The Influence of Design and Operational Factors on the Removal of Personal Care Products by Constructed Wetlands
Abstract
:1. Introduction
2. Methods
3. Results and Discussion
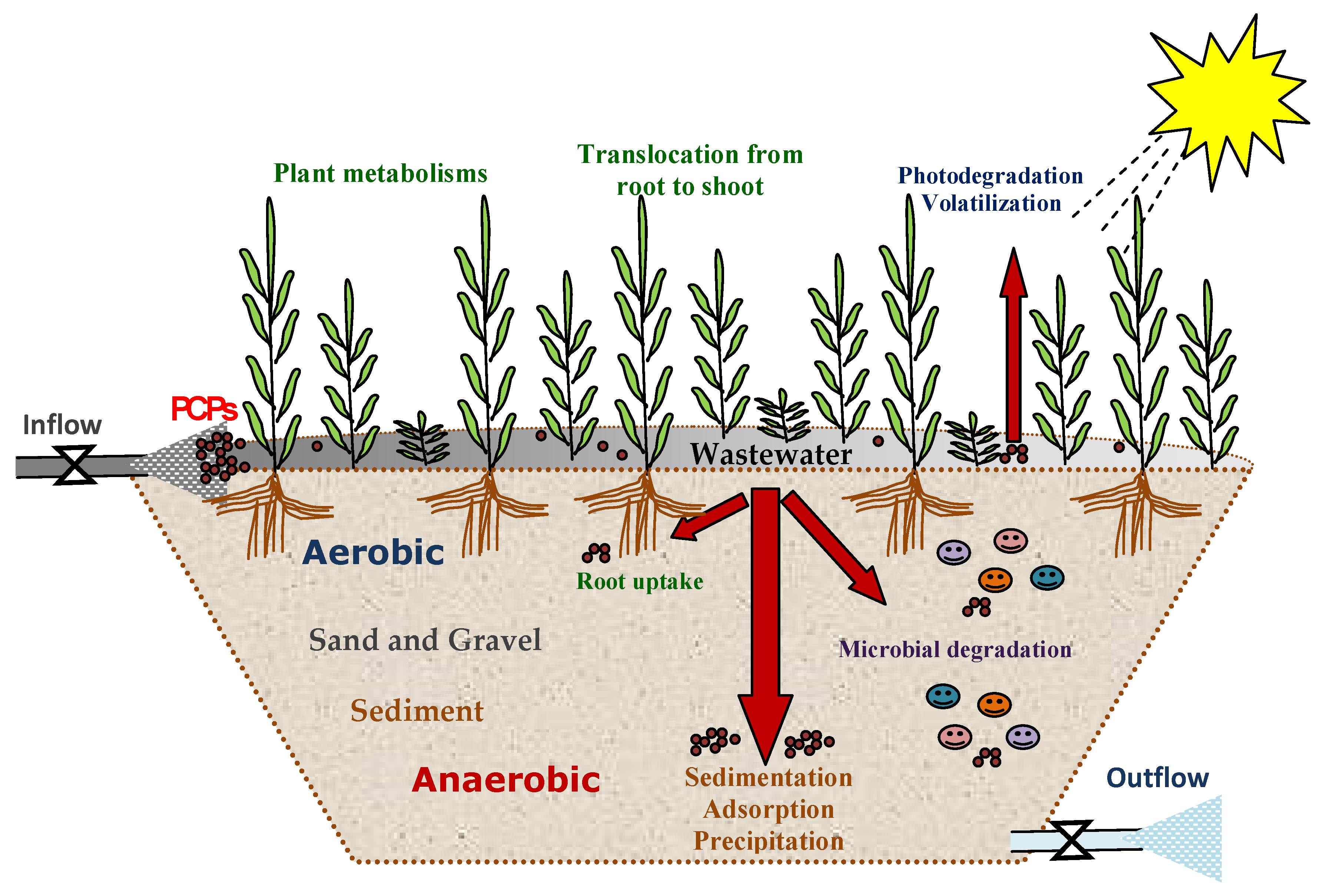
3.1. Removal Mechanisms of PCPs in CWs
3.2. Influence of Design and Operational Factors of CWs on the Removal of PCPs
3.2.1. Depth
3.2.2. Area
3.2.3. HLR
3.2.4. OLR
3.2.5. HRT
3.3. Influence of Physicochemical Parameters of CWs on the Removal of PCPs
3.3.1. pH
3.3.2. Temperature
3.3.3. Effluent DO
3.4. Effect of Plants and Support Matrix of CWs on the Removal of PCPs
3.4.1. Effect of Plants
3.4.2. Effect of Support Matrix
3.5. Effect of Seasonality on the Removal of PCPs
4. Conclusions
- The design and operational parameters are important governing factors in CWs performance for the removal of PCPs. HLR and HRT showed a significant correlation with the removal efficiency of three out of six studied PCPs, whereas, depth and area were significantly correlated with the removal efficiency of two of the studied PCPs, and OLR was significantly correlated with the removal efficiency of one of the studied PCPs. Nevertheless, the correlation was not significant with the removal efficiency of the same PCPs, which demonstrates that the removal efficiency of PCPs is not affected by only one design and operational parameter but directly or indirectly influenced by all parameters. For instance, the removal efficiency of PCPs showed a significant correlation with three factors such as methylparaben (area, HLR, and OLR) and oxybenzone (depth, HLR, and HRT); two factors such as tonalide (depth and HRT); and one factor such as galaxolide (HRT), methyl dihydrojasmonate (area), and triclosan (HLR).
- The temperature and effluent DO exhibit a significant correlation with the removal efficiency of most of the studied PCPs (three in both cases), which indicates the importance of DO and temperature for the enhancement of biodegradation, and subsequent removal of PCPs, which are better removed under aerobic conditions. Temperature and effluent DO both showed a significant correlation with the removal efficiency of galaxolide and tonalide. However, the correlation was not significant with the removal efficiency of the same PCPs, which is evident in the case of methyl dihydrojasmonate (temperature) and triclosan (effluent DO). Although pH did not show a significant correlation with the removal efficiency of any of the studied PCPs, the available evidence indicates that pH is an important parameter because it controls several biotic processes (e.g., plants development, nitrification, and heterotrophic production) and abiotic processes (e.g., the attachment of ionizable PCPs to soil/sediment via ion exchange).
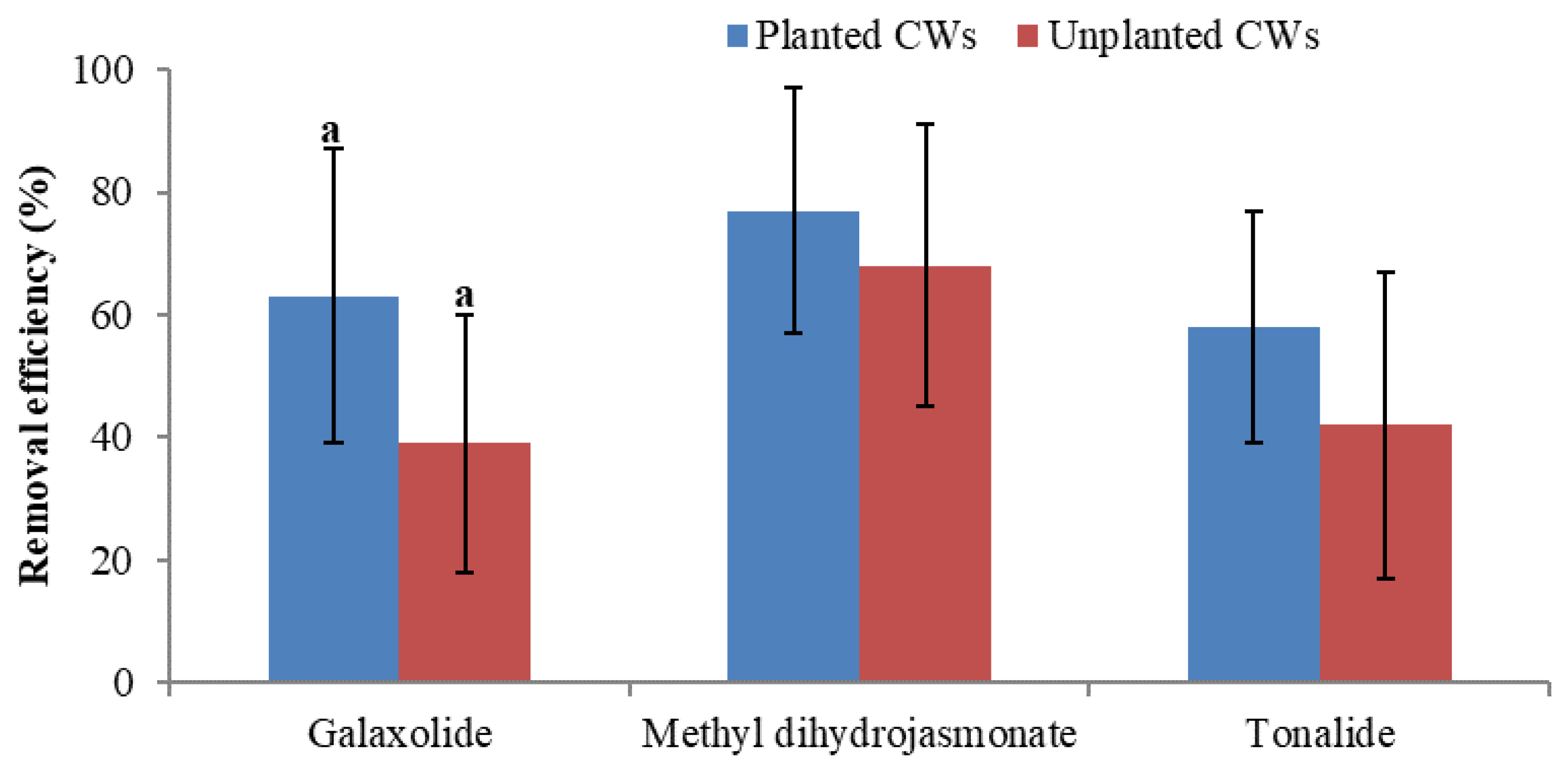
- The effect of plants in CWs is explicit by direct uptake of PCPs (methylparaben, propylparaben, and methyl dihydrojasmonate), as well as indirect positive effects such as enhancement in aerobic biodegradation, which was considered as one of the major removal mechanisms of nine out of 15 studied PCPs. The contribution of plants (direct and indirect) in CWs is also evident by the higher removal efficiency of PCPs (galaxolide, tonalide, methyl dihydrojasmonate, and triclosan) in planted CWs compared with unplanted CWs.
- The enhanced performance of CWs can be achieved by using the substrate material of high adsorption capacity, especially for those PCPs (triclosan, cashmeran, galaxolide, tonalide, and oxybenzone), which are mainly removed by adsorption onto the substrate. Furthermore, the substrate material providing a larger surface area for microbial growth and higher oxygen is also suggested to improve the removal efficiency of PCPs, which are mainly removed via aerobic biodegradation pathways (acesulfame, methylparaben, propylparaben, methyl dihydrojasmonate, and sulizobenzone).
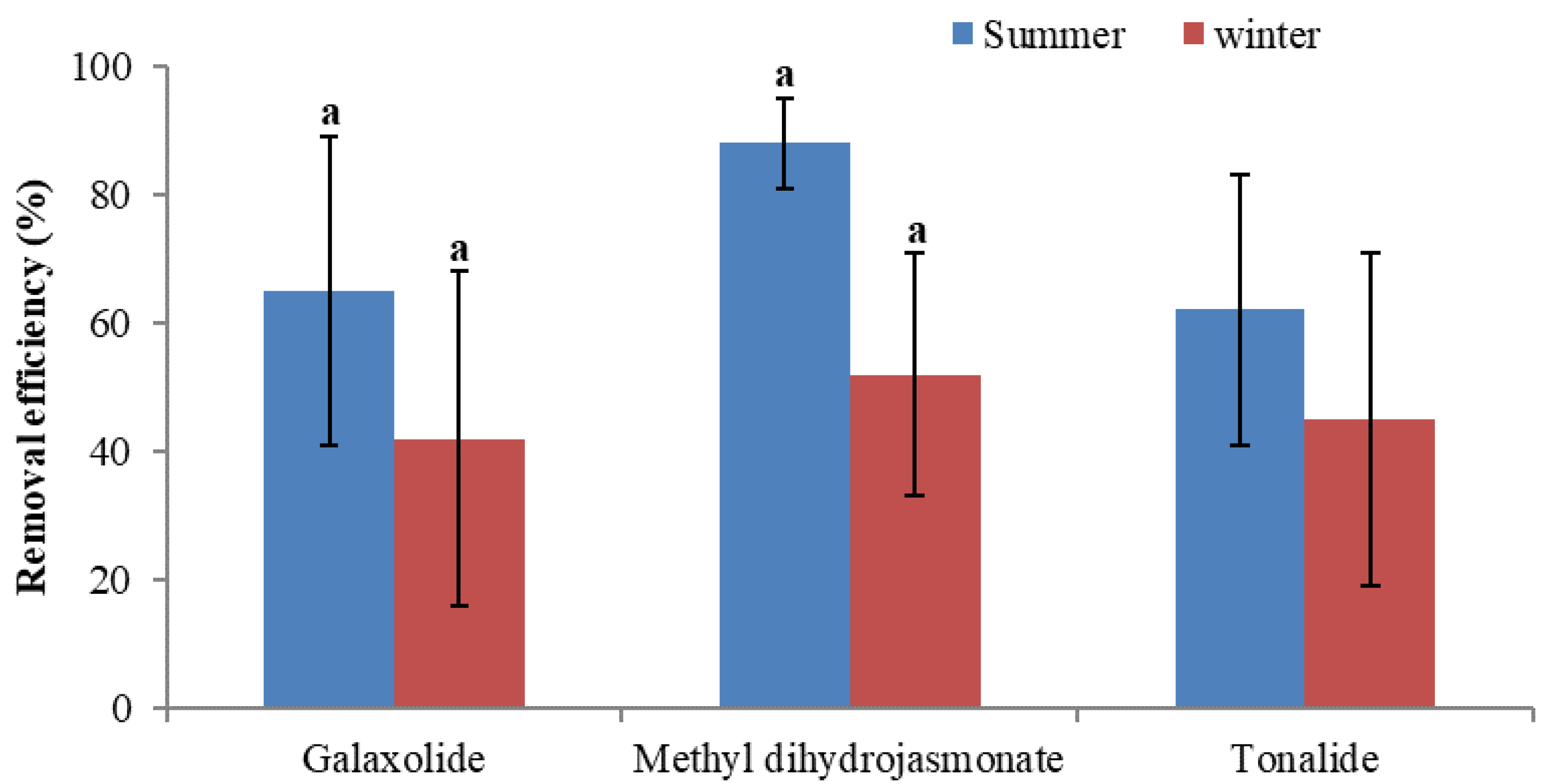
- The comparatively higher removal efficiency of most of the examined PCPs in summer than in winter might be due to the contribution of more removal mechanisms such as biodegradation, plant uptake, and photodegradation at warm temperature. On the contrary, in winter only adsorption/sorption processes are more dominant at low temperature. The removal efficiency of almost all of the studied PCPs demonstrated seasonal differences, but significant difference in the removal efficiency during summer and winter was established in the case of galaxolide and methyl dihydrojasmonate.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Matamoros, V.; Salvadó, V. Evaluation of the seasonal performance of a water reclamation pond-constructed wetland system for removing emerging contaminants. Chemosphere 2012, 86, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Hijosa-Valsero, M.; Matamoros, V.; Sidrach-Cardona, R.; Martin-Villacorta, J.; Becares, E.; Bayona, J.M. Comprehensive assessment of the design configuration of constructed wetlands for the removal of pharmaceuticals and personal care products from urban wastewaters. Water Res. 2010, 44, 3669–3678. [Google Scholar] [CrossRef] [PubMed]
- Hijosa-Valsero, M.; Matamoros, V.; Pedescoll, A.; Martín-Villacorta, J.; Bécares, E.; García, J.; Bayona, J.M. Evaluation of primary treatment and loading regimes in the removal of pharmaceuticals and personal care products from urban wastewaters by subsurface-flow constructed wetlands. Int. J. Environ. Anal. Chem. 2011, 91, 632–653. [Google Scholar] [CrossRef]
- Vymazal, J.; Březinová, T.D.; Koželuh, M.; Kule, L. Occurrence and removal of pharmaceuticals in four full-scale constructed wetlands in the Czech Republic—the first year of monitoring. Ecol. Eng. 2017, 98, 354–364. [Google Scholar] [CrossRef]
- Matamoros, V.; Arias, C.; Brix, H.; Bayona, J.M. Removal of pharmaceuticals and personal care products (PPCPs) from urban wastewater in a pilot vertical flow constructed wetland and a sand filter. Environ. Sci. Technol. 2007, 41, 8171–8177. [Google Scholar] [CrossRef] [PubMed]
- Ávila, C.; Nivala, J.; Olsson, L.; Kassa, K.; Headley, T.; Mueller, R.A.; Bayona, J.M.; García, J. Emerging organic contaminants in vertical subsurface flow constructed wetlands: Influence of media size, loading frequency and use of active aeration. Sci. Total Environ. 2014, 494–495, 211–217. [Google Scholar] [CrossRef]
- Ávila, C.; Matamoros, V.; Reyes-Contreras, C.; Piña, B.; Casado, M.; Mita, L.; Rivetti, C.; Barata, C.; García, J.; Bayona, J.M. Attenuation of emerging contaminants in a hybrid constructed wetland system under different hydraulic loading rates and their associated toxicological effects in wastewater. Sci. Total Environ. 2014, 470–471, 1272–1280. [Google Scholar] [CrossRef]
- Dai, Y.; Tao, R.; Tai, Y.; Tam, N.F.; Dan, A.; Yang, Y. Application of a full-scale newly developed stacked constructed wetland and an assembled bio-filter for reducing phenolic endocrine disrupting chemicals from secondary effluent. Ecol. Eng. 2017, 99, 496–503. [Google Scholar] [CrossRef]
- Matamoros, V.; Rodríguez, Y.; Bayona, J.M. Mitigation of emerging contaminants by full-scale horizontal flow constructed wetlands fed with secondary treated wastewater. Ecol. Eng. 2017, 99, 222–227. [Google Scholar] [CrossRef]
- Matamoros, V.; García, J.; Bayona, J.M. Organic micropollutant removal in a full-scale surface flow constructed wetland fed with secondary effluent. Water Res. 2008, 42, 653–660. [Google Scholar] [CrossRef]
- Herrera-Cardenas, J.; Navarro, A.E.; Torres, E. Effects of porous media, macrophyte type and hydraulic retention time on the removal of organic load and micropollutants in constructed wetlands. J. Environ. Sci. Health Part A 2016, 51, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Vystavna, Y.; Frkova, Z.; Marchand, L.; Vergeles, Y.; Stolberg, F. Removal efficiency of pharmaceuticals in a full scale constructed wetland in East Ukraine. Ecol. Eng. 2017, 108, 50–58. [Google Scholar] [CrossRef]
- Salcedo, J.J.P.; Montes, G.H.E.; Frómeta, A.E.N.; Osorio, A.C.; Negrete, J.M. Removal of Organic Micropollutants from Riverine Waters using Constructed Wetlands: A Mesocosms Experiment. Int. J. Appl. Eng. Res. 2018, 13, 15740–15748. [Google Scholar]
- Ávila, C.; Reyes, C.; Bayona, J.M.; García, J. Emerging organic contaminant removal depending on primary treatment and operational strategy in horizontal subsurface flow constructed wetlands: Influence of redox. Water Res. 2013, 47, 315–325. [Google Scholar] [CrossRef]
- Matamoros, V.; Rodríguez, Y.; Albaigés, J.A. comparative assessment of intensive and extensive wastewater treatment technologies for removing emerging contaminants in small communities. Water Res. 2016, 88, 777–785. [Google Scholar] [CrossRef]
- Chen, Y.; Vymazal, J.; Březinová, T.; Koželuh, M.; Kule, L.; Huang, J.; Chen, Z. Occurrence, removal and environmental risk assessment of pharmaceuticals and personal care products in rural wastewater treatment wetlands. Sci. Total Environ. 2016, 566–567, 1660–1669. [Google Scholar] [CrossRef]
- Kahl, S.; Nivala, J.; Afferden, M.V.; Müller, R.A.; Reemtsma, T. Effect of design and operational conditions on the performance of subsurface flow treatment wetlands: Emerging organic contaminants as indicators. Water Res. 2017, 125, 490–500. [Google Scholar] [CrossRef]
- Li, J.; Zhou, Q.; Campos, L.C. Removal of selected emerging PPCP compounds using greater duckweed (Spirodela polyrhiza) based lab-scale free water constructed wetland. Water Res. 2017, 126, 252–261. [Google Scholar] [CrossRef] [Green Version]
- Nivala, J.; Kahl, S.; Boog, J.; Afferden, M.; Reemtsma, T.; Müller, R.A. Dynamics of emerging organic contaminant removal in conventional and intensified subsurface flow treatment wetlands. Sci. Total Environ. 2019, 649, 1144–1156. [Google Scholar] [CrossRef]
- Hijosa-Valsero, M.; Reyes-Contreras, C.; Domínguez, C.; Bécares, E.; Bayona, J.M. Behaviour of pharmaceuticals and personal care products in constructed wetland compartments: Influent, effluent, pore water, substrate and plant roots. Chemosphere 2016, 145, 508–517. [Google Scholar] [CrossRef]
- Reyes-Contreras, C.; Hijosa-Valsero, M.; Sidrach-Cardona, R.; Bayona, J.M.; Bécares, E. Temporal evolution in PPCP removal from urban wastewater by constructed wetlands of different configuration: A medium-term study. Chemosphere 2012, 88, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Carranza-Diaz, O.; Schultze-Nobre, L.; Moeder, M.; Nivala, J.; Kuschk, P.; Koeser, H. Removal of selected organic micropollutants in planted and unplanted pilot-scale horizontal flow constructed wetlands under conditions of high organic load. Ecol. Eng. 2014, 71, 234–245. [Google Scholar] [CrossRef]
- Button, M.; Cosway, K.; Sui, J.; Weber, K. Impacts and fate of triclosan and sulfamethoxazole in intensified re-circulating vertical flow constructed wetlands. Sci. Total Environ. 2019, 649, 1017–1028. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Yang, Y.; Liu, J.; Kang, Y.; Zhang, J.; Hu, Z.; Liang, S. Enhanced triclosan and nutrient removal performance in vertical up-flow constructed wetlands with manganese oxides. Water Res. 2018, 143, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Verlicchi, P.; Zambello, E. How efficient are constructed wetlands in removing pharmaceuticals from untreated and treated urban wastewaters? A review. Sci. Total Environ. 2014, 470–471, 1281–1306. [Google Scholar] [CrossRef]
- Zhang, D.; Gersberg, R.M.; Ng, W.J.; Tan, S.K. Removal of pharmaceuticals and personal care products in aquatic plant-based systems: A review. Environ. Pollut. 2014, 184, 620–639. [Google Scholar] [CrossRef]
- Verlicchi, P.; Zambello, E.; Aukidy, M.A. Removal of Personal Care Products in Constructed Wetlands. In Personal Care Products in the Aquatic Environment; Díaz-Cruz, M.S., Barcelό, D., Eds.; The Handbook of Environmental Chemistry; Springer: Cham, Switzerland, 2015; Volume 36, pp. 319–354. [Google Scholar] [CrossRef]
- Vo, H.-N.-P.; Bui, X.-T.; Nguyen, T.-M.-H.; Koottatep, T.; Bandyopadhyay, A. Insights of the Removal Mechanisms of Pharmaceutical and Personal Care Products in Constructed Wetlands. Curr. Pollut. Rep. 2018, 4, 93–103. [Google Scholar] [CrossRef]
- Gorito, A.M.; Ribeiro, A.R.; Almeida, C.M.R.; Silva, A.M.T. A review on the application of constructed wetlands for the removal of priority substances and contaminants of emerging concern listed in recently launched EU legislation. Environ. Pollut. 2017, 227, 428–443. [Google Scholar] [CrossRef]
- Ilyas, H.; van Hullebusch, E.D. Performance comparison of different constructed wetlands designs for the removal of personal care products. Int. J. Environ. Res. Public Health 2020, 17, 3091. [Google Scholar] [CrossRef]
- Anjos, M.L.; Isique, W.D.; Albertin, L.L.; Matsumoto, T.; Henares, M.N.P. Parabens Removal from Domestic Sewage by Free-Floating Aquatic Macrophytes. Waste Biomass Valor. 2019, 10, 2221–2226. [Google Scholar] [CrossRef]
- Petrie, B.; Rood, S.; Smith, B.D.; Proctor, K.; Youdan, J.; Barden, R.; Kasprzyk-Hordern, B. Biotic phase micropollutant distribution in horizontal sub-surface flow constructed wetlands. Sci. Total Environ. 2018, 630, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, Y.-S.; Deng, W.-J.; Ying, G.-G. Removal of steroid hormones and biocides from rural wastewater by an integrated constructed wetland. Sci. Total Environ. 2019, 660, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Sgroi, M.; Pelissari, C.; Roccaro, P.; Sezerino, P.H.; García, J.; Vagliasindi, F.G.A.; Ávila, C. Removal of organic carbon, nitrogen, emerging contaminants and fluorescing organic matter in different constructed wetland configurations. Chem. Eng. J. 2018, 332, 619–627. [Google Scholar] [CrossRef] [Green Version]
- Yi, X.; Tran, N.H.; Yin, T.; He, Y.; Gin, K.Y.-H. Removal of selected PPCPs, EDCs, and antibiotic resistance genes in landfill leachate by a full-scale constructed wetlands system. Water Res. 2017, 121, 46–60. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, J.; Zhao, C.; Hay, A.G.; Xie, H.; Zhan, J. Triclosan removal in wetlands constructed with different aquatic plants. Appl. Microbiol. Biotechnol. 2016, 100, 1459–1467. [Google Scholar] [CrossRef]
- Wang, Y.; Yin, T.; Kelly, B.C.; Gin, K.Y.-H. Bioaccumulation behaviour of pharmaceuticals and personal care products in a constructed wetland. Chemosphere 2019, 222, 275–285. [Google Scholar] [CrossRef]
- Ávila, C.; Bayona, J.M.; Martín, I.; Salas, J.J.; García, J. Emerging organic contaminant removal in a full-scale hybrid constructed wetland system for wastewater treatment and reuse. Ecol. Eng. 2015, 80, 108–116. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Xie, H.; Xu, J.; Xu, X.; Zhang, J.; Hu, Z.; Liu, C.; Liang, S.; Wang, Q.; Wang, J. Bacterial community variation and microbial mechanism of triclosan (TCS) removal by constructed wetlands with different types of plants. Sci. Total Environ. 2015, 505, 633–639. [Google Scholar] [CrossRef]
- Park, N.; Vanderford, B.J.; Snyder, S.A.; Sarp, S.; Kim, S.D.; Cho, J. Effective controls of micro-pollutants included in wastewater effluent using constructed wetlands under anoxic condition. Ecol. Eng. 2009, 35, 418–423. [Google Scholar] [CrossRef]
- Francini, A.; Mariotti, L.; Gregorio, S.D.; Sebastiani, L.; Andreucci, A. Removal of micro-pollutants from urban wastewater by constructed wetlands with Phragmites australis and Salix matsudana. Environ. Sci. Pollut. Res. 2018, 25, 36474–36484. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, H. The fate and risk of selected pharmaceutical and personal care products in wastewater treatment plants and a pilot-scale multistage constructed wetland system. Environ. Sci. Pollut. Res. 2014, 21, 1466–1479. [Google Scholar] [CrossRef] [PubMed]
- Hijosa-Valsero, M.; Matamoros, V.; Martin-Villacorta, J.; Becares, E.; Bayona, J.M. Assessment of full-scale natural systems for the removal of PPCPs from wastewater in small communities. Water Res. 2010, 44, 1429–1439. [Google Scholar] [CrossRef] [PubMed]
- Rühmland, S.; Wick, A.; Ternes, T.A.; Barjenbruch, M. Fate of pharmaceuticals in a subsurface flow constructed wetland and two ponds. Ecol. Eng. 2015, 80, 125–139. [Google Scholar] [CrossRef]
- Auvinen, H.; Gebhardt, W.; Linnemann, V.; Laing, G.D.; Rousseau, D.P.L. Laboratory- and full-scale studies on the removal of pharmaceuticals in an aerated constructed wetland: Effects of aeration and hydraulic retention time on the removal efficiency and assessment of the aquatic risk. Water Sci. Technol. 2017, 76, 1457–1465. [Google Scholar] [CrossRef] [Green Version]
- Dan, A.; Yang, Y.; Dai, Y.-N.; Chen, C.-X.; Wang, S.-Y.; Tao, R. Removal and factors influencing removal of sulfonamides and trimethoprim from domestic sewage in constructed wetlands. Bioresour. Technol. 2013, 146, 363–370. [Google Scholar] [CrossRef]
- Truu, M.; Juhanson, J.; Truu, J. Microbial biomass, activity and community composition in constructed wetlands. Sci. Total Environ. 2009, 407, 3958–3971. [Google Scholar] [CrossRef]
- Dordio, A.V.; Carvalho, A.J.P. Constructed wetlands with light expanded clay aggregates for agricultural wastewater treatment. Sci. Total Environ. 2013, 463–464, 454–461. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, P.N.; Araújo, J.L.; Mucha, A.P.; Basto, M.C.P.; Almeida, C.M.R. Potential of constructed wetlands microcosms for the removal of veterinary pharmaceuticals from livestock wastewater. Bioresour. Technol. 2013, 134, 412–416. [Google Scholar] [CrossRef]
- Ávila, C.; Pedescoll, A.; Matamoros, V.; Bayona, J.M.; García, J. Capacity of a horizontal subsurface flow constructed wetland system for the removal of emerging pollutants: An injection experiment. Chemosphere 2010, 81, 1137–1142. [Google Scholar] [CrossRef]
- Pilon-Smits, E. Phytoremediation. Ann. Rev. Plant Biol. 2005, 56, 15–39. [Google Scholar] [CrossRef]
- Liu, L.; Liu, Y.-H.; Wang, Z.; Liu, C.-X.; Huang, X.; Zhu, G.-F. Behavior of tetracycline and sulfamethazine with corresponding resistance genes from swine wastewater in pilot-scale constructed wetlands. J. Hazard. Mater. 2014, 278, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Dordio, A.V.; Teimão, J.; Ramalho, I.; Palace Carvalho, A.J.; Candeias, A.J.E. Selection of a support matrix for the removal of some phenoxyacetic compounds in constructed wetlands systems. Sci. Total Environ. 2007, 380, 237–246. [Google Scholar] [CrossRef]
- Dordio, A.V.; Candeias, A.J.E.; Pinto, A.P.; da Costa, C.T.; Carvalho, A.J.P. Preliminary media screening for application in the removal of clofibric acid, carbamazepine and ibuprofen by SSF-constructed wetlands. Ecol. Eng. 2009, 35, 290–302. [Google Scholar] [CrossRef]
- Dordio, A.V.; Gonçalves, P.; Texeira, D.; Candeias, A.J.; Castanheiro, J.E.; Pinto, A.P.; Carvalho, A.J.P. Pharmaceuticals sorption behaviour in granulated cork for the selection of a support matrix for a constructed wetlands system. Int. J. Environ. Anal. Chem. 2011, 91, 615–631. [Google Scholar] [CrossRef]

| Design, Operational, and Physicochemical Parameters | References |
|---|---|
| Operational Factors | |
| Hydraulic loading rate | Matamoros et al. [5]; Ávila et al. [6,7]; Dai et al. [8] |
| Organic loading rate | Matamoros et al. [9] |
| Hydraulic retention time | Matamoros et al. [10]; Matamoros and Salvadó [1]; Ávila et al. [6]; Herrera-Cárdenas et al. [11]; Vymazal et al. [4]; Vystavna et al. [12]; Salcedo et al. [13] |
| Physicochemical Parameters | |
| pH | Hijosa-Valsero et al. [2,3] |
| Temperature | Hijosa-Valsero et al. [2,3]; Ávila et al. [14]; Matamoros et al. [9,15]; Vymazal et al. [4] |
| Dissolved oxygen | Hijosa-Valsero et al. [2]; Ávila et al. [6,7,14]; Chen et al. [16]; Kahl et al. [17]; Li et al. [18]; Vymazal et al. [4]; Nivala et al. [19] |
| Planted and Unplanted CWs | Hijosa-Valsero et al. [2,3,20]; Reyes-Contreras et al. [21]; Carranza-Diaz et al. [22]; Salcedo et al. [13]; Button et al. [23] |
| Role of Support Matrix | Ávila et al. [6]; Salcedo et al. [13]; Xie et al. [24]; Nivala et al. [19] |
| Effect of Seasonality (summer and winter) | Matamoros et al. [10,15]; Hijosa-Valsero et al. [2,3]; Reyes- Contreras et al. [21] |
| No. of Categories | Category | Personal Care Products |
|---|---|---|
| 1 | Artificial sweetener | Sucralose, Acesulfame |
| 2 | Preservatives | Methylparaben, Propylparaben |
| 3 | Insect repellent | N, N-diethyl-3-methyl benzoylamide, N, N-diethyl-3-methylbenzamide, N, N-diethyl-meta-toluamide |
| 4 | Antiseptics | Triclosan, Triclocarban |
| 5 | Fragrances | Cashmeran, Celestolide, Galaxolide, Methyl dihydrojasmonate, Tonalide |
| 6 | Flame retardants | Tributyl phosphate, Triphenyl phosphate, Tris (2-chloroethyl) phosphate |
| 7 | Sunscreen agents | Hydrocinnamic acid, Oxybenzone, Sulisobenzone |
| Design, Operational, and Physicochemical Parameters | FWSCW | HFCW | VFCW | HCW |
|---|---|---|---|---|
| Number of CWs | 24 | 57 | 12 | 38 |
| Number of studies | 8.0 | 15 | 8.0 | 19 |
| Scale of application | Lab, Pilot, Full | Lab, Pilot, Full | Lab, Pilot | Lab, Pilot, Full |
| Type of treatment | Primary, Secondary, Tertiary | Primary, Secondary, Tertiary | Primary, Secondary | Primary, Secondary, Tertiary |
| Depth (m) | 0.8 ± 0.9 | 0.5 ± 0.1 | 0.8 ± 0.1 | 0.8 ± 0.4 |
| Area (m2 PE−1) | 12 ± 8 | 6.6 ± 5.7 | 4.3 ± 3.4 | 9.0 ± 6.9 |
| HLR (m3 m−2 d−1) | 0.1 ± 0.1 | 0.5 ± 1.1 | 0.07 ± 0.03 | 0.1 ± 0.3 |
| OLR (g COD m−2 d−1) | 15 ± 18 | 32 ± 20 | 27 ± 20 | 24 ± 30 |
| HRT (days) | 6.1 ± 9.4 | 4.9 ± 4.6 | 3.2 ± 3.2 | 4.7 ± 7.8 |
| pH | 6.9 ± 0.3 | 7.4 ± 0.6 | 7.4 ± 0.7 | 7.4 ± 0.4 |
| Temperature (°C) | 13 ± 6 | 17 ± 6 | 19 ± 2 | 15 ± 6 |
| Effluent DO (mg L−1) | 1.4 ± 2.4 | 1.7 ± 2.2 | 5.4 ± 3.0 | 2.0 ± 1.9 |
| Class/PCPs | Possible Removal Mechanism | References | Dominant Removal Mechanism * |
|---|---|---|---|
| Artificial sweeteners | |||
| Acesulfame | Biodegradation (aerobic) | Kahl et al. [17]; Nivala et al. [19] | Biodegradation (aerobic) |
| Preservatives | |||
| Methylparaben | Plant uptake | Anjos et al. [31]; Petrie et al. [32] | Plant uptake; Biodegradation (aerobic); Photodegradation ** |
| Biodegradation (aerobic) | Matamoros et al. [9,15]; Anjos et al. [31]; Chen et al. [33] | ||
| Photodegradation | Chen et al. [33] | ||
| Hydrolysis | Chen et al. [33] | ||
| Volatilization | Chen et al. [33] | ||
| Propylparaben | Plant uptake | Anjos et al. [31] | Plant uptake; Biodegradation (aerobic); Photodegradation ** |
| Biodegradation (aerobic) | Anjos et al. [31] | ||
| Photodegradation | NA | ||
| Insect repellents | |||
| N, N-diethyl-meta-toluamide | Biodegradation (aerobic) | Li et al. [18]; Sgroi et al. [34] | Biodegradation (anaerobic) ** |
| Biodegradation (anaerobic) | Yi et al. [35]; Sgroi et al. [34] | ||
| Antiseptics | |||
| Triclosan | Adsorption | Carranza-Diaz et al. [22]; Chen et al. [16]; Liu et al. [36]; Xie et al. [24]; Button et al. [23]; Wang et al. [37] | Adsorption; Biodegradation (aerobic); Photodegradation |
| Sorption | Ávila et al. [7]; Vystavna et al. [12] | ||
| Biodegradation (aerobic) | Ávila et al. [6,7,38]; Zhang et al. [26]; Zhao et al. [39]; Chen et al. [16]; Liu et al. [36]; Li et al. [18]; Vymazal et al. [4]; Xie et al. [24]; Button et al. [23]; Chen et al. [33]; Wang et al. [37] | ||
| Biodegradation (anaerobic) | Park et al. [40]; Vystavna et al. [12] | ||
| Photodegradation | Matamoros and Salvadó [1]; Zhang et al. [26]; Ávila et al. [7,38]; Matamoros et al. [15]; Li et al. [18]; Vymazal et al. [4]; Vystavna et al. [12]; Francini et al. [41]; Chen et al. [33] | ||
| Plant uptake | Zhang et al. [26]; Liu et al. [36]; Dai et al. [8]; Li et al. [18]; Vymazal et al. [4]; Francini et al. [41]; Xie et al. [24] | ||
| Triclocarban | Sorption | Zhu and Chen [42]; Vymazal et al. [4] | Sorption ** |
| Fragrances | |||
| Methyl dihydro-jasmonate | Biodegradation (aerobic) | Matamoros et al. [5,15]; Hijosa-Valsero et al. [2,3,20,43]; Reyes- Contreras et al. [21] | Biodegradation (aerobic); Plant uptake |
| Biodegradation (anaerobic) | Hijosa-Valsero et al. [2] | ||
| Plant uptake | Hijosa-Valsero et al. [2,20]; Reyes-Contreras et al. [21]; Salcedo et al. [13] | ||
| Retention processes | Hijosa-Valsero et al. [20] | ||
| Cashmeran | Sorption | Matamoros and Salvadó [1] | Sorption **; Adsorption ** |
| Adsorption | NA | ||
| Galaxolide | Plant uptake | Hijosa-Valsero et al. [2,20]; Reyes-Contreras et al. [21]; Salcedo et al. [13] | Sorption; Adsorption |
| Adsorption | Hijosa-Valsero et al. [2,20]; Reyes-Contreras et al. [21] | ||
| Retention processes | Hijosa-Valsero et al. [20] | ||
| Sorption onto organic surfaces | Matamoros et al. [5,15]; Hijosa-Valsero et al. [2,3]; Matamoros and Salvadó [1]; Carranza-Diaz et al. [22] | ||
| Tonalide | Plant uptake | Hijosa-Valsero et al. [2,20]; Reyes-Contreras et al. [21] | Sorption; Adsorption |
| Adsorption | Hijosa-Valsero et al. [2,20]; Reyes-Contreras et al. [21] | ||
| Retention processes | Hijosa-Valsero et al. [20] | ||
| Sorption onto organic surfaces | Matamoros et al. [5,15]; Hijosa-Valsero et al. [2,3]; Matamoros and Salvadó [1]; Ávila et al. [6,14,38]; Carranza-Diaz et al. [22] | ||
| Photodegradation | Ávila et al. [7,38] | ||
| Flame retardants | |||
| Tributyl phosphate | Biodegradation | Matamoros et al. [15] | Sorption **; Biodegradation (aerobic) ** |
| Sorption | NA | ||
| Triphenyl phosphate | Biodegradation | Matamoros et al. [15] | Biodegradation (aerobic) **; Sorption ** |
| Sorption | NA | ||
| Tris (2-chloroethyl) phosphate | Recalcitrant to biodegradation | Matamoros and Salvadó [1]; Matamoros et al. [9,15] | Sorption ** |
| Sorption | NA | ||
| Plant uptake | NA | ||
| Sunscreen agents | |||
| Oxybenzone | Biodegradation (aerobic) | Matamoros and Salvadó [1]; Ávila et al. [6,7,14] | Adsorption **; Biodegradation (aerobic); Sorption |
| Sorption | Matamoros and Salvadó [1] | ||
| Adsorption | NA | ||
| Sulisobenzone | NA | NA | Biodegradation (aerobic) ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ilyas, H.; Hullebusch, E.D.v. The Influence of Design and Operational Factors on the Removal of Personal Care Products by Constructed Wetlands. Water 2020, 12, 1367. https://doi.org/10.3390/w12051367
Ilyas H, Hullebusch EDv. The Influence of Design and Operational Factors on the Removal of Personal Care Products by Constructed Wetlands. Water. 2020; 12(5):1367. https://doi.org/10.3390/w12051367
Chicago/Turabian StyleIlyas, Huma, and Eric D. van Hullebusch. 2020. "The Influence of Design and Operational Factors on the Removal of Personal Care Products by Constructed Wetlands" Water 12, no. 5: 1367. https://doi.org/10.3390/w12051367
APA StyleIlyas, H., & Hullebusch, E. D. v. (2020). The Influence of Design and Operational Factors on the Removal of Personal Care Products by Constructed Wetlands. Water, 12(5), 1367. https://doi.org/10.3390/w12051367






