Development of Biochars Derived from Water Bamboo (Zizania latifolia) Shoot Husks Using Pyrolysis and Ultrasound-Assisted Pyrolysis for the Treatment of Reactive Black 5 (RB5) in Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Analytical Methods
2.2. BC and UBC Preparation and RB5 Adsorption-Capacity Comparison
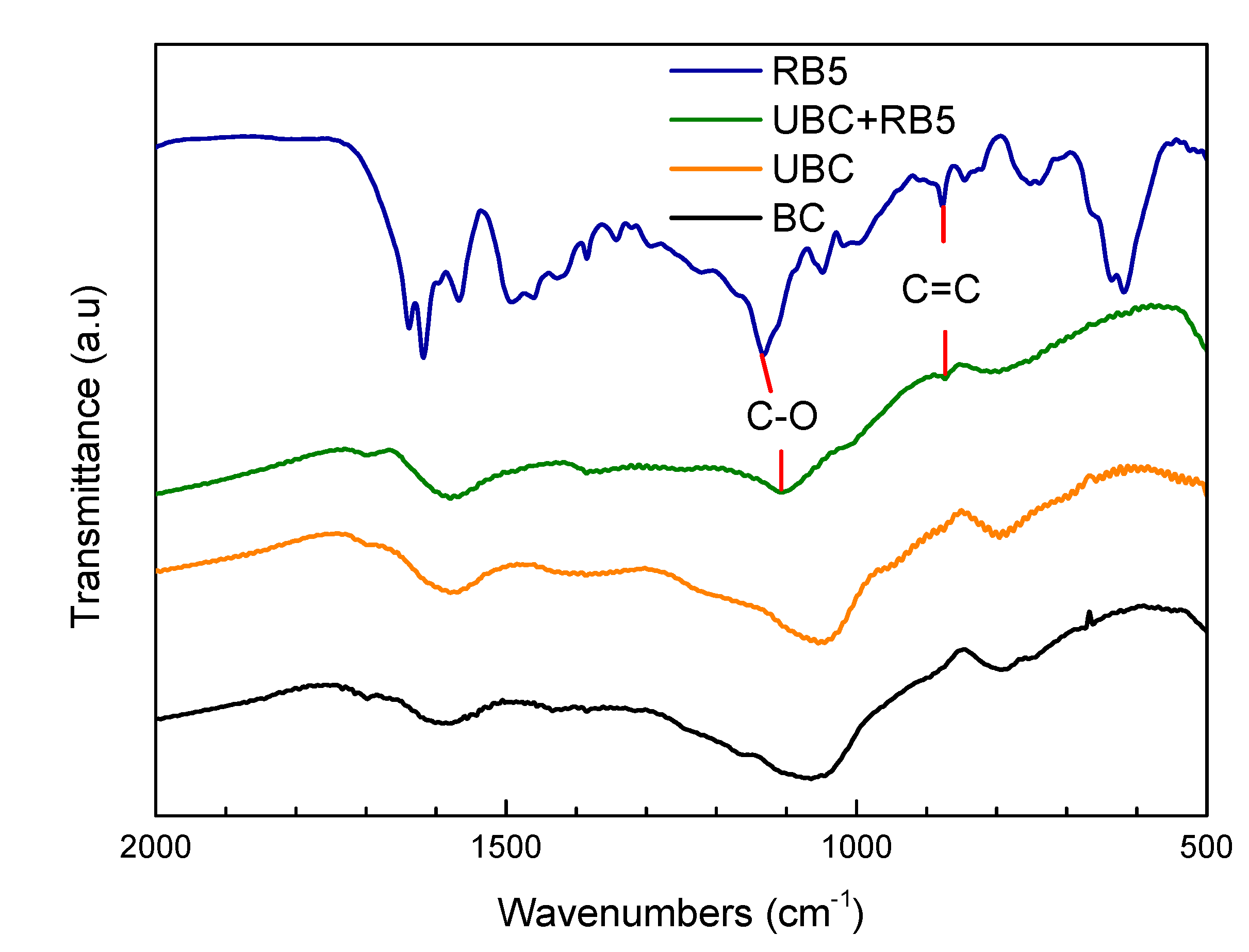
2.3. Characterization of BC and UBC
2.4. Adsorption Equilibrium Isotherm Experiments
2.5. Adsorption Kinetics Studies
3. Results and Discussion
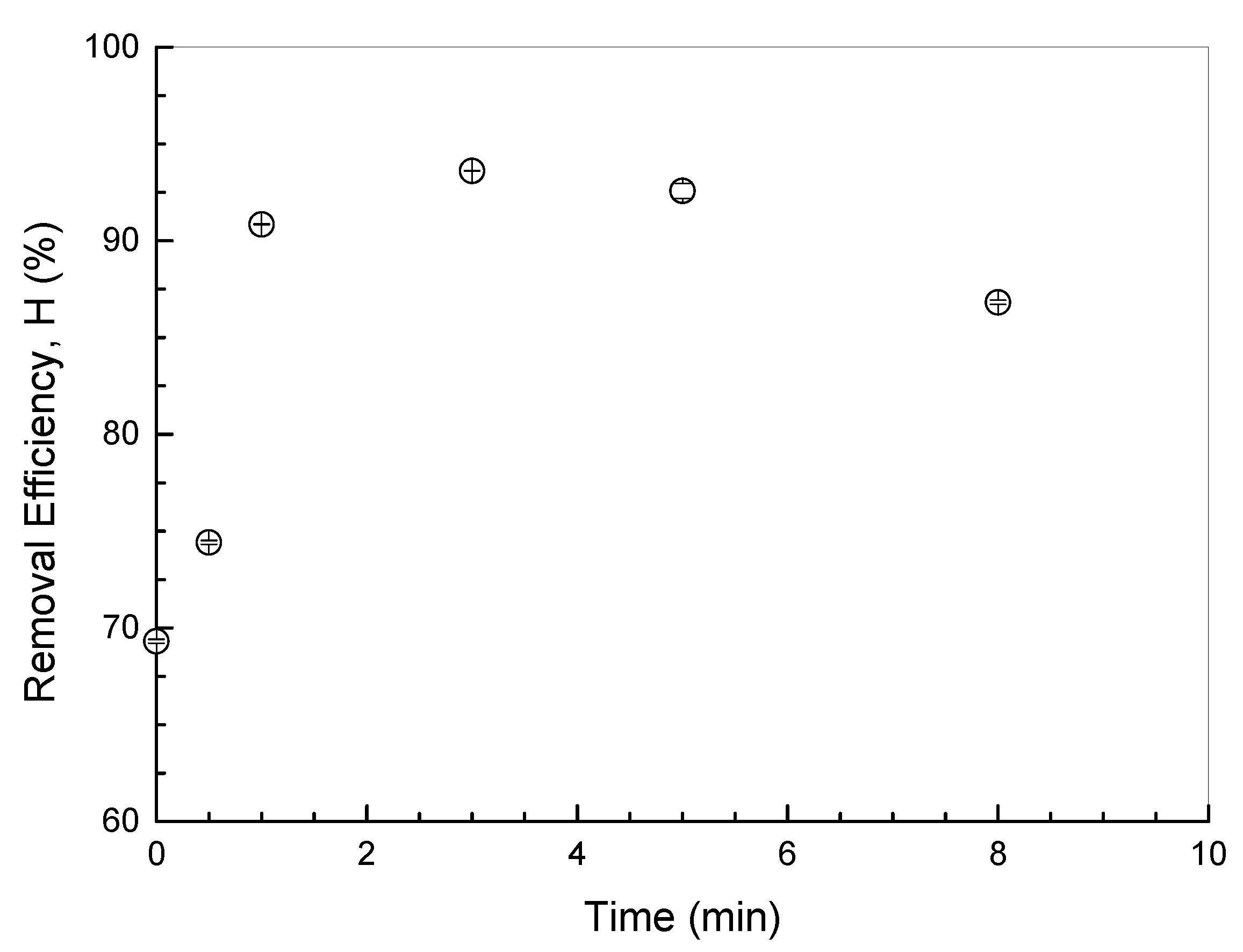
3.1. Effect of Ultrasonic Irradiation Time on UBC Properties
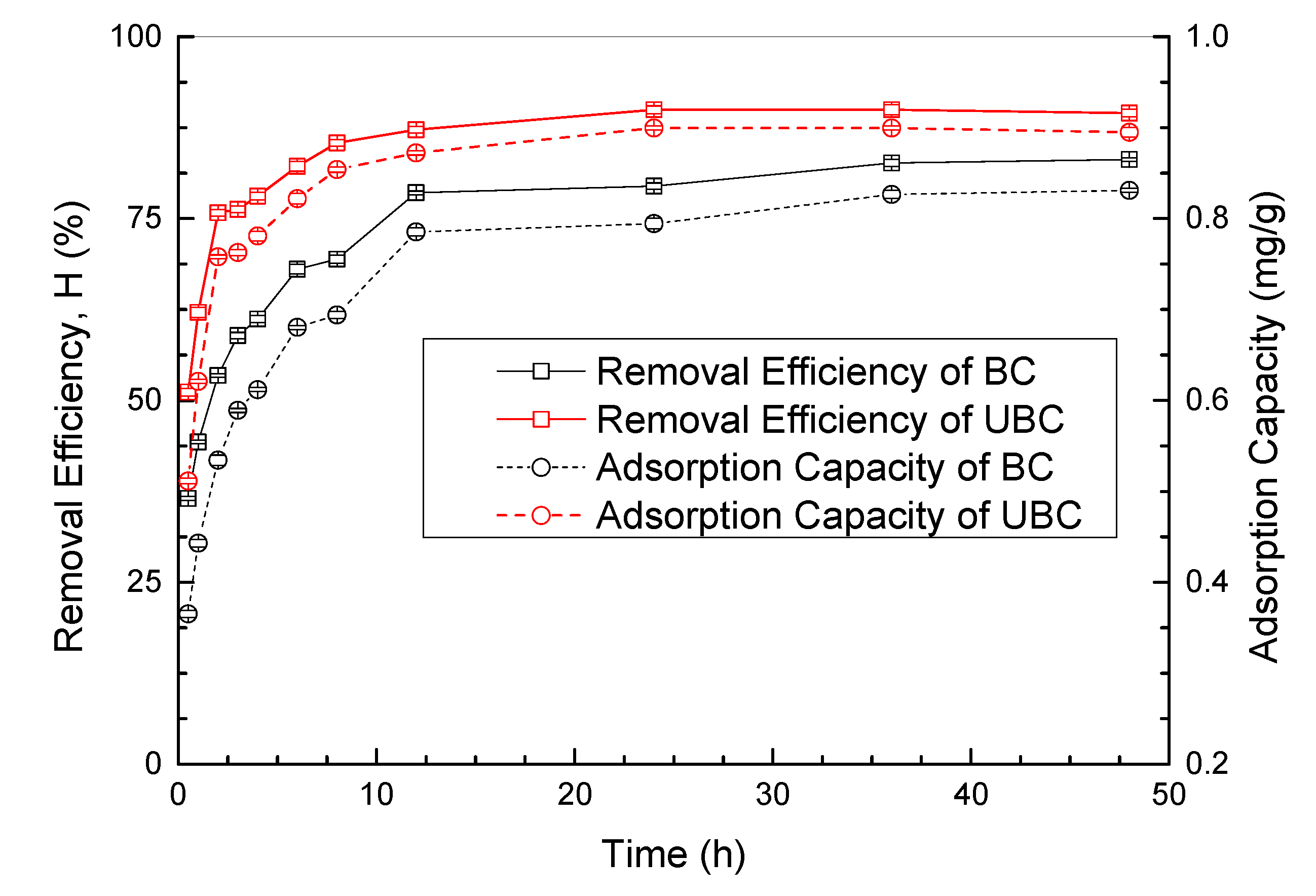
3.2. Comparison of RB5 Adsorption Capacity by BC and UBC
3.3. Effects of Operating Conditions on RB5 Adsorption of UBC
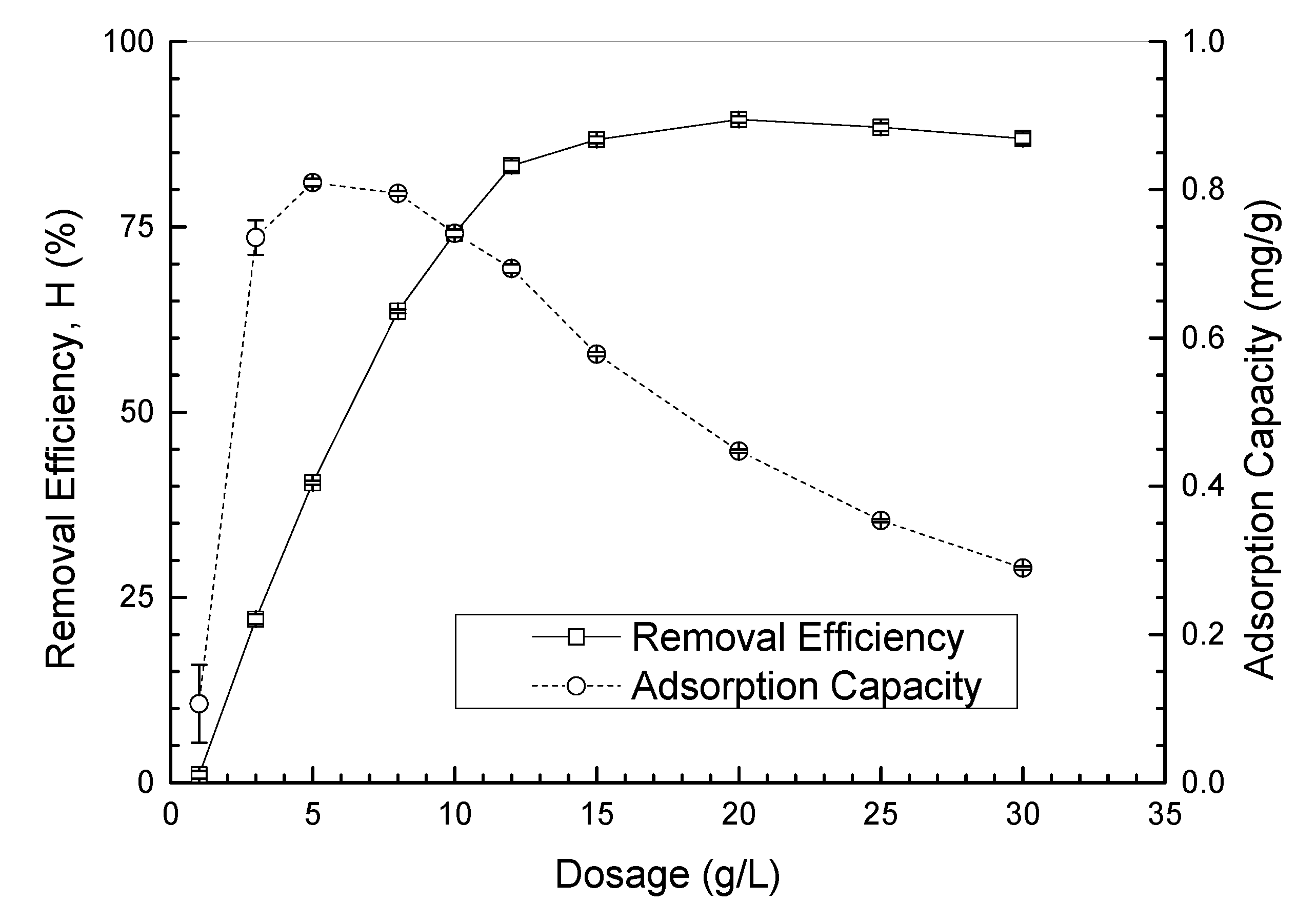
3.3.1. UBC Dosage
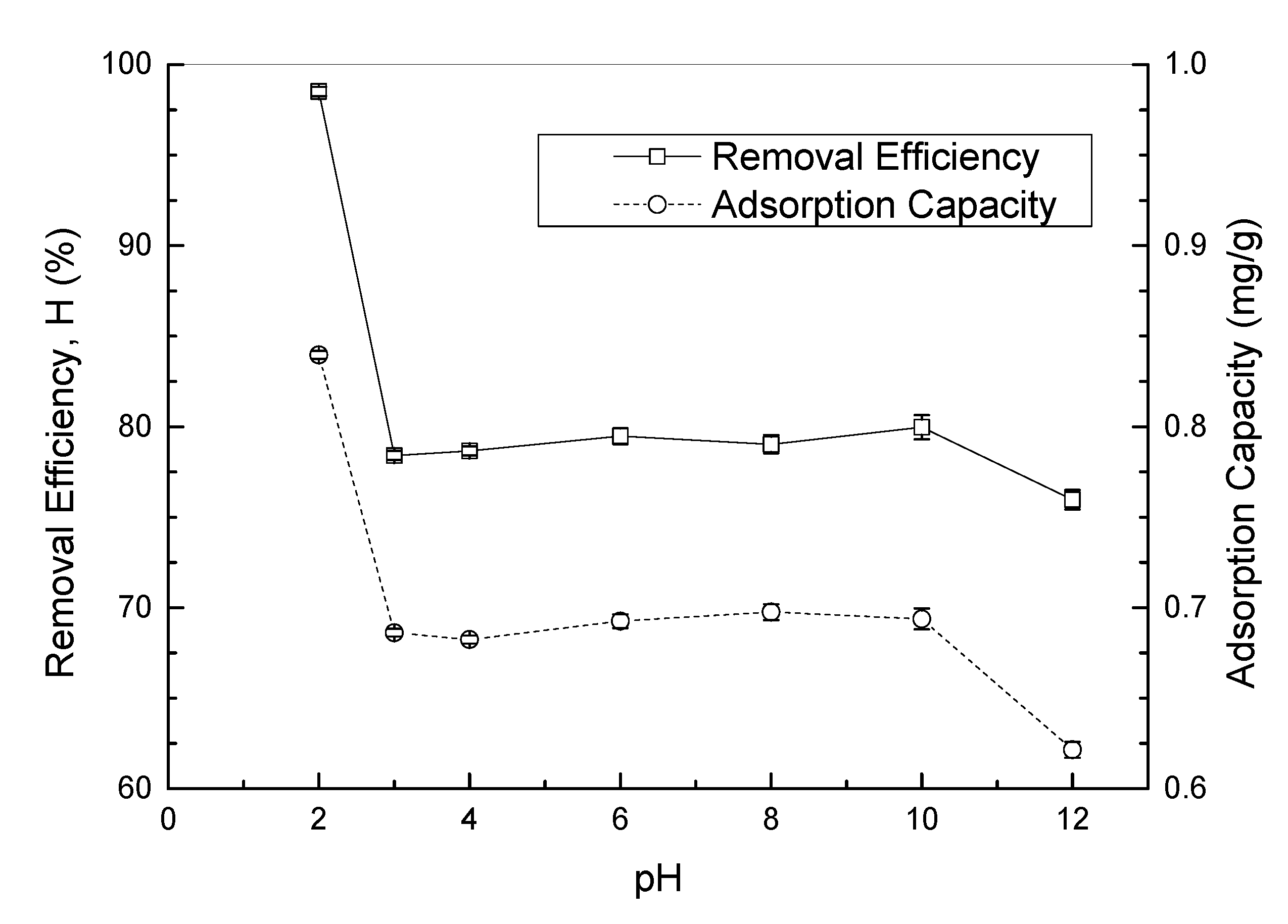
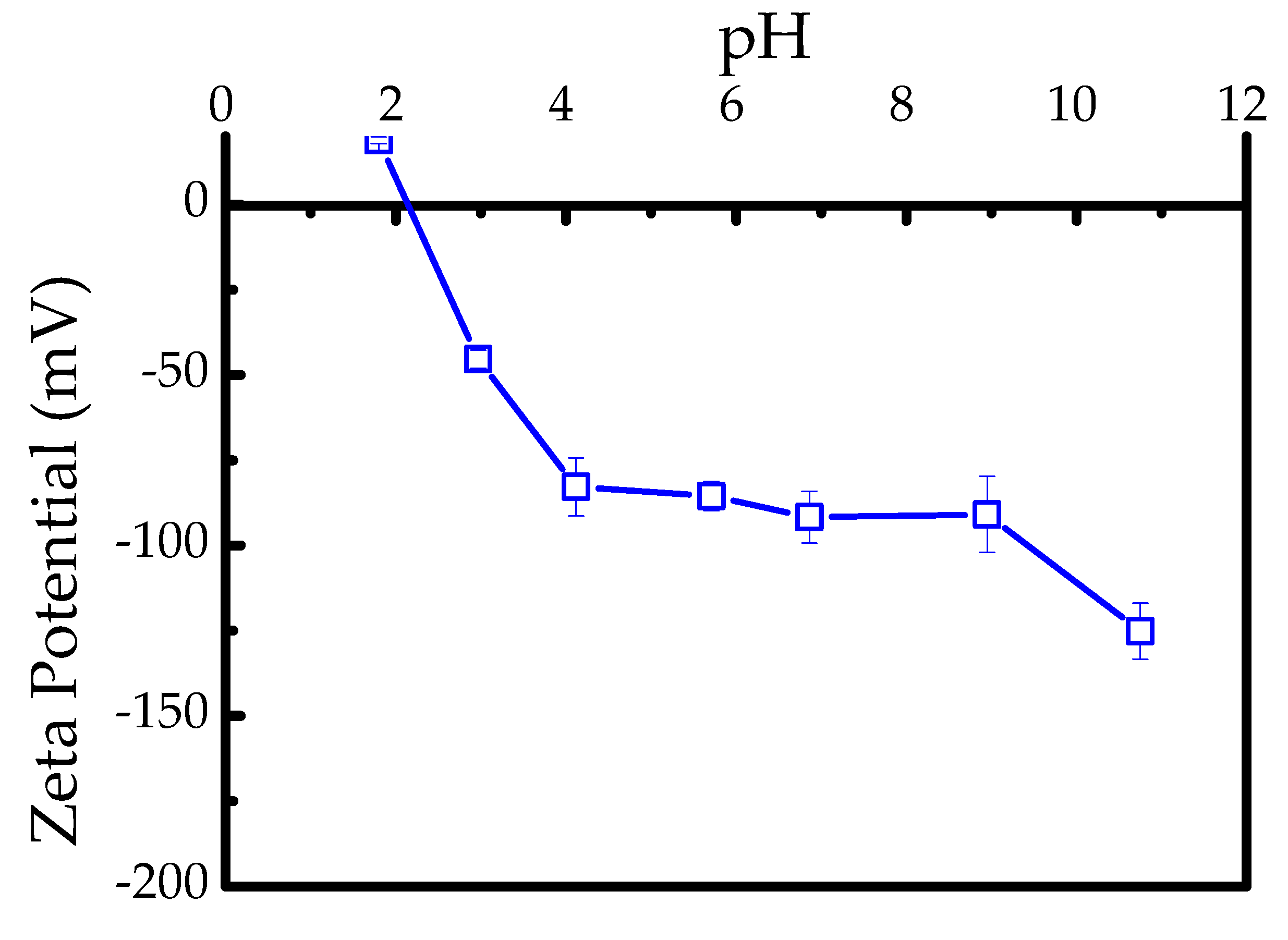
3.3.2. Solution pH
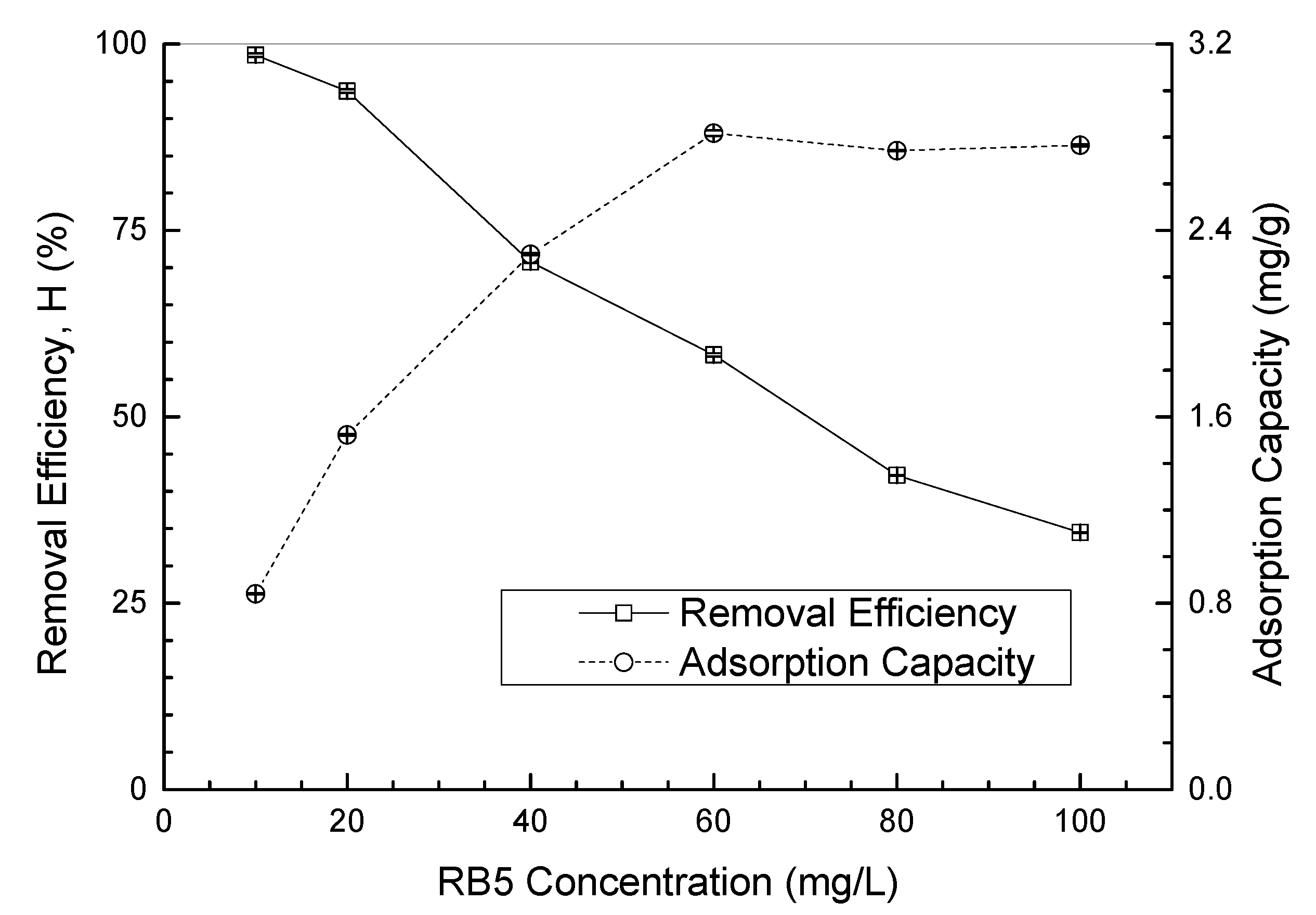
3.3.3. Initial RB5 Concentration
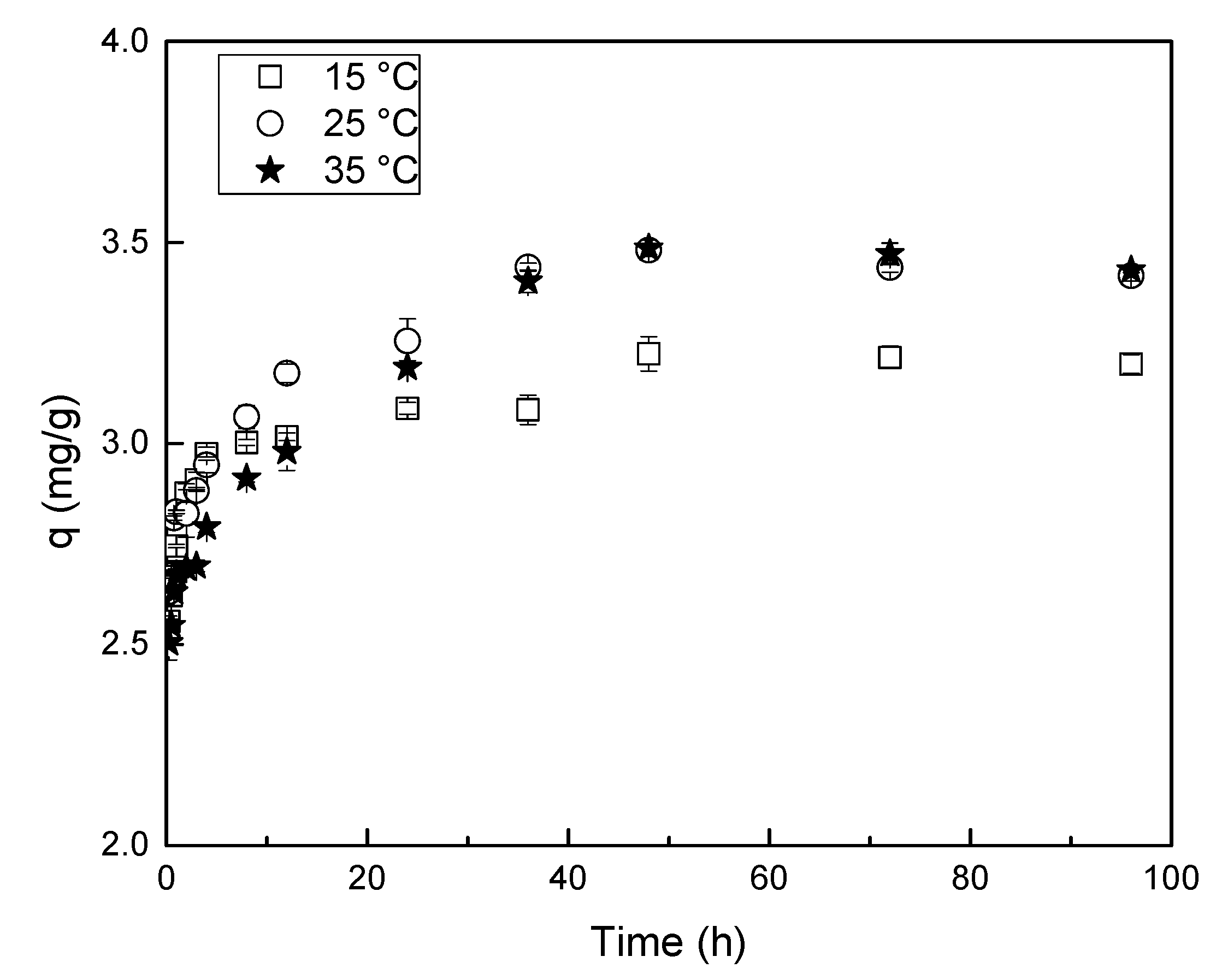
3.3.4. Temperature and Reaction Time
3.4. Study on Adsorption Mechanism
3.4.1. Adsorption Isotherms
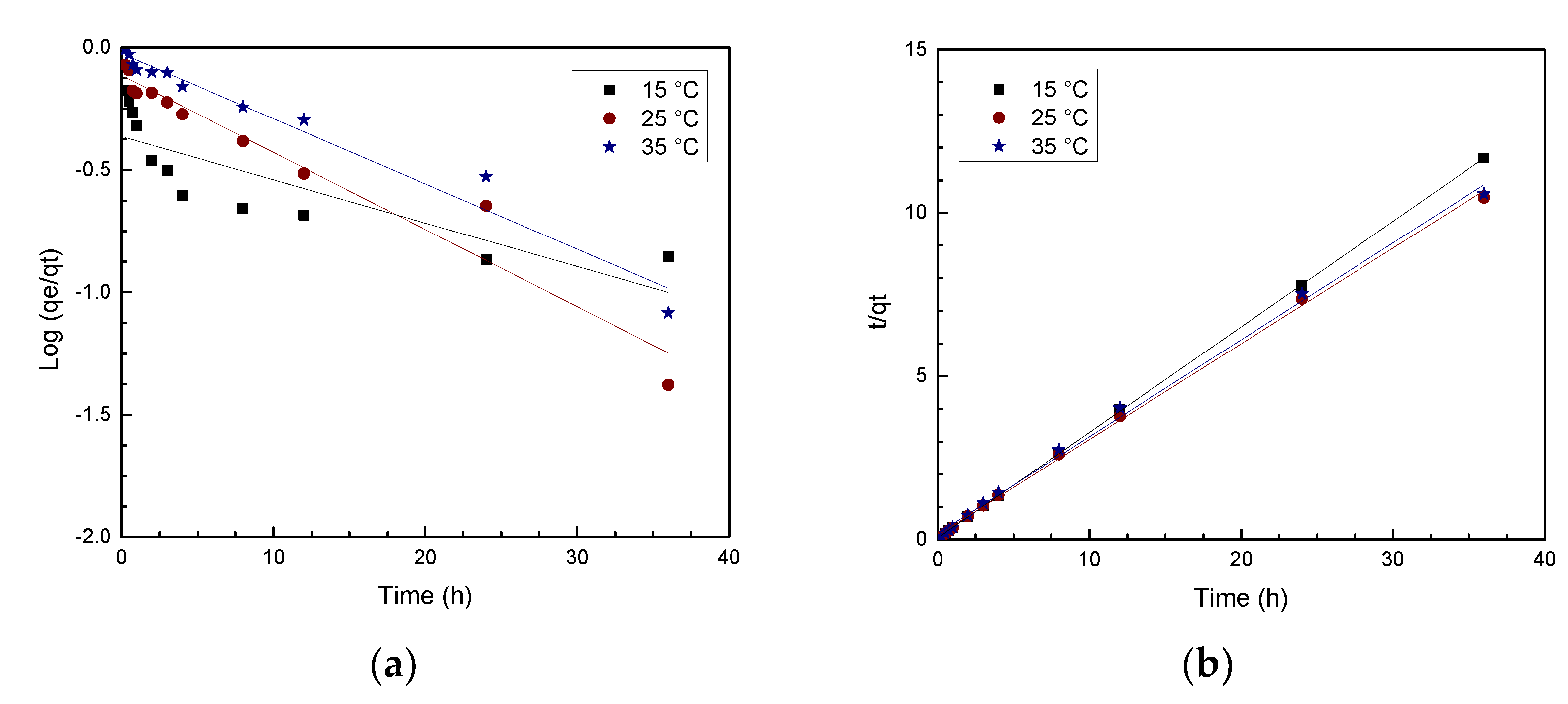
3.4.2. Adsorption Kinetics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cederlund, H.; Börjesson, E.; Stenström, J. Effects of a wood-based biochar on the leaching of pesticides chlorpyrifos, diuron, glyphosate and MCPA. J. Environ. Manag. 2017, 191, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Doan, T.T.; Henry-Des-Tureaux, T.; Rumpel, C.; Janeau, J.L.; Jouquet, P. Impact of compost, vermicompost and biochar on soil fertility, maize yield and soil erosion in Northern Vietnam: A three year mesocosm experiment. Sci. Total Environ. 2015, 514, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Gámiz, B.; Velarde, P.; Spokas, K.A.; Hermosín, M.C.; Cox, L. Biochar soil additions affect herbicide fate: Importance of application timing and feedstock species. J. Agric. Food Chem. 2017, 65, 3109–3117. [Google Scholar] [CrossRef] [PubMed]
- Streubel, J.D.; Collins, H.P.; Garcia-Perez, M.; Tarara, J.; Granatstein, D.; Kruger, C.E. Influence of contrasting biochar types on five soils at increasing rates of application. Soil Sci. Soc. Am. J. 2011, 75, 1402–1413. [Google Scholar] [CrossRef]
- Yousaf, B.; Liu, G.; Wang, R.; Abbas, Q.; Imtiaz, M.; Liu, R. Investigating the biochar effects on C-mineralization and sequestration of carbon in soil compared with conventional amendments using the stable isotope (δ13C) approach. GCB Bioenergy 2017, 9, 1085–1099. [Google Scholar] [CrossRef]
- Case, S.D.C.; McNamara, N.P.; Reay, D.S.; Stott, A.W.; Grant, H.K.; Whitaker, J. Biochar suppresses N2O emissions while maintaining N availability in a sandy loam soil. Soil Biol. Biochem. 2015, 81, 178–185. [Google Scholar] [CrossRef]
- Cayuela, M.L.; Jeffery, S.; van Zwieten, L. The molar H: Corg ratio of biochar is a key factor in mitigating N2O emissions from soil. Agric. Ecosyst. Environ. 2015, 202, 135–138. [Google Scholar] [CrossRef]
- Sheng, Y.; Zhu, L. Biochar alters microbial community and carbon sequestration potential across different soil pH. Sci. Total Environ. 2018, 622–623, 1391–1399. [Google Scholar] [CrossRef]
- Smith, P. Soil carbon sequestration and biochar as negative emission technologies. Glob. Chang. Biol. 2016, 22, 1315–1324. [Google Scholar] [CrossRef]
- Dehkhoda, A.M.; West, A.H.; Ellis, N. Biochar based solid acid catalyst for biodiesel production. Appl. Catal. A Gen. 2010, 382, 197–204. [Google Scholar] [CrossRef]
- Dong, T.; Gao, D.; Miao, C.; Yu, X.; Degan, C.; Garcia-Pérez, M.; Rasco, B.; Sablani, S.S.; Chen, S. Two-step microalgal biodiesel production using acidic catalyst generated from pyrolysis-derived biochar. Energy Convers. Manag. 2015, 105, 1389–1396. [Google Scholar] [CrossRef]
- Fang, G.; Liu, C.; Gao, J.; Dionysiou, D.D.; Zhou, D. Manipulation of persistent free radicals in biochar to activate persulfate for contaminant degradation. Environ. Sci. Technol. 2015, 49, 5645–5653. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, P.A.; Backes, R.C.; Martins, A.; De Carvalho, C.T.; Da Silva, R.A.B. Biochar: A low-cost electrode modifier for electrocatalytic, sensitive and selective detection of similar organic compounds. Electroanalysis 2018, 30, 2233–2236. [Google Scholar] [CrossRef]
- Basri, N.H.; Deraman, M.; Kanwal, S.; Talib, I.A.; Manjunatha, J.G.; Aziz, A.A.; Farma, R. Supercapacitors using binderless composite monolith electrodes from carbon nanotubes and pre-carbonized biomass residues. Biomass Bioenergy 2013, 59, 370–379. [Google Scholar] [CrossRef]
- Farma, R.; Deraman, M.; Awitdrus, A.; Talib, I.A.; Taer, E.; Basri, N.H.; Manjunatha, J.G.; Ishak, M.M.; Dollah, B.N.M.; Hashmi, S.A. Preparation of highly porous binderless activated carbon electrodes from fibres of oil palm empty fruit bunches for application in supercapacitors. Bioresour. Technol. 2013, 132, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Goodman, P.A.; Li, H.; Gao, Y.; Lu, Y.F.; Stenger-Smith, J.D.; Redepenning, J. Preparation and characterization of high surface area, high porosity carbon monoliths from pyrolyzed bovine bone and their performance as supercapacitor electrodes. Carbon N. Y. 2013, 55, 291–298. [Google Scholar] [CrossRef]
- Tang, L.; Yu, J.; Pang, Y.; Zeng, G.; Deng, Y.; Wang, J.; Ren, X.; Ye, S.; Peng, B.; Feng, H. Sustainable efficient adsorbent: Alkali-acid modified magnetic biochar derived from sewage sludge for aqueous organic contaminant removal. Chem. Eng. J. 2018, 336, 160–169. [Google Scholar] [CrossRef]
- Zhang, K.; Sun, P.; Zhang, Y. Decontamination of Cr(VI) facilitated formation of persistent free radicals on rice husk derived biochar. Front. Environ. Sci. Eng. 2019, 13, 22. [Google Scholar] [CrossRef]
- Reguyal, F.; Sarmah, A.K.; Gao, W. Synthesis of magnetic biochar from pine sawdust via oxidative hydrolysis of FeCl2 for the removal sulfamethoxazole from aqueous solution. J. Hazard. Mater. 2017, 321, 868–878. [Google Scholar] [CrossRef]
- Chatterjee, R.; Sajjadi, B.; Mattern, D.L.; Chen, W.Y.; Zubatiuk, T.; Leszczynska, D.; Leszczynski, J.; Egiebor, N.O.; Hammer, N. Ultrasound cavitation intensified amine functionalization: A feasible strategy for enhancing CO2 capture capacity of biochar. Fuel 2018, 225, 287–298. [Google Scholar] [CrossRef]
- Li, H.; Dong, X.; Da Silva, E.B.; de Oliveira, L.M.; Chen, Y.; Ma, L.Q. Mechanisms of metal sorption by biochars: Biochar characteristics and modifications. Chemosphere 2017, 178, 466–478. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, modification and environmental application of biochar: A review. J. Clean. Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Wang, C.; Wang, H.; Cao, Y. Pb(II) sorption by biochar derived from Cinnamomum camphora and its improvement with ultrasound-assisted alkali activation. Colloids Surf. A Physicochem. Eng. Asp. 2018, 556, 177–184. [Google Scholar] [CrossRef]
- Shrestha, R. Ultrasound-Assisted Post-Pyrolysis Magnetization of Microporous Ultrasound-Assisted Post-Pyrolysis Magnetization of Microporous Biochar for Effective Removal of Heavy Metals Biochar for Effective Removal of Heavy Metals. Undergraduate Thesis, The University of Mississippi, University, MS, USA, 2020. Available online: https://egrove.olemiss.edu/hon_thesis (accessed on 15 June 2020).
- Chatterjee, R.; Sajjadi, B.; Chen, W.Y.; Mattern, D.L.; Egiebor, N.O.; Hammer, N.; Raman, V. Low frequency ultrasound enhanced dual amination of biochar: A nitrogen-enriched sorbent for CO2 Capture. Energy Fuels 2019, 33, 2366–2380. [Google Scholar] [CrossRef]
- Sajjadi, B.; Chen, W.Y.; Adeniyi, A.; Mattern, D.L.; Mobley, J.; Huang, C.P.; Fan, R. Variables governing the initial stages of the synergisms of ultrasonic treatment of biochar in water with dissolved CO2. Fuel 2019, 235, 1131–1145. [Google Scholar] [CrossRef]
- Ahmad, A.A.; Hameed, B.H. Fixed-bed adsorption of reactive azo dye onto granular activated carbon prepared from waste. J. Hazard. Mater. 2010, 175, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Al-Degs, Y.S.; El-Barghouthi, M.I.; El-Sheikh, A.H.; Walker, G.M. Effect of solution pH, ionic strength, and temperature on adsorption behavior of reactive dyes on activated carbon. Dyes Pigments 2008, 77, 16–23. [Google Scholar] [CrossRef]
- Sathian, S.; Rajasimman, M.; Radha, G.; Shanmugapriya, V.; Karthikeyan, C. Performance of SBR for the treatment of textile dye wastewater: Optimization and kinetic studies. Alex. Eng. J. 2014, 53, 417–426. [Google Scholar] [CrossRef]
- Padmesh, T.V.N.; Vijayaraghavan, K.; Sekaran, G.; Velan, M. Biosorption of Acid Blue 15 using fresh water macroalga Azolla filiculoides: Batch and column studies. Dyes Pigments 2006, 71, 77–82. [Google Scholar] [CrossRef]
- Heibati, B.; Rodriguez-Couto, S.; Amrane, A.; Rafatullah, M.; Hawari, A.; Al-Ghouti, M.A. Uptake of Reactive Black 5 by pumice and walnut activated carbon: Chemistry and adsorption mechanisms. J. Ind. Eng. Chem. 2014, 20, 2939–2947. [Google Scholar] [CrossRef]
- Güzel, F.; Sayʇili, H.; Sayʇili, G.A.; Koyuncu, F. New low-cost nanoporous carbonaceous adsorbent developed from carob (Ceratonia siliqua) processing industry waste for the adsorption of anionic textile dye: Characterization, equilibrium and kinetic modeling. J. Mol. Liq. 2015, 206, 244–255. [Google Scholar] [CrossRef]
- Shih, Y.F. Mechanical and thermal properties of waste water bamboo husk fiber reinforced epoxy composites. Mater. Sci. Eng. A 2007, 445–446, 289–295. [Google Scholar] [CrossRef]
- Greluk, M.; Hubicki, Z. Kinetics, isotherm and thermodynamic studies of Reactive Black 5 removal by acid acrylic resins. Chem. Eng. J. 2010, 162, 919–926. [Google Scholar] [CrossRef]
- Barrer, R.M. Expanded clay minerals: A major class of molecular sieves. J. Incl. Phenom. 1986, 4, 109–119. [Google Scholar] [CrossRef]
- Inglezakis, V.J.; Loizidou, M.D.; Grigoropoulou, H.P. Equilibrium and kinetic ion exchange studies of Pb2+, Cr3+, Fe3+ and Cu2+ on natural clinoptilolite. Water Res. 2002, 36, 2784–2792. [Google Scholar] [CrossRef]
- Ahmad, A.A.; Idris, A.; Hameed, B.H. Organic dye adsorption on activated carbon derived from solid waste. Desalin. Water Treat. 2013, 51, 2554–2563. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Asakura, Y.; Koda, S.; Yasuda, K. Dependence of cavitation, chemical effect, and mechanical effect thresholds on ultrasonic frequency. Ultrason. Sonochem. 2017, 39, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, K.; Nguyen, T.T.; Asakura, Y. Measurement of distribution of broadband noise and sound pressures in sonochemical reactor. Ultrason. Sonochem. 2018, 43, 23–28. [Google Scholar] [CrossRef]
- Yasuda, K. Sonochemical green technology using active bubbles: Degradation of organic substances in water. Curr. Opin. Green Sustain. Chem. 2021, 27, 100411. [Google Scholar] [CrossRef]
- Ip, A.W.M.; Barford, J.P.; McKay, G. A comparative study on the kinetics and mechanisms of removal of Reactive Black 5 by adsorption onto activated carbons and bone char. Chem. Eng. J. 2010, 157, 434–442. [Google Scholar] [CrossRef]
- Peter, A.; Chabot, B.; Loranger, E. Enhancing surface properties of softwood biochar by ultrasound assisted slow pyrolysis. In Proceedings of the 2019 IEEE International Ultrasonics Symposium (IUS), Glasgow, UK, 6–9 October 2019; pp. 2477–2480. [Google Scholar] [CrossRef]
- Cardoso, N.F.; Pinto, R.B.; Lima, E.C.; Calvete, T.; Amavisca, C.V.; Royer, B.; Cunha, M.L.; Fernandes, T.H.; Pinto, I.S. Removal of remazol black B textile dye from aqueous solution by adsorption. Desalination 2011, 269, 92–103. [Google Scholar] [CrossRef]
- Vakili, M.; Zwain, H.M.; Mojiri, A.; Wang, W.; Gholami, F.; Gholami, Z.; Giwa, A.S.; Wang, B.; Cagnetta, G.; Salamatinia, B. Effective adsorption of reactive black 5 onto hybrid hexadecylamine impregnated chitosan-powdered activated carbon beads. Water 2020, 12, 2242. [Google Scholar] [CrossRef]
- Zhang, P.; Lo, I.; O’Connor, D.; Pehkonen, S.; Cheng, H.; Hou, D. High efficiency removal of methylene blue using SDS surface-modified ZnFe2O4 nanoparticles. J. Colloid Interface Sci. 2017, 508, 39–48. [Google Scholar] [CrossRef]
- Santhosh, C.; Daneshvar, E.; Kollu, P.; Peräniemi, S.; Grace, A.N.; Bhatnagar, A. Magnetic SiO2@CoFe2O4 nanoparticles decorated on graphene oxide as efficient adsorbents for the removal of anionic pollutants from water. Chem. Eng. J. 2017, 322, 472–487. [Google Scholar] [CrossRef]
- Banerjee, S.; Chattopadhyaya, M.C. Adsorption characteristics for the removal of a toxic dye, tartrazine from aqueous solutions by a low cost agricultural by-product. Arab. J. Chem. 2017, 10, S1629–S1638. [Google Scholar] [CrossRef]
- Subramaniam, R.; Kumar Ponnusamy, S. Novel adsorbent from agricultural waste (cashew NUT shell) for methylene blue dye removal: Optimization by response surface methodology. Water Resour. Ind. 2015, 11, 64–70. [Google Scholar] [CrossRef]
- Santhy, K.; Selvapathy, P. Removal of reactive dyes from wastewater by adsorption on coir pith activated carbon. Bioresour. Technol. 2006, 97, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Mook, W.T.; Aroua, M.K.; Szlachta, M. Palm shell-based activated carbon for removing reactive black 5 dye: Equilibrium and kinetics studies. BioResources 2016, 11, 1432–1447. [Google Scholar] [CrossRef]
- Malik, P.K. Use of activated carbons prepared from sawdust and rice-husk for adsoprtion of acid dyes: A case study of acid yellow 36. Dyes Pigments 2003, 56, 239–249. [Google Scholar] [CrossRef]
- Hsueh, C.L.; Lu, Y.W.; Hung, C.C.; Huang, Y.H.; Chen, C.Y. Adsorption kinetic, thermodynamic and desorption studies of C.I. Reactive Black 5 on a novel photoassisted Fenton catalyst. Dyes Pigments 2007, 75, 130–135. [Google Scholar] [CrossRef]
- Zhang, P.; O’Connor, D.; Wang, Y.; Jiang, L.; Xia, T.; Wang, L.; Tsang, D.C.; Ok, Y.S.; Hou, D. A green biochar/iron oxide composite for methylene blue removal. J. Hazard. Mater. 2020, 384, 121286. [Google Scholar] [CrossRef] [PubMed]
- Ai, L.; Zhang, C.; Liao, F.; Wang, Y.; Li, M.; Meng, L.; Jiang, J. Removal of methylene blue from aqueous solution with magnetite loaded multi-wall carbon nanotube: Kinetic, isotherm and mechanism analysis. J. Hazard. Mater. 2011, 198, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Watthanaphanit, A.; Saito, N. Simple Solution Plasma Synthesis of Hierarchical Nanoporous MnO2 for Organic Dye Removal. ACS Sustain. Chem. Eng. 2017, 5, 5842–5851. [Google Scholar] [CrossRef]
- Katsumi, T. Soil excavation and reclamation in civil engineering: Environmental aspects. Soil Sci. Plant. Nutr. 2015, 61, 22–29. [Google Scholar] [CrossRef]
- Fytianos, K.; Voudrias, E.; Kokkalis, E. Sorption-desorption behaviour of 2,4-dichlorophenol by marine sediments. Chemosphere 2000, 40, 3–6. [Google Scholar] [CrossRef]
- Isah, U.A.; Abdulraheem, G.; Bala, S.; Muhammad, S.; Abdullahi, M. Kinetics, equilibrium and thermodynamics studies of C.I. Reactive Blue 19 dye adsorption on coconut shell based activated carbon. Int. Biodeterior. Biodegrad. 2015, 102, 265–273. [Google Scholar] [CrossRef]
- Schimmel, D.; Fagnani, K.C.; Dos Santos, J.B.O.; Barros, M.A.S.D.; Da Silva, E.A. Adsorption of turquoise blue qg reactive dye on commercial activated carbon in batch reactor: Kinetic and equilibrium studies. Braz. J. Chem. Eng. 2010, 27, 289–298. [Google Scholar] [CrossRef]

| Langmuir | Freundlich | |||||
|---|---|---|---|---|---|---|
| Root-Mean-Square Deviation (RMSE) | R2 (p-Value) | R2 (p-Value) | ||||
| 2.811 | 2.814 | 0.362 | 0.999 (5.590 | 1.134 | 0.202 | 0.964 () |
| Temperature | 1st Order | 2nd Order | ||
|---|---|---|---|---|
| k1 | R2 (p-Value) | k2 | R2 (p-Value) | |
| 15 °C | 0.072 | ) | 0.007 | ) |
| 25 °C | 0.061 | ) | 0.055 | ) |
| 35 °C | 0.041 | ) | 0.105 | ) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.T.; Chen, H.-H.; To, T.H.; Chang, Y.-C.; Tsai, C.-K.; Chen, K.-F.; Tsai, Y.-P. Development of Biochars Derived from Water Bamboo (Zizania latifolia) Shoot Husks Using Pyrolysis and Ultrasound-Assisted Pyrolysis for the Treatment of Reactive Black 5 (RB5) in Wastewater. Water 2021, 13, 1615. https://doi.org/10.3390/w13121615
Nguyen TT, Chen H-H, To TH, Chang Y-C, Tsai C-K, Chen K-F, Tsai Y-P. Development of Biochars Derived from Water Bamboo (Zizania latifolia) Shoot Husks Using Pyrolysis and Ultrasound-Assisted Pyrolysis for the Treatment of Reactive Black 5 (RB5) in Wastewater. Water. 2021; 13(12):1615. https://doi.org/10.3390/w13121615
Chicago/Turabian StyleNguyen, Thanh Tam, Hung-Hsiang Chen, Thi Hien To, Yu-Chen Chang, Cheng-Kuo Tsai, Ku-Fan Chen, and Yung-Pin Tsai. 2021. "Development of Biochars Derived from Water Bamboo (Zizania latifolia) Shoot Husks Using Pyrolysis and Ultrasound-Assisted Pyrolysis for the Treatment of Reactive Black 5 (RB5) in Wastewater" Water 13, no. 12: 1615. https://doi.org/10.3390/w13121615
APA StyleNguyen, T. T., Chen, H.-H., To, T. H., Chang, Y.-C., Tsai, C.-K., Chen, K.-F., & Tsai, Y.-P. (2021). Development of Biochars Derived from Water Bamboo (Zizania latifolia) Shoot Husks Using Pyrolysis and Ultrasound-Assisted Pyrolysis for the Treatment of Reactive Black 5 (RB5) in Wastewater. Water, 13(12), 1615. https://doi.org/10.3390/w13121615






