Fluoride Polluted Groundwaters in Calabria Region (Southern Italy): Natural Source and Remediation
Abstract
1. Introduction
2. Materials and Methods
2.1. Site of Origin of F-Rich Samples
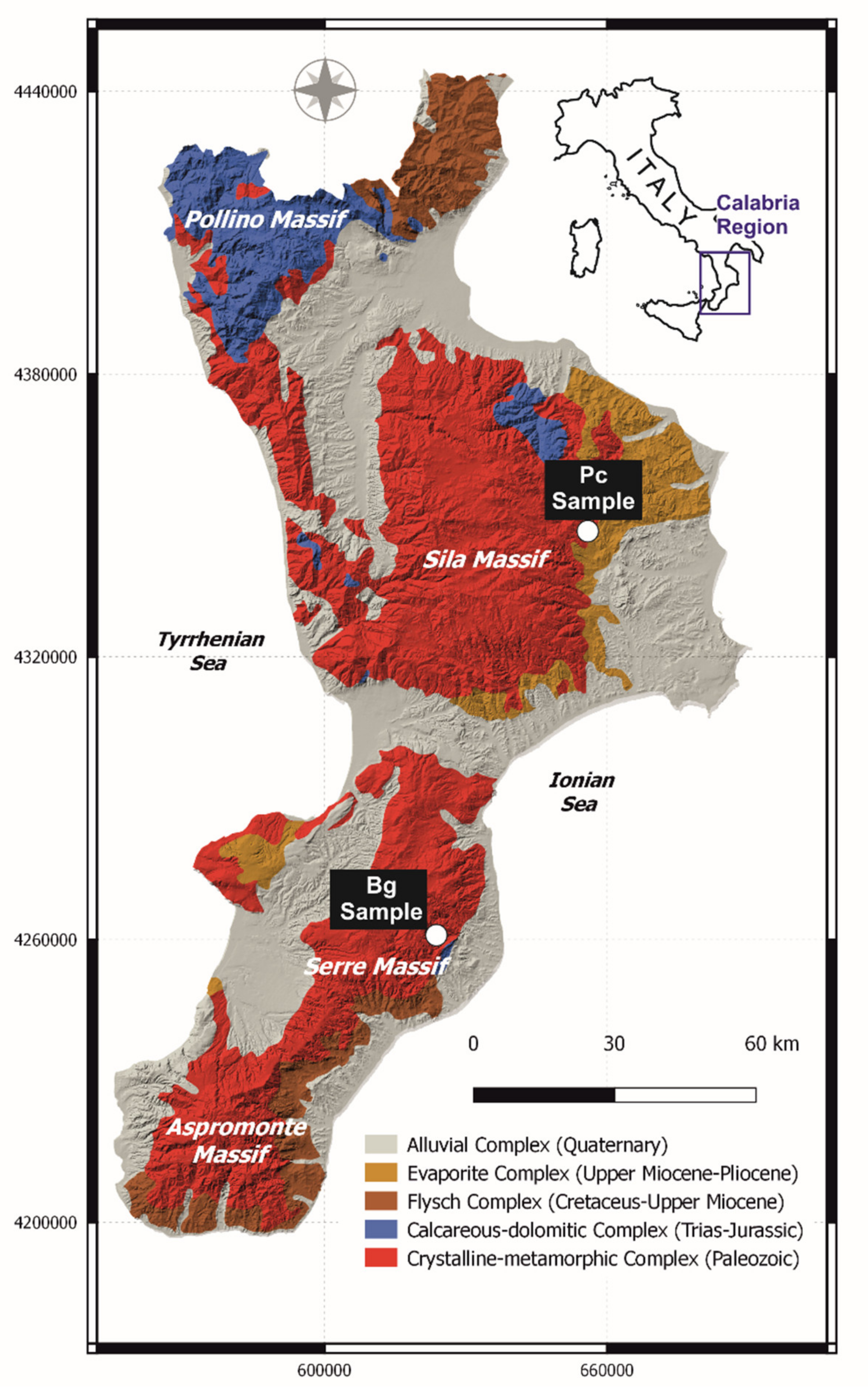
2.2. Water Sampling and Analyses
2.3. Experimental Set-Up for NF/RO Tests
3. Results and Discussion
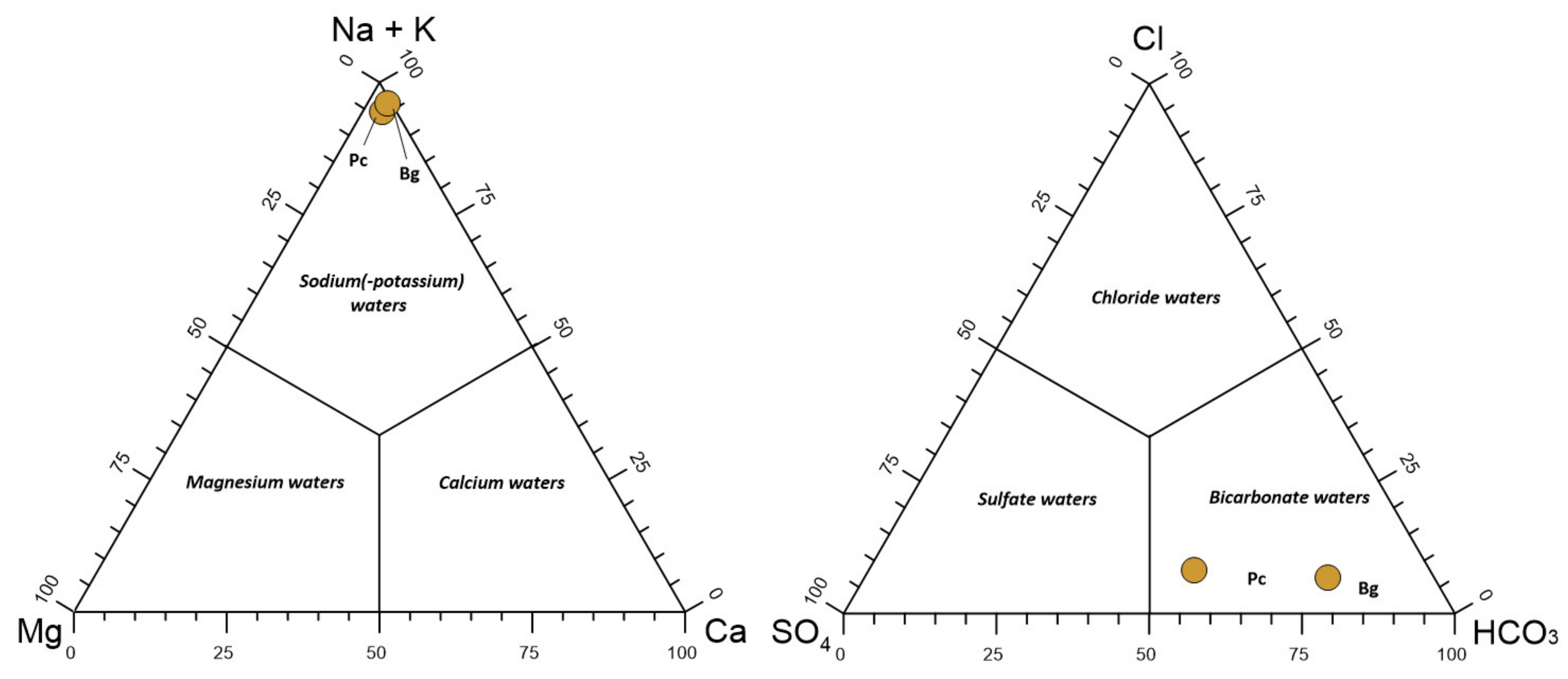
3.1. Water Characterization Before Membrane Treatment Tests
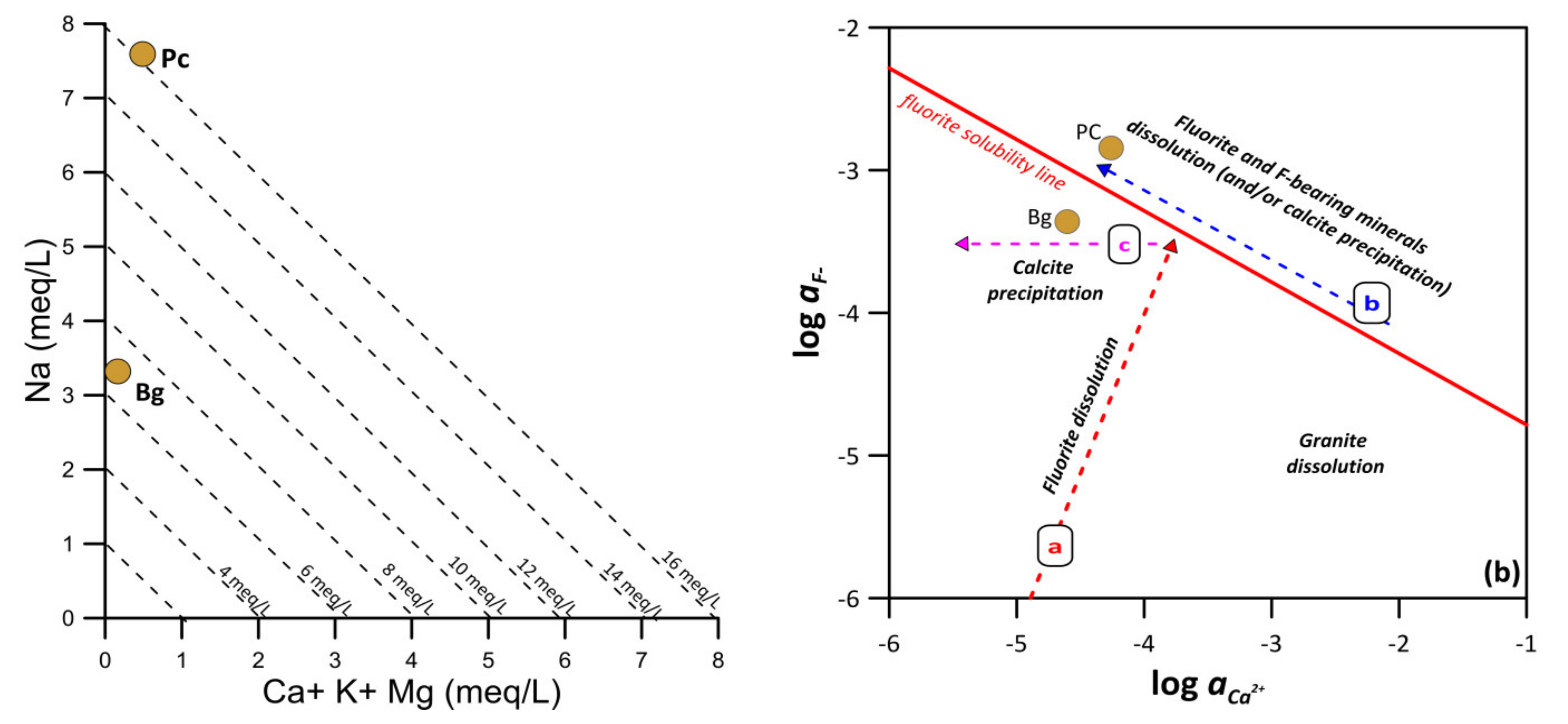
3.2. Source of Fluoride
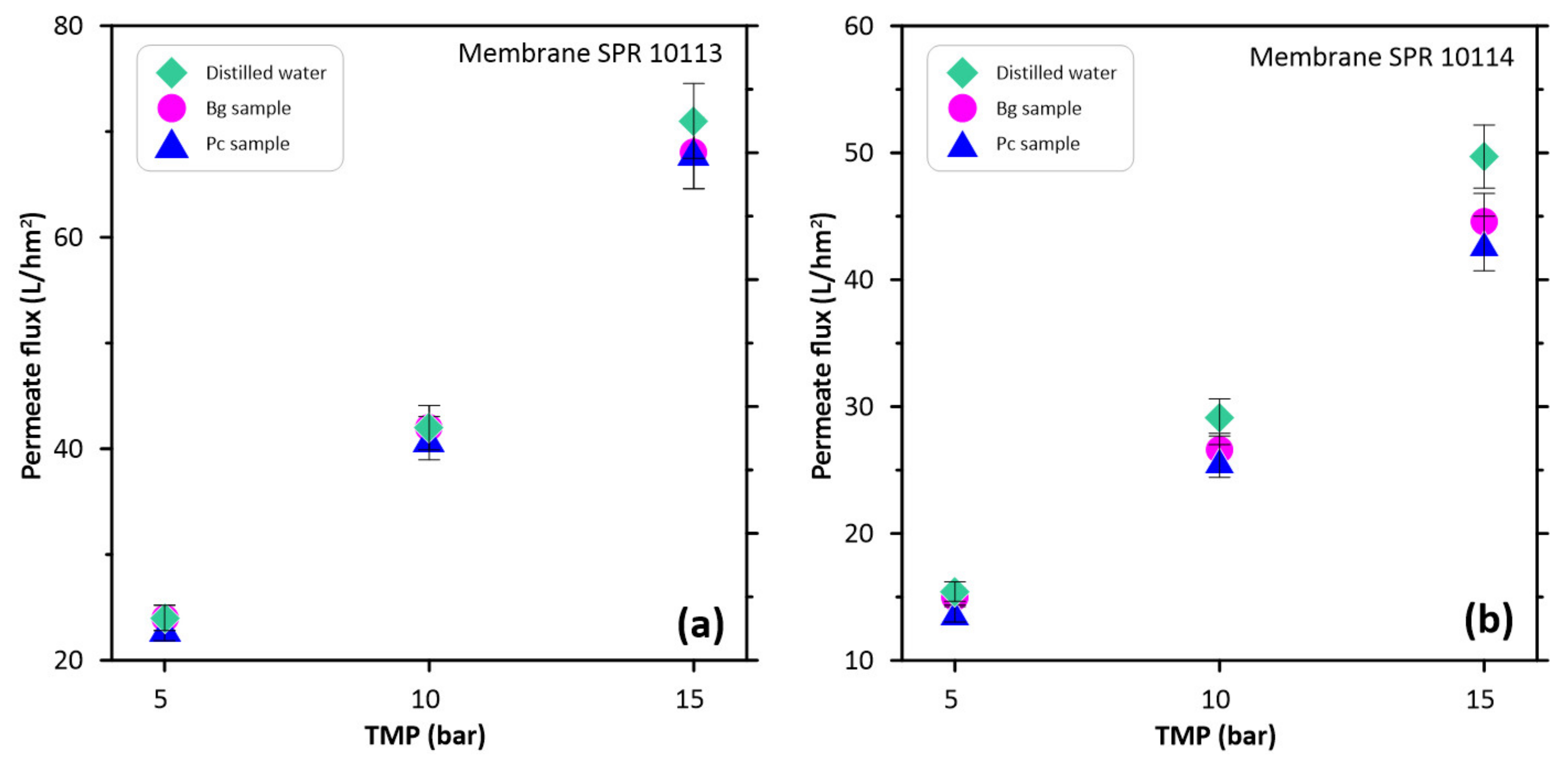
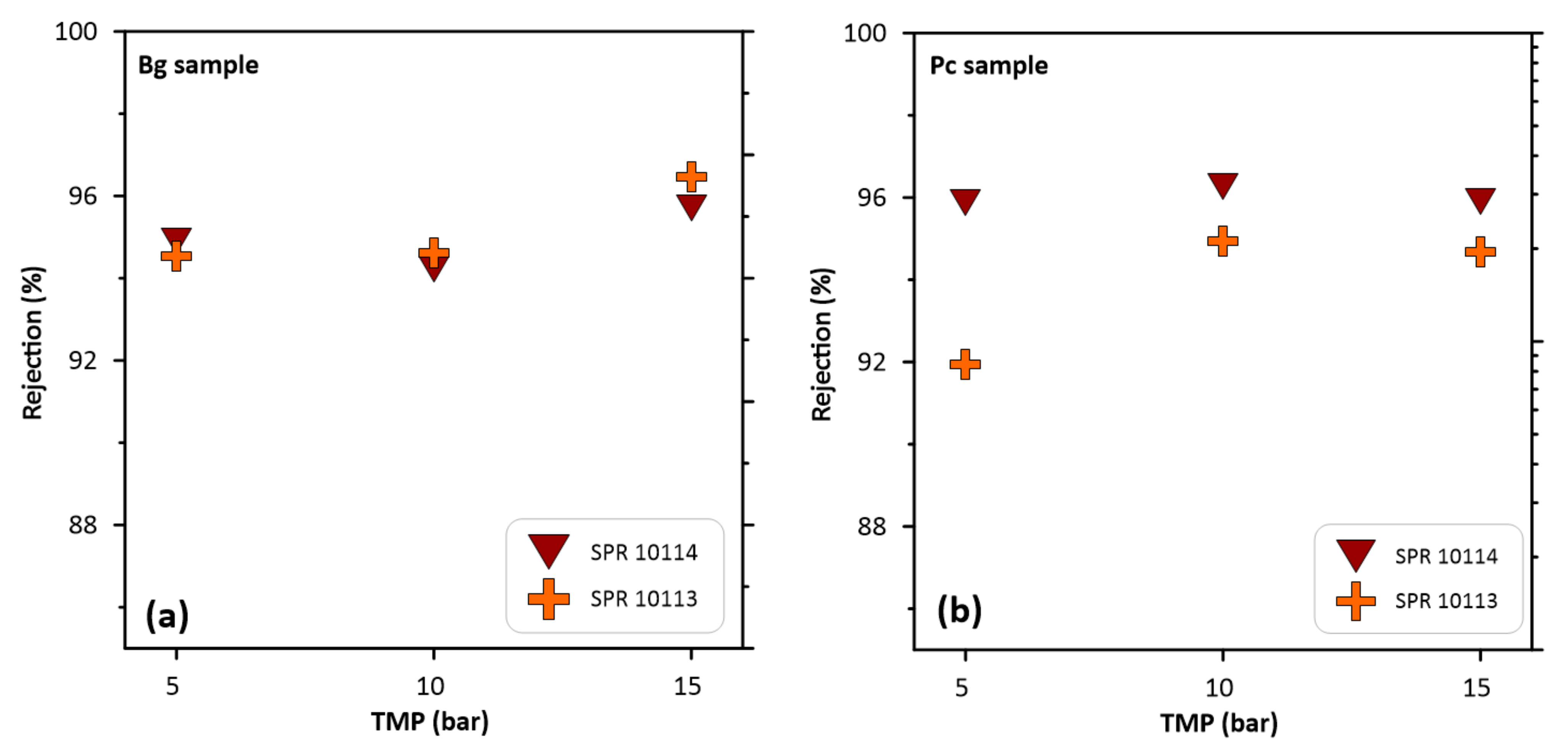
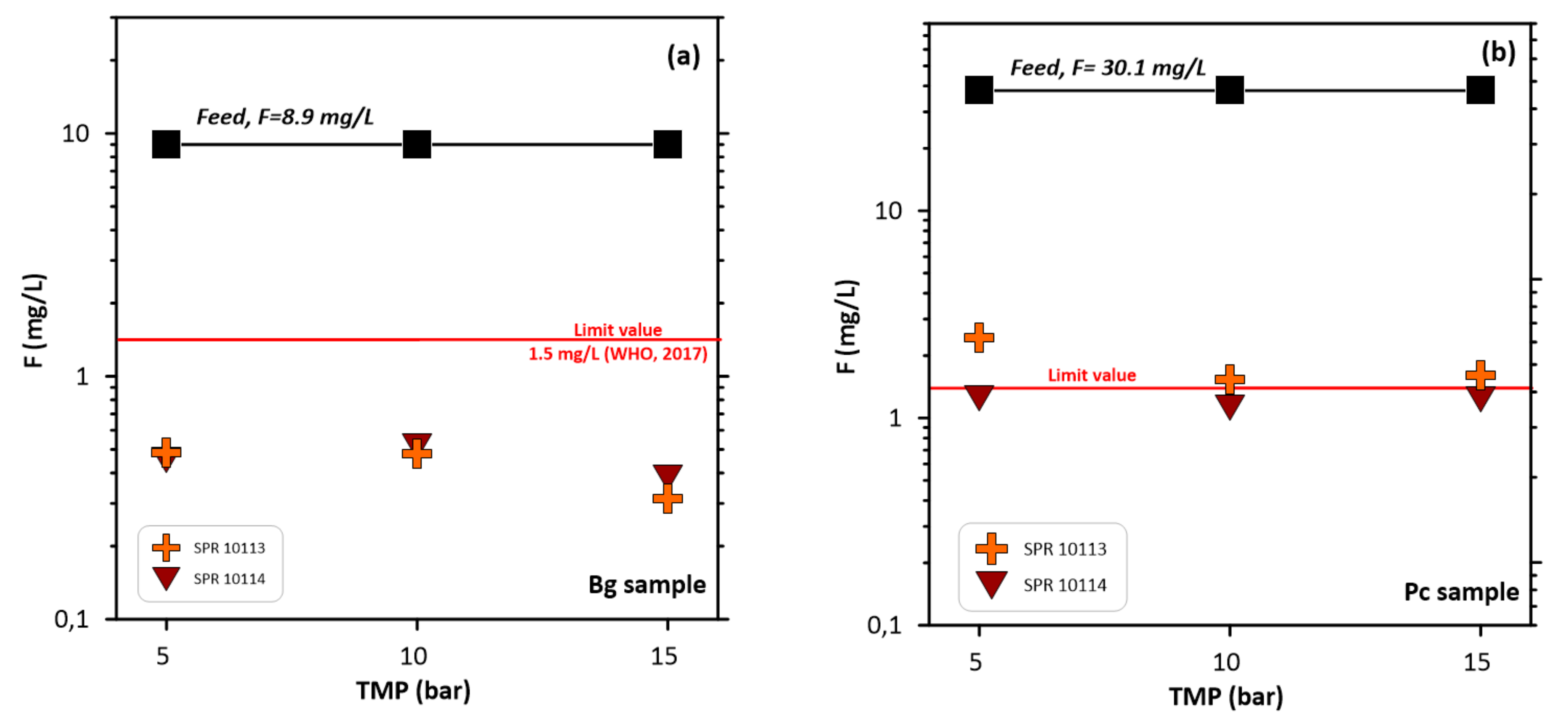
3.3. Permeate Flux and Fluorine Rejection
3.4. Comparison with Literature Data
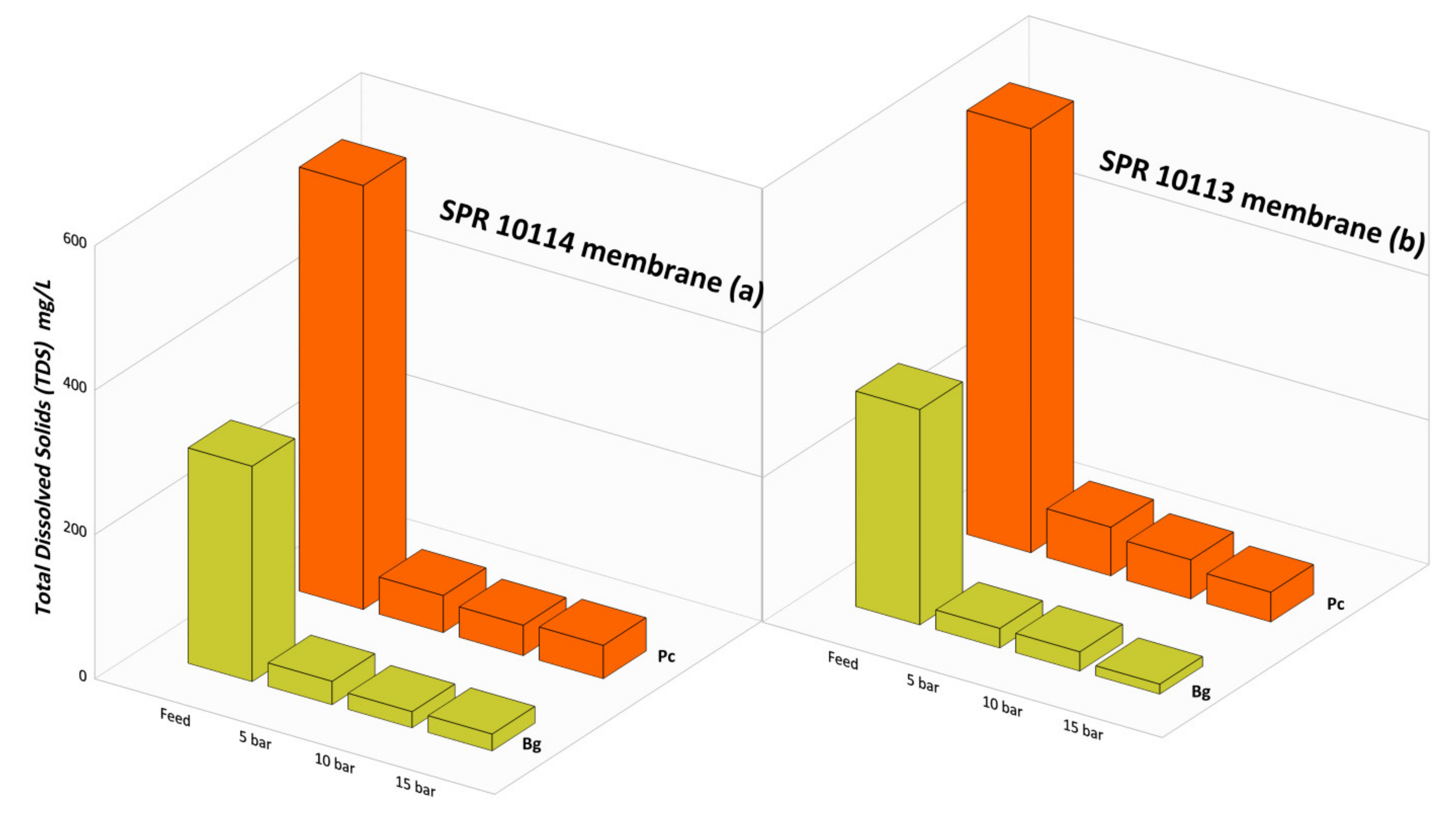
3.5. Total Dissolved Solid (TDS) Rejection
4. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Edmunds, W.M.; Smedley, P.L. Fluoride in natural waters. In Essentials of Medical Geology; Springer: Dordrecht, The Netherlands, 2013; pp. 311–336. [Google Scholar]
- World Health Organization. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating First Addendum, 4th ed + 1st add. 2017. Available online: https://apps.who.int/iris/handle/10665/254637 (accessed on 4 May 2021).
- Chae, G.T.; Yun, S.T.; Mayer, B.; Kim, K.H.; Kim, S.Y.; Kwon, J.S.; Kim, K.; Koh, Y.K. Fluorine geochemistry in bedrock groundwater of South Korea. Sci. Total Environ. 2007, 385, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Fawell, J.; Bailey, K.; Chilton, J.; Dahi, E.; Magara, Y. Fluoride in Drinking-Water; IWA Publishing: London, UK, 2006. [Google Scholar]
- Nordstrom, D.K.; Jenne, E.A. Fluorite solubility equilibria in selected geothermal waters. Geochim. Cosmochim. Acta 1997, 41, 175–188. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Tossou, Y.Y.J.; Orban, P.; Gesels, J.; Otten, J.; Yessoufou, S.; Boukari, M.; Brouyère, S. Hydrogeochemical mechanisms governing the mineralization and elevated fluoride F-contents in Precambrian crystalline aquifer groundwater in central Benin, Western Africa. Environ. Earth Sci. 2017, 76, 1–14. [Google Scholar] [CrossRef]
- Apambire, W.B.; Boyle, D.R.; Michel, F.A. Geochemistry, genesis, and health implications of fluoriferous groundwaters in the upper regions of Ghana. Environ. Geol. 1997, 33, 13–24. [Google Scholar] [CrossRef]
- Fuoco, I.; Figoli, A.; Criscuoli, A.; Brozzo, G.; De Rosa, R.; Gabriele, B.; Apollaro, C. Geochemical modeling of chromium release in natural waters and treatment by RO/NF membrane processes. Chemosphere 2020, 254, 126696. [Google Scholar] [CrossRef] [PubMed]
- Figoli, A.; Fuoco, I.; Apollaro, C.; Chabane, M.; Mancuso, R.; Gabriele, B.; De Rosa, R.; Vespasiano, G.; Barca, D.; Criscuoli, A. Arsenic-contaminated groundwaters remediation by nanofiltration. Sep. Purif. Technol. 2020, 238, 116461. [Google Scholar] [CrossRef]
- Velizarov, S.; Crespo, J.G.; Reis, M.A. Removal of inorganic anions from drinking water supplies by membrane bio/processes. Rev. Environ. Sci. Bio/Technol. 2004, 3, 361–380. [Google Scholar] [CrossRef]
- Kut, K.M.K.; Sarswat, A.; Srivastava, A.; Pittman, C.U., Jr.; Mohan, D. A review of fluoride in african groundwater and local remediation methods. Groundw. Sustain. Dev. 2016, 2, 190–212. [Google Scholar] [CrossRef]
- Velizarov, S.; Oehmen, A.; Reis, M.A.; Crespo, J.G. Electro-membrane Processes for the Removal of Trace Toxic Ions from Water. In Membrane Technologies for Water Treatment: Removal of Toxic Trace Elements with Emphasis on Arsenic, Fluoride and Uranium; CRC Press: Boca Raton, FL, USA, 2016; pp. 73–87. [Google Scholar]
- Figoli, A.; Criscuoli, A. (Eds.) Sustainable Membrane Technology for Water and Wastewater Treatment; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Bey, S.; Criscuoli, A.; Simone, S.; Figoli, A.; Benamor, M.; Drioli, E. Hydrophilic PEEK-WC hollow fibre membrane contactors for chromium (VI) removal. Desalination 2011, 283, 16–24. [Google Scholar] [CrossRef]
- Bouhadjar, S.I.; Kopp, H.; Britsch, P.; Deowan, S.A.; Hoinkis, J.; Bundschuh, J. Solar powered nanofiltration for drinking water production from fluoride-containing groundwater–A pilot study towards developing a sustainable and low-cost treatment plant. J. Environ. Manag. 2019, 231, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Richards, B.S.; Schäfer, A.I. Renewable energy powered membrane technology: Case study of St. Dorcas borehole in Tanzania demonstrating fluoride removal via nanofiltration/reverse osmosis. Sep. Purif. Technol. 2016, 170, 445–452. [Google Scholar] [CrossRef]
- Shen, J.; Schäfer, A.I. Factors affecting fluoride and natural organic matter (NOM) removal from natural waters in Tanzania by nanofiltration/reverse osmosis. Sci. Total Environ. 2015, 527, 520–529. [Google Scholar] [CrossRef]
- Nasr, A.B.; Charcosset, C.; Amar, R.B.; Walha, K. Defluoridation of water by nanofiltration. J. Fluor. Chem. 2013, 150, 92–97. [Google Scholar] [CrossRef]
- Chakrabortty, S.; Roy, M.; Pal, P. Removal of fluoride from contaminated groundwater by cross flow nanofiltration: Transport modeling and economic evaluation. Desalination 2013, 313, 115–124. [Google Scholar] [CrossRef]
- Tahaikt, M.; Haddou, A.A.; El Habbani, R.; Amor, Z.; Elhannouni, F.; Taky, M.; Kharif, M.; Boughriba, A.; Hafsi, M.; Elmidaoui, A. Comparison of the performances of three commercial membranes in fluoride removal by nanofiltration. Continuous operations. Desalination 2008, 225, 209–219. [Google Scholar] [CrossRef]
- Dolar, D.; Košutić, K.; Vučić, B. RO/NF treatment of wastewater from fertilizer factory—Removal of fluoride and phosphate. Desalination 2011, 265, 237–241. [Google Scholar] [CrossRef]
- Paugam, L.; Diawara, C.K.; Schlumpf, J.P.; Jaouen, P.; Quéméneur, F. Transfer of monovalent anions and nitrates especially through nanofiltration membranes in brackish water conditions. Sep. Purif. Technol. 2004, 40, 237–242. [Google Scholar] [CrossRef]
- Van Dijk, J.P.; Bello, M.; Brancaleoni, G.P.; Cantarella, G.; Costa, V.; Frixa, A.; Golfetto, F.; Merlini, S.; Riva, M.; Torricelli, S.; et al. A regional structural model for the northern sector of the Calabrian Arc (southern Italy). Tectonophysics 2000, 324, 267–320. [Google Scholar] [CrossRef]
- Ietto, F.; Perri, F.; Cella, F. Geotechnical and landslide aspects in weathered granitoid rock masses (Serre Massif, southern Calabria, Italy). Catena 2016, 145, 301–315. [Google Scholar] [CrossRef]
- Angì, G.; Cirrincione, R.; Fazio, E.; Fiannacca, P.; Ortolano, G.; Pezzino, A. Metamorphic evolution of preserved Hercynian crustal section in the Serre Massif (Calabria–Peloritani Orogen, southern Italy). Lithos 2010, 115, 237–262. [Google Scholar] [CrossRef]
- Apollaro, C.; Accornero, M.; Marini, L.; Barca, D.; De Rosa, R. The impact of dolomite and plagioclase weathering on the chemistry of shallow groundwaters circulating in a granodiorite-dominated catchment of the Sila Massif (Calabria, Southern Italy). Appl. Geochem. 2009, 24, 957–979. [Google Scholar] [CrossRef]
- Perri, F.; Scarciglia, F.; Apollaro, C.; Marini, L. Characterization of granitoid profiles in the Sila Massif (Calabria, southern Italy) and reconstruction of weathering processes by mineralogy, chemistry, and reaction path modeling. J. Soils Sediments 2015, 15, 1351–1372. [Google Scholar] [CrossRef]
- Bertolani, M.; Foggia, F. La formazione Kinzigitica della Sila Greca. Boll. Soc. Geol. It. 1975, 94, 329–345. [Google Scholar]
- Allocca, V.; Celico, F.; Celico, P.; De Vita, P.; Fabbrocino, S.; Mattia, S.; Monacelli, G.; Musilli, I.; Piscopo, V.; Scalise, A.R.; et al. Note Illustrative della Carta Idrogeologica dell’Italia Meridionale; Istituto Poligrafico e Zecca dello Stato: Rome, Italy, 2007; p. 211. ISBN 88-448-0223-6. [Google Scholar]
- Apollaro, C.; Tripodi, V.; Vespasiano, G.; De Rosa, R.; Dotsika, E.; Fuoco, I.; Critelli, S.; Muto, F. Chemical, isotopic and geotectonic relations of the warm and cold waters of the Galatro and Antonimina thermal areas, southern Calabria, Italy. Mar. Pet. Geol. 2019, 109, 469–483. [Google Scholar] [CrossRef]
- Apollaro, C.; Buccianti, A.; Vespasiano, G.; Vardè, M.; Fuoco, I.; Barca, D.; Bloise, A.; Miriello, D.; Cofone, F.; Servidio, A.; et al. Comparative geochemical study between the tap waters and the bottled mineral waters in Calabria (Southern Italy) by compositional data analysis (CoDA) developments. Appl. Geochem. 2019, 107, 19–33. [Google Scholar] [CrossRef]
- Apollaro, C.; Fuoco, I.; Bloise, L.; Calabrese, E.; Marini, L.; Vespasiano, G.; Muto, F. Geochemical Modeling of Water-Rock Interaction Processes in the Pollino National Park. Geofluids 2021, 2021. [Google Scholar] [CrossRef]
- Apollaro, C.; Perri, F.; Le Pera, E.; Fuoco, I.; Critelli, T. Chemical and minero-petrographical changes on granulite rocks affected by weathering processes. Front. Earth Sci. 2019, 13, 247–261. [Google Scholar] [CrossRef]
- Apollaro, C.; Caracausi, A.; Paternoster, M.; Randazzo, P.; Aiuppa, A.; De Rosa, R.; Fuoco, I.; Mongelli, G.; Muto, F.; Vanni, E.; et al. Fluid geochemistry in a low-enthalpy geothermal field along a sector of southern Apennines chain (Italy). J. Geochem. Explor. 2020, 219, 106618. [Google Scholar] [CrossRef]
- Apollaro, C.; Fuoco, I.; Brozzo, G.; De Rosa, R. Release and fate of Cr (VI) in the ophiolitic aquifers of Italy: The role of Fe (III) as a potential oxidant of Cr (III) supported by reaction path modelling. Sci. Total Environ. 2019, 660, 1459–1471. [Google Scholar] [CrossRef] [PubMed]
- Apollaro, C.; Vespasiano, G.; Muto, F.; De Rosa, R.; Barca, D.; Marini, L. Use of mean residence time of water, flowrate, and equilibrium temperature indicated by water geothermometers to rank geothermal resources. Application to the thermal water circuits of Northern Calabria. J. Volcanol. Geotherm. Res. 2016, 328, 147–158. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C.A.J. Description of Input and Examples for PHREEQC Version 3—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations: U.S. Geological Survey Techniques and Methods; USGS: Reston, VA, USA, 2013; Book 6; Chapter A43; p. 497. [Google Scholar]
- Charlton, S.R.; Parkhurst, D.L. PhreeqcI-A Graphical User Interface to the Geochemical Model PHREEQC; Fact Sheet FS-031-02; U.S. Geological Survey: Reston, VA, USA, 2002; 2p. [CrossRef]
- Kumar, M.; Goswami, R.; Patel, A.K.; Srivastava, M.; Das, N. Scenario, perspectives and mechanism of arsenic and fluoride co-occurrence in the groundwater: A review. Chemosphere 2020, 249, 126126. [Google Scholar] [CrossRef] [PubMed]
- Deer, W.A.; Howie, R.A.; Zussman, J. An Introduction to the Rock- Forming Minerals, 3rd ed.; The Mineralogical Society: London, UK, 2013; 498p. [Google Scholar]
- Bonardi, G.; De Vivo, B.; Giunta, G.; Lima, A.; Perrone, V.; Zuppetta, A. Mineralizzazioni dell’Arco Calabro Peloritano. Ipotesi Genetiche e Quadro Evolutivo. Boll. Soc. Geol. It. 1982, 141–155. [Google Scholar]
- Zucchetti, S. I Giacimenti di Fluorite dell’area Peninsulare Italiana. Atti della Giornata di Studio su «LE FLUORITI ITALIANE» Torino. 1972. Available online: https://www.researchgate.net/profile/Stefano-Zucchetti/publication/303854674 (accessed on 4 May 2021).
- Castaldo, G. Memoria Illustrativa della Carta Mineraria D’italia. SCALA 1: 1000000. Carta Geologica d’Italia; Servizio Geologico D’ Italia: Roma, Italy, 1975; Volume XIV. [Google Scholar]
- Chae, G.T.; Yun, S.T.; Kim, K.; Mayer, B. Hydrogeochemistry of sodium-bicarbonate type bedrock groundwater in the Pocheon spa area, South Korea: Water–rock interaction and hydrologic mixing. J. Hydrol. 2006, 321, 326–343. [Google Scholar] [CrossRef]
- Chae, G.T.; Yun, S.T.; Kwon, M.J.; Kim, Y.S.; Mayer, B. Batch dissolution of granite and biotite in water: Implication for fluorine geochemistry in groundwater. Geochem. J. 2006, 40, 95–102. [Google Scholar] [CrossRef]
- Critelli, T.; Marini, L.; Schott, J.; Mavromatis, V.; Apollaro, C.; Rinder, T.; De Rosa, R.; Oelkers, E.H. Dissolution rates of actinolite and chlorite from a whole-rock experimental study of metabasalt dissolution from 2≤ pH≤ 12 at 25 C. Chem. Geol. 2014, 390, 100–108. [Google Scholar] [CrossRef]
- Gogoi, R.R.; Khanikar, L.; Gogoi, J.; Neog, N.; Deka, D.J.; Sarma, K.P. Geochemical sources, hydrogeochemical behaviour of fluoride release and its health risk assessment in some fluorosis endemic areas of the Brahmaputra valley of Assam, India. Appl. Geochem. 2021, 127, 104911. [Google Scholar] [CrossRef]
- Shen, J.; Jeihanipour, A.; Richards, B.S.; Schäfer, A.I. Renewable energy powered membrane technology: Experimental investigation of system performance with variable module size and fluctuating energy. Sep. Purif. Technol. 2019, 221, 64–73. [Google Scholar] [CrossRef]
- Owusu-Agyeman, I.; Reinwald, M.; Jeihanipour, A.; Schäfer, A.I. Removal of fluoride and natural organic matter from natural tropical brackish waters by nanofiltration/reverse osmosis with varying water chemistry. Chemosphere 2019, 217, 47–58. [Google Scholar] [CrossRef]
- Owusu-Agyeman, I.; Shen, J.; Schäfer, A.I. Renewable energy powered membrane technology: Impact of pH and ionic strength on fluoride and natural organic matter removal. Sci. Total Environ. 2018, 621, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Jeihanipour, A.; Shen, J.; Abbt-Braun, G.; Huber, S.A.; Mkongo, G.; Schäfer, A.I. Seasonal variation of organic matter characteristics and fluoride concentration in the Maji ya Chai River (Tanzania): Impact on treatability by nanofiltration/reverse osmosis. Sci. Total Environ. 2018, 637, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Pontié, M.; Dach, H.; Leparc, J.; Hafsi, M.; Lhassani, A. Novel approach combining physico-chemical characterizations and mass transfer modelling of nanofiltration and low pressure reverse osmosis membranes for brackish water desalination intensification. Desalination 2008, 221, 174–191. [Google Scholar] [CrossRef]
- Shen, J.; Mkongo, G.; Abbt-Braun, G.; Ceppi, S.L.; Richards, B.S.; Schäfer, A.I. Renewable energy powered membrane technology: Fluoride removal in a rural community in northern Tanzania. Sep. Purif. Technol. 2015, 149, 349–361. [Google Scholar] [CrossRef]
- Van der Aa, M. Classification of mineral water types and comparison with drinking water standards. Environ. Geol. 2003, 44, 554–563. [Google Scholar] [CrossRef]
- Verma, K.C.; Kushwaha, A.S. Demineralization of drinking water: Is it prudent? Med. J. Armed Forces Ind. 2014, 70, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Kozisek, F. Health risks from drinking demineralised water. Nutr. Drink. Water 2005, 1, 148–163. [Google Scholar]
| SPR 10113 | SRP 10114 | |
|---|---|---|
| Type | NF | NF |
| Materials | Polyamide-TFC | Polyamide-TFC |
| Manufacturer | SEPRA | SEPRA |
| Configuration | flat plate | flat plate |
| MWCO | 100–250 Da | 150–300 Da |
| MgSO4 rejection | 99.50% | 99% |
| NaCl rejection | 55% | 40% |
| pH operating range | 3–10 | 3–10 |
| Sample | pH | T | EC | Eh | SiO2 | Ca | Mg | K | Na | Cl | SO4 | HCO3 | NH4 | F |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sorensen’s Scale | °C | µS/cm | mV | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| Pc | 9.6 | 20.1 | 1059.0 | −119.9 | 11.25 | 5.30 | 2.20 | 1.65 | 174.55 | 23.35 | 75.61 | 259.34 | 3.80 | 30.12 |
| Bg | 10.1 | 21.4 | 363.5 | −214.6 | 23.30 | 2.39 | 0.26 | 0.99 | 76.31 | 8.66 | 9.08 | 168.58 | - | 8.90 |
| Membrane Type | Water Sample | F- Concentration Before Treatment | Applied TMP | Flux | F Rejection | F Concentration After Treatment | Cond | Ref. |
|---|---|---|---|---|---|---|---|---|
| mg/L | bar | L/m2h | % | mg/L | µS/cm | |||
| NF5 | Louza 2 | 3.39 | 8 | 91 a | 57.2 | 1.45 | - | [19] |
| NF9 | Louza 2 | 3.39 | 8 | 57.2 a | 88.8 | 0.38 | - | [19] |
| NF 90 4040 | BGW | 55 b | 6 | 29 *b | 98 b | 0.64 b | 2990 b | [16] |
| NF-1 | GW Birbhum | 20 | 9.8 | 106 | 95 | 1 * | 671 | [20] |
| NF-2 | GW Birbhum | 20 | 9.8 | 124 | 78 | 4.4 * | 671 | [20] |
| NF-20 | GW Birbhum | 20 | 9.8 | 116 | 86 | 2.8 * | 671 | [20] |
| NF-90 | St. Dorcas borehole | 49.1 b | 4.8 | 20.8 | 98.2 | 0.9 | 3935 b | [17] |
| NF-270 | St. Dorcas borehole | 49.1 b | 4.8 | 29.8 | 71.9 | 14.2 | 3935 b | [17] |
| NF270 | feed water | 2.32 | 10 | - | 82 * | 0.4 | 1492 | [21] |
| TR60 | feed water | 2.32 | 10 | - | 74 * | 0.6 | 1492 | [21] |
| NF90 | feed water | 2.32 | 10 | - | 98 * | 0.05 | 1492 | [21] |
| SPR 10113 | Bg | 8.9 | 10 | 42 | 94.6 | 0.5 | 364 | This work |
| SPR 10114 | Bg | 8.9 | 10 | 27 | 94.2 | 0.5 | 364 | This work |
| SPR 10113 | Pc | 30.1 | 10 | 41 | 94.9 | 1.5 | 1059 | This work |
| SPR 10114 | Pc | 30.1 | 10 | 26 | 96 | 1.1 | 1059 | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuoco, I.; Apollaro, C.; Criscuoli, A.; De Rosa, R.; Velizarov, S.; Figoli, A. Fluoride Polluted Groundwaters in Calabria Region (Southern Italy): Natural Source and Remediation. Water 2021, 13, 1626. https://doi.org/10.3390/w13121626
Fuoco I, Apollaro C, Criscuoli A, De Rosa R, Velizarov S, Figoli A. Fluoride Polluted Groundwaters in Calabria Region (Southern Italy): Natural Source and Remediation. Water. 2021; 13(12):1626. https://doi.org/10.3390/w13121626
Chicago/Turabian StyleFuoco, Ilaria, Carmine Apollaro, Alessandra Criscuoli, Rosanna De Rosa, Svetlozar Velizarov, and Alberto Figoli. 2021. "Fluoride Polluted Groundwaters in Calabria Region (Southern Italy): Natural Source and Remediation" Water 13, no. 12: 1626. https://doi.org/10.3390/w13121626
APA StyleFuoco, I., Apollaro, C., Criscuoli, A., De Rosa, R., Velizarov, S., & Figoli, A. (2021). Fluoride Polluted Groundwaters in Calabria Region (Southern Italy): Natural Source and Remediation. Water, 13(12), 1626. https://doi.org/10.3390/w13121626











