Explaining Variation in Abundance and Species Diversity of Avian Cestodes in Brine Shrimps in the Salar de Atacama and Other Chilean Wetlands
Abstract
1. Introduction
2. Materials and Methods
2.1. Brine Shrimp Populations and Study Sites
2.2. Sampling
2.3. Parasitological Examination
2.4. Bird Data
2.5. Statistical Analyses
3. Results
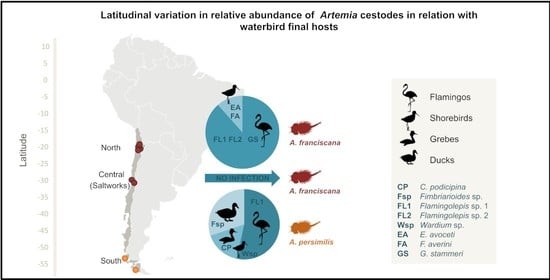
3.1. Taxonomic Composition of Cestode Communities in Artemia in Chile
3.2. General Infection Levels
3.3. Cestode Distribution in A. franciscana
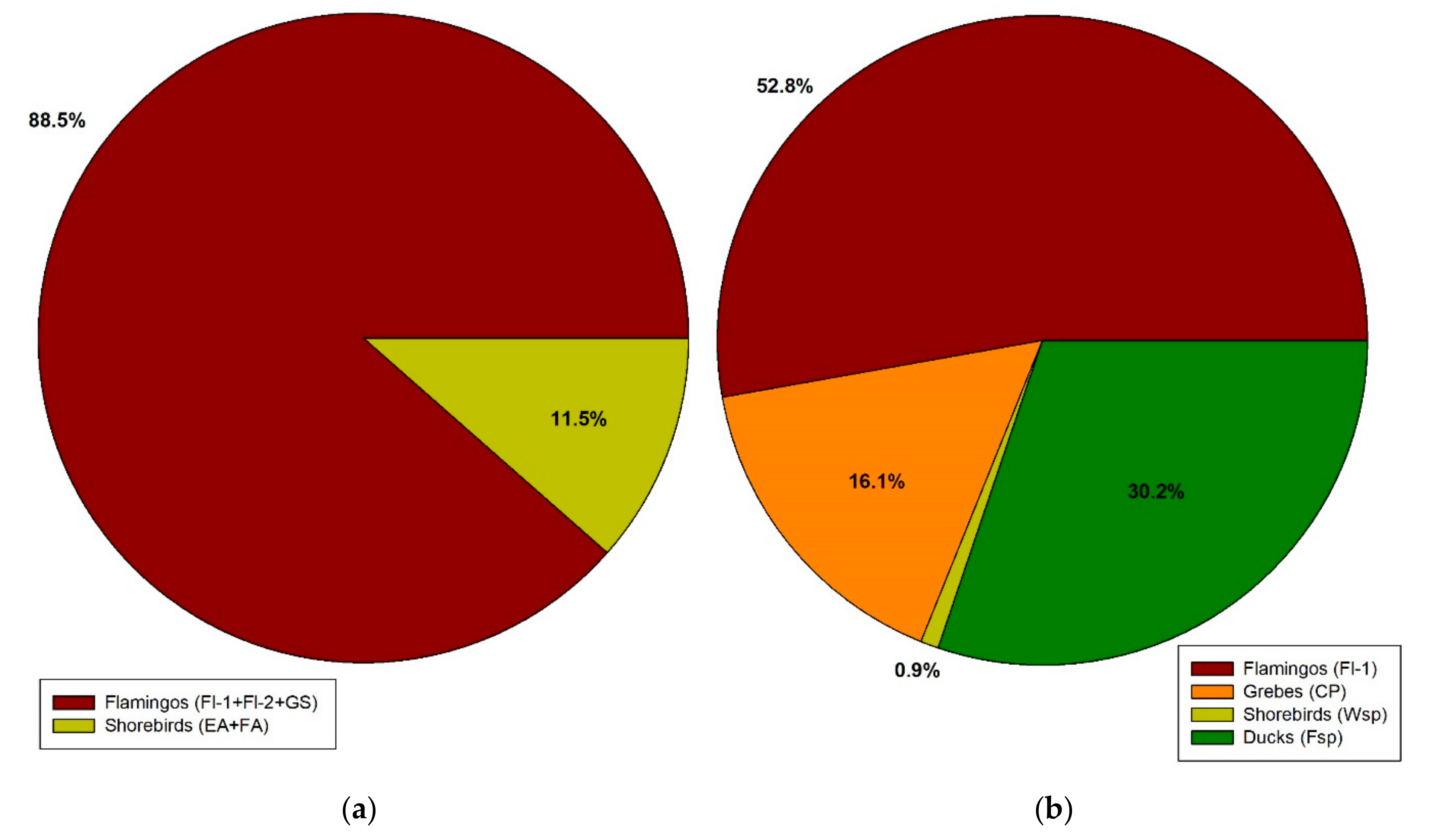
3.4. Bird Data
3.5. Wetland Size and Salinity Effects
3.6. Sex Effects
4. Discussion
4.1. Latitudinal, Regional, and Local Effects on Cestode Communities
4.2. The Effects of Season and Host Sex on Cestode Populations
4.3. Relationship with North American Cestodes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brooks, D.R.; Hoberg, E.P. Triage for the Biosphere: The Need and Rationale for Taxonomic Inventories and Phylogenetic Studies of Parasites. Comp. Parasitol. 2000, 67, 1–25. [Google Scholar]
- Poulin, R. Latitudinal gradients in parasite diversity: Bridging the gap between temperate and tropical areas. Neotrop. Helminthol. 2010, 4, 169–177. [Google Scholar]
- Preisser, W. Latitudinal gradients of parasite richness: A review and new insights from helminths of cricetid rodents. Ecography 2019, 42, 1315–1330. [Google Scholar] [CrossRef]
- Poulin, R.; Leung, T.L.F. Latitudinal gradient in the taxonomic composition of parasite communities. J. Helminthol. 2011, 85, 228–233. [Google Scholar] [CrossRef]
- Sehgal, R.N. Manifold habitat effects on the prevalence and diversity of avian blood parasites. Int. J. Parasitol. Parasites Wildl. 2015, 4, 421–430. [Google Scholar] [CrossRef]
- Merino, S.; Moreno, J.; Vásquez, R.A.; Martínez, J.; Sánchez-Monsálvez, I.; Estades, C.F.; Ippi, S.; Sabat, P.; Rozzi, R.; Mcgehee, S. Haematozoa in forest birds from southern Chile: Latitudinal gradients in prevalence and parasite lineage richness. Austral Ecol. 2008, 33, 329–340. [Google Scholar] [CrossRef]
- Clark, N.J. Phylogenetic Uniqueness, Not Latitude, Explains the Diversity of Avian Blood Parasite Communities Worldwide. Glob. Ecol. Biogeogr. 2018, 27, 744–755. [Google Scholar] [CrossRef]
- Cuevas, E.; Vianna, J.A.; Botero-Delgadillo, E.; Doussang, D.; González-Acuña, D.; Barroso, O.; Rozzi, R.; Vásquez, R.A.; Quirici, V. Latitudinal gradients of haemosporidian parasites: Prevalence, diversity and drivers of infection in the Thorn-tailed Rayadito (Aphrastura Spinicauda). Int. J. Parasitol. Parasites Wildl. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Fecchio, A.; Bell, J.A.; Bosholn, M.; Vaughan, J.A.; Tkach, V.V.; Lutz, H.L.; Cueto, V.R.; Gorosito, C.A.; González-Acuña, D.; Stromlund, C.; et al. An inverse latitudinal gradient in infection probability and phylogenetic diversity for Leucocytozoon blood parasites in New World birds. J. Anim. Ecol. 2020, 89, 423–435. [Google Scholar] [CrossRef]
- Redón, S.; Vasileva, G.P.; Georgiev, B.B.; Gajardo, G. Exploring parasites in extreme environments of high conservational importance: Artemia franciscana (Crustacea: Branchiopoda) as intermediate host of avian cestodes in Andean hypersaline lagoons from Salar de Atacama, Chile. Parasitol. Res. 2020, 119, 3377–3390. [Google Scholar] [CrossRef]
- Jarecka, L. Development of Hymenolepis arctowskii Jarecka et Ostas, 1984 (Cestoda, Hymenolepididae) in the intermediate host Branchinecta gaini Daday (Branchiopoda) of the Antarctic. Acta Parasitol. Pol. 1984, 29, 337–342. [Google Scholar]
- Bondarenko, S.; Kontrimavichus, V. Life-cycles of cestodes of the genus Branchiopodataenia Bondarenko & Kontrimavichus, 2004 (Cestoda: Hymenolepididae) from gulls in Chukotka. Syst. Parasitol. 2004, 57, 191–199. [Google Scholar] [PubMed]
- Caira, J.N.; Jensen, K.; Georgiev, B.B.; Kuchta, R.; Littlewood, D.T.J.; Mariaux, J.; Scholz, T.; Tkach, V.; Waeschenbach, A. An overview of tapeworms from vertebrate bowels of the earth. In Planetary Biodiversity Inventory (2008–2017): Tapeworms from Vertebrate Bowels of the Earth; Caira, J.N., Jensen, K., Eds.; Natural History Museum, University of Kansas: Lawrence, KS, USA, 2017; Volume 25, pp. 1–20. [Google Scholar]
- Mariaux, J.; Tkach, V.V.; Vasileva, G.P.; Waeschenbach, A.; Beveridge, I.; Dimitrova, Y.D.; Haukisalmi, V.; Greiman, S.E.; Littlewood, D.T.J.; Makarikov, A.A.; et al. Cyclophyllidea van Beneden in Braun, 1900. In Planetary Biodiversity Inventory (2008–2017): Tapeworms from Vertebrate Bowels of the Earth; Caira, J.N., Jensen, K., Eds.; Natural History Museum, University of Kansas: Lawrence, KS, USA, 2017; Volume 25, pp. 77–148. [Google Scholar]
- Redón, S.; Vasileva, G.P.; Georgiev, B.B.; Gajardo, G. First report of cestode infection in the crustacean Artemia persimilis from Southern Chilean Patagonia and its relation with the Neotropical aquatic birds. PeerJ 2019, 7, e7395. [Google Scholar] [CrossRef] [PubMed]
- Guagliardo, S.E.; Graff, M.E.; Gigola, G.; Tanzola, R.D. Biological aspects of the life history of Confluaria podicipina (Cestoda, Hymenolepididae) from a hypersaline pampasic lagoon. Pan-Am. J. Aquat. Sci. 2020, 15, 54–63. [Google Scholar]
- Poulin, R. Parasite biodiversity revisited: Frontiers and constraints. Int. J. Parasitol. 2014, 44, 581–589. [Google Scholar] [CrossRef]
- Sánchez, M.I.; Nikolov, P.N.; Georgieva, D.D.; Georgiev, B.B.; Vasileva, G.P.; Pankov, P.; Paracuellos, M.; Lafferty, K.D.; Green, A.J. High prevalence of cestodes in Artemia spp. throughout the annual cycle: Relationship with abundance of avian final hosts. Parasitol. Res. 2013, 112, 1913–1923. [Google Scholar] [CrossRef]
- Redón, S.; Amat, F.; Sánchez, M.I.; Green, A.J. Comparing cestode infections and their consequences for host fitness in two sexual branchiopods: Alien Artemia franciscana and native A. salina from syntopic-populations. PeerJ 2015, 3, e1073. [Google Scholar] [CrossRef]
- Redón, S.; Green, A.J.; Georgiev, B.B.; Vasileva, G.P.; Amat, F. Influence of developmental stage and sex on infection of the American brine shrimp Artemia franciscana Kellogg, 1906 by avian cestodes in Ebro Delta salterns, Spain. Aquat. Invasions 2015, 10, 415–423. [Google Scholar] [CrossRef]
- Gajardo, G.; Redón, S. Hypersaline Lagoons from Chile, the Southern Edge of the World. In Lagoon Environments around the World-A Scientific Perspective; Manning, A.J., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Gajardo, G.; Redón, S. Andean hypersaline lakes in the Atacama Desert, northern Chile: Between lithium exploitation and unique biodiversity conservation. Conserv. Sci. Pract. 2019, 1, e94. [Google Scholar] [CrossRef]
- Gajardo, G.; Colihueque, N.; Parraguez, M.; Sorgeloos, P. International study on Artemia LVIII. Morphologic differentiation and reproductive isolation of Artemia populations from South America. Int. J. Salt Lake Res. 1998, 7, 133–151. [Google Scholar] [CrossRef]
- Gajardo, G.; Crespo, J.; Triantafyllidis, A.; Tzika, A.; Baxevanis, A.D.; Kappas, I.; Abatzopoulos, T.J. Species identification of Chilean Artemia populations based on mitochondrial DNA RFLP analysis. J. Biogeogr. 2004, 31, 547–555. [Google Scholar] [CrossRef]
- Amat, F.; Cohen, R.G.; Hontoria, F.; Navarro, J.C. Further evidence and characterization of Artemia franciscana populations in Argentina. J. Biogeogr. 2004, 31, 1–15. [Google Scholar] [CrossRef]
- De los Ríos, P. Review of the biogeography of Artemia Leach, 1819 (Crustacea: Anostraca) in Chile. Int. J. Artemia Biol. 2013, 3, 64–67. [Google Scholar]
- Yasuhara, M.; Hunt, G.; Dowsett, H.J.; Robinson, M.M.; Stoll, D.K. Latitudinal species diversity gradient of marine zooplankton for the last three million years. Ecol. Lett. 2012, 15, 1174–1179. [Google Scholar] [CrossRef]
- Sebastián-González, E.; Green, A.J. Habitat use by waterbirds in relation to pond size, water depth, and isolation: Lessons from a restoration in southern Spain. Restor. Ecol. 2014, 22, 311–318. [Google Scholar] [CrossRef]
- Caziani, S.M.; Derlindati, E.J.; Tálamo, A.; Sureda, A.L.; Trucco, C.E.; Nicolossi, G. Waterbird richness in altiplano wetlands of northwestern Argentina. Waterbirds 2001, 24, 103–117. [Google Scholar] [CrossRef]
- Browne, R.A.; Sallee, S.E.; Grosch, D.S.; Segreti, W.O.; Purser, S.M. Partitioning genetic and environmental components of reproduction and lifespan in Artemia. Ecology 1984, 65, 949–960. [Google Scholar] [CrossRef]
- Marazuela, M.A.; Vazquez-Sune, E.; Ayora, C.; Garcia-Gil, A.; Palma, T. Hydrodynamics of salt flat basins: The Salar de Atacama example. Sci. Total Environ. 2019, 651, 668–683. [Google Scholar] [CrossRef]
- Caziani, S.M.; Olivio, O.R.; Ramirez, E.R.; Romano, M.; Derlindati, E.J.; Tálamo, A.; Ricalde, D.; Quiroga, C.; Contreras, J.P.; Valqui, M.; et al. Seasonal distribution, abundance, and nesting of Puna, Andean, and Chilean Flamingos. Condor 2007, 109, 276–287. [Google Scholar] [CrossRef]
- Derlindati, E.J.; Romano, M.C.; Cruz, N.N.; Barison, C.; Arengo, F.; Barberis, I.M. Seasonal activity patterns and abundance of Andean flamingo (Phoenicoparrus andinus) at two contrasting wetlands in Argentina. Ornitol. Neotrop. 2014, 25, 317–331. [Google Scholar]
- Vilina, Y.A.; Cofre, H.L.; Silva-Garcia, C.; Garcia, M.D.; Perez-Friedenthal, C. Effects of El Niño on abundance and breeding of Black-Necked Swans on El Yali wetland in Chile. Waterbirds 2002, 25, 123–127. [Google Scholar]
- Fuentes-González, N.; Gajardo, G. A Glimpse to Laguna de Los Cisnes, a field laboratory and Natural Monument in the Chilean Patagonia. Lat. Am. J. Aquat. Res. 2017, 45, 491–495. [Google Scholar] [CrossRef]
- Georgiev, B.B.; Sánchez, M.I.; Green, A.J.; Nikolov, P.N.; Vasileva, G.P.; Mavrodieva, R.S. Cestodes from Artemia parthenogenetica (Crustacea, Branchiopoda) in the Odiel Marshes, Spain: A systematic survey of cysticercoids. Acta Parasitol. 2005, 50, 105–117. [Google Scholar]
- Redón, S.; Berthelemy, N.J.; Mutafchiev, Y.; Amat, F.; Georgiev, B.B.; Vasileva, G.P. Helminth parasites of Artemia franciscana (Crustacea: Branchiopoda) in the Great Salt Lake, Utah: First data from the native range of this invader of European wetlands. Folia Parasitol. 2015, 62, 030. [Google Scholar] [CrossRef]
- Bush, A.O.; Lafferty, K.D.; Lotz, J.M.; Shostak, A.W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 1997, 83, 575–583. [Google Scholar] [CrossRef]
- Geobiota. Cambios y Mejoras en la Operación Minera del Salar de Atacama. Región Antofagasta. Informe de Seguimiento Ambiental (GEOB.SQM611). 2021. Available online: https://snifa.sma.gob.cl/ (accessed on 5 June 2021).
- eBird. eBird Basic Dataset; Cornell Lab of Ornithology: Ithaca, NY, USA, 2021. [Google Scholar]
- Poulin, R.; Mouritsen, K.N. Large-scale determinants of trematode infections in intertidal gastropods. Mar. Ecol. Prog. Ser. 2003, 254, 187–198. [Google Scholar] [CrossRef]
- Thieltges, D.W.; Ferguson, M.A.D.; Jones, C.S.; Noble, L.R.; Poulin, R. Biogeographical patterns of marine larval trematode parasites in two intermediate snail hosts in Europe. J. Biogeogr. 2009, 36, 1493–1501. [Google Scholar] [CrossRef]
- Andrade, B.; Grau, S. La Laguna de Cáhuil, Un Ejemplo de Estuario Estacional En Chile Central. Rev. Geogr. Norte Gd. 2005, 33, 59–72. [Google Scholar]
- Georgiev, B.B.; Angelov, A.; Vasileva, G.P.; Sánchez, M.I.; Hortas, F.; Mutafchiev, Y.; Pankov, P.; Green, A.J. Larval helminths in the invasive American brine shrimp Artemia franciscana throughout its annual cycle. Acta Parasitol. 2014, 59, 380–389. [Google Scholar] [CrossRef]
- Boyle, T.P.; Caziani, S.M.; Waltermire, R.G. Landsat TM inventory and assessment of waterbird habitat in the southern altiplano of South America. Wetl. Ecol. Manag. 2004, 12, 563–573. [Google Scholar] [CrossRef]
- Gutiérrez, J.S.; Soriano-Redondo, A. Wilson’s Phalaropes can double their feeding rate by associating with Chilean flamingos. Ardea 2018, 106, 131–139. [Google Scholar] [CrossRef]
- Hurlbert, S.H.; López, M.; Keith, J.O. Wilson’s phalarope in the Central Andes and its interaction with the Chilean flamingo. Rev. Chil. Hist. Nat. 1984, 57, 47–57. [Google Scholar]
- Garay, G.L.; Johnson, W.E.; Franklin, W.L. Relative abundance of aquatic birds and their use of wetlands in the Patagonia of southern Chile. Rev. Chil. Hist. Nat. 1991, 64, 127–137. [Google Scholar]
- Garay, G.; Guineo, O.; Mutschke, E.; Ríos, C. Tamaño, estructura y distribución estacional de poblaciones de aves acuáticas en el Fiordo Última Esperanza y Canal Señoret, Región de Magallanes. An. Inst. Patagon. 2008, 36, 33–43. [Google Scholar] [CrossRef]
- Gibbons, J.; Vilina, Y.A.; Cárcamo, J. Distribución y abundancia de Cisne Coscoroba (Coscoroba coscoroba), Cisne de Cuello Negro (Cygnus melancoryphus) y del Flamenco Chileno (Phoenicopterus chilensis) en la Región de Magallanes. An. Inst. Patagon. 2007, 35, 53–58. [Google Scholar]
- Ramírez, I. Catastro de Fauna Monumento Natural Laguna de Los Cisnes; Departamento de Áreas Protegidas y Medio Ambiente, Ministerio de Agricultura: Santiago, Chile, 2009; 24p.
- Tobar, C.; Rau, J.R.; Iriarte, A.; Villalobos, R.; Lagos, N.; Cursach, J.; Díaz, C.; Fuentes, N.; Gantz, A. Composition, diversity and size of diatoms consumed by the Andean Flamingo (Phoenicoparrus andinus) in salar de Punta Negra, Antofagasta Region, northern Chile. Ornitol. Neotrop. 2012, 23, 243–250. [Google Scholar]
- Polla, W.M.; Di Pasquale, V.; Rasuk, M.C.; Barberis, I.; Romano, M.; Manzo, R.A.; Paggi, J.C.; Farias, M.E.; Contreras, M.; Devercelli, M. Diet and feeding selectivity of the Andean Flamingo Phoenicoparrus andinus and Chilean Flamingo Phoenicopterus chilensis in lowland wintering areas. Wildfowl 2018, 68, 3–29. [Google Scholar]
- Medina, G.R.; Goenaga, J.; Hontoria, F.; Cohen, G.; Amat, F. Effects of temperature and salinity on prereproductive life span and reproductive traits of two species of Artemia (Branchiopoda, Anostraca) from Argentina: Artemia franciscana and A. persimilis. Hydrobiologia 2007, 579, 41–53. [Google Scholar] [CrossRef]
- Van Stappen, G. Zoogeography. In Artemia: Basic and Applied Biology; Springer: Dordrecht, The Netherlands, 2002; pp. 171–224. [Google Scholar]
- Gajardo, G.; Beardmore, J.A. The brine shrimp Artemia: Adapted to critical life conditions. Front. Physiol. 2012, 3, 1–8. [Google Scholar] [CrossRef]
- Vignatti, A.M.; Cabrera, G.C.; Pilati, A.; Echaniz, S.A. Biology of Artemia persimilis Piccinelli and Prosdocimi, 1968 (Crustacea: Anostraca) at the highest salinities reported for the species under natural conditions. Int. J. Artemia Biol. 2014, 4, 38–43. [Google Scholar]
- De los Ríos-Escalante, P.; Gajardo, G. Potential heterogeneity in crustacean zooplankton assemblages in southern Chilean saline lakes. Braz. J. Biol. 2010, 70, 1031–1032. [Google Scholar] [CrossRef][Green Version]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Suppression of Artemia spp. (Crustacea, Anostraca) populations by predators in the Crimean hypersaline lakes: A review of the evidence. Int. Rev. Hydrobiol. 2019, 104, 5–13. [Google Scholar] [CrossRef]
- Bucher, E.H.; Curto, E. Influence of long-term climatic changes on breeding of the Chilean flamingo in Mar Chiquita, Córdoba, Argentina. Hydrobiologia 2012, 697, 127–137. [Google Scholar] [CrossRef]
- Maksimova, A.P. Morphology and life cycle on the cestode Confluaria podicipina (Cestoda: Hymenolepididae). Parazitologiya 1981, 15, 325–331. (In Russian) [Google Scholar]
- Redón, S.; Amat, F.; Hontoria, F.; Vasileva, G.P.; Nikolov, P.N.; Georgiev, B.B. Participation of metanauplii and juvenile individuals of Artemia parthenogenetica (Branchiopoda) in the circulation of avian cestodes. Parasitol. Res. 2011, 108, 905–912. [Google Scholar] [CrossRef]
- Amarouyache, M.; Derbal, F.; Kara, M.H. The parasitism of Flamingolepis liguloides (Gervais, 1847) (Cestoda, Hymenolepididae) in Artemia salina (Crustacea, Branchiopoda) in two saline lakes in Algeria. Acta Parasitol. 2009, 54, 330–334. [Google Scholar] [CrossRef]
- Lin, Q.; Xu, L.; Hou, J.; Liu, Z.; Jeppesen, E.; Han, B.P. Responses of trophic structure and zooplankton community to salinity and temperature in Tibetan lakes: Implication for the effect of climate warming. Water Res. 2017, 124, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Pavón-Jordán, D.; Clausen, P.; Dagys, M.; Devos, K.; Encarnacao, V.; Fox, A.D.; Frost, T.; Gaudard, C.; Hornman, M.; Keller, V.; et al. Habitat- and species-mediated short- and long-term distributional changes in waterbird abundance linked to variation in European winter weather. Divers. Distrib. 2019, 25, 225–239. [Google Scholar] [CrossRef]
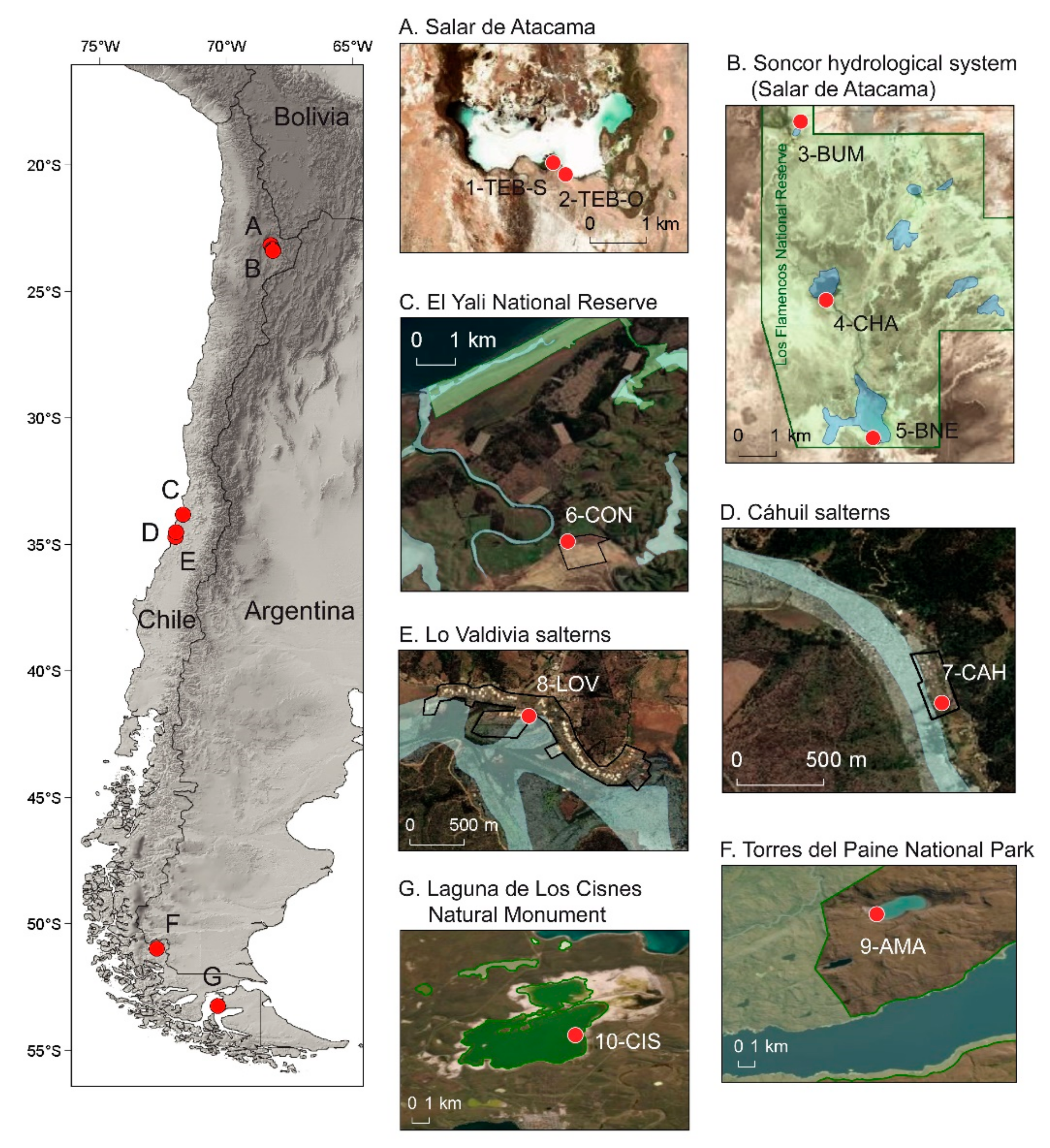
| Locality | Coordinates | Sampling Date | Temperature (°C) | Salinity (g/L) | pH | Depth (cm) | Size (ha) | Artemia Species (n) |
|---|---|---|---|---|---|---|---|---|
| North Chile—II Region-Antofagasta | ||||||||
| Laguna Tebenquiche-Origen TEB-O (Salar de Atacama, El Loa Province) | 23°08′24.7″ S, 68°15′15.2″ W | 3 November 2017 2 June 2018 | 20.6 21.0 | 320 180 | 7.4 7.8 | 20 30 | 217 | AF * AF (206) |
| Laguna Tebenquiche-Sendero TEB-S (Salar de Atacama, El Loa Province) | 23°08′18.9″ S, 68°15′22.2″ W | 3 November 2017 2 June 2018 | 16.0 23.9 | 230 240 | 7.7 7.4 | 20 30 | 217 | AF (200) AF (148) |
| Estanque Burro Muerto BUM (Salar de Atacama, El Loa Province) | 23°17′16.5″ S, 68°10′26.4″ W | 1 June 2018 | 13.6 | 60 | 7.8 | 50 | 0.01 | AF (200) |
| Laguna Chaxas CHA (Salar de Atacama, El Loa Province) | 23°19′49.9″ S, 68°10′02.9″ W | 5 November 2017 31 May 2018 | 21.9 18.6 | 119 80 | 7.9 7.8 | 10 10 | 39 | AF * AF (150) |
| Laguna Barros Negros BNE (Salar de Atacama, El Loa Province) | 23°21′51.8″ S, 68°09′20.8″ W | 5 November 2017 31 May 2018 | 34.8 15.8 | 200 132 | 7.6 7.7 | 25 25 | 121 | - AF (220) |
| Central Chile—V Region-Valparaiso | ||||||||
| Salinas de El Convento CON (Santo Domingo, San Antonio Province) | 33°48′00″ S, 71°42′53.4″ W | 4 February 2018 | 25.4 | 320 | 7.6 | 5 | 97 | AF * |
| Central Chile—VI Region- Libertador Bernardo O´Higgins | ||||||||
| Salinas de Cáhuil CAH (Pichilemu, Cardenal Caro Province) | 34°29′43.7″ S, 71°59′56″ W | 2 February 2018 | 27.6 | 130 | 8.6 | 30 | 7.5 | AF (200) |
| Salinas de Lo Valdivia (Paredones, Cardenal Caro Province) | 34°41′49.2″ S, 72°00′53.7″ W | 3 February 2018 | 28.0 | 150 | 7.9 | 35 | 69 | AF (200) |
| South Chile—XII Region Magallanes and Chilean Antarctica | ||||||||
| Laguna Amarga AMA (Última Esperanza Province) | 50°58′22.3″ S, 72º43′58.85″ W | 26 November 2017 18 April 2018 | 10.6 - | 86 81 | 9.2 - | 25–50 - | 487 | AP (500) 2 AP (200) 1 |
| Laguna de Los Cisnes (Porvenir, Tierra del Fuego Island) | 53°14′48.82″ S, 70°21′8.33″ W | 29 November 2017 17 April 2018 | 13.7 - | 55 51 | 9.1 - | 15–25 - | 1561 | AP (200) 1 AP (200) 1 |
| North (Latitude 23° S) AF-Salar de Atacama | Centre (Latitude 34° S) AF-Coastal Saltworks | South (Latitude 50° S) AP-Amarga Lagoon | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Family Cestode Species | Cysts | P% | MI ± SE | MA ± SE | Cysts | P% | MI ± SE | MA ± SE | Cysts | P% | MI ± SE | MA ± SE |
| Hymenolepididae | ||||||||||||
| CP | 0 | 0.0 | - | 0.00 | 0 | 0.0 | - | 0.00 | 5 | 1.7 | 1.00 ± 0.00 | 0.02 ± 0.01 |
| Fsp | 0 | 0.0 | - | 0.00 | 0 | 0.0 | - | 0.00 | 3 | 1.0 | 1.00 ± 0.00 | 0.01 ± 0.01 |
| Fl-1 | 49 | 3.1 | 1.4 | 0.044 ± 0.01 | 0 | 0.0 | - | 0.00 | 0 | 0.0 | 0.00 | 0.00 ± 0.00 |
| Fl-2 | 3 | 0.3 | 1.00 ± 0.00 | 0.003 ± 0.002 | 0 | 0.0 | - | 0.00 | 0 | 0.0 | 0.00 | 0.00 ± 0.00 |
| Dilepididae | ||||||||||||
| EA | 2 | 0.2 | 1.00 ± 0.00 | 0.002 ± 0.001 | 0 | 0.0 | - | 0.00 | 0 | 0.0 | 0.00 | 0.00 |
| FA | 5 | 0.4 | 1.00 ± 0.00 | 0.004 ± 0.002 | 0 | 0.0 | - | 0.00 | 0 | 0.0 | 0.00 | 0.00 |
| Progynotaeniidae | ||||||||||||
| GS | 2 | 0.2 | 1.00 ± 0.00 | 0.002 ± 0.002 | 0 | 0.0 | - | 0.00 | 0 | 0.0 | 0.00 | 0.00 |
| Total | 61 | 4.1 | 1.33 ± 0.24 | 0.054 ± 0.013 | 0 | 0.0 | - | 0.00 | 8 | 2.7 | 1.00 ± 0.00 | 0.027 ± 0.01 |
| No. Artemia examined | 1124 | 400 | 300 | |||||||||
| No. infected | 46 | 0 | 8 | |||||||||
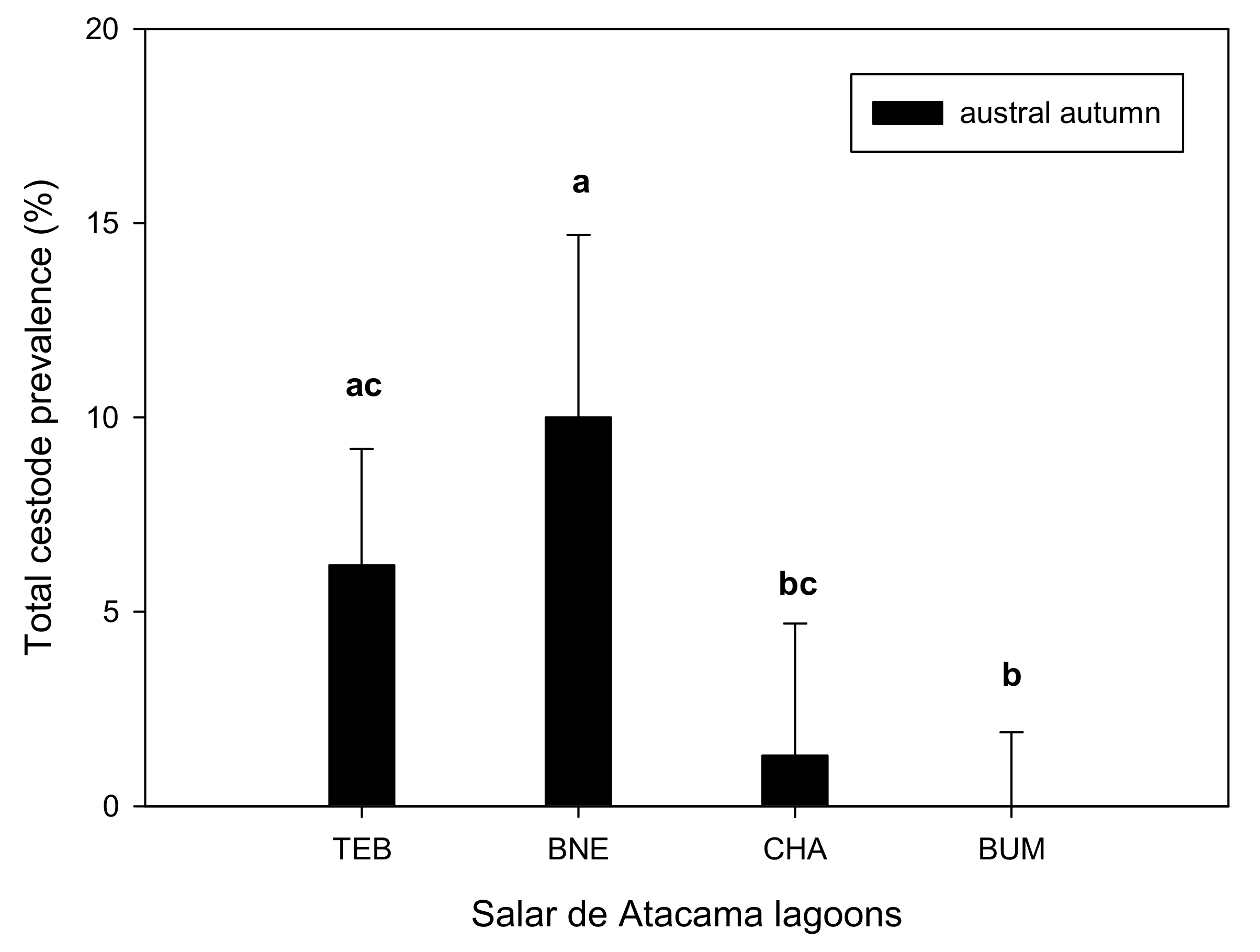
| Localities | |||||||
|---|---|---|---|---|---|---|---|
| Cestode Species | TEB S = 180–240 g/L | BNE S = 132 g/L | CHA S = 80 g/L | BUM S = 60 g/L | χ2 | K-W (H) | |
| Fl-1 | P% | 4.0 ab | 8.6 b | 1.3 a | 0.0 a | 24.572 ** | |
| MI ± SE | 1.93 ± 0.71 | 1.05 ± 0.05 | 1.00 ± 0.00 | - | 3.909 | ||
| MA ± SE | 0.08 ± 0.03 ac | 0.09 ± 0.02 a | 0.013 ± 0.01 bc | 0.00 b | 24.388 ** | ||
| RA (%) | 75 | 86.4 | 100 | 0.0 | |||
| Fl-2 | P% | 0.8 | 0.0 | 0.0 | 0.0 | 2.710 | |
| MI ± SE | 1.00 ± 0.00 | - | - | - | no test | ||
| MA ± SE | 0.009 ± 0.01 | 0.00 | 0.00 | 0.00 | 4.841 | ||
| RA (%) | 8.3 | 0.0 | 0.0 | 0.0 | |||
| EA | P% | 0.3 | 0.5 | 0.0 | 0.0 | 1.557 | |
| MI ± SE | 1.00 | 1.00 | - | - | 0.000 | ||
| MA ± SE | 0.003 ± 0.003 | 0.005 ± 0.00 | 0.00 | 0.00 | 1.407 | ||
| RA (%) | 2.8 | 4.5 | 0.0 | 0.0 | |||
| FA | P% | 1.4 | 0.0 | 0.0 | 0.0 | 5.047 | |
| MI ± SE | 1.00 ± 0.00 | - | - | - | no test | ||
| MA ± SE | 0.01 ± 0.006 | 0.00 | 0.00 | 0.00 | 8.086 * | ||
| RA (%) | 13.9 | 0.0 | 0.0 | 0.0 | |||
| GS | P% | 0.0 | 0.9 | 0.0 | 0.0 | 3.904 | |
| MI ± SE | - | 1.00 ± 0.00 | - | - | no test | ||
| MA ± SE | 0.00 | 0.009 ± 0.01 | 0.00 | 0.00 | 6.407 | ||
| RA (%) | 0.0 | 9.1 | 0.0 | 0.0 | |||
| Total | P% | 6.2 ac | 10 a | 1.3 bc | 0.0 b | 27.563 ** | |
| MI ± SE | 1.64 ± 0.5 | 1.05 ± 0.05 | 1.00 ± 0.00 | - | 2.730 | ||
| MA ± SE | 0.10 ± 0.04 a | 0.11 ± 0.02 a | 0.013 ± 0.01 b | 0.00 b | 27.437 ** | ||
| No. cestodes | 36 | 23 | 2 | 0 | |||
| No. Artemia examined | 354 | 220 | 150 | 200 | |||
| Cestode Species | TEB-S S = 240 g/L | TEB-O S = 180 g/L | Z | U | p-Value | |
|---|---|---|---|---|---|---|
| Fl-1 | P% | 0.0 | 6.8 | 2.961 | 0.003 | |
| MI ± SE | - | 1.93 ± 0.71 | no test | - | ||
| MA ± SE | 0.00 | 0.013 ± 0.06 | 14,208 | 0.001 | ||
| RA (%) | 0.0 | 87.1 | ||||
| Fl-2 | P% | 0.0 | 1.5 | 0.917 | 0.359 | |
| MI ± SE | - | 1.00 ± 0.00 | no test | - | ||
| MA ± SE | 0.00 | 0.015 ± 0.01 | 15,022 | 0.195 | ||
| RA (%) | 0.0 | 9.7 | ||||
| EA | P% | 0.0 | 0.5 | −0.139 | 0.890 | |
| MI ± SE | - | 1.00 | no test | - | ||
| MA ± SE | 0.00 | 0.005 ± 0.005 | 15,170 | 0.579 | ||
| RA (%) | 0.0 | 3.2 | ||||
| FA | P% | 3.4 | 0.0 | 2.210 | 0.027 | |
| MI ± SE | 1.00 ± 0.00 | - | no test | - | ||
| MA ± SE | 0.025 ± 0.01 | 0.00 | 14,729 | 0.011 | ||
| RA (%) | 100 | 0.0 | ||||
| Total | P% | 3.4 | 8.3 | 1.656 | 0.098 | |
| MI ± SE | 1.00 ± 0.00 | 1.8 ± 0.64 | 32.5 | 0.325 | ||
| MA ± SE | 0.025 ± 0.01 | 0.15 ± 0.06 | 14,491 | 0.028 | ||
| SR | 0.03 ± 0.015 | 0.09 ± 0.02 | 14,498 | 0.036 | ||
| No. cestodes | 5 | 31 | ||||
| No. Artemia examined | 148 | 206 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Redón, S.; Gajardo, G.; Vasileva, G.P.; Sánchez, M.I.; Green, A.J. Explaining Variation in Abundance and Species Diversity of Avian Cestodes in Brine Shrimps in the Salar de Atacama and Other Chilean Wetlands. Water 2021, 13, 1742. https://doi.org/10.3390/w13131742
Redón S, Gajardo G, Vasileva GP, Sánchez MI, Green AJ. Explaining Variation in Abundance and Species Diversity of Avian Cestodes in Brine Shrimps in the Salar de Atacama and Other Chilean Wetlands. Water. 2021; 13(13):1742. https://doi.org/10.3390/w13131742
Chicago/Turabian StyleRedón, Stella, Gonzalo Gajardo, Gergana P. Vasileva, Marta I. Sánchez, and Andy J. Green. 2021. "Explaining Variation in Abundance and Species Diversity of Avian Cestodes in Brine Shrimps in the Salar de Atacama and Other Chilean Wetlands" Water 13, no. 13: 1742. https://doi.org/10.3390/w13131742
APA StyleRedón, S., Gajardo, G., Vasileva, G. P., Sánchez, M. I., & Green, A. J. (2021). Explaining Variation in Abundance and Species Diversity of Avian Cestodes in Brine Shrimps in the Salar de Atacama and Other Chilean Wetlands. Water, 13(13), 1742. https://doi.org/10.3390/w13131742