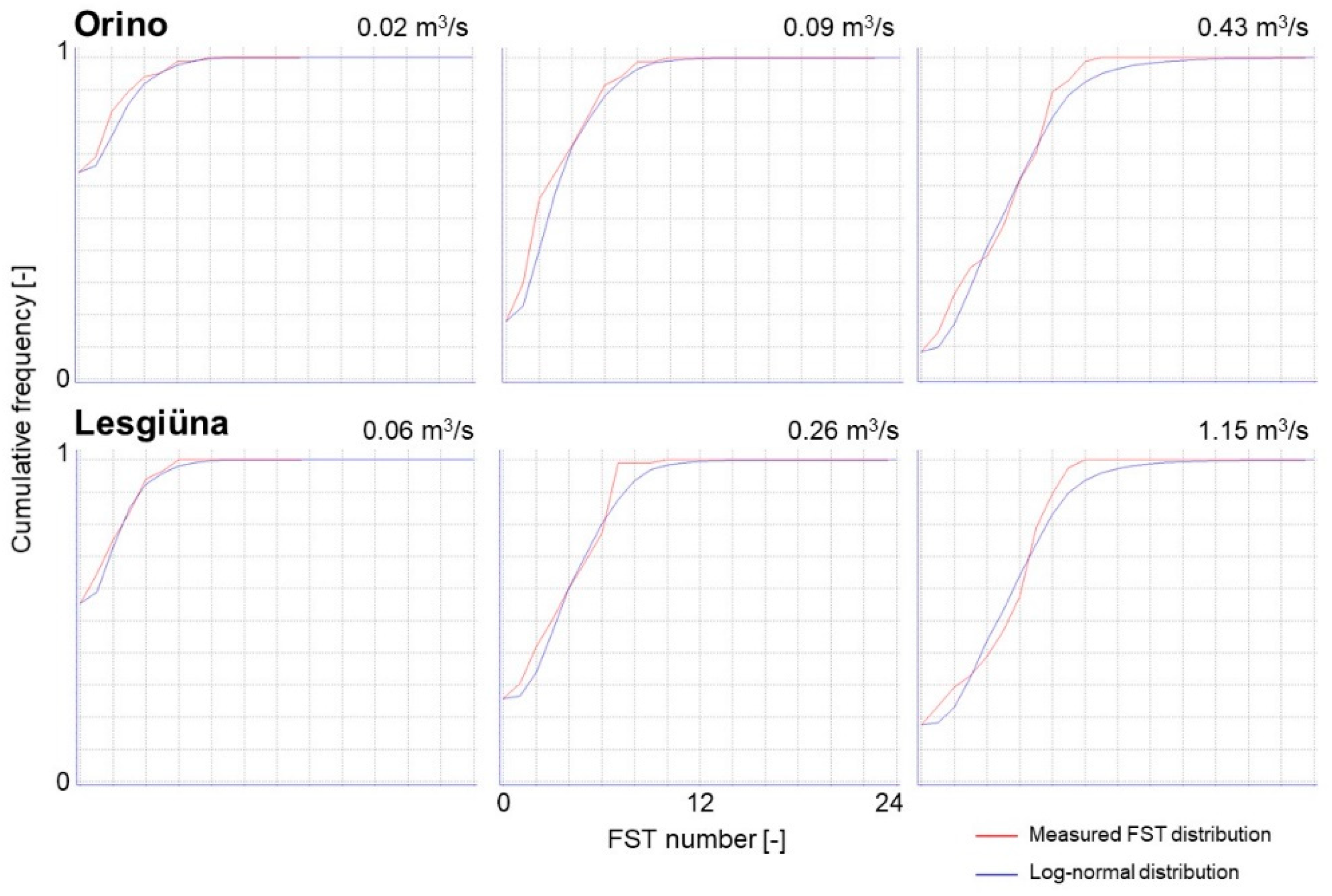
Appendix B
| WUA [m2] | HHS | |||||
|---|---|---|---|---|---|---|
| Discharge | 0.02 m3 s−1 | 0.09 m3 s−1 | 0.44 m3 s−1 | 0.02 m3 s−1 | 0.09 m3 s−1 | 0.44 m3 s−1 |
| Taxa | ||||||
| Baetis alpinus | 1559.90 | 2379.01 | 3017.02 | 0.55 | 0.74 | 0.84 |
| Baetis rhodani | 2525.79 | 3035.85 | 3176.58 | 0.89 | 0.94 | 0.88 |
| Baetis muticus | 2418.04 | 3003.01 | 3081.16 | 0.87 | 0.93 | 0.86 |
| Epeorus sylvicola | 118.77 | 361.48 | 694.93 | 0.04 | 0.11 | 0.19 |
| Rhitrogena semicolorata | 463.54 | 1386.96 | 2272.84 | 0.16 | 0.43 | 0.63 |
| Ecdyonurus spp. | 2738.47 | 2874.96 | 2765.21 | 0.96 | 0.89 | 0.77 |
| Isoperla spp. | 1202.48 | 1973.67 | 2702.45 | 0.42 | 0.61 | 0.75 |
| Leuctra spp. | 1082.58 | 1343.66 | 1659.67 | 0.38 | 0.42 | 0.46 |
| Protonemura spp. | 63.63 | 203.62 | 439.97 | 0.02 | 0.06 | 0.12 |
| Hydropsyche spp. | 358.25 | 654.61 | 1035.15 | 0.13 | 0.20 | 0.29 |
| Rhyacophila spp. | 663.79 | 1426.78 | 2210.17 | 0.23 | 0.44 | 0.61 |
| Orthocladiinae | 2737.82 | 3164.39 | 3546.89 | 0.96 | 0.98 | 0.99 |
| Tanypodinae | 2732.76 | 2836.49 | 2586.80 | 0.96 | 0.88 | 0.72 |
| Simuliidae | 171.24 | 465.64 | 856.20 | 0.06 | 0.14 | 0.24 |
| FFGs | ||||||
| Grazers | 2279.42 | 2834.50 | 3324.77 | 0.80 | 0.88 | 0.92 |
| Shredders | 1232.08 | 1523.85 | 1856.78 | 0.43 | 0.47 | 0.52 |
| P. filter feeders | 564.83 | 1000.55 | 1509.66 | 0.20 | 0.31 | 0.42 |
| Detritus C.G. | 2519.10 | 3024.19 | 3448.24 | 0.88 | 0.93 | 0.96 |
| Predators | 2033.22 | 2487.05 | 2950.82 | 0.71 | 0.77 | 0.82 |
| Comm. metrics | ||||||
| Tot. abundance | 2563.62 | 3054.06 | 3473.86 | 0.90 | 0.94 | 0.97 |
| Tot. biomass | 2455.64 | 2948.32 | 3398.62 | 0.86 | 0.91 | 0.95 |
| Taxa richness | 2636.61 | 3074.33 | 3482.67 | 0.93 | 0.95 | 0.97 |
| Shannon index | 2004.65 | 2304.48 | 2619.02 | 0.70 | 0.71 | 0.73 |
| Simpson index | 2366.18 | 2743.91 | 3109.11 | 0.83 | 0.85 | 0.86 |
| Resources | ||||||
| CPOM | 317.33 | 388.42 | 557.32 | 0.11 | 0.12 | 0.15 |
| FPOM | 2817.32 | 3147.32 | 3449.35 | 0.99 | 0.97 | 0.96 |
| WUA [m2] | HHS | |||||
|---|---|---|---|---|---|---|
| Discharge | 0.07 m3 s−1 | 0.26 m3 s−1 | 1.15 m3 s−1 | 0.07 m3 s−1 | 0.26 m3 s−1 | 1.15 m3 s−1 |
| Taxa | ||||||
| Baetis alpinus | 2636.38 | 4970.42 | 7198.79 | 0.58 | 0.75 | 0.81 |
| Baetis rhodani | 4102.96 | 6096.36 | 7821.66 | 0.90 | 0.92 | 0.88 |
| Baetis muticus | 4044.52 | 5996.58 | 7588.22 | 0.89 | 0.91 | 0.85 |
| Epeorus sylvicola | 233.07 | 848.19 | 1598.28 | 0.05 | 0.13 | 0.18 |
| Rhitrogena semicolorata | 923.63 | 3084.90 | 5226.90 | 0.20 | 0.47 | 0.59 |
| Ecdyonurus spp. | 4353.69 | 5704.12 | 6972.5 | 0.95 | 0.86 | 0.78 |
| Isoperla spp. | 2055.81 | 4209.87 | 6404.58 | 0.45 | 0.64 | 0.72 |
| Leuctra spp. | 1756.25 | 2812.96 | 4039.43 | 0.38 | 0.43 | 0.45 |
| Protonemura spp. | 124.33 | 500.14 | 1011.87 | 0.03 | 0.08 | 0.11 |
| Hydropsyche spp. | 621.15 | 1456.32 | 2431.77 | 0.14 | 0.22 | 0.27 |
| Rhyacophila spp. | 1202.68 | 3136.64 | 5148.59 | 0.26 | 0.47 | 0.58 |
| Orthocladiinae | 4402.18 | 6480.70 | 8742.53 | 0.96 | 0.98 | 0.98 |
| Tanypodinae | 4343.69 | 5564.64 | 6561.37 | 0.95 | 0.84 | 0.74 |
| Simuliidae | 325.72 | 1081.13 | 1977.75 | 0.07 | 0.16 | 0.22 |
| FFGs | ||||||
| Grazers | 3708.03 | 5853.05 | 8109.54 | 0.81 | 0.88 | 0.91 |
| Shredders | 1998.90 | 3176.06 | 4522.08 | 0.44 | 0.48 | 0.51 |
| P. filter feeders | 975.95 | 2194.06 | 3554.71 | 0.21 | 0.33 | 0.40 |
| Detritus C.G. | 4076.37 | 6204.29 | 8452.97 | 0.89 | 0.94 | 0.95 |
| Predators | 3295.45 | 5154.41 | 7206.70 | 0.72 | 0.78 | 0.81 |
| Comm. metrics | ||||||
| Total abundance | 4143.03 | 6263.97 | 8523.34 | 0.91 | 0.95 | 0.96 |
| Total biomass | 3971.58 | 6068.91 | 8327.91 | 0.87 | 0.92 | 0.94 |
| Taxa richness | 4244.45 | 6309.89 | 8570.50 | 0.93 | 0.95 | 0.96 |
| Shannon index | 3218.10 | 4739.16 | 6454.89 | 0.70 | 0.72 | 0.73 |
| Simpson index | 3805.70 | 5629.09 | 7653.01 | 0.83 | 0.85 | 0.86 |
| Resources | ||||||
| CPOM | 509.03 | 853.80 | 1351.71 | 0.11 | 0.13 | 0.15 |
| FPOM | 4506.49 | 6420.34 | 8547.81 | 0.99 | 0.97 | 0.96 |
Appendix C
| TAXA | Orino | Lesgiüna |
|---|---|---|
| Aeschinidae | x | |
| Cordulegaster spp. | x | |
| Ephemera danica | x | |
| Habroleptoides confusa | x | x |
| Epeorus alpicola | x | |
| Epeorus sylvicola | x | x |
| Ecdyonurus spp. | x | x |
| Rhytrogena degrangei | x | |
| Rhytrogena semicolorata | x | x |
| Baetis alpinus | x | x |
| Baetis rhodani | x | x |
| Alanites muticus | x | x |
| Siphlonurus lacustris | x | |
| Leptoblephia spp. early instar (?) | x | |
| Capnia nigra | x | |
| Leuctra spp. | x | x |
| Isoperla spp. | x | x |
| Isoperla grammatica | x | x |
| Isoperla rivulorum | x | x |
| Perla grandis | x | x |
| Protonemoura spp. | x | x |
| Protonemoura nimborum | x | x |
| Amphinemoura sulcicollis | x | x |
| Nemoura mortoni | x | |
| Limnephilidae | x | x |
| Hydroptila spp. | x | x |
| Silo/Lithax | x | x |
| Hydropsyche spp. | x | x |
| Allogamus auricollis | x | x |
| Ryacophila spp. | x | x |
| Sericostoma spp. | x | |
| Polycentropus spp. | x | |
| Drusus spp. | x | |
| Potamophylax spp. | x | |
| Philopotamus ludificatus | x | |
| Philopotamus montanus | x | |
| Philopotamus spp. | x | |
| Odontoceridae | x | x |
| Enoycila pusilla | x | |
| Psychodidae | x | |
| Stratiomidae | x | |
| Thaumaleidae | x | |
| Clinocera spp. | x | x |
| Prosimulium spp. | x | x |
| Simulium spp. | x | x |
| Dicranota spp. | x | x |
| Hexatoma spp. | x | x |
| Tipulidae | x | |
| Atherix marginata | x | x |
| Atherix ibis | x | |
| Tanypodinae | x | x |
| Orthocladiinae | x | x |
| Tanytarsini | x | x |
| Chironomini | x | x |
| Riolus spp. | x | x |
| Esolus spp. | x | x |
| Elmis spp. | x | x |
| Dryops spp. | x | x |
| Coleoptera | x | |
| Planaria | x | x |
| Oligochaeta | x | x |
| Hydracaridae | x | x |
| Nematomorpha | x | x |
| Mollusc (bivalve) | x |
References
- Bunn, S.E.; Arthington, A.H. Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environ. Manag. 2002, 30, 492–507. [Google Scholar] [CrossRef] [Green Version]
- Poff, N.L.; Zimmerman, J.K.H. Ecological responses to altered flow regimes: A literature review to inform the science and management of environmental flows. Freshw. Biol. 2010, 55, 194–205. [Google Scholar] [CrossRef]
- Gabbud, C.; Robinson, C.T.; Lane, S.N. Summer is in winter: Disturbance-driven shifts in macroinvertebrate communities following hydroelectric power exploitation. Sci. Total Environ. 2019, 650, 2164–2180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wohl, E.; Bledsoe, B.P.; Jacobson, R.B.; Poff, N.L.; Rathburn, S.L.; Walters, D.M.; Wilcox, A.C. The natural sediment regime in rivers: Broadening the foundation for ecosystem management. Bioscience 2015, 65, 358–371. [Google Scholar] [CrossRef] [Green Version]
- Zarfl, C.; Lumsdon, A.E.; Berlekamp, J.; Tydecks, L.; Tockner, K. A global boom in hydropower dam construction. Aquat. Sci. 2015, 77, 161–170. [Google Scholar] [CrossRef]
- Glachant, J.-M.; Saguan, M. Regimes for Granting the Right to Use Hydropower in Europe; European University Institute: Florence, Italy, 2015. [Google Scholar]
- Lange, K.; Meier, P.; Trautwein, C.; Schmid, M.; Robinson, C.T.; Weber, C.; Brodersen, J. Basin-scale effects of small hydropower on biodiversity dynamics. Front. Ecol. Environ. 2018, 16, 397–404. [Google Scholar] [CrossRef]
- Arthington, A.H.; Bhaduri, A.; Bunn, S.E.; Jackson, S.E.; Tharme, R.E.; Tickner, D.; Young, B.; Acreman, M.; Baker, N.; Capon, S.; et al. The Brisbane Declaration and Global Action Agenda on Environmental Flows (2018). Front. Environ. Sci. 2018, 6, 45. [Google Scholar] [CrossRef] [Green Version]
- Shenton, W.; Bond, N.R.; Yen, J.D.L.; Mac Nally, R. Putting the “ecology” into environmental flows: Ecological dynamics and demographic modelling. Environ. Manag. 2012, 50, 1–10. [Google Scholar] [CrossRef]
- Acreman, M.C.; Dunbar, M.J. Defining environmental river flow requirements? a review. Hydrol. Earth Syst. Sci. Discuss. Eur. Geosci. Union 2004, 8, 861–876. [Google Scholar] [CrossRef]
- Bovee, K.D. A Guide to stream habitat analysis using the Instream Flow Incremental Methodology. Instream Flow Inf. Pap. 1982, 12, 248. [Google Scholar]
- Rinaldi, M.; Belletti, B.; Van deBund, W.; Bertoldi, W.; Gurnell, A.; Buijse, T.; Mosselman, E. Review on Ecohydromorphological Methods; Deliverable of the EU FP7 REFORM project. 2013. Available online: http://www.reformrivers.eu/ (accessed on 31 July 2021).
- Conallin, J.; Boegh, E.; Jensen, J.K. Instream physical habitat modelling types: An analysis as stream hydromorphological modelling tools for EU water resource managers. Int. J. River Basin Manag. 2010, 8, 93–107. [Google Scholar] [CrossRef] [Green Version]
- Lamouroux, N.; Fuchs, U.; Mérigoux, S. Modelling the hydraulic preferences of benthic macroinvertebrates in small European streams. Freshw. Biol. 2007, 52, 145–164. [Google Scholar]
- Power, M.E.; Stout, R.J.; Cushing, C.E.; Harper, P.P.; Hauer, F.R.; Matthews, W.J.; Moyle, P.B.; Statzner, B. Biotic and abiotic controls in river and stream communities. J. North. Am. Benthol. Soc. 1988, 7, 456–479. [Google Scholar] [CrossRef]
- Bruder, A.; Salis, R.K.; Jones, P.E.; Matthaei, C.D. Biotic interactions modify multiple-stressor effects on juvenile brown trout in an experimental stream food web. Glob. Chang. Biol. 2017, 23, 3882–3894. [Google Scholar] [CrossRef]
- Jowett, I.G. Hydraulic constraints on habitat suitability for benthic invertebrates in gravel-bed rivers. River Res. Appl. 2003, 19, 495–507. [Google Scholar] [CrossRef]
- Statzner, B.; Gore, J.A.; Resh, V.H. Hydraulic stream ecology: Observed patterns and potential applications. J. North. Am. Benthol. Soc. 1988, 7, 307–360. [Google Scholar] [CrossRef]
- Hart, D.D.; Finelli, C.M. Physical-biological coupling in streams: The pervasive effects of flow on benthic organisms. Annu. Rev. Ecol. Syst. 1999, 30, 363–395. [Google Scholar] [CrossRef]
- Statzner, B.; Higler, B. Stream hydraulics as a major determinant of benthic invertebrate zonation patterns. Freshw. Biol. 1986, 16, 127–139. [Google Scholar] [CrossRef]
- Lepori, F.; Palm, D.; Malmqvist, B. Effects of stream restoration on ecosystem functioning: Detritus retentiveness and decomposition. J. Appl. Ecol. 2005, 42, 228–238. [Google Scholar] [CrossRef]
- Dewson, Z.S.; James, A.B.W.; Death, R.G. Stream ecosystem functioning under reduced flow conditions. Ecol. Appl. 2007, 17, 1797–1808. [Google Scholar] [CrossRef] [Green Version]
- Hynes, H.B.N. The Ecology of Stream Insects. Annu. Rev. Entomol. 1970, 15, 25–42. [Google Scholar] [CrossRef]
- Moog, O. Fauna Aquatica Austriaca, 2nd ed.; Wasserwirtschaftskataster, Bundesministerium für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Vienna, Austria, 2002. [Google Scholar]
- Cummins, K.W.; Klug, M.J. Feeding ecology of stream invertebrates. Annu. Rev. Ecol. Syst. 1979, 10, 147–172. [Google Scholar] [CrossRef]
- Mangadze, T.; Wasserman, R.J.; Froneman, P.W.; Dalu, T. Macroinvertebrate functional feeding group alterations in response to habitat degradation of headwater Austral streams. Sci. Total Environ. 2019, 695, 133910. [Google Scholar] [CrossRef] [PubMed]
- Small, M.J.; Doyle, M.W.; Fuller, R.L.; Manners, R.B. Hydrologic versus geomorphic limitation on CPOM storage in stream ecosystems. Freshw. Biol. 2008, 53, 1618–1631. [Google Scholar] [CrossRef]
- Webster, J.R.; Benfield, E.F.; Golladay, S.W.; Hill, B.H.; Hornick, L.E.; Kazmierczak, R.F.; Perry, W.B. Experimental studies of physical factors affecting seston transport in streams. Limnol. Oceanogr. 1987, 32, 848–863. [Google Scholar] [CrossRef]
- Biggs, B.J.F.; Close, M.E. Periphyton biomass dynamics in gravel bed rivers: The relative effects of flows and nutrients. Freshw. Biol. 1989, 22, 209–231. [Google Scholar] [CrossRef]
- Lamouroux, N.; Dolédec, S.; Gayraud, S. Biological traits of stream macroinvertebrate communities: Effects of microhabitat, reach, and basin filters. J. North. Am. Benthol. Soc. 2004, 23, 449–466. [Google Scholar] [CrossRef]
- Townsend, C.R.; Hildrew, A.G. Species traits in relation to a habitat templet for river systems. Freshw. Biol. 1994, 31, 265–275. [Google Scholar] [CrossRef]
- Mérigoux, S.; Dolédec, S. Hydraulic requirements of stream communities: A case study on invertebrates. Freshw. Biol. 2004, 49, 600–613. [Google Scholar] [CrossRef]
- Mérigoux, S.; Lamouroux, N.; Olivier, J.M.; Dolédec, S. Invertebrate hydraulic preferences and predicted impacts of changes in discharge in a large river. Freshw. Biol. 2009, 54, 1343–1356. [Google Scholar] [CrossRef]
- Jowett, I.G.; Richardson, J. Microhabitat preferences of benthic invertebrates in a New Zealand river and the development of in-stream flow-habitat models of Deleatidium spp. N. Zeal. J. Mar. Freshw. Res. 1990, 24, 19–30. [Google Scholar] [CrossRef]
- Truffer, B.; Markard, J.; Bratrich, C.; Wehrli, B. Green electricity from alpine hydropower plants. Mt. Res. Dev. 2001, 21, 19–24. [Google Scholar] [CrossRef]
- Lamouroux, N.; Mérigoux, S.; Dolédec, S.; Snelder, T.H. Transferability of hydraulic preference models for aquatic macroinvertebrates. River Res. Appl. 2013, 29, 933–937. [Google Scholar] [CrossRef]
- Mérigoux, S.; Schneider, M. Invertebrates and near-bed hydraulic forces: Combining data from different EU countries to better assess habitat suitability. In Proceedings of the COST 626—European Aquatic Modelling Network: Proceedings from the Final Meeting, Silkeborg, Denmark, 19–20 May 2005; pp. 241–248. [Google Scholar]
- Meier, W.; Bonjour, C.; Wüest, A.; Reichert, P. Modeling the effect of water diversion on the temperature of mountain streams. J. Environ. Eng. 2003, 129, 755–764. [Google Scholar] [CrossRef]
- Bratrich, C.; Truffer, B.; Jorde, K.; Markard, J.; Meier, W.; Peter, A.; Schneider, M.; Wehrli, B. Green hydropower: A new assessment procedure for river management. River Res. Appl. 2004, 20, 865–882. [Google Scholar] [CrossRef]
- Statzner, B.; Müller, R. Standard hemispheres as indicators of flow characteristics in lotic benthic research. Freshw. Biol. 1989, 21, 445–459. [Google Scholar] [CrossRef]
- Statzner, B.; Kohmann, F.; Hildrew, A.G. Calibration of FST-hemispheres against bottom shear stress in a laboratory flume. Freshw. Biol. 1991, 26, 227–231. [Google Scholar] [CrossRef]
- Kopecki, I. Calculational Approach to FST- Hemispheres for Multiparametrical Benthos Habitat Modelling; Universität Stuttgart: Stuttgart, Germany, 2008. [Google Scholar]
- Dittrich, A.; Schmedtje, U. Indicating shear stress with FST-hemisphere—Effects of stream bottom topography and water depth. Freshw. Biol. 1995, 34, 107–121. [Google Scholar] [CrossRef]
- Scherer, M. Typisierungskonzept zur Festlegung Einer Oekologisch Begruendeten Mindestwassermenge; Inst. fuer Wasserwirtschaft und Kulturtechnik, Fakultaet fuer Bauingenieur- und Vermessungswesen; Karlsruhe University: Karlsruhe, Germany, 1999. [Google Scholar]
- Tachet, H.; Richoux, P.; Bournaud, M.; Usseglio-Polatera, P. Invertébrés d’eau Douce: Systématique, Biologie, Écologie, 2nd ed.; CNRS Editions: Paris, France, 2010; ISBN 2271057450. [Google Scholar]
- Belfiore, C. Guide per il Riconoscimento Delle specie ANIMALI delle Acque Interne Italiane. 24. Efemerotteri (Ephemeroptera); CNR, Consiglio Nazionale delle Ricerche: Verona, Italy, 1983. [Google Scholar]
- Consiglio, C. Guide per il Riconoscimento Delle Specie Animali Delle Acque Interne Italiane. 9. Plecotteri (Plecoptera); CNR, Consiglio Nazionale delle Ricerche: Verona, Italy, 1980. [Google Scholar]
- Moretti, G. Guide per il Riconoscimento Delle Specie Animali Delle Acque Interne Italiane. 19. Tricotteri (Tricoptera); CNR, Consiglio Nazionale delle Ricerche: Verona, Italy, 1983. [Google Scholar]
- Bauernfeind, E.; Lechthaler, W. Ephemeroptera: Key to larvae from Central Europe; CD-Edition: Vienna, Austria, 2014. [Google Scholar]
- Lechthaler, W.; Stockinger, W. Trichoptera: Key to larvae from Central Europe; CD-Edition: Vienna, Austria, 2005. [Google Scholar]
- Meyer, E.I. The relationship between body length parameters and dry mass in running water invertebrates. Arch. Fur Hydrobiol. 1989, 117, 191–203. [Google Scholar]
- Burgherr, P.; Meyer, E.I. Regression analysis of linear body dimensions vs. dry mass in stream macroinvertebrates. Arch. Fur Hydrobiol. 1997, 139, 101–112. [Google Scholar] [CrossRef]
- Benke, A.C.; Huryn, A.D.; Smock, L.A.; Wallace, J.B. Length-mass relationships for freshwater macroinvertebrates in North America with particular reference to the southeastern United States. J. North. Am. Benthol. Soc. 1999, 18, 308–343. [Google Scholar] [CrossRef] [Green Version]
- Johnston, T.A.; Cunjak, R.A. Dry mass-length relationships for benthic insects: A review with new data from Catamaran Brook, New Brunswick, Canada. Freshw. Biol. 1999, 41, 653–674. [Google Scholar] [CrossRef]
- Gonzalez, J.M.; Basaguren, A.; Pozo, J. Size-mass relationships of stream invertebrates in a northern Spain stream. Hydrobiology 2002, 489, 131–137. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Jorde, K. Bottom shear stress pattern and its ecological impact. Int. J. Sediment. Transp. 1997, 12, 369–378. [Google Scholar]
- Jorde, K.; Bratrich, C. Influence of river bed morphology and flow regulations in diverted streams on bottom shear stress pattern and hydraulic habitat. In Advances in River Bottom Ecology IV; Bretschko, G., Helesic, J., Eds.; Backhuys: Kerkwerve, The Netherlands, 1998; pp. 47–63. [Google Scholar]
- Kopecki, I.; Schneider, M. Handbuch für das Habitatsimulationsmodell CASiMiR; Modul: CASiMiR-Benthos, Institut für Wasserbau der Universität Stuttgart: Stuttgart, Germany, 2010; p. 26. [Google Scholar]
- Lamouroux, N.; Statzner, B.; Fuchs, U.; Kohmann, F.; Schmedtje, U. An unconventional approach to modeling spatial and temporal variability of local shear stress in stream segments. Water Resour. Res. 1992, 28, 3251–3258. [Google Scholar] [CrossRef]
- Lancaster, J.; Hildrew, A.G. Flow refugia and the microdistribution of lotic macroinvertebrates. J. North. Am. Benthol. Soc. 1993, 12, 385–393. [Google Scholar] [CrossRef]
- Imhof, A. Habitatansprüche und Verhalten von Perla grandis rambur (Plecoptera: Perlidae) und Anderen Räuberischen Steinfliegenlarven. Ph.D. Thesis, ETH Zurich, Zürich, Switzerland, 1994. [Google Scholar]
- Brown, B.L. Spatial heterogeneity reduces temporal variability in stream insect communities. Ecol. Lett. 2003, 6, 316–325. [Google Scholar] [CrossRef]
- Lepori, F.; Palm, D.; Brännäs, E.; Malmqvist, B. Does restoration of structural heterogeneity in streams enhance fish and macroinvertebrate diversity? Ecol. Appl. 2005, 15, 2060–2071. [Google Scholar] [CrossRef]
- Collier, K.J. Flow preferences of larval chironomidae (diptera) in Tongariro river, New Zealand. N. Zeal. J. Mar. Freshw. Res. 1993, 27, 219–226. [Google Scholar] [CrossRef]
- Hynes, H.B.N. The Ecology of Running Waters; University of Toronto Press: Toronto, ON, Canada, 1970. [Google Scholar]
- Fonseca, D.M.; Hart, D.D. Colonization history masks habitat preferences in local distributions of stream insects. Ecology 2001, 82, 2897–2910. [Google Scholar] [CrossRef]
- Townsend, C.R.; Hildrew, A.G. Field experiments on the drifting, colonization and continuous redistribution of stream benthos. J. Anim. Ecol. 1976, 45, 759. [Google Scholar] [CrossRef]
- Wallace, J.B.; Merritt, R.W. Filter-feeding ecology of aquatic insects. Annu. Rev. Entomol. 1980, 25, 103–132. [Google Scholar] [CrossRef] [Green Version]
- Lytle, D.A.; Poff, N.L. Adaptation to natural flow regimes. Trends Ecol. Evol. 2004, 19, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.T.; Siebers, A.R.; Ortlepp, J. Long-term ecological responses of the River Spöl to experimental floods. Freshw. Sci. 2018, 37, 433–447. [Google Scholar] [CrossRef] [Green Version]
- Bruno, D.; Belmar, O.; Maire, A.; Morel, A.; Dumont, B.; Datry, T. Structural and functional responses of invertebrate communities to climate change and flow regulation in alpine catchments. Glob. Chang. Biol. 2019, 25, 1612–1628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Parliament and Council of the European Union Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. Eur. Communities 2000, 372, 1–73.
- Acreman, M.C.; Ferguson, A.J.D. Environmental flows and the European Water Framework Directive. Freshw. Biol. 2010, 55, 32–48. [Google Scholar] [CrossRef]
- Buffagni, A.; Cazzola, M.; López-Rodríguez, M.J.; Alba-Tercedor, J.; Armanini, D.G. Distribution and Ecological Preferences of European freshwater Organisms. Volume 3—Ephemeroptera; Schmidt-Kloiber, A., Hering, D., Eds.; Pensoft Publishers: Sofia, Bulgaria, 2009; ISBN 9789546424419. [Google Scholar]
- Sagnes, P.; Mérigoux, S.; Péru, N. Hydraulic habitat use with respect to body size of aquatic insect larvae: Case of six species from a French Mediterranean type stream. Limnol.-Ecol. Manag. Inl. Waters 2008, 38, 23–33. [Google Scholar] [CrossRef] [Green Version]
- Koljonen, S.; Louhi, P.; Mäki-Petäys, A.; Huusko, A.; Muotka, T. Quantifying the effects of in-stream habitat structure and discharge on leaf retention: Implications for stream restoration. Freshw. Sci. 2012, 31, 1121–1130. [Google Scholar] [CrossRef]
- Frutiger, A.; SCHIB, J. Limitations of FST hemispheres in lotic benthos research. Freshw. Biol. 1993, 463–474. [Google Scholar] [CrossRef]
- Möbes-Hansen, B.; Waringer, J.A. The influence of hydraulic stress on microdistribution patterns of zoobenthos in a sandstone brook (Weidlingbach, Lower Austria). Int. Rev. Hydrobiol. 1998, 5–6, 381–396. [Google Scholar] [CrossRef]
- Jowett, I.G.; Richardson, J.; Biggs, B.J.F.; Hickey, C.W.; Quinn, J.M. Microhabitat preferences of benthic invertebrates and the development of generalized Deleatidium spp habitat suitability curves, applied to 4 New Zealand rivers. N. Zeal. J. Mar. Freshw. Res. 1991, 25, 187–199. [Google Scholar] [CrossRef] [Green Version]
- Poff, N.L.; Ward, J.V. Implications of streamflow variability and predictability for lotic community structure: A regional analysis of streamflow patterns. Can. J. Fish. Aquat. Sci. 1989, 46, 1805–1818. [Google Scholar] [CrossRef]
- Rosenfeld, J. Developing flow–ecology relationships: Implications of nonlinear biological responses for water management. Freshw. Biol. 2017, 62, 1305–1324. [Google Scholar] [CrossRef] [Green Version]
- Bruder, A.; Frainer, A.; Rota, T.; Primicerio, R. The importance of ecological networks in multiple-stressor research and management. Front. Environ. Sci. 2019, 7, 59. [Google Scholar] [CrossRef] [Green Version]
- Poff, N.L.; Allan, J.D.; Bain, M.B.; Karr, J.R.; Prestegaard, K.L.; Richter, B.D.; Sparks, R.E.; Stromberg, J.C. The Natural Flow Regime: A paradigm for river conservation and restoration. Bioscience 1997, 47, 769–784. [Google Scholar] [CrossRef]
- Poff, N.L.; Ward, J.V. Physical habitat template of lotic systems: Recovery in the context of historical pattern of spatiotemporal heterogeneity. Environ. Manag. 1990, 14, 629–645. [Google Scholar] [CrossRef]
- Yarnell, S.M.; Petts, G.E.; Schmidt, J.C.; Whipple, A.A.; Beller, E.E.; Dahm, C.N.; Goodwin, P.; Viers, J.H. Functional flows in modified riverscapes: Hydrographs, habitats and opportunities. Bioscience 2015, 65, 963–972. [Google Scholar] [CrossRef] [Green Version]
- Acreman, M.C.; Overton, I.C.; King, J.; Wood, P.J.; Cowx, I.G.; Dunbar, M.J.; Kendy, E.; Young, W.J. The changing role of ecohydrological science in guiding environmental flows. Hydrol. Sci. J. 2014, 59, 433–450. [Google Scholar] [CrossRef]
- Davies, P.M.; Naiman, R.J.; Warfe, D.M.; Pettit, N.E.; Arthington, A.H.; Bunn, S.E. Flow–ecology relationships: Closing the loop on effective environmental flows. Mar. Freshw. Res. 2012, 133–141. [Google Scholar] [CrossRef]

| DOC mg C/L | k20 °C µs/cm2 | pH | TN mg /L | PO4 –P µg /L | TP µg /L | DO 1 % | t 1 °C | |
|---|---|---|---|---|---|---|---|---|
| Orino | 3.3 ± 0.4 | 68 ± 32 | 7.1 ± 0.5 | 1.1 ± 0.4 | 1.5 ± 0.6 | 5.7 ± 2.6 | 100.4 ± 1.4 | 6.4 ± 3.7 |
| Lesgiüna | 2.0 ± 0.5 | 46 ± 17 | 7.0 ± 0.0 | 0.8 ± 0.2 | 1.5 ± 0.2 | 3.2 ± 2.0 | 101.2 ± 2.3 | 4.7 ± 2.5 |
| Discharge (m3·s−1) | Wetted Area (m2) | FSTμ (N) | Vμ (ms−1) | Dμ (m) | DGS (mm) | |
|---|---|---|---|---|---|---|
| Orino | ||||||
| Winter (baseflow) | 0.02 | 2848 | 1 ± 1.7 | 0.06 ± 0.07 | 0.06 ± 0.04 | 64–128 |
| Early spring | 0.09 | 3236 | 3 ± 2.4 | 0.11 ± 0.11 | 0.07 ± 0.05 | 64–128 |
| Late spring | 0.43 | 3596 | 5 ± 3.0 | 0.19 ± 0.15 | 0.15 ± 0.06 | 64–128 |
| Lesgiüna | ||||||
| Winter (baseflow) | 0.06 | 4567 | 1 ± 1.8 | 0.07 ± 0.06 | 0.07 ± 0.03 | 64–128 |
| Early spring | 0.26 | 6617 | 3 ± 2.4 | 0.16 ± 0.14 | 0.10 ± 0.10 | 64–128 |
| Late spring | 1.15 | 8885 | 5 ± 3.1 | 0.20 ± 0.18 | 0.15 ± 0.07 | 128–256 |
| FST | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R2 | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | |
| Taxa | ||||||||||||||||||
| E. sylvicola +*** | 0.39 | 0 | 0.03 | 0.07 | 0.1 | 0.13 | 0.16 | 0.20 | 0.24 | 0.27 | 0.31 | 0.35 | 0.39 | 0.43 | 0.47 | 0.52 | 0.56 | 0.6 |
| Ecdyonurus spp.* | 0.10 | 1 | 0.98 | 0.95 | 0.92 | 0.88 | 0.84 | 0.79 | 0.73 | 0.67 | 0.60 | 0.53 | 0.44 | 0.36 | 0.27 | 0.17 | 0.06 | 0.05 |
| R.semicolorata *** | 0.29 | 0.01 | 0.15 | 0.30 | 0.43 | 0.55 | 0.66 | 0.75 | 0.82 | 0.89 | 0.94 | 0.97 | 0.99 | 1 | 0.99 | 0.97 | 0.94 | 0.89 |
| B. alpinus *** | 0.48 | 0.42 | 0.55 | 0.66 | 0.76 | 0.84 | 0.90 | 0.95 | 0.98 | 1 | 0.99 | 0.98 | 0.96 | 0.91 | 0.85 | 0.77 | 0.68 | 0.57 |
| B. rhodani ** | 0.20 | 0.84 | 0.91 | 0.95 | 0.99 | 1 | 0.99 | 0.98 | 0.94 | 0.89 | 0.82 | 0.73 | 0.062 | 0.50 | 0.36 | 0.20 | 0.03 | 0 |
| A.muticus | 0.04 | 0.82 | 0.90 | 0.95 | 0.99 | 1 | 0.99 | 0.97 | 0.92 | 0.86 | 0.77 | 0.66 | 0.54 | 0.39 | 0.22 | 0.04 | 0 | 0 |
| Leuctra spp. +* | 0.12 | 0.36 | 0.37 | 0.39 | 0.41 | 0.42 | 0.44 | 0.46 | 0.48 | 0.51 | 0.53 | 0.55 | 0.58 | 0.60 | 0.63 | 0.66 | 0.69 | 0.72 |
| Isoperla spp. ** | 0.17 | 0.30 | 0.41 | 0.52 | 0.61 | 0.70 | 0.77 | 0.84 | 0.89 | 0.93 | 0.96 | 0.99 | 1 | 0.99 | 0.98 | 0.97 | 0.94 | 0.90 |
| Protonemura spp. +*** | 0.54 | 0 | 0.02 | 0.03 | 0.05 | 0.07 | 0.09 | 0.12 | 0.14 | 0.17 | 0.21 | 0.24 | 0.28 | 0.32 | 0.36 | 0.41 | 0.45 | 0.50 |
| Hydropsyche spp. +*** | 0.40 | 0.08 | 0.12 | 0.15 | 0.19 | 0.23 | 0.26 | 0.30 | 0.34 | 0.37 | 0.41 | 0.45 | 0.49 | 0.52 | 0.56 | 0.60 | 0.64 | 0.68 |
| Rhyacophila spp.*** | 0.34 | 0.10 | 0.22 | 0.33 | 0.44 | 0.53 | 0.62 | 0.69 | 0.76 | 0.82 | 0.87 | 0.92 | 0.95 | 0.98 | 0.99 | 1 | 0.99 | 0.98 |
| Simuliidae +*** | 0.43 | 0.01 | 0.05 | 0.09 | 0.13 | 0.17 | 0.21 | 0.25 | 0.29 | 0.33 | 0.38 | 0.42 | 0.46 | 0.50 | 0.54 | 0.58 | 0.62 | 0.66 |
| Tanypodinae | 0.10 | 1 | 0.98 | 0.96 | 0.92 | 0.88 | 0.82 | 0.76 | 0.68 | 0.60 | 0.50 | 0.39 | 0.28 | 0.15 | 0.01 | 0 | 0 | 0 |
| Orthocladiinae | 0.01 | 0.95 | 0.96 | 0.97 | 0.98 | 0.99 | 0.99 | 1 | 1 | 1 | 1 | 1 | 0.99 | 0.99 | 0.98 | 0.97 | 0.96 | 0.94 |
| FFGs | ||||||||||||||||||
| Grazers *** | 0.33 | 0.75 | 0.80 | 0.84 | 0.88 | 0.92 | 0.94 | 0.97 | 0.98 | 0.99 | 1 | 1 | 0.99 | 0.98 | 0.97 | 0.95 | 0.92 | 0.89 |
| Shredders +** | 0.20 | 0.41 | 0.43 | 0.45 | 0.46 | 0.48 | 0.50 | 0.52 | 0.54 | 0.56 | 0.58 | 0.60 | 0.63 | 0.65 | 0.68 | 0.70 | 0.73 | 0.76 |
| P. filter feeders +*** | 0.46 | 0.13 | 0.19 | 0.24 | 0.30 | 0.35 | 0.40 | 0.45 | 0.49 | 0.54 | 0.58 | 0.62 | 0.66 | 0.70 | 0.73 | 0.77 | 0.80 | 0.83 |
| Detritus C.G. ** | 0.17 | 0.85 | 0.89 | 0.92 | 0.94 | 0.96 | 0.98 | 0.99 | 1 | 1 | 1 | 0.99 | 0.98 | 0.96 | 0.94 | 0.92 | 0.89 | 0.85 |
| Predators ** | 0.14 | 0.68 | 0.71 | 0.74 | 0.76 | 0.79 | 0.81 | 0.84 | 0.86 | 0.88 | 0.90 | 0.91 | 0.93 | 0.94 | 0.96 | 0.97 | 0.98 | 1 |
| Comm. metrics | ||||||||||||||||||
| Tot. biomass *** | 0.26 | 0.83 | 0.86 | 0.89 | 0.91 | 0.94 | 0.95 | 0.97 | 0.98 | 0.99 | 1 | 1 | 1 | 1 | 0.99 | 0.98 | 0.96 | 0.95 |
| Tot. abundance ** | 0.20 | 0.87 | 0.90 | 0.93 | 0.95 | 0.97 | 0.98 | 0.99 | 1 | 1 | 1 | 1 | 0.99 | 0.98 | 0.96 | 0.94 | 0.92 | 0.89 |
| Taxa richness * | 0.16 | 0.90 | 0.91 | 0.92 | 0.94 | 0.95 | 0.96 | 0.97 | 0.98 | 0.99 | 1 | 1 | 1 | 1 | 1 | 0.99 | 0.99 | 0.98 |
| Shannon index * | 0.16 | 0.70 | 0.70 | 0.70 | 0.71 | 0.71 | 0.72 | 0.73 | 0.73 | 0.74 | 0.75 | 0.76 | 0.77 | 0.79 | 0.80 | 0.81 | 0.83 | 0.84 |
| Simpson index * | 0.12 | 0.82 | 0.83 | 0.84 | 0.85 | 0.85 | 0.86 | 0.87 | 0.87 | 0.88 | 0.89 | 0.90 | 0.90 | 0.91 | 0.92 | 0.93 | 0.94 | 0.95 |
| Resources | ||||||||||||||||||
| CPOM * | 0.18 | 0.10 | 0.10 | 0.10 | 0.11 | 0.11 | 0.12 | 0.14 | 0.16 | 0.18 | 0.20 | 0.23 | 0.26 | 0.30 | 0.34 | 0.38 | 0.43 | 0.48 |
| FPOM | <0.01 | 1 | 0.99 | 0.98 | 0.97 | 0.97 | 0.96 | 0.95 | 0.94 | 0.94 | 0.93 | 0.92 | 0.91 | 0.90 | 0.89 | 0.88 | 0.87 | 0.86 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Consoli, G.; Lepori, F.; Robinson, C.T.; Bruder, A. Predicting Macroinvertebrate Responses to Water Abstraction in Alpine Streams. Water 2021, 13, 2121. https://doi.org/10.3390/w13152121
Consoli G, Lepori F, Robinson CT, Bruder A. Predicting Macroinvertebrate Responses to Water Abstraction in Alpine Streams. Water. 2021; 13(15):2121. https://doi.org/10.3390/w13152121
Chicago/Turabian StyleConsoli, Gabriele, Fabio Lepori, Christopher T. Robinson, and Andreas Bruder. 2021. "Predicting Macroinvertebrate Responses to Water Abstraction in Alpine Streams" Water 13, no. 15: 2121. https://doi.org/10.3390/w13152121
APA StyleConsoli, G., Lepori, F., Robinson, C. T., & Bruder, A. (2021). Predicting Macroinvertebrate Responses to Water Abstraction in Alpine Streams. Water, 13(15), 2121. https://doi.org/10.3390/w13152121





