Historical Changes in the Ecological Connectivity of the Seine River for Fish: A Focus on Physical and Chemical Barriers Since the Mid-19th Century
Abstract
1. Introduction
2. Materials and Methods
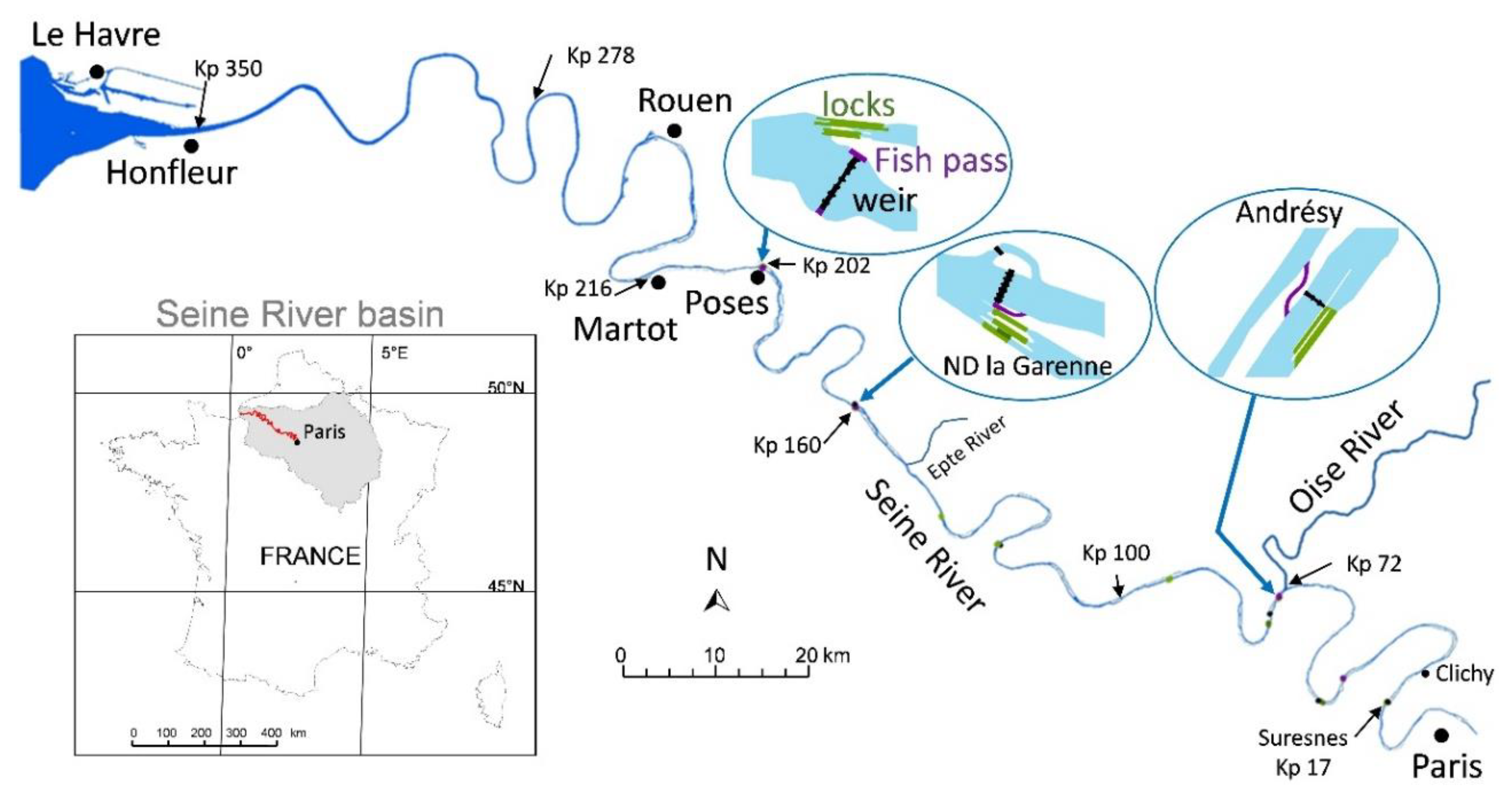
2.1. Context of the Study Area
2.2. Selected Fish Species
2.3. Historical Data
2.3.1. Physical Features and Infrastructures
2.3.2. Chemical Barriers
2.3.3. Fish Historical Distribution
2.4. Functional Connectivity Modeling
3. Results
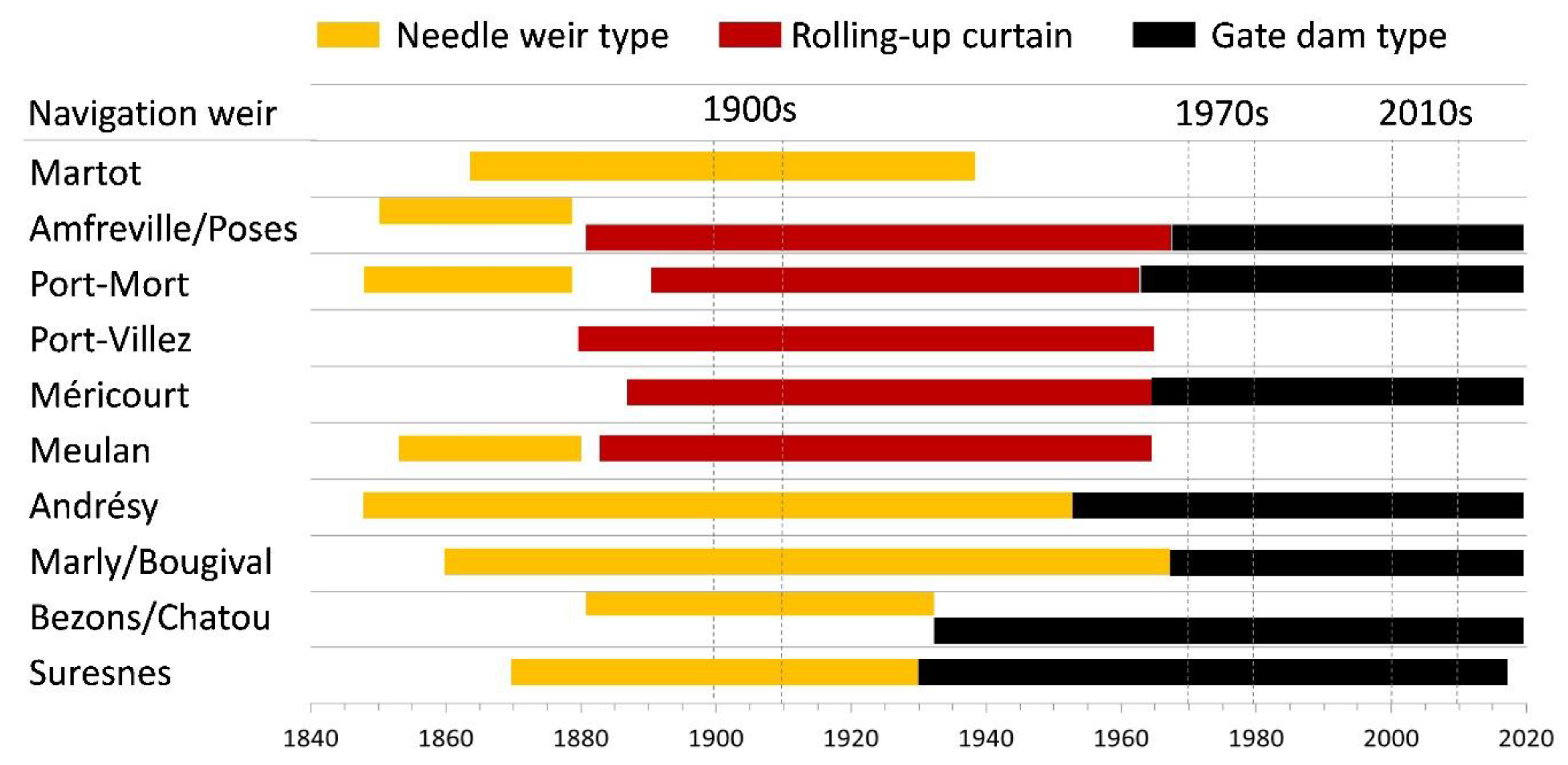
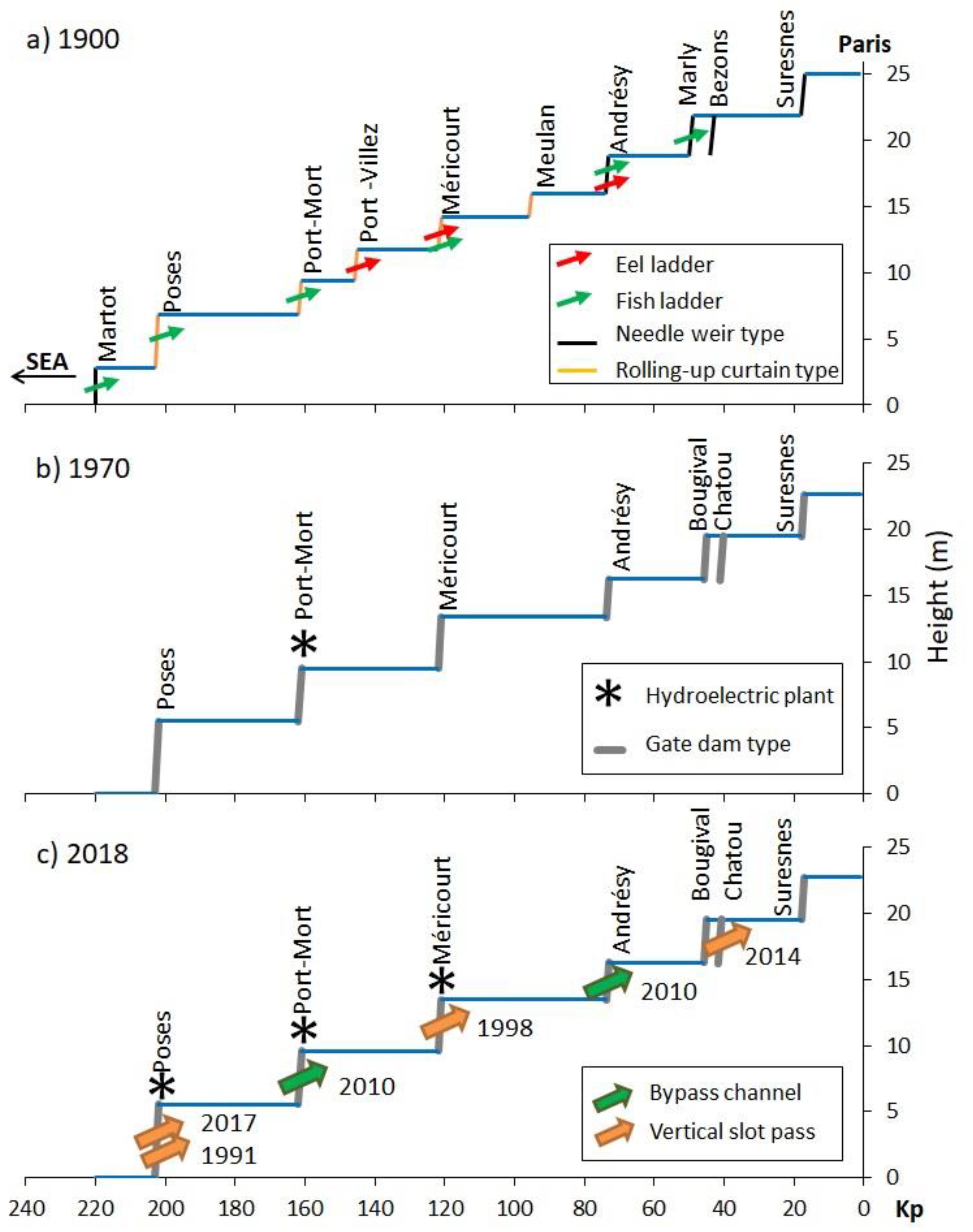
3.1. Historical Timeline of Weirs and Locks
3.2. History of Fish Passes
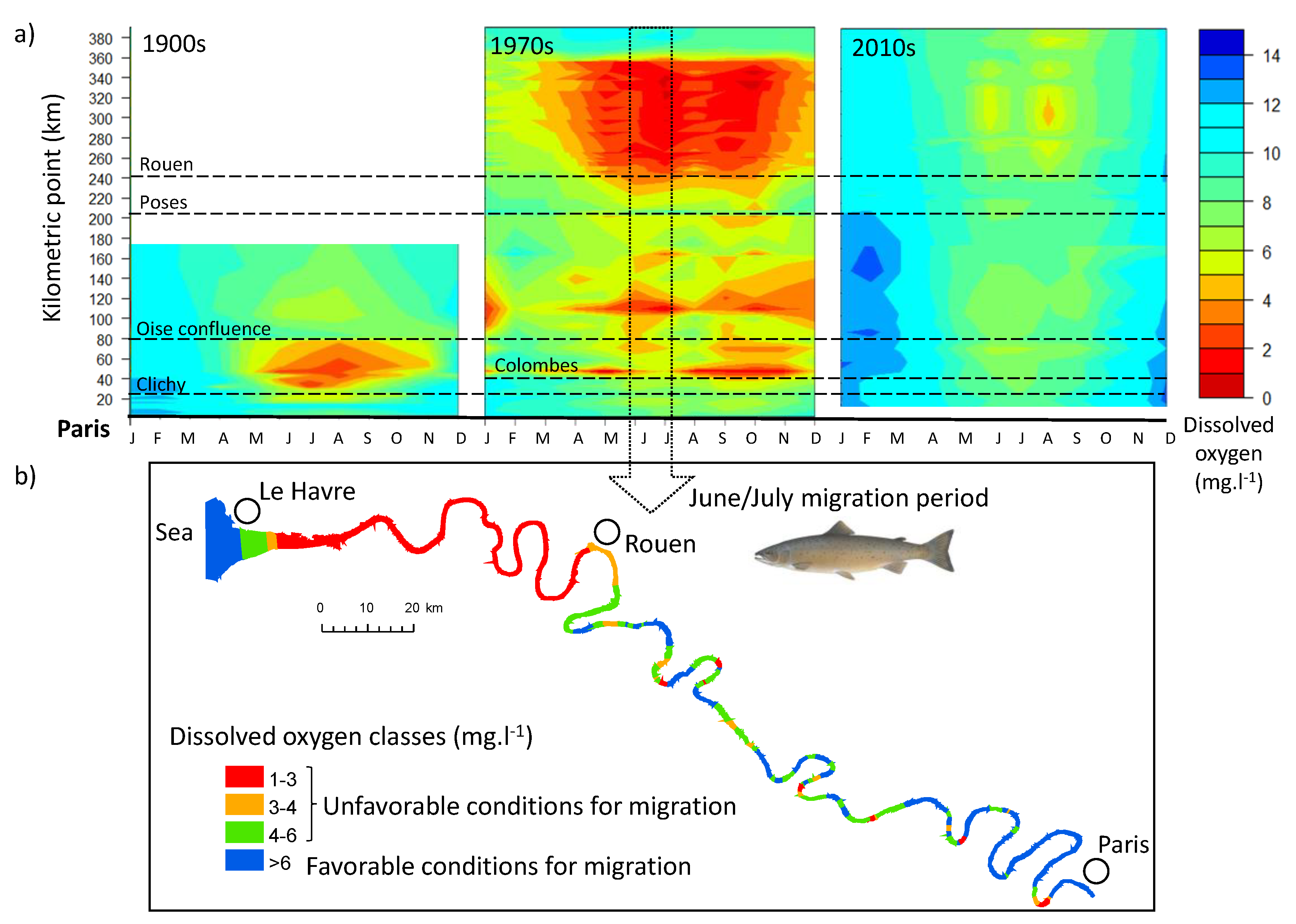
3.3. Dissolved Oxygen Evolution
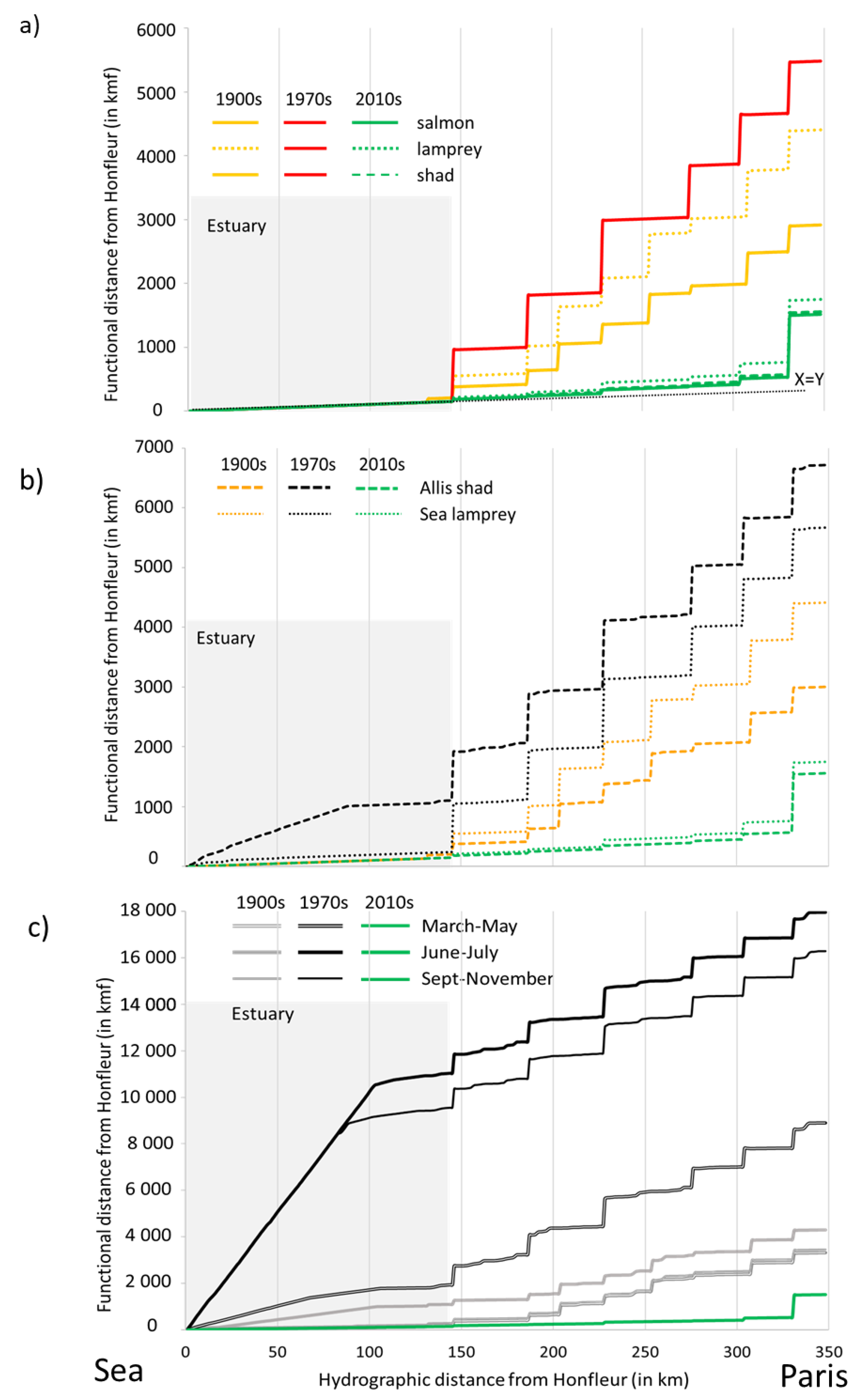
3.4. Accessibility: Comparison of Functional Distances
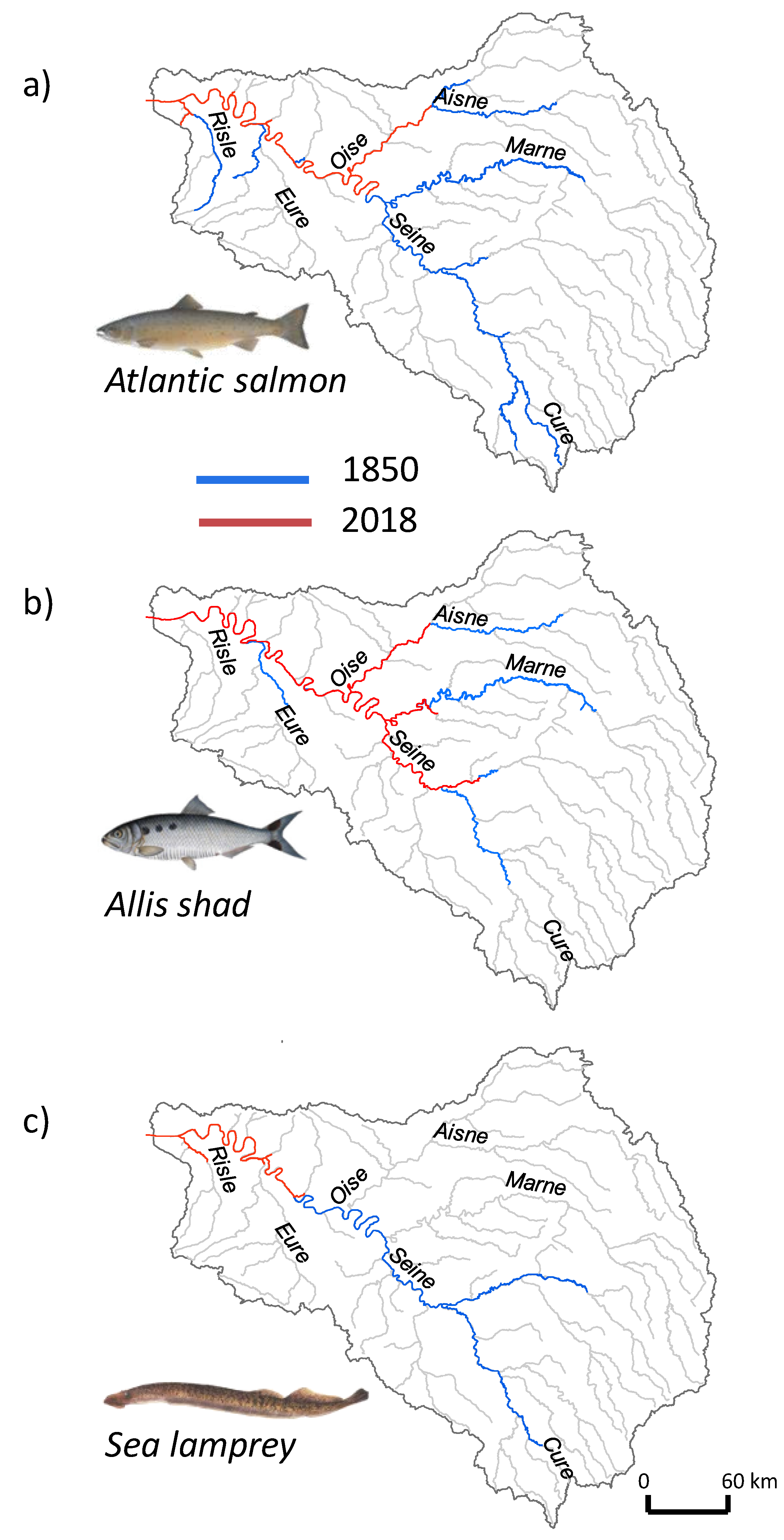
3.5. Historical Fish Distribution
4. Discussion
4.1. Reconstructing the Tide of History
4.2. The Modeling Approach
4.3. Management Implications and Perspectives
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thorp, J.H.; Thoms, M.C.; Delong, M.D. The riverine ecosystem synthesis: Biocomplexity in river networks across space and time. River Res. Appl. 2006, 22, 123–147. [Google Scholar] [CrossRef]
- WWF. Living Planet Report-2018: Aiming Higher; Grooten, M., Almond, R.E.A., Eds.; WWF International: Gland, Switzerland, 2018; 148p. [Google Scholar]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.-I.; Knowler, D.J.; Lévêque, C.; Naiman, R.J.; Prieur-Richard, A.-H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater biodiversity: Importance, threats, status and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef] [PubMed]
- Dynesius, M.; Nilsson, C. Fragmentation and flow regulation of river systems in the northern third of the world. Science 1994, 266, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Malaj, E.; Von der Ohe, P.C.; Grote, M.; Kühne, R.; Mondy, C.P.; Usseglio-Polatera, P.; Brack, W.; Schäfer, R.B. Organic chemicals jeopardize the health of freshwater ecosystems on the continental scale. Proc. Natl. Acad. Sci. USA 2014, 111, 9549–9554. [Google Scholar] [CrossRef]
- Fuller, M.R.; Doyle, M.W.; Strayer, D.L. Causes and consequences of habitat fragmentation in river networks. Ann. N. Y. Acad. Sci. 2015, 1355, 31–51. [Google Scholar] [CrossRef]
- Vörösmarty, C.J.; McIntyre, P.B.; Gessner, M.O.; Dudgeon, D.; Prusevich, A.; Green, P.; Glidden, S.; Bunn, S.E.; Sullivan, C.A.; Liermann, C.R. Global threats to human water security and river biodiversity. Nature 2010, 467, 555. [Google Scholar] [CrossRef]
- Duncan, J.R.; Lockwood, J.L. Extinction in a field of bullets: A search for causes in the decline of the world’s freshwater fishes. Biol. Conserv. 2001, 102, 97–105. [Google Scholar] [CrossRef]
- Fullerton, A.H.; Burnett, K.M.; Steel, E.A.; Flitcroft, R.L.; Pess, G.R.; Feist, B.E.; Torgersen, C.E.; Miller, D.J.; Sanderson, B.L. Hydrological connectivity for riverine fish: Measurement challenges and research opportunities. Freshw. Biol. 2010, 55, 2215–2237. [Google Scholar] [CrossRef]
- Pringle, C.M. Hydrologic connectivity and the management of biological reserves: A global perspective. Ecol. Appl. 2001, 11, 981–998. [Google Scholar] [CrossRef]
- Falke, J.A.; Dunham, J.B.; Jordan, C.E.; McNyset, K.M.; Reeves, G.H. Spatial ecological processes and local factors predict the distribution and abundance of spawning by Steelhead (Oncorhynchus mykiss) across a complex riverscape. PLoS ONE 2013, 8, e79232. [Google Scholar] [CrossRef]
- Belliard, J.; Beslagic, S.; Delaigue, O.; Tales, E. Reconstructing long-term trajectories of fish assemblages using historical data: The Seine River basin (France) during the last two centuries. Environ. Sci. Pollut. Res. 2018, 25, 23430–23450. [Google Scholar] [CrossRef] [PubMed]
- Haidvogl, G.; Hoffmann, R.; Pont, D. Historical ecology of riverine fish in Europe. Aquat. Sci. 2015, 77, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Lenders, H.; Chamuleau, T.; Hendriks, A.; Lauwerier, R.; Leuven, R.; Verberk, W. Historical rise of waterpower initiated the collapse of salmon stocks. Sci. Rep. 2016, 6, 29269. [Google Scholar] [CrossRef] [PubMed]
- Lobón-Cerviá, J.; Elvira, B.; Rincón, P. Historical Changes in the Fish Fauna of the River Duero Basin. In Historical Change of Large Alluvial Rivers; Western Europe; Petts, G.E., Möller, H., Roux, A.L., Eds.; John Wiley and Sons: Chichester, UK, 1989; pp. 221–232. [Google Scholar]
- Segurado, P.; Branco, P.; Avelar, A.P.; Ferreira, M.T. Historical changes in the functional connectivity of rivers based on spatial network analysis and the past occurrences of diadromous species in Portugal. Aquat. Sci. 2015, 77, 427–440. [Google Scholar] [CrossRef]
- Van Puijenbroek, P.J.T.M.; Buijse, A.D.; Kraak, M.H.S.; Verdonschot, P.F.M. Species and river specific effects of river fragmentation on European anadromous fish species. River Res. Appl. 2019, 35, 68–77. [Google Scholar] [CrossRef]
- Tétard, S.; Feunteun, E.; Bultel, E.; Gadais, R.; Bégout, M.-L.; Trancart, T.; Lasne, E. Poor oxic conditions in a large estuary reduce connectivity from marine to freshwater habitats of a diadromous fish. Estuar. Coast. Shelf Sci. 2016, 169, 216–226. [Google Scholar] [CrossRef]
- Maes, J.; Stevens, M.; Breine, J. Poor water quality constrains the distribution and movements of twaite shad Alosa fallax fallax (Lacépède, 1803) in the watershed of river Scheldt. In Fish and Diadromy in Europe (Ecology, Management, Conservation); Dufour, S., Prévost, E., Rochard, E., Williot, P., Eds.; Developments in Hydrobiology; Springer: Dordrecht, The Netherlands, 2008; Volume 200, pp. 129–143. [Google Scholar]
- McDowall, R.M. Diadromy in Fishes: Migrations between Freshwater and Marine Environments; Croom Helm: London, UK, 1988; 250p. [Google Scholar]
- Dufour, S.; Prévost, E.; Rochard, E.; Williot, P. Fish and Diadromy in Europe (Ecology, Management, Conservation); Developments in Hydrobiology; Springer: Dordrecht, The Netherlands, 2008; Volume 200, 186p. [Google Scholar]
- Cote, D.; Kehler, D.; Bourne, C.; Wiersma, Y. A new measure of longitudinal connectivity for stream networks. Landsc. Ecol. 2009, 24, 101–113. [Google Scholar] [CrossRef]
- Adriaensen, F.; Chardon, J.P.; De Blust, G.; Swinnen, E.; Villalba, S.; Gulinck, H.; Matthysen, E. The application of ‘least-cost’ modelling as a functional landscape model. Landsc. Urban Plan. 2003, 64, 233–247. [Google Scholar] [CrossRef]
- Foubert, A.; Le Pichon, C.; Mingelbier, M.; Farrell, J.M.; Morin, J.; Lecomte, F. Modeling the effective spawning and nursery habitats of northern pike within a large spatiotemporally variable river landscape (St. Lawrence River, Canada). Limnol. Oceanogr. 2019, 64, 803–819. [Google Scholar] [CrossRef]
- Caldwell, I.; Gergel, S. Thresholds in seascape connectivity: Influence of mobility, habitat distribution, and current strength on fish movement. Landsc. Ecol. 2013, 28, 1937–1948. [Google Scholar] [CrossRef]
- Shaffer, H.B.; Fisher, R.N.; Davidson, C. The role of natural history collections in documenting species declines. Trends Ecol. Evol. 1998, 13, 27–30. [Google Scholar] [CrossRef]
- Guillerme, A. Le testament de la Seine/The legacy of the Seine. Géocarrefour 1990, 65, 240–250. [Google Scholar] [CrossRef]
- Meybeck, M.; Lestel, L.; Briand, C. La Seine sous Surveillance: Les Analyses des Impacts de L’agglomération Parisienne par L’observatoire de Montsouris de 1876 à 1937. In Les Rivières Urbaines et Leur Pollution; Lestel, L., Carré, C., Eds.; Collection Indisciplines, Editions Quae: Versailles, France, 2017; pp. 32–43. [Google Scholar]
- Lafite, R.; Romaña, L.-A. A man-altered macrotidal estuary: The Seine estuary (France): Introduction to the special issue. Estuaries Coasts 2001, 24, 939. [Google Scholar] [CrossRef]
- Barles, S. Urban metabolism and river systems: An historical perspective–Paris and the Seine, 1790–1970. Hydrol. Earth Syst. Sci. 2007, 4, 1845–1878. [Google Scholar] [CrossRef]
- Meybeck, M.; Lestel, L.; Carré, C.; Bouleau, G.; Garnier, J.; Mouchel, J.M. Trajectories of river chemical quality issues over the Longue Durée: The Seine River (1900s–2010). Environ. Sci. Pollut. Res. 2018, 25, 23468–23484. [Google Scholar] [CrossRef] [PubMed]
- Belliard, J.; Boet, P.; Allardi, J. Évolution à long terme du peuplement piscicole du bassin de la Seine. Bull. Fr. Pêche Piscic. 1995, 83–91. [Google Scholar] [CrossRef]
- Belliard, J.; Beslagic, S.; Tales, E. Changes in Fish Communities of the Seine Basin over a Long-Term Perspective. In The Seine River Basin; Flipo, N., Labadie, P., Lestel, L., Eds.; Handbook of Environmental Chemistry; Springer: Cham, Switzerland, 2020. [Google Scholar]
- Cavailler, P. Le fief de la Mothe ou maîtrise de l’eau de Corbeil au début du XVIIe siècle. In Paris et Ile-de-France, Mémoires, t. 45, La Seine et son Histoire en Ile-de-France; Fédération des sociétés historiques et archéologiques de Paris et Ile-de-France: Paris, France, 1994; pp. 169–192. [Google Scholar]
- Benoit, P.; Loridant, F.; Matteoni, O. Pêche et Pisciculture en eau douce: La Rivière et L’étang au Moyen Age. In Actes des 1es Rencontres Internationales de Liessies (conference proceedings); Conseil général du Nord: Lille, France, 2004; pp. 4–5. [Google Scholar]
- UICN; MNHN; AFB. Liste Rouge des Espèces Menacées en France, les Poissons D’eau Douce de France Métropolitaine; UICN: Montreuil, France, 2019; 16p. [Google Scholar]
- Rochard, E.; Pellegrini, P.; Marchal, J.; Béguer, M.; Ombredane, D.; Lassalle, G.; Menvielle, E.; Baglinière, J. Identification of diadromous fish species on which to focus river restoration: An example using an eco-anthropological approach (the Seine basin, France). In Proceedings of the Challenges for Diadromous Fishes in a Dynamic Global Environment, Halifax, NS, Canada, 18–21 June 2007; pp. 691–711. [Google Scholar]
- Belliard, J.; Marchal, J.; Ditche, J.-M.; Tales, E.; Sabatié, R.; Baglinière, J.-L. Return of adult anadromous allis shad (Alosa alosa L.) in the river Seine, France: A sign of river recovery? River Res. Appl. 2009, 25, 788–794. [Google Scholar] [CrossRef]
- Baudoin, J.; Burgun, V.; Chanseau, M.; Larinier, M.; Ovidio, M.; Sremski, W.; Steinbach, P.; Voegtle, B. Evaluer le franchissement des obstacles par les poissons. In Principes et Méthodes; Onema: Paris, France, 2014. [Google Scholar]
- Claridge, P.; Potter, I. Oxygen consumption, ventilatory frequency and heart rate of lampreys (Lampetra fluviatilis) during their spawning run. J. Exp. Biol. 1975, 63, 193–206. [Google Scholar]
- Cardin; Babin. Carte de L’estuaire de la Seine Indiquant les Variations du Chenal de 1880 à 1900; 1907. [Google Scholar]
- AESN. Tome 2: Besoins et Utilisation d’eau, Pollution. Fascicule 6—Hydraulique Fluviale et Voies Navigables; Les Bassins de la Seine et des cours d’eau Normands: Paris, France, 1976; 140p. [Google Scholar]
- Agence Financière de Bassin Seine Normandie; Notre fleuve: La Seine, France, 1980; 32p.
- Lescure, S.; Arnaud-Fassetta, G.; Cordier, S. Sur quelques modifications hydromorphologiques dans le Val de Seine (Bassin parisien, France) depuis 1830: Quelle part accorder aux facteurs hydrologiques et anthropiques? EchoGéo 2011, 8, 1–18. [Google Scholar] [CrossRef]
- Leynaud, G.; Trocheris, F.; Larinier, M. Les obstacles à la réalisation du cycle vital des poissons. La Houille Blanche 1987, 39–44. [Google Scholar] [CrossRef]
- Beslagic, S. Histoire des Interactions Entre les Sociétés Humaines et le Milieu Aquatique Durant L’anthropocene. Évolutions des Peuplements Piscicoles dans le Bassin de la Seine. Ph.D. Thesis, Université Paris 1-Panthéon-Sorbonne, Paris, France, 2013. [Google Scholar]
- Carbonaro-Lestel, L.; Meybeck, M. La mesure de la qualité chimique de l’eau, 1850–1970. La Houille Blanche 2009, 3, 25–30. [Google Scholar] [CrossRef]
- Meybeck, M.; Lestel, L.; Briand, C. L’impact de l’agglomération parisienne sur le milieu aquatique de 1876 à 1937, dans les travaux de l’Observatoire de Montsouris. In Les Rivères Urbaines et Leur Pollution; Lestel, L., Carré, C., Eds.; Collection Indisciplines, Editions Quae: Versailles, France, 2017; pp. 32–42. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Akima, H.; Gebhardt, A.; Petzold, T.; Maechler, M. Akima: Interpolation of Irregularly and Regularly Spaced Data. R Package Version 0.6-2. 2016. Available online: https://www.google.com.hk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwiZ0om8uajpAhUPQd4KHXcKAawQFjAAegQIBBAB&url=https%3A%2F%2Fcran.r-project.org%2Fweb%2Fpackages%2Fakima%2Fakima.pdf&usg=AOvVaw2l4Omur7FN7TZ3F_f2hbUk (accessed on 10 May 2020).
- Akima, H. A method of bivariate interpolation and smooth surface fitting for irregularly distributed data points. ACM Trans. Math. Software (TOMS) 1978, 4, 148–159. [Google Scholar] [CrossRef]
- Cleveland, W.S. Visualizing Data; Hobart Press: Summit, NJ, USA, 1993; 360p. [Google Scholar]
- Beslagic, S.; Marinval, M.-C.; Belliard, J. CHIPS: A database of historic fish distribution in the Seine River basin (France). Cybium 2013, 37, 75–93. [Google Scholar]
- Merg, M.L.; Dézerald, O.; Kreutzenberger, K.; Demski, S.; Reyjol, Y.; Belliard, J. Modeling diadromous fish loss from historical data: Identification of anthropogenic drivers and testing of mitigation scenarios. PLoS ONE. (under review).
- Le Pichon, C.; Gorges, G.; Faure, T.; Boussard, H. Anaqualand 2.0: Freeware of distances calculations with Frictions on a Corridor; Cemagref Antony. 2006. Available online: https://www6.rennes.inra.fr/sad/Outils-Produits/Outils-informatiques/Anaqualand (accessed on 10 May 2020).
- De Mas, F.B. Chapitre III: Barrage mobile à pont supérieur. In Cours de Navigation Intérieure de l’Ecole Nationale des Ponts et Chaussées; Rivières Canalisées, Librairie Polytechnique: Paris, France, 1903; pp. 131–143. [Google Scholar]
- De Guibert, P. Impact des aménagements pour la navigation sur les niveaux d’eau de la Seine en aval de Paris. La Houille Blanche 1997, 8, 48–50. [Google Scholar] [CrossRef]
- Larinier, M. Upstream and downstream fish passage experience in France. In Fish Migration and Fish Bypasses; Jungwirth, M., Schmutz, S., Weiss, S., Eds.; Fishing News Books: Oxford, UK, 1998; pp. 127–145. [Google Scholar]
- Katopodis, C.; Williams, J.G. The development of fish passage research in a historical context. Ecol. Eng. 2012, 48, 8–18. [Google Scholar] [CrossRef]
- Lestel, L.; Carré, C. Les Rivières Urbaines et Leur Pollution; Collection Indisciplines, Editions Quæ: Versailles, France, 2017; 286p. [Google Scholar]
- Romero, E.; Le Gendre, R.; Garnier, J.; Billen, G.; Fisson, C.; Silvestre, M.; Riou, P. Long-term water quality in the lower Seine: Lessons learned over 4 decades of monitoring. Environ. Sci. Policy 2016, 58, 141–154. [Google Scholar] [CrossRef]
- Arrignon, J. Comportement de l’espèce “Salmo trutta” dans le bassin de la Seine Suite (1). Bull. Fr. Piscic. 1968, 228, 77–101. [Google Scholar] [CrossRef]
- Lansing, J.S. Complex adaptive systems. Annu. Rev. Anthropol. 2003, 32, 183–204. [Google Scholar] [CrossRef]
- Meybeck, M.; Lestel, L. A Western European River in the Anthropocene: The Seine, 1870–2010. In Rivers of the Anthropocene; Kelly, J.M., Scarpino, P., Berry, H., Syvitski, J., Meybeck, M., Eds.; University of California Press: Oakland, CA, USA, 2017; pp. 84–100. [Google Scholar]
- Soetaert, K.; Middelburg, J.J.; Heip, C.; Meire, P.; Van Damme, S.; Maris, T. Long-term change in dissolved inorganic nutrients in the heterotrophic Scheldt estuary (Belgium, The Netherlands). Limnol. Oceanogr. 2006, 51, 409–423. [Google Scholar] [CrossRef]
- Fernandes, M.R.; Aguiar, F.; Martins, M.; Rivaes, R.; Ferreira, M. Long-term human-generated alterations of Tagus River: Effects of hydrological and land-use changes in distinct river zones. Catena 2020, 188, 1–14. [Google Scholar] [CrossRef]
- Winiwarter, V.; Schmid, M.; Dressel, G. Looking at half a millennium of co-existence: The Danube in Vienna as a socio-natural site. Water Hist. 2013, 5, 101–119. [Google Scholar] [CrossRef]
- Limburg, K.E.; Waldman, J.R. Dramatic declines in North Atlantic diadromous fishes. BioScience 2009, 59, 955–965. [Google Scholar] [CrossRef]
- Lavollée, G. Le saumon en Seine. Bull. Soc. Cent. Aquic. Pêche 1902, 14, 221–234. [Google Scholar]
- Tales, E.; Jérôme, B.; Beslagic, S.; Stefani, F.; Tartari, G.; Wolter, C. Réponse des Peuplements de Poissons à L’urbanisation et aux Altérations Anthropiques à Long Terme des Cours d’eau. In Les Rivières Urbaines et Leur Pollution; Lestel, L., Carré, C., Eds.; Collection Indisciplines, Editions Quæ: Versailles, France, 2017; pp. 242–252. [Google Scholar]
- Maes, J.; Stevens, M.; Breine, J. Modelling the migration opportunities of diadromous fish species along a gradient of dissolved oxygen concentration in a European tidal watershed. Estuar. Coast. Shelf Sci. 2007, 75, 151–162. [Google Scholar] [CrossRef]
- Moreau, E. Les poissons du département de l’Yonne. Bull. Soc. Sci. Hist. Nat. Yonne 1898, 52, 3–82. [Google Scholar]
- Garnier, J.; Billen, G.; Cébron, A. Modelling nitrogen transformations in the lower Seine river and estuary (France): Impact of wastewater release on oxygenation and N²O emission. Hydrobiologia 2007, 588, 291–302. [Google Scholar] [CrossRef]
- Euzenat, G.; Penil, C.; Allardi, J. Migr’en Seine. Stratégie Pour le Retour du Saumon en Seine; Rapport Conseil Supérieur de la Pêche/SIAAP: Paris, France, 1992; 38p. [Google Scholar]
- Guillard, J.; Colon, M. First results on migrating shad (Alosa fallax) and mullet (Mugil cephalus) echocounting in a lock on the Rhône River (France) using a split-beam sounder, and relationships with environmental data and fish caught. Aquat. Living Resour. 2000, 13, 327–330. [Google Scholar] [CrossRef]
- Garcier, R. Rivers we can’t bring ourselves to clean–historical insights into the pollution of the Moselle River (France), 1850–2000. Hydrol. Earth Syst. Sci. 2007, 11, 1731–1745. [Google Scholar] [CrossRef]
- Van Dijk, G.; Marteijn, E.; Schulte-Wülwer-Leidig, A. Ecological rehabilitation of the River Rhine: Plans, progress and perspectives. Regul. Rivers Res. Manag. 1995, 11, 377–388. [Google Scholar] [CrossRef]
- Roy, M.L.; Le Pichon, C. Modelling functional fish habitat connectivity in rivers: A case study for prioritizing restoration actions targeting brown trout. Aquat. Conserv. Mar. Freshw. Ecosyst. 2017, 27, 927–937. [Google Scholar] [CrossRef]
- Beier, P.; Majka, D.R.; Spencer, W.D. Forks in the road: Choices in procedures for designing wildland linkages. Conserv. Biol. 2008, 22, 836–851. [Google Scholar] [CrossRef] [PubMed]
- Le Pichon, C.; Coustillas, J.; Zahm, A.; Bunel, M.; Gazeau-Nadin, C.; Rochard, E. Summer use of the tidal freshwaters of the River Seine by three estuarine fish: Coupling telemetry and GIS spatial analysis. Estuar. Coast. Shelf Sci. 2017, 196, 83–96. [Google Scholar] [CrossRef]
- Baisez, A.; Bach, J.-M.; Leon, C.; Parouty, T.; Terrade, R.; Hoffmann, M.; Laffaille, P. Migration delays and mortality of adult Atlantic salmon Salmo salar en route to spawning grounds on the River Allier, France. Endanger. Species Res. 2011, 15, 265–270. [Google Scholar] [CrossRef]
- Haidvogl, G.; Winiwarter, V.; Brumat, S. Natural History of the Danube Region. In Danube: Future Interdisciplinary School Proceedings 2017; Hanus, C., Steiner, G., Eds.; Edition Donau Universitat Krems: Krems an der Donau, Austria, 2019; pp. 43–54. [Google Scholar]
- Hein, T.; Funk, A.; Pletterbauer, F.; Graf, W.; Zsuffa, I.; Haidvogl, G.; Schinegger, R.; Weigelhofer, G. Management challenges related to long-term ecological impacts, complex stressor interactions, and different assessment approaches in the Danube River Basin. River Res. Appl. 2019, 35, 500–509. [Google Scholar] [CrossRef]
- Bouleau, G. La catégorisation politique des eaux sous l’angle de la political ecology: Le patrimoine piscicole et la pollution en France. Lespace Geogr. 2017, 46, 214–230. [Google Scholar] [CrossRef]
- Violette, A. La disparition du saumon. Bull. Soc. Cent. Aquic. Pêche 1902, 14, 181–197. [Google Scholar]
- Boet, P.; Duvoux, B.; Allardi, J.; Belliard, J. Incidence des orages estivaux sur le peuplement piscicole de la Seine à l’aval de l’agglomération parisienne (Bief Andresy-Mericourt). La Houille Blanche 1994, 141–147. [Google Scholar] [CrossRef]
- Moore, H.E.; Rutherfurd, I.D. Lack of maintenance is a major challenge for stream restoration projects. River Res. Appl. 2017, 33, 1387–1399. [Google Scholar] [CrossRef]
- Wang, S.; Flipo, N.; Romary, T. Assimilating high-frequency data in a hydro-biogeochemical model of river systems, the ProSe-PA approach. Geophys. Res. Abstr. 2019, 21, 1. [Google Scholar]
- Ducharne, A.; Eric, S.; Habets, F.; Deque, M.; Gascoin, S.; Hachour, A.; Martin, E.; Oudin, L.; Page, C.; Terray, L.; et al. Potential evolution of the Seine River flood regime under climate change. La Houille Blanche 2011, 1, 51–57. [Google Scholar] [CrossRef][Green Version]
| Allis Shad | Atlantic Salmon | Sea Lamprey | |
|---|---|---|---|
| National status 2019 | CR | NT, VU (Allier River population) | EN |
| Dissolved O2 (mg·L−1) | >4 | ≥6 | >3 * |
| Swimming capacities: burst speed, (m·s−1) | 3.5–5.0 | 4.5–6.5 | 3.0–4.5 |
| Behavior to cross obstacle | Swimming | Swimming, high jumping ability | Swimming and “burst and attach” (suction cup mouth) |
| Migration size [Seine River] | 45–70 cm | 50–100 cm [1SW < 75 cm, 2SW < 90 cm] | 60–90 cm |
| Migration period(s) (Seine River) | March to June |
| March to June |
| Resistance Values | ||||||
|---|---|---|---|---|---|---|
| Type of Barrier | Biological Cost/Risk | Longitudinal Barriers Thickness (m) | A. Salmon | S. Lamprey | A. Shad | |
| Physical | Hydroelectric dam | Energetic cost/predation | 10 | 80,000 | 80,000 | 80,000 |
| Gate dam | 10 | 80,000 | 80,000 | 80,000 | ||
| Lateral fish ladder | 10 | 80,000 | 80,000 | 80,000 | ||
| Operating/closed lock | 100 | 8000 | 8000 | 8000 | ||
| Needle weir | 10 | 40,000 | 60,000 | 40,000 | ||
| Rolling curtain weir | 10 | 40,000 | 60,000 | 40,000 | ||
| Fish ladder | 10 | 10,000 | 20,000 | 10,000 | ||
| Fish pass: vertical slot | 20 | 1000 | 2000 | 1000 | ||
| Fish pass: secondary channel | 200 | 50 | 200 | 150 | ||
| Disused open lock | Minimal cost | 100 | 2 | 2 | 2 | |
| Chemical | Reach oxygen class 1–3 mg·L−1 | Mortality/physiologic cost | 1000 | 100 * | 10 * | 20 * |
| Reach oxygen class 3–4 mg·L−1 | 1000 | 20 * | 1 * | 10 * | ||
| Reach oxygen class 4–6 mg·L−1 | Minimal cost | 1000 | 10 * | 1 | 1 | |
| Reach oxygen class > 6 mg·L−1 | No cost assumed | 1000 | 1 | 1 | 1 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le Pichon, C.; Lestel, L.; Courson, E.; Merg, M.-L.; Tales, E.; Belliard, J. Historical Changes in the Ecological Connectivity of the Seine River for Fish: A Focus on Physical and Chemical Barriers Since the Mid-19th Century. Water 2020, 12, 1352. https://doi.org/10.3390/w12051352
Le Pichon C, Lestel L, Courson E, Merg M-L, Tales E, Belliard J. Historical Changes in the Ecological Connectivity of the Seine River for Fish: A Focus on Physical and Chemical Barriers Since the Mid-19th Century. Water. 2020; 12(5):1352. https://doi.org/10.3390/w12051352
Chicago/Turabian StyleLe Pichon, Céline, Laurence Lestel, Emeric Courson, Marie-Line Merg, Evelyne Tales, and Jérôme Belliard. 2020. "Historical Changes in the Ecological Connectivity of the Seine River for Fish: A Focus on Physical and Chemical Barriers Since the Mid-19th Century" Water 12, no. 5: 1352. https://doi.org/10.3390/w12051352
APA StyleLe Pichon, C., Lestel, L., Courson, E., Merg, M.-L., Tales, E., & Belliard, J. (2020). Historical Changes in the Ecological Connectivity of the Seine River for Fish: A Focus on Physical and Chemical Barriers Since the Mid-19th Century. Water, 12(5), 1352. https://doi.org/10.3390/w12051352





