Chromium Removal from Tannery Wastewater by Electrocoagulation: Optimization and Sludge Characterization
Abstract
:1. Introduction
Electrocoagulation Mechanism
2. Materials and Methods
2.1. Tannery Wastewater
2.2. Wastewater Characterization
2.3. Experimental Design
2.4. Experimental Setup
3. Results and Discussion
3.1. RSM Optimization of the Experimental Parameters
3.2. Analysis of Variance and Model Fitting
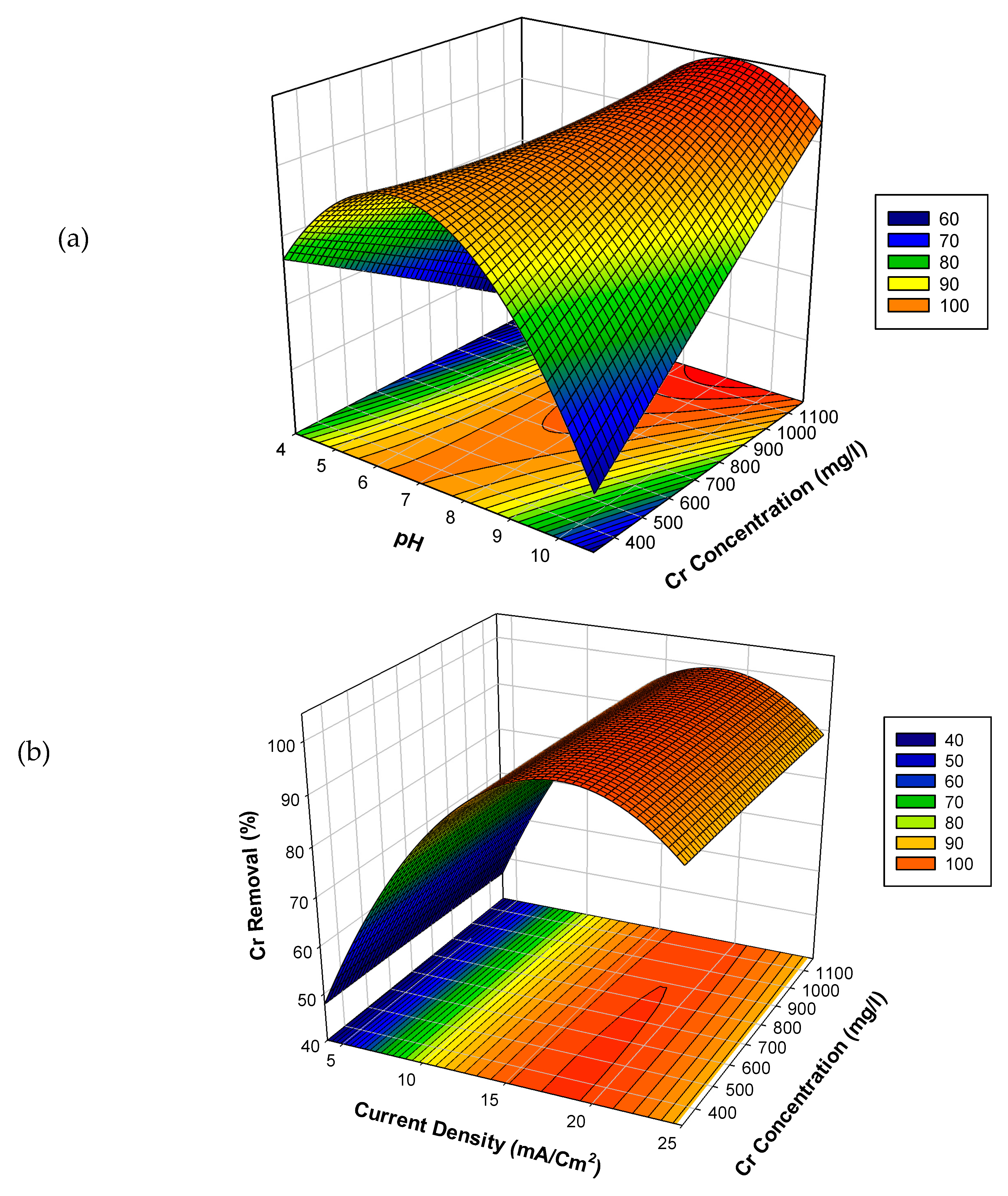
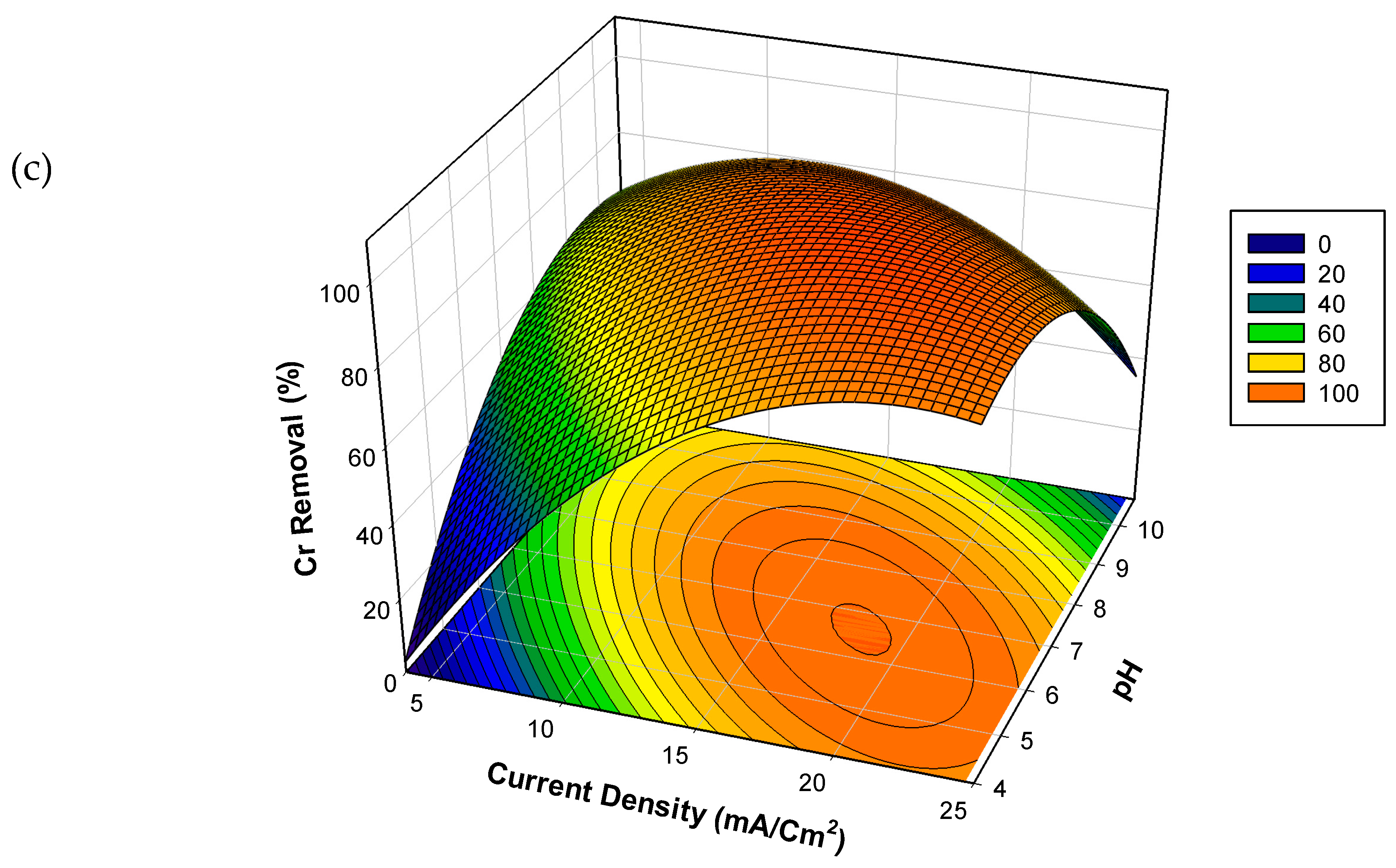
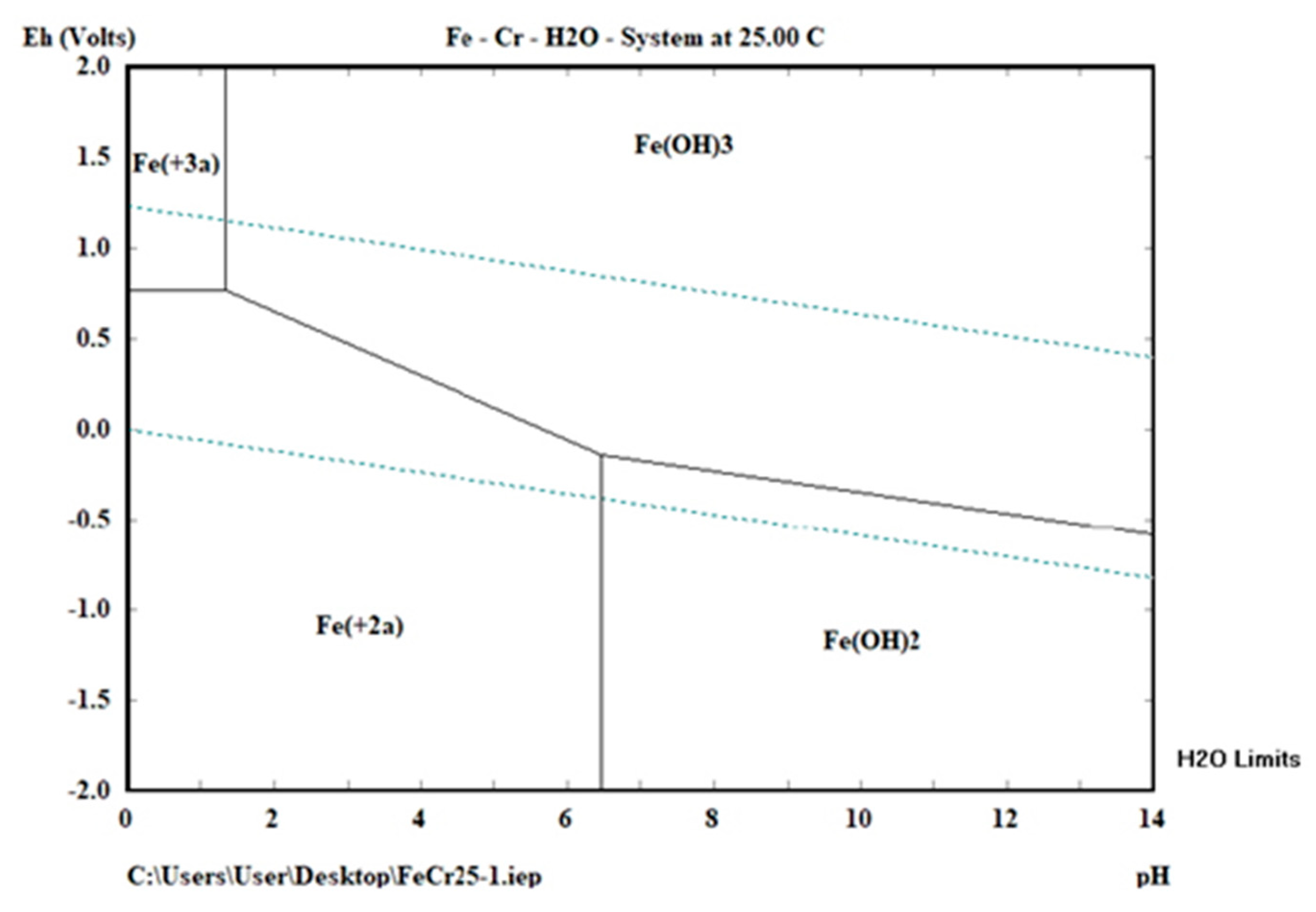
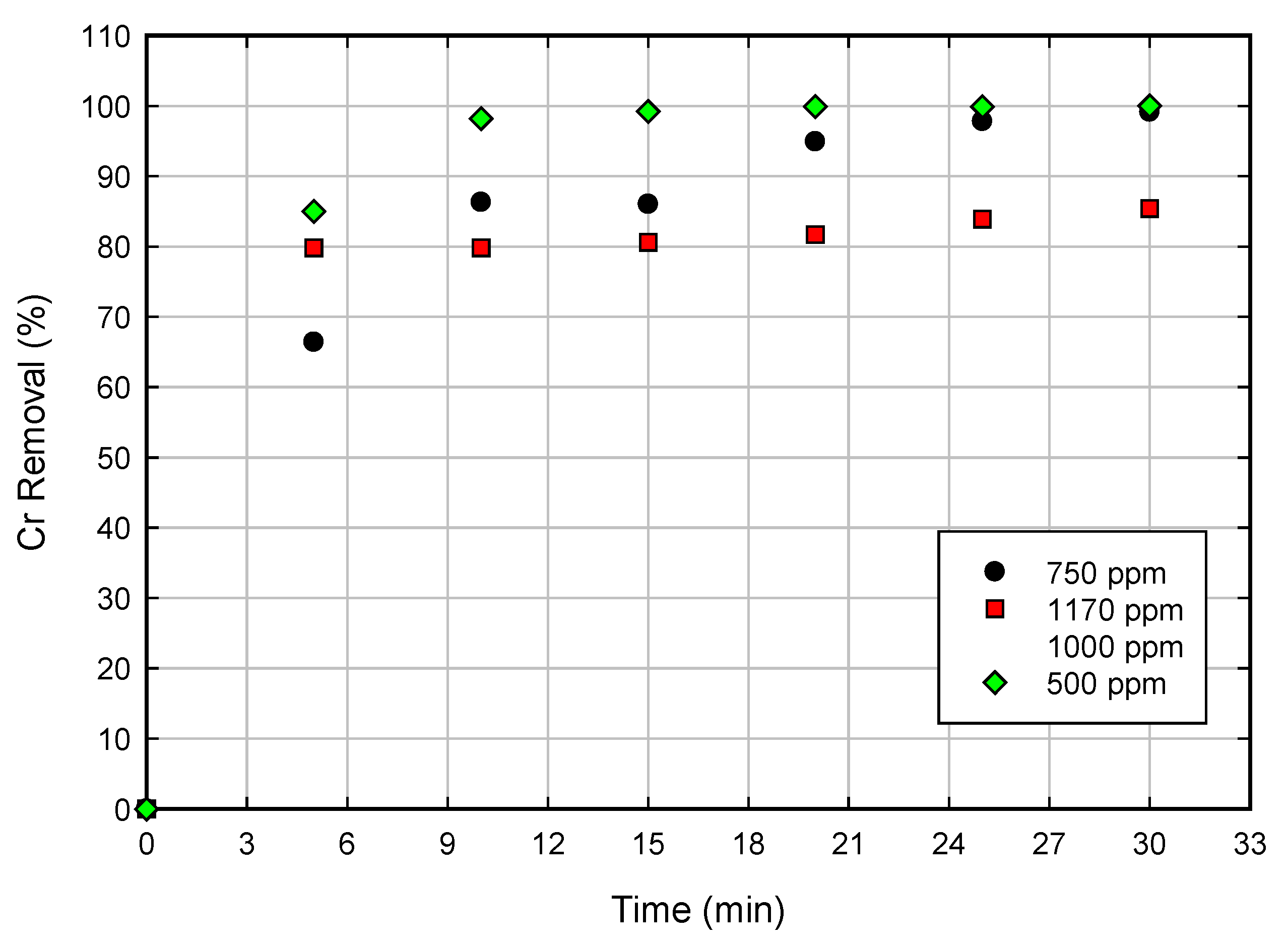
3.3. Effects of Experimental Parameters on the Percentage Cr Removal
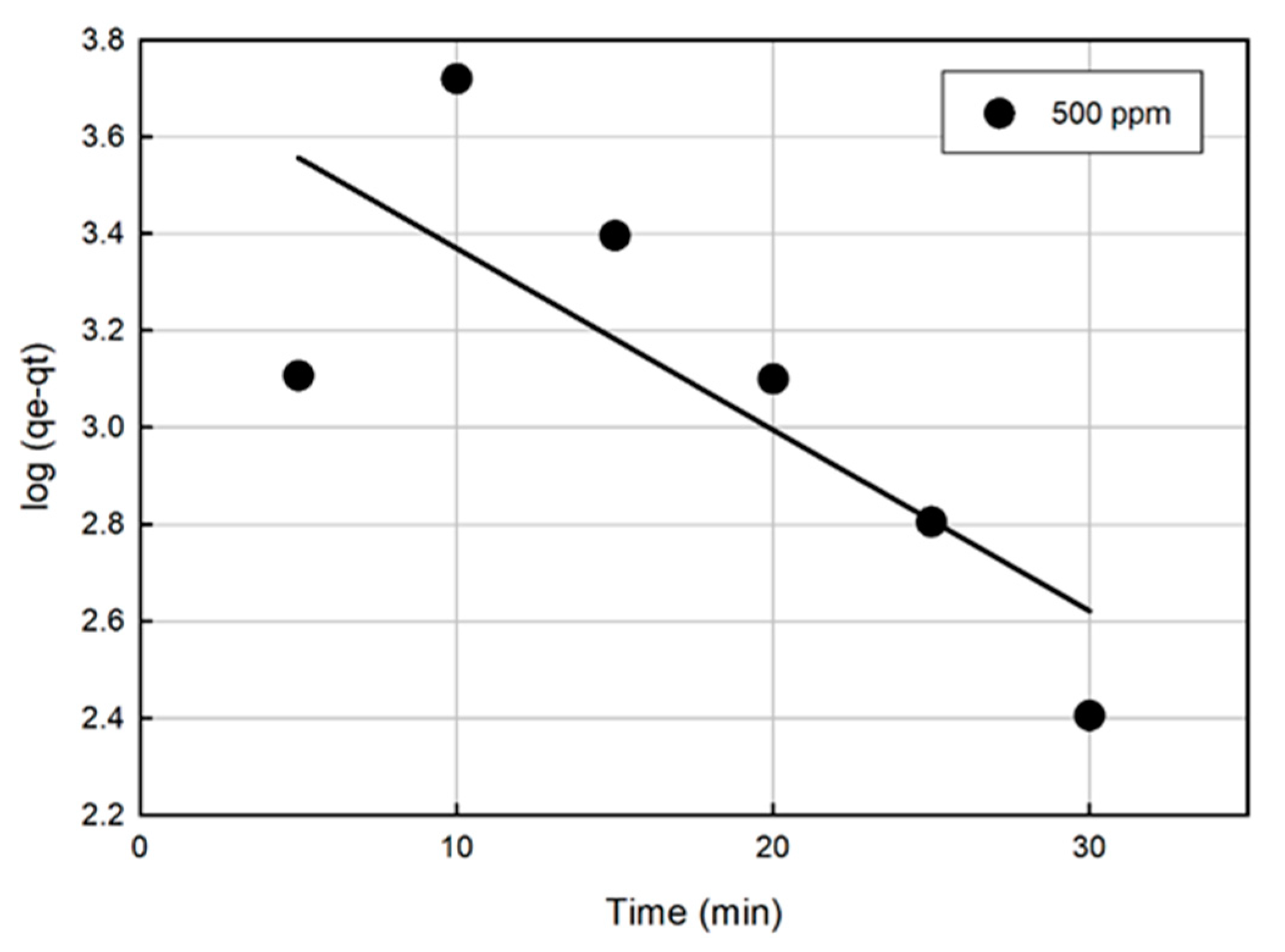
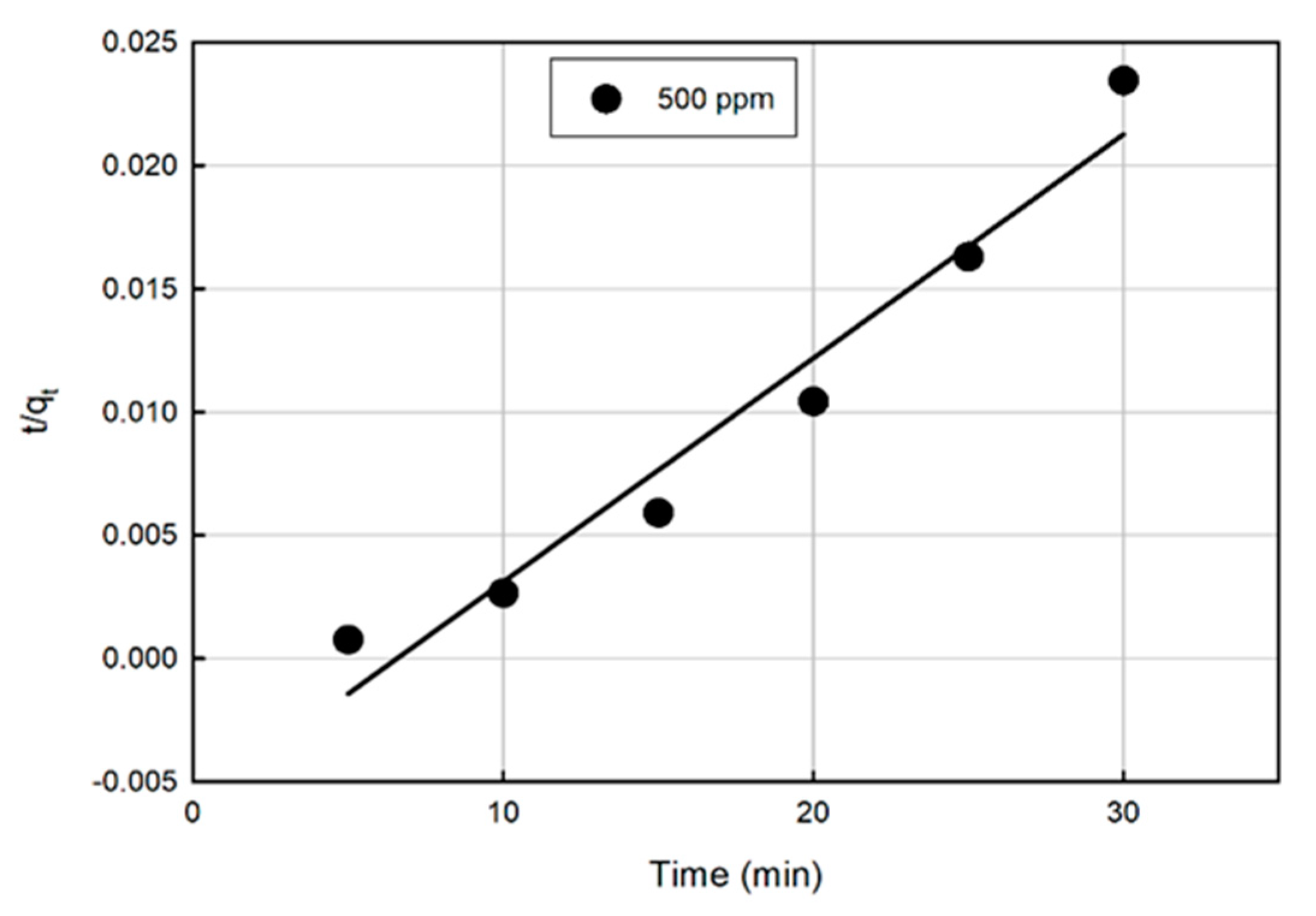
3.4. Kinetic Modeling
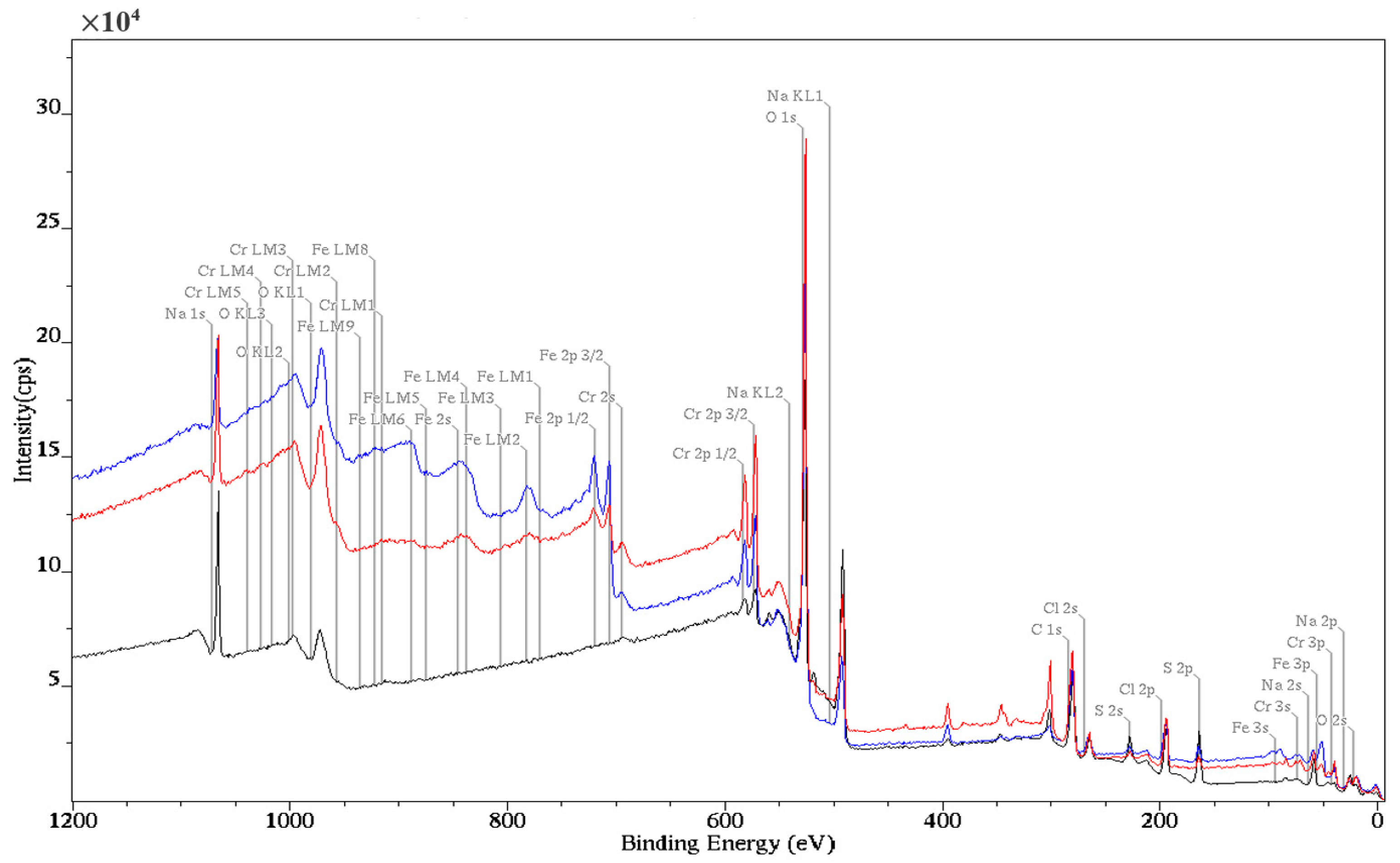
3.5. X-ray Photoelectron Spectroscopy (XPS)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Anderson, R.A. Nutritional role of chromium. Sci. Total Environ. 1981, 17, 13–29. [Google Scholar] [CrossRef]
- OSHA. Fact Sheet Health Effects of Hexavalent Chromium Hexavalent; OSHA: Washington, DC, USA, 2006.
- Saha, R.; Nandi, R.; Saha, B. Sources and toxicity of hexavalent chromium. J. Coord. Chem. 2011, 64, 1782–1806. [Google Scholar] [CrossRef]
- Megharaj, M.; Avudainayagam, S.; Naidu, R. Toxicity of hexavalent chromium and its reduction by bacteria isolated from soil contaminated with tannery waste. Curr. Microbiol. 2003, 47, 51–54. [Google Scholar] [CrossRef]
- Kleber, R.J.; Helz, G.R. Indirect Photoreduction of Aqueous Chromium(VI). Environ. Sci. Technol. 1992, 26, 307–312. [Google Scholar]
- Saha, B.; Orvig, C. Biosorbents for hexavalent chromium elimination from industrial and municipal effluents. Coord. Chem. Rev. 2010, 254, 2959–2972. [Google Scholar] [CrossRef]
- Electronic Code of Federal Regulations, Environmental Protection Agency. Protection of Environment, Part 425—Leather Tanning and Finishing Point Source Category. Available online: https://www.govinfo.gov/app/details/CFR-2012-title40-vol31/CFR-2012-title40-vol31-part425 (accessed on 2 May 2020).
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Moussa, D.T.; El-Naas, M.H.; Nasser, M.; Al-Marri, M.J. A comprehensive review of electrocoagulation for water treatment: Potentials and challenges. J. Environ. Manag. 2017, 186, 24–41. [Google Scholar] [CrossRef]
- Sayali, .D.A.; Chavan, F.I.; Hussain, M. Electrocoagulation of Waste Water by Using Iron and Aluminium. In Proceedings of the International Conference Proceeding ICGTETM, Jalgaon, India, 4–6 January 2016; pp. 15–21. [Google Scholar]
- Song, Z.; Williams, C.J.; Edyvean, R.G.J. Sedimentation of tannery wastewater. Water Res. 2000, 34, 2171–2176. [Google Scholar] [CrossRef]
- Mwinyihija, M.; Meharg, A.; Dawson, J.; Strachan, N.J.C.; Killham, K. An ecotoxicological approach to assessing the impact of tanning industry effluent on river health. Arch. Environ. Contam. Toxicol. 2006, 50, 316–324. [Google Scholar] [CrossRef]
- Körbahti, B.K.; Tanyolaç, A. Electrochemical treatment of simulated industrial paint wastewater in a continuous tubular reactor. Chem. Eng. J. 2009, 148, 444–451. [Google Scholar] [CrossRef]
- Moisés, T.-P.; Patricia, B.-H.; Barrera-Díaz, C.E.; Gabriela, R.-M.; Natividad-Rangel, R. Treatment of industrial effluents by a continuous system: Electrocoagulation–Activated sludge. Bioresour. Technol. 2010, 101, 7761–7766. [Google Scholar] [CrossRef]
- Hamdan, S.S.; El-Naas, M.H. An electrocoagulation column (ECC) for groundwater purification. J. Water Process Eng. 2014, 4, 25–30. [Google Scholar] [CrossRef]
- Hamdan, S.S.; El-Naas, M.H. Characterization of the removal of Chromium(VI) from groundwater by electrocoagulation. J. Ind. Eng. Chem. 2014, 20, 2775–2781. [Google Scholar] [CrossRef]
- Un, U.T.; Onpeker, S.E.; Ozel, E. The treatment of chromium containing wastewater using electrocoagulation and the production of ceramic pigments from the resulting sludge. J. Environ. Manag. 2017, 200, 196–203. [Google Scholar]
- Golder, A.K.; Samanta, A.N.; Ray, S. Removal of trivalent chromium by electrocoagulation. Sep. Purif. Technol. 2007, 53, 33–41. [Google Scholar] [CrossRef]
- Akbal, F.; Camcı, S. Copper, chromium and nickel removal from metal plating wastewater by electrocoagulation. Desalination 2011, 269, 214–222. [Google Scholar] [CrossRef]
- Mouedhen, G.; Feki, M.; de Petris-Wery, M.; Ayedi, H.F. Electrochemical removal of Cr(VI) from aqueous media using iron and aluminum as electrode materials: Towards a better understanding of the involved phenomena. J. Hazard. Mater. 2009, 168, 983–991. [Google Scholar] [CrossRef]
- Daneshvar, N.; Oladegaragoze, A.; Djafarzadeh, N. Decolorization of basic dye solutions by electrocoagulation: An investigation of the effect of operational parameters. J. Hazard. Mater. 2006, 129, 116–122. [Google Scholar] [CrossRef]
- Yıldız, Y.Ş.; Koparal, A.S.; Keskinler, B. Effect of initial pH and supporting electrolyte on the treatment of water containing high concentration of humic substances by electrocoagulation. Chem. Eng. J. 2008, 138, 63–72. [Google Scholar] [CrossRef]
- Lyklema, H. 1-Introduction to Colloid Science. In Particulate Colloids 4; Lyklema, J., Ed.; Academic Press: Cambridge, MA, USA, 2005; pp. 1–16. [Google Scholar]
- Rebhun, M.; Lurie, M. Control of Organic Matter by Coagulation and Floc Separation. Water Sci. Technol. 1993, 27, 1–20. [Google Scholar] [CrossRef]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, Part 1000; American Public Health Association: Washington, DC, USA, 1999. [Google Scholar]
- Kuznetsov, M.V.; Volkov, V.L.; Zhakharova, G.S.; Gubanov, V.A. XPS study of catalytic compounds H2V12−xMexO31−δ nH2O (Me: Mo, Cr). J. Electron Spectros. Relat. Phenomena 1994, 68, 579–588. [Google Scholar] [CrossRef]
- Sleigh, C.; Pijpers, A.P.; Jaspers, A.; Coussens, B.; Meier, R.J. On the determination of atomic charge via ESCA including application to organometallics. J. Electron Spectros. Relat. Phenomena 1996, 77, 41–57. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef]
- El-Naas, M.H.; Mohammad, A.F.; Suleiman, M.I.; Al-Musharfy, M.; Al-Marzouqi, A.H. Statistical Analysis and Optimization of a Process for CO2 capture. World Acad. Sci. Eng. Technol. Int. J. Chem. Mol. Eng. 2016, 10, 397–404. [Google Scholar]
- Sedlak, D.L.; Chan, P.G. Reduction of hexavalent chromium by ferrous iron. Geochim. Cosmochim. Acta 1997, 61, 2185–2192. [Google Scholar] [CrossRef]
- Schlautman, M.A.; Han, I. Effects of pH and dissolved oxygen on the reduction of hexavalent chromium by dissolved ferrous iron in poorly buffered aqueous systems. Water Res. 2001, 35, 1534–1546. [Google Scholar] [CrossRef]
- Pettine, M.; D’Ottone, L.; Campanella, L.; Millero, F.J.; Passino, R. The reduction of chromium (VI) by iron (II) in aqueous solutions. Geochim. Cosmochim. Acta 1998, 62, 1509–1519. [Google Scholar] [CrossRef]
- Buerge, I.J.; Hug, S.J. Kinetics and pH dependence of chromium(VI) reduction by iron(II). Environ. Sci. Technol. 1997, 31, 1426–1432. [Google Scholar] [CrossRef]
- Yoosefian, M.; Ahmadzadeh, S.; Aghasi, M.; Dolatabadi, M. Optimization of electrocoagulation process for efficient removal of ciprofloxacin antibiotic using iron electrode; kinetic and isotherm studies of adsorption. J. Mol. Liq. 2017, 225, 544–553. [Google Scholar] [CrossRef]
- El-Shazly, A.H.; Al-Zahrani, A.A.; Alhamed, Y.A. Kinetics and performance analysis of batch electrocoagulation unit used for the removal of a mixture of phosphate and nitrate ions from industrial effluents. Int. J. Electrochem. Sci. 2013, 8, 3176–3185. [Google Scholar]

| Parameter | Unit | Value Range |
|---|---|---|
| pH | - | 3.4–3.7 |
| Conductivity | mS cm−1 | 44–55 |
| COD | mg L−1 | 5250–9600 |
| Total Dissolved Solids | mg L−1 | 38,200–39,400 |
| Total Suspended Solids | mg L−1 | 256–289 |
| Sulphate | mg L−1 | 9987–11,527 |
| Chloride | mg L−1 | 26,513–31,103 |
| Total Organic Carbon | mg L−1 | 2060–2710 |
| Total Nitrogen | mg L−1 | 115–136 |
| Chromium | mg L−1 | 2705–3800 |
| Parameter | Unit | Range |
|---|---|---|
| pH | - | 2.29–10.7 |
| Cr concentration | ppm | 329.55–1170.45 |
| Current density | mA/cm2 | 1.2–24.7 |
| Run No. | CD mA/cm2 | pH | Concentration ppm | Cr Removal % | |
|---|---|---|---|---|---|
| Experimental | Predicted | ||||
| 1 | 20.00 | 4.00 | 1000.00 | 71.40 | 70.56 |
| 2 | 20.00 | 9.00 | 1000.00 | 99.90 | 99.99 |
| 3 | 13.00 | 6.50 | 750.00 | 98.50 | 95.20 |
| 4 | 13.00 | 6.50 | 1170.45 | 84.60 | 85.90 |
| 5 | 06.00 | 4.00 | 500.00 | 24.50 | 22.76 |
| 6 | 20.00 | 9.00 | 500.00 | 95.00 | 95.62 |
| 7 | 13.00 | 6.50 | 750.00 | 94.80 | 95.20 |
| 8 | 13.00 | 6.50 | 750.00 | 94.60 | 95.20 |
| 9 | 13.00 | 10.70 | 750.00 | 100.00 | 100.30 |
| 10 | 13.00 | 6.50 | 750.00 | 94.60 | 94.70 |
| 11 | 13.00 | 6.50 | 750.00 | 94.50 | 94.70 |
| 12 | 01.22 | 6.50 | 750.00 | 23.30 | 23.12 |
| 13 | 24.77 | 6.50 | 750.00 | 93.00 | 93.06 |
| 14 | 06.00 | 4.00 | 1000.00 | 2.80 | 3.54 |
| 15 | 13.00 | 6.50 | 750.00 | 94.30 | 94.70 |
| 16 | 06.00 | 9.00 | 500.00 | 86.00 | 86.17 |
| 17 | 20.00 | 4.00 | 500.00 | 92.50 | 93.31 |
| 18 | 13.00 | 6.50 | 329.55 | 99.90 | 100.30 |
| 19 | 13.00 | 2.29 | 750.00 | 19.50 | 19.90 |
| 20 | 06.00 | 9.00 | 1000.00 | 90.00 | 91.11 |
| Source | Degrees of Freedom (DF) | Adjusted Sum Square | Adjusted Mean Square | F-Value | P-Value | Significance |
|---|---|---|---|---|---|---|
| Model | 9 | 19,152.5 | 2128.05 | 0579.51 | 0.000 | Significant |
| Linear | 3 | 12,960.0 | 4319.99 | 1176.42 | 0.000 | Significant |
| CD | 1 | 5439.6 | 5439.58 | 1481.31 | 0.000 | Significant |
| pH | 1 | 7255.6 | 7255.64 | 1975.86 | 0.000 | Significant |
| Conc | 1 | 0264.8 | 264.76 | 72.10 | 0.000 | Significant |
| Square | 3 | 4121.6 | 1373.88 | 374.14 | 0.000 | Significant |
| CD × CD | 1 | 2353.5 | 353.45 | 640.89 | 0.000 | Significant |
| pH × pH | 1 | 2137.3 | 2137.28 | 582.02 | 0.000 | Significant |
| Conc × Conc | 1 | 6.8 | 6.81 | 1.86 | 0.203 | Not significant |
| 2-Way Interaction | 3 | 2070.9 | 690.29 | 187.98 | 0.000 | Significant |
| CD × pH | 1 | 1740.5 | 1740.50 | 473.97 | 0.000 | Significant |
| CD × Conc | 1 | 0.1 | 0.13 | 0.03 | 0.857 | Not significant |
| pH × Conc | 1 | 330.2 | 330.25 | 89.93 | 0.000 | Significant |
| Error | 10 | 36.7 | 3.67 | |||
| Lack-of-Fit | 5 | 23.7 | 0004.73 | 1.81 | 0.265 | Not significant |
| Pure Error | 5 | 13.1 | 0002.61 |
| Initial Cr Concentration | 500 ppm (CD: 20 mA/cm2 and pH: 9) | 1000 ppm (CD: 6 mA/cm2 and pH: 9) | ||
|---|---|---|---|---|
| Peak | Position BE (ev) | Atomic Conc. % | Position BE (ev) | Atomic Conc. % |
| C 1S | 281.00 | 30.02 | 281.00 | 33.13 |
| O 1S | 527.00 | 44.61 | 527.00 | 45.64 |
| Cr 2P | 573.00 | 5.63 | 573.00 | 7.02 |
| S 2S | 165.00 | 1.42 | 165.00 | 1.09 |
| Cl 2S | 196.00 | 3.51 | 196.00 | 3.97 |
| Na 1S | 1068.00 | 4.32 | 1067.00 | 5.59 |
| Fe 2S | 708.00 | 10.49 | 707.00 | 3.56 |
| 500 ppm | 1000 ppm | |||
|---|---|---|---|---|
| Peak | Position BE (ev) | Atomic Concentration % | Position BE (ev) | Atomic Concentration % |
| Cr 2P Cr-O | 586.417 | 75.98 | 586.816 | 79.28 |
| Cr 2 P Cr-OH | 589.568 | 24.02 | 589.697 | 20.72 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Genawi, N.M.; Ibrahim, M.H.; El-Naas, M.H.; Alshaik, A.E. Chromium Removal from Tannery Wastewater by Electrocoagulation: Optimization and Sludge Characterization. Water 2020, 12, 1374. https://doi.org/10.3390/w12051374
Genawi NM, Ibrahim MH, El-Naas MH, Alshaik AE. Chromium Removal from Tannery Wastewater by Electrocoagulation: Optimization and Sludge Characterization. Water. 2020; 12(5):1374. https://doi.org/10.3390/w12051374
Chicago/Turabian StyleGenawi, Nahid M., Mohamed H. Ibrahim, Muftah H. El-Naas, and Awad E. Alshaik. 2020. "Chromium Removal from Tannery Wastewater by Electrocoagulation: Optimization and Sludge Characterization" Water 12, no. 5: 1374. https://doi.org/10.3390/w12051374
APA StyleGenawi, N. M., Ibrahim, M. H., El-Naas, M. H., & Alshaik, A. E. (2020). Chromium Removal from Tannery Wastewater by Electrocoagulation: Optimization and Sludge Characterization. Water, 12(5), 1374. https://doi.org/10.3390/w12051374






