Dependence of Long-Term Dynamics of Zooplankton in the Ob River on Interannual Changes in Hydrological and Hydrochemical Parameters
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Zooplankton
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Savkin, V.M.; Dvurechenskaya, S.Y. Resources-related and water-environmental problems of the integrated use of the Novosibirsk reservoir. Water Resour. 2014, 41, 446–453. [Google Scholar] [CrossRef]
- Modern State of Water Resources and Functioning of Water Management Complex of the Basin of the Ob and Irtysh; Vinokurov, Y.I., Puzanov, A.V., Bezmaternykh, D.M., Eds.; Publishing House of the SB RAS: Novosibirsk, Russia, 2012; 236p. (In Russian) [Google Scholar]
- Savichev, O.G. Rivers of the Tomsk Region: Condition, Protection and Use; Tomsk Polytechnic University: Tomsk, Russia, 2003; 170p. (In Russian) [Google Scholar]
- Viroux, L. Zooplankton distribution in flowing waters and its implications for sampling: Case studies in the River Meuse (Belgium) and the River Moselle (France, Luxembourg). J. Plankton Res. 1999, 21, 1231–1248. [Google Scholar] [CrossRef]
- Combs, S.A. Protecting freshwater ecosystem in the face of global climate change. In Buying Time: A User’s Manual for Building Resistance to Climate Change in Natural Systems; Hoffman, L.J., Ed.; WWF: Morges, Switzerland, 2003; pp. 177–216. [Google Scholar]
- Massicotte, P.; Frenette, J.J.; Proulx, R.; Pinel-Alloul, B.; Bertolo, A. Riverscape heterogeneity explains spatial variation in zooplankton functional evenness and biomass in a large river ecosystem. Landsc. Ecol. 2014, 29, 67–79. [Google Scholar] [CrossRef]
- Baranyi, C.; Hein, T.; Holarek, C.; Keckeis, S.; Schiemer, F. Zooplankton Biomass and Community Structure in a Danube River Floodplain System: Effects of Hydrology. Freshw. Biol. 2002, 47, 1–10. [Google Scholar] [CrossRef]
- Napiórkowski, P.; Napiórkowska, T. The diversity and longitudinal changes of zooplankton in the lower course of a large, regulated European river (the lower Vistula River, Poland). Biologia 2013, 68, 1163–1170. [Google Scholar] [CrossRef]
- Bertani, I.; Segers, H.; Rossetti, G. Biodiversity down by the flow: New records of monogonont rotifers for Italy found in the Po River. J. Limnol. 2010, 69, 321–328. [Google Scholar] [CrossRef][Green Version]
- Lair, N. A review of regulation mechanisms of metazoan plankton in riverine ecosystems: Aquatic habitat versus biota. River Res. Appl. 2006, 22, 567–593. [Google Scholar] [CrossRef]
- Steinberg, D.K.; Condon, R.H. Zooplankton of the York River. J. Coast. Res. 2009, 57, 66–79. [Google Scholar] [CrossRef]
- Zhao, K.; Song, K.; Pan, Y.; Wang, L.; Da, L.; Wang, Q. Metacommunity structure of zooplankton in river networks: Roles of environmental and spatial factors. Ecol. Indic. 2017, 73, 96–104. [Google Scholar] [CrossRef]
- Tóth, F.; Zsuga, K.; Kerepeczki, É.; Berzi-Nagy, L.; Körmöczi, L.; Lövei, G.L. Seasonal differences in taxonomic diversity of rotifer communities in a Hungarian lowland oxbow lake exposed to aquaculture effluent. Water 2020, 12, 1300. [Google Scholar] [CrossRef]
- Sindt, A.R.; Wolf, M.C. Spatial and temporal trends of Minnesota River phytoplankton and zooplankton. River Res. Appl. 2021, 1–20. [Google Scholar] [CrossRef]
- Gundrizer, A.N.; Zalozny, N.A.; Golubykh, O.S.; Popkova, L.A.; Ruzanova, A.I. Studies of hydrobionts from Middle Ob. Sib. Ecol. J. 2000, 3, 315–322. (In Russian) [Google Scholar]
- Turner, J. Zooplankton Feeding Ecology: Contents of Fecal Pellets of the Copepod Centropages velificatus from Waters near the Mouth of the Mississippi River. Biol. Bull. 1987, 173, 377–386. [Google Scholar] [CrossRef]
- Turner, J. Zooplankton feeding ecology: Does a diet of Phaeocystis support good copepod grazing, survival, egg production and egg hatching success? J. Plankton Res. 2002, 24, 1185–1195. [Google Scholar] [CrossRef]
- Dickerson, K.D.; Medley, K.A.; Havel, J.E. Spatial variation in zooplankton community structure is related to hydrologic flow units in the Missouri river, USA. River Res. Appl. 2010, 26, 605–618. [Google Scholar] [CrossRef]
- Porter, K.G.; Robbins, E.I. Zooplankton Fecal Pellets Link Fossil Fuel and Phosphate Deposits. Science 1981, 212, 931–933. [Google Scholar] [CrossRef] [PubMed]
- Gutelmakher, B.L. Plankton metabolism as a whole. In Trophometabolic Interactions of Zoo- and Phytoplankton L.; Zoological Institute of USSR Academy of Sciences: Saint Petersburg, Russia, 1986; 155p. (In Russian) [Google Scholar]
- Yermolaeva, N.I. The role of zooplankton in the formation of sapropel in the lakes south of Western Siberia. World Sci. Cult. Educ. 2013, 6, 545–549. (In Russian) [Google Scholar]
- Yermolaeva, N.I.; Zarubina, E.Y.; Puzanov, A.V.; Romanov, R.E.; Leonova, G.A. Hydrobiological conditions of sapropel formation in lakes in the South of Western Siberia. Water Resour. 2016, 43, 129–140. [Google Scholar] [CrossRef]
- Abdulwahab, S.; Rabee, A.M. Ecological factors affecting the distribution of the zooplankton community in the Tigris River at Baghdad region, Iraq. Egypt. J. Aquat. Res. 2015, 41, 187–196. [Google Scholar] [CrossRef]
- Lim, B.; Han, S.; Choi, I.; Yoon, J.; Lee, J.; Cheon, S.; Cho, K. Evaluation of physico-chemical parameters regulating zooplankton community structure in the Geum River, Korea. Iran. J. Fish. Sci. 2020, 19, 352–371. [Google Scholar] [CrossRef]
- Ejsmont-Karabin, J. The usefulness of zooplankton as lake ecosystem indicators: Rotifer trophic state index. Pol. J. Ecol. 2012, 60, 339–350. [Google Scholar]
- Sládeček, V. Rotifers as indicators of water quality. Hydrobiologia 1983, 100, 169–201. [Google Scholar] [CrossRef]
- Krupa, E.; Romanova, S.; Berkinbaev, G.; Yakovleva, N.; Sadvakasov, E. Zooplankton as Indicator of the Ecological State of Protected Aquatic Ecosystems (Lake Borovoe, Burabay National Nature Park, Northern Kazakhstan). Water 2020, 12, 2580. [Google Scholar] [CrossRef]
- Pashkova, O.V. Zooplankton as Indicator of Organic and Toxic Pollution and Ecological State of Aquatic Ecosystems (a Review). Hydrobiol. J. 2013, 49, 3–20. [Google Scholar] [CrossRef]
- Ermolaeva, N.I.; Dvurechenskaya, S.Y. Regional indices of the indicator significance of zooplanktonic organisms in water bodies of Southern Western Siberia. Russ. J. Ecol. 2013, 44, 527–531. [Google Scholar] [CrossRef]
- Saunders, J.F., III; Lewis, W.M.J. Zooplankton abundance in the lower Orinoco River, Venezuela. Limnol. Oceanogr. 1989, 34, 397–409. [Google Scholar] [CrossRef]
- Burger, D.F.; Hogg, I.D.; Green, J.D. Distribution and abundance of zooplankton in the Waikato River, New Zealand. Hydrobiologia 2002, 479, 31–38. [Google Scholar] [CrossRef]
- Rossetti, G.; Viaroli, P.; Ferrari, I. Role of abiotic and biotic factors in structuring the metazoan plankton community in a lowland river. River Res. Appl. 2009, 25, 814–835. [Google Scholar] [CrossRef]
- Deksne, R.; Škute, A.; Gruberts, D.; Paidere, J. Effects of climate change on zooplankton community structure of the middle stretch of the Daugava river over the last 50 years. Ecohydrol. Hydrobiol. 2011, 11, 79–95. [Google Scholar] [CrossRef]
- Shevtsova, L.V.; Guleykova, L.V. Long-term dynamics of zooplankton of the Desna river. Hydrobiol. J. 2005, 41, 3–14. [Google Scholar] [CrossRef]
- Lansac-Tôha, F.A.; Bonecker, C.C.; Velho, L.F.M.; Simões, N.R.; Dias, J.D.; Alves, G.M.; Takahashi, E.M. Biodiversity of zooplankton communities in the Upper Paraná River floodplain: Interannual variation from Long-Term studies. Braz. J. Biol. 2009, 69 (Suppl. 2), 539–549. [Google Scholar] [CrossRef] [PubMed]
- Deksne, R.; Škute, A. The influence of ecohydrological factors on the cenosis of the Daugava River zooplankton. Acta Zool. Litu. 2011, 21, 133–144. [Google Scholar] [CrossRef]
- Kulakov, D.V. Seasonal and interannual changes of the zooplankton of the Neman River. Principy èkologii 2018, 7, 87–102. [Google Scholar] [CrossRef]
- Chará-Serna, A.M.; Casper, A. How do large river zooplankton communities respond to abiotic and biotic drivers over time? A complex and spatially dependent example. Freshw. Biol. 2021, 66, 391–405. [Google Scholar] [CrossRef]
- Shvartsev, S.L.; Savichev, O.G.; Vertman, G.G.; Zarubina, R.F.; Nalyvayko, N.G.; Trifonova, N.G.; Turov Yu., G.; Friesen, L.F.; Yankovsky, V.V. Ecological and geochemical state of river waters in the Middle Ob. Water Resour. 1996, 23, 723–731. [Google Scholar]
- Savichev, O.G. Spatial and temporal changes in the chemical composition of the river waters of the Central Ob basin. Geogr. I Prir. Resur. 2000, 2, 60–66. (In Russian) [Google Scholar]
- Savichev, O.G. The hydrochemical drain of the middle Ob river basin. Bull. Tomsk Polytech. Univ. 2007, 310, 27–31. (In Russian) [Google Scholar]
- Sorokovikova, L.M.; Netsvetaeva, O.G.; Khodzher, T.V.; Kobeleva, N.A.; Chebykin, E.P.; Golobokova, L.P.; Chubarov, M.P. Chemical composition and water quality of the Ob River. In Ecological and Biogeochemical Studies in the Ob River Basin; Zuev, V.V., Kurovsky, A.V., Shvartsev, S.L., Eds.; RASKO Publishing House, LLC: Tomsk, Russia, 2002; pp. 21–26. (In Russian) [Google Scholar]
- Goncharov, A.V.; Isaev, V.A.; Lobchenko, E.E.; Nichiporova, I.P. Oxygen regime of rivers in Volga, Ob, and Lena basins. Water Resour. 2011, 38, 608–614. [Google Scholar] [CrossRef]
- Goncharov, A.V.; Zaslavskaya, M.B.; Isaev, V.A.; Lobchenko, E.E.; Nichiporova, I.P. Types of oxygen’s regime of Ob’ basin rivers. Geogr. Nat. Resour. 2013, 3, 69–76. (In Russian) [Google Scholar]
- Uvarova, V.I. Hydrochemical description of the low Ob waterways. Vestnik ekologii, lesovedeniya i landshaftovedeniya. Int. J. Environ. Stud. 2011, 11, 132–142. (In Russian) [Google Scholar]
- Lapin, S.A. Features of freshwater runoff formation in the estuarine systems of the Ob and Yenisei. Proc. Russ. Sci. Res. Inst. Fish. Oceanogr. 2017, 166, 139–150. [Google Scholar]
- Pipko, I.I.; Pugach, S.P.; Semiletov, I.P.; Savichev, O.G.; Shakhova, N.E.; Moiseeva, Y.A.; Repina, I.A.; Barskov, K.V.; Sergienko, V.I. Dynamics of dissolved inorganic carbon and CO2 fluxes between the water and the atmosphere in the main channel of the Ob river. Dokl. Chem. 2019, 484, 52–57. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Manasypov, R.M.; Loiko, S.; Shirokova, L.S.; Krickov, I.A.; Pokrovsky, B.G.; Kolesnichenko, L.G.; Kopysov, S.G.; Zemtzov, V.A.; Kulizhsky, S.P.; et al. Permafrost coverage, watershed area and season control of dissolved carbon and major elements in Western Siberian rivers. Biogeosciences 2015, 12, 6301–6320. [Google Scholar] [CrossRef]
- Novikova, O.D. Rotifera, Cladocera and Copepoda of the Middle Ob Basin. Ph.D. Thesis, Tomsk State University, Tomsk, Russia, 1973; 19p. (In Russian). [Google Scholar]
- Fish Ecology of the Ob-Irtysh Basin; Pavlov, D.S., Ed.; KMK Scientific Press: Moscow, Russia, 2006; 596p. (In Russian) [Google Scholar]
- Key to Freshwater Invertebrates of Russia and Adjacent Territories. Vol. 1. Lower Invertebrates; Tsalolikhin, S.Y., Ed.; Nauka: St Petersburg, Russia, 1994; 395p. [Google Scholar]
- Key to Freshwater Invertebrates of Russia and Adjacent Territories. Vol. 2. Crustaceans; Tsalolikhin, S.Y., Ed.; Nauka: St Petersburg, Russia, 1995; p. 629. [Google Scholar]
- Key to Freshwater Invertebrates of the European Part of the USSR. Plankton and Benthos; Kutikova, L.A., Starobogatova, Y.I., Eds.; Hydrometeoizdat: Leningrad, Russia, 1977; 511p. [Google Scholar]
- Kutikova, L.A. Kolovratki Fauny SSSR. [Rotifer Fauna of USSR]; Nauka: Leningrad, Russia, 1970; 742p. (In Russian) [Google Scholar]
- Manuylova, E.F. Cladocera of fauna the USSR; Nauka: Leningrad, Russia, 1964; 326p. (In Russian) [Google Scholar]
- Rylov, V.M. Cyclopoida of Fresh Water. Fauna of the USS; New Series, N 35; Publishing House of the USSR Academy of Sciences: Leningrad, Russia, 1948; 416p. (In Russian) [Google Scholar]
- Rylov, V.M. Freshwater Calanoida of the USSR. Freshwater Fauna. Issue 1; Institute of Fisheries and Commercial Research: Moscow, Russia, 1930; 318p. (In Russian) [Google Scholar]
- Smirnov, N.N. Fauna of the USSR. Crustaceans. Volume 1, Issue. 2; Chydoridae of the Fauna of the World; Nauka: Moscow, Russia, 1971; 268p. (In Russian) [Google Scholar]
- Smirnov, N.N. Fauna of the USSR. Crustaceans. Volume 1, Issue 3; Macrothricidae and Moinidae of the Fauna of the World; Nauka, Leningrad Branch: Moscow, Russia, 1976; 237p. (In Russian) [Google Scholar]
- Borutsky, E.V. Determinant of Free-Living Freshwater Copepod Crayfish of the USSR and Neighboring Countries by Fragments in Fish Intestines; Publishing House of the USSR Academy of Sciences: Moscow, Russia, 1960; 219p. (In Russian) [Google Scholar]
- Borutsky, E.V.; Stepanova, L.A.; Kos, M.S. Key to Freshwater Calanoida of the USSR; Nauka: St. Petersburg, Russia, 1991; 503p. (In Russian) [Google Scholar]
- Balushkina, E.V.; Vinberg, G.G. Relationship between Length and Weight of Planktonic Crustaceans. In Eksperimental’nye I Polevye Issledovaniya Biologicheskikh Osnov Produktivnosti Ozer (Experimental and Field Studies of Biological Bases of Productivity of Lakes); Izd. Zool. Inst. AN SSSR: Leningrad, Russia, 1979; pp. 169–172. (In Russian) [Google Scholar]
- Bakanov, A.I. Quantitative assessment of domination in ecological communities. In Quantitative Methods of Ecology and Hydrobiology: Book of Papers Dedicated to A.I. Bakanov’s Memory; Publishing House of Samar Scientific Center of RAS: Togliatti, Russia, 2005; p. 44. (In Russian) [Google Scholar]
- Slàdeček, V. System of water quality from the biological point of view. Arch. Hydrobiolgia. Ergeb. Der Limnol. 1973, 7, 1–218. [Google Scholar]
- Annual Data on the Regime and Resources of Land Surface Waters; Part 1; Rivers and canals. RSFSR. The Ob Basins (Without the Irtysh basin), Nadym, Pura, and Taza; West Siberian Regional Research Hydrometeorological Institute: Novosibirsk, Russia, 2009. (In Russian)
- Magritsky, D.V. Heat Runoff to the Russian Arctic Seas and Its Changes; Vestnik Moskovskogo Unviersiteta: Moscow, Russia, 2009; Volume 5, pp. 69–77. (In Russian) [Google Scholar]
- Bulavina, A.S. Reconstruction of the Ob river runoff according to meteorological observations data. Proc. Kola Sci. Cent. RAS 2020, 11, 17–27. [Google Scholar] [CrossRef]
- Basu, B.K.; Pick, F.R. Phytoplankton and Zooplankton Development in a Lowland Temperature River. J. Plankton Res. 1997, 19, 237–253. [Google Scholar] [CrossRef]
- Canale, R.P.; Vogel, A.H. Effects of Temperature on Phytoplankton Growth. J. Environ. Eng. Div. 1974, 100, 229–241. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Walsby, A.E. Water blooms. Biol. Rev. Camb. Philos. Soc. 1975, 50, 437–481. [Google Scholar] [CrossRef]
- Baird, M.E.; Emsley, S.M.; Mcglade, J.M. Modelling the interacting effects of nutrient uptake, light capture and temperature on phytoplankton growth. J. Plankton Res. 2001, 23, 829–840. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, S.; Niu, X. Effect of water temperature on the dynamic behavior of phytoplankton–zooplankton model. Appl. Math. Comput. 2020, 378, 125211. [Google Scholar] [CrossRef]
- Işkın, U.; Filiz, N.; Cao, Y.; Neif, É.M.; Öğlü, B.; Lauridsen, T.L.; Davidson, T.A.; Søndergaard, M.; Tavşanoğlu, Ü.N.; Beklioğlu, M.; et al. Impact of Nutrients, Temperatures, and a Heat Wave on Zooplankton Community Structure: An Experimental Approach. Water 2020, 12, 3416. [Google Scholar] [CrossRef]
- Alimov, A.F. The fundamentals of the theory of the functioning of aquatic ecosystems. Hydrobiol. J. 1990, 26, 3–12. (In Russian) [Google Scholar]
- Ivanova, M.B. Influence of active reaction and general water salinity on the formation of zooplankton community in lakes when the values of these factors approach extreme ones. Proc. Zool. Inst. RAS 1997, 272, 71–86. (In Russian) [Google Scholar]
- Ivanova, M.B. Dependence of the number of species in zooplankton of lakes on the total mineralization of water and the pH value. Inland Water Biol. 2005, 1, 64–68. (In Russian) [Google Scholar]
- Ivanova, M.B.; Kazantseva, T.I. Influence of active reaction and total salinity of water on the species diversity of pelagic zooplankton in lakes (statistical analysis). Russ. J. Ecol. 2006, 4, 294–300. (In Russian) [Google Scholar]
- Brett, M.T. Zooplankton communities and acidification process (a review). Water Air Soil Pollut. 1989, 44, 387–414. [Google Scholar] [CrossRef]
- Havens, K.E. Summer zooplankton dynamics in the limnetic and littoral zones of a humic acid lake. Hydrobiologia 1991, 215, 21–29. [Google Scholar] [CrossRef]
- Havens, K.E.; Carlson, R.E. Functional complementarity in plankton communities along a gradient of acid stress. Environ. Pollut. 1998, 101, 427–436. [Google Scholar] [CrossRef]
- Fischer, J.M.; Frost, T.M.; Ives, A.R. Compensatory Dynamics in Zooplankton Community Responses to Acidification: Measurement and Mechanisms. Ecol. Appl. 2001, 11, 1060–1072. [Google Scholar] [CrossRef]
- Dyga, A.K.; Zolotareva, V.I. Influence of the active reaction of water on individual representatives of zooplankton and macrozoobenthos in connection with industrial pollution of water bodies. Water Resour. 1983, 5, 104–107. (In Russian) [Google Scholar]
- Havens, K.E.; Hanazato, T. Zooplankton community responses to chemical stressors: A comparison of results from acidification and pesticide contamination research. Environ. Pollut. 1993, 82, 277–288. [Google Scholar] [CrossRef]
- Hansen, B.W.; Andersen, C.M.B.; Hansen, P.J.; Nielsen, T.G.; Vismann, B.; Tiselius, P. In situ and experimental evidence for effects of elevated pH on protistan and metazoan grazers. J. Plankton Res. 2019, 41, 257–271. [Google Scholar] [CrossRef]
- Imant, E.N.; Novoselov, A.P. Dynamics of Zooplankton Composition in the Lower Northern Dvina River and Some Factors Determining Zooplankton Abundance. Russ. J. Ecol. 2021, 52, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Dvurechenskaya, S.Y.; Yermolaeva, N.I. Interrelations between Chemical Composition of Water and Characteristics of Zooplankton in the Novosibirsk Water Storage Basin. Contemp. Probl. Ecol. 2014, 7, 464–472. [Google Scholar] [CrossRef]
- Khamitova, M.F. Study of Changes in Hydrobiological Characteristics under Conditions of Local Pollution in the Region of the Middle Volga. Ph.D. Thesis, Kazan University, Kazan, Russia, 2017; 24p. (In Russian). [Google Scholar]
- Aleksevnina, M.S.; Pozdeev, I.V. Sanitary Hydrobiology with the Basics of Aquatic Toxicology; Perm National Research University: Perm, Russia, 2016; 205p. (In Russian) [Google Scholar]
- Frutos, S.M.; Poi de Neiff, A.S.G.; Neiff, J.J. Zooplankton of the Paraguay River: A comparison between sections and hydrological phases. Ann. De Limnol. Int. J. Limnol. 2006, 42, 277–288. [Google Scholar] [CrossRef]
- Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Sedell, J.R.; Cushing, C.E. The river continuum concept. Can. J. Fish. Aquat. Sci. 1980, 37, 130–137. [Google Scholar] [CrossRef]
- Górski, K.; Collier, K.J.; Duggan, I.C.; Taylor, C.M.; Hamilton, D.P. Connectivity and complexity of floodplain habitats govern zooplankton dynamics in a large temperate river system. Freshw. Biol. 2013, 58, 1458–1470. [Google Scholar] [CrossRef]
- De Ruyter van Steveninck, E.D.; Admiraal, W.; Breebaart, L.; Tubbing, G.M.J.; van Zanten, B. Plankton in the River Rhine: Structural and functional changes observed during downstream transport. J. Plankton Res. 1992, 14, 1351–1368. [Google Scholar] [CrossRef]
- Scherwass, A.; Bergfeld, T.; Schoel, A.; Weitere, M.; Arndt, H. Changes in the plankton community along the length of the River Rhine: Lagrangian sampling during spring. J. Plankton Res. 2010, 32, 32–491. [Google Scholar] [CrossRef]
- Otyukova, N.G.; Tselmovich, O.L.; Krylov, A.V. The Effect of the Precipation Amount and Flow Regulation on Chemical Composition of Water and Zooplankton of a Small River. Inland Water Biol. 2007, 3, 48–55. (In Russian) [Google Scholar]
- Berggren, M.; Bengtson, P.; Soares, A.R.A.; Karlsson, J. Terrestrial support of zooplankton biomass in northern rivers. Limnol Oceanogr. 2018, 63, 2479–2492. [Google Scholar] [CrossRef]
- Groeneveld, M.; Catalán, N.; Attermeyer, K.; Hawkes, J.; Einarsdóttir, K.; Kothawala, D.; Bergquist, J.; Tranvik, L. Selective adsorption of terrestrial dissolved organic matter to inorganic surfaces along a boreal inland water continuum. J. Geophys. Res. Biogeosciences 2020, 125, e2019JG005236. [Google Scholar] [CrossRef]
- Duggan, I.C. The ecology of periphytic rotifers. Hydrobiologia 2001, 446–447, 139–148. [Google Scholar] [CrossRef]
- Lucena-Moya, P.; Duggan, I.C. Macrophyte architecture affects the abundance and diversity of littoral microfauna. Aquat. Ecol. 2011, 45, 279–287. [Google Scholar] [CrossRef]
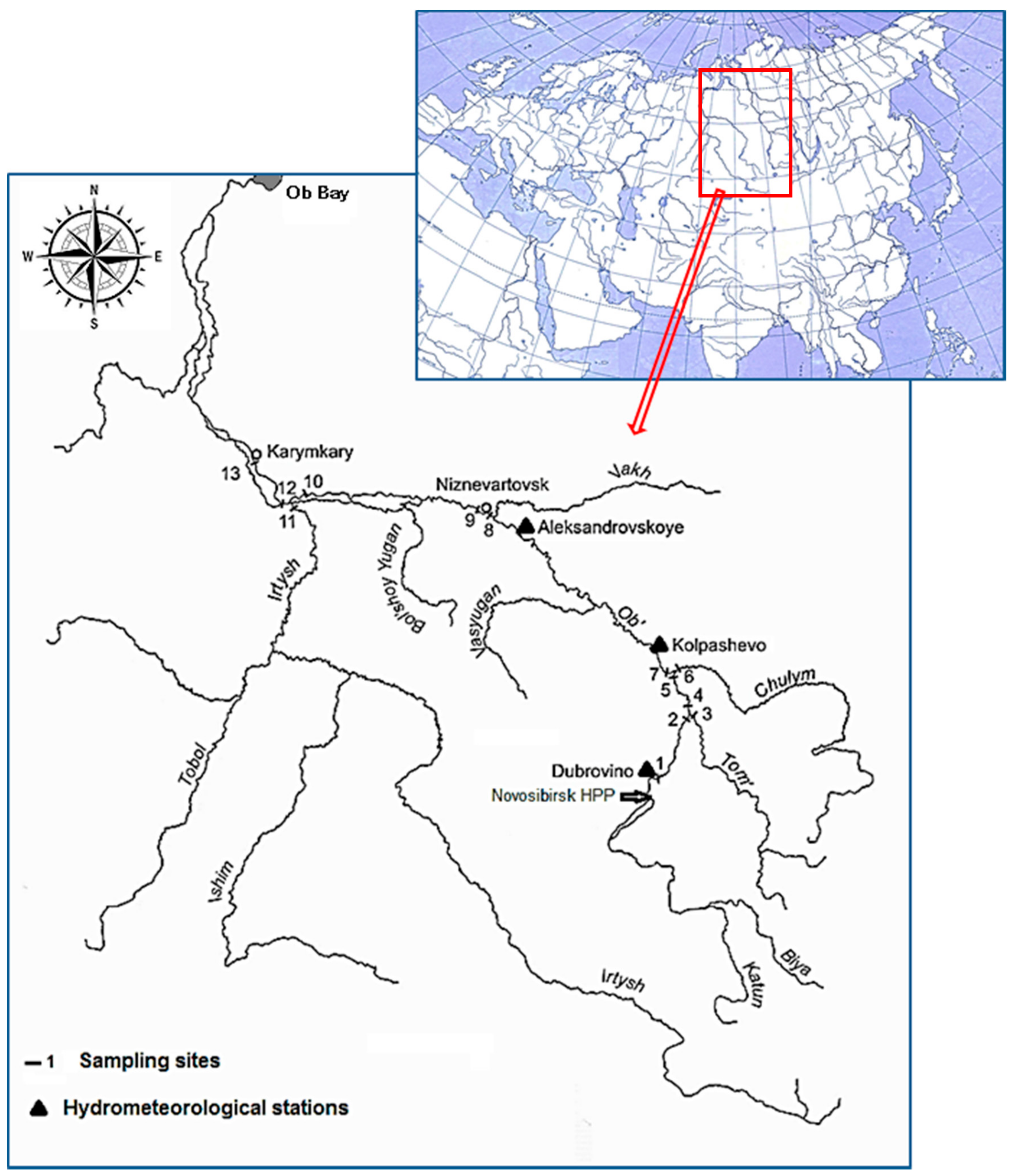
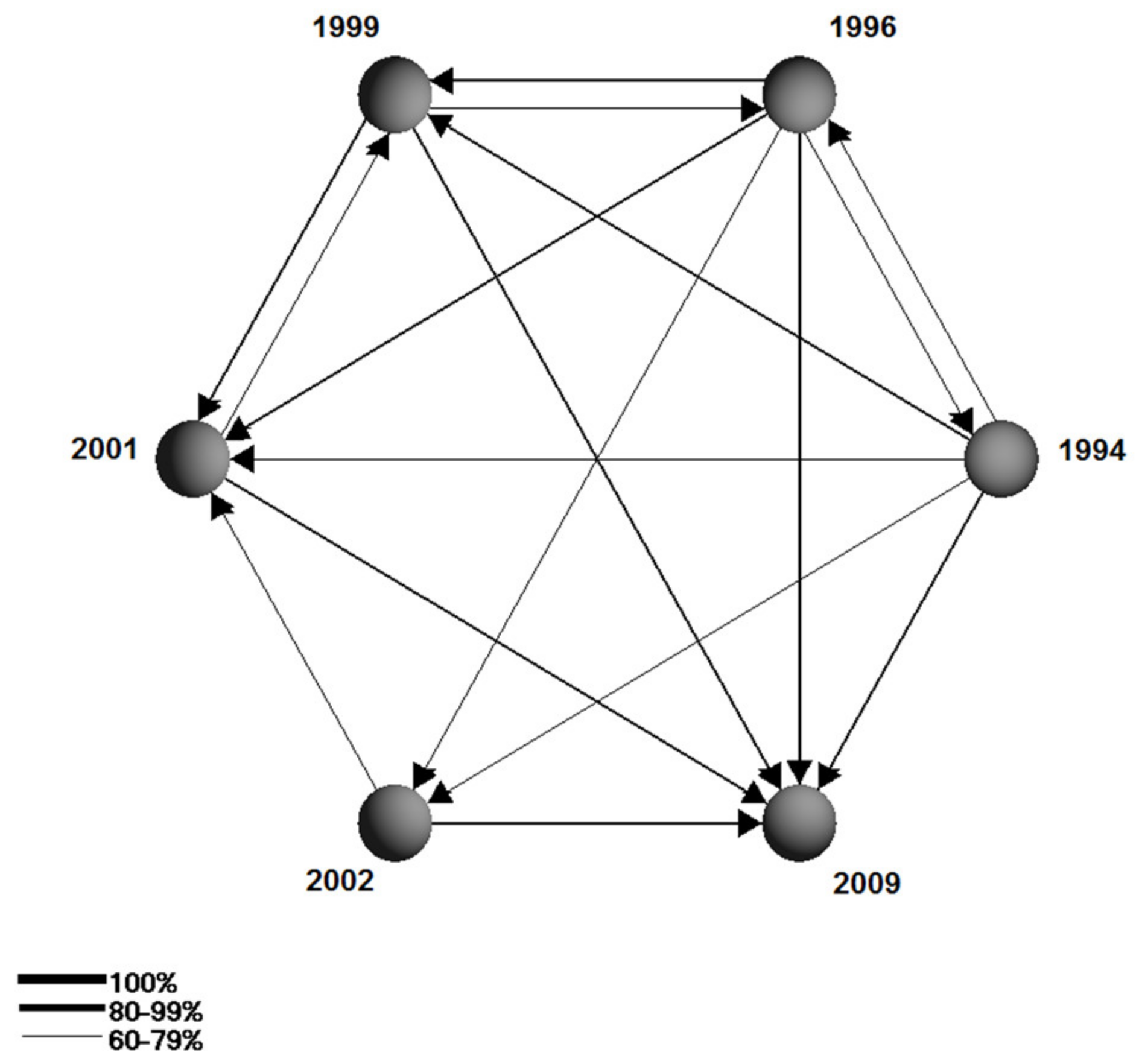
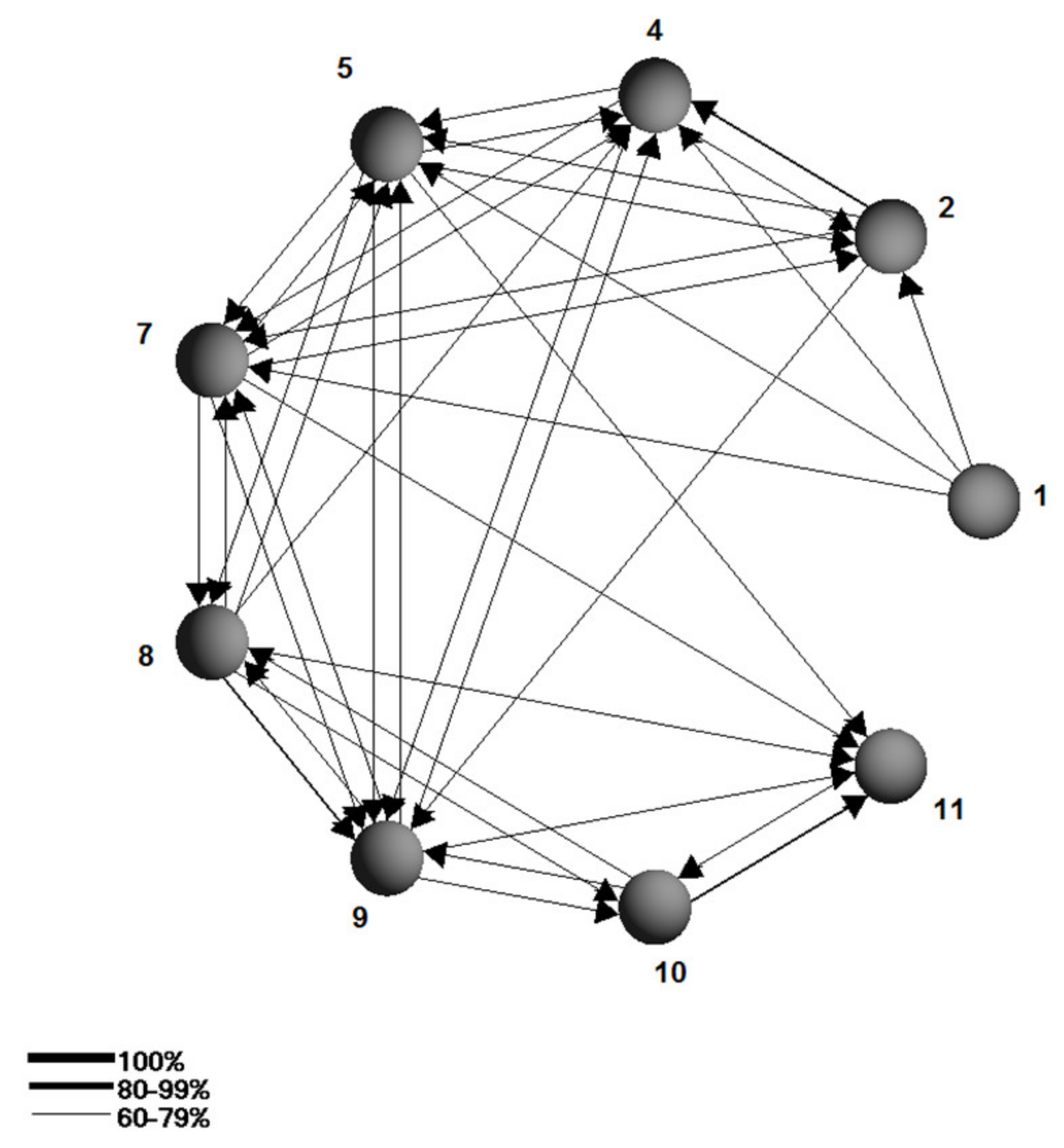
| No | Sampling Site Names | Sampling Site Coordinates | Km From the OB Source |
|---|---|---|---|
| Hydrochemical and hydrobiological samples | |||
| 0 | Novosibirsk HPP | 54°50′51″ N 82°59′30″ E | 678 |
| 1 | Dubrovino | 55°28′42″ N 83°16′36″ E | 785 |
| 2 | 7 km above Tom River mouth | 56°44′56″ N 84°24′47″ E | 963 |
| 3 | Tom River mouth | 56°53′21″ N 84°27′42″ E | 987 |
| 4 | 10 km below Tom River mouth | 56°58′14″ N 84°24′08″ E | 997 |
| 5 | 1 km above R.Chulym mouth | 57°42′58″ N 83°51′15″ E | 1132 |
| 6 | R.Chulym mouth | 57°43′30″ N 83°49′35″ E | 1133 |
| 7 | 3 km below R.Chulym mouth | 57°42′40″ N 83°46′28″ E | 1136 |
| 8 | 1 km above Nizhnevartovsk | 60°50′37″ N 76°38′28″ E | 1940 |
| 9 | 1 km below Nizhnevartovsk | 60°51′33″ N 76°23′55″ E | 1956 |
| 10 | 30 km above R.Irtysh mouth | 60°10′46″ N 69°11′17″ E | 2430 |
| 11 | R.Irtysh mouth | 61 04′48″ N 68 49′50″ E | 2500 |
| Hydrometeorological stations | |||
| Dubrovino | 55°28′42″ N 83°16′36″ E | 785 | |
| Kolpashevo | 58°18′04″ N 82°54′19″ E | 1250 | |
| Aleksandovskoye | 60°26′36″ N 77°52′57″ E | 1840 | |
| Variables | 1994 | 1996 | 1999 | 2001 | 2002 | 2009 |
|---|---|---|---|---|---|---|
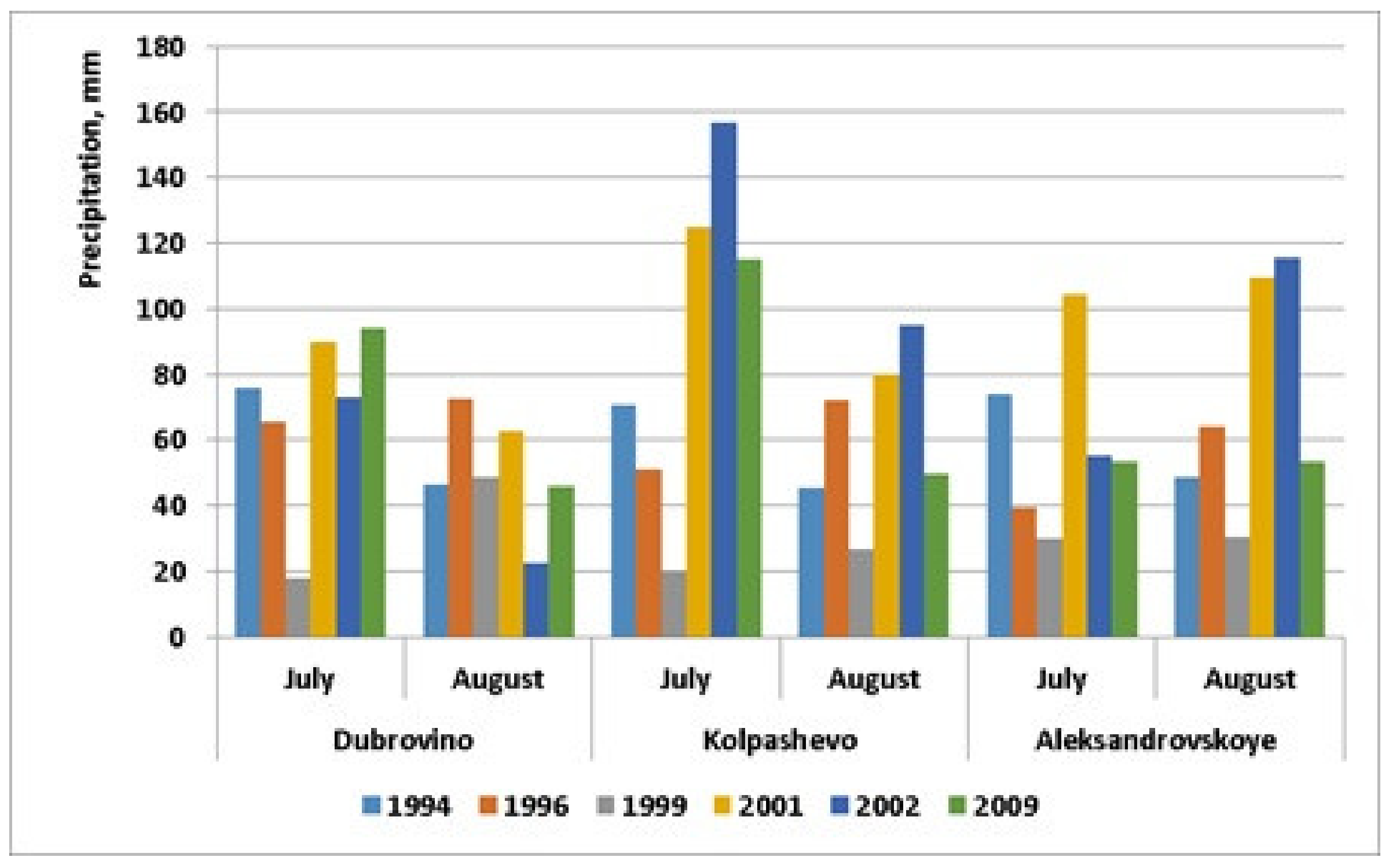
| Precipitation, mm (July), | 99.8 ± 31.1 | 48.2 ± 5.8 | 22.6 ± 2.6 | 101.1 ± 8.9 | 105.7 ± 24.7 | 79.9 ± 14.8 |
| Precipitation, mm (August) | 79.0 ± 22.9 | 75.6 ± 5.9 | 33.6 ± 5.1 | 84.5 ± 9.7 | 71.3 ± 20.9 | 50.4 ± 1.7 |
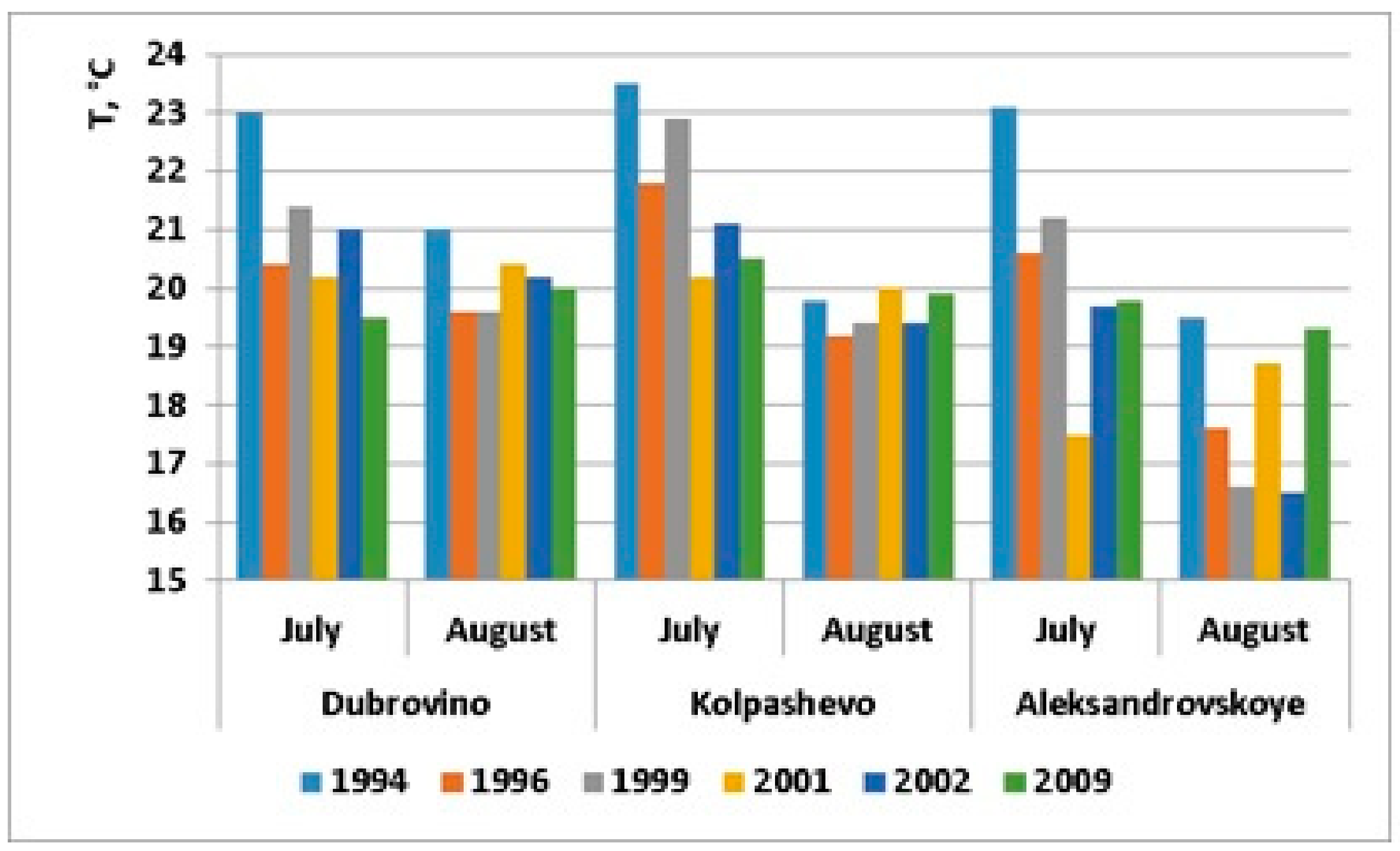
| Temperature, °C (July) | 23.2 ± 0.2 | 21.1 ± 0.4 | 21.8 ± 0.5 | 19.3 ± 0.9 | 20.6 ± 0.5 | 19.9 ± 0.3 |
| Temperature, °C (August) | 20.1 ± 0.5 | 18.8 ± 0.4 | 18.5 ± 0.9 | 19.7 ± 0.5 | 18.7 ± 1.1 | 19.7 ± 0.2 |
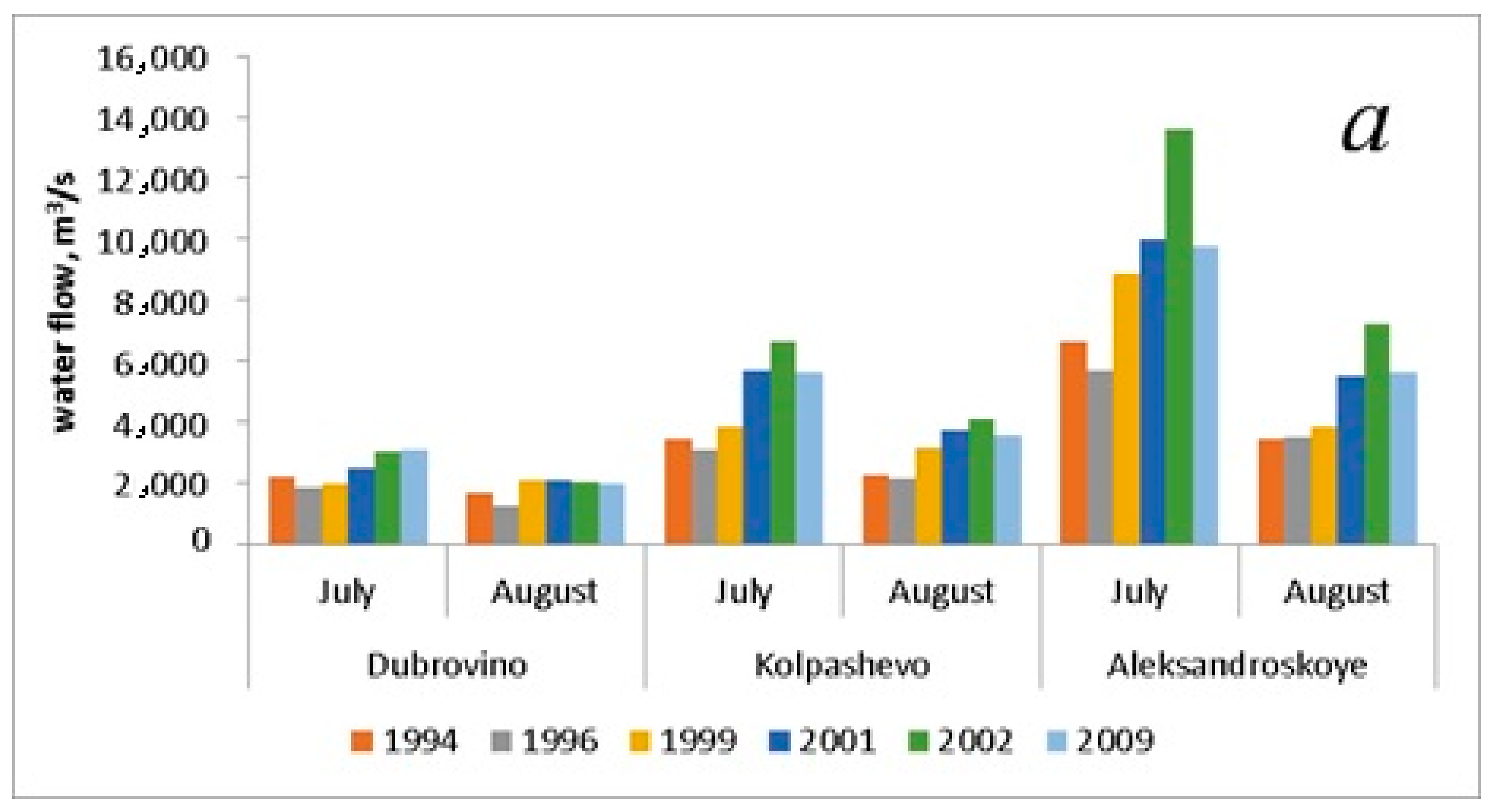
| Discharge, m3/s (July) | 4083.3 ± 1310.7 | 3530.0 ± 1137.7 | 4883.3 ± 2069.9 | 6040.0 ± 2164.3 | 7740.0 ± 3108.8 | 6140.0 ± 1947.9 |
| Discharge, m3/s (August) | 2446.7 ± 522.2 | 2283.3 ± 646.7 | 3023.3 ± 521.2 | 3756.7 ± 984.4 | 4440.0 ± 1510.7 | 3713.3 ± 1065.9 |
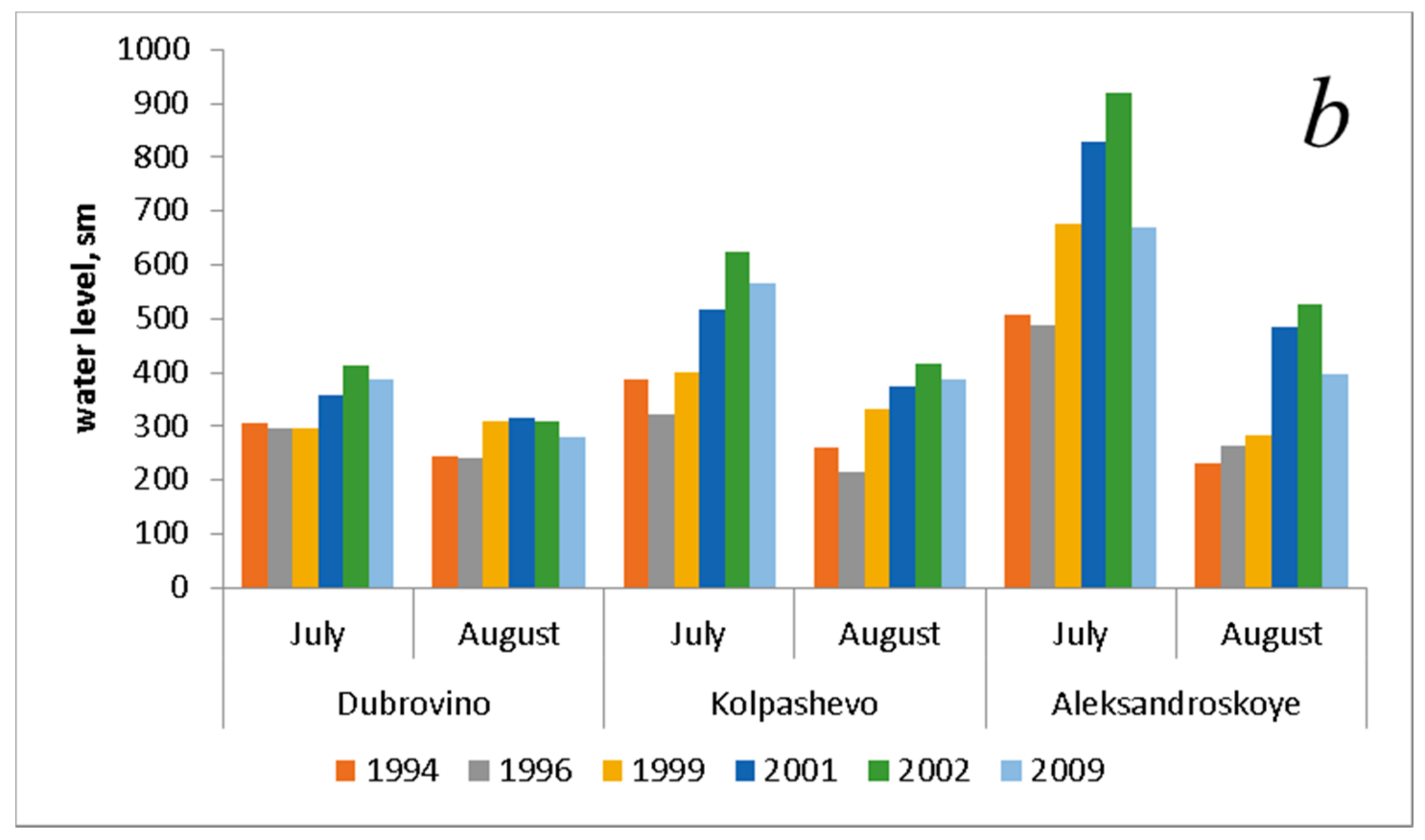
| Water level, sm * (July) | 401.0 ± 58.6 | 370.0 ± 59.5 | 458.0 ± 113.9 | 567.7 ± 138.0 | 653.0 ± 146.4 | 541.3 ± 82.9 |
| Water level, sm * (August) | 244.0 ± 8.4 | 240.7 ± 14.8 | 308.3 ± 14.2 | 392.3 ± 50.0 | 418.7 ± 62.6 | 355.0 ± 38.1 |
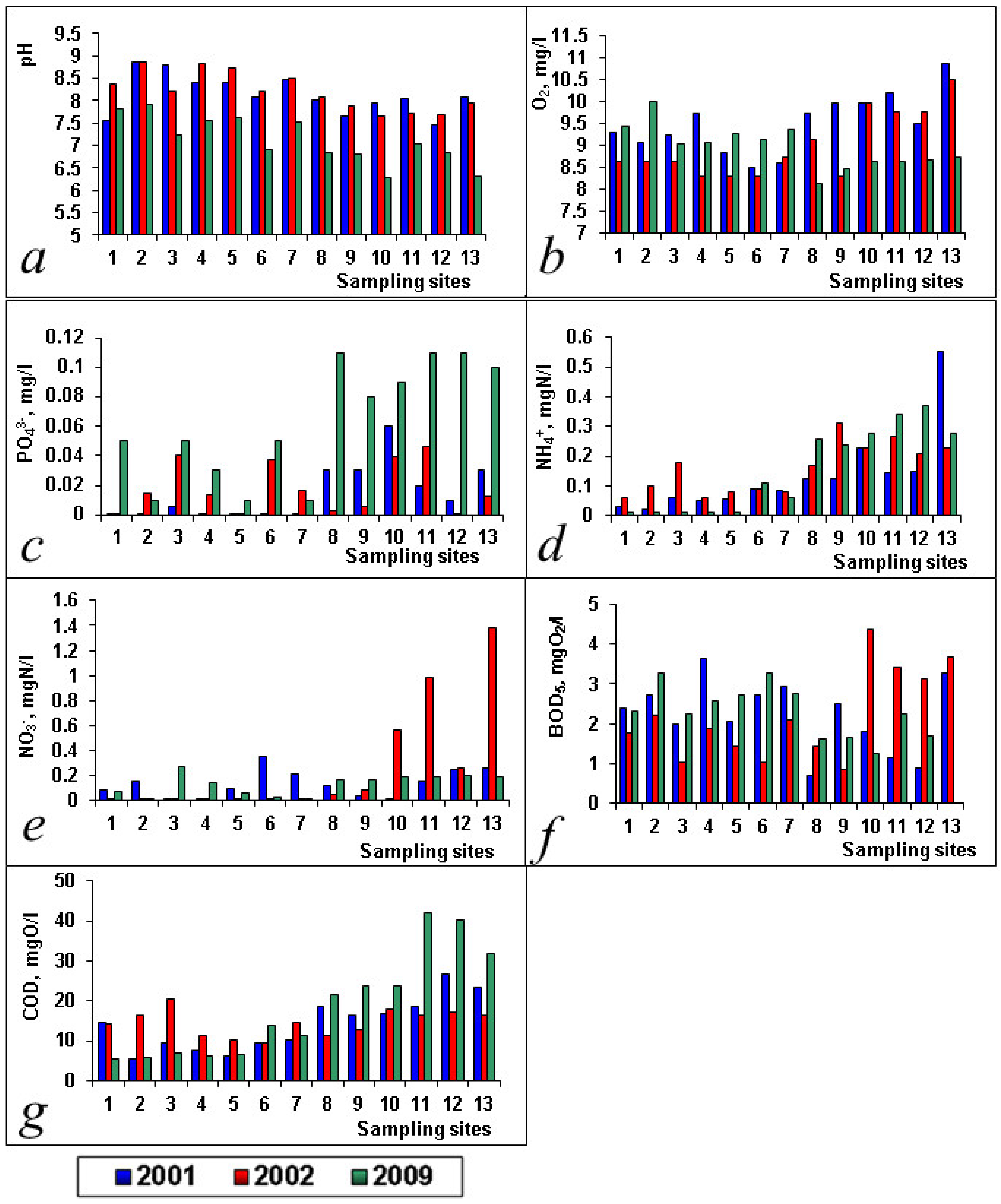
| PO43−, mg/L | 0.153 ± 0.048 | 0.036 ± 0.022 | 0.015 ± 0.005 | 0.018 ± 0.004 | 0.062 ± 0.011 | |
| NH4+, mgN/L | 0.133 ± 0.038 | 0.159 ± 0.024 | 0.153 ± 0.040 | |||
| NO2−, mgN/L | 0.038 ± 0.014 | 0.021 ± 0.001 | 0.014 ± 0.012 | 0.020 ± 0.010 | ||
| NO3−, mgN/L | 0.133 ± 0.031 | 0.257 ± 0.125 | 0.132 ± 0.023 | |||
| O2, mg/L | 9.50 ± 0.19 | 8.99 ± 0.21 | 8.97 ± 0.14 | |||
| pH | 8.13 ± 0.12 | 8.20 ± 0.12 | 7.13 ± 0.15 | |||
| BOD5, mgO2/L | 2.21 ± 0.25 | 2.18 ± 0.31 | 2.30 ± 0.19 | |||
| COD, mgO/L | 14.13 ± 1.85 | 14.61 ± 0.92 | 18.41 ± 3.66 |
| Sites | 1994 | 1996 | 1999 | 2001 | 2002 | 2009 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | B | n | N | B | n | N | B | n | N | B | n | N | B | n | N | B | n | |
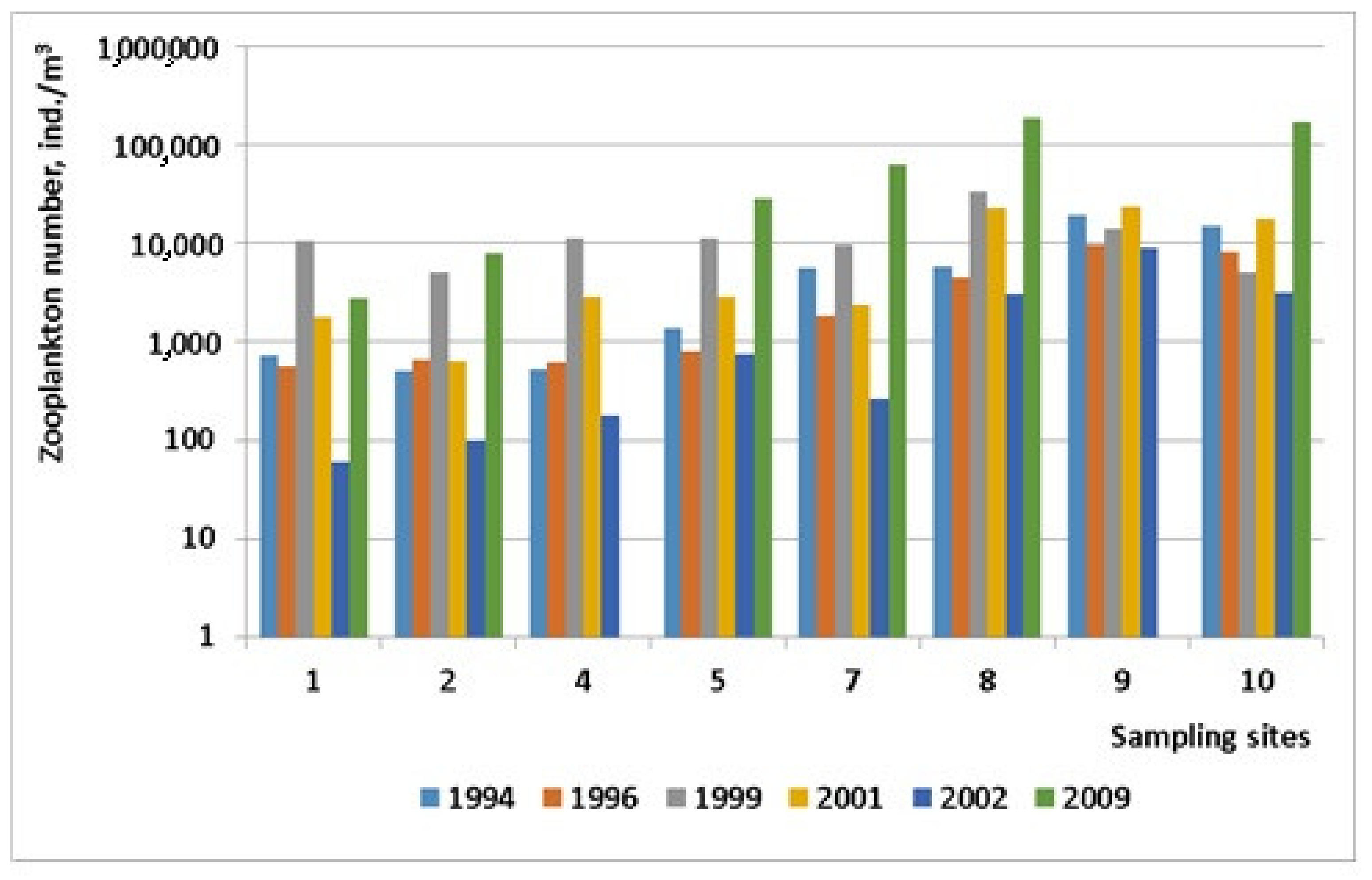
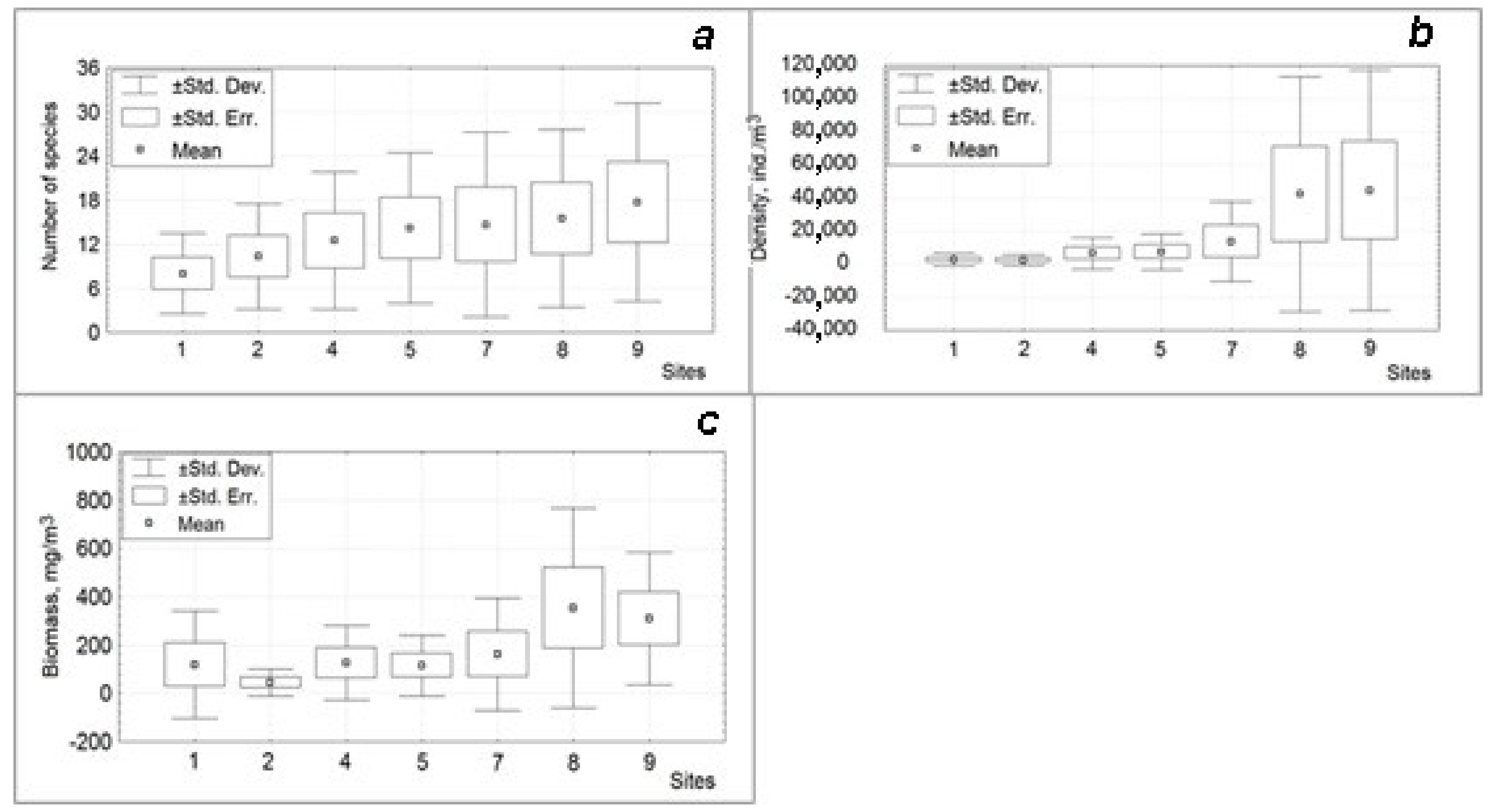
| 1 | 734 | 22.57 | 8 | 570 | 9.6 | 5 | 10.550 | 573.31 | 9 | 1780 | 64.40 | 6 | 60 | 1.78 | 2 | 2810 | 41.10 | 18 |
| 2 | 510 | 9.11 | 10 | 660 | 15.37 | 8 | 5080 | 139.11 | 10 | 640 | 12.30 | 7 | 100 | 1.88 | 3 | 8080 | 90.90 | 24 |
| 3 | 2887 | 29.71 | 8 | 340 | 8.61 | 7 | 2800 | 47.31 | 11 | 1860 | 52.30 | 9 | 400 | 6.24 | 5 | 3460 | 13.80 | 17 |
| 4 | 533 | 8.95 | 8 | 610 | 14.57 | 11 | 11.200 | 314.62 | 10 | 2880 | 83.30 | 10 | 180 | 5.33 | 5 | 24.220 | 329.90 | 31 |
| 5 | 1355 | 26.05 | 9 | 800 | 24.10 | 10 | 11.200 | 238.63 | 12 | 2880 | 83.30 | 10 | 740 | 13.87 | 9 | 28.320 | 302.60 | 35 |
| 6 | 667 | 5.71 | 6 | 685 | 6.30 | 9 | 2700 | 61.71 | 11 | 720 | 21.10 | 9 | 600 | 27.24 | 7 | 32.000 | 81.50 | 23 |
| 7 | 5688 | 87.20 | 9 | 1860 | 38.50 | 12 | 9580 | 184.87 | 11 | 2400 | 40.10 | 10 | 260 | 1.01 | 6 | 62.810 | 621.00 | 40 |
| 8 | 5778 | 76.68 | 10 | 4430 | 81.26 | 12 | 33.000 | 714.69 | 12 | 23.140 | 172.20 | 10 | 3100 | 46.33 | 9 | 185.510 | 1021.90 | 40 |
| 9 | 19.553 | 314.25 | 10 | 9700 | 196.22 | 14 | 14.200 | 296.12 | 14 | 23.500 | 130.60 | 11 | 8940 | 84.04 | 12 | 192.200 | 835.90 | 45 |
| 10 | 14.955 | 348.98 | 9 | 8100 | 78.90 | 10 | 5150 | 94.97 | 11 | 17.620 | 88.70 | 8 | 3200 | 48.59 | 11 | 169.860 | 384.80 | 35 |
| 11 | 1689 | 62.88 | 9 | 1580 | 31.50 | 11 | 3200 | 46.51 | 7 | 21.660 | 90.70 | 10 | 2320 | 45.50 | 6 | 61.210 | 72.30 | 45 |
| Means ± St.Dev. | Copepoda | Cladocera | Rotifera | Total Zooplankton | |
|---|---|---|---|---|---|
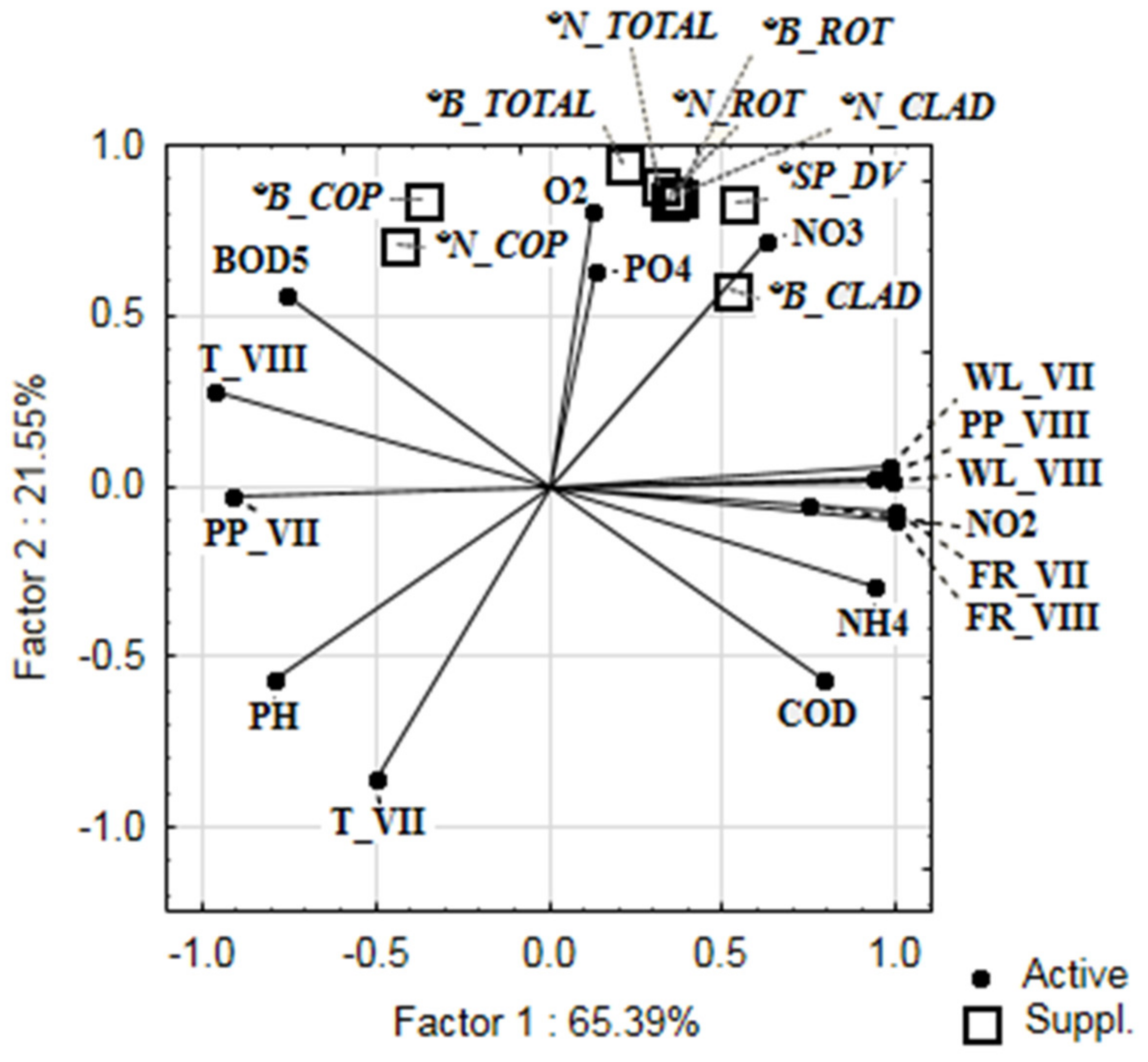
| Precipitation (July) | 110.28 ± 42.55 | 0.146 | −0.164 | −0.187 | −0.167 |
| Precipitation (August) | 99.93 ± 15.86 | −0.584 | 0.485 | 0.479 | 0.433 |
| Temperature (July) | 19.63 ± 1.536 | −0.333 | −0.940 | −0.952 | −0.954 |
| Temperature (August) | 18.65 ± 1.53 | 0.586 | −0.069 | −0.075 | −0.032 |
| Flow rate (July) | 8960.0 ± 3599.82 | −0.478 | 0.273 | 0.280 | 0.240 |
| Flow rate (August) | 5130.0 ± 1574.27 | −0.482 | 0.232 | 0.240 | 0.201 |
| Water level (July) | 722.5 ± 183.74 | −0.499 | 0.466 | 0.467 | 0.424 |
| Water level (August) | 451.75 ± 68.15 | −0.523 | 0.420 | 0.421 | 0.378 |
| PO43− | 0.012 ± 0.014 | −0.041 | 0.893 | 0.874 | 0.861 |
| NH4+ | 0.15 ± 0.14 | −0.497 | −0.016 | −0.005 | −0.042 |
| NO2− | 0.02 ± 0.02 | −0.003 | −0.066 | −0.036 | −0.044 |
| NO3− | 0.04 ± 0.06 | 0.072 | 0.944 | 0.948 | 0.935 |
| O2 | 9.23 ± 0.68 | 0.835 | 0.479 | 0.514 | 0.551 |
| pH | 8.30 ± 0.33 | −0.183 | −0.648 | −0.672 | −0.665 |
| BOD5 | 2.20 ± 1.17 | 0.913 | 0.078 | 0.093 | 0.148 |
| COD | 11.04 ± 2.88 | −0.874 | −0.119 | −0.129 | −0.183 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yermolaeva, N.; Dvurechenskaya, S.; Kirillov, V.; Puzanov, A. Dependence of Long-Term Dynamics of Zooplankton in the Ob River on Interannual Changes in Hydrological and Hydrochemical Parameters. Water 2021, 13, 1910. https://doi.org/10.3390/w13141910
Yermolaeva N, Dvurechenskaya S, Kirillov V, Puzanov A. Dependence of Long-Term Dynamics of Zooplankton in the Ob River on Interannual Changes in Hydrological and Hydrochemical Parameters. Water. 2021; 13(14):1910. https://doi.org/10.3390/w13141910
Chicago/Turabian StyleYermolaeva, Nadezhda, Serafima Dvurechenskaya, Vladimir Kirillov, and Aleksandr Puzanov. 2021. "Dependence of Long-Term Dynamics of Zooplankton in the Ob River on Interannual Changes in Hydrological and Hydrochemical Parameters" Water 13, no. 14: 1910. https://doi.org/10.3390/w13141910
APA StyleYermolaeva, N., Dvurechenskaya, S., Kirillov, V., & Puzanov, A. (2021). Dependence of Long-Term Dynamics of Zooplankton in the Ob River on Interannual Changes in Hydrological and Hydrochemical Parameters. Water, 13(14), 1910. https://doi.org/10.3390/w13141910






