The Effects of Sodium Percarbonate Generated Free Oxygen on Daphnia—Implications for the Management of Harmful Algal Blooms
Abstract
:1. Introduction
2H2O2 → 2H2O + O2
2. Materials and Methods
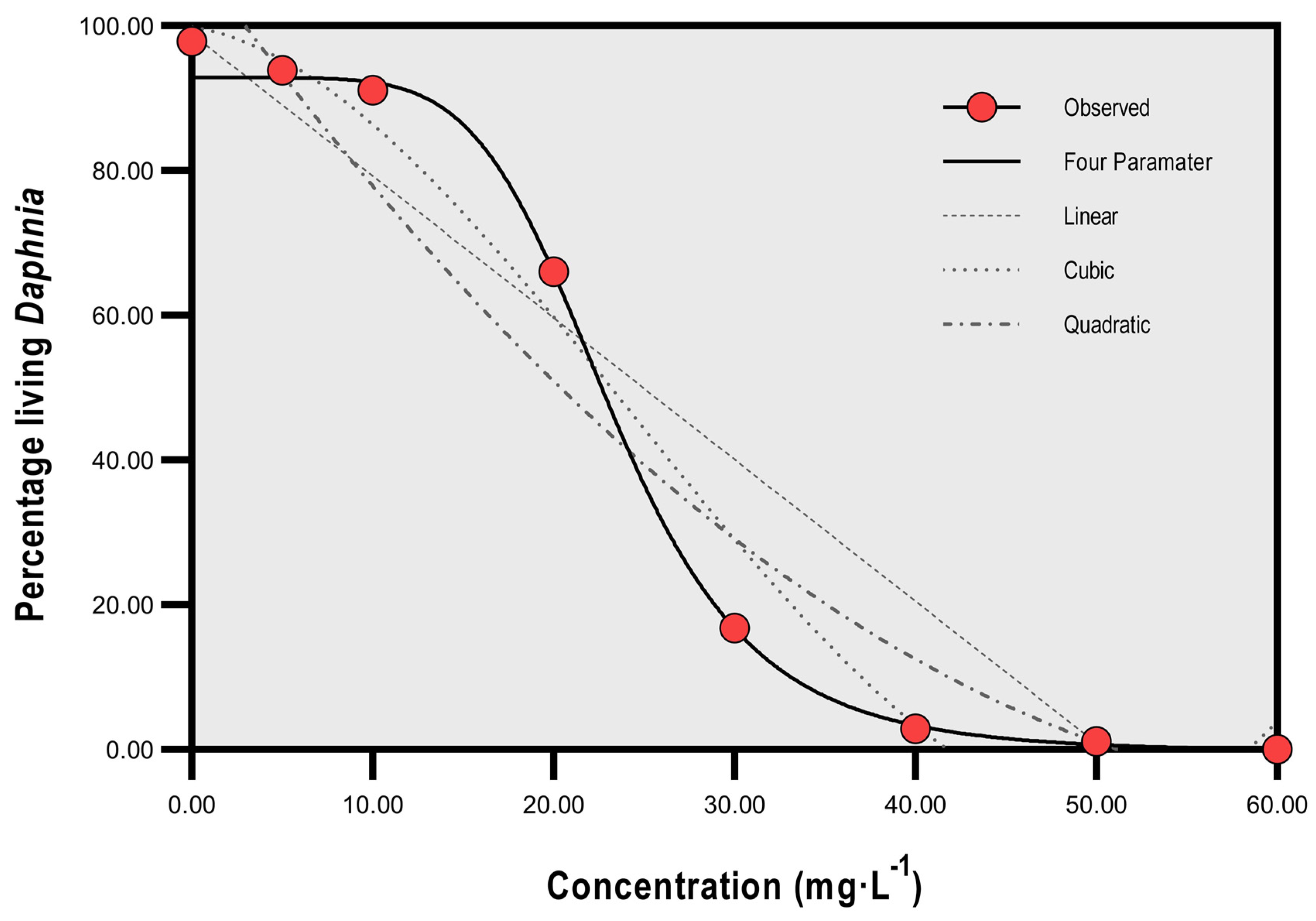
3. Results
Total Mortality and Concentration of SP
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shurin, J.B.; Gruner, D.S.; Hillebrand, H. All wet or dried up? Real differences between aquatic and terrestrial food webs. Proc. R. Soc. B Biol. Sci. 2005, 273, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Field, C.B.; Behrenfeld, M.J.; Randerson, J.T.; Falkowski, P. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 1998, 281, 237–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, D.M. Prevention, control, and mitigation of harmful algal blooms: Multiple approaches to HAB management. In Harmful Algae Management mitigate, Heng Mui Keng, Terrace, Singapore, 21 December 2004; Etheridge, S., Anderson, D., Kleindinst, J., Zhu, M., Zou, Y., Eds.; Asia Pacific Economic Cooperation: Heng Mui Keng Terrace, Singapore, 2004; pp. 123–130. [Google Scholar]
- Barrington, D.; Xiao, X.; Coggins, L.; Ghadouani, A. Control and management of harmful algal blooms. In Climate Change and Marine and Freshwater Toxins Botana; Louzao, L.M., Vilariño, M.C., Eds.; N. De Gruyter: Berlin, Germany, 2015; pp. 313–358. ISBN 978-3-11-033303-9. [Google Scholar]
- Meriluoto, J.; Blaha, L.; Bojadzija, G.; Bormans, M.; Brient, L.; Codd, G.A.; Drobac, D.; Faassen, E.J.; Fastner, J.; Hiskia, A.; et al. Toxic cyanobacteria and cyanotoxins in european waters—Recent Progress Achieved through the CYANOCOST Action and Challenges for Further Research. Adv. Oceanogr. Limnol 2017, 8, 161–178. [Google Scholar] [CrossRef] [Green Version]
- Medrano, E.A.; Uittenbogaard, R.; Wiel, B.V.D.; Pires, L.D.; Clercx, H. An alternative explanation for cyanobacterial scum formation and persistence by oxygenic photosynthesis. Harmful Algae 2016, 60, 27–35. [Google Scholar] [CrossRef]
- Tiling, K.; Proffitt, C.E. Effects of Lyngbya Majuscula blooms on the seagrass Halodule Wrightii and resident invertebrates. Harmful Algae 2017, 62, 104–112. [Google Scholar] [CrossRef]
- Ghadouani, A.; Pinel-Alloul, B.; Prepas, E.E. Effects of experimentally induced cyanobacterial blooms on crustacean zooplankton communities. Freshw. Biol. 2003, 48, 363–381. [Google Scholar] [CrossRef]
- Weenink, E.F.J.; Luimstra, V.M.; Schuurmans, J.M.; Herk, M.J.V.; Visser, P.M.; Matthijs, H.C.P. Combatting Cyanobacteria with Hydrogen Peroxide: A laboratory study on the consequences for phytoplankton community and diversity. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [Green Version]
- Dokulil, M.T.; Teubner, K. Cyanobacterial dominance in lakes. Hydrobiologia 2000, 438, 1−12. [Google Scholar] [CrossRef]
- Matthijs, H.C.P.; Jančula, D.; Visser, P.M.; Maršálek, B. Existing and emerging cyanocidal compounds: New perspectives for cyanobacterial bloom mitigation. Aquat. Ecol. 2016, 50, 443–460. [Google Scholar] [CrossRef] [Green Version]
- Drábková, M.; Admiraal, W.; Maršálek, B. Combined exposure to hydrogen peroxide and light selective effects on cyanobacteria, green algae, and diatoms. Environ. Sci. Technol. 2007, 41, 309–314. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Veljovic-Jovanovic, S. Active oxygen species and photosynthesis: Mehler and ascorbate peroxidase reactions. Iugoslav Physiol. Pharmacol. Acta 1998, 34, 503–522. [Google Scholar]
- Matthijs, H.C.; Visser, P.M.; Reeze, B.; Meeuse, J.; Slot, P.C.; Wijn, G.; Talens, R.; Huisman, J. Selective suppression of harmful cyanobacteria in an entire lake with hydrogen peroxide. Water Res. 2012, 46, 1460–1472. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.K.; Eggleton, M.A.; Lochmann, R.T. An environmentally friendly approach for mitigating cyanobacterial bloom and their toxins in hypereutrophic ponds: Potentiality of a newly developed granular hydrogen peroxide-based compound. Sci. Total Environ. 2018, 637–638, 524–537. [Google Scholar] [CrossRef]
- Massachusetts Department of Environmental Protection—Hydrogen Peroxide, Peracetic Acid and Sodium Percarbonate. Available online: http://www.mass.gov/eea/docs/agr/pesticides/aquatic/sodium-carbonate-peroxyhydrate-and-hydrogen-peroxide.pdf (accessed on 10 October 2016).
- Wilson, A.E.; Sarnelle, O.; Tillmanns, A.R. Effects of cyanobacterial toxicity and morphology on the population growth of freshwater zooplankton: Meta-analyses of laboratory experiments. Limnol. Oceanogr. 2006, 51, 1915–1924. [Google Scholar] [CrossRef] [Green Version]
- Lampert, W. Laboratory studies on zooplankton-cyanobacteria interactions. N. Z. J. Mar. Freshw. Res. 1987, 21, 483–490. [Google Scholar] [CrossRef]
- Lampert, W. Zooplankton research: The contribution of limnology to general ecological paradigms. Aquat. Ecol. 1997, 31, 19–27. [Google Scholar] [CrossRef]
- Sarnelle, O. Daphnia as keystone predators: Effects on phytoplankton diversity and grazing sesistance. J. Plankton Res. 2005, 27, 1229–1238. [Google Scholar] [CrossRef]
- Wogram, J.; Liess, M. Rank ordering of macroinvertebrate species sensitivity to toxic compounds by comparison with that of Daphnia Magna. Bull. Environ. Contam. Toxicol. 2001, 67, 360–367. [Google Scholar]
- Wetzel, R. Limnology, 3rd ed.; Academic Press: Cambridge, MA, USA, 2001; p. 1006. [Google Scholar]
- Pedersen, F.; Hansen, P. Effects of high pH on the growth and survival of six marine heterotrophic protists. Mar. Ecol. Prog. Ser. 2003, 260, 33–41. [Google Scholar] [CrossRef] [Green Version]
- Yin, X.W.; Niu, C.J. Effect of pH on Survival, Reproduction, Egg Viability and Growth Rate of Five Closely Related Rotifer Species. Aquat. Ecol. 2007, 42, 607–616. [Google Scholar] [CrossRef]
- Beklioglu, M.; Moss, B. The Impact of pH on interactions among phytoplankton algae, zooplankton and perch (Perca Fluviatilis) in a Shallow, Fertile Lake. Freshw. Biol. 1995, 33, 497–509. [Google Scholar] [CrossRef]
- Reynolds, C. The Ecology of Phytoplankton; Cambridge University Press: Cambridge, UK; Melbourne, Australia; Madrid, Spain; Cape Town, South Africa; Singapore; São Paulo, Brazil, 2006; p. 551. [Google Scholar]
- McQueen, D.J.; Post, J.R.; Mills, E.L. Trophic relationships in freshwater pelagic ecosystems. Can. J. Fish. Aquat. Sci. 1986, 43, 1571–1581. [Google Scholar] [CrossRef]
- Christoffersen, K.; Riemann, B.; Klysner, A.; Søndergaard, M. Potential role of fish predation and natural populations of zooplankton in structuring a plankton community in eutrophic lake water. Limnol. Oceanogr. 1993, 38, 561–573. [Google Scholar] [CrossRef]
- Envolab Fine Chemicals Nadwęglan sodu. Available online: https://envolab.pl/sklep/nadweglan-sodu-12kg/ (accessed on 1 January 2016).
- Chapman, M.A.; Lewis, M.H.; Stout, V.M. Introduction to the freshwater crustacea of New Zealand; Collins: Auckland, New Zealand, 1976. [Google Scholar]
- McDonald, J.H. Regressions. In Handbook of Biological Statistics; Sparky House Publishing: Baltimore, MD, USA, 2014; Volume 2, pp. 173–181. [Google Scholar]
- Kreyling, J.; Schweiger, A.H.; Bahn, M.; Ineson, P.; Migliavacca, M.; Morel-Journel, T.; Christiansen, J.R.; Schtickzelle, N.; Larsen, K.S. To replicate, or not to replicate—that is the question: How to tackle nonlinear responses in ecological experiments. Ecol. Lett. 2018, 21, 1629–1638. [Google Scholar] [CrossRef]
- Meinertz, J.R.; Greseth, S.L.; Gaikowski, M.P.; Schmidt, L.J. Chronic toxicity of hydrogen peroxide to Daphnia magna in a continuous exposure, flow-through test system. Sci. Total Environ. 2008, 392, 225–232. [Google Scholar] [CrossRef]
- Reichwaldt, E.S.; Zheng, L.; Barrington, D.J.; Ghadouani, A. Acute toxicological response of Daphnia and Moina to hydrogen peroxide. J. Environ. Eng. 2011, 138, 607–611. [Google Scholar] [CrossRef] [Green Version]
- Wasielak, K. The Influence of Atomic Oxygen on Chemical Properties and Microbial Activity in Aquatic Ecosystem; The University of Warsaw: Warsaw, Poland, 2016. [Google Scholar]
- OECD SIDS Initial Assessment Report for SIAM 15—sodium Carbonate. Available online: https://hpvchemicals.oecd.org/ui/handler.axd?id=5A6538BE-AA30-4A72-AD1C-906D9B5413BD (accessed on 19 March 2020).
- Bogart, S.J.; Woodman, S.; Steinkey, D.; Meays, C.; Pyle, G.G. Rapid changes in water hardness and alkalinity: Calcite formation is lethal to Daphnia magna. Sci. Total Environ. 2016, 559, 182–191. [Google Scholar] [CrossRef]
- Warne, M.S.D.; Schifko, A.D. Toxicity of laundry detergent components to a freshwater cladoceran and their contribution to detergent toxicity. Ecotoxicol. Environ. Saf. 1999, 44, 196–206. [Google Scholar] [CrossRef]
- Geer, T.D.; Kinley, C.M.; Iwinski, K.J.; Calomeni, A.J.; Rodgers, J.H. Comparative toxicity of sodium carbonate peroxyhydrate to freshwater organisms. Ecotoxicol. Environ. Saf. 2016, 132, 202–211. [Google Scholar] [CrossRef]
- Pirow, R.; Wollinger, F.; Paul, R.J. The sites of respiratory gas exchange in the planktonic crustacean Daphnia magna: An in vivo study employing blood haemoglobin as an internal oxygen probe. J. Exp. Biol. 1999, 202, 3089–3099. [Google Scholar] [PubMed]
- Broönmark, C.; Hansson, L.-A. The Biology of Lakes and Ponds; Oxford University Press: Oxford, UK, 2018. [Google Scholar]
- Pirow, R.; Buchen, I. The dichotomous oxyregulatory behaviour of the planktonic crustacean Daphnia Magna. J. Exp. Biol. 2004, 207, 683–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebert, D. Ecology, Epidemiology, and Evolution of Parasitism in Daphnia; National Library of Medicine (US): Bethesda, MD, USA; National Center for Biotechnology Information: Basel, Switzerland, 2004.
- Pirow, R.; Wollinger, F.; Paul, R.J. The importance of the feeding current for oxygen uptake in the water flea Daphnia magna. J. Exp. Biol. 1999, 202, 553–562. [Google Scholar] [PubMed]
- Burns, C.W. Relation between filtering rate, temperature, and body size in four Species of Daphnia. Limnol. Oceanogr. 1969, 14, 693–700. [Google Scholar] [CrossRef]
- Egloff, D.A.; Palmer, D.S. Size relations of the filtering area of two Daphnia species. Limnol. Oceanogr. 1971, 16, 900–905. [Google Scholar] [CrossRef]
- Crafton, E.A.; Cutright, T.J.; Bishop, W.M.; Ott, D.W. Modulating the effect of iron and total organic carbon on the efficiency of a hydrogen peroxide-based algaecide for suppressing cyanobacteria. Water Air Soil Pollut. 2019, 230, 56. [Google Scholar] [CrossRef]
- Kędzierska, A. Methods of Mitigation of Cyanobacterial Blooms Basing on Various-Scale Experiments. Master’s Thesis, University of Warsaw, Warsaw, Poland, 2016. [Google Scholar]

| Treatment | Sodium Percarbonate (SP) (2Na2CO3·3H2O2) | Hydrogen Peroxide (HP) (H2O2) |
|---|---|---|
| Control | 0.0 mg·L−1 | 0.0 mg·L−1 |
| T1 | 5.0 mg·L−1 | 1.4 mg·L−1 |
| T2 | 10.0 mg·L−1 | 2.8 mg·L−1 |
| T3 | 20.0 mg·L−1 | 5.5 mg·L−1 |
| T4 | 30.0 mg·L−1 | 8.3 mg·L−1 |
| T5 | 40.0 mg·L−1 | 11.1 mg·L−1 |
| T6 | 50.0 mg·L−1 | 13.8 mg·L−1 |
| T7 | 60.0 mg·L−1 | 16.6 mg·L−1 |
| Species | Control (Mean) | T1: 5 SP | T2: 10 SP | T3: 20 SP | T4: 30 SP | T5: 40 SP | T6: 50 SP | T7: 60 SP |
|---|---|---|---|---|---|---|---|---|
| Daphnia sp. | 79.6 SD 47 | 280 | 292 | 150 | 185 | 104 | 95 | 265 |
| Species | Control (Mean) | T 1: 5 SP | T2: 10 SP | T3: 20 SP | T4: 30 SP | T5: 40 SP | T6: 50 SP | T7: 60 SP |
|---|---|---|---|---|---|---|---|---|
| Percentage living Daphnia | 97.9 SD 1.23 | 93.9 | 91.1 | 66.0 | 16.8 | 2.9 | 1.1 | 0.0 |
| Living | 77 | 263 | 266 | 99 | 31 | 3 | 1 | 0 |
| Dead | 2.6 | 17 | 26 | 51 | 154 | 101 | 95 | 265 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thoo, R.; Siuda, W.; Jasser, I. The Effects of Sodium Percarbonate Generated Free Oxygen on Daphnia—Implications for the Management of Harmful Algal Blooms. Water 2020, 12, 1304. https://doi.org/10.3390/w12051304
Thoo R, Siuda W, Jasser I. The Effects of Sodium Percarbonate Generated Free Oxygen on Daphnia—Implications for the Management of Harmful Algal Blooms. Water. 2020; 12(5):1304. https://doi.org/10.3390/w12051304
Chicago/Turabian StyleThoo, Robin, Waldemar Siuda, and Iwona Jasser. 2020. "The Effects of Sodium Percarbonate Generated Free Oxygen on Daphnia—Implications for the Management of Harmful Algal Blooms" Water 12, no. 5: 1304. https://doi.org/10.3390/w12051304
APA StyleThoo, R., Siuda, W., & Jasser, I. (2020). The Effects of Sodium Percarbonate Generated Free Oxygen on Daphnia—Implications for the Management of Harmful Algal Blooms. Water, 12(5), 1304. https://doi.org/10.3390/w12051304




