Facile Detection of Vitamin B12 with Copper Oxide Nanocrystal Graphenic Composite Electrode
Abstract
:1. Introduction
2. Experimental
2.1. Reagents and Chemicals
2.2. Apparatus
2.3. Preparation of the GUITAR Electrode
2.4. Preparation of the Copper Oxide-Modified GUITAR Electrode
2.5. Electrochemical Measurements
2.6. Preparation of Standard Solution
2.7. Interference Elimination of the Biological Sample
2.8. Standard Addition Method
2.9. Method Validation with HPLC
3. Results and Discussion
3.1. Characterization of Electrodes
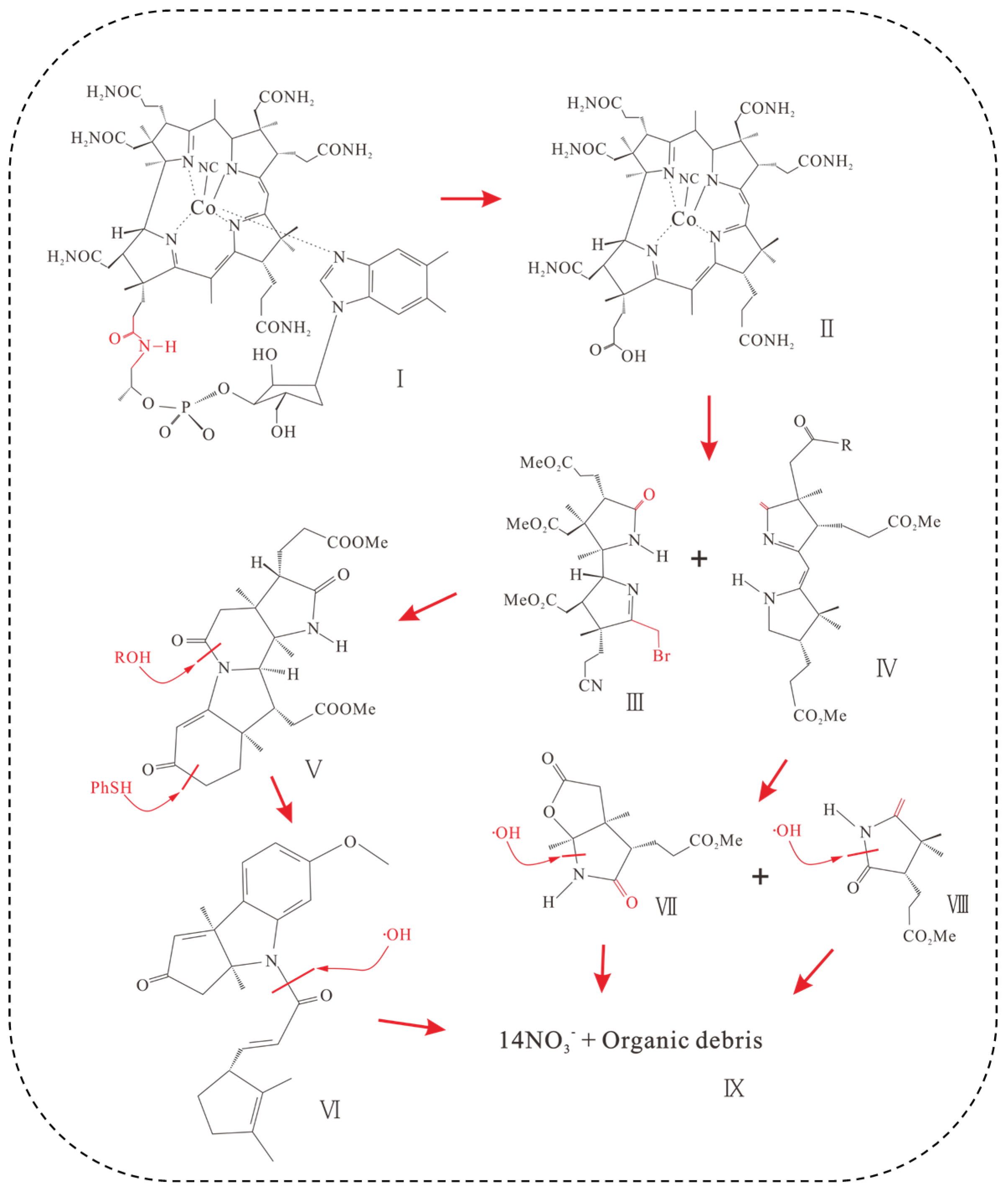
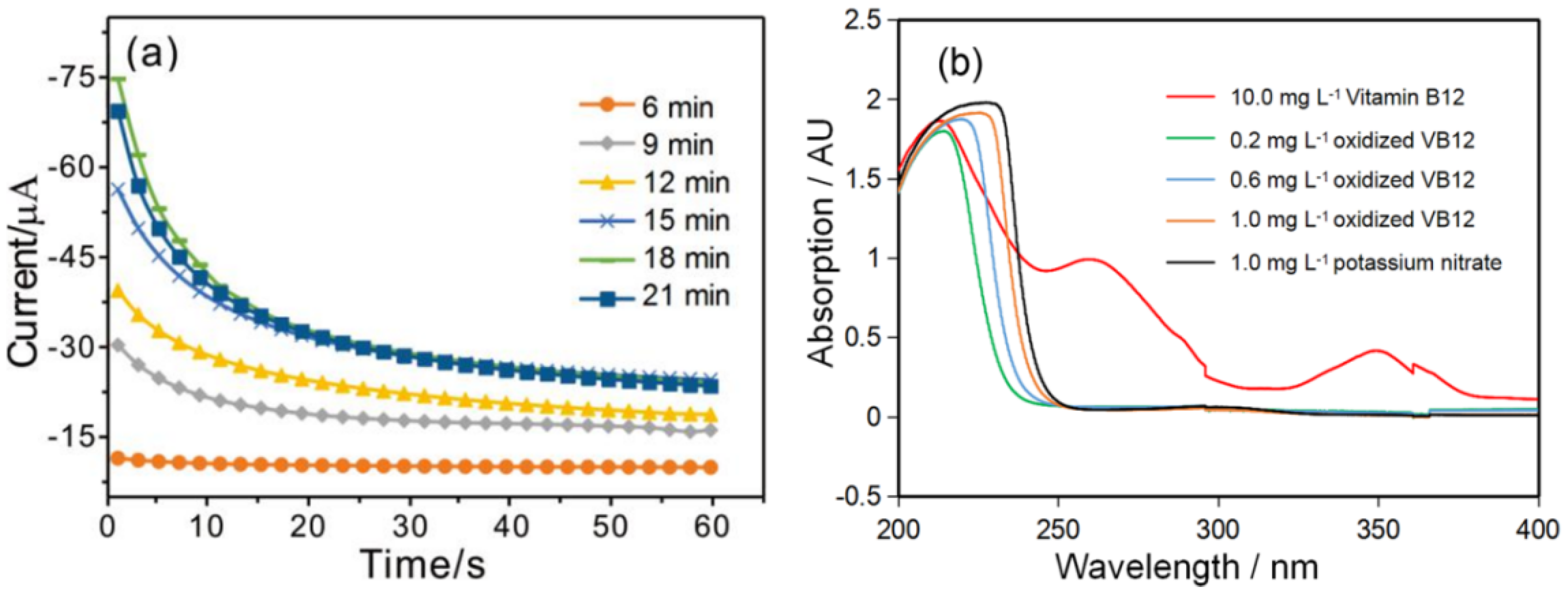
3.2. Electrochemical Oxidation of VB12
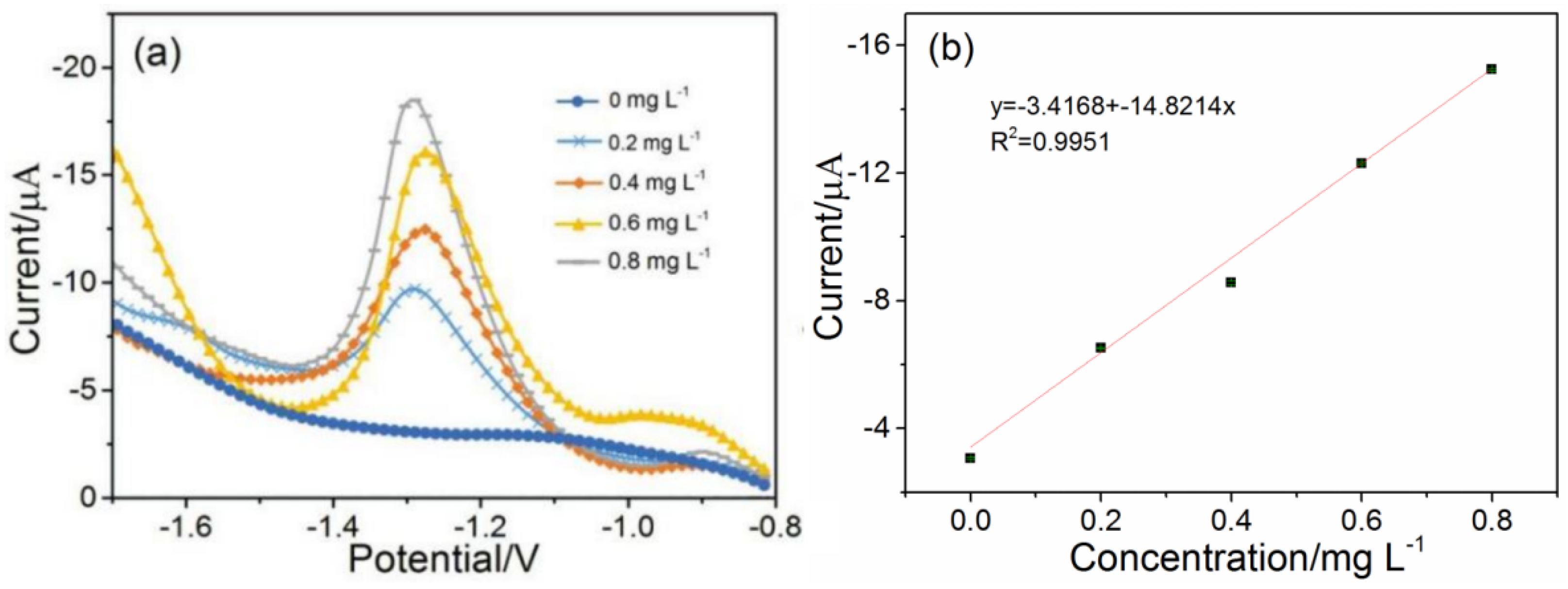
3.3. The Establishment of Calibration Curves
3.4. Interference Study
3.5. Quantification of VB12 in Biological Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brown, K.L. Chemistry and enzymology of vitamin B12. Chem. Rev. 2005, 105, 2075–2149. [Google Scholar] [CrossRef]
- Giedyk, M.; Goliszewska, K.; Gryko, D. Vitamin B12 catalysed reactions. Chem. Soc. Rev. 2015, 44, 3391–3404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Padmanabhan, S.; Jost, M.; Drennan, C.; Elías-Arnanz, M. A new facet of vitamin B12: Gene regulation by cobalamin-based photoreceptors. Annu. Rev. Biochem. 2017, 86, 485–514. [Google Scholar] [CrossRef] [PubMed]
- Bastet, L.; Dubé, A.; Massé, E.; Lafontaine, D. New insights into riboswitch regulation mechanisms. Mol. Microbiol. 2011, 80, 1148–1154. [Google Scholar] [CrossRef]
- Shelton, A.N.; Seth, E.C.; Mok, K.C.; Han, A.W.; Jackson, S.N.; Haft, D.R.; Taga, M.E. Uneven distribution of cobamide biosynthesis and dependence in bacteria predicted by comparative genomics. ISME J. 2018, 13, 789–804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.B.; Chen, F. Determination of vitamin B12 in pharmaceutical preparations by a highly sensitive fluorimetric method. Fresen. J. Anal. Chem. 2000, 368, 836–838. [Google Scholar] [CrossRef]
- Castilho, S.; Borrego, A.; Henriques, C.; Ribeiro, M.F.; Fernandes, A. Monitoring cobalt ions siting in BEA and FER zeolites by in-situ UV–Vis spectroscopy: A DRS study. Inorg. Chim. Acta 2017, 455, 568–574. [Google Scholar] [CrossRef]
- Moreno, P.; Salvado, V. Determination of eight water- and fat-soluble vitamins in multi-vitamin pharmaceutical formulations by high-performance liquid chromatography. J. Chromatogr. A 2000, 870, 207–215. [Google Scholar] [CrossRef]
- Li, H.B.; Chen, F.; Jiang, Y. Determination of vitamin B12 in multivitamin tablets and fermentation medium by high-performance liquid chromatography with fluorescence detection. J. Chromatogr. A 2000, 891, 243–247. [Google Scholar] [CrossRef]
- Lambert, D.; Adjalla, C.; Felden, F.; Benhayoun, S.; Nicolas, J.P.; Guéant, J.L. Identification of vitamin B12 and analogues by high-performance capillary electrophoresis and comparison with high-performance liquid chromatography. J. Chromatogr. A 1992, 608, 311–315. [Google Scholar] [CrossRef]
- Shokrollahi, A.; Ebrahimi, F. Supramolecular-based ultrasonic-assisted dispersion solidification liquid-liquid microextraction of copper and cobalt prior to their flame atomic absorption spectrometry determination. J. Aoac Int. 2017, 100, 1861–1868. [Google Scholar] [CrossRef]
- Kumar, S.S.; Chouhan, R.S.; Thakur, M.S. Trends in analysis of vitamin B12. Anal. Biochem. 2010, 398, 139–149. [Google Scholar] [CrossRef]
- Kou, Y.; Lu, J.; Jiang, X.; Tian, B.; Xue, Y.; Wang, M.; Tan, L. Electrochemical determination of vitamin B12 based on Cu2+-involved fenton-like reaction. Electroanalysis 2019, 31, 1–10. [Google Scholar] [CrossRef]
- Kreft, G.L.; Braga, O.C.D.; Spinelli, A. Analytical electrochemistry of vitamin B12 on a bismuth-film electrode surface. Electrochim. Acta 2012, 83, 125–132. [Google Scholar] [CrossRef]
- Parvin, M.H.; Azizi, E.; Arjomandi, J.; Lee, J.Y. Highly sensitive and selective electrochemical sensor for detection of vitamin B12 using an Au/PPy/FMNPs@TD-modified electrode. Sensor. Actuat. B Chem. 2018, 261, 335–344. [Google Scholar] [CrossRef]
- Kuralay, F.; Vural, T.; Bayram, C.; Denkbas, E.B.; Abaci, S. Carbon nanotube-chitosan modified disposable pencil graphite electrode for vitamin B12 analysis. Colloid. Surface. B 2011, 87, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Wan, Q.; Wang, X. Voltammetry of vitamin b12 on a thin self-assembled monolayer modified electrode. Electrochim. Acta 2005, 50, 2175–2180. [Google Scholar] [CrossRef]
- Tomčik, P.; Banks, C.E.; Davies, T.J. A self-catalytic carbon paste electrode for the detection of vitamin B12. Anal. Chem. 2004, 76, 161–165. [Google Scholar] [CrossRef]
- Pala, B.B.; Vural, T.; Kuralay, F.; Cirak, T.; Bolat, G.; Abaci, S.; Denkbas, E.B. Disposable pencil graphite electrode modified with peptide nanotubes for vitamin B12 analysis. Appl. Surf. Sci. 2014, 303, 37–45. [Google Scholar] [CrossRef]
- Pereira, D.F.; Santana, E.R.; Piovesan, J.V.; Spinelli, A. A novel electrochemical strategy for determination of vitamin B12 by Co(i/ii) redox pair monitoring with boron-doped diamond electrode. Diam. Relat. Mater. 2020, 105, 107793. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, W.; Jia, R.; Yu, Y.; Zhang, B. Unveiling the activity origin of a copper-based electrocatalyst for selective nitrate reduction to ammonia. Angew. Chem. Int. Edit. 2020, 59, 5350–5354. [Google Scholar] [CrossRef] [PubMed]
- Reyter, D.; Bélanger, D.; Roué, L. Study of the electroreduction of nitrate on copper in alkaline solution. Electrochim. Acta 2008, 53, 5977–5984. [Google Scholar] [CrossRef]
- Badea, G.E. Electrocatalytic reduction of nitrate on copper electrode in alkaline solution. Electrochim. Acta 2009, 54, 996–1001. [Google Scholar] [CrossRef]
- Pignatello, J.J.; Oliveros, E.; MacKay, A. Advanced oxidation processes for organic contaminant destruction based on the fenton reaction and related chemistry. Crit. Rev. Environ. Sci. Technol. 2006, 36, 1–84. [Google Scholar] [CrossRef]
- Babuponnusami, A.; Muthukumar, K. A review on fenton and improvements to the fenton process for wastewater treatment. J. Environ. Chem. Eng. 2014, 2, 557–572. [Google Scholar] [CrossRef]
- Hoigné, J. Inter-calibration of oh radical sources and water quality parameters. Water Sci. Technol. 1997, 35, 1–8. [Google Scholar] [CrossRef]
- Cheng, I.F.; Xie, Y.; Gyan, I.O.; Nicholas, N.W. Highest measured anodic stability in aqueous solutions: Graphenic electrodes from the thermolyzed asphalt reaction. Rsc Adv. 2013, 3, 2379–2384. [Google Scholar] [CrossRef]
- Cheng, I.F.; Xie, Y.; Gonzales, R.A.; Brejna, P.R.; Sundararajan, J.P.; Kengne, B.A.F.; Aston, D.E.; McIlroy, D.N.; Foutch, J.D.; Griffiths, P.R. Synthesis of graphene paper from pyrolyzed asphalt. Carbon 2011, 49, 2852–2861. [Google Scholar] [CrossRef]
- Hamam, A.; Oukil, D.; Dib, A.; Hammache, H.; Makhloufi, L.; Saidani, B. Polypyrrole coated cellulosic substrate modified by copper oxide as electrode for nitrate electroreduction. Surf. Rev. Lett. 2015, 22, 1550065. [Google Scholar] [CrossRef]
- Okbamichael, M.; Sanudo-Wilhelmy, S.A. A new method for the determination of vitamin B12 in seawater. Anal. Chim. Acta 2004, 517, 33–38. [Google Scholar] [CrossRef]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902. [Google Scholar] [CrossRef]
- Rao, C.N.R.; Sood, A.K.; Subrahmanyam, K.S.; Govindaraj, A. Graphene: The new two-dimensional nanomaterial. Angew. Chem. Int. Edit. 2010, 48, 7752–7777. [Google Scholar] [CrossRef] [PubMed]
- Achary, L.S.K.; Nayak, P.S.; Barik, B.; Kumar, A.; Dash, P. Ultrasonic-assisted green synthesis of β-amino carbonyl compounds by copper oxide nanoparticles decorated phosphate functionalized graphene oxide via Mannich reaction. Catal. Today 2020, 348, 137–147. [Google Scholar] [CrossRef]
- Collins, D.A.; Hogenkamp, H.P. Adionuclide Labeling of Vitamin B12 and Coenzymes Thereof. US Patent 5,739,313, 1998. [Google Scholar]
- Davies, M.J. The oxidative environment and protein damage. Biochim. Biophys. Acta 2005, 1703, 93–109. [Google Scholar] [CrossRef]
- Sante-Lhoutellier, V.; Aubry, L.; Gatellier, P. Effect of oxidation on in vitro digestibility of skeletal muscle myofibrillar proteins. J. Agr. Food Chem. 2007, 55, 5343–5348. [Google Scholar] [CrossRef] [PubMed]
- Brillas, E.; Bastida, R.M.; Llosa, E.; Casado, J. Electrochemical destruction of aniline and 4-chloroaniline for wastewater treatment using a carbon-ptfe O2-fed cathode. J. Electrochem. Soc. 1995, 142, 1733–1741. [Google Scholar] [CrossRef]
- Cheng, Y.; Ma, B.; Tan, C.P.; Lai, O.M.; Panpipat, W.; Cheong, L.Z.; Shen, C. Hierarchical macro-microporous zif-8 nanostructures as efficient nano-lipase carriers for rapid and direct electrochemical detection of nitrogenous diphenyl ether pesticides. Sensor. Actuat. B Chem. 2020, 321, 128477. [Google Scholar] [CrossRef]
- Manivel, P.; Madasamy, K.; Suryanarayanan, V.; Nesakumar, N.; Kulandaisamy, A.J.; Kathiresan, M.; Parthiban, M.; Velayutham, D.; Lee, K.M. Cu(HBTC) (4,4′-bipy)·3DMF nanorods supported on platinum electrode as an electrochemical sensing platform for efficient vitamin B12 detection. J. Taiwan Inst. Chem. Eng. 2019, 96, 1–10. [Google Scholar] [CrossRef]



| Electrode | Sample Source | LOD | Detection Element | Reference |
|---|---|---|---|---|
| GCE(ea)/PTH a | Injection sample | 2 nmol L−1 | Cobalt | [13] |
| BiFE b | Pharmaceutical | 33.1 nmol L−1 | Cobalt | [14] |
| Au/PPy/FMNPs@TD c | Food samples | 0.62 nmol L−1 | Cobalt | [15] |
| SWCNT-PGE d | Pharmaceutical | 0.89 nmol L−1 | Cobalt | [16] |
| MMA/SAM/Au e | Pharmaceutical | 1.0 nmol L−1 | Cobalt | [17] |
| DBCH-CPE f | Pharmaceutical | 0.85 nmol L−1 | Cobalt | [18] |
| PNT-PGE g | Pharmaceutical | 93 nmol L−1 | Cobalt | [19] |
| Boron-doped diamond electrode | Toothpaste | 86 nmol L−1 | Cobalt | [20] |
| Copper oxide-modified GUITAR electrode | Microbial sample | 0.59 nmol L−1 | Nitrogen | This work |
| Sample | Added/nM | Found/nM | Recovery, % | RSD, % N = 3 |
|---|---|---|---|---|
| 1021 cell N | 105.00 | 107.72 | 102.59 | 2.3 |
| cobD cell N | 5.50 | 5.83 | 106.00 | 2.1 |
| 1021 cell H | 60.00 | 59.17 | 98.62 | 1.7 |
| cobD cell H | 3.50 | 3.54 | 101.14 | 2.0 |
| Sample | This Work/nM | HPLC/nM | Error/nM |
|---|---|---|---|
| 1021 cell N | 107.72 | 106.47 | +1.25 |
| cobD cell N | 5.83 | 5.24 | +0.59 |
| 1021 cell H | 59.17 | 57.77 | +1.40 |
| cobD cell H | 3.54 | 3.17 | +0.37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, C.; Zhao, N.; Jiang, X.; Wan, D.; Xie, Y. Facile Detection of Vitamin B12 with Copper Oxide Nanocrystal Graphenic Composite Electrode. Water 2021, 13, 1790. https://doi.org/10.3390/w13131790
Tian C, Zhao N, Jiang X, Wan D, Xie Y. Facile Detection of Vitamin B12 with Copper Oxide Nanocrystal Graphenic Composite Electrode. Water. 2021; 13(13):1790. https://doi.org/10.3390/w13131790
Chicago/Turabian StyleTian, Chao, Na Zhao, Xinyue Jiang, Duanji Wan, and Yuqun Xie. 2021. "Facile Detection of Vitamin B12 with Copper Oxide Nanocrystal Graphenic Composite Electrode" Water 13, no. 13: 1790. https://doi.org/10.3390/w13131790




