Highest Composition Dissimilarity among Phytoplankton Communities at Intermediate Environmental Distances across High-Altitude Tropical Lakes
Abstract
1. Introduction
2. Materials and Methods
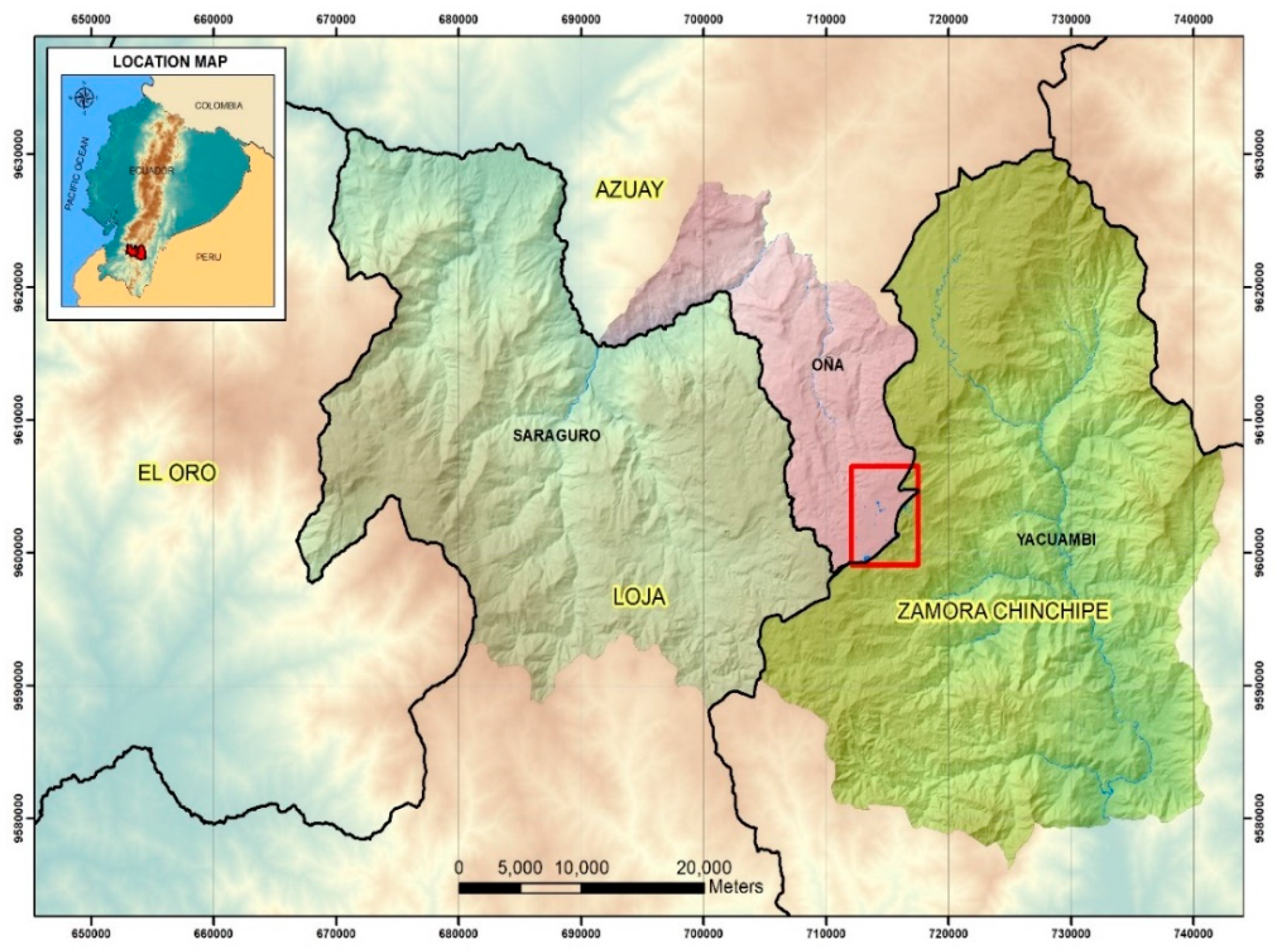
2.1. Studied Lakes
2.2. Sampling and Physicochemical Analyses
2.3. Phytoplankton Composition
2.4. Community Dissimilarity
2.5. Geographical Distances
2.6. Environmental Distances
2.7. Data Analysis
3. Results
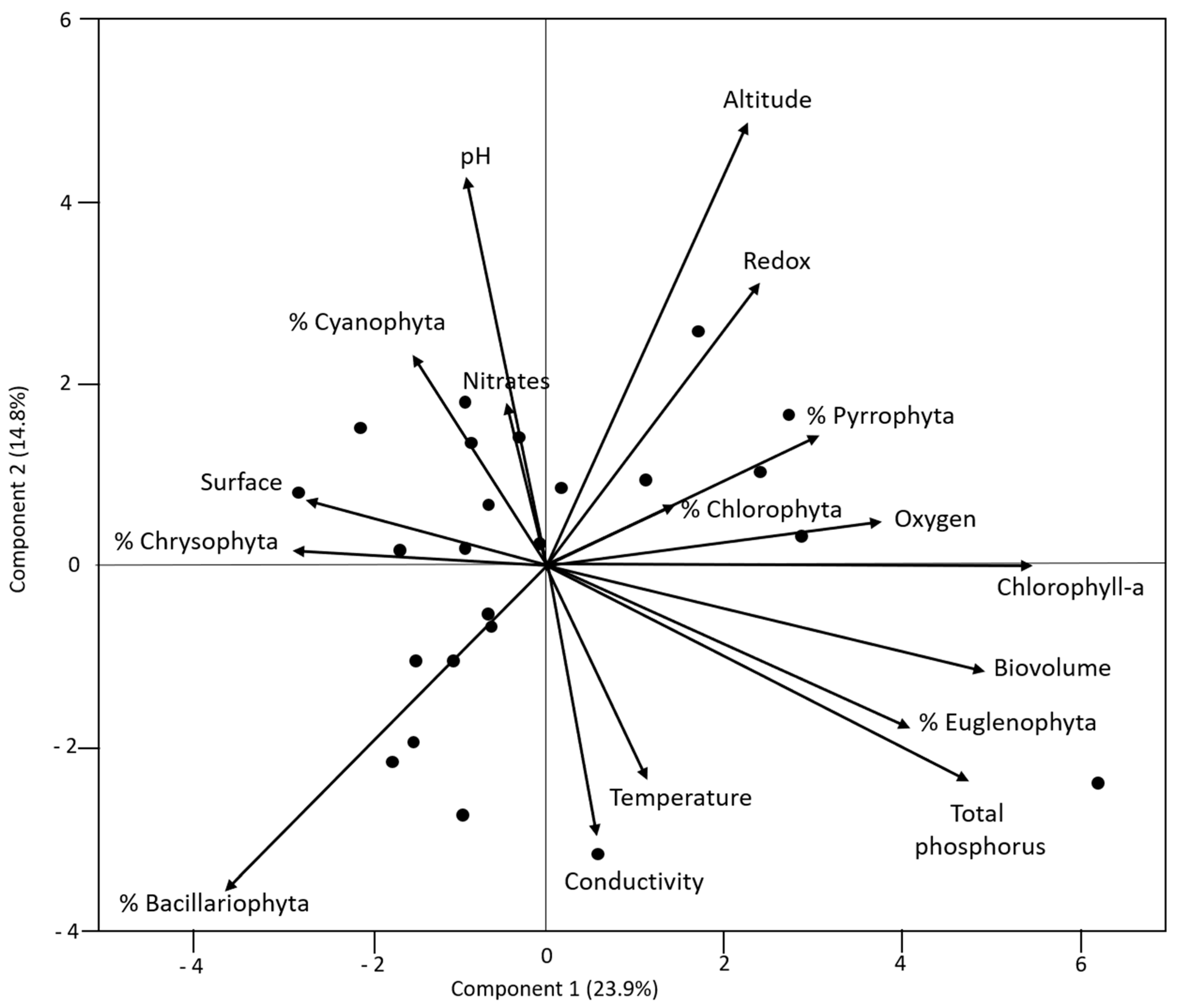
3.1. Links amongst Variables
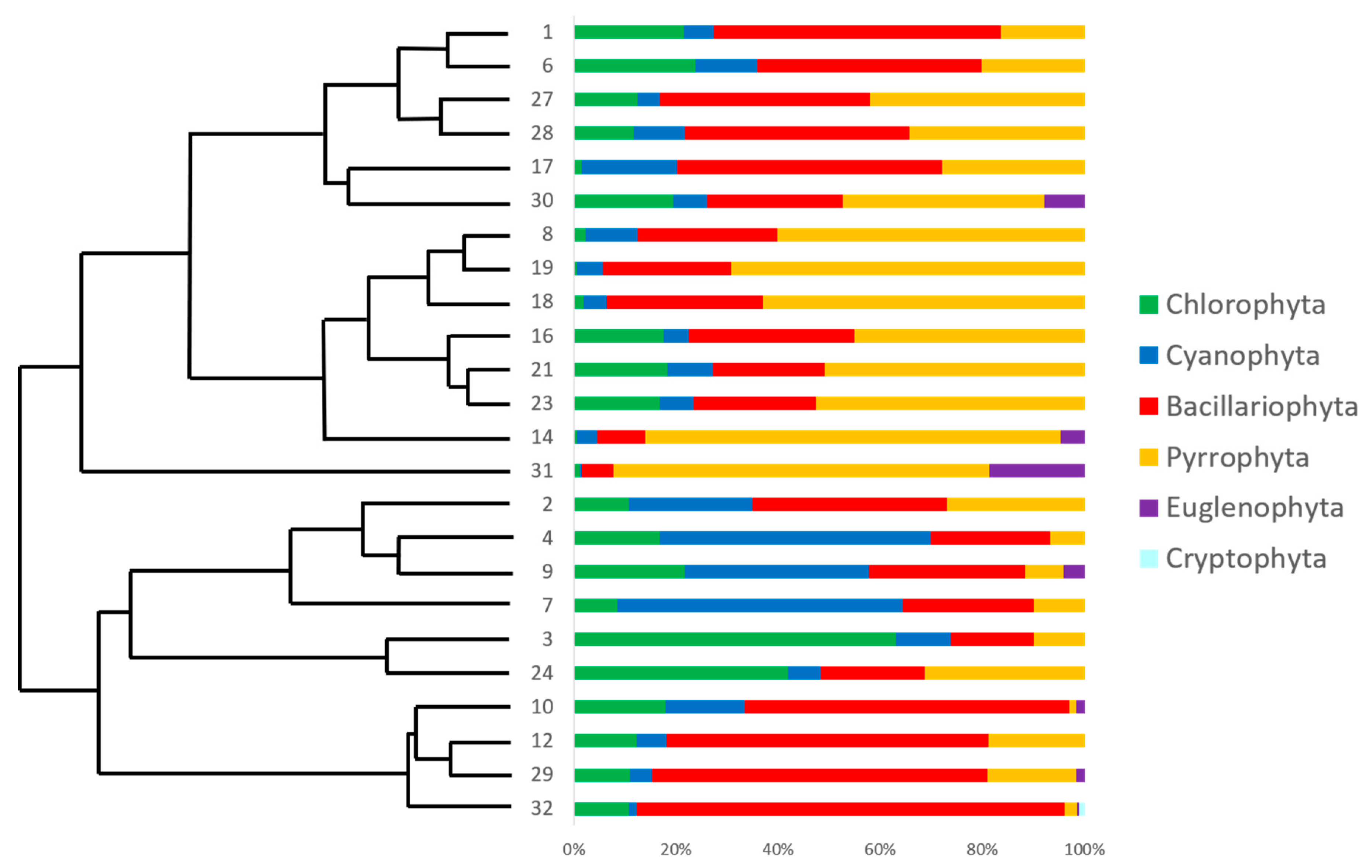
3.2. Phyla Community Structure
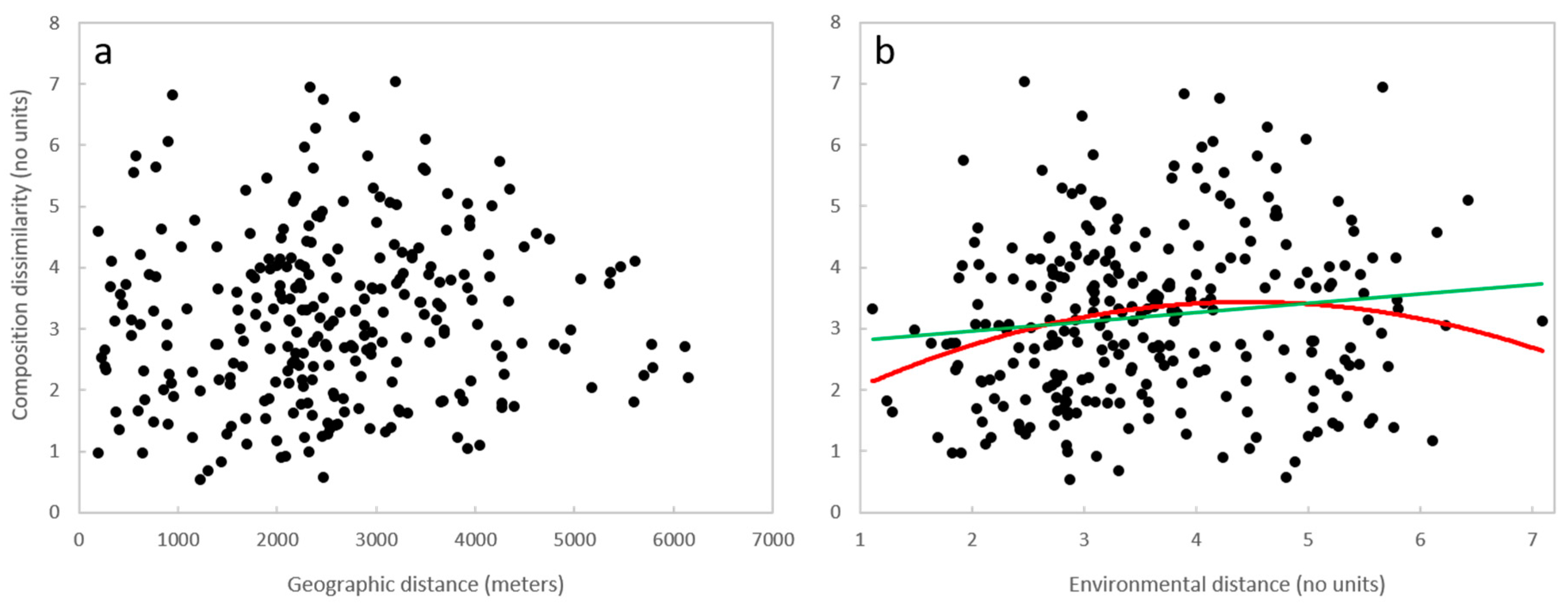
3.3. Geographic Distance, Environmental Distance, and Community Similarity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huston, M.A. Biological Diversity: The Coexistence of Species on Changing Landscapes; Cambridge University Press: Cambridge, UK, 1994; 681p. [Google Scholar]
- Chesson, P. Mechanisms of maintenance of species diversity. Annu. Rev. Ecol. Syst. 2000, 31, 343–366. [Google Scholar] [CrossRef]
- Wright, J.S. Plant diversity in tropical forests: A review of mechanisms of species coexistence. Oecologia 2002, 130, 1–14. [Google Scholar] [CrossRef]
- Nekola, J.C.; White, P.S. The distance decay of similarity in biogeography and ecology. J. Biogeogr. 1999, 26, 867–878. [Google Scholar] [CrossRef]
- Soininen, J.; McDonald, R.; Hillebrand, H. The distance decay of similarity in ecological communities. Ecography 2007, 30, 3–12. [Google Scholar] [CrossRef]
- Leibold, M.A.; Holyoak, M.; Mouquet, N.; Amarasekare, P.; Chase, J.M.; Hoopes, M.F.; Holt, R.D.; Shurin, J.B.; Law, R.; Tilman, D.; et al. The metacommunity concept: A framework for multi-scale community ecology. Ecol. Lett. 2004, 7, 601–613. [Google Scholar] [CrossRef]
- Hubbell, S.P. The Unified Neutral Theory of Biodiversity and Biogeography; Princeton University Press: Princeton, NJ, USA, 2001; 392p. [Google Scholar]
- Cottenie, K. Integrating environmental and spatial processes in ecological community dynamics. Ecol. Lett. 2005, 8, 1175–1182. [Google Scholar] [CrossRef]
- Thompson, R.; Townsend, C. A truce with neutral theory: Local deterministic factors, species traits and dispersal limitation together determine patterns of diversity in stream invertebrates. J. Anim. Ecol. 2006, 75, 476–484. [Google Scholar] [CrossRef]
- De Wit, R.; Bouvier, T. ‘Everything is everywhere, but, the environment selects’; what did Baas Becking and Beijerinck really say? Environ. Microbiol. 2006, 8, 755–758. [Google Scholar] [CrossRef] [PubMed]
- Finlay, B.J.; Fenchel, T. Cosmopolitan metapopulations of free-living microbial eukaryotes. Protist 2004, 155, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Finlay, B.J. Global dispersal of free-living microbial eukaryote species. Science 2002, 296, 1061–1063. [Google Scholar] [CrossRef] [PubMed]
- Green, J.L.; Holmes, A.J.; Westoby, M.; Oliver, I.; Briscoe, D.; Dangerfield, M.; Gillings, M.; Beattie, A.J. Spatial scaling of microbial eukaryote diversity. Nature 2004, 432, 747–750. [Google Scholar] [CrossRef] [PubMed]
- Heino, J.; Bini, L.M.; Karjalainen, S.M.; Mykrä, H.; Soininen, J.; Vieira, L.C.G.; Diniz-Filho, J.A.F. Geographical patterns of micro-organismal community structure: Are diatoms ubiquitously distributed across boreal streams? Oikos 2010, 119, 129–137. [Google Scholar] [CrossRef]
- Martiny, J.B.H.; Bohannan, B.J.M.; Brown, J.H.; Colwell, R.K.; Fuhram, J.A.; Green, J.L.; Horner-Devine, C.; Kane, M. Adams Krumins, J.; Ruske, C.R.; et al. Microbial biogeography: Putting microorganisms on the map. Nat. Rev. Microbiol. 2006, 4, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Telford, R.J.; Vandvik, V.; Birks, H.J.B. Dispersal limitations matter for microbial morphospecies. Science 2006, 312, 1015. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, R.J.; Grogan, D.W.; Taylor, J.W. Geographic barriers isolate endemic populations of hyperthermophilic archaea. Science 2003, 301, 976–978. [Google Scholar] [CrossRef]
- Astorga, A.; Oksanen, J.; Luoto, M.; Soininen, J.; Virtanen, R.; Muotka, T. Distance decay of similarity in freshwater communities: Do macro-and microorganisms follow the same rules? Glob. Ecol. Biogeogr. 2012, 21, 365–375. [Google Scholar] [CrossRef]
- Mazaris, A.D.; Moustaka-Gouni, M.; Michaloudi, E.; Bobori, D.C. Biogeographical patterns of freshwater micro-and macroorganisms: A comparison between phytoplankton, zooplankton and fish in the eastern Mediterranean. J. Biogeogr. 2010, 37, 1341–1351. [Google Scholar] [CrossRef]
- Goldenberg Vilar, A.; van Dam, H.; van Loon, E.E.; Vonk, J.A.; van Der Gest, H.G.; Admiraal, W. Eutrophication decreases distance decay of similarity in diatom communities. Freshw. Biol. 2014, 59, 1522–1531. [Google Scholar] [CrossRef]
- Soininen, J.; Kokocinski, M.; Estlander, S.; Kotanen, J.; Heino, J. Neutrality, niches, and determinants of plankton metacommunity structure across boreal wetland ponds. Ecoscience 2007, 14, 146–154. [Google Scholar] [CrossRef]
- Soininen, J.; Korhonen, J.J.; Karhu, J.; Vetterli, A. Disentangling the spatial patterns in community composition of prokaryotic and eukaryotic lake plankton. Limnol. Oceanogr. 2011, 56, 508–520. [Google Scholar] [CrossRef]
- Wetzel, C.E.; Bicudo, D.C.; Ector, L.; Lobo, E.A.; Soininen, J.; Landeiro, V.L.; Bini, L.M. Distance decay of similarity in neotropical diatom communities. PLoS ONE 2012, 7, e45071. [Google Scholar] [CrossRef] [PubMed]
- Pastorino, P.; Prearo, M. High-mountain lakes, indicators of global change: Ecological characterization and environmental pressures. Diversity 2020, 12, 260. [Google Scholar] [CrossRef]
- Buytaert, W.; Célleri, R.; De Bièvre, B.; Cisneros, F.; Wyseure, G.; Deckers, J.; Hofstede, R. Human impact on the hydrology of the Andean páramos. Earth Sci. Rev. 2006, 79, 53–72. [Google Scholar] [CrossRef]
- Van Colen, W.R.; Mosquera, P.V.; Vanderstukken, M.; Goiris, K.; Carrasco, M.-C.; Decaestecker, E.; Alonso, M.; León- Tamariz, F.; Muylaert, K. Limnology and trophic status of glacial lakes in the tropical Andes (Cajas National Park, Ecuador). Freshw. Biol. 2017, 62, 458–473. [Google Scholar] [CrossRef]
- Cartuche, A.; Guan, Z.; Ibelings, B.W.; Venail, P. Phytoplankton diversity relates negatively with productivity in tropical high-altitude lakes from southern Ecuador. Sustainability 2019, 11, 5235. [Google Scholar] [CrossRef]
- Hillebrand, H.; Dürselen, C.-D.; Kirschtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- Çelekli, A.; Öztürk, B.; Kapi, M. Relationship between phytoplankton composition and environmental variables in an artificial pond. Algal Res. 2014, 5, 37–41. [Google Scholar] [CrossRef]
- Chen, X.; Bu, Z.; Stevenson, M.A.; Cao, Y.; Zeng, L.; Qin, B. Variations in diatom communities at genus and species levels in peatlands (central China) linked to microhabitats and environmental factors. Sci. Total Environ. 2016, 568, 137–146. [Google Scholar] [CrossRef]
- Van der Gucht, K.; Cottenie, K.; Muylaert, K.; Vloemans, N.; Cousin, S.; Declerck, S.; Jeppesen, E.; Conde-Porcuna, J.-M.; Schwenk, K.; Zwart, G.; et al. The power of species sorting: Local factors drive bacterial community composition over a wide range of spatial scales. Proc. Natl. Acad. Sci. USA 2007, 104, 20404–20409. [Google Scholar] [CrossRef]
- Korhonen, J.J.; Soininen, J.; Hillebrand, H. A quantitative analysis of temporal turnover in aquatic species assemblages across ecosystems. Ecology 2010, 91, 508–517. [Google Scholar] [CrossRef]
- Barta, B.; Mouillet, C.; Espinosa, R.; Andino, P.; Jacobsen, D.; Christoffersen, K.S. Glacial-fed and páramo lake ecosystems in the tropical high Andes. Hydrobiologia 2018, 813, 19–32. [Google Scholar] [CrossRef]
- Michelutti, N.; Wolfe, A.P.; Cooke, C.A.; Hobbs, W.O.; Vuille, M.; Smol, J.P. Climate change forces new ecological states in tropical Andean lakes. PLoS ONE 2015, 10, e0115338. [Google Scholar] [CrossRef] [PubMed]
- Mouillet, C.; Barta, B.; Espinosa, R.; Andino, P.; Christoffersen, K.S.; Jacobsen, D. Ecological effects of introduced rainbow trout (Oncorhynchus mykiss) in pristine Ecuadorian high Andean lakes. Fundam. Appl. Limnol. 2018, 191, 323–337. [Google Scholar] [CrossRef]
| Variable | Minimum | Maximum | Mean |
|---|---|---|---|
| Area (ha) | 0.51 | 12.41 | 2.01 |
| Altitude (m.a.s.l.) | 3288 | 3362 | 3323 |
| Maximum depth (m) | 1.4 | 8.7 | 2.9 |
| Temperature (°C) | 10.7 | 15.29 | 13.21 |
| pH | 3.45 | 5.45 | 4.36 |
| Oxygen (mg L−1) | 6.7 | 7.48 | 7.07 |
| Redox (mv) | 189 | 321 | 248 |
| Conductivity (μS cm−1) | 23.6 | 51.4 | 36.4 |
| Total phosphorus (µg L−1) | 0.01 | 0.15 | 0.058 |
| Nitrates/nitrites (mg L−1) | 0.0015 | 0.0194 | 0.0080 |
| Chlorophyll-a (µg L−1) | 1.49 | 5.05 | 3.01 |
| Phytoplankton biovolume (µm3 mL−1 106) | 3.63 | 241.67 | 32.39 |
| Structure Variable | Environmental Variable | Coefficient of Correlation |
|---|---|---|
| % Bacillariophyta | Redox potential | −0.592 |
| % Bacillariophyta | Altitude | −0.466 |
| % Cyanophyta | Temperature | −0.466 |
| % Euglenophyta | Total phosphorus | 0.452 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cartuche, A.; Manoylov, K.M.; Ibelings, B.W.; Venail, P. Highest Composition Dissimilarity among Phytoplankton Communities at Intermediate Environmental Distances across High-Altitude Tropical Lakes. Water 2021, 13, 1378. https://doi.org/10.3390/w13101378
Cartuche A, Manoylov KM, Ibelings BW, Venail P. Highest Composition Dissimilarity among Phytoplankton Communities at Intermediate Environmental Distances across High-Altitude Tropical Lakes. Water. 2021; 13(10):1378. https://doi.org/10.3390/w13101378
Chicago/Turabian StyleCartuche, Alonso, Kalina M. Manoylov, Bastiaan W. Ibelings, and Patrick Venail. 2021. "Highest Composition Dissimilarity among Phytoplankton Communities at Intermediate Environmental Distances across High-Altitude Tropical Lakes" Water 13, no. 10: 1378. https://doi.org/10.3390/w13101378
APA StyleCartuche, A., Manoylov, K. M., Ibelings, B. W., & Venail, P. (2021). Highest Composition Dissimilarity among Phytoplankton Communities at Intermediate Environmental Distances across High-Altitude Tropical Lakes. Water, 13(10), 1378. https://doi.org/10.3390/w13101378






