Towards the Removal of Antibiotics Detected in Wastewaters in the POCTEFA Territory: Occurrence and TiO2 Photocatalytic Pilot-Scale Plant Performance
Abstract
:Highlights:
- Antibiotics mass loadings range from 11,332 mg/day·1000 inhabitants to undetectable levels.
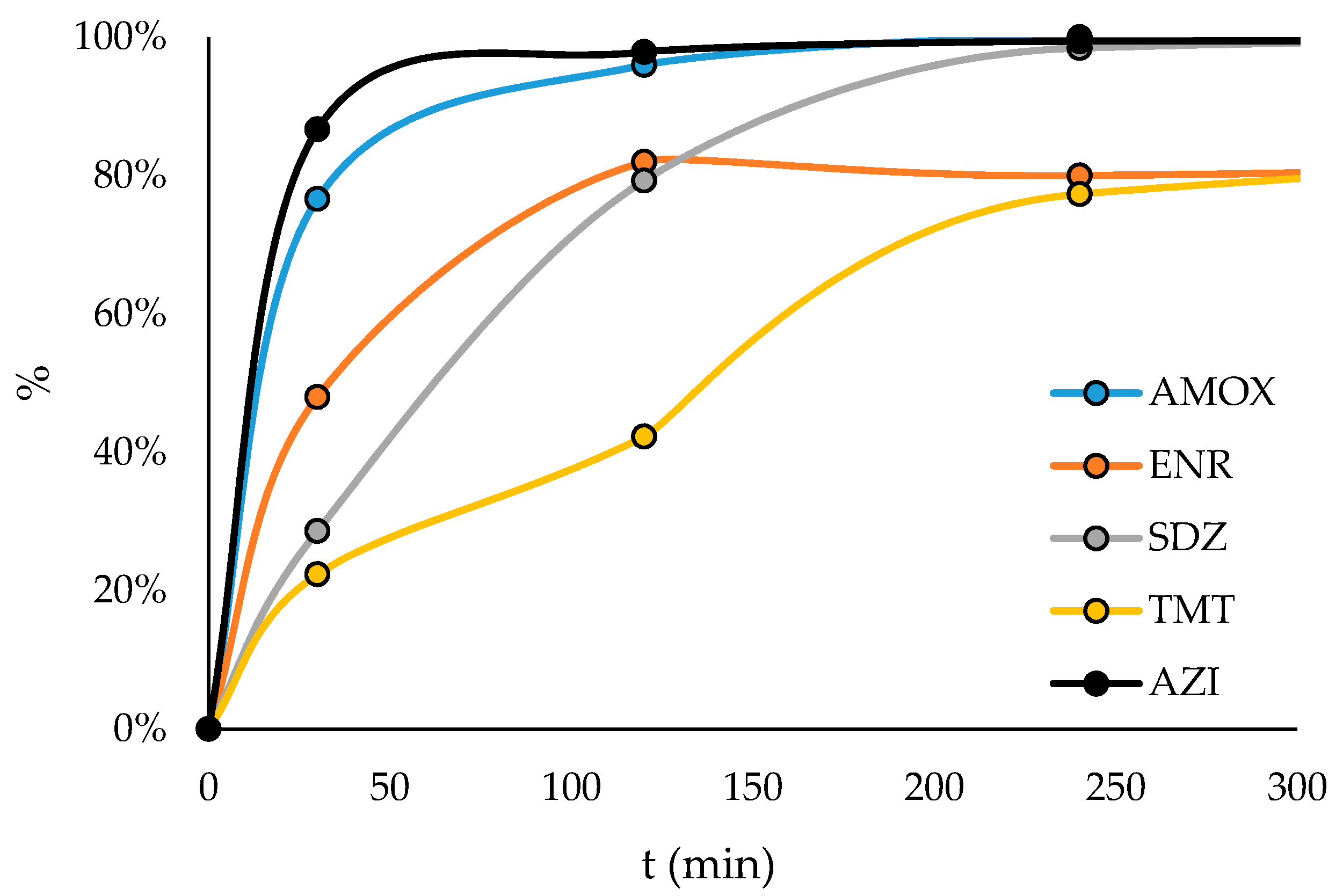
- Sulfadiazine, amoxicillin and azithromycin can be removed from wastewaters, while 80% of trimethoprim and enrofloxacin removal can be achieved after the photocatalytic treatment.
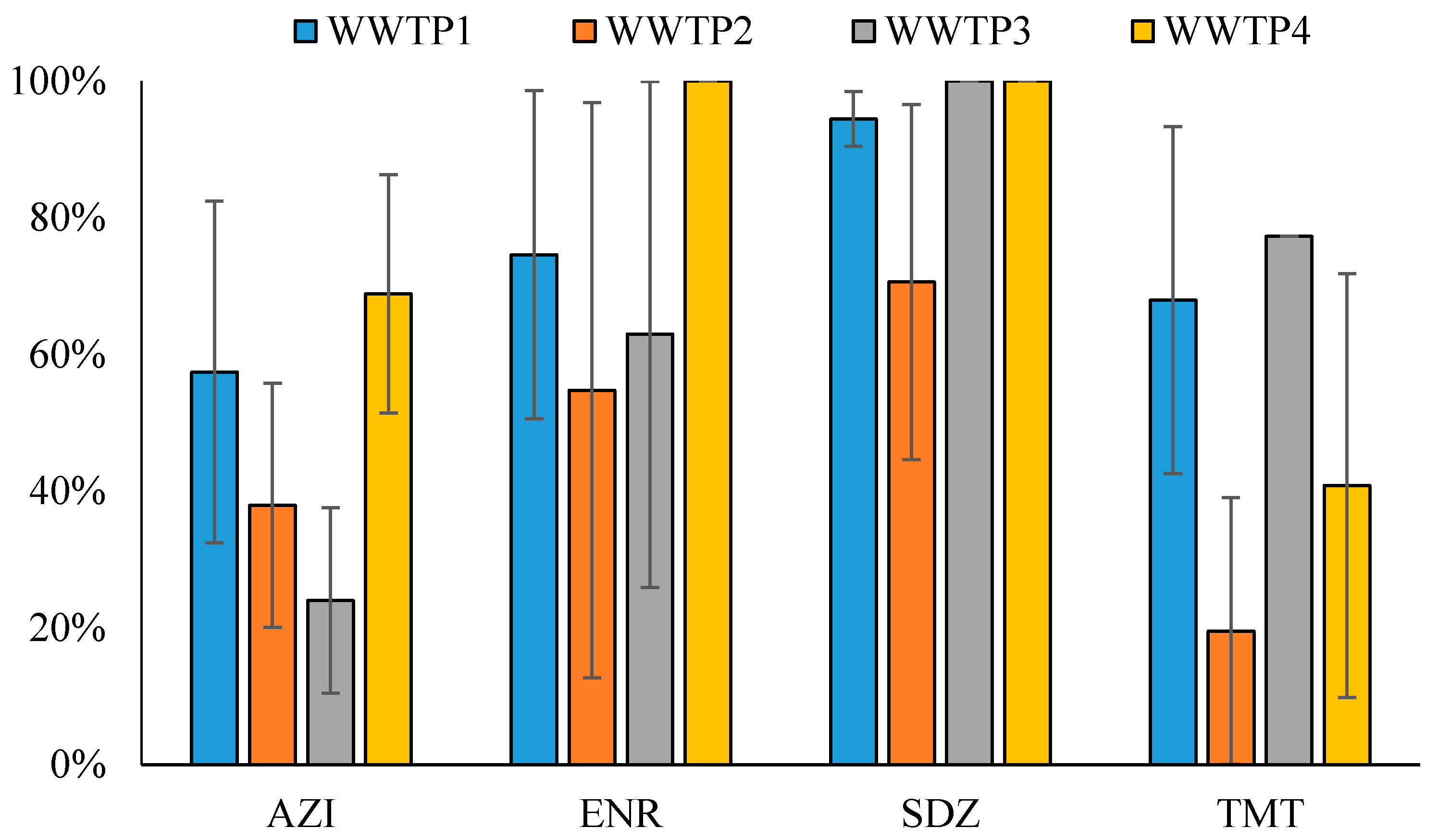
- The facilities provided with trickling filters proved to be more effective in removing antibiotics from wastewaters.
1. Introduction
2. Materials and Methods
2.1. Site Description and Sample Collection
2.2. Antibiotic Characterization
2.3. Total Ti Assessment in the Effluent
2.4. Photocatalytic Oxidation Experiment
3. Results
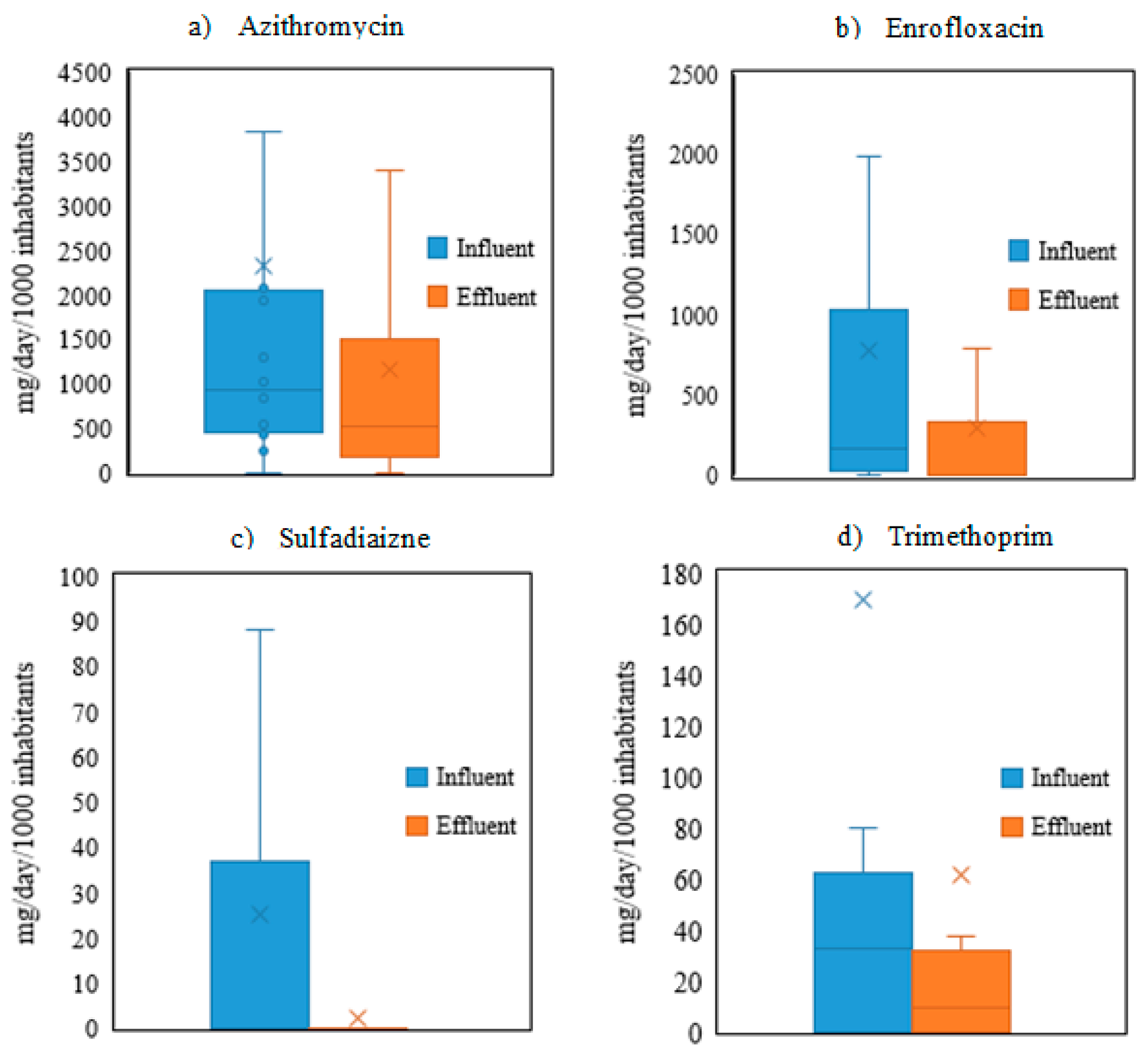
3.1. Occurrence of Target Antibiotics in Urban Wastewaters
3.2. Removal Efficiency of Selected WWTPs
3.3. Photocatalytic Oxidation of Antibiotics
3.4. Ti Assessment in the Effluent of the Photocatalytic Treatment Plant
4. Conclusions
- The mass loadings of the antibiotics ranged from 11,332 mg/day·1000 inhabitants to undetectable levels. Azithromycin had the highest mass loadings, followed by enrofloxacin, trimethoprim, sulfadiazine and amoxicillin.
- The use of enrofloxacin and azithromycin increased in the locations of the WWTPs during the period of this study.
- Sulfadiazine, amoxicillin and azithromycin were totally removed from wastewaters in the TiO2 photocatalytic pilot-scale plant, while 80% removal of trimethoprim and enrofloxacin was achieved by the treatment. Moreover, the facility was able to recover the catalyst after the treatment, minimizing the Ti released into the environment and allowing catalyst reuse.
- Although WWTPs are not designed to remove antibiotics, they do reduce them. This research shows that biological treatments have a significant influence on antibiotic removal. In particular, the presence of a trickling filter in the water treatment line of the WWTPs has been demonstrated to lead to a higher degree of antibiotic removal. However, the efficiency of the antibiotic removal depends on the physicochemical properties of the antibiotics and on the characteristics of the wastewater.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klein, E.Y.; Van Boeckel, T.P.; Martinez, E.M.; Pant, S.; Gandra, S.; evin, S.A.; Goossens, H.; Laxminarayan, R. Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc. Natl. Acad. Sci. USA 2018, 115, E3463–E3470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, L.H.; Gros, M.; Rodriguez-Mozaz, S.; Delerue-Matos, C.; Pena, A.; Barceló, D.; Montenegro, M.C. Contribution of hospital effluents to the load of pharmaceuticals in urban wastewaters: Identification of ecologically relevant pharmaceuticals. Sci. Total Environ. 2013, 461–462, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Mceneff, G.; Barron, L.; Kelleher, B.; Paull, B.; Quinn, B. A year-long study of the spatial occurrence and relative distribution of pharmaceutical residues in sewage effluent, receiving marine waters and marine bivalves. Sci. Total Environ. 2014, 476–477, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Kuehn, B.M. Antibiotic-Resistant “Superbugs” May Be Transmitted From Animals to Humans. Med. News Perspect Futur. 2007, 298, 2125–2126. [Google Scholar] [CrossRef] [PubMed]
- Watkinson, A.J.; Murby, E.J.; Costanzo, S.D. Removal of antibiotics in conventional and advanced wastewater treatment: Implications for environmental discharge and wastewater recycling. Water Res. 2007, 41, 4164–4176. [Google Scholar] [CrossRef] [PubMed]
- EMA. Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2017 Trends from 2010 to 2017. Ema/294674/2019. 2019. Available online: https://www.ema.europa.eu/en/documents/report/sales-veterinary-antimicrobial-agents-31-european-countries-2017_en.pdf (accessed on 18 February 2020).
- García-Galán, M.J.; Garrido, T.; Fraile, J.; Ginebreda, A.; Díaz-Cruz, M.S.; Barceló, D. Simultaneous occurrence of nitrates and sulfonamide antibiotics in two ground water bodies of Catalonia (Spain). J. Hydrol. 2010, 383, 93–101. [Google Scholar] [CrossRef]
- Jurado, A.; Walther, M.; Díaz-Cruz, M.S. Occurrence, fate and environmental risk assessment of the organic microcontaminants included in the Watch Lists set by EU Decisions 2015/495 and 2018/840 in the groundwater of Spain. Sci. Total Environ. 2019, 663, 285–296. [Google Scholar] [CrossRef]
- Boy-Roura, M.; Mas-Pla, J.; Petrovic, M.; Gros, M.; Soler, D.; Brusi, D.; Menció, A. Towards the understanding of antibiotic occurrence and transport in groundwater: Findings from the Baix Fluvià alluvial aquifer (NE Catalonia, Spain). Sci. Total Environ. 2018, 612, 1387–1406. [Google Scholar] [CrossRef]
- García-Gil, A.; Garrido Schneider, E.; Mejías, M.; Barceló, D.; Vázquez-Suñé, E.; Díaz-Cruz, S. Occurrence of pharmaceuticals and personal care products in the urban aquifer of Zaragoza (Spain) and its relationship with intensive shallow geothermal energy exploitation. J. Hydrol. 2018, 566, 629–642. [Google Scholar] [CrossRef] [Green Version]
- Senta, I.; Terzic, S.; Ahel, M. Occurrence and fate of dissolved and particulate antimicrobials in municipal wastewater treatment. Water Res. 2013, 47, 705–714. [Google Scholar] [CrossRef] [Green Version]
- Babić, S.; Ašperger, D.; Mutavdžić, D.; Horvat, A.J.M.; Kaštelan-Macan, M. Solid phase extraction and HPLC determination of veterinary pharmaceuticals in wastewater. Talanta 2006, 70, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Golovko, O.; Kumar, V.; Fedorova, G.; Randak, T.; Grabic, R. Seasonal changes in antibiotics, antidepressants/psychiatric drugs, antihistamines and lipid regulators in a wastewater treatment plant. Chemosphere 2014, 111, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Al Aukidy, M.; Verlicchi, P.; Jelic, A.; Petrovic, M.; Barcelò, D. Monitoring release of pharmaceutical compounds: Occurrence and environmental risk assessment of two WWTP effluents and their receiving bodies in the Po Valley, Italy. Sci. Total Environ. 2012, 438, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Tuc Dinh, Q.; Alliot, F.; Moreau-Guigon, E.; Eurin, J.; Chevreuil, M.; Labadie, P. Measurement of trace levels of antibiotics in river water using on-line enrichment and triple-quadrupole LC-MS/MS. Talanta 2011, 85, 1238–1245. [Google Scholar] [CrossRef]
- Rossmann, J.; Schubert, S.; Gurke, R.; Oertel, R.; Kirch, W. Simultaneous determination of most prescribed antibiotics in multiple urban wastewater by SPE-LC-MS/MS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 969, 162–170. [Google Scholar] [CrossRef]
- Tamtam, F.; Mercier, F.; Le Bot, B.; Eurin, J.; Dinh, Q.T.; Clément, M.; Chevreuil, M. Occurrence and fate of antibiotics in the Seine River in various hydrological conditions. Sci. Total Environ. 2008, 393, 84–95. [Google Scholar] [CrossRef]
- Wagil, M.; Kumirska, J.; Stolte, S.; Puckowski, A.; Maszkowska, J.; Stepnowski, P.; Białk-Bielińska, A. Development of sensitive and reliable LC-MS/MS methods for the determination of three fluoroquinolones in water and fish tissue samples and preliminary environmental risk assessment of their presence in two rivers in northern Poland. Sci. Total Environ. 2014, 493, 1006–1013. [Google Scholar] [CrossRef]
- Dan, A.; Zhang, X.; Dai, Y.; Chen, C.; Yang, Y. Occurrence and removal of quinolone, tetracycline, and macrolide antibiotics from urban wastewater in constructed wetlands. J. Clean Prod. 2020, 252, 119677. [Google Scholar] [CrossRef]
- Samir, A.; Abdel-Moein, K.A.; Zaher, H.M. Emergence of penicillin-macrolide-resistant Streptococcus pyogenes among pet animals: An ongoing public health threat. Comp. Immunol. Microbiol. Infect. Dis. 2020, 68, 101390. [Google Scholar] [CrossRef]
- Milaković, M.; Vestergaard, G.; González-Plaza, J.J.; Petrić, I.; Šimatović, A.; Senta, I.; Kublik, S.; Schloter, M.; Smalla, K.; Udiković-Kolić, N. Pollution from azithromycin-manufacturing promotes macrolide-resistance gene propagation and induces spatial and seasonal bacterial community shifts in receiving river sediments. Environ. Int. 2019, 123, 501–511. [Google Scholar] [CrossRef]
- Yao, N.; Li, C.; Yu, J.; Xu, Q.; Wei, S.; Tian, Z.; Yang, Z.; Yang, W.; Shen, J. Insight into adsorption of combined antibiotic-heavy metal contaminants on graphene oxide in water. Sep. Purif. Technol. 2019, 236, 116278. [Google Scholar] [CrossRef]
- Pan, S.F.; Zhu, M.P.; Chen, J.P.; Yuan, Z.H.; Zhong, L.B.; Zheng, Y.M. Separation of tetracycline from wastewater using forward osmosis process with thin film composite membrane—Implications for antibiotics recovery. Sep. Purif. Technol. 2015, 153, 76–83. [Google Scholar] [CrossRef]
- Moles, S.; Valero, P.; Escuadra, S.; Mosteo, R.; Gómez, J.; Ormad, M.P. Performance comparison of commercial TiO2: Separation and reuse for bacterial photo-inactivation and emerging pollutants photo-degradation. Environ. Sci. Pollut. Res. 2020, 1–15. [Google Scholar] [CrossRef] [PubMed]
- van Grieken, R.; Marugán, J.; Pablos, C.; Furones, L.; López, A. Comparison between the photocatalytic inactivation of Gram-positive E. faecalis and Gram-negative E. coli faecal contamination indicator microorganisms. Appl. Catal. B Environ. 2010, 100, 212–220. [Google Scholar] [CrossRef]
- Gumy, D.; Rincon, A.G.; Hajdu, R.; Pulgarin, C. Solar photocatalysis for detoxification and disinfection of water: Different types of suspended and fixed TiO2 catalysts study. Sol. Energy 2006, 80, 1376–1381. [Google Scholar] [CrossRef]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Bernabeu, A.; Vercher, R.F.; Santos-Juanes, L.; Simón, P.J.; Lardín, C.; Martínez, M.A.; Vicente, J.A.; González, R.; Llosá, C.; Arques, A.; et al. Solar photocatalysis as a tertiary treatment to remove emerging pollutants from wastewater treatment plant effluents. Catal. Today 2011, 161, 235–240. [Google Scholar] [CrossRef]
- Malesic-Eleftheriadou, N.; Evgenidou, E.; Kyzas, G.Z.; Bikiaris, D.N.; Lambropoulou, D.A. Removal of antibiotics in aqueous media by using new synthesized bio-based poly(ethylene terephthalate)-TiO2 photocatalysts. Chemosphere 2019, 234, 746–755. [Google Scholar] [CrossRef]
- Biancullo, F.; Moreira, N.F.; Ribeiro, A.R.; Manaia, C.M.; Faria, J.L.; Nunes, O.C.; Castro-Silva, S.M.; Silva, A.M. Heterogeneous photocatalysis using UVA-LEDs for the removal of antibiotics and antibiotic resistant bacteria from urban wastewater treatment plant effluents. Chem. Eng. J. 2019, 36, 304–313. [Google Scholar] [CrossRef]
- Cai, Q.; Hu, J. Decomposition of sulfamethoxazole and trimethoprim by continuous UVA/LED/TiO2 photocatalysis: Decomposition pathways, residual antibacterial activity and toxicity. J. Hazard. Mater. 2017, 323, 527–536. [Google Scholar] [CrossRef]
- Englert, B. Method 1694: Pharmaceuticals and Personal Care Products in Water, Soil, Sediment, and Biosolids by HPLC/MS/MS; US Environmental Protection Agency (EPA): Washington, DC, USA, 2007. [Google Scholar]
- Mirzaei, R.; Yunesian, M.; Nasseri, S.; Gholami, M.; Jalilzadeh, E.; Shoeibi, S.; Bidshahi, H.S.; Mesdaghinia, A. An optimized SPE-LC-MS/MS method for antibiotics residue analysis in ground, surface and treated water samples by response surface methodology- central composite design. J. Environ. Health Sci. Eng. 2017, 15, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Bao, M.; Barreiro, R.; Miranda, J.M.; Cepeda, A.; Regal, P. Fast HPLC-MS/MS method for determining penicillin antibiotics in infant formulas using molecularly imprinted solid-phase extraction. J. Anal. Methods Chem. 2015, 2015, 959675. [Google Scholar] [CrossRef] [PubMed]
- Gros, M.; Petrovié, M.; Barceló, D. Multi-residue analytical methods using LC-tandem MS for the determination of pharmaceuticals in environmental and wastewater samples: A review. Anal. Bioanal. Chem. 2006, 386, 941–952. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Antimicrobial Consumption. ECDC. Annual Epidemiological Report for 2017; ECDC: Stockholm, Sweden, 2018.
- Lei, K.; Zhu, Y.; Chen, W.; Pan, H.Y.; Cao, Y.X.; Zhang, X.; Guo, B.B.; Sweetman, A.; Lin, C.Y.; Ouyang, W.; et al. Spatial and seasonal variations of antibiotics in river waters in the Haihe River Catchment in China and ecotoxicological risk assessment. Environ. Int. 2019, 130, 104919. [Google Scholar] [CrossRef]
- Lamm, A.; Gozlan, I.; Rotstein, A.; Avisar, D. Detection of amoxicillin-diketopiperazine-2’, 5’ in wastewater samples. J. Environ. Sci. Health Part A Toxic/Hazardous Subst Environ. Eng. 2009, 44, 1512–1517. [Google Scholar] [CrossRef]
- Gozlan, I.; Rotstein, A.; Avisar, D. Amoxicillin-degradation products formed under controlled environmental conditions: Identification and determination in the aquatic environment. Chemosphere 2013, 91, 985–992. [Google Scholar] [CrossRef]
- Li, L.; Guo, C.; Ai, L.; Dou, C.; Wang, G.; Sun, H. Research on degradation of penicillins in milk by β-lactamase using ultra-performance liquid chromatography coupled with time-of-flight mass spectrometry. J. Dairy Sci. 2014, 97, 4052–4061. [Google Scholar] [CrossRef] [Green Version]
- Xia, S.; Jia, R.; Feng, F.; Xie, K.; Li, H.; Jing, D.; Xu, X. Effect of solids retention time on antibiotics removal performance and microbial communities in an A/O-MBR process. Bioresour. Technol. 2012, 106, 36–43. [Google Scholar] [CrossRef]
- Abegglen, C.; Joss, A.; McArdell, C.S.; Fink, G.; Schlüsener, M.P.; Ternes, T.A.; Siegrist, H. The fate of selected micropollutants in a single-house MBR. Water Res. 2009, 43, 2036–2046. [Google Scholar] [CrossRef]
- Tran, N.H.; Chen, H.; Reinhard, M.; Mao, F.; Gin, K.Y.H. Occurrence and removal of multiple classes of antibiotics and antimicrobial agents in biological wastewater treatment processes. Water Res. 2016, 104, 461–472. [Google Scholar] [CrossRef]
- Thompson, A.; Griffin, P.; Stuetz, R.; Cartmell, E. The Fate and Removal of Triclosan during Wastewater Treatment. Water Environ Res. 2005, 77, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Drewes, J.E.; Heberer, T.; Reddersen, K. Fate of pharmaceuticals during indirect potable reuse. Water Sci. Technol. 2002, 46, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res. 2009, 43, 363–380. [Google Scholar] [CrossRef] [PubMed]
- Awfa, D.; Ateia, M.; Fujii, M.; Johnson, M.S.; Yoshimura, C. Photodegradation of pharmaceuticals and personal care products in water treatment using carbonaceous-TiO2 composites: A critical review of recent literature. Water Res. 2018, 142, 26–45. [Google Scholar] [CrossRef] [PubMed]
- Conde-Cid, M.; Álvarez-Esmorís, C.; Paradelo-Núñez, R.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Álvarez-Rodríguez, E.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A. Occurrence of tetracyclines and sulfonamides in manures, agricultural soils and crops from different areas in Galicia (NW Spain). J. Clean Prod. 2018, 197, 491–500. [Google Scholar] [CrossRef]


| #WWTP | Population Equivalent | Total Inlet Flow (m3/day) | Water Treatment Line |
|---|---|---|---|
| 1 | 695,232 | 129,600 | Grit and grease separator/Activated Sludge/Decanter |
| 2 | 82,500 | 22,150 | Grit and grease separator/Decanter/Trickling filter (first stage) /Decanter/Trickling filter (second stage)/Decanter |
| 3 | 10,470 | 10,995 | Decanter/Trickling filter (first stage) /Decanter |
| 4 | 51,336 | 7500 | Grit and grease separator/Decanter/ Moving bed biofilm reactor/Decanter |
| Antibiotic | LOD (ng/L) | LOQ (ng/L) |
|---|---|---|
| Sulfadiazine | 0.8 | 2.5 |
| Trimethoprim | 0.8 | 2.5 |
| Amoxicillin | 10 | 30 |
| Enrofloxacin | 1.2 | 3.7 |
| Azithromycin | 2.0 | 6.5 |
| Antibiotic | Group | Chemical Abstracts Service Registry Number (CAS Nr.) | MW (g/mol) |
|---|---|---|---|
| Sulfadiazine | Sulfonamide | 68-35-9 | 250 |
| Trimethoprim | Trimethoprim | 738-70-5 | 290 |
| Amoxicillin | β-lactam | 26787-78-0 | 365 |
| Enrofloxacin | Fluoroquinolone | 93106-60-6 | 359 |
| Azithromycin | Macrolide | 83905-01-5 | 749 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moles, S.; Mosteo, R.; Gómez, J.; Szpunar, J.; Gozzo, S.; Castillo, J.R.; Ormad, M.P. Towards the Removal of Antibiotics Detected in Wastewaters in the POCTEFA Territory: Occurrence and TiO2 Photocatalytic Pilot-Scale Plant Performance. Water 2020, 12, 1453. https://doi.org/10.3390/w12051453
Moles S, Mosteo R, Gómez J, Szpunar J, Gozzo S, Castillo JR, Ormad MP. Towards the Removal of Antibiotics Detected in Wastewaters in the POCTEFA Territory: Occurrence and TiO2 Photocatalytic Pilot-Scale Plant Performance. Water. 2020; 12(5):1453. https://doi.org/10.3390/w12051453
Chicago/Turabian StyleMoles, Samuel, Rosa Mosteo, Jairo Gómez, Joanna Szpunar, Sebastiano Gozzo, Juan R. Castillo, and María P. Ormad. 2020. "Towards the Removal of Antibiotics Detected in Wastewaters in the POCTEFA Territory: Occurrence and TiO2 Photocatalytic Pilot-Scale Plant Performance" Water 12, no. 5: 1453. https://doi.org/10.3390/w12051453
APA StyleMoles, S., Mosteo, R., Gómez, J., Szpunar, J., Gozzo, S., Castillo, J. R., & Ormad, M. P. (2020). Towards the Removal of Antibiotics Detected in Wastewaters in the POCTEFA Territory: Occurrence and TiO2 Photocatalytic Pilot-Scale Plant Performance. Water, 12(5), 1453. https://doi.org/10.3390/w12051453





