Effects of Diversity, Coverage and Biomass of Submerged Macrophytes on Nutrient Concentrations, Water Clarity and Phytoplankton Biomass in Two Restored Shallow Lakes
Abstract
:1. Introduction
2. Methods
2.1. Study Area
2.2. Restoration Measures
2.3. Sampling and Laboratory Analyses
2.4. Data Analyses
3. Results
3.1. Changes in the Transplanted Macrophyte Communities
3.2. Water Quality after Macrophyte Transplantation
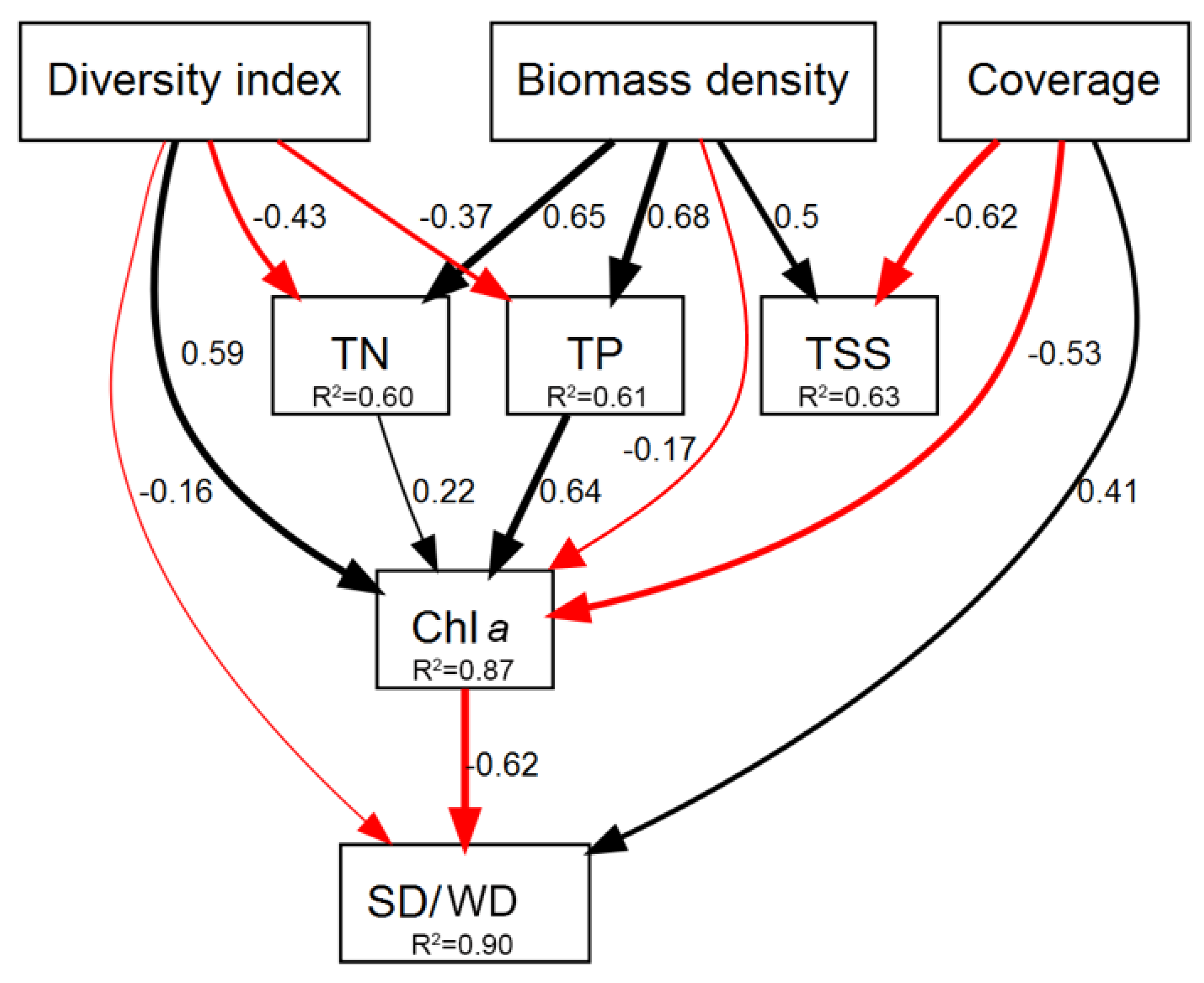
3.3. Path Analysis between Macrophytes Community and Water Quality Parameters
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gulati, R.D.; Lammens, E.; Meijer, M.L.; Vandonk, E. Biomanipulation, tool for water management. Hydrobiologia 1990, 200–201, 1–628. [Google Scholar]
- Hansson, L.A.; Annadotter, H.; Bergman, E.; Hamrin, S.F.; Jeppesen, E.; Kairesalo, T.; Luokkanen, E.; Nilsson, P.A.; Søndergaard, M.; Strand, J. Biomanipulation as an application of food-chain theory: Constraints, synthesis, and recommendations for temperate lakes. Ecosystems 1998, 1, 558–574. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Søndergaard, M.; Lauridsen, T.L. Response of fish and plankton to nutrient loading reduction in eight shallow Danish lakes with special emphasis on seasonal dynamics. Freshw. Biol. 2005, 50, 1616–1627. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Lauridsen, T.L.; Davidson, T.A.; Liu, Z.; Mazzeo, N.; Trochine, C.; Ozkan, K.; Jensen, H.S.; Trolle, D.; et al. Biomanipulation as a restoration tool to combat eutrophication: Recent advances and future challenges. Adv. Ecol. Res. 2012, 47, 411–488. [Google Scholar]
- Chen, F.; Shu, T.; Jeppesen, E.; Liu, Z.; Chen, Y. Restoration of a subtropical eutrophic shallow lake in China: Effects on nutrient concentrations and biological communities. Hydrobiologia 2013, 718, 59–71. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Jensen, J.P.; Havens, K.E.; Anneville, O.; Carvalho, L.; Coveney, M.F.; Deneke, R.; Dokulil, M.T.; Foy, B.; et al. Lake responses to reduced nutrient loading—an analysis of contemporary long-term data from 35 case studies. Freshw. Biol. 2005, 50, 1747–1771. [Google Scholar] [CrossRef]
- Hilt, S.; Gross, E.M.; Hupfer, M.; Morscheid, H.; Maehlmann, J.; Melzer, A.; Poltz, J.; Sandrock, S.; Scharf, E.M.; Schneider, S.; et al. Restoration of submerged vegetation in shallow eutrophic lakes—a guideline and state of the art in Germany. Limnologica 2006, 36, 155–171. [Google Scholar] [CrossRef] [Green Version]
- Sand-Jensen, K.; Pedersen, N.L.; Thorsgaard, I.; Moeslund, B.; Borum, J.; Brodersen, K.P. 100 years of vegetation decline and recovery in Lake Fure, Denmark. J. Ecol. 2008, 96, 260–271. [Google Scholar] [CrossRef]
- Sand-Jensen, K.; Bruun, H.H.; Baastrup-Spohr, L. Decade-long time delays in nutrient and plant species dynamics during eutrophication and re-oligotrophication of Lake Fure 1900-2015. J. Ecol. 2017, 105, 690–700. [Google Scholar] [CrossRef] [Green Version]
- Bakker, E.S.; Sarneel, J.M.; Gulati, R.D.; Liu, Z.; van Donk, E. Restoring macrophyte diversity in shallow temperate lakes: Biotic versus abiotic constraints. Hydrobiologia 2013, 710, 23–37. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Hu, J.; Zhong, P.; Zhang, X.; Ning, J.; Larsen, S.E.; Chen, D.; Gao, Y.; He, H.; Jeppesen, E. Successful restoration of a tropical shallow eutrophic lake: Strong bottom-up but weak top-down effects recorded. Water Res. 2018, 146, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Qin, B. Approaches to Mechanisms and Control of Eutrophication of Shallow Lakes in the Middle and Lower Reaches of the Yangtze River. Lake Sci. 2002, 14, 193–202. (in Chinese). [Google Scholar]
- Mehner, T.; Arlinghaus, R.; Berg, S.; Dorner, H.; Jacobsen, L.; Kasprzak, P.; Koschel, R.; Schulze, T.; Skov, C.; Wolter, C.; et al. How to link biomanipulation and sustainable fisheries management: A step-by-step guideline for lakes of the European temperate zone. Fish. Manage. Ecol. 2004, 11, 261–275. [Google Scholar] [CrossRef]
- Ke, X.S.; Li, W. Germination requirement of Vallisneria natans seeds: Implications for restoration in Chinese lakes. Hydrobiologia 2006, 559, 357–362. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Søndergaard, M.; Lauridsen, T.; Landkildehus, F. Trophic structure, species richness and biodiversity in Danish lakes: Changes along a phosphorus gradient. Freshw. Biol. 2000, 45, 201–218. [Google Scholar] [CrossRef]
- Hilt, S.; Alirangues Nunez, M.M.; Bakker, E.S.; Blindow, I.; Davidson, T.A.; Gillefalk, M.; Hansson, L.A.; Janse, J.H.; Janssen, A.B.G.; Jeppesen, E.; et al. Response of submerged macrophyte communities to external and internal restoration measures in north temperate shallow lakes. Front. Plant. Sci. 2018, 9, 194. [Google Scholar] [CrossRef] [Green Version]
- Scheffer, M.; Vandenberg, M.; Breukelaar, A.; Breukers, C.; Coops, H.; Doef, R.; Meijer, M.L. Vegetated areas with clear water in turbid shallow lakes. Aquat. Bot. 1994, 49, 193–196. [Google Scholar] [CrossRef]
- James, W.F.; Barko, J.W.; Butler, M.G. Shear stress and sediment resuspension in relation to submersed macrophyte biomass. Hydrobiologia 1994, 515, 181–191. [Google Scholar] [CrossRef]
- Hargeby, A.; Blindow, I.; Andersson, G. Long-term patterns of shifts between clear and turbid states in Lake Krankesjon and Lake Takern. Ecosystems 2007, 10, 29–36. [Google Scholar] [CrossRef]
- Sayer, C.D.; Burgess, A.; Kari, K.; Davidson, T.A.; Peglar, S.; Yang, H.; Rose, N. Long-term dynamics of submerged macrophytes and algae in a small and shallow, eutrophic lake: Implications for the stability of macrophyte-dominance. Freshw. Biol. 2010, 55, 565–583. [Google Scholar] [CrossRef]
- Sayer, C.D.; Davidson, T.A.; Jones, J.I. Seasonal dynamics of macrophytes and phytoplankton in shallow lakes: A eutrophication-driven pathway from plants to plankton? Freshw. Biol. 2010, 55, 500–513. [Google Scholar] [CrossRef]
- Rodrigo, M.A.; Rojo, C.; Segura, M.; Alonso-Guillen, J.L.; Martin, M.; Vera, P. The role of charophytes in a Mediterranean pond created for restoration purposes. Aquat. Bot. 2015, 120, 101–111. [Google Scholar] [CrossRef]
- Gopal, B.; Goel, U. Competition and allelopathy in aquatic plant communities. Bot. Rev. 1993, 59, 155–210. [Google Scholar] [CrossRef]
- Yu, J.; Liu, Z.; He, H.; Zhen, W.; Guan, B.; Chen, F.; Li, K.; Zhong, P.; Teixeira-de Mello, F.; et al. Submerged macrophytes facilitate dominance of omnivorous fish in a subtropical shallow lake: Implications for lake restoration. Hydrobiologia 2016, 775, 97–107. [Google Scholar] [CrossRef]
- Ebina, J.; Tsutsui, T.; Shirai, T. Simultaneous determination of total nitrogen and total phosphorus in water using peroxodisulfate oxidation. Water Res. 1983, 17, 1721–1726. [Google Scholar] [CrossRef]
- Parsons, T.R. Discussion of spectrophotometric determination of marine-plant pigments with revised equations for ascertaining chlorophylls and carotenoids. J. Mar. Res. 1963, 21, 155–163. [Google Scholar]
- Yu, J.; Liu, Z.; Li, K.; Chen, F.; Guan, B.; Hu, Y.; Zhong, P.; Tang, Y.; Zhao, X.; He, H.; et al. Restoration of shallow lakes in subtropical and tropical China: Response of nutrients and water clarity to biomanipulation by fish removal and submerged plant transplantation. Water 2016, 8, 438. [Google Scholar] [CrossRef] [Green Version]
- Duffy, J.E.; Lefcheck, J.S.; Stuart-Smith, R.D.; Navarrete, S.A.; Edgar, G.J. Biodiversity enhances reef fish biomass and resistance to climate change. Proc. Natl Acad. Sci. USA 2016, 113, 6230–6235. [Google Scholar] [CrossRef] [Green Version]
- R Development Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing 2016, Vienna, Austria. Available online: http://www.R-project.org (accessed on 15 May 2020).
- Fox, J. Structural equation modeling with the sem package in R. Struct. Equ. Modeling 2006, 13, 465–486. [Google Scholar] [CrossRef] [Green Version]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2009. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. Available online: https://cran.r-project.org/web/packages/vegan (accessed on 15 May 2020).
- Qiu, D.R.; Wu, Z.B.; Liu, B.Y.; Deng, J.Q.; Fu, G.P.; He, F. The restoration of aquatic macrophytes for improving water quality in a hypertrophic shallow lake in Hubei Province, China. Ecol. Eng. 2001, 18, 147–156. [Google Scholar] [CrossRef]
- Zeng, L.; He, F.; Dai, Z.; Xu, D.; Liu, B.; Zhou, Q.; Wu, Z. Effect of submerged macrophyte restoration on improving aquatic ecosystem in a subtropical, shallow lake. Ecol. Eng. 2017, 106, 578–587. [Google Scholar] [CrossRef]
- Chen, K.; Bao, C.; Zhou, W. Ecological restoration in eutrophic Lake Wuli: A large enclosure experiment. Ecol. Eng. 2009, 35, 1646–1655. [Google Scholar] [CrossRef]
- Blindow, I.; Hargeby, A.; Hilt, S. Facilitation of clear-water conditions in shallow lakes by macrophytes: Differences between charophyte and angiosperm dominance. Hydrobiologia 2014, 737, 99–110. [Google Scholar] [CrossRef]
- Engelhardt, K.A.M.; Ritchie, M.E. Effects of macrophyte species richness on wetland ecosystem functioning and services. Nature 2001, 411, 687–689. [Google Scholar] [CrossRef]
- Kalff, J. Limnology: Inland Water Ecosystems; Prentice Hall: Upper Saddle River, NJ, USA, 2002. [Google Scholar]
- Van, T.K.; Wheeler, G.S.; Center, T.D. Competition between Hydrilla verticillata and Vallisneria americana as influenced by soil fertility. Aquat. Bot. 1999, 62, 225–233. [Google Scholar] [CrossRef]
- Van den Berg, M.S.; Coops, H.; Meijer, M.L.; Scheffer, M.; Simons, J. Clear water associated with a dense Chara vegetation in the shallow and turbid Lake Veluwemeer, the Netherlands. In Structuring Role of Submerged Macrophytes in Lakes, 1st ed.; Jeppesen, E., Søndergaard, M., Søndergaard, M., Christoffersen, K., Eds.; Ecological Studies Series; Springer: New York, NY, USA, 1998; pp. 339–352. [Google Scholar]
- Kufel, L.; Kufel, I. Chara beds acting as nutrient sinks in shallow lakes—A review. Aquat. Bot. 2002, 72, 249–260. [Google Scholar] [CrossRef]
- Yu, J.; Zhen, W.; Guan, B.; Zhong, P.; Jeppesen, E.; Liu, Z. Dominance of Myriophyllum spicatumin submerged macrophyte communities associated with grass carp. Knowl. Manag. Aquat. Ecos. 2016, 417, 24. [Google Scholar] [CrossRef] [Green Version]


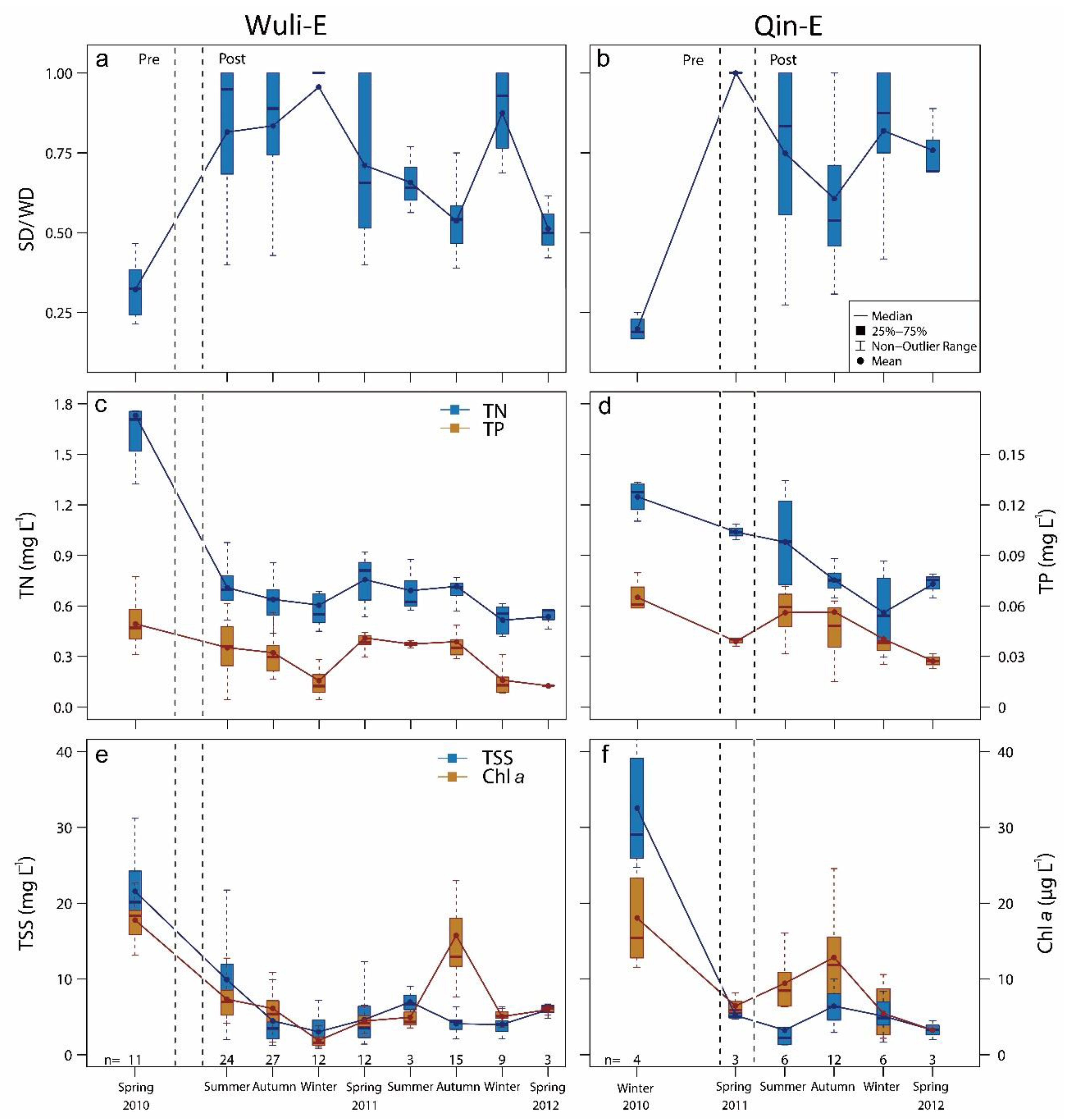
| Study Area | Main Area | Restored Area | Depth of Restored Area (m) | TN (mg L−1) | TP (mg L−1) | SD (cm) | Sediment Parameters (mg/g) | ||
|---|---|---|---|---|---|---|---|---|---|
| TN | TP | TOC | |||||||
| Lake Wuli | Wuli-C 860 ha | Wuli-E 5 ha | 2 | 1.7 ± 0.2 | 0.06 ± 0.01 | 30 | 1.64 ± 0.33 | 0.716 ± 0.30 | 14.7 ± 4.5 |
| Lake Qinhu | Qin-C 130 ha | Qin-E 8 ha | 1.5 | 1.3 ± 0.2 | 0.06 ± 0.01 | 20 | 1.55 ± 0.07 | 0.768 ± 0.22 | 13.3 ± 3.2 |
| Source of Variation | TN | TP | TSS | Chl a | SD/WD | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| F | p | F | p | F | p | F | p | F | p | |
| Between subjects | ||||||||||
| Restored area | 14.94 | 0.018 | 9.90 | 0.035 | 21.99 | 0.009 | 0.34 | 0.591 | 17.719 | 0.014 |
| Unrestored area | 6.52 | 0.063 | 2.64 | 0.179 | 0.44 | 0.542 | 6.50 | 0.063 | 2.08 | 0.190 |
| Within subjects | ||||||||||
| Month | 34.51 | 0.001 | 9.54 | 0.012 | 1.80 | 0.240 | 14.40 | 0.012 | 7.02 | 0.012 |
| Month × Restored area | 9.06 | 0.019 | 0.95 | 0.417 | 0.93 | 0.414 | 0.73 | 0.461 | 1.39 | 0.302 |
| Month × Unrestored area | 9.36 | 0.009 | 11.13 | 0.016 | 0.74 | 0.477 | 7.23 | 0.043 | 0.41 | 0.555 |
| Differences | TN | TP | TSS | Chl a | SD/WD |
|---|---|---|---|---|---|
| Restored Wuli-E—Qin-E | −0.091 * | −0.016 * | −2.705 ** | −1.316 | −0.126 * |
| Unrestored Wuli-C—Qin-C | 0.18 | −0.011 | 0.493 | 5.167 | 0.013 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, Y.; Yin, C.; Zhao, Y.; Liu, Z.; Liu, P.; Zhen, W.; Hu, Y.; Yu, J.; Wang, Z.; Guan, B. Effects of Diversity, Coverage and Biomass of Submerged Macrophytes on Nutrient Concentrations, Water Clarity and Phytoplankton Biomass in Two Restored Shallow Lakes. Water 2020, 12, 1425. https://doi.org/10.3390/w12051425
Gao Y, Yin C, Zhao Y, Liu Z, Liu P, Zhen W, Hu Y, Yu J, Wang Z, Guan B. Effects of Diversity, Coverage and Biomass of Submerged Macrophytes on Nutrient Concentrations, Water Clarity and Phytoplankton Biomass in Two Restored Shallow Lakes. Water. 2020; 12(5):1425. https://doi.org/10.3390/w12051425
Chicago/Turabian StyleGao, Yiming, Chunyu Yin, Yu Zhao, Zhengwen Liu, Pingping Liu, Wei Zhen, Yaohui Hu, Jinlei Yu, Zhaoxia Wang, and Baohua Guan. 2020. "Effects of Diversity, Coverage and Biomass of Submerged Macrophytes on Nutrient Concentrations, Water Clarity and Phytoplankton Biomass in Two Restored Shallow Lakes" Water 12, no. 5: 1425. https://doi.org/10.3390/w12051425
APA StyleGao, Y., Yin, C., Zhao, Y., Liu, Z., Liu, P., Zhen, W., Hu, Y., Yu, J., Wang, Z., & Guan, B. (2020). Effects of Diversity, Coverage and Biomass of Submerged Macrophytes on Nutrient Concentrations, Water Clarity and Phytoplankton Biomass in Two Restored Shallow Lakes. Water, 12(5), 1425. https://doi.org/10.3390/w12051425





