Microwave Irradiation in Technologies of Wastewater and Wastewater Sludge Treatment: A Review
Abstract
1. Introduction
2. Materials and Methods
3. Results
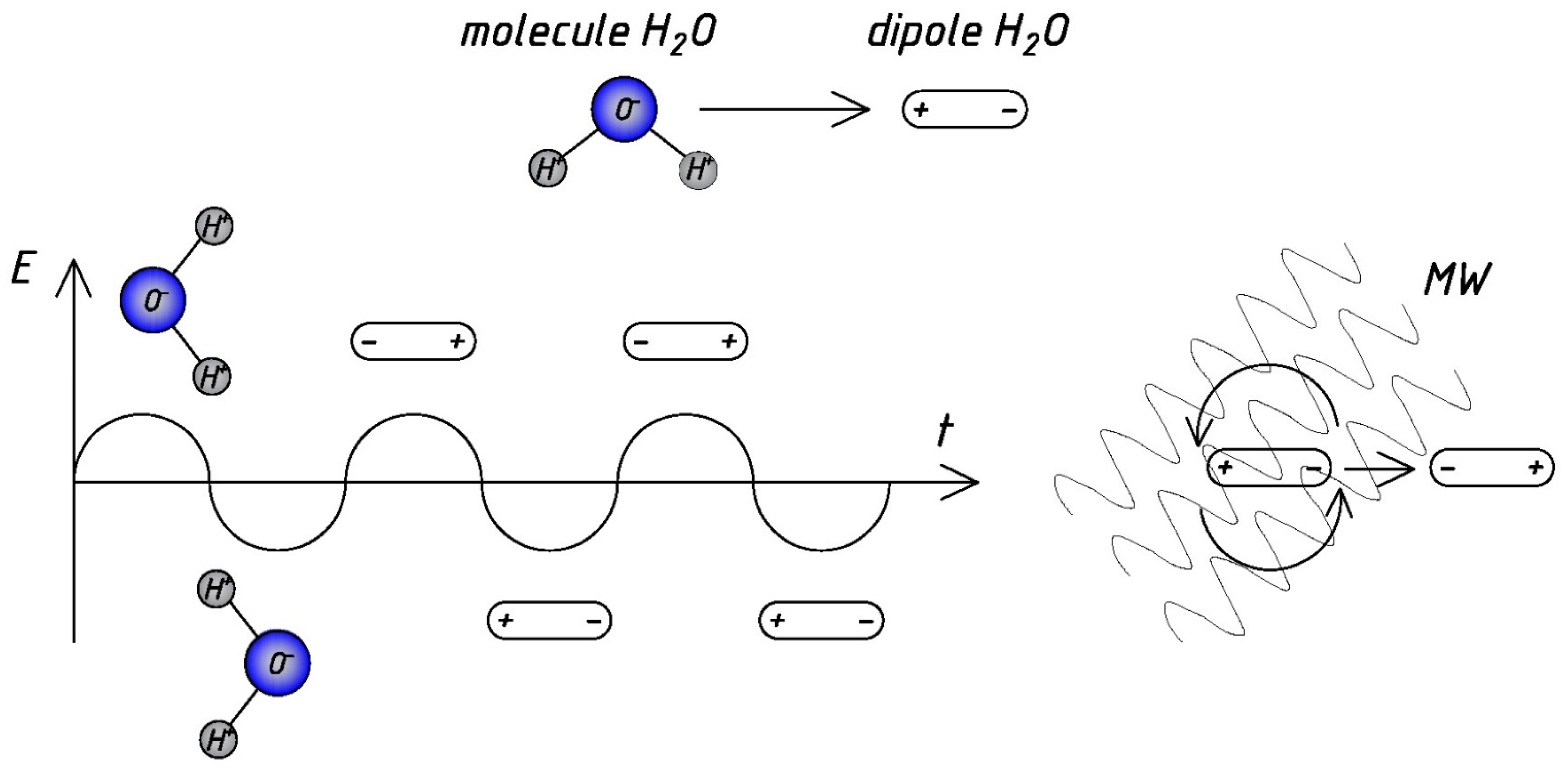
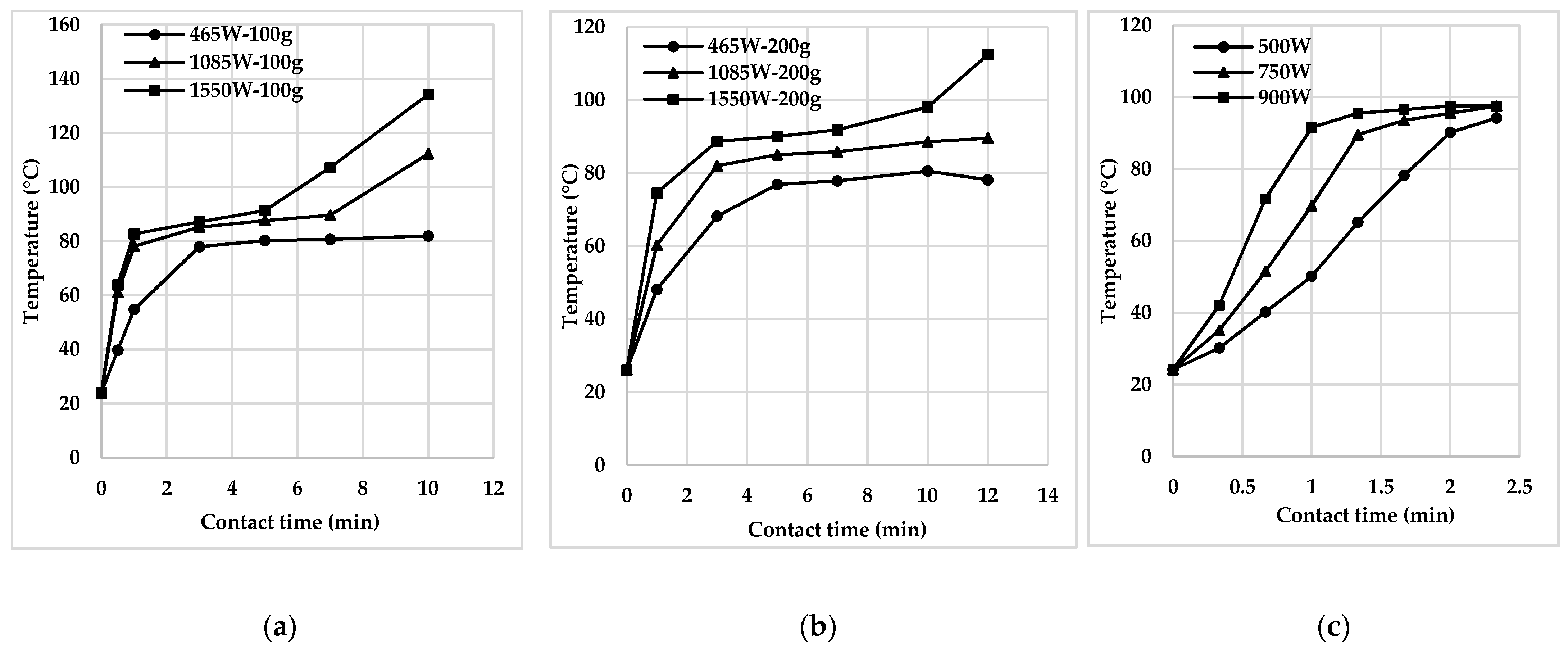
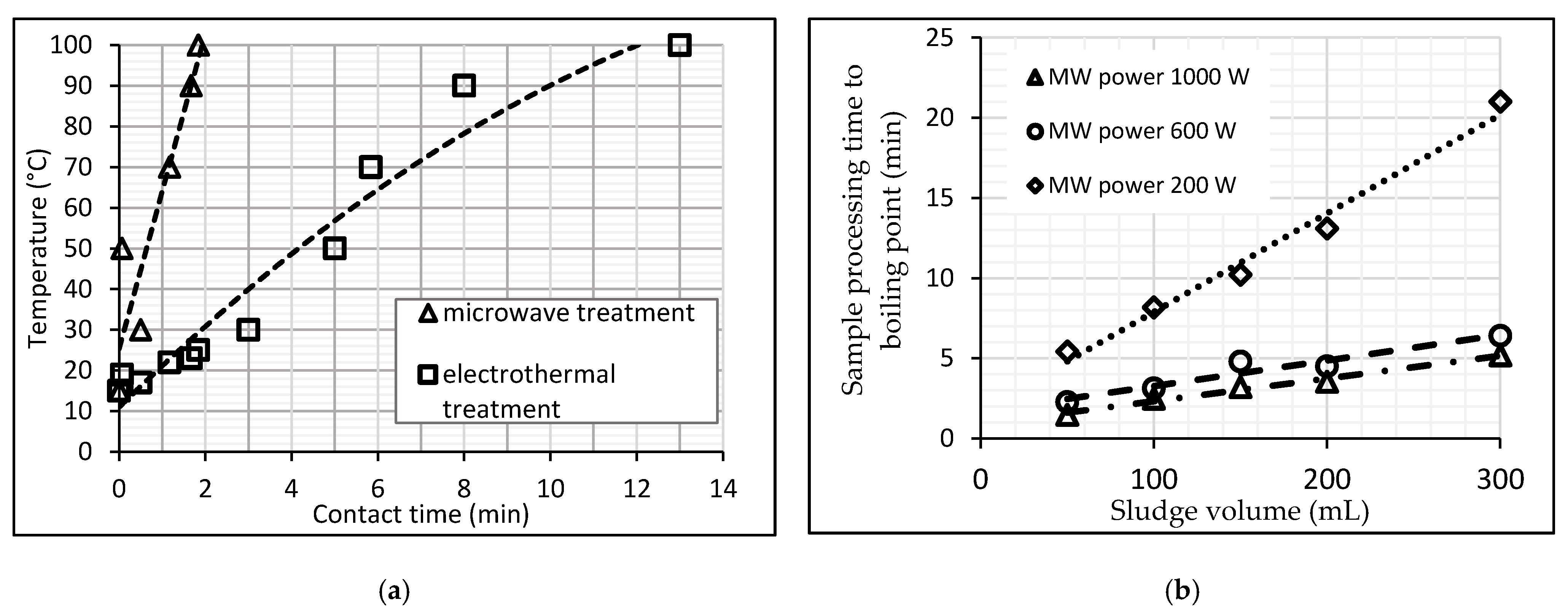
3.1. Heating and Thermal Treatment
3.2. Properties Change
3.2.1. Wastewater
3.2.2. Wastewater Sludge
3.3. Decontamination (Disinfection)
3.4. Decomposition of Organic Substances
3.5. Demulsification
3.6. Extraction of Heavy Metals
3.7. Biogas Processing
3.8. Pyrolysis of Wastewater Sludge
3.9. Modification of Sorbents
3.10. Devices for MW-Processing of WW and WWS
4. Conclusions and Considerations for Future Research
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Priroda, S.U. Nature, Ecology and Environment. Available online: http://priroda.su/ (accessed on 4 May 2021).
- Harris, P.W.; McCabe, B.K. Review of pre-treatments used in anaerobic digestion and their potential application in high-fat cattle slaughterhouse wastewater. Appl. Energy 2015, 155, 560–575. [Google Scholar] [CrossRef]
- Tyagi, V.; Lo, S.-L. Microwave irradiation: A sustainable way for sludge treatment and resource recovery. Renew. Sustain. Energy Rev. 2013, 18, 288–305. [Google Scholar] [CrossRef]
- Zhen, G.; Lu, X.; Kato, H.; Zhao, Y.; Li, Y.-Y. Overview of pretreatment strategies for enhancing sewage sludge disintegration and subsequent anaerobic digestion: Current advances, full-scale application and future perspectives. Renew. Sustain. Energy Rev. 2017, 69, 559–577. [Google Scholar] [CrossRef]
- Elagroudy, S.; El-Gohary, F. Microwave Pretreatment of Mixed Sludge for Anaerobic Digestion Enhancement. Environ. Eng. 2013, 5, 105–111. [Google Scholar] [CrossRef]
- Kichigin, V.; Samara State Technical University; Zemlyanova, M.; Vyalkova, E.; Tumen State Industrial University. Study of the Possibility of Using Microwave Radiation for the Treatment of Liquid Municipal Waste. Urban Constr. Arch. 2018, 8, 44–49. [Google Scholar] [CrossRef]
- Mudhoo, A.; Sharma, S.K.; Sharma, S.K. Microwave Irradiation Technology in Waste Sludge and Wastewater Treatment Research. Crit. Rev. Environ. Sci. Technol. 2011, 41, 999–1066. [Google Scholar] [CrossRef]
- Wiesbrock, F.; Hoogenboom, R.; Schubert, U.S. Microwave-Assisted Polymer Synthesis: State-of-the-Art and Future Perspectives. Macromol. Rapid Commun. 2004, 25, 1739–1764. [Google Scholar] [CrossRef]
- Remya, N.; Lin, J.-G. Current status of microwave application in wastewater treatment—A review. Chem. Eng. J. 2011, 166, 797–813. [Google Scholar] [CrossRef]
- Gole, V.L.; Gogate, P.R. Degradation of brilliant green dye using combined treatment strategies based on different irradiations. Sep. Purif. Technol. 2014, 133, 212–220. [Google Scholar] [CrossRef]
- Herrero, M.A.; Kremsner, J.M.; Kappe, C.O. Nonthermal Microwave Effects Revisited: On the Importance of Internal Temperature Monitoring and Agitation in Microwave Chemistry. J. Org. Chem. 2008, 73, 36–47. [Google Scholar] [CrossRef]
- Yang, L.; Chen, Z.; Yang, J.; Liu, Y.; Wang, J.; Yu, Y.; Gao, X. Removal of volatile fatty acid in landfill leachate by the microwave-hydrothermal method. Desalination Water Treat. 2013, 52, 4423–4429. [Google Scholar] [CrossRef]
- Wei, R.; Wang, P.; Zhang, G.; Wang, N.; Zheng, T. Microwave-responsive catalysts for wastewater treatment: A review. Chem. Eng. J. 2020, 382, 122781. [Google Scholar] [CrossRef]
- Vialkova, E.; Zemlyanova, M.; Danilov, O. Energy efficiency in municipal waste treatment. MATEC Web Conf. 2018, 170, 04020. [Google Scholar] [CrossRef][Green Version]
- Hidaka, H.; Saitou, A.; Honjou, H.; Hosoda, K.; Moriya, M.; Serpone, N. Microwave-assisted dechlorination of polychlorobenzenes by hypophosphite anions in aqueous alkaline media in the presence of Pd-loaded active carbon. J. Hazard. Mater. 2007, 148, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Kappe, C.O. Controlled Microwave Heating in Modern Organic Synthesis. Angew. Chem. Int. Ed. 2004, 43, 6250–6284. [Google Scholar] [CrossRef]
- Mishra, R.R.; Sharma, A.K. Microwave–material interaction phenomena: Heating mechanisms, challenges and opportunities in material processing. Compos. Part A Appl. Sci. Manuf. 2016, 81, 78–97. [Google Scholar] [CrossRef]
- Clark, D.E.; Folz, D.C.; West, J.K. Processing materials with microwave energy. Mater. Sci. Eng. A 2000, 287, 153–158. [Google Scholar] [CrossRef]
- Horikoshi, S.; Serpone, N. Photochemistry with microwaves: Catalysts and environmental applications. J. Photochem. Photobiol. C Photochem. Rev. 2009, 10, 96–110. [Google Scholar] [CrossRef]
- Baghurst, D.R.; Mingos, D.M.P. Superheating effects associated with microwave dielectric heating. J. Chem. Soc. Chem. Commun. 1992, 674–677. [Google Scholar] [CrossRef]
- Gabriel, C.; Gabriel, S.; Grant, E.H.; Halstead, B.S.J.; Mingos, D.M.P. Dielectric parameters relevant to microwave dielectric heating. Chem. Soc. Rev. 1998, 27, 213–224. [Google Scholar] [CrossRef]
- Zhang, L.; Guo, X.; Yan, F.; Su, M.; Li, Y. Study of the degradation behaviour of dimethoate under microwave irradiation. J. Hazard. Mater. 2007, 149, 675–679. [Google Scholar] [CrossRef] [PubMed]
- Akbari, S.; Nour, A.; Jamari, S.; Rajabi, A. Demulsification of Water-in-Crude Oil Emulsion via Conventional Heating and Microwave Heating Technology in Their Optimum Conditions. Aust. J. Basic Appl. Sci. 2016, 10, 66–74. [Google Scholar]
- Vialkova, E.; Zemlyanova, M.; Fugaeva, A. Treatment and utilization of liquid communal waste in the cities. MATEC Web Conf. 2018, 212, 03005. [Google Scholar] [CrossRef][Green Version]
- Yu, Q.; Lei, H.; Li, Z.; Li, H.; Chen, K.; Zhang, X.; Liang, R. Physical and chemical properties of waste-activated sludge after microwave treatment. Water Res. 2010, 44, 2841–2849. [Google Scholar] [CrossRef] [PubMed]
- Mawioo, P.M.; Rweyemamu, A.; Garcia, H.A.; Hooijmans, C.M.; Brdjanovic, D. Evaluation of a microwave based reactor for the treatment of blackwater sludge. Sci. Total Environ. 2016, 548–549, 72–81. [Google Scholar] [CrossRef]
- Mawioo, P.M.; Hooijmans, C.M.; Garcia, H.A.; Brdjanovic, D. Microwave treatment of faecal sludge from intensively used toilets in the slums of Nairobi, Kenya. J. Environ. Manag. 2016, 184, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Tigyi, J.; Masszi, G.; Koszorus, L. A Simple Microwave Argument about the Changes of Structure of Water in Solutions of Macromolecules. In Interactions of Water in Ionic and Nonionic Hydrates; Springer: Berlin/Heidelberg, Germany, 1987; pp. 195–198. [Google Scholar]
- Chaplin, M. A proposal for the structuring of water. Biophys. Chem. 2000, 83, 211–221. [Google Scholar] [CrossRef]
- Asakuma, Y.; Kanazawa, Y.; Parmar, H.; Phan, C.; Pareek, V.; Evans, G. Surface Tension Profiles under Various Microwave Radiation Modes. J. Energy Power Eng. 2014, 8, 585–588. [Google Scholar]
- Parmar, H.; Asada, M.; Kanazawa, Y.; Asakuma, Y.; Phan, C.M.; Pareek, V.; Evans, G.M. Influence of Microwaves on the Water Surface Tension. Langmuir 2014, 30, 9875–9879. [Google Scholar] [CrossRef]
- Asakuma, Y.; Munenaga, T.; Nakata, R. Observation of bubble formation in water during microwave irradiation by dynamic light scattering. Heat Mass Transf. 2016, 52, 1833–1840. [Google Scholar] [CrossRef]
- Wong, T.W.; Iskhandar, A.; Kamal, M.; Jumi, S.J.; Kamarudin, N.H.; Zin, N.Z.M.; Salleh, N.H.M. Effects of Microwave on Water and Its Influence on Drug Dissolution. Prog. Electromagn. Res. C 2009, 11, 121–136. [Google Scholar] [CrossRef]
- Zemlyanova, M.V. Intensification of Household Wastewater Sludge Treatment by Ultra-High Frequency Electromagnetic Radiation. Ph.D. Thesis, Samarskiy Gosudarstvennyy Arkhitekturno-Stroitel’nyy Universitet, Samara, Russia, 2015. Available online: http://www.dslib.net/vodosnabzhenie/intensifikacija-processov-obrabotki-osadkov-gorodskih-stochnyh-vod-s-pomowju.html (accessed on 3 May 2021).
- Zhang, J.; Pang, Q.; He, Z.; Tian, C.; Wu, T. Treatment of Blast Furnace Gas Washing Water by Utilization of Coagulation Associated with Microwave. In Applications of Process Engineering Principles in Materials Processing, Energy and Environmental Technologies; Wang, S., Free, M.L., Alam, S., Zhang, M., Taylor, P.R., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 555–563. [Google Scholar]
- Zemlyanova, M.; Vialkova, E. Investigation of Combined Influence of Super-High-Frequency Electromagnetic Radiation on the Sewage Sediment Properties. Ecol. Ind. Russ. 2018, 22, 20–25. [Google Scholar] [CrossRef]
- Liu, J.; Wei, Y.; Li, K.; Tong, J.; Wang, Y.; Jia, R. Microwave-acid pretreatment: A potential process for enhancing sludge dewaterability. Water Res. 2016, 90, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Wojciechowska, E. Application of microwaves for sewage sludge conditioning. Water Res. 2005, 39, 4749–4754. [Google Scholar] [CrossRef]
- Doğan, I.; Sanin, F.D. Alkaline solubilization and microwave irradiation as a combined sludge disintegration and minimization method. Water Res. 2009, 43, 2139–2148. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Chang, J.; Chen, J.; Kong, H. Impacts of Microwave on Dewater Ability and Morphological Feature of Sewage Sludge. J. Civ. Archit. Environ. Eng. 2013, 35, 135–139. [Google Scholar] [CrossRef]
- Mawioo, P.M.; Garcia, H.A.; Hooijmans, C.M.; Velkushanova, K.; Simonič, M.; Mijatović, I.; Brdjanovic, D. A pilot-scale microwave technology for sludge sanitization and drying. Sci. Total Environ. 2017, 601–602, 1437–1448. [Google Scholar] [CrossRef]
- Rao, B.; Su, X.; Lu, X.; Wan, Y.; Huang, G.; Zhang, Y.; Xu, P.; Qiu, S.; Zhang, J. Ultrahigh pressure filtration dewatering of municipal sludge based on microwave pretreatment. J. Environ. Manag. 2019, 247, 588–595. [Google Scholar] [CrossRef]
- Makarov, P.O. Biophysical Bases of the Action of Ultraviolet and Ultrasonic Radiation and Ultrahigh-Frequency Electromagnetic Field. Lectures on Biophysics; LSU Publishing House: Leningrad, Russia, 1968; pp. 209–233. [Google Scholar]
- Devyatkov, N.D. Influence of Millimeter-band Electromagnetic Radiation on Biological Objects. Sov. Phys. Uspekhi 1974, 16, 568–569. [Google Scholar] [CrossRef]
- Belitsky, B.I. Study of the effect of the microwave field on microorganisms in pulse and continuous mode. Biophysics 1982, 27, 923–927. [Google Scholar]
- Panasenko, V.I. The Effect of a Powerful EMF with a Frequency of 2375 Mhz on Microorganisms. Biological Effect of Electromagnetic Fields; USSR: Pushchino, Russia, 1982; pp. 26–27. [Google Scholar]
- Benjamin, E.; Reznik, A.; Williams, A.L. Mathematical Models for Conventional and Microwave Thermal Deactivation of Enterococcus Faecalis, Staphylococcus aureus and Escherichia coli. Cell Mol. Biol. 2007, 53, 42–48. [Google Scholar]
- Rosenberg, H.C. System for Purifying Liquids. U.S. Patent 4013558A, 22 March 1977. [Google Scholar]
- Zabolotskij, L.L.; Klimarev, S.I.; Lobanov, A.G. Device for Disinfection and Heating of Aqueous Media. Patent SU1139439A1, 1985. [Google Scholar]
- Klimarev, S.I.; Grigorev, A.A.; Sinyak, Y.E. Method for Liquids Disinfection and Heating, and Device for Its Implementation. Russian Patent RU2627899, 14 August 2017. [Google Scholar]
- Ahmedova, O.O.; Stepanov, S.F.; Soshinov, A.G.; Bahtiarov, K.H. Increase of Efficiency of Local Treatment Facilities of Sewage for the Account of Application of the Combined Electrophysical Methods of Influence. Mod. Probl. Sci. Educ. 2009, 5, 56–60. [Google Scholar]
- Karlsson, M.; Carlsson, H.; Idebro, M.; Eek, C. Microwave Heating as a Method to Improve Sanitation of Sewage Sludge in Wastewater Plants. IEEE Access 2019, 7, 142308–142316. [Google Scholar] [CrossRef]
- Ara, E.; Sartaj, M.; Kennedy, K. Effect of microwave pre-treatment of thickened waste activated sludge on biogas production from co-digestion of organic fraction of municipal solid waste, thickened waste activated sludge and municipal sludge. Waste Manag. Res. 2014, 32, 1200–1209. [Google Scholar] [CrossRef]
- Kenge, A.; Liao, P.H.; Lo, K.V. Solubilization of municipal anaerobic sludge using microwave-enhanced advanced oxidation process. J. Environ. Sci. Health Part A 2009, 44, 502–506. [Google Scholar] [CrossRef]
- Hong, S.M.; Park, J.K.; Teeradej, N.; Lee, Y.O.; Cho, Y.K.; Park, C.H. Pretreatment of Sludge with Microwaves for Pathogen Destruction and Improved Anaerobic Digestion Performance. Water Environ. Res. 2006, 78, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Kuglarz, M.; Karakashev, D.B.; Angelidaki, I. Microwave and thermal pretreatment as methods for increasing the biogas potential of secondary sludge from municipal wastewater treatment plants. Bioresour. Technol. 2013, 134, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Worthington, P. Organic Micropollutants in the Aqueous Environment. In Studies in Environmental Science; Pawlowski, L., Alaerts, G., Lacy, W.J., Eds.; Chemistry for Protection of the Environment 1985; Elsevier: Amsterdam, The Netherlands, 1986; Volume 29, pp. 235–244. [Google Scholar]
- Randtke, S.J. Organic Contaminant Removal by Coagulation and Related Process Combinations. J. Am. Water Work. Assoc. 1988, 80, 40–56. [Google Scholar] [CrossRef]
- Kuzubova, L.I.; Morozov, S.V. Organic contaminant of drinking water. Ecology. World Lit. Rev. Ser. 1993, 1, 1–167. [Google Scholar]
- Garcia-Costa, A.L.; Zazo, J.A.; Casas, J.A. Microwave-assisted catalytic wet peroxide oxidation: Energy optimization. Sep. Purif. Technol. 2019, 215, 62–69. [Google Scholar] [CrossRef]
- Cheng, G.; Lin, J.; Lu, J.; Zhao, X.; Cai, Z.; Fu, J. Advanced Treatment of Pesticide-Containing Wastewater Using Fenton Reagent Enhanced by Microwave Electrodeless Ultraviolet. BioMed Res. Int. 2015, 2015, 1–8. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, N.; Sun, X.; Zhao, Q.; Wang, P. Treatment of polymer-flooding wastewater by a modified coal fly ash-catalysed Fenton-like process with microwave pre-enhancement: System parameters, kinetics, and proposed mechanism. Chem. Eng. J. 2021, 406, 126734. [Google Scholar] [CrossRef]
- Lin, L.; Chen, J.; Xu, Z.; Yuan, S.; Cao, M.; Liu, H.; Lu, X. Removal of ammonia nitrogen in wastewater by microwave radiation: A pilot-scale study. J. Hazard. Mater. 2009, 168, 862–867. [Google Scholar] [CrossRef]
- Ravera, M.; Buico, A.; Gosetti, F.; Cassino, C.; Musso, D.; Osella, D. Oxidative degradation of 1,5-naphthalenedisulfonic acid in aqueous solutions by microwave irradiation in the presence of H2O2. Chemosphere 2009, 74, 1309–1314. [Google Scholar] [CrossRef]
- Jung, S.C. The microwave-assisted photo-catalytic degradation of organic dyes. Water Sci. Technol. 2011, 63, 1491–1498. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-C.; Lo, S.-L.; Chiueh, P.-T.; Chang, D.-G. Efficient decomposition of perfluorocarboxylic acids in aqueous solution using microwave-induced persulfate. Water Res. 2009, 43, 2811–2816. [Google Scholar] [CrossRef]
- Han, D.-H.; Cha, S.-Y.; Yang, H.-Y. Improvement of oxidative decomposition of aqueous phenol by microwave irradiation in UV/H2O2 process and kinetic study. Water Res. 2004, 38, 2782–2790. [Google Scholar] [CrossRef] [PubMed]
- Ta, N.; Hong, J.; Liu, T.; Sun, C. Degradation of atrazine by microwave-assisted electrodeless discharge mercury lamp in aqueous solution. J. Hazard. Mater. 2006, 138, 187–194. [Google Scholar] [CrossRef]
- Hong, J.; Sun, C.; Yang, S.-G.; Liu, Y.-Z. Photocatalytic degradation of methylene blue in TiO2 aqueous suspensions using microwave powered electrodeless discharge lamps. J. Hazard. Mater. 2006, 133, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Horikoshi, S.; Hidaka, H.; Serpone, N. Environmental remediation by an integrated microwave/UV illumination technique: VI. A simple modified domestic microwave oven integrating an electrodeless UV-Vis lamp to photodegrade environmental pollutants in aqueous media. J. Photochem. Photobiol. A Chem. 2004, 161, 221–225. [Google Scholar] [CrossRef]
- Horikoshi, S.; Tokunaga, A.; Hidaka, H.; Serpone, N. Environmental remediation by an integrated microwave/UV illumination method: VII. Thermal/non-thermal effects in the microwave-assisted photocatalyzed mineralization of bisphenol-A. J. Photochem. Photobiol. A Chem. 2004, 162, 33–40. [Google Scholar] [CrossRef]
- Zhanqi, G.; Shaogui, Y.; Na, T.; Cheng, S. Microwave assisted rapid and complete degradation of atrazine using TiO2 nanotube photocatalyst suspensions. J. Hazard. Mater. 2007, 145, 424–430. [Google Scholar] [CrossRef]
- Verma, P.; Samanta, S.K. Microwave-enhanced advanced oxidation processes for the degradation of dyes in water. Environ. Chem. Lett. 2018, 16, 969–1007. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Li, G.; Qu, J. Oxidative decomposition of azo dye C.I. Acid Orange 7 (AO7) under microwave electrodeless lamp irradiation in the presence of H2O2. J. Hazard. Mater. 2006, 134, 183–189. [Google Scholar] [CrossRef]
- Hong, J.; Ta, N.; Yang, S.-G.; Liu, Y.-Z.; Sun, C. Microwave-assisted direct photolysis of bromophenol blue using electrodeless discharge lamps. Desalination 2007, 214, 62–69. [Google Scholar] [CrossRef]
- Wang, N.; Wang, P. Study and application status of microwave in organic wastewater treatment—A review. Chem. Eng. J. 2016, 283, 193–214. [Google Scholar] [CrossRef]
- Ju, Y.; Yang, S.; Ding, Y.; Sun, C.; Gu, C.; He, Z.; Qin, C.; He, H.; Xu, B. Microwave-enhanced H2O2-based process for treating aqueous malachite green solutions: Intermediates and degradation mechanism. J. Hazard. Mater. 2009, 171, 123–132. [Google Scholar] [CrossRef]
- Yang, S.; Wang, P.; Yang, X.; Wei, G.; Zhang, W.; Shan, L. A novel advanced oxidation process to degrade organic pollutants in wastewater: Microwave-activated persulfate oxidation. J. Environ. Sci. 2009, 21, 1175–1180. [Google Scholar] [CrossRef]
- Zhang, X.; Li, G.; Wang, Y. Microwave assisted photocatalytic degradation of high concentration azo dye Reactive Brilliant Red X-3B with microwave electrodeless lamp as light source. Dye. Pigment. 2007, 74, 536–544. [Google Scholar] [CrossRef]
- Anshuman, A.; Saremi-Yarahmadi, S.; Vaidhyanathan, B. Enhanced catalytic performance of reduced graphene oxide–TiO2 hybrids for efficient water treatment using microwave irradiation. RSC Adv. 2018, 8, 7709–7715. [Google Scholar] [CrossRef]
- Mishra, S.; Meda, V.; Dalai, A.K.; Headley, J.V.; Peru, K.M.; McMartin, D.W. Microwave treatment of naphthenic acids in water. J. Environ. Sci. Health Part A 2010, 45, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- Evdokimov, I.N.; Novikov, M.A. Structural features of industrial water-crude oil emulsions. Microwave studies. Chem. Technol. Fuels Oils 2007, 43, 46–50. [Google Scholar] [CrossRef]
- Kovaleva, L.A.; Zinnatullin, R.R.; Mullayanov, A.; Mavletov, M.V.; Blagochinnov, V.N. Microstructure evolution of water-oil emulsions in high-frequency and microwave electromagnetic fields. High Temp. 2013, 51, 870–872. [Google Scholar] [CrossRef]
- Saifuddin, N.M.; Chua, K.H. Treatment of Oily Waste Water Emulsions from Metallurgical Industries Using Microwave Irradiation. Biotechnology 2006, 5, 308–314. [Google Scholar] [CrossRef]
- Kuo, C.-H.; Lee, C.-L. Treatment of oil/water emulsions using seawater-assisted microwave irradiation. Sep. Purif. Technol. 2010, 74, 288–293. [Google Scholar] [CrossRef]
- Saifuddin, N.M.; Hussein, R. Separation of Water from Very Stable Water-in-Oil Emulsion Using Microwave Radiation with Presence of Inorganic Salts. Int. J. Chem. 2014, 35, 1512–1521. [Google Scholar]
- Abdurahman, N.; Yunus, R.; Azhari, N.; Said, N.; Hassan, Z. The Potential of Microwave Heating in Separating Water-in-Oil (w/o) Emulsions. Energy Procedia 2017, 138, 1023–1028. [Google Scholar] [CrossRef]
- Akbari, S.; Nour, A.; Fayaz, F.; Halimi, M.; Nour, A. Analysis of Microwave Heating Process for Demulsification of Water-in-Crude Oil Emulsions. Aust. J. Basic Appl. Sci. 2016, 10, 1–10. [Google Scholar]
- Basak, T. Role of resonances on microwave heating of oil-water emulsions. AIChE J. 2004, 50, 2659–2675. [Google Scholar] [CrossRef]
- Saitov, R.I.; Abdeev, E.R.; Khasanova, A.F.; Abdeev, R.G.; Rukomoynikov, A.A. Development of Energy-Efficient Equipment and Technology for Environmentally Friendly Microwave Processing of Oil Sludge. Int. J. Eng. Adv. Technol. 2020, 9, 1683–1687. [Google Scholar]
- Lamble, K.J.; Hill, S.J. Microwave digestion procedures for environmental matrices. Analyst 1998, 123, 103–133. [Google Scholar] [CrossRef]
- Kutseva, N.K.; Kryuchkova, S.L.; Pirogova, S.V.; Naumova, S.V.; Kryuchkov, V.A.; Chamaev, A.V. Microwave Sample Preparation in the Determination of Metals in Waste Water. J. Anal. Chem. 2000, 55, 1142–1147. [Google Scholar] [CrossRef]
- Kütük, M.A.; Aksoy, M. A Case Study on Sewage Sludge Incineration Plant: Gaski. In Proceedings of the Second International Conference on Water, Energy and the Environment, Kusadası, Turkey, 21–24 September 2013; pp. 1–6. [Google Scholar]
- Blagojevic, V.; Sustersic, V.; Bozickovic, S.; Veselin, B.; Vanja, Š.; Siniša, B. Pyrolysis and gasification in the process of sewage sludge treatment. Zast. Mater. 2017, 58, 305–312. [Google Scholar] [CrossRef]
- Donatello, S.; Cheeseman, C.R. Recycling and recovery routes for incinerated sewage sludge ash (ISSA): A review. Waste Manag. 2013, 33, 2328–2340. [Google Scholar] [CrossRef] [PubMed]
- Khramenkov, S.V.; Pakhomov, A.N.; Khrenov, K.E.; Streltsov, S.A.; Khamidov, M.G.; Belov, N.A. Utilization of Biogas and Creation of Autonomous Sources of Power Supply at Treatment Facilities. Water Supply Sanit. Tech. 2010, 10-1, 48–53. [Google Scholar]
- Fericelli, P.D. Comparison of Sludge Treatment by Gasification vs. Incineration. In Proceedings of the Ninth LACCEI Latin American and Caribbean Conference (LACCEI’2011), Medellín, Colombia, 3–5 August 2011; p. 10. [Google Scholar]
- Danilovich, D.A. Crisis of competence in the design of wastewater treatment facilities. Best Available Technol. Water Supply Sanit. 2018, 4, 5–13. [Google Scholar]
- Coelho, N.M.G.; Droste, R.L.; Kennedy, K.J. Evaluation of continuous mesophilic, thermophilic and temperature phased anaerobic digestion of microwaved activated sludge. Water Res. 2011, 45, 2822–2834. [Google Scholar] [CrossRef]
- Hong, S.-M. Enhancement of Pathogen Destruction and Anaerobic Digestibility Using Microwaves. Ph.D. Thesis, University of Wisconsin, Madison, WI, USA, 2002. [Google Scholar]
- Eskicioglu, C.; Kennedy, K.J.; Droste, R.L. Enhancement of batch waste activated sludge digestion by microwave pretreatment. Water Environ. Res. 2007, 79, 2304–2317. [Google Scholar] [CrossRef]
- Eskicioglu, C.; Droste, R.L.; Kennedy, K.J. Performance of Anaerobic Waste Activated Sludge Digesters after Microwave Pretreatment. Water Environ. Res. 2007, 79, 2265–2273. [Google Scholar] [CrossRef] [PubMed]
- Toreci, I.; Kennedy, K.J.; Droste, R.L. Effect of High-Temperature Microwave Irradiation on Municipal Thickened Waste Activated Sludge Solubilization. In Proceedings of the 11th Conference on Process Integration, Modeling and Optimization for Energy Saving and Pollution Reduction (PRES), Prague, Czech Republic, 24–28 August 2008. [Google Scholar]
- Toreci, I.; Kennedy, K.J.; Droste, R.L. Evaluation of continuous mesophilic anaerobic sludge digestion after high temperature microwave pretreatment. Water Res. 2009, 43, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Mehdizadeh, S.N.; Eskicioglu, C.; Bobowski, J.; Johnson, T. Conductive heating and microwave hydrolysis under identical heating profiles for advanced anaerobic digestion of municipal sludge. Water Res. 2013, 47, 5040–5051. [Google Scholar] [CrossRef] [PubMed]
- Haranghy, L.; Kertesz, S.Z.; Vereb, G.; Laszlo, Z.S.; Vagvolgyi, A.; Jakoi, Z.; Czupy, I.; Hodur, C.; Rakhely, G.; Beszedes, S. Intensification of the biodegradation of wastewater sludge by microwave irradiation. Geosci. Eng. 2020, 8, 322–333. Available online: https://www.researchgate.net/publication/346483507 (accessed on 3 May 2021).
- Yanin, E.P. Domestic wastewater sludge burning (Problems and Methods). Resour. Sav. Technol. 2006, 24, 3–29. [Google Scholar]
- Zaker, A.; Chen, Z.; Wang, X.; Zhang, Q. Microwave-assisted pyrolysis of sewage sludge: A review. Fuel Process. Technol. 2019, 187, 84–104. [Google Scholar] [CrossRef]
- Menéndez, J.; Inguanzo, M.; Pis, J. Microwave-induced pyrolysis of sewage sludge. Water Res. 2002, 36, 3261–3264. [Google Scholar] [CrossRef]
- Malyshkina, E.S.; Vyalkova, E.I.; Osipova, E.Y. Water Purification with Natural Sorbents. Vestn. Tomsk. Gos. arkhitekturno-stroitel’nogo Univ. J. Constr. Arch. 2019, 21, 188–200. [Google Scholar] [CrossRef]
- Bakhia, T.; Khamizov, R.K.; Bavizhev, M.D.; Konov, M.A. The effect of microwave treatment of clinoptilolite on its ion-exchange kinetic properties. Sorpt. Chromatogr. Process. 2016, 16, 803–812. [Google Scholar]
- Berdonosov, S.S. Microwave chemistry. Soros Educ. J. 2001, 7, 32–38. [Google Scholar]
- Bannova, E.A.; Kitaeva, N.K.; Merkov, S.M.; Muchkina, M.V.; Zaloznaya, E.P.; Martynov, P.N. Study of a method for obtaining a hydrophobic sorbent based on modified peat. Sorpt. Chromatogr. Process. 2013, 13, 60–68. [Google Scholar]
- Danilov, O.S.; Mikheyev, V.A.; Moskalenko, T.V. Research of Electromagnetic Microwave Radiation Influence on the Solid Fuels. Izv. Samara Sci. Cent. Russ. Acad. Sci. 2011, 13, 1264–1267. [Google Scholar]
- Belchinskaya, L.I.; Khodosova, N.A.; Novikova, L.A. Effect of different mechanisms of heating of layered aluminosilicate on sorption processes Communication 1. Effect of preliminary thermal and electromagnetic (microwave) heating of montmorillonite on sorption of water. Sorption and chromatographic processes. Coрбциoнные Хрoматoграфические Прoцессы 2017, 17, 781–791. (In Russian) [Google Scholar] [CrossRef]
- Faizal, A.M.; Kutty, S.R.M.; Ezechi, E.H. Removal of Oil from Water by Column Adsorption Method Using Microwave Incinerated Rice Husk Ash (MIRHA). InCIEC 2014 2015, 963–971. [Google Scholar] [CrossRef]
- Myuller, R.F.; Ol’shanskaya, V.P.; Rumyantsev, A.I. Method of Regeneration of Sorbents by Non-Thermal Exposure to Ultra-High-Frequency Electromagnetic Radiation. Russian Patent RU2438774C1, 10 January 2012. [Google Scholar]
- Yuen, F.K.; Hameed, B.H. Recent developments in the preparation and regeneration of activated carbons by microwaves. Adv. Colloid Interface Sci. 2009, 149, 19–27. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Microwave-assisted regeneration of activated carbon. Bioresour. Technol. 2012, 119, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Stenanov, S.F.; Ahmedova, O.O.; Soshimov, A.G. Sewage Treatment Installation. Russian Patent RU116851U1, 10 June 2012. [Google Scholar]
- Gunich, S.V.; Valycheva, T.I. Method of Processing of Domestic and Industrial Wastes to Furnace Fuel and Hydrocarbon Substance and Device to This End. Russian Patent RU2552259C2, 10 June 2015. [Google Scholar]
- Naumov, V.V.; Zdraevskij, D.A.; Glazunov, V.I.; Berezovskij, O.M. Method of Processing and Utilisation of Wastes. Russian Patent RU2573137C1, 10 January 2016. [Google Scholar]
- Levin, E.V. Installation of Microwave Processing of Sewage Sludge. Russian Patent RU2582415C2, 27 April 2016. [Google Scholar]
- Zemlyanova, M.V.; Vialkova, E.I.; Obukhov, L.V. Method for Treatment and Decontamination of Waste Water and Sediments Thereof, and Device for Method Implementing. Russian Patent RU2693783C1, 4 July 2019. [Google Scholar]
- Kovalev, D.A.; Kovalev, A.A.; Sobchenko, Y.A. Method and Plant for Anaerobic Processing of Liquid Organic Wastes. Russian Patent RU2687415C1, 13 May 2019. [Google Scholar]
- Bogdanov, A.V.; Perevalova, N.I.; Migunov, M.I.; Tarasevich, S.A.; Khrushchev, V.V.; Grekhov, I.V.; Kovaleva, L.A.; Zinnatullin, R.R.; Sultanguzhin, R.F.; Gabdrafikov, A.F. System and Method for Electromagnetic Phase Separation of Water-Oil Emulsion. Russian Patent RU2710181C1, 24 December 2019. [Google Scholar]
- Sivokon, V.N.; Tishechkin, N.N.; Klyushnik, A.A.; Dyatlov, A.V.; Smirnov, A.A. Wastewater Treatment Plant by Irradiation with Ultrahigh Frequency Waves and Ultraviolet Light. Russian Patent RU193171U1, 15 October 2019. [Google Scholar]
- Zobnin, B.B.; Kochetkov, V.V.; Vozhegov, A.V.; Semiachkov, A.I.; Ponomarev, O.P.; Matevosian, M.B. Method of Purifying Acid Mine Water and Mobile Process Complex for Implementation Thereof. Russian Patent RU2739259C1, 22 December 2020. [Google Scholar]
| Section | Direction of MW Research | WW | WWS | [Ref./No] |
|---|---|---|---|---|
| Section 3.1 | Heating and thermal treatment | + | + | [3,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27] |
| Section 3.2 | Properties change | + | + | [28,29,30,31,32,33,34,35,36,37,38,39,40,41,42] |
| Section 3.3 | Decontamination | + | + | [14,26,28,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56] |
| Section 3.4 | Decomposition of organic substances | + | - | [9,13,22,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81] |
| Section 3.5 | Demulsification | + | - | [82,83,84,85,86,87,88,89,90] |
| Section 3.6 | Extraction of heavy metals | + | + | [6,14,24,91,92] |
| Section 3.7 | Generation of biogas | - | + | [93,94,95,96,97,98,99,100,101,102,103,104,105,106] |
| Section 3.8 | Pyrolysis of sewage sludge | - | + | [107,108,109] |
| Section 3.9 | Sorbent modification | + | - | [73,110,111,112,113,114,115,116,117,118,119] |
| Section 3.10 | Devices for WW and WWS MW processing | + | + | [120,121,122,123,124,125,126,127,128] |
| Paper’s Category | Number | [Ref./No] | Country * |
|---|---|---|---|
| Magazine Site | 1 | [1] | Russia |
| Reviews | 28 | [2,3,4,7,8,9,13,14,16,17,21,43,51,57,58,59,73,76,90,91,93,94,95,98,107,108,112,119] | Australia, Taiwan, Japan, China, India, The Netherlands, Russia, Austria, UK, Malaysia, and Serbia |
| Research papers | 86 | [5,6,10,11,12,15,18,19,20,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,44,45,46,47,52,53,54,55,56,60,61,62,63,64,65,66,67,68,69,70,71,72,74,75,77,78,79,80,81,82,83,84,85,86,87,88,89,92,96,97,99,100,101,102,103,104,105,106,109,110,111,113,114,115,116,119] | Egypt, Russia, India, Austria, China, Japan, Italy, USA, UK, Malaysia, Afghanistan, The Netherlands, Hungary, Australia, Japan, Poland, Turkey, South Africa, Slovenia, Sweden, Canada, Korea, Denmark, Spain |
| Patents | 13 | [48,49,50,117,120,121,122,123,124,125,126,127,128] | USA, and Russia |
| Properties | Mechanism, Reason | Power MW | Duration MW | Description of Results | [Ref./No] |
|---|---|---|---|---|---|
| Structure | Water molecules’ excitation and mobility | 300 W | 10 min | More mobile, less ordered water structure and corrugated water clusters | [28,29] |
| Surface tension | Decrease in viscosity and increase in water molecules’ activity | 30–600 W | 10–120 s | Water surface tension rapidly decreased at about 20–30% | [30,31,32] |
| pH | Extraction of carbon dioxide | 300 W 800 W | 10 min 5–8 min | pH: from 4.81 to 4.91 pH: from 6.04 to 7.33 | [33,34] |
| Electrical conductivity | Increase in water molecules’ activity | 300 W | 10 min | Electrical conductivity of water samples increased by 7.8% | [33] |
| Nanobubbles formation | Surface tension change | 30–100 W 100–500 W | 60 s 10 s | Nanobubbles size: 200–2500 nm 400–4000 nm | [32] |
| Coagulation of suspended solids | MW treatment, reagent addition and sedimentation | 396 W | 2 min | Particles’ growth process, high deposition rate | [25,35] |
| Change in quality indicators of WW | Decomposition of substances | 800 W | 5–10 min | Decrease in WW pollution (Table 4) | [14,34] |
| Wastewater Indexes | Inlet Concentration before MW, mg/L | Outlet Concentration after MW, mg/L | Description of Results |
|---|---|---|---|
| Suspended matters | 940 | 850 | The concentration decreases from 940 to 450 mg/L (after first stage) and then increases to 850 mg/L (after second stage). Efficient is 9.5% |
| Chemical oxygen demand (COD) | 1240 | 490 | Efficient is 60.5% |
| Biological oxygen demand (BOD) | 430 | 250 | The concentration decreases from 430 to 250 mg/L (after first stage) and then increases to 280 mg/L (after second stage). Efficient is 35% |
| Ammonium-ion | 200 | 150 | Efficient is 25% |
| Nitrate-ion | 0 | 3.8 | The concentration increases by reason of the destruction of organic matters and transformation ammonium nitrogen into nitrate–ion |
| Phosphate-ion | 29 | 19 | Efficient is 34.5% |
| Sulfates | 14 | 2 | Efficient is 85.7% |
| Chlorides | 4.8 | 2.8 | Efficient is 41.7% |
| pH | 6.04 | 7.33 | The environment became more neutral |
| WWS Properties | WWS Form, Processing Method | MW Power | Duration MW | Description of Results | [Ref./No] |
|---|---|---|---|---|---|
| Structure change | Activated sludge; a mixture of activated sludge and raw sediment | 450–900 W | 1–10 min | Sediment’s structure changes: the flakes firstly expand than collapse | [25,34,36] |
| Temperature | Activated sludge; a mixture of activated sludge and raw sediment | 300–900 W | 1–10 min | Rise in temperature and rapid boiling at 100 °C | [25,34,36] |
| Resistivity (fluidity) | Mixed sediments | 800 W | 3–4 min | Decrease in viscosity and resistivity by 5 times | [25,34,36,37] |
| Raw sediment, mixed sediments | 550 W | 6 min | A decrease of 73–84% from the original value for raw sediment and a mixture of sediments | [38] | |
| Humidity | Activated sludge, raw sediment, and mixed sediments | 450–800 W | 1–8 min | Humidity decreases by 2–3% | [34,36] |
| Moisture output | Mixture of sediments; activated sludge with the addition of an acid solution and heating to 100 °C | 600–900 W | 1–5 min | The time of capillary absorption of the sediment mixture is reduced by 1.2–1.3 times; when the pH changes to 2.0–2.4 of the activated sludge, a reduction of 4 times is achieved | [25,34,36,37,39] |
| Raw sediment, mixed sediment, fermented sediment | 500–600 W | 1–1.5 min | The minimum capillary suction time is achieved only in the first 1–2 min of treatment to a temperature of 60–80 °C, then it begins to increase | [38,40,41] | |
| Compaction (sedimentation) | Activated sludge | 900 W 750 W | 60 s 80 s | The settling velocity increases (the settling time is 24 h) | [25,34,36,37] |
| Mineralisation | A mixture of raw sediment and activated sludge; activated sludge | 800 W | 8 min | Increase in the ash content of the wastewater sludge mixture by 6.1%; intensification of solubilisation of organic matter | [31,32,33,34] |
| Dry matter content in the cake * | Dehydrated municipal sediment | 550 W | 0.5–4 min | In the raw sediment, the dry matter content increases by 8%; in the mixture and in the fermented—by 2%. | [38] |
| Cake * humidity | Dehydrated municipal sediment | 400 W | 2 min | May achieve 28% | [38] |
| Decontamination | Raw sediment, mixed sediments | 800–900 W | 1–8 min | Reducing the content of pathogenic microorganisms by 99% | [14,26,27,31,33,34,37,42,52] |
| Type of an Organic Substance | Sample Volume | Concentration | Oxidizing Agent, Catalyst, pH | MW Power | MW Duration, Temperature | Effect | [Ref./No] |
|---|---|---|---|---|---|---|---|
| Sample was only MW-treated | |||||||
| Ammonia (laboratory installation) | 100 mL | 0.5–12 g/L | Air 1 L/min pH = 11 | 750 W | 3 min 80 °C | D * 98.4–96.1% | [63] |
| Ammonia (pilot plant) | 28,000 mL | 2.4–11 g/L | Air 30 L/min pH = 11.6–12 | 4.8 kW | 60 min 80–100 °C | D * 80% | [63] |
| With an addition of the oxidizer: MW + OX | |||||||
| Naphthalene Disulfonic Acid | 10 mL | 1.0 mmol/L | H2O2 | 300 W | 20 min 30 min 80 °C | D * 90% M ** 50% | [64] |
| Dimethoate (phosphoric compound) | No Data | 0.1 mmol/L | K2S2O8 pH = 6.8 | 750 W | 4 min 100 °C | D * 100% | [22] |
| Perflurooctanic acid | 50 mL | 0.25 mmol/L | Na2S2O8 | 800 W | 240 min 60–130 °C | D * 99.3% M ** 74.3% | [66] |
| The photo-Fenton process: MW + OX + C + UV | |||||||
| Polyacrylamide (PAA) | No Data | 150 mg/L | H2O2/AC pH = 3 | 70 W 490 W | 6 min | D * 20% D * 80% | [61] |
| Pesticides (dimethoate, triazophos, malathion) | 1000 mL | 6.11–31.65 mg/L | H2O2 Fe2+; pH = 5 | 80 W | 120 min 25 °C | M ** 72.1% | [62] |
| Direct photolysis: MW + OX + EDL | |||||||
| Phenol | 50 mL | 200 mg/L | H2O2 | 1000 W | 9 min 30 min 50 °C | D * 90% M ** 95% | [67] |
| Atrazine | 50 mL | 50 mg/L | pH = 6.3 | 900 W | 30 min 30 °C | D * 100% | [68] |
| Photocatalysis: MW + OX + UV + TiO2 | |||||||
| Methylene Blue (aromatic compound) | 50 mL | 100 mg/L | TiO2 load pH = 7 | 900 W | 15 min 100 °C | D * 96% M ** 50% | [69] |
| 2,4–D chlorophenoxyacetic herbicide | 10 mL | 0.04 mmol/L | TiO2 load pH = 4.9 | 700 W | 20 min 200 °C | D * 100% | [70] |
| Bisphenol A (Endocrine disruptor) | 30 mL | 0.1 mM | TiO2 load pH = 6.7 | 1500 W | 90 min 150 °C | M ** 100% | [71] |
| Phenol | 50 mL | 10 mg/L | TiO2/AC | 900 W | 30 min 1000 °C | D * 87% | [72] |
| Atrazine | 50 mL | 20 mg/L | TiO2 nanotubes pH = 8.1 | 900 W | 5 min 20 min | D * 100% M ** 98.5% | [72] |
| Emulsion Type | Water Content | Catalyst (C) | MW Power and Frequency | MW Duration | Results | [Ref./No] |
|---|---|---|---|---|---|---|
| Waste water, sludge and oil-in-water emulsions: sample + C + MW+ 60 min settling | ||||||
| Waste emulsions of the metallurgical industry | No Data | NaOH (6 M) HCl (0.12–0.6 M) | 230–930 W 2.45 GHz | 1–4 min | 65–90% | [84] |
| Waste oil water emulsions after metalworking | 99% | Sea water 20% | 700 W 2.45 GHz | 40 s | 92–93.2% | [85] |
| Slurries, emulsions, and liquid waste of the water-in-oil type: sample + C/none + MW + 50–70 min settling | ||||||
| Crude oil | 20 (60)% | none | 700 W 2.45 GHz | 42 s | Efficiency 1/td 0.085 (0.04) | [82] |
| Crude oil | 40–60% | Sodium Acetate 0.2 M | 360–450 W 2.45 GHz | 2–3 min | 93–100% | [23,86] |
| Crude oil | 50 (20)% | none | 900 W 2.45 GHz | 1–3.5 min 2–4.5 min | 85% 75% | [87] |
| Low-temperature separation of emulsions | ||||||
| Oil Sludge | 40% | none | 100 W 10 GHz | 2–3 min | Visual destruction of oil globules | [90] |
| Oil/petroleum- emulsions | 30% | none | 45 W 0.5 GHz | 10 s | Visual enlargement of water droplets in oil | [83] |
| Indicators | Content of Metal Ions in Decanted Water, mg/L | Increase in Concentration of Metals in Water | |
|---|---|---|---|
| without MW Treatment | Following MW Processing | ||
| Arsenic | <0.002 | 0.004 ± 0.002 | 2 times |
| Nickel | 0.011 ± 0.003 | 0.020 ± 0.006 | 1.8 times |
| Mercury | 0.013 ± 0.008 | 0.017 ± 0.010 | 1.3 times |
| Lead | 0.002 ± 0.001 | 0.002 ± 0.001 | Not observed |
| Chrome (6+) | 0.114 ± 0.0052 | 0.218 ± 0.100 | 1.9 times |
| Sediment | MW Power and Frequency | Temperature | Sample Processing Conditions | Description of Results | [Ref./No] |
|---|---|---|---|---|---|
| Domestic wastewater sludge mixture, ratio 48:52 | 1250 W 2.45 GHz | 96 °C | 500 mL samples heated in a home MW furnace to the boiling point, then subjected to anaerobic digestion in laboratory reactors for 5–18 days; biogas output recorded. | With thermophilic fermentation, the gas output increased by 17–26%. | [99] |
| Dehydrated WWS | 1200 W 2.45 GHz | 80–160 °C | Samples heated and kept at a set temperature for 1 min, then cooled for 25 min. Heating speed 7.5 °C/min. Further, the samples were subjected to anaerobic digestion in laboratory reactors for 5–20 days; biogas output was recorded. | Maximal biogas output: At 160 °C- on the fifth day of the fermentation process. At 120 °C on the tenth day. | [105] |
| Sludge mixture | 300–600 W 2.45 GHz | no data | Microwave pre-treatments were carried out in a semi–pilot MW unit in which the flow rate varied in the range of 5–60 L/h. Next, anaerobic digestion of the nitrogen-treated sludge mixture was carried out at a temperature of 37 degrees. | The biogas production improved by 174–210% (depending on the MW power and irradiated energy) | [106] |
| Sorbent | Sorbent Preparation Process | MW Power | Duration | Temperature | Description of Results | [Ref./No] |
|---|---|---|---|---|---|---|
| Peat | MW heat | 60–600 W | 60 min | No data | Oil capacity 2.5–2.73 g/g. With increasing power, the adsorption of iodine increases by 1.2–1.4 times (from 115 to 150 mg/g), and for methylene blue it decreases by 2 times (from 55 to 28 mg/g) | [113] |
| Peat | MW heat | 900 W | 12 min | 450 °C | Iodine adsorption activity increased from 11.4% to 19.1% | [114] |
| Brown coal | MW heat | 900 W | 22.5 min | 315 °C | Iodine adsorption activity increased from 18.0% to 34.9% | [114] |
| Montmorillonite | MW heat | 800 W | 4 min | 154 °C | Water vapor adsorption increased from 0.67 to 3.66 mmol/g | [115] |
| Pine sawdust | Grinding, drying and MW heat | 600 W | 2 min | 40 °C | Increase in the sorption capacity for petroleum products by 3.7–4 times for initial concentrations of less than 5 mg/L and by 1.2 times for initial concentrations of 16–35 mg/L | [110] |
| Rice husk | Combustion in the MW furnace | No data | 288 h 384 h | 500 °C 800 °C | Removal of petroleum products: 78% 98% | [116] |
| Device/Installation | Application Domain: Treatment of | Type of Action | Parameters: Power N, Frequency F, Throughput Q, Temperature T | Patent Number, (Year) | MW Process Setting | [Ref./No] | ||
|---|---|---|---|---|---|---|---|---|
| WW | WWS | 1 * | 2 * | |||||
| Sewage treatment installation | + | - | - | + | No Data | RU 116 851 (2012) | Combined UV and MW water disinfection | [120] |
| Industrial and domestic sewage handling equipment | - | + | + | - | T 430–1000 °C f 2.5 GHz | RU 2 552 259 (2015) | Decomposition of waste under the combined influence of thermal and electromagnetic fields without oxygen access | [121] |
| Waste incineration chamber | - | + | + | - | T 1200–1400 °C | RU 2 573 137 (2016) | Heating of the disposed waste to the combustion temperature | [122] |
| Sewage sludge MW treatment plant | - | + | - | + | N 5440 W Q 1.37 t/h | RU 2 582 415 (2016) | Disinfection of industrial, domestic, and agricultural sewage sludge | [123] |
| Wastewater and sediments MW treatment and decontamination device | + | + | - | + | N 2000 W f 2.45 GHz Q 0.1 m3/h T 50–85 °C | RU 2 693 783 (2019) | MW pretreatment of sediments prior to anaerobic fermentation | [124] |
| Anaerobic processing plant for liquid organic waste | - | + | + | - | T 60–70 °C | RU 2 687 415 (2019) | MW pretreatment of WWS prior to anaerobic fermentation | [125] |
| Electromagnetic phase separation system for oil–water emulsion | - | + | - | + | f 2.45 GHz | RU 2 710 181 (2019) | Destruction of oil–water emulsions (sludge from oil fields and oil refineries) | [126] |
| Wastewater treatment by irradiation with ultra-high-frequency waves and ultraviolet light plant | + | - | - | + | No Data | RU 193 171 U1 (2019) | Combined UV and MW wastewater disinfection | [127] |
| MW mobile technological complex treatment of acid mine water | + | - | + | - | N 1000 W f 1.38 GHz Q 150 m3/h | RU 2 739 259 (2020) | MW is used to transfer ionic and molecular components to the condensed phase and remove them from the treated water | [128] |
| No | Practical Application Field | Promising Areas of Practical Application of MW in Wastewater and Wastewater Sludge Treatment Technologies |
|---|---|---|
| 1. | Municipal local services | Wastewater treatment of all types (reduction of organic matter concentration, improvement of coagulation processes, and decomposition of chemicals), wastewater sludge treatment (reduction of volumes and organic matter content), an increase of biogas production during wastewater sludge fermentation on an urban spit, disinfection of all forms of wastewater and wastewater sludge, and intensification of pyrolysis and production of secondary raw materials |
| 2. | Production of drinking water | Water disinfection. Treatment of water sludge in order to reduce the volume, reducing the organic component |
| 3. | Agricultural industry | Improvement of biogas output in the fermentation of manure, sewage sludge, and other liquid industrial waste; decontamination of all types of wastewater and sediments; and decomposition of pesticides in surface wastewater |
| 4. | Medical institutions, pharmaceutical industry | Disinfection of all types of wastewater and waste; decomposition of medicines in wastewater and sediments |
| 5. | Metallurgical industry and machine-building | Industrial wastewater coagulation, extraction of metals from wastewater and sediments |
| 6. | Oil and petroleum industry | Demulsification of oil-water emulsions and oil-containing industrial wastewater, decomposition and reduction of oil sludge volumes |
| 7. | Dairy industry | Organic matter oxidation in industrial wastewater, sludge treatment to reduce volumes and organic matter, and decontamination of all types of wastewater and sludge |
| 8. | Chemical industry | New substances synthesis; chemical decomposition in industrial wastewater and sediments |
| 9. | Textile industry | Dye decomposition in industrial wastewater, improving the coagulation of contaminants; decontamination of all types of wastewater and sludge |
| 10. | Recycling and disposal of industrial waste | Decomposition of chemical, organic, and radioactive substances; reduction of liquid waste; and decontamination |
| 11. | Sorbent manufacture | Modification, regeneration, and activation of phyto–sorbents for sewage purification |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vialkova, E.; Obukhova, M.; Belova, L. Microwave Irradiation in Technologies of Wastewater and Wastewater Sludge Treatment: A Review. Water 2021, 13, 1784. https://doi.org/10.3390/w13131784
Vialkova E, Obukhova M, Belova L. Microwave Irradiation in Technologies of Wastewater and Wastewater Sludge Treatment: A Review. Water. 2021; 13(13):1784. https://doi.org/10.3390/w13131784
Chicago/Turabian StyleVialkova, Elena, Marina Obukhova, and Larisa Belova. 2021. "Microwave Irradiation in Technologies of Wastewater and Wastewater Sludge Treatment: A Review" Water 13, no. 13: 1784. https://doi.org/10.3390/w13131784
APA StyleVialkova, E., Obukhova, M., & Belova, L. (2021). Microwave Irradiation in Technologies of Wastewater and Wastewater Sludge Treatment: A Review. Water, 13(13), 1784. https://doi.org/10.3390/w13131784






