Isotope Signs (234U/238U, 2H, 18O) of Groundwater: An Investigation of the Existence of Paleo-Permafrost in European Russia (Pre-Volga Region)
Abstract
:1. Introduction
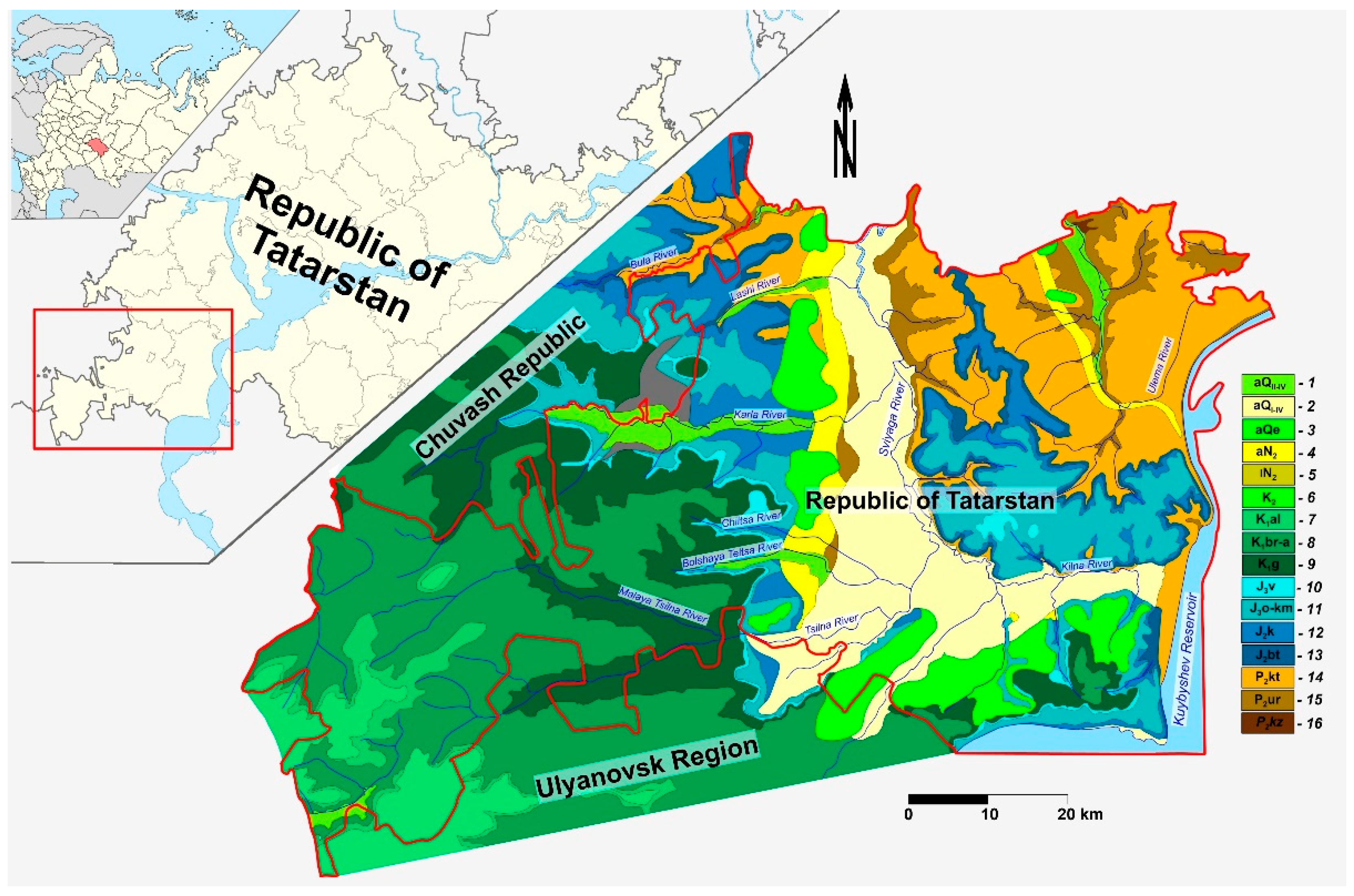
2. Characterization of the Studied Area
3. Materials and Methods
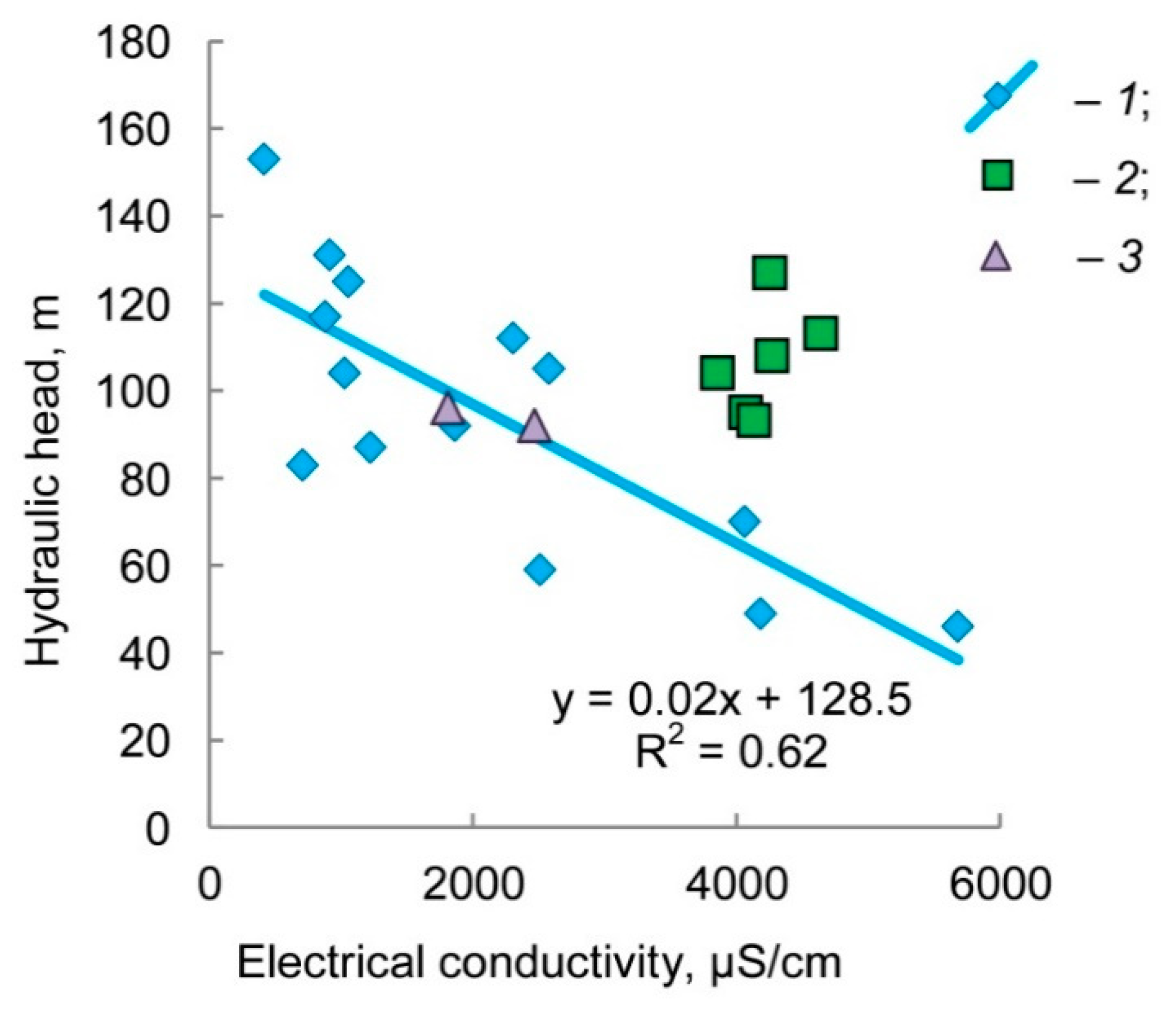
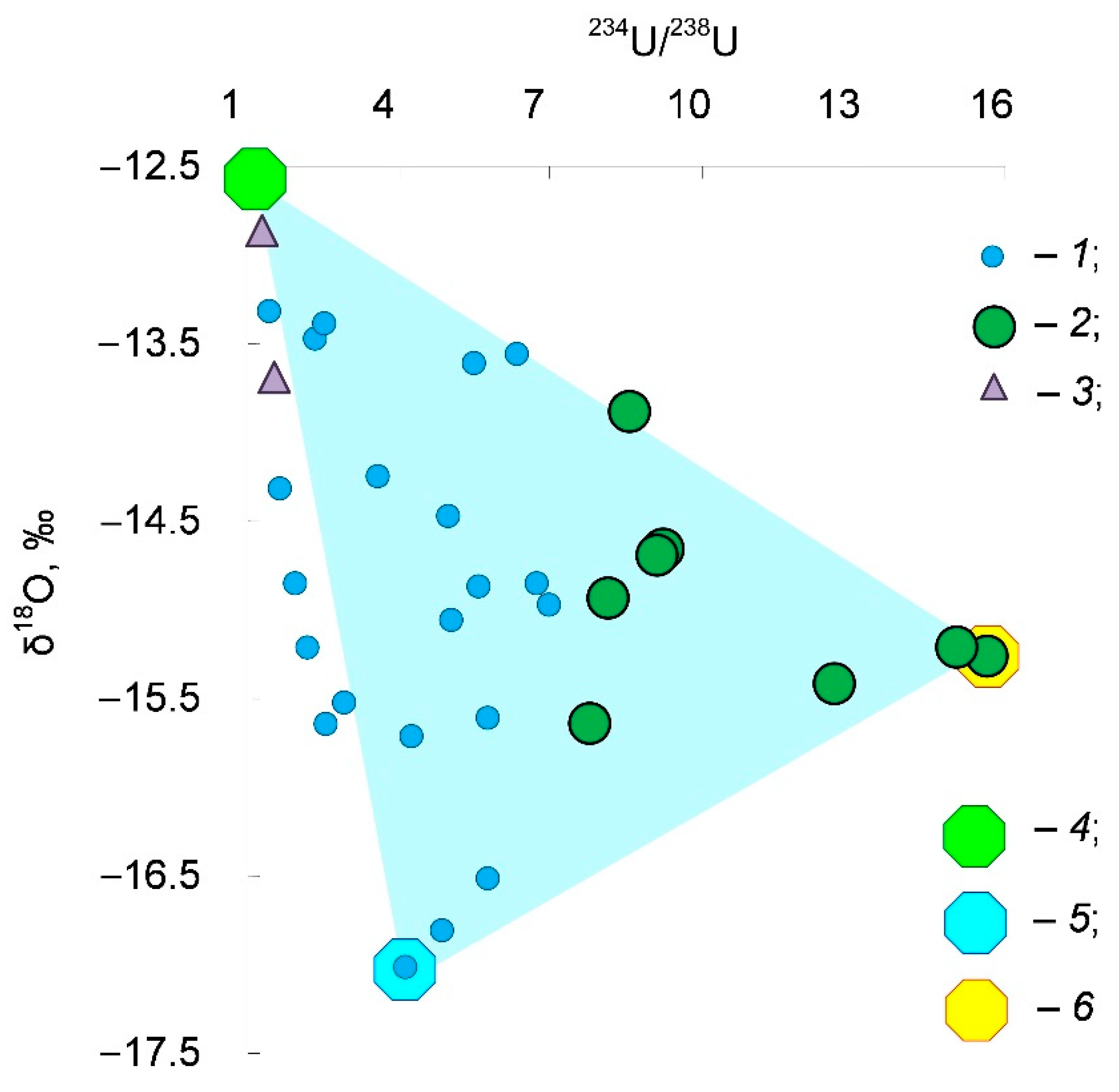
4. Results and Discussion
| S = 0.4–1.0 | HCO3(70–96) NO3(1–18) Cl(1–6) SO4(1–6) | pH = 7.5–7.8. |
| Ca(22–69) Mg(7–49) Na(2–37) |
| S = 0.9–1.8 | SO4(36–63) HCO3(27–52) Cl(2–17) | pH = 7.3–8.1. |
| Na(14–86) Mg(9–27) Ca(7–17) |
| S = 2.0–6.9 | SO4(46–85) Cl(7–51) HCO3(8–39) | pH = 7.1–7.8. |
| Na(37–80) Ca(11–35) Mg(9–28) |
| S = 1.8–4 | Cl(34–71) SO4(18–32) HCO3(2–7) | pH = 7.5–8.5. |
| Ca(45–74) Mg(6–21) Na(12–34) |
- Modern and/or young (Holocene age) recharge water with salinity S < 0.5 g/L. It has the least depletion in the isotopic composition of hydrogen and oxygen δ18O → −12.9 ‰ and δ2H → −90 ‰, which is close to the modern precipitation of the region, and practically the equilibrium isotope composition of uranium 234U/238U → 1.
- Water of the late and/or postglacial genesis. It has a slight increase in salinity S ≈ 0.5–1.5 g/L, the isotopically lightest composition δ18O → −17.0 ‰ and δ2H → −119 ‰ (maybe water of a proglacial lake?), and a small excess of uranium-234 234U/238U ≈ 4.
- The glacial meltwater formed due to the permafrost thawing. It is the brackish water S → 3 g/L (for these 33 samples under consideration). This water has the intermediate composition of the stable isotopes δ18O ≈ −15.0 ‰ and δ2H ≈ −110 ‰ (ice in permafrost, most likely, formed from mixture water of the Valdai cryochron and the previous interstadial), and maximum disequilibrium in uranium 234U/238U → 15.7.
- reduced mineralization, as salts must be lost in the freezing process;
- an isotope composition of hydrogen and oxygen with less depletion than the recharge water (points must have a shift to the right from the meteoric line on the δ2H vs. δ18O diagram).
5. Conclusions
- Modern and/or young recharge water of the infiltration origin has the lowest salinity S < 500 mg/L, the heaviest stable isotope composition δ18O → −12.9 ‰ and δ2H → −90 ‰ close to modern precipitation, and practically equilibrium uranium 234U/238U → 1;
- Late and/or postglacial water, which has a slight increase in mineralization S ≈ 0.5–1.5 g/L, the lightest isotopic composition δ18O → −17.0 ‰ and δ2H → −119 ‰, and a small excess of uranium-234 234U/238U ≈ 4;
- Glacial meltwater (groundwater formed from thawing permafrost) has increased salinity S → 3 g/L, an intermediate composition of the stable isotopes δ18O ≈ −15.0 ‰ and δ2H ≈ −110 ‰, and maximum excess of uranium-234 234U/238U → 15.7.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Sample ID | Sites of Groundwater Collection | Coordinates | Geological Age | |
|---|---|---|---|---|
| Latitude | Longitude | |||
| PRV-1 | Yablonovka Village | 54.67640 | 47.33320 | K2 |
| PRV-2 | Yablonovka Village | 54.67738 | 47.33272 | K2 |
| PRV-3 | Nemchinovka Village | 54.87359 | 48.38206 | P2t |
| PRV-4 | Chuvash Kischak Village | 54.77169 | 48.16469 | P2kz |
| PRV-5 | Yaskul Village | 54.76633 | 48.30734 | P2kz |
| PRV-6 | Old Tinchali Village | 54.84544 | 48.15256 | P2kz |
| PRV-7 | Mullanur Vakhitov Village | 54.93067 | 48.12241 | P2t |
| PRV-8 | Volny Stan Village | 54.87190 | 47.80623 | P2kz |
| PRV-9 | Kamennyi Brod Village | 55.14796 | 48.17735 | P2t |
| PRV-10 | Tatar Belovozhka Village | 55.03437 | 48.10233 | P2t |
| PRV-11 | Malye Shikhardans Village | 54.95153 | 47.85416 | P2t |
| PRV-12 | Embulatovo Village | 54.93251 | 47.95070 | P2t |
| PRV-13 | Staroe Ilmovo Village | 54.68475 | 47.57681 | P2kz |
| PRV-14 | Novoe Ilmovo Village | 54.67081 | 47.61837 | P2kz |
| PRV-15 | Novie Chukaly Village | 54.63715 | 47.63321 | P2kz |
| PRV-16 | Starie Chukaly Village | 54.65622 | 47.55037 | P2kz |
| PRV-17 | Bolshaya Aksa Village | 54.63486 | 47.50940 | P2kz |
| PRV-18 | Gorodishe Village | 54.57689 | 47.51139 | P2kz |
| PRV-19 | Novye Ishli Village | 54.79223 | 47.56261 | P2kz |
| PRV-20 | Staryie Ishli Village | 54.80350 | 47.49523 | P2kz |
| PRV-21 | Novye Kakerly Village | 54.82065 | 47.60283 | P2kz |
| PRV-22 | Malyi Ubei Village | 54.78862 | 47.79419 | J2k-km |
| PRV-23 | Alyoshkin Saplyk Village | 54.74966 | 47.63657 | P2kz |
| PRV-24 | Shlanga Village | 54.81568 | 47.40597 | P2kz |
| PRV-25 | Kyzyl-Chishma Village | 54.99799 | 47.80966 | P2t |
| PRV-26 | Kyzyl-Chishma Village | 54.99772 | 47.81442 | P2t |
| PRV-26/1 | Kyzyl-Chishma Village | 54.99750 | 47.81577 | Q |
| PRV-26/2 | Kyzyl-Chishma Village | 54.99178 | 47.79538 | Q |
| PRV-27 | Rifulka tract | 54.66155 | 47.29987 | K2 |
| PRV-28 | Yantikovo Village | 55.09047 | 47.86835 | P2t |
| PRV-29 | Ishmurzino-Surinsk Village | 55.05441 | 47.89444 | P2t |
| PRV-30 | Yamanchurino Village | 54.97296 | 47.96838 | P2t |
| PRV-31 | Shemalakovo Village | 54.97709 | 47.90161 | P2t |
References
- Gerasimov, I.P.; Markov, K.K. Ice Age on the USSR. In Proceedings of the Institute of Geography; Academy of Sciences of the USSR: Moscow, Russia, 1939; p. 462. [Google Scholar]
- Velichko, A.A. Natural process in the Pleistocene; Nauka Publishers: Moscow, Russia, 1973; p. 256. (In Russian) [Google Scholar]
- Butakov, G.P. Pleistocene peglacial in the east of the Russian Plain; Kazan State University, Kazan Publishing House: Kazan, Russia, 1986; p. 144. [Google Scholar]
- Hubberten, H.W.; Andreev, A.; Astakhov, V.I.; Demidov, I.; Dowdeswell, J.A.; Henriksen, M.; Hjort, C.; Houmark-Nielsen, M.; Jakobsson, M.; Kuzmina, S.; et al. The periglacial climate and environment in northern Eurasia during the Last Glaciation. Quat. Sci. Rev. 2004, 23, 1333–1357. [Google Scholar] [CrossRef]
- Houmark-Nielsen, M.; Demidov, I.; Funder, S.; Grøsfjeld, K.; Kjær, K.H.; Larsen, E.; Lavrova, N.; Lyså, A.; Nielsen, J.K. Early and Middle Valdaian glaciations, ice-dammed lakes and periglacial interstadials in northwest Russia: New evidence from the Pyoza River area. Glob. Planet. Chang. 2001, 31, 215–237. [Google Scholar] [CrossRef]
- Kalm, V.; Gorlach, A. Impact of bedrock surface topography on spatial distribution of Quaternary sediments and on the flow pattern of late Weichselian glaciers on the East European Craton (Russian Plain). Geomorphology 2014, 207, 1–9. [Google Scholar] [CrossRef]
- Lasberg, K.; Kalm, V.; Kihno, K. Ice-free interval corresponding to marine isotope stages 4 and 3 at the last glacial maximum position at Kileshino, Valdaj upland, Russia. Estonian J. Earth Sci. 2014, 63, 88–96. [Google Scholar] [CrossRef]
- Velichko, A.A.; Faustova, M.A.; Gribchenko, Y.N.; Pisareva, V.V.; Sudakova, N.G. Glaciations of the East European Plain—distribution and chronology. In Developments in Quaternary Sciences; Ehlers, J., Gibbard, P.L., Eds.; Elsevier: Amsterdam, The Nederland, 2004; pp. 337–354. [Google Scholar] [CrossRef]
- Velichko, A.A.; Morozova, T.D.; Nechaev, V.P.; Rutter, N.W.; Dlusskii, K.G.; Little, E.C.; Catto, N.R.; Semenov, V.V.; Evans, M.E. Loess/paleosol/cryogenic formation and structure near the northern limit of loess deposition, East European Plain, Russia. Quat. Int. 2006, 152–153, 14–30. [Google Scholar] [CrossRef]
- Velichko, A.A.; Faustova, M.A.; Pisareva, V.V.; Gribchenko, Y.U.N.; Sudakova, N.G.; Lavrentiev, N.V. Glaciations of the east European plain—Distribution and chronology. Dev. Quat. Sci. 2011, 15, 337–359. [Google Scholar] [CrossRef]
- Svendsen, J.I.; Alexanderson, H.; Astakhov, V.I.; Demidov, I.; Dowdeswell, J.A.; Funder, S.; Gataullin, V.; Henriksen, M.; Hjort, C.; Houmark-Nielsen, M.; et al. Late Quaternary ice sheet history of northern Eurasia. Quat. Sci. Rev. 2004, 23, 11–13. [Google Scholar] [CrossRef]
- Lisiecki, L.E.; Raymo, M.E. A Pliocene-Pleistocene stack of 57 globally distributed benthic δ18O records. Paleoceanogr. Paleoclimatol. 2005, 20, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Velichko, A.A.; Zelikson, E.M. Landscape, climate and mammoth food resources in the East European Plain during the Late Paleolithic epoch. Quat. Int. 2005, 126–128, 137–151. [Google Scholar] [CrossRef]
- Linge, H.; Larsen, E.; Kjær, K.H.; Demidov, I.; Brook, E.J.; Raisbeck, G.M.; Yiou, F. Cosmogenic 10Be exposure age dating across Early to Late Weichselian ice-marginal zones in northwestern Russia. Boreas 2006, 35, 576–586. [Google Scholar] [CrossRef]
- Ehlers, J.; Astakhov, V.; Gibbard, P.L.; Mangerud, J.; Svendsen, J.I. Glaciations| Late Pleistocene Glaciations in Europe. In Encyclopedia of Quaternary Science; Elias, S.A., Ed.; Elsevier: Amsterdam, The Netherlands; pp. 1085–1095. [CrossRef]
- Sycheva, S.; Sedov, S. Paleopedogenesis during the Mikulino interglacial (MIS 5e) in the East-European plain: Buried toposequence of the key-section «Alexandrov quarry». Boletín de la Sociedad Geológica Mexicana 2012, 64, 189–197. [Google Scholar] [CrossRef]
- Yanina, T.A. Correlation of the Late Pleistocene paleogeographical events of the Caspian Sea and Russian Plain. Quat. Int. 2012, 271, 120–129. [Google Scholar] [CrossRef]
- Helmens, K.F. The last interglacial-glacial cycle (MIS 5-2) re-examined based on long proxy records from central and northern Europe. Quat. Sci. Rev. 2014, 86, 115–123. [Google Scholar] [CrossRef]
- Hughes, A.L.C.; Gyllencreutz, R.; Lohne, Ø.S.; Mangerud, J.; Svendsen, J.I. The last Eurasian ice sheets—a chronological database and time-slice reconstruction, DATED-1. Boreas 2016, 45, 1–45. [Google Scholar] [CrossRef]
- Astakhov, V.; Shkatova, V.; Zastrozhnov, A.; Chuyko, M. Glaciomorphological Map of the Russian Federation. Quat. Int. 2016, 420, 4–14. [Google Scholar] [CrossRef]
- Andreicheva, L.N.; Ponomarev, D.V. Litho- and biostratigraphy of the middle neopleistocene of the European northeast of Russia. Stratigr. Geol. Correl. 2018, 26, 584–597. [Google Scholar] [CrossRef]
- Zastrozhnov, A.; Danukalova, G.; Shick, S.; Kolfshoten, T. State of stratigraphic knowledge of Quaternary deposits in European Russia: Unresolved issues and challenges for further research. Quat. Int. 2018, 478, 4–26. [Google Scholar] [CrossRef]
- Sycheva, S.; Pushkina, P.; Khokhlova, O.; Ukrainsky, P. Interrelations of the Bryansk paleosol (end of MIS 3) with the Holocene surface soils in micro-depressions of the central forest-steppe within the Russian upland. Catena 2019, 172, 619–633. [Google Scholar] [CrossRef]
- Ponomarev, D.; Andreicheva, L. Middle-upper quaternary stratigraphy in the northeast of European Russia inferred from rodent record and lithology of tills. Quat. Int. 2019, 534, 60–72. [Google Scholar] [CrossRef]
- Astakhov, V.I. The Northern Pleistocene of Russia; Cambridge Scholars Publishing; Lady Stephenson Library: Newcastle upon Tyne, UK, 2020; p. 610. [Google Scholar]
- Makeev, A.; Rusakov, A.; Khokhlova, O.; Kust, P.; Mikhaylova, D.; Aseyeva, E.; Kurbanova, F.; Rusakova, E.; Mihailov, E. Dataset on spatial variability of soil properties: Tokhmeyevo archaeological site of the bronze age, Chuvashia (southern fringe of the forest zone, the Russian plain). Data Brief 2020, 33, 106489. [Google Scholar] [CrossRef]
- Andreev, A.A.; Gromig, R.; Wagner, B.; Wennrich, V.; Melles, M.; Shumilovskikh, L.S.; Savelieva, L.A.; Fedorov, G.B.; Ludikova, A.; Brill, D. Environmental conditions in northwestern Russia during MIS 5 inferred from the pollen stratigraphy in a sediment core from Lake Ladoga. Boreas 2019, 48, 377–386. [Google Scholar] [CrossRef]
- Bolikhovskaya, N.S.; Molodkov, A.N. East European loess–palaeosol sequences: Palynology, stratigraphy and correlation. Quat. Int. 2006, 149, 24–36. [Google Scholar] [CrossRef]
- Atlas of the Republic of Tatarstan (2005) Cartographic Edition; Cartography: Moscow, Russia, 2005; 215p. (In Russian)
- Markova, A.; Kolfschoten, V.T.; Puzachenko, A.Y. Evolution of the European Ecosystems during Pleistocene–Holocene Transition (24–8 kyr BP); GEOS: Moscow, Russia, 2008; p. 277. [Google Scholar]
- Markova, A.K.; Simakova, A.N.; Puzachenko, A.Y. Ecosystems of Eastern Europe at the time of maximum cooling of the Valdai glaciation (24–18 kyr BP) inferred from data on plant communities and mammal assemblages. Quat. Int. 2009, 201, 53–59. [Google Scholar] [CrossRef]
- Markova, A.K.; Puzachenko, A.Y. European small mammal faunas during Dnieper (Saale) glaciation and transition to the Mikulino (Eem) interglacial. Quat. Int. 2020. [Google Scholar] [CrossRef]
- Novenko, E.Y. Late Valdai pollen flora from loess sediments in the central East-European Plain. Paleoenvironmental reconstruction. Quat. Int. 2006, 152–153, 146–152. [Google Scholar] [CrossRef]
- Novenko, E.Y.; Olchev, A.V. Early Holocene vegetation and climate dynamics in the central part of the East European Plain (Russia). Quat. Int. 2015, 388, 12–22. [Google Scholar] [CrossRef]
- Puzachenko, A.Y.; Markova, A.K. Diversity dynamics of large- and medium-sized mammals in the Late Pleistocene and the Holocene on the East European Plain: Systems approach. Quat. Int. 2016, 420, 391–401. [Google Scholar] [CrossRef]
- Simakova, A.N. The vegetation of the Russian Plain during the second part of the Late Pleistocene (33–18 ka). Quat. Int. 2006, 149, 110–114. [Google Scholar] [CrossRef]
- Little, E.C.; Lian, O.B.; Velichko, A.A.; Morozova, T.D.; Nechaev, V.P.; Dlussky, K.G.; Rutter, N.W. Quaternary stratigraphy and optical dating of loess from the east European Plain (Russia). Quat. Sci. Rev. 2002, 21, 1745–1762. [Google Scholar] [CrossRef]
- Panin, P.G.; Timireva, S.N.; Morozova, T.D.; Kononov, Y.M.; Velichko, A.A. Morphology and micromorphology of the loess-paleosol sequences in the south of the East European plain (MIS 1–MIS 17). Catena 2018, 168, 79–101. [Google Scholar] [CrossRef]
- Panin, A.V.; Astakhov, V.I.; Lotsari, E.; Komatsu, G.; Lang, J.; Winsemann, J. Middle and Late Quaternary glacial lake-outburst floods, drainage diversions and reorganization of fluvial systems in northwestern Eurasia. Earth Sci. Rev. 2020, 201, 103069. [Google Scholar] [CrossRef]
- Sidorchuk, A.Y.; Panin, A.V.; Borisova, O.K. Morphology of river channels and surface runoff in the Volga River basin (East European Plain) during the Late Glacial period. Geomorphology 2009, 113, 137–157. [Google Scholar] [CrossRef]
- Velichko, A.A. (Ed.) Paleoclimates and paleolandscapes of the extratropical space of the Northern Hemisphere. In Late Pleistocene–Holocene; GEOS: Moscow, Russia, 2009; p. 120. (In Russian) [Google Scholar]
- Rusakov, A.; Nikonov, A.; Savelieva, L.; Simakova, A.; Sedov, S.; Maksimov, F.; Kuznetsov, V.; Savenko, V.; Starikova, A.; Korkka, M.; et al. Landscape evolution in the periglacial zone of Eastern Europe since MIS5: Proxies from paleosols and sediments of the Cheremoshnik key site (Upper Volga, Russia). Quat. Int. 2015, 365, 26–41. [Google Scholar] [CrossRef]
- Rusakov, A.; Sedov, S.; Sheinkman, V.; Dobrynin, D.; Zinovyev, E.; Trofimova, S.; Maksimov, F.; Kuznetsov, V.; Korkka, M.; Levchenko, S. Late Pleistocene paleosols in the extra-glacial regions of Northwestern Eurasia: Pedogenesis, post-pedogenic transformation, paleoenvironmental inferences. Quat. Int. 2019, 501, 174–192. [Google Scholar] [CrossRef]
- Simonova, J.; Rusakov, A.; Ryumin, A.; Mirin, D.; Lemeshko, N.; Popov, A.; Rusakova, E. The response of salt-affected hydromorphic soils of the Nero Lake basin to the recent climate change within the Upper Volga Region, Russia. Soil Tillage Res. 2021, 207, 104871. [Google Scholar] [CrossRef]
- Sidorchuk, A.; Borisova, O.; Panin, A. Fluvial response to the Late Valdai/Holocene environmental change on the East European Plain. Glob. Planet. Chang. 2001, 28, 303–318. [Google Scholar] [CrossRef]
- Zaretskaya, N.; Panin, A.; Molod’kov, A.; Trofimova, S.; Simakova, A.; Baranov, D. Pleistocene stratigraphy of the Vychegda River basin, European North-East. Quat. Int. 2020, 546, 185–195. [Google Scholar] [CrossRef]
- Zaretskaya, N.E.; Panin, A.V.; Karpukhina, N.V. The SIS limits and related proglacial events in the Severnaya Dvina basin, northwestern Russia: Review and new data. Bull. Geol. Soc. Finl. 2018, 90, 301–313. [Google Scholar] [CrossRef]
- Cohen, K.M.; Gibbard, P.L. Global chronostratigraphical correlation table for the last 2.7 million years, version 2019 QI-500. Quat. Int. 2019, 500, 20–31. [Google Scholar] [CrossRef]
- Jones, M.D.; Dee, S.; Anderson, L.; Baker, A.; Bowen, G.; Noone, D.C. Water isotope systematics: Improving our palaeoclimate interpretations. Quaternary Sci. Rev. 2016, 131, 243–249. [Google Scholar] [CrossRef]
- Miller, G.H.; Brigham-Grette, J.; Alley, R.B.; Anderson, L.; Bauch, H.A.; Douglas, M.S.V.; Edwards, M.E.; Elias, S.A.; Finney, B.P.; Fitzpatrick, J.J.; et al. Temperature and precipitation history of the Arctic. Quaternary Sci. Rev. 2010, 29, 1679–1715. [Google Scholar] [CrossRef]
- Négrel, P.; Petelet-Giraud, E. Isotopes in groundwater as indicators of climate changes. Trends Analytical Chem. 2011, 30, 1279–1290. [Google Scholar] [CrossRef] [Green Version]
- Paillard, D. Quaternary glaciations: From observations to theories. Quat. Sci. Rev. 2015, 107, 11–24. [Google Scholar] [CrossRef]
- Ferronsky, V.I.; Vlasova, L.S.; Esikov, A.D.; Zorin, L.V.; Borisova, Z.K.; Polyakov, V.A.; Seletsky, Y.B.; Punning, Y.-M.K.; Vaikmäe, R.A. Variations in the isotopic composition of groundwater, atmospheric precipitation and organic matter in alluvial sediments due to climate fluctuations. Water Res. 1982, 5, 6–23. [Google Scholar]
- Suksi, U.J.; Rasilainen, K.; Ruskeeniemi, T.; Marcos, N.; Hellmuth, K.H. Natural U occurrences as a palaeo-hydrogeological indicator—observations from the Palmottu natural analogue site, Finland. In Uranium in the Aquatic Environment; Merkel, B.J., Planer-Friedrich, B., Wolkersdorfer, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar] [CrossRef]
- Francke, A.; Dosseto, A.; Just, J.; Wagner, B.; Jones, B.G. Assessment of the controls on (234U/238U) activity ratios recorded in detrital lacustrine sediments. Chem. Geol. 2020, 550, 119698. [Google Scholar] [CrossRef]
- Kayzar, T.M.; Villa, A.C.; Lobaugh, M.L.; Gaffney, A.M.; Williams, R.W. Investigating uranium distribution in surface sediments and waters: A case study of contamination from the Juniper Uranium Mine, Stanislaus National Forest, CA. J. Environ. Radioactivity 2014, 136, 85–97. [Google Scholar] [CrossRef]
- Tokarev, I.V.; Bubnov, Y.P.; Kharkhordin, I.L. Conditions for the formation of resources and the quality of groundwater in the southwest of the Republic of Tatarstan according to geochemical and isotope data. In Materials of the International Conference “The Future of Hydrogeology: Current Trends and Prospects”; St. Petersburg State University: St. Petersburg, Russia, 2008; pp. 196–208. [Google Scholar]
- Tokarev, I.V.; Zubkov, A.A.; Rumynin, V.G.; Pozdnyakov, S.P.; Polyakov, V.A.; Kuznetsov, V.Y. Assessment of the long-term safety of radioactive waste disposal: 1. Paleoreconstruction of groundwater formation conditions. Water Res. 2009, 36, 206–213. [Google Scholar] [CrossRef]
- Tokarev, I.V.; Zubkov, A.A.; Rumynin, V.G.; Pozdnyakov, S.P. Assessment of the long-term safety of radioactive waste disposal: 2. Isotopic study of water exchange in a multilayer system. Water Res. 2009, 36, 345–356. [Google Scholar] [CrossRef]
- Tokarev, I.V.; Kipfer, R.; Tomonaga, Y.; Brennwald, M.S.; Vereschagina, E.A. Comparison of 4He and 14C dating, noble-gas temperatures and stable isotope (d2H, d18O) data for groundwater in stratified aquifers (Tomsk-7, S.E. Siberia). Mineral. Mag. 2011, 75, 2018. [Google Scholar] [CrossRef]
- Manakov, A.V. Kuibyshev Reservoir; Nauka Publishers: Leningrad, Russia, 1983; p. 214. (In Russian) [Google Scholar]
- Sycheva, S.A. Paleo-permafrost events in the periglacial area of the Central Russian Upland at the end of the Middle and Late Pleistocene. Earth’s Cryosphere 2012, 16, 45–56. [Google Scholar] [CrossRef]
- Geology of the Republic of Tatarstan; Kazan State University: Kazan, Russia, 2007; p. 74. (In Russian)







| MIS | Stage | Age, ka | Paleo-relief forms, cryotextures | Frosty conditions |
|---|---|---|---|---|
| 5e | Mikulino | 128–117 | Small wedges data | Seasonal permafrost |
| 5d | Early Valdai Stadium 1 | 117–100 | Plastic deformations, small pseudomorphs on wedges, etc. | Discontinuous permafrost |
| 5c | Early Valdai Interstadial 1 | 100–91 | Ground veins—humus tongues | Seasonal freezing and drying out |
| 5b | Early Valdai Stadium 2 | 91–77 | Small wedges | Discontinuous permafrost |
| 5a | Early Valdai Interstadial 2 | 77–71 | Soil veins—humus tongues-cosmas | Seasonal freezing and drying out |
| 4 | Early Valdai Stadium 3 | 71–58 | Pseudomorphs on ice wedges, 0.6 m wide, 0.5 m deep, and post-cryogenic textures | Continuous and discontinuous permafrost |
| 3 | Middle Valdai interstadial 1 | 58–49.5 | Small cracks, gleying | Deep seasonal freezing |
| 3 | Middle Valdai Stadium 1 | 49.5–32.0 | Plastic deformations and small wedges | Discontinuous permafrost |
| 3 | Middle Valdai Interstadial 2 | 32.0–24.0 | Wedges | Deep seasonal freezing |
| 2 | Maximum glaciation and late glacial | 24.0–12.2 | Large wedge-shaped structures. Dells. | Continuous permafrost, then intermittent and seasonal permafrost |
| 1 | Holocene | 11.6–0 | Seasonal freezing |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yakovlev, E.; Tokarev, I.; Zykov, S.; Iglovsky, S.; Ivanchenko, N. Isotope Signs (234U/238U, 2H, 18O) of Groundwater: An Investigation of the Existence of Paleo-Permafrost in European Russia (Pre-Volga Region). Water 2021, 13, 1838. https://doi.org/10.3390/w13131838
Yakovlev E, Tokarev I, Zykov S, Iglovsky S, Ivanchenko N. Isotope Signs (234U/238U, 2H, 18O) of Groundwater: An Investigation of the Existence of Paleo-Permafrost in European Russia (Pre-Volga Region). Water. 2021; 13(13):1838. https://doi.org/10.3390/w13131838
Chicago/Turabian StyleYakovlev, Evgeny, Igor Tokarev, Sergey Zykov, Stanislav Iglovsky, and Nikolay Ivanchenko. 2021. "Isotope Signs (234U/238U, 2H, 18O) of Groundwater: An Investigation of the Existence of Paleo-Permafrost in European Russia (Pre-Volga Region)" Water 13, no. 13: 1838. https://doi.org/10.3390/w13131838
APA StyleYakovlev, E., Tokarev, I., Zykov, S., Iglovsky, S., & Ivanchenko, N. (2021). Isotope Signs (234U/238U, 2H, 18O) of Groundwater: An Investigation of the Existence of Paleo-Permafrost in European Russia (Pre-Volga Region). Water, 13(13), 1838. https://doi.org/10.3390/w13131838





