Factors Controlling of Thermal Water Hydrogeochemical Characteristics in Tatun Volcano Group, Taiwan
Abstract
1. Introduction
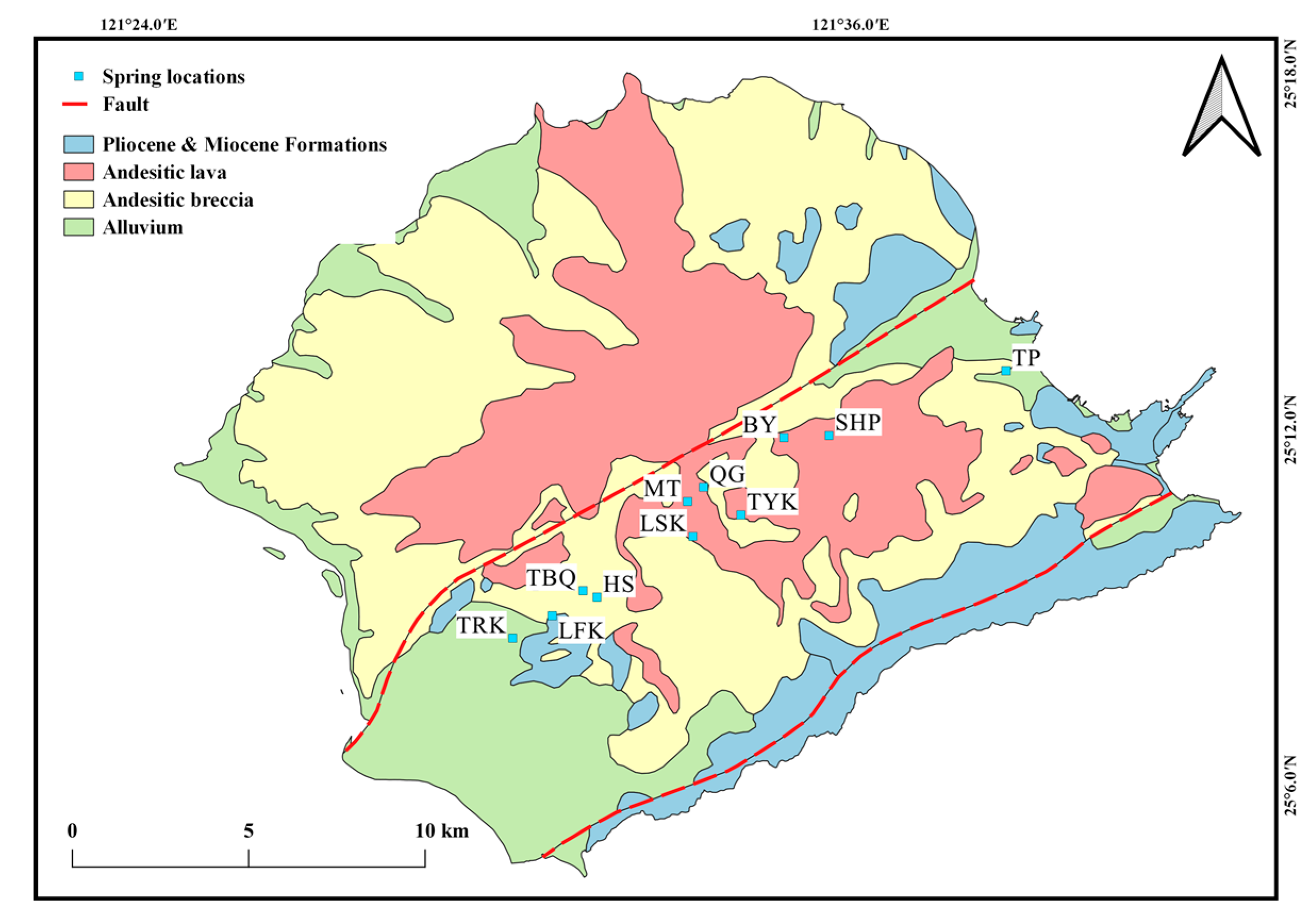
2. Study Area
3. Materials and Methods
3.1. Geothermometry
3.1.1. Chemical Geothermometry
3.1.2. Multicomponent Geothermometry
3.2. Oxygen and Hydrogen Isotopes
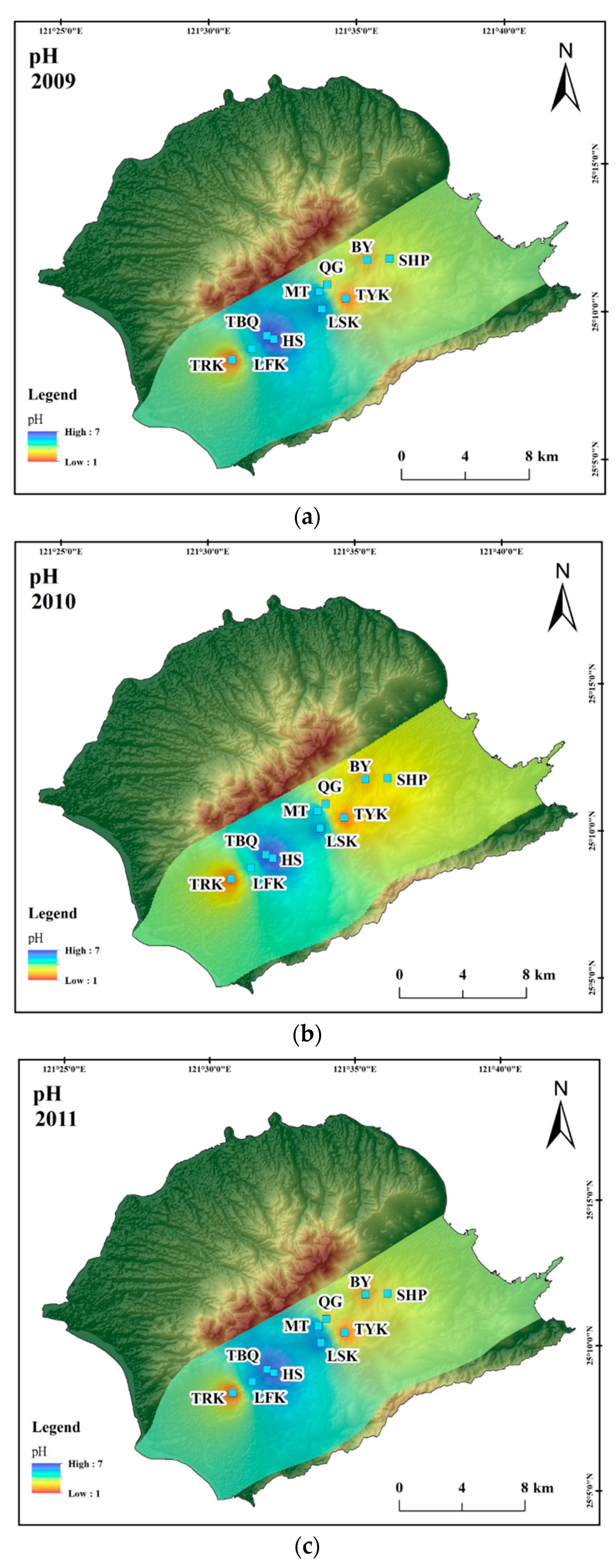
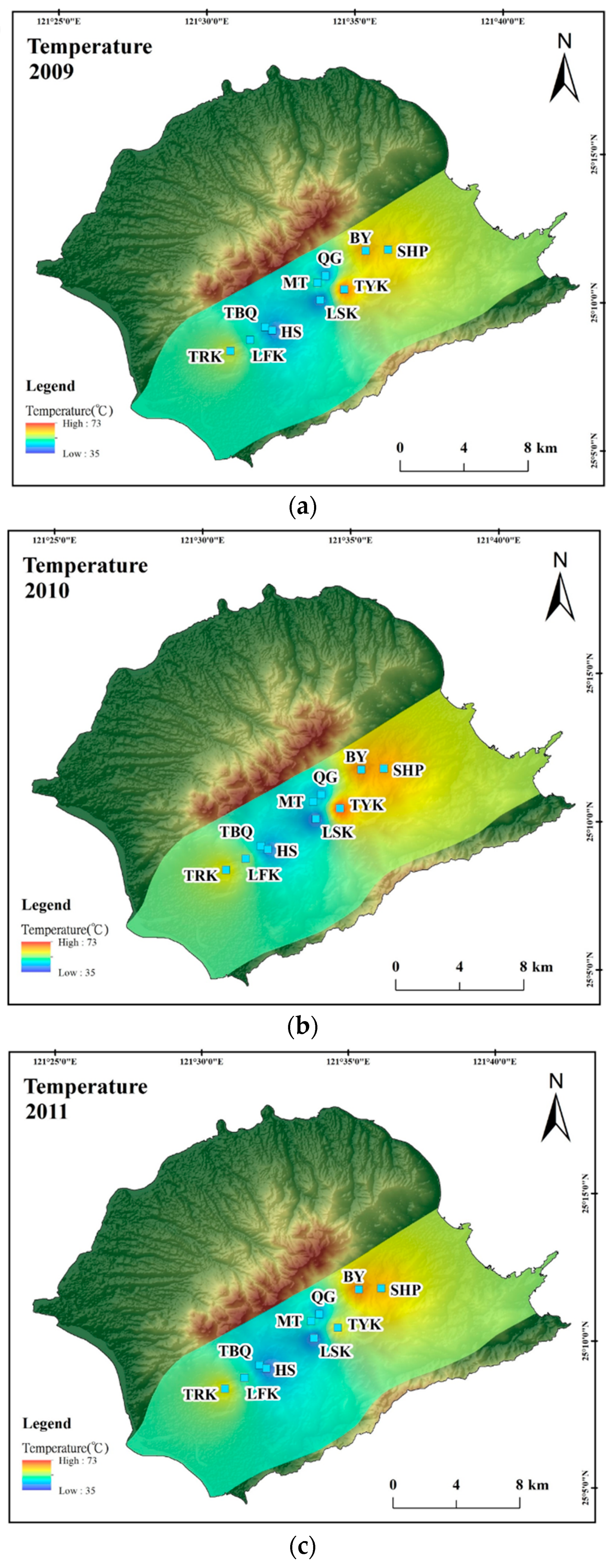
4. Results and Discussion
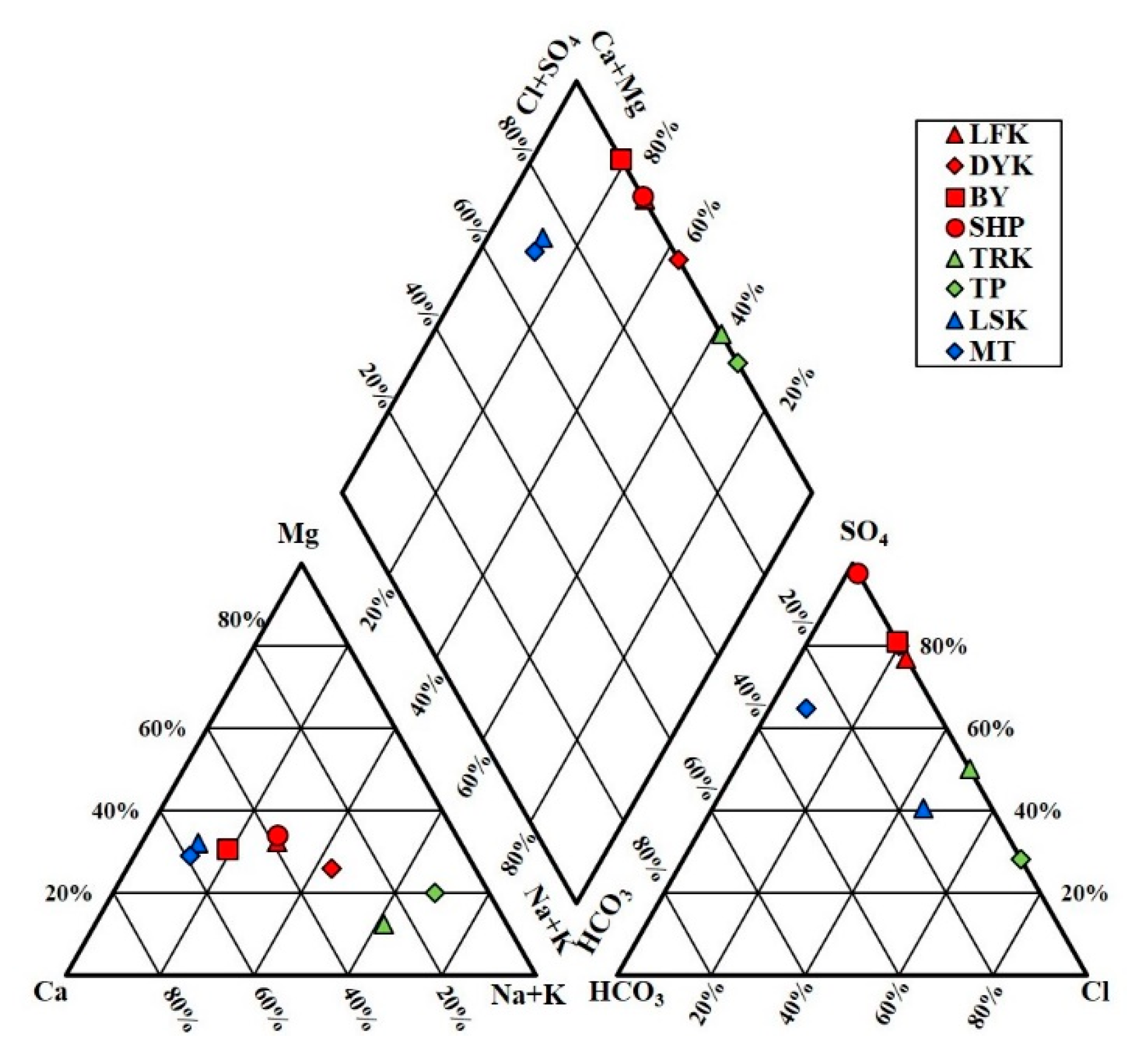
4.1. Hydrogeochemistry
4.2. Classical Geothermometry
4.3. Multicomponent Geothermometry
4.4. Isotope Characteristics
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, C.-M.; Song, S.-R.; Chen, Y.-L.; Tsao, S. Characteristics and Origins of Hot Springs in the Tatun Volcano Group in Northern Taiwan. Terr. Atmos. Ocean. Sci. 2011, 22, 475. [Google Scholar] [CrossRef]
- Blasco, M.; Gimeno, M.J.; Auqué, L.F. Low Temperature Geothermal Systems in Carbonate-Evaporitic Rocks: Mineral Equilibria Assumptions and Geothermometrical Calculations. Insights from the Arnedillo Thermal Waters (Spain). Sci. Total. Environ. 2018, 615, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Ba, J.; Chen, Q.; Pan, X. Hydrogeochemistry and Environmental Isotope Compositions and Their Evolution in Geothermal Groundwater in Yizhang, Hunan Province, China. Environ. Eng. Manag. J. 2019, 18, 765–774. [Google Scholar] [CrossRef]
- Mroczek, E.; Milicich, S.D.; Bixley, P.; Sepulveda, F.; Bertrand, E.; Soengkono, S.; Rae, A. Ohaaki Geothermal System: Refinement of a Conceptual Reservoir Model. Geothermics 2016, 59, 311–324. [Google Scholar] [CrossRef]
- Gao, Z.; Liu, J.; Li, F.; Wang, M.; Feng, J.; Wu, G. Hydrochemical Characteristics and Temporal Variations of Geothermal Water Quality in Tangtou, Shandong, China. Water 2019, 11, 1643. [Google Scholar] [CrossRef]
- Mohammadi, Z.; Bagheri, R.; Jahanshahi, R. Hydrogeochemistry and Geothermometry of Changal Thermal Springs, Zagros Region, Iran. Geothermics 2010, 39, 242–249. [Google Scholar] [CrossRef]
- Li, J.; Yang, G.; Sagoe, G.; Li, Y. Major Hydrogeochemical Processes Controlling the Composition of Geothermal Waters in the Kangding Geothermal Field, Western Sichuan Province. Geothermics 2018, 75, 154–163. [Google Scholar] [CrossRef]
- Wang, X.; Qiao, W.; Chen, J.; Liu, X.; Yang, F. Understanding the Burial and Migration Characteristics of Deep Geothermal Water Using Hydrogen, Oxygen, and Inorganic Carbon Isotopes. Water 2017, 10, 7. [Google Scholar] [CrossRef]
- Vespasiano, G.; Apollaro, C.; De Rosa, R.; Muto, F.; LaRosa, S.; Fiebig, J.; Mulch, A.; Marini, L. The Small Spring Method (SSM) for the Definition of Stable Isotope–Elevation Relationships in Northern Calabria (Southern Italy). Appl. Geochem. 2015, 63, 333–346. [Google Scholar] [CrossRef]
- Apollaro, C.; Vespasiano, G.; Muto, F.; De Rosa, R.; Barca, D.; Marini, L. Use of Mean Residence Time of Water, Flowrate, and Equilibrium Temperature Indicated by Water Geothermometers to Rank Geothermal Resources. Application to the Thermal Water Circuits of Northern Calabria. J. Volcanol. Geotherm. Res. 2016, 328, 147–158. [Google Scholar] [CrossRef]
- Doveri, M.; Lelli, M.; Marini, L.; Raco, B. Revision, Calibration, and Application of the Volume Method to Evaluate the Geothermal Potential of Some Recent Volcanic Areas of Latium, Italy. Geothermics 2010, 39, 260–269. [Google Scholar] [CrossRef]
- Chambefort, I.; Buscarlet, E.; Wallis, I.; Sewell, S.; Wilmarth, M. Ngatamariki Geothermal Field, New Zealand: Geology, Geophysics, Chemistry and Conceptual Model. Geothermics 2016, 59, 266–280. [Google Scholar] [CrossRef]
- Alam, B.Y.C.S.S.S.; Itoi, R.; Taguchi, S.; Saibi, H.; Yamashiro, R. Hydrogeochemical and Isotope Characterization of Geothermal Waters from the Cidanau Geothermal Field, West Java, Indonesia. Geothermics 2019, 78, 62–69. [Google Scholar] [CrossRef]
- Vespasiano, G.; Apollaro, C.; Muto, F.; Dotsika, E.; De Rosa, R.; Marini, L. Chemical and Isotopic Characteristics of the Warm and Cold Waters of the Luigiane Spa near Guardia Piemontese (Calabria, Italy) in a Complex Faulted Geological Framework. Appl. Geochem. 2014, 41, 73–88. [Google Scholar] [CrossRef]
- Xu, T.; Hou, Z.; Jia, X.; Spycher, N.; Jiang, Z.; Feng, B.; Na, J.; Yuan, Y. Classical and Integrated Multicomponent Geothermometry at the Tengchong Geothermal Field, Southwestern China. Environ. Earth Sci. 2016, 75, 1502. [Google Scholar] [CrossRef]
- Martínez-Florentino, A.K.; Esteller, M.; Domínguez-Mariani, E.; Expósito, J.L.; Paredes, J. Hydrogeochemistry, Isotopes and Geothermometry of Ixtapan de la Sal–Tonatico Hot Springs, Mexico. Environ. Earth Sci. 2019, 78, 600. [Google Scholar] [CrossRef]
- Aydin, H.; Karakuş, H.; Mutlu, H. Hydrogeochemistry of Geothermal Waters in Eastern Turkey: Geochemical and Isotopic Constraints on Water-Rock Interaction. J. Volcanol. Geotherm. Res. 2020, 390, 106708. [Google Scholar] [CrossRef]
- Karolytė, R.; Serno, S.; Johnson, G.; Gilfillan, S.M.V. The Influence of Oxygen Isotope Exchange between CO2 and H2O in Natural CO2 -Rich Spring Waters: Implications for Geothermometry. Appl. Geochem. 2017, 84, 173–186. [Google Scholar] [CrossRef]
- Chen, C.H.; Lin, S.B. Eruptions Younger than 20 ka of the Tatun Volcano Group as Viewed from the Sediments of the Sungshan Formation in Taipei Basin. West. Pac. Earth Sci. 2002, 2, 191–204. [Google Scholar]
- Yang, T.; Sano, Y.; Song, S. 3He/4He Ratios of Fumaroles and Bubbling Gases of Hot Springs in Tatun Volcano Group, North Taiwan. Il Nuovo Cim. C 1999, 22, 281–286. [Google Scholar]
- Lee, H.-F.; Yang, T.F.; Lan, T.F.; Chen, C.-H.; Song, S.-R.; Tsao, S. Temporal Variations of Gas Compositions of Fumaroles in the Tatun Volcano Group, Northern Taiwan. J. Volcanol. Geotherm. Res. 2008, 178, 624–635. [Google Scholar] [CrossRef]
- Konstantinou, K.I.; Lin, C.-H.; Liang, W.-T. Seismicity Characteristics of a Potentially Active Quaternary Volcano: The Tatun Volcano Group, Northern Taiwan. J. Volcanol. Geotherm. Res. 2007, 160, 300–318. [Google Scholar] [CrossRef]
- Ohba, T.; Sawa, T.; Taira, N.; Yang, T.F.; Lee, H.F.; Lan, T.F.; Ohwada, M.; Morikawa, N.; Kazahaya, K. Magmatic Fluids of Tatun Volcanic Group, Taiwan. Appl. Geochem. 2010, 25, 513–523. [Google Scholar] [CrossRef]
- Dobson, P.; Gasperikova, E.; Spycher, N.; Lindsey, N.J.; Guo, T.R.; Chen, W.-S.; Liu, C.H.; Wang, C.-J.; Chen, S.-N.; Fowler, A.P. Conceptual Model of the Tatun Geothermal System, Taiwan. Geothermics 2018, 74, 273–297. [Google Scholar] [CrossRef]
- Chen, C.H.; Wu, Y.J. Volcanic Geology of the Tatun Geothermal Area, Northern Taiwan. Proc. Geol. Soc. China 1971, 14, 5–20. [Google Scholar]
- Mining Research and Service Organization. The Geothermal Exploration of the Tatun Volcano Group (III); MRSO Report; MRSO: Taipei, Taiwan, 1971; p. 111. (In Chinese) [Google Scholar]
- Song, M.; Guan, Y. The Electronic Government Performance of Environmental Protection Administrations in Anhui Province, China. Technol. Forecast. Soc. Chang. 2015, 96, 79–88. [Google Scholar] [CrossRef]
- Industrial Technology Research Institute. High Performance Geothermal Power Generation Technology Research Project. 103 D0102. Research Institution Energy Technology Development Program 2014 Executive Report; Ministry of Economic Affairs: Taipei, Taiwan, 2014. (In Chinese)
- Tsai, J.H. Oxygen and Hydrogen Isotope Studies of Surface Water and Groundwaters in the Tatun Volcano Group. Master’s Thesis, Chinese Culture University, Taipei, Taiwan, 2013. (In Chinese). [Google Scholar]
- Fournier, R. Chemical Geothermometers and Mixing Models for Geothermal Systems. Geothermics 1977, 5, 41–50. [Google Scholar] [CrossRef]
- Fournier, R.O.; Potter, R. Revised and Expanded Silica (Quartz) Geothermometer. Geotherm. Resour. Counc. Bull. 1982, 11, 10. [Google Scholar]
- Fournier, R.O. A Revised Equation for the Na/K Geothermometer. J. Water. Resour. Prot. 1979, 3, 221–224. [Google Scholar]
- Fournier, R.O. Water Geothermometers Applied to Geothermal Energy. In Application of Geochemistry in Geothermal Reservoir Development; Center on Small Energy Resources: Rome, Italy, 1991; pp. 37–69. [Google Scholar]
- Reed, M.; Spycher, N. Calculation of pH and Mineral Equilibria in Hydrothermal Waters with Application to Geothermometry and Studies of Boiling and Dilution. Geochim. Cosmochim. Acta 1984, 48, 1479–1492. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C. Description of Input and Examples for PHREEQC Version 3: A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; US Geological Survey: Reston, VA, USA, 2013. [CrossRef]
- Charlton, S.R.; Parkhurst, D.L. Modules Based on the Geochemical Model Phreeqc for Use in Scripting and Programming Languages. Comput. Geosci. 2011, 37, 1653–1663. [Google Scholar] [CrossRef]
- Yeh, H.-F.; Lin, H.-I.; Lee, C.-H.; Hsu, K.-C.; Wu, C.-S. Identifying Seasonal Groundwater Recharge Using Environmental Stable Isotopes. Water 2014, 6, 2849–2861. [Google Scholar] [CrossRef]
- Craig, H. Isotopic Variations in Meteoric Waters. Science 1961, 133, 1702–1703. [Google Scholar] [CrossRef]
- Van der Straaten, C.M.; Mook, W.G. Stable Isotopic Composition of Precipitation and Climatic Variability. In Palaeoclimates and Palaeowaters: A Collection of Environ- Mental Isotope Studies; IAEA: Vienna, Austria, 1983. [Google Scholar]
- Peng, T.R.; Wang, C.H.; Huang, C.C.; Fei, L.Y.; Chen, C.T.A.; Hwong, J.L. Stable Isotopic Characteristic of Taiwan’s Precipitation: A Case Study of Western Pacific Monsoon Region. Earth Planet. Sci. Lett. 2010, 289, 357–366. [Google Scholar] [CrossRef]
- Piper, A.M. A Graphic Procedure in the Geochemical Interpretation of Water-Analyses. Trans. Am. Geophys. Union 1944, 25, 914–928. [Google Scholar] [CrossRef]
- Ohsawa, S.; Lee, H.F.; Liang, B.; Komori, S.; Chen, C.H.; Kagiyama, T. Geochemical Characteristics and Origins of Acid Hot Spring Waters in Tatun Volcanic Group, Taiwan. J. Hot Spring Sci. 2013, 62, 282–293. [Google Scholar]
- Tseng, H.C. 3D Geological Structure and Potentials of Geothermal Power in the Tatun Volcano Group. Master’s Thesis, National Taiwan University, Taipei, Taiwan, 2014. (In Chinese). [Google Scholar]
- Giggenbach, W.F. Geothermal Solute Equilibria. Derivation of Na-K-Mg-Ca Geoindicators. Geochim. Cosmochim. Acta 1988, 52, 2749–2765. [Google Scholar] [CrossRef]
- Liu, K.K.; Chen, C.C.; Shieh, Y.N.; Chiang, S.C. Hydrogen and Oxygen Isotopic Compositions of Meteoric Waters from the Tatun Shan Area, Northern Taiwan. Bull. Inst. Earth Sci. 1984, 4, 159–175. (In Chinese) [Google Scholar]
- Ignacio, S.T.-A.; Muharrem, S.; Daniel, P.Z.; Peter, B. Stable Isotope Composition of Hydrothermally Altered Rocks and Hydrothermal Minerals at the Los Azufres Geothermal Field, Mexico. Turk. J. Earth Sci. 2012, 21, 127–143. [Google Scholar]


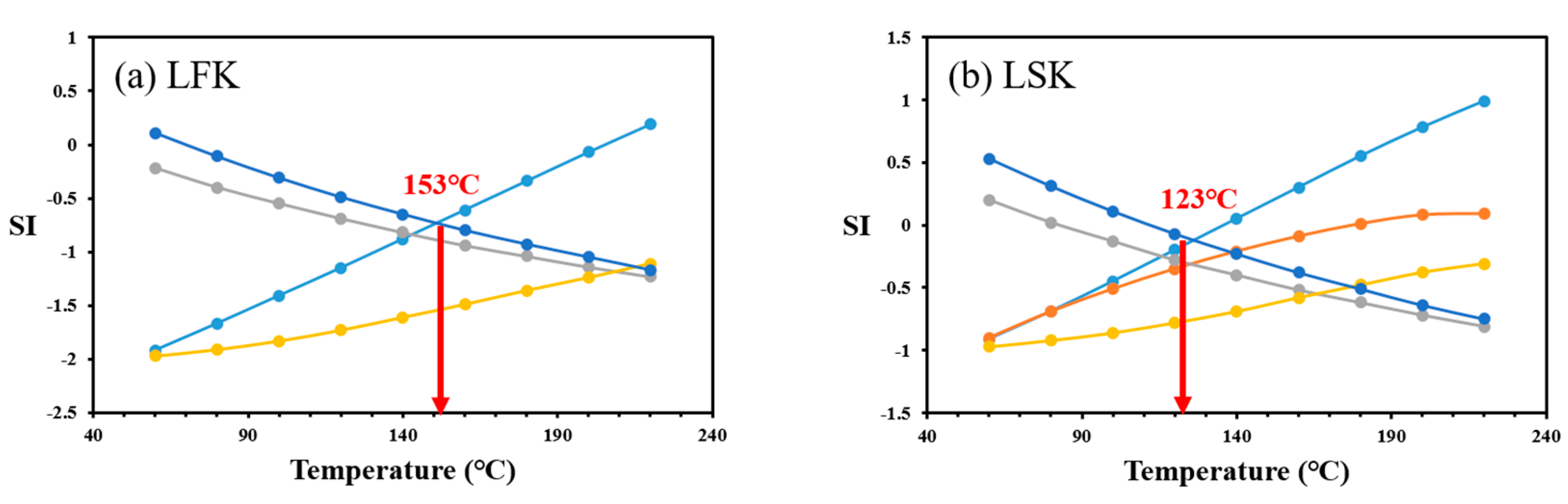
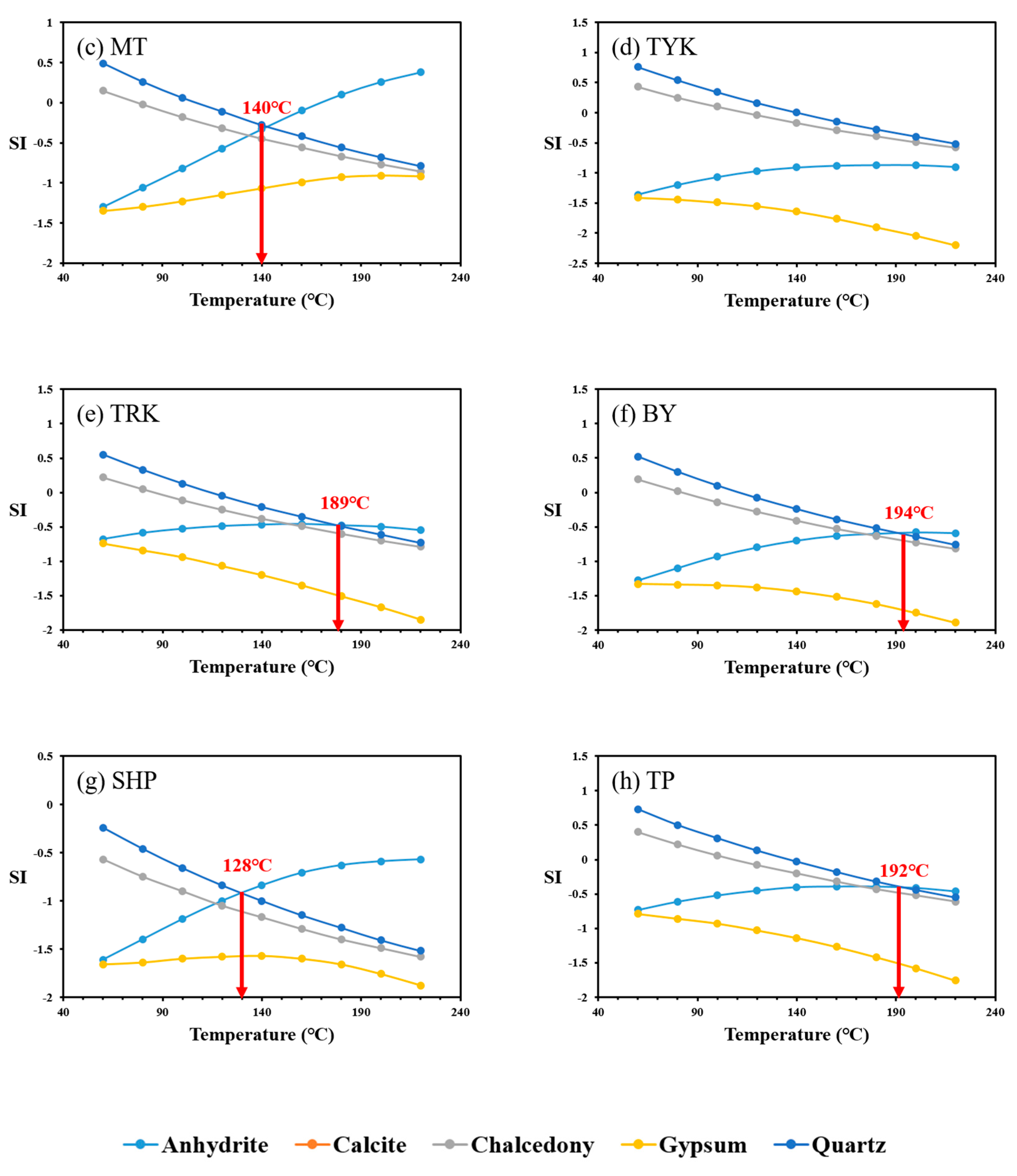
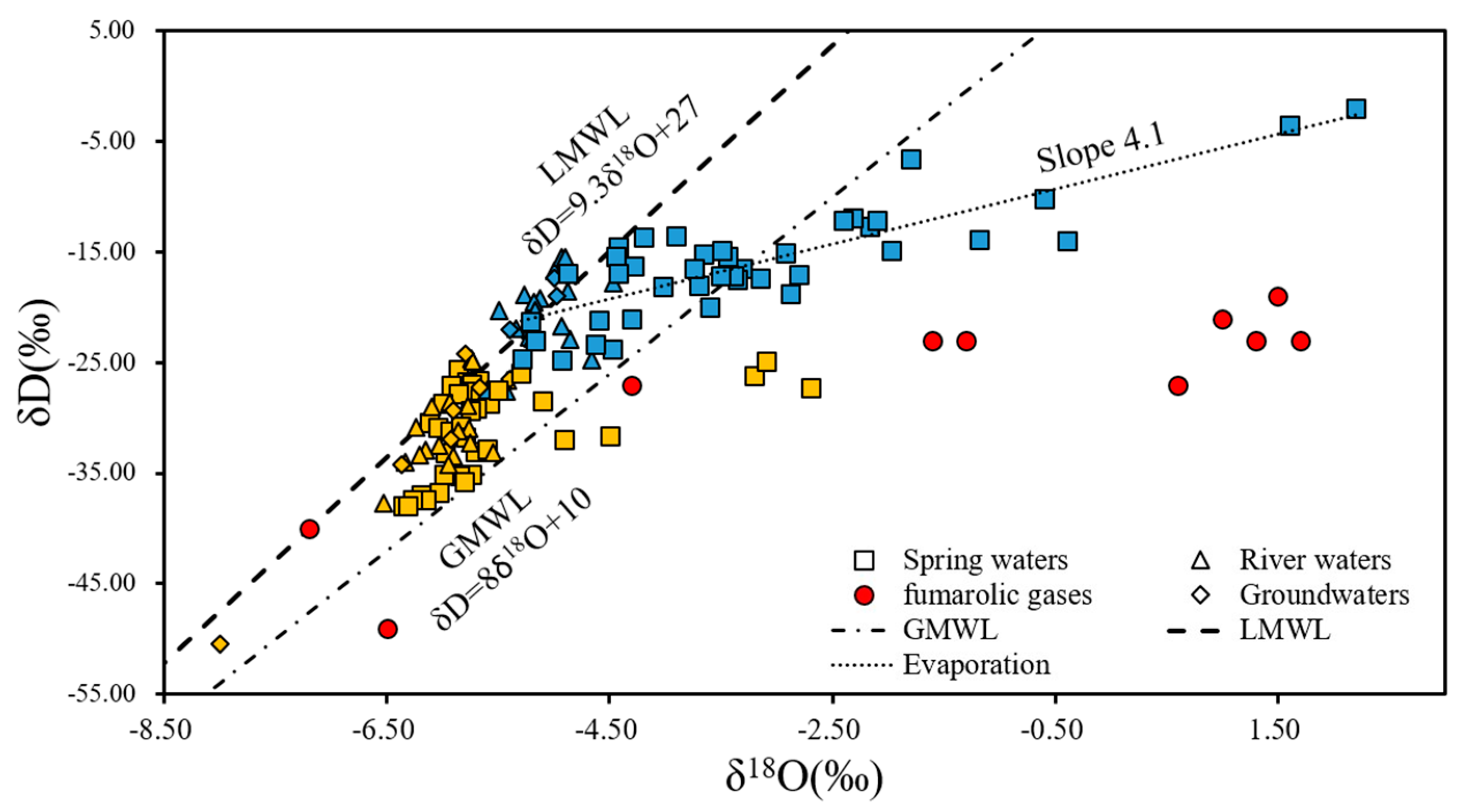
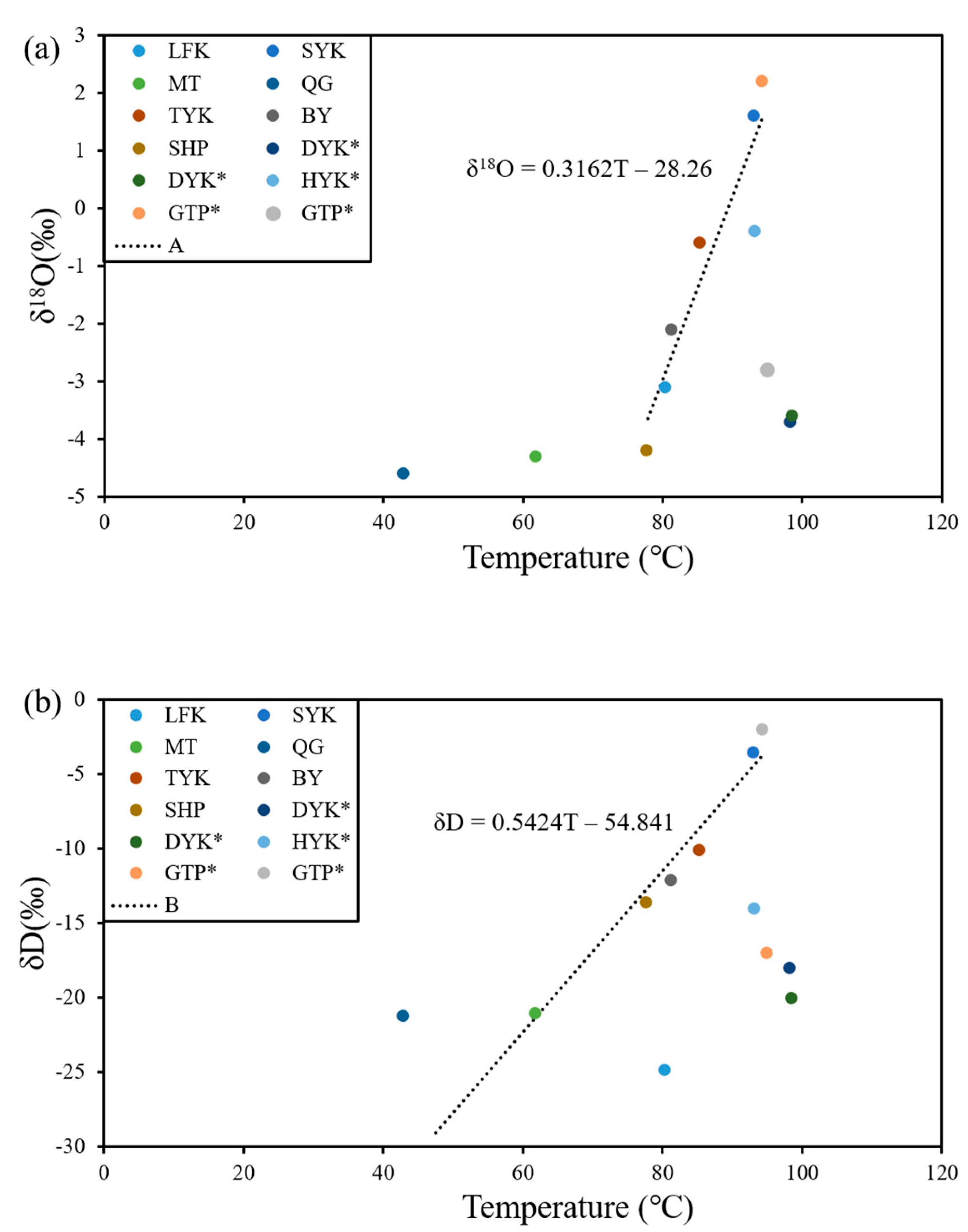
| Geothermometer | Equation |
|---|---|
| Chalcedony [30] | |
| Quartz [31] | |
| Na-K [32] | |
| K-Mg [33] |
| Location | Temp (°C) | EC | pH | TDS | CI− | SO42− | HCO3− | SiO2 | Na+ | Ca2+ | K+ | Mg2+ |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LFK | 49.10 | 295.00 | 5.71 | 185.00 | 23.11 | 104.20 | - | 23.50 | 11.60 | 20.50 | 9.84 | 10.30 |
| LSK | 40.20 | 1321.00 | 6.03 | 900.00 | 187.50 | 312.50 | 255.30 | 61.20 | 30.50 | 172.00 | 19.40 | 60.20 |
| MT | 53.40 | 611.00 | 3.93 | 392.00 | 11.26 | 178.60 | 104.30 | 55.40 | 10.30 | 71.30 | 10.10 | 21.20 |
| TYK | 73.70 | 12,960.00 | 2.00 | 10,740.00 | 935.20 | 5128.00 | - | 102.00 | 20.30 | 20.20 | 21.30 | 10.40 |
| TRK | 55.80 | 19,540.00 | 1.58 | 17,080.00 | 2684.00 | 3658.00 | - | 62.40 | 451.00 | 213.00 | 212.00 | 61.20 |
| BY | 65.20 | 2043.00 | 2.50 | 1424.00 | 104.70 | 611.70 | - | 60.30 | 10.40 | 41.30 | 12.50 | 15.30 |
| SHP | 63.80 | 976.00 | 2.91 | 646.00 | 6.54 | 421.10 | - | 10.40 | 11.30 | 20.70 | 10.30 | 11.30 |
| TP | 69.80 | 22.10 | 1.87 | 19.30 | 4284.00 | 2270.00 | - | 91.70 | 1524.00 | 251.00 | 278.00 | 261.00 |
| Sample | Spring T (°C) | Chalcedony (°C) | Quartz (°C) | Na-K (°C) | K2-Mg (°C) |
|---|---|---|---|---|---|
| LFK | 49.1 | 37.7 | 69.9 | 533.0 | 6.7 |
| LSK | 40.2 | 82.3 | 111.7 | 471.4 | 12.8 |
| MT | 53.4 | 77.1 | 107.0 | 568.1 | 16.9 |
| TYK | 73.7 | 111.7 | 138.4 | 585.6 | −13.9 |
| TRK | 55.8 | 83.3 | 112.7 | 416.0 | −45.0 |
| BY | 65.2 | 81.5 | 111.0 | 622.9 | 5.5 |
| SHP | 63.8 | 7.8 | 39.1 | 550.1 | 6.8 |
| TP | 69.8 | 105.2 | 132.5 | 285.8 | −35.9 |
| Location | Anhydrite | Chalcedony | Chrysotile | Gypsum | Halite | Quartz | Sepiolite | Sylvite | Talc |
|---|---|---|---|---|---|---|---|---|---|
| LFK | −2.05 | −0.11 | −12.74 | −1.99 | −8.14 | 0.24 | −9.72 | −7.86 | −8.98 |
| LSK | −1.12 | 0.39 | −9.02 | −0.98 | −6.86 | 0.78 | −6.11 | −6.69 | −4.35 |
| MT | −1.37 | 0.22 | −21.47 | −1.36 | −8.52 | 0.56 | −5.92 | −8.21 | −17.01 |
| TYK | −1.25 | 0.31 | −33.09 | −1.43 | −6.47 | 0.61 | −23.33 | −6.22 | −28.25 |
| TRK | −0.71 | 0.26 | −34.83 | −0.72 | −4.67 | 0.60 | −24.04 | −4.69 | −30.25 |
| BY | −1.23 | 0.14 | −29.75 | −1.33 | −7.59 | 0.46 | −21.13 | −7.23 | −25.32 |
| SHP | −1.57 | −0.61 | −29.19 | −1.66 | −8.74 | −0.29 | −21.97 | −8.49 | −26.27 |
| TP | −0.67 | 0.31 | −29.48 | −0.82 | −3.97 | 0.61 | −20.81 | −4.46 | −24.67 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, H.-H.; Yeh, H.-F. Factors Controlling of Thermal Water Hydrogeochemical Characteristics in Tatun Volcano Group, Taiwan. Water 2020, 12, 2473. https://doi.org/10.3390/w12092473
Hsu H-H, Yeh H-F. Factors Controlling of Thermal Water Hydrogeochemical Characteristics in Tatun Volcano Group, Taiwan. Water. 2020; 12(9):2473. https://doi.org/10.3390/w12092473
Chicago/Turabian StyleHsu, Hung-Hsiang, and Hsin-Fu Yeh. 2020. "Factors Controlling of Thermal Water Hydrogeochemical Characteristics in Tatun Volcano Group, Taiwan" Water 12, no. 9: 2473. https://doi.org/10.3390/w12092473
APA StyleHsu, H.-H., & Yeh, H.-F. (2020). Factors Controlling of Thermal Water Hydrogeochemical Characteristics in Tatun Volcano Group, Taiwan. Water, 12(9), 2473. https://doi.org/10.3390/w12092473






