The Effect of Human Impact on the Water Quality and Biocoenoses of the Soft Water Lake with Isoetids: Lake Jeleń, NW Poland
Abstract
1. Introduction
2. Materials and Methods
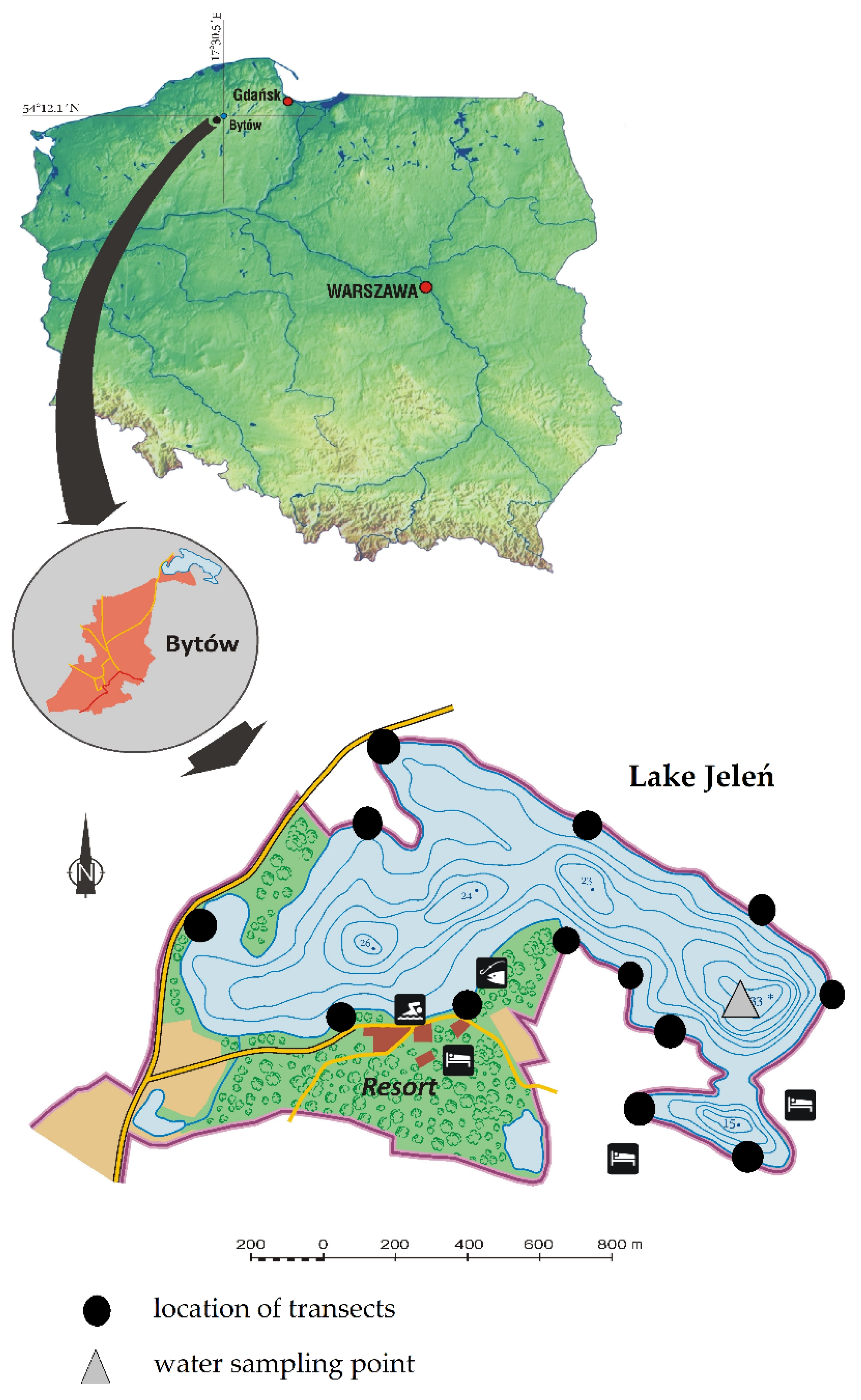
2.1. Study Site
2.2. Methods
3. Results
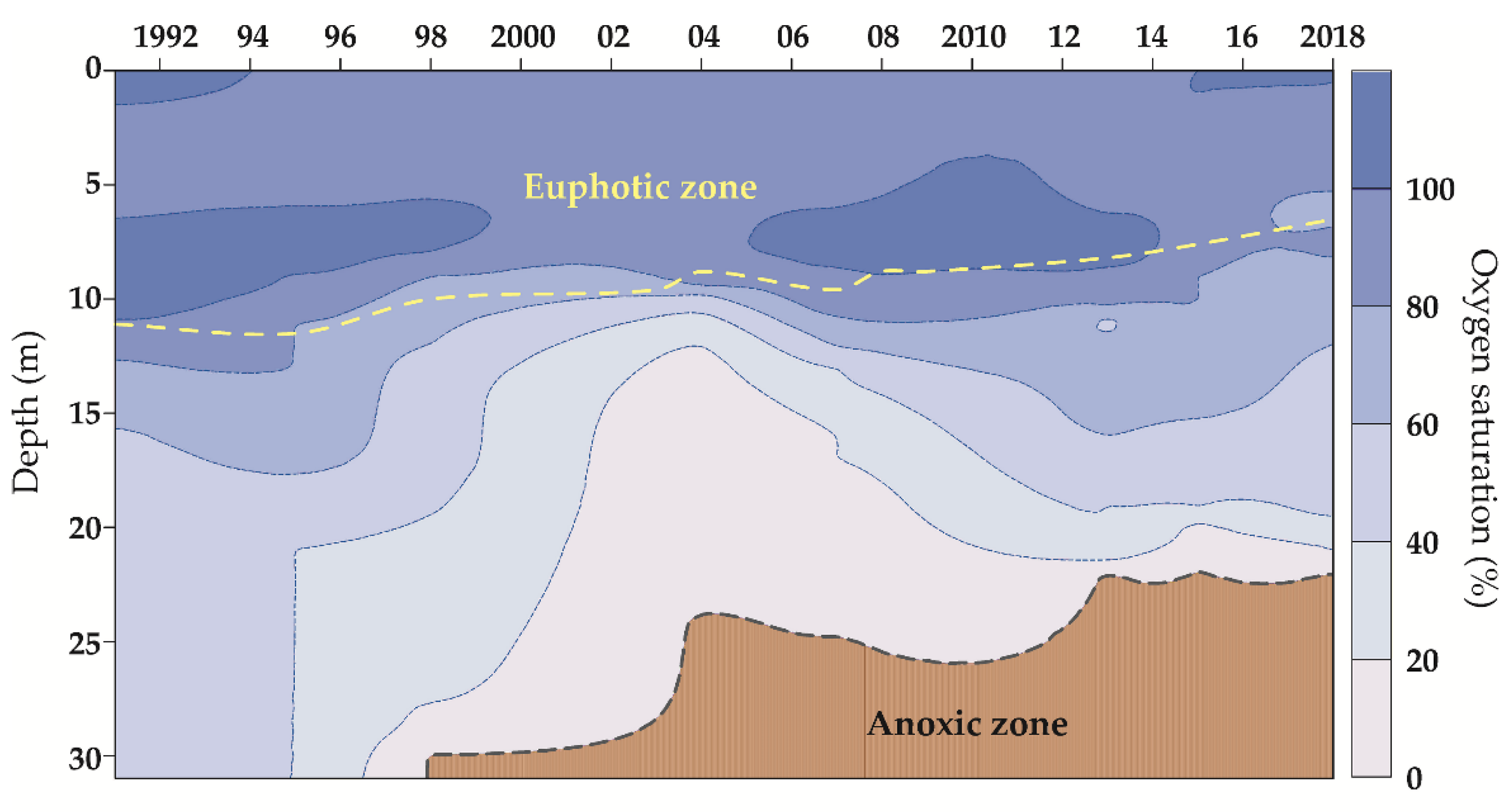
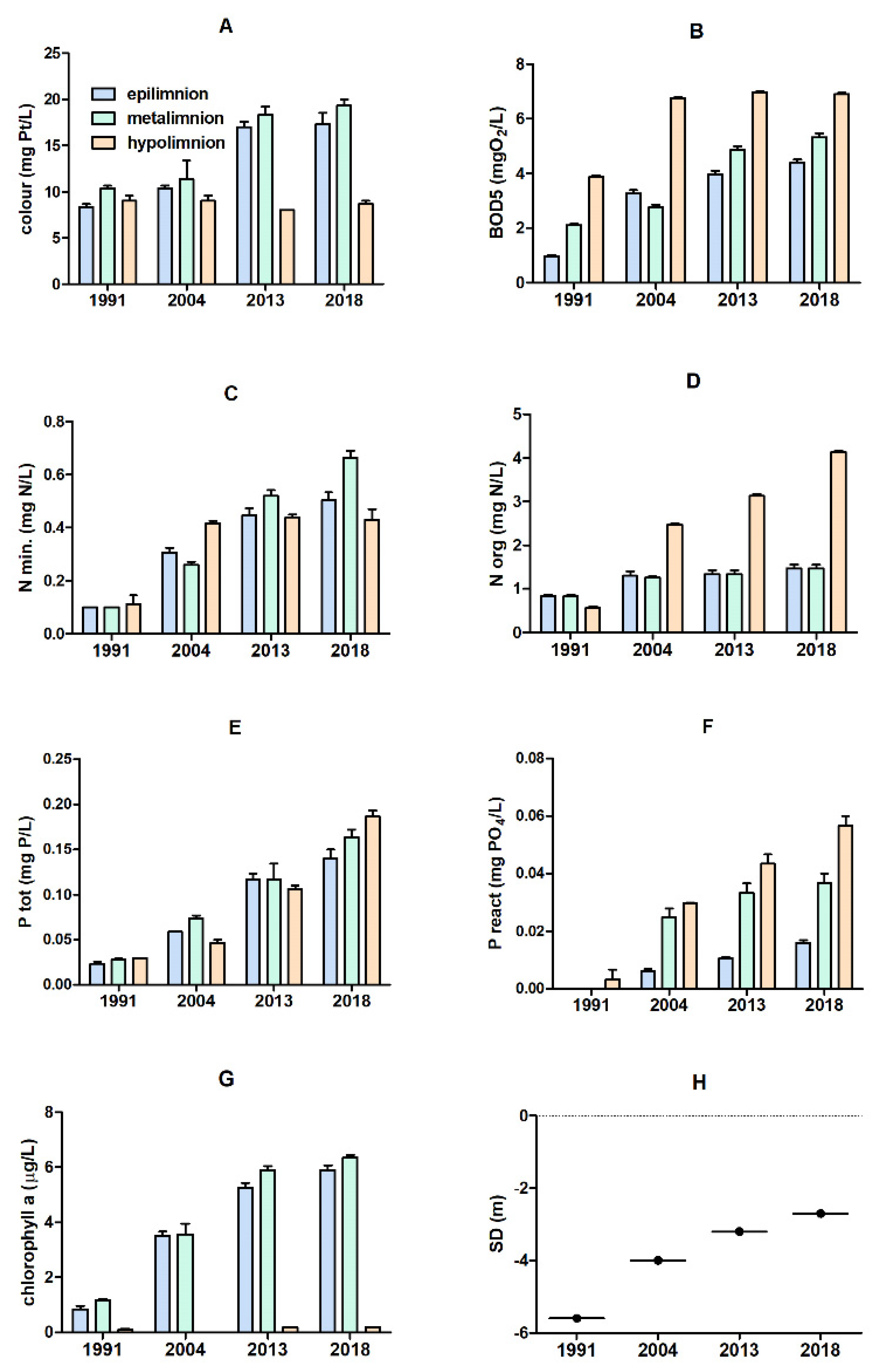
3.1. Physical and Chemical Parameters of the Lake Water
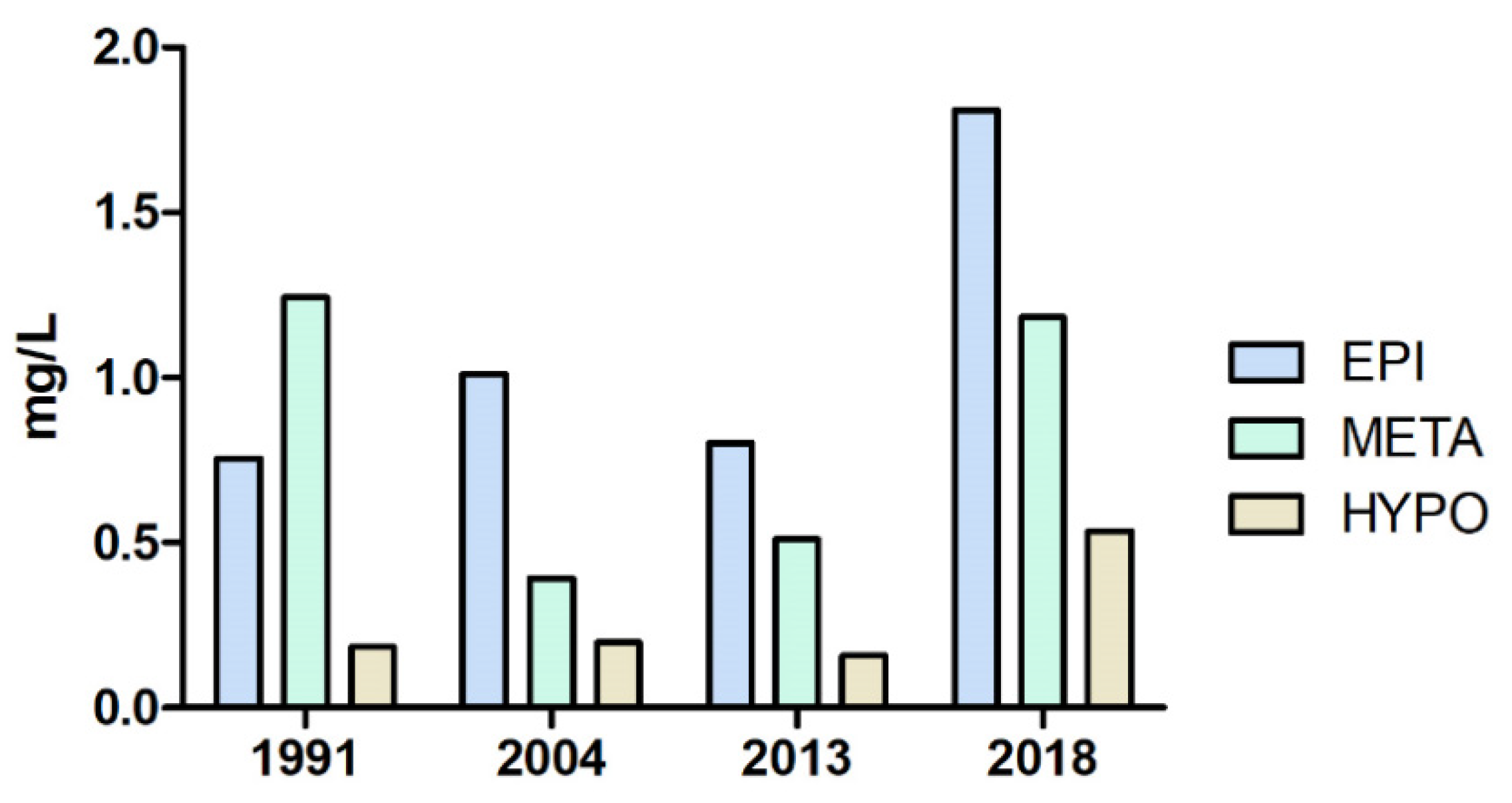
3.2. Phytoplankton
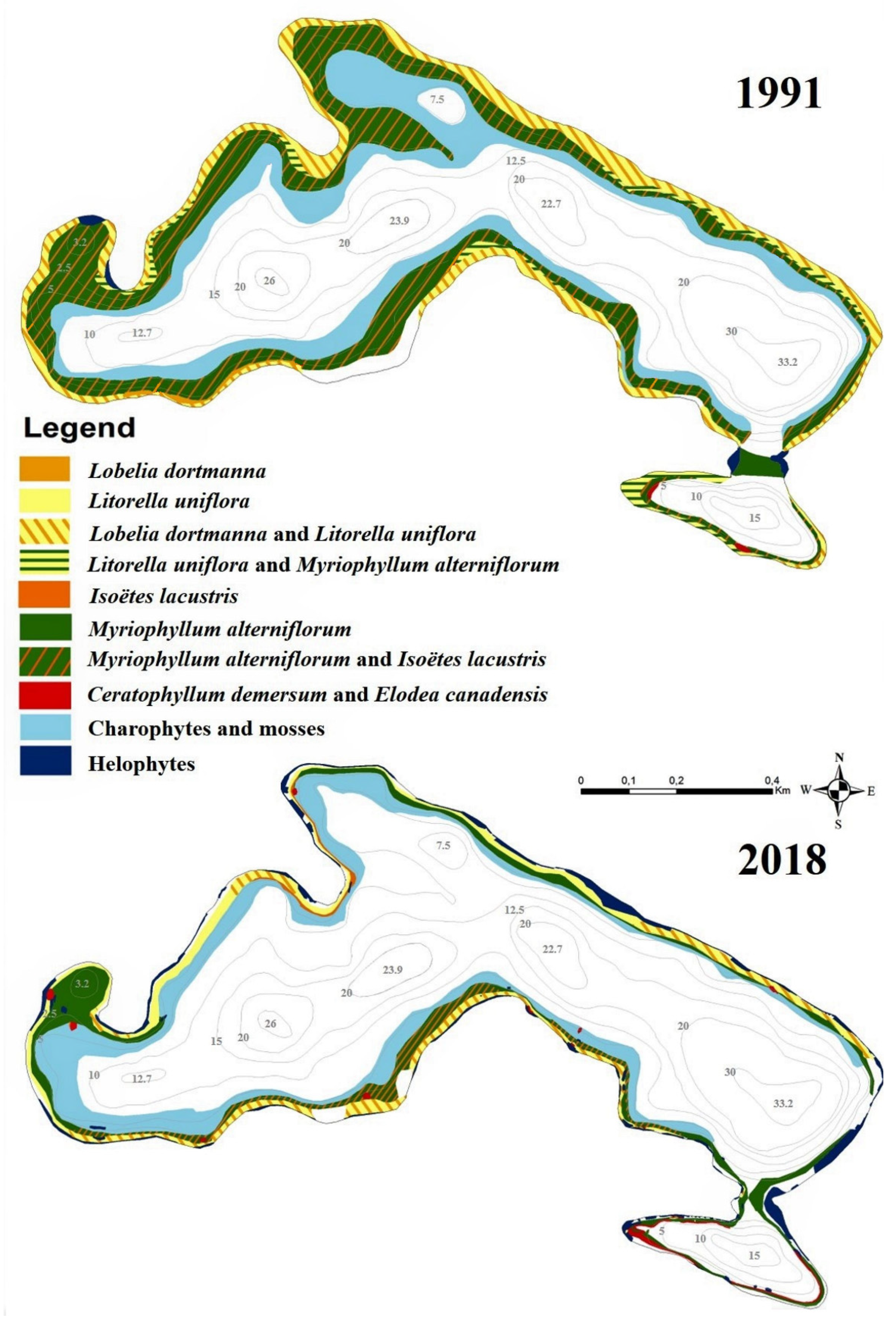
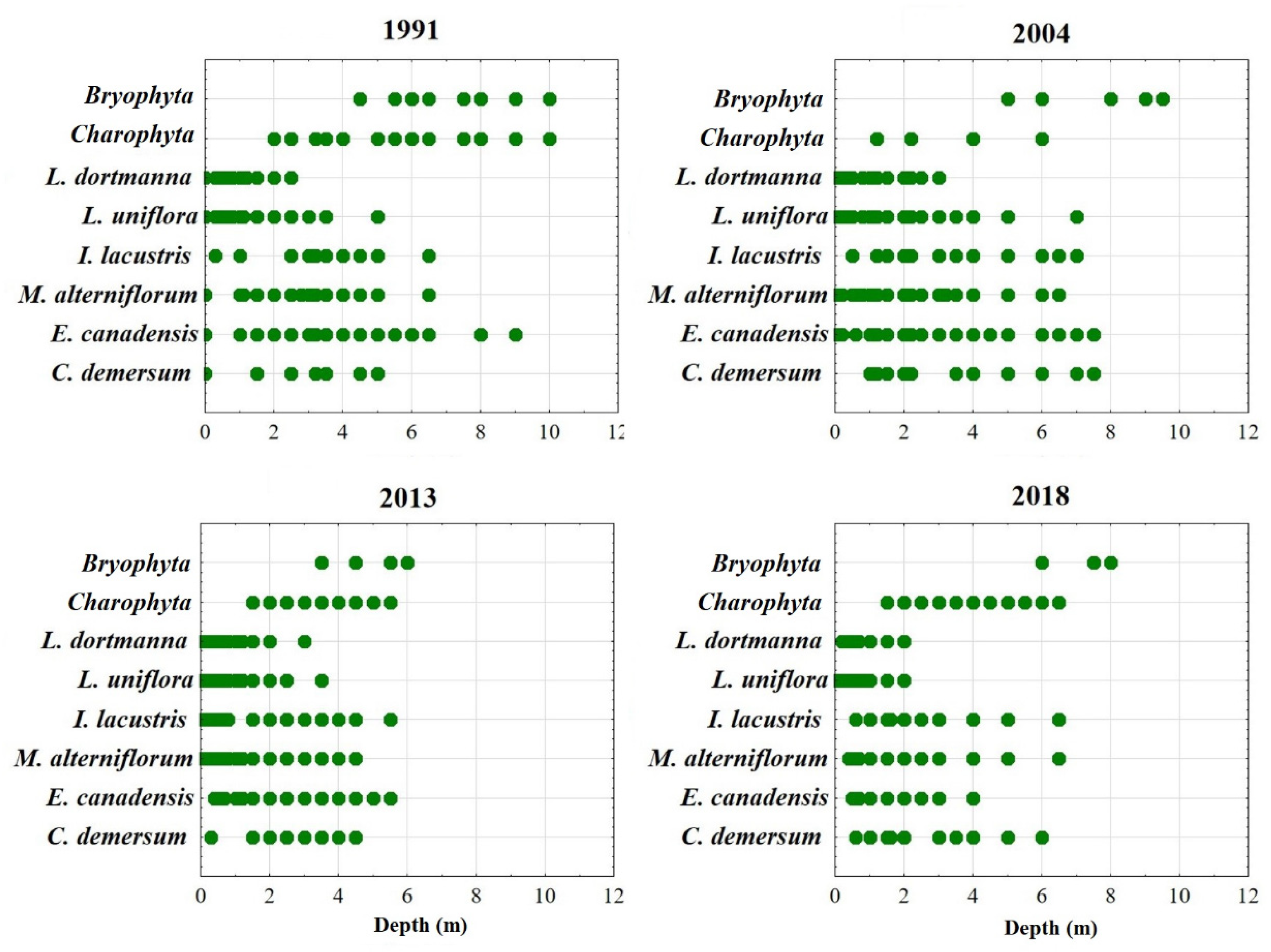
3.3. Macrophytes
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Arts, G.H.P. Deterioration of atlantic soft water macrophyte communities by acidification, eutrophication and alkalinisation. Aquat. Bot. 2002, 73, 373–393. [Google Scholar] [CrossRef]
- Smolders, A.J.P.; Lucassen, E.C.H.E.T.; Roelofs, J.G.M. The isoetid environment: Biogeochemistry and threats. Aquat. Bot. 2002, 73, 325–350. [Google Scholar] [CrossRef]
- Spierenburg, P.; Lucassen, E.C.H.E.T.; Lotter, A.F.; Roelofs, J.G.M. Competition between isoetids and invading elodeids at different concentrations of aquatic carbon dioxide. Freshw. Biol. 2010, 55, 1274–1287. [Google Scholar] [CrossRef]
- European Commission, DG Environment, Nature ENV B.3, B. European Commission, Interpretation Manual of European Union Habitats–EUR28; European Commission: Brussels, Belgium, 2013. [Google Scholar]
- Kolada, A.; Piotrowicz, R.; Wilk-Woźniak, E.; Dynowski, P.; Klimaszyk, P. Conservation status of the Natura 2000 habitat 3110 in Poland: Monitoring, classification and trends. Limnol. Rev. 2017, 17, 215–222. [Google Scholar] [CrossRef]
- Kraska, M. Jeziora lobeliowe (Lobelia lakes). In Siedliska Przyrodnicze. Poradniki Ochrony Siedlisk i Gatunków Natura 2000–Podręcznik Metodyczny. Tom 2: Wody Słodkie i Torfowiska (Natural Habitats. Guides for the Protection of Habitats and Natura 2000 Species–A Methodological Handbook; Herbich, J., Ed.; Ministerstwo Środowiska: Warsaw, Poland, 2004; Volume 2, pp. 29–36. [Google Scholar]
- Borowiak, D.; Piotrowicz, R.; Nowiński, K.; Klimaszyk, P. Soft-water lobelia lakes in Poland. In Polish River Basins and Lakes–Part I. The Handbook of Environmental Chemistry; Krzeniewska, E., Harnisz, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; pp. 89–118. [Google Scholar]
- Free, G.; Bowman, J.; McGarrigle, M.; Caroni, R.; Donnelly, K.; Tierney, D.; Trodd, W.; Little, R. The identification, characterization and conservation value of isoetid lakes in Ireland. Aquat. Conserv. Mar. Freshw. Ecosyst. 2009, 19, 264–273. [Google Scholar] [CrossRef]
- Kraska, M.; Klimaszyk, P.; Piotrowicz, R. Anthropogenic changes in properties of the water and spatial structure of the vegetation of the lobelia lake Lake Modre in the Bytów Lakeland. Oceanol. Hydrobiol. Stud. 2013, 42, 302–313. [Google Scholar] [CrossRef]
- Klimaszyk, P.; Rzymski, P. Catchment vegetation can trigger lake dystrophy through changes in runoff water quality. Ann. Limnol.-Int. J. Limnol. 2013, 49, 191–197. [Google Scholar] [CrossRef]
- Klimaszyk, P.; Piotrowicz, R.; Rzymski, P. Changes in physico-chemical conditions and macrophyte abundance in a shallow soft-water lake mediated by a Great Cormorant roosting colony. J. Limnol. 2015, 74, 114–122. [Google Scholar] [CrossRef]
- Joniak, T.; Klimaszyk, P.; Kraska, M. Diel dynamics of vertical changes of chlorophyll and bacteriochlorophyll in small humic lakes. Oceanol. Hydrobiol. Stud. 2010, 39, 103–111. [Google Scholar] [CrossRef]
- Naiman, R.J.; Decamps, H. The Ecology and Management of Aquatic-Terestrial Ecotones; UNESCO/MAB(05)/M2/v.4: Paris, French, 1990. [Google Scholar]
- Serafin, A.; Sender, J.; Bronowicka-Mielniczuk, U. Potential of shrubs, shore vegetation and macrophytes of a lake to function as a phytogeochemical barrier against biogenic substances of various origin. Water 2019, 11, 290. [Google Scholar] [CrossRef]
- Murphy, K.J. Plant communities and plant diversity in softwater lakes of northern Europe. Aquat. Bot. 2002, 73, 287–324. [Google Scholar] [CrossRef]
- Szyper, H.; Gołdyn, R.; Romanowicz, W.; Kraska, M. Możliwości ochrony oligotroficznego jeziora Jeleń przed czynnikami antropogennymi (Possibilities of protecting the oligotrophic Lake Jeleń against influence of man). In Jeziora Lobeliowe. Charakterystyka, Funkcjonowanie i Ochrona. Cz. I.; Kraska, M., Ed.; Sorus: Poznań, Poland, 1994; pp. 105–115. [Google Scholar]
- Rosińska, J.; Piotrowicz, R.; Celiński, K.; Dabert, M.; Rzymski, P.; Klimaszyk, P. The reappearance of an extremely rare and critically endangered Nitella translucens (Charophyceae) in Poland. J. Phycol. 2019, 55, 1412–1415. [Google Scholar] [CrossRef] [PubMed]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 17th ed.; American Public Health Association: Washington, DC, USA, 1991. [Google Scholar]
- Carlson, R.E. A trophic state index for lakes. Limnol. Ocean. 1977, 22, 361–369. [Google Scholar] [CrossRef]
- Borowiak, D. Właściwości Optyczne wód Jeziornych Pomorza (Optical properties of the Pomeranian Lake Waters); Wydawnictwo Uniwersytetu Gdańskiego: Gdańsk, Poland, 2011. [Google Scholar]
- Ficek, D. Właściwości Biooptyczne wód jezior Pomorza oraz ich poróWnanie z Właściwościami wód Innych Jezior i Morza Bałtyckiego (Bio-Optical Properties of Waters in Pomeranian Lakes and Their Comparison to Optical Properties of Other Lake Waters and the Baltic Sea); Rozpr. Mon.; PAN IO: Sopot, Poland, 2013. [Google Scholar]
- Wetzel, R.G.; Likens, G.E. Limnological Analyses, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Szeląg-Wasielewska, E.; Gołdyn, R.; Bernaciak, A. Fitoplankton a stan trofii wód jeziora Jeleń na Pojezierzu Bytowskim (Phytoplankton versus the trophic state of lake Jeleń in the Pomeranian Lakeland). In Badania Fizjograficzne nad Polską Zachodnią, Seria B–Botanika; PTPN: Poznań, Poland, 1999; pp. 203–223. [Google Scholar]
- Hörnström, E. Trophic characterization of lakes by means of qualitative phytoplankton analysis. Limnologica 1981, 13, 249–261. [Google Scholar]
- Braun-Blanquet, J. Pflanzensoziologie; Springer: Berlin/Heidelberg, Germany, 1964. [Google Scholar]
- Jensén, S. An objective method for sampling the macrophyte vegetation in lakes. Vegetatio 1977, 33, 107–118. [Google Scholar] [CrossRef]
- Kraska, M.; Piotrowicz, R. Roślinność wybranych jezior lobeliowych na tle warunków fizyczno-chemicznych ich wód. In Jeziora Lobeliowe. Charakterystyka, Funkcjonowanie i Ochrona. Cz. I.; Kraska, M., Ed.; Sorus: Poznań, Poland, 1994; pp. 37–83. [Google Scholar]
- Madsen, T.V.; Olesen, B.; Bagger, J. Carbon acquisition and carbon dynamics by aquatic isoetids. Aquat. Bot. 2002, 73, 351–372. [Google Scholar] [CrossRef]
- Brouwer, E.; Bobbink, R.; Roelofs, J.G.M. Restoration of aquatic macrophyte vegetation in acidified and eutrophied softwater lakes: An overview. Aquat. Bot. 2002, 73, 405–431. [Google Scholar] [CrossRef]
- Szmeja, J. Evolution and conservation of lobelia lakes in Poland. Fragm. Flor. Geobot. 1997, 40, 89–94. [Google Scholar]
- Kraska, M.; Piotrowicz, R. Lobelia lakes: Specificity, trophy, vegetation and protection. In Protection of Lakes and Wetlands of Pomerania Region; Malinowski, B., Ed.; Towarzystwo Ekologiczno-Kulturalne: Bobolice, Poland, 2000; pp. 48–52. [Google Scholar]
- Doms, F.A. Ein neuer Standort von Isoetes lacustris (L.) Dur. (A new location for Isoetes lacustris (L.) Dur.). Werh. Bot. Ver. Pr. Brand. 1862, 3–4, 387–388. [Google Scholar]
- Szmal, Z. Badania hydrochemiczne jezior lobeliowych Pojezierza Zachodniego. PTPN 1959, XIX, 106. [Google Scholar]
- Roelofs, J.G.M. Soft-water macrophytes and ecosystems: Why are they so vulnerable to environmental changes? Introduction. Aquat. Bot. 2002, 73, 285–286. [Google Scholar] [CrossRef]
- Piotrowicz, R.; Kraska, M.; Klimaszyk, P.; Szyper, H.; Joniak, T. Vegetation richness and nutrient loads in 16 lakes of Drawieński National Park (northern Poland). Polish J. Environ. Stud. 2006, 15, 467–478. [Google Scholar]
- Szeląg-Wasielewska, E.; Gołdyn, R. Zbiorowiska glonów w pelagialu jezior lobeliowych (Algal communities in the pelagial zone of lobelian lakes. In Jeziora Lobeliowe. Charakterystyka, Funkcjonowanie i ochrona. Cz. I.; Kraska, M., Ed.; Sorus: Poznań, Poland, 1994; pp. 37–65. [Google Scholar]
- Szeląg-Wasielewska, E. Vertical distribution of phototrophs in the pelagic zone of three small Lobelia lakes. Oceanol. Hydrobiol. Stud. 2010, 39, 121–133. [Google Scholar] [CrossRef]
- Oleksowicz, A. Phytoplankton communities of Lobelia–type lakes in the Kashubian Lake District (Pomerania, northern Poland). Acta Hydrobiol. 1989, 31, 259–271. [Google Scholar]
- Luścińska, M.; Oleksowicz, A.S. The structure and production of algal communities in the lakes of the Bory Tucholskie region. In Some Ecological Processes of the Biological Systems in North Poland; Bohr, R., Nienartowicz, A., Witkoń-Michalska, J., Eds.; N. Copernicus University Press: Toruń, Poland, 1992; pp. 283–297. [Google Scholar]
- Szeląg-Wasielewska, E. Phytoplankton community structure in non-stratified lakes of Pomerania (NW Poland). Hydrobiologia 2003, 506–509, 229–236. [Google Scholar] [CrossRef]
- Szeląg-Wasielewska, E. Phytoplankton structure of two small lakes–changes after a decade. Oceanol. Hydrobiol. Stud. 2007, 36, 113–120. [Google Scholar]
- Pełechata, A. Zbiorowiska fitoplanktonu jezior lobeliowych Pojezierza Pomorskiego (Phytoplankton communities of lobelia lakes of the Pomeranian Lake District). In Jeziora Lobeliowe w Drugiej Dekadzie XXI Wieku; Bociąg, K., Borowiak, D., Eds.; Fundacja Rozwoju Uniwersytetu Gdańskiego: Gdańsk, Poland, 2016; pp. 80–96. [Google Scholar]
- Szeląg-Wasielewska, E. Phytoplankton structure in lakes with a low trophic level in north-west Poland. In Physicochemical Problems of Natural Waters Ecology; Gurgul, H., Ed.; Wydawnictwo Naukowe Uniwersytetu Szczecińskiego: Szczecin, Poland, 2005; pp. 281–296. [Google Scholar]
- Heinonen, P. Quantity and composition of phytoplankton in Finnish inland waters [eutrophication, water quality, diversity, indicator species, odour index]. Water Res. Inst. 1980, 37, 1–91. [Google Scholar]
- Komárek, J.; Komárkova, J. Taxonomic review of the cyanoprokaryotic genera Planktothrix and Planktothricoides. Czech. Phycol. 2004, 4, 1–18. [Google Scholar]
- Welker, M.; Christiansen, G.; von Döhren, H. Diversity of coexisting Planktothrix (Cyanobacteria) chemotypes deduced by mass spectral analysis of microystins and other oligopeptides. Arch. Microbiol. 2004, 182, 288–298. [Google Scholar] [CrossRef]
- Rosińska, J.; Gołdyn, R. Response of vegetation to growing recreational pressure in the shallow Raczyńskie Lake. Knowl. Manag. Aquat. Ecosyst 2018, 419, 1. [Google Scholar] [CrossRef]
- O’Hare, M.T.; Aguiar, F.C.; Asaeda, T.; Bakker, E.S.; Chambers, P.A.; Clayton, J.S.; Elger, A.; Ferreira, T.M.; Gross, E.M.; Gunn, I.D.M.; et al. Plants in aquatic ecosystems: Current trends and future directions. Hydrobiologia 2018, 812, 1–11. [Google Scholar] [CrossRef]
- Klimaszyk, P.; Kraska, M.; Piotrowicz, R. Human impact on the functioning lobelia lake: Lake Jeleń (Bytów Lakeland). In Diagnosing the State of the Environment, Methods and Prognoses; Garbacz, J.K., Ed.; Bydgoskie Towarzystwo Naukowe: Bydgoszcz, Poland, 2014; pp. 131–138. [Google Scholar]
- Chappuis, E.; Lumbreras, A.; Ballesteros, E.; Gacia, E. Deleterious interaction of light impairment and organic matter enrichment on Isoetes lacustris (Lycopodiophyta, Isoetales). Hydrobiologia 2015, 760, 145–158. [Google Scholar] [CrossRef][Green Version]
- Ribaudo, C.; Tison-Rosebery, J.; Buquet, D.; Gwilherm, J.; Jamoneau, A.; Abril, G.; Anschutz, P.; Bertrin, V. Invasive aquatic plants as ecosystem engineers in an oligo-mesotrophic shallow lake. Front. Plant. Sci. 2018, 9, 1781. [Google Scholar] [CrossRef] [PubMed]
- Sand-Jensen, K.; Claus, L.; Borum, J. High resistance of oligotrophic isoetid to oxic and anoxic dark exposure. Freshwat. Biol. 2015, 60, 1044–1051. [Google Scholar] [CrossRef]
- Srivastava, J.; Gupta, A.; Chandra, H. Managing water quality with aquatic macrophytes. Rev. Environ. Sci. Biotechnol. 2008, 7, 255. [Google Scholar] [CrossRef]
- Kuczyńska-Kippen, N.; Klimaszyk, P. Diel microdistribution of physical and chemical parameters within the dense Chara bed and their impact on zooplankton. Biologia 2007, 4, 432–437. [Google Scholar] [CrossRef]
- Borowiak, D.; Bociąg, K.; Nowiński, K.; Borowiak, M. Light requirements of water lobelia (Lobelia dortmanna L.). Limnol. Rev. 2017, 17, 171–182. [Google Scholar] [CrossRef]
- Brouwer, E.; Roelofs, J.G.M. Degraded softwater lakes: Possibilities for restoration. Restor. Ecol. 2001, 9, 155–166. [Google Scholar] [CrossRef]
- Kłosowski, S.; Szańkowski, M. Habitat differentiation of the Myriophyllum alterniflorum and Littorella uniflora phytocoenoses in Poland. Acta Soc. Bot. Pol. 2011, 73, 79–86. [Google Scholar] [CrossRef][Green Version]
- Dierssen, K. Littorelletea uniflorae. In Prodromus der Europäischen Pflanzengesellschaften; Tuxen, R., Ed.; Vaduz, Liechtenstein, 1975. [Google Scholar]
- Roelofs, J.G.M. Restoration of eutrophied shallow softwater lakes based upon carbon and phosphorus limitation. Neth. J. Aquat. Ecol. 1996, 30, 197–202. [Google Scholar] [CrossRef]
- Pulido, C.; Keijsers, D.J.H.; Lucassen, E.C.H.E.T.; Pedersen, O.; Roelofs, J.G.M. Elevated alkalinity and sulfate adversely affect the aquatic macrophyte Lobelia dortmanna. Aquat. Ecol. 2012, 46, 283–295. [Google Scholar] [CrossRef][Green Version]
- Kochanowski, J.; Tobolski, K. A new locality of lobelia in Lake Krzywce Wielkie: Bory Tucholskie National Park. Stud. Lim. Tel. 2010, 4, 61–64. [Google Scholar]
- Riera, J.L.; Ballesteros, E.; Pulido, C.; Chappuis, E.; Gacia, E. Recovery of submersed vegetation in a high mountain oligotrophic soft-water lake over two decades after impoundment. Hydrobiologia 2017, 794, 139–151. [Google Scholar] [CrossRef]
- Zalewski, M.; Robarts, R. Ecohydrology–a new paradigm for integrated water resources management. SILnews 2003, 40, 1–5. [Google Scholar]
- Romo, S.; Van Donk, E.; Gylstra, R.; Gulati, R. A multivariate analysis of phytoplankton and foodweb changes in a shallow biomanipulated lake. Freshw. Biol. 1996, 36, 683–696. [Google Scholar] [CrossRef][Green Version]
- Rosińska, J.; Romanowicz-Brzozowska, W.; Kozak, A.; Gołdyn, R. Zooplankton changes during bottom-up and top-down control due to sustainable restoration in a shallow urban lake. Environ. Sci. Pollut. Res. 2019, 26, 19575–19587. [Google Scholar] [CrossRef]
- Larssen, T.; Cosby, B.J.; Lund, E.; Wright, R.F. Modeling future acidification and fish populations in Norwegian surface waters. Environ. Sci. Technol. 2010, 44, 5345–5351. [Google Scholar] [CrossRef]
- Lucassen, C.H.E.T.; Smolders, A.J.P.; Roelofs, J.G.M. Liming induces changes in the macrophyte vegetation of Norwegian softwater lakes by mitigating carbon limitation: Results from a field experiment. App. Veget. Sci. 2012, 15, 166–174. [Google Scholar] [CrossRef]
- Lindmark, G.K. Acidified lakes sediment treatment with sodium carbonate, a remedy? Hydrobiologia 1987, 92, 537–547. [Google Scholar]
- Sand-Jensen, K.; Borum, J.; Binzer, T. Oxygen stress and reduced growth of Lobelia dortmanna in sandy lake sediments subject to organic enrichment. Freshw. Biol. 2005, 9, 1–11. [Google Scholar] [CrossRef]
- Bellemakers, M.J.S.; Maessen, M.; Verheggen, G.M.; Roelofs, J.G.M. Effect of liming on shallow acidified moorland pools: A culture and seed bank experiment. Aquat Bot. 1996, 54, 37–50. [Google Scholar] [CrossRef]
- Rybak, M.; Kołodziejczyk, A.; Joniak, T.; Ratajczak, I.; Gąbka, M. Bioaccumulation and toxicity studies of macroalgae (Charophyceae) treated with aluminium: Experimental studies in the context of lake restoration. Ecotoxicol. Environ. Saf. 2017, 145, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Rybak, M.; Gąbka, M.; Ratajczak, I.; Woźniak, M.; Sobczyński, T.; Joniak, T. In-situ behavioural response and ecological stoichiometry adjustment of macroalgae (Characeae, Charophyceae) to iron overload: Implications for lake restoration. Water Res. 2020, 173, 115602. [Google Scholar] [CrossRef] [PubMed]

| Parameter | 1991 | 2004 | 2013 | 2018 | ANOVA | |
|---|---|---|---|---|---|---|
| color | mgPt/L | 9.2 ± 1.09 | 10.2 ± 2.1 | 13 ± 3.16 | 15.1 ± 5.1 | ** |
| pH | 6.79 ± 0.527 | 7.02 ± 0.31 | 7.18 ± 0.58 | 7.7 ± 0.93 | * | |
| BOD | mgO2/L | 2.3 ± 1.27 | 4.3 ± 1.89 | 4.8 ± 1.43 | 5.54 ± 1.1 | *** |
| N-NH4 | mgN/L | 0.08 ± 0.03 | 0.2 ± 0.03 | 0.23 ± 0.043 | 0.3 ± 0.074 | *** |
| N-NO3 | mgN/L | 0.02 ± 0.044 | 0.13 ± 0.08 | 0.2 ± 0.034 | 0.24 ± 0.066 | *** |
| N-NO2 | mgN/L | 0 | 0.001± 0.0002 | 0 | 0.001± 0.0004 | |
| N min. | mgN/L | 0.1 ± 0.03 | 0.33 ± 0.07 | 0.42 ± 0.058 | 0.53 ± 0.11 | *** |
| N org. | mgN/L | 0.74 ± 0.14 | 1.68 ± 0.6 | 1.76 ± 0.6 | 2.36 ± 1.33 | ** |
| P tot. | mgP/L | 0.027 ± 0.004 | 0.06 ± 0.012 | 0.088 ± 0.011 | 0.159 ± 0.02 | *** |
| PO4 | mgPO4/L | 0.001 ± 0.003 | 0.02 ± 0.011 | 0.027 ± 0.013 | 0.037 ± 0.01 | *** |
| Ca | mgCa/L | 7.02 ± 0.3 | 8.9 ± 0.9 | 7.2 ± 0.7 | 7.7 ± 0.9 | |
| EC | µSm/cm | 71.9 ± 13.2 | 79.4 ± 10.4 | 77.9 ± 9.98 | 85.4 ± 6.2 | |
| Chl | µg/L | 0.7 ± 0.49 | 2.36 ± 1.8 | 3.46 ± 2.5 | 4.15 ± 2.9 | * |
| TSI | 111 | 146 | 155 | 169 | ||
| No. | Taxon | Biomass (mg/L) |
|---|---|---|
| 1991 | ||
| 1 | Gymnodinium uberrimum (G.J.Allman) Kofoid & Swezy | 0.898 (m) |
| 2 | Gymnodinium oligoplacatum Skuja | 0.138 (e) |
| 3 | Coenococcus planktonicus Korshikov | 0.104 (e) |
| 2004 | ||
| 1 | Planktothrix agardhii (Gomont) Anagnostidis & Komárek | 0.707 (e) |
| 2 | Gymnodinium uberrimum (G.J.Allman) Kofoid & Swezy | 0.090 (e) |
| 3 | Eudorina elegans Ehrenberg | 0.053 (m) |
| 2013 | ||
| 1 | Gymnodinium sp. | 0.212 (e) |
| 2 | Ceratium hirundinella (O.F. Müller) Dujardin | 0.167 (m) |
| 3 | Peridinium inconspicuum Lemmermann | 0.139 (e) |
| 2018 | ||
| 1 | Dolichospermum spiroides (Klebhan) Wacklin, Hoffmann & Komárek | 1.312 (e) |
| 2 | Planktothrix agardhii (Gomont) Anagnostidis & Komárek | 0.157 (h) |
| 3 | Dolichospermum affine (Lemmermann) Wacklin, Hoffmann & Komárek | 0.116 (e) |
| Taxon/Year | 1991 | 2004 | 2013 | 2018 |
|---|---|---|---|---|
| Cyanobacteria | 6 | 13 | 9 | 13 |
| Euglenophyceae | 3 | 1 | 0 | 1 |
| Cryptophyceae | 6 | 4 | 5 | 5 |
| Dinophyceae | 5 | 4 | 4 | 4 |
| Chrysophyceae | 5 | 3 | 4 | 3 |
| Bacillariophyceae | 2 | 5 | 2 | 4 |
| Xanthophyceae | 1 | 1 | 0 | 0 |
| Chlorophyceae | 13 | 13 | 17 | 13 |
| Conjugatophyceae | 1 | 8 | 7 | 8 |
| Total | 42 | 52 | 48 | 51 |
| Year | Epilimnion | Metalimnion | Hypolimnion | Average Trophic State Index |
|---|---|---|---|---|
| 1991 | 1.32 | 1.17 | 1.75 | 1.41 |
| 2004 | 2.10 | 2.16 | 1.84 | 2.03 |
| 2013 | 1.57 | 2.11 | 2.17 | 1.95 |
| 2018 | 2.62 | 2.45 | 2.53 | 2.58 |
| Name of Species | 1991 | 2004 | 2013 | 2018 |
|---|---|---|---|---|
| Emergent plants (amphiphytes and helophytes) | ||||
| Acorus calamus L. | − | + | + | + |
| Alisma plantago-aquatica L. | + | + | + | + |
| Calla palustris L. | − | + | − | − |
| Carex acutiformis L. | − | + | + | + |
| Carex paniculata L. | − | − | + | + |
| Carex rostrata Stokes | + | + | + | − |
| Eleocharis palustris (L.) R. et Schr. | + | + | + | + |
| Epilobium palustre L. | + | + | − | − |
| Equisetum limosum L. | + | + | − | + |
| Glyceria plicata Fries | + | + | + | + |
| Hydrocotyle vulgaris L. | + | + | + | + |
| Iris pseudoacorus L. | + | + | − | + |
| Juncus bulbosus L. | − | − | + | − |
| Juncus effusus L. | − | + | + | + |
| Lycopus europaeus L. | + | + | − | + |
| Lysimachia thyrsiflora L. | + | + | − | − |
| Lysimachia vulgaris L. | − | + | + | + |
| Lythrum salicaria L. | + | + | + | + |
| Mentha aquatica L. | + | + | + | − |
| Mentha verticillata L. | + | + | − | − |
| Myosostis palustris (L.) Nathorst | + | + | + | + |
| Phalaris arundinacea L. | − | + | + | + |
| Phragmites australis (Cav.) Trin. ex Steud | + | + | + | + |
| Scutellaria galericulata L. | + | + | − | + |
| Sparganium erectum Huds. | − | + | + | + |
| Sparganium emersum Huds. | + | + | + | − |
| Typha angustifolia L. | − | + | + | + |
| Floating-leaved macrophytes (pleustophytes and nymphaeids) | ||||
| Lemna minor L. | + | + | + | + |
| Luronium natans (L.) Raf. | + | + | + | − |
| Polygonum amphibium L. | + | + | + | + |
| Sparganium angustifolium Michx | − | + | + | + |
| Submerged macrophytes (elodeids) | ||||
| Ceratophyllum demersum L. | + | + | + | + |
| Elodea canadensis Rich. | + | + | + | + |
| Myriophyllum alterniflorum DC. | + | + | + | + |
| Potamogeton obtusifolius Mert.et Koch. | + | + | − | − |
| Potamogeton crispus L. | − | − | + | − |
| Stuckenia pectinata (L.) Börner | − | − | + | + |
| Isoetids | ||||
| Isoëtes lacustris L. | + | + | + | + |
| Littorella uniflora (L.) Aschers. | + | + | + | + |
| Lobelia dortmanna L. | + | + | + | + |
| “Underwater meadows” (Charophytes and Bryophytes) | ||||
| Chara delicatula Agardh | + | + | + | − |
| Nitella flexilis (L.) Agardh | + | + | + | − |
| Nitella translucens (Persoon) Agardh | − | − | + | + |
| Nitellopsis obtusa (Desvaux) J. Groves | + | − | − | + |
| Drepanocladus sendtneri (Schimp. ex H.Müll.) Warnst. | + | + | + | + |
| Fontinalis antipyretica Hedw. | + | − | + | − |
| Total number of species | 32 | 39 | 35 | 32 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klimaszyk, P.; Borowiak, D.; Piotrowicz, R.; Rosińska, J.; Szeląg-Wasielewska, E.; Kraska, M. The Effect of Human Impact on the Water Quality and Biocoenoses of the Soft Water Lake with Isoetids: Lake Jeleń, NW Poland. Water 2020, 12, 945. https://doi.org/10.3390/w12040945
Klimaszyk P, Borowiak D, Piotrowicz R, Rosińska J, Szeląg-Wasielewska E, Kraska M. The Effect of Human Impact on the Water Quality and Biocoenoses of the Soft Water Lake with Isoetids: Lake Jeleń, NW Poland. Water. 2020; 12(4):945. https://doi.org/10.3390/w12040945
Chicago/Turabian StyleKlimaszyk, Piotr, Dariusz Borowiak, Ryszard Piotrowicz, Joanna Rosińska, Elżbieta Szeląg-Wasielewska, and Marek Kraska. 2020. "The Effect of Human Impact on the Water Quality and Biocoenoses of the Soft Water Lake with Isoetids: Lake Jeleń, NW Poland" Water 12, no. 4: 945. https://doi.org/10.3390/w12040945
APA StyleKlimaszyk, P., Borowiak, D., Piotrowicz, R., Rosińska, J., Szeląg-Wasielewska, E., & Kraska, M. (2020). The Effect of Human Impact on the Water Quality and Biocoenoses of the Soft Water Lake with Isoetids: Lake Jeleń, NW Poland. Water, 12(4), 945. https://doi.org/10.3390/w12040945






