Long-Term Consequences of Water Pumping on the Ecosystem Functioning of Lake Sekšu, Latvia
Abstract
1. Introduction
2. Materials and Methods
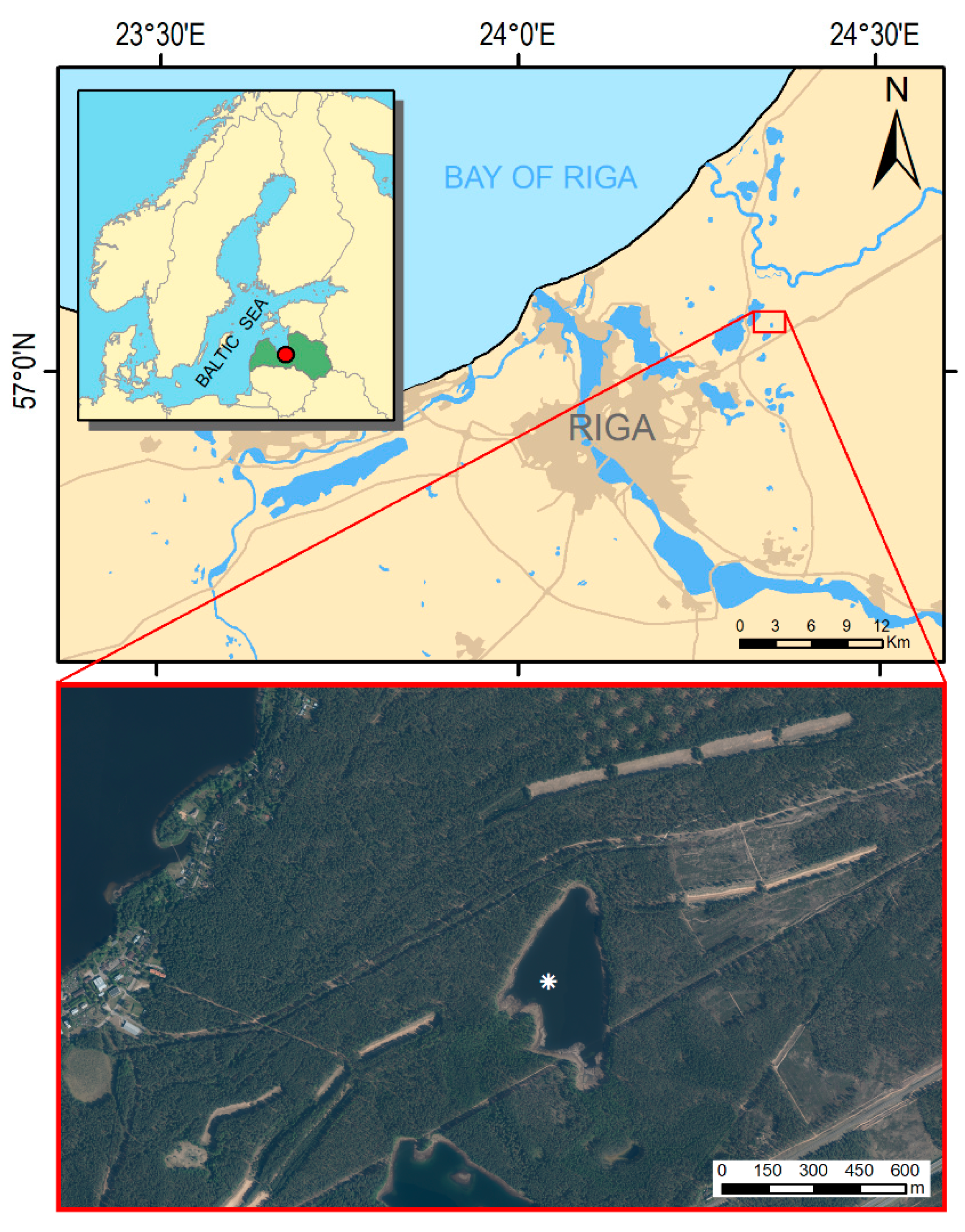
2.1. Study Site
2.2. Materials and Methods
2.2.1. Lake Sediment Coring
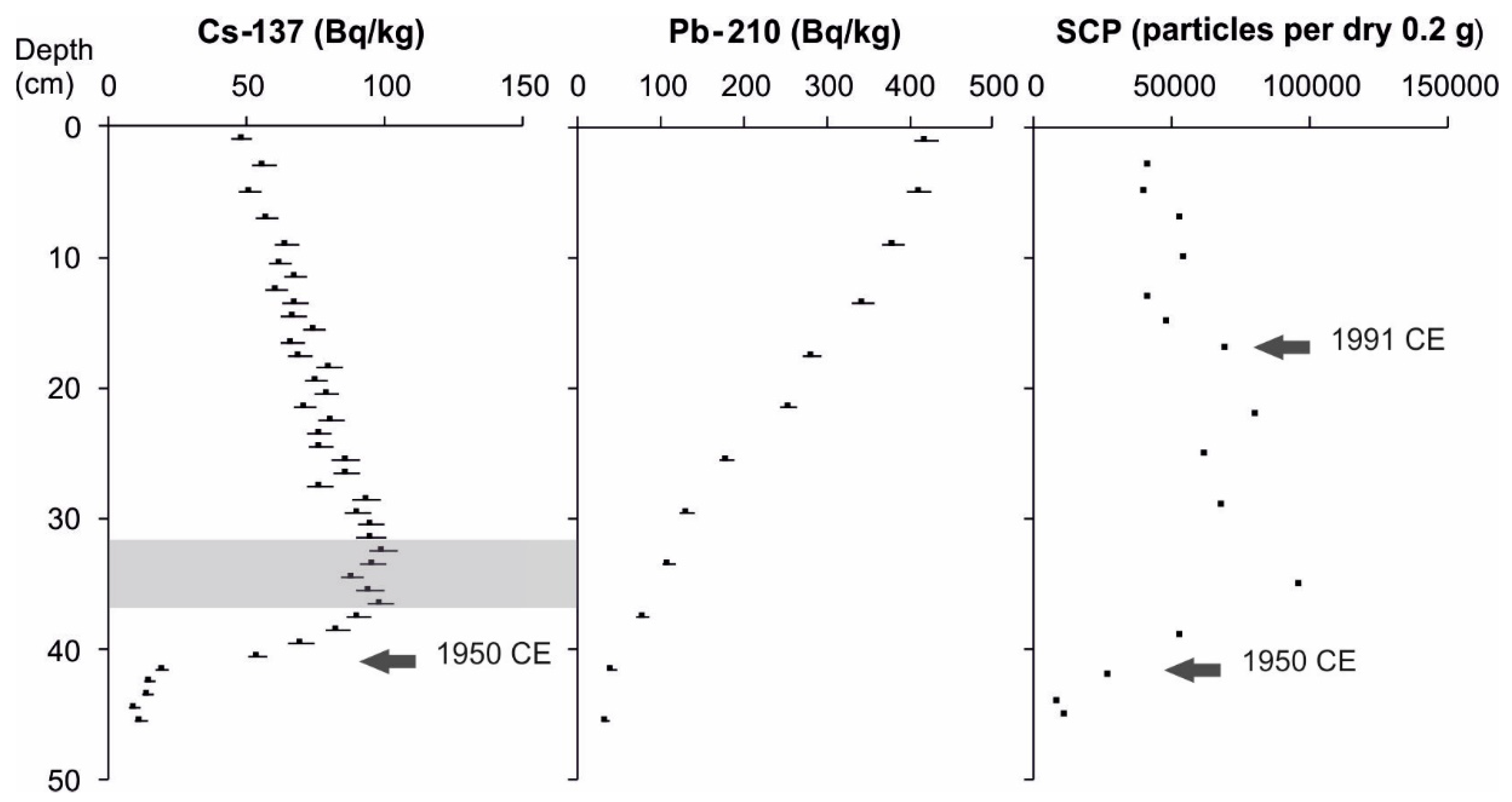
2.2.2. Core Chronology
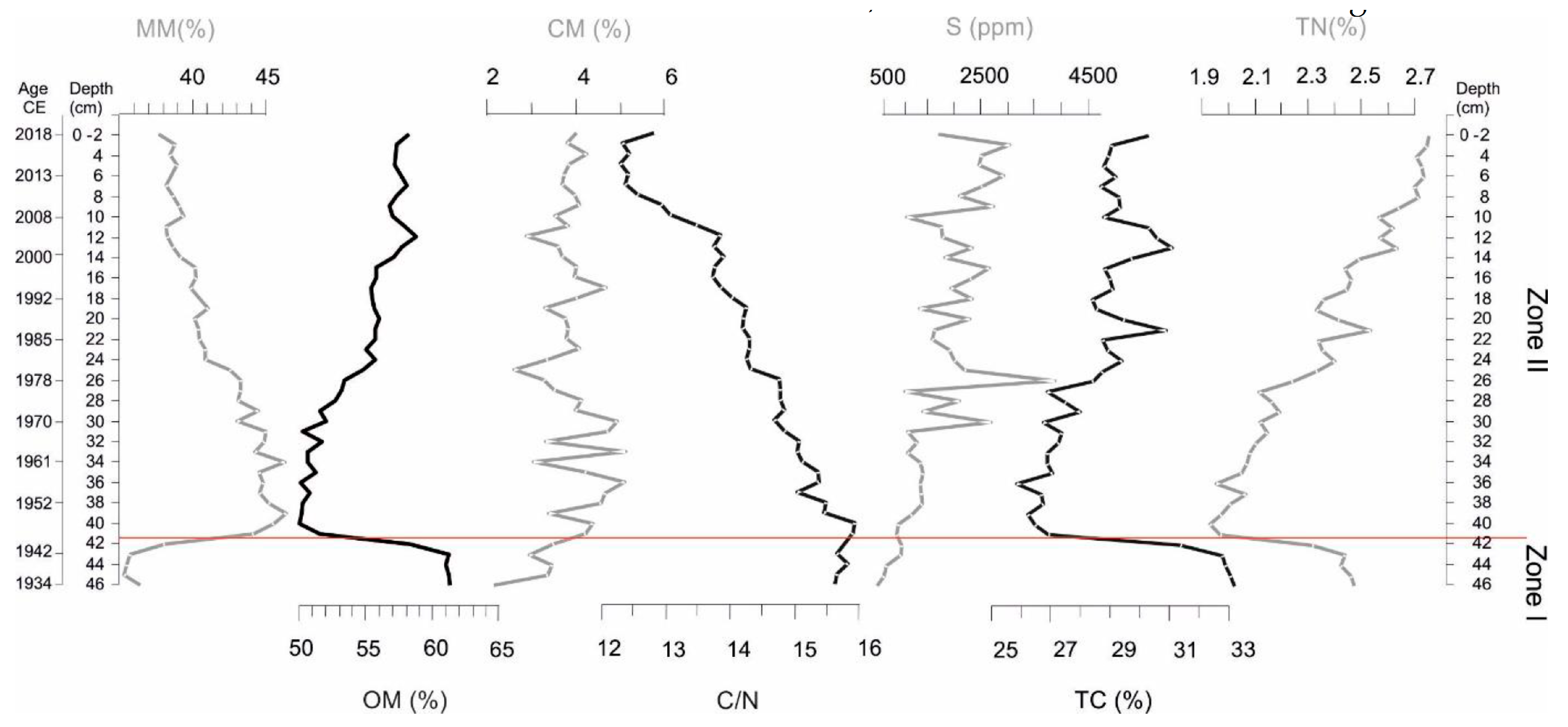
2.2.3. Physical and Chemical Sediment Analyses
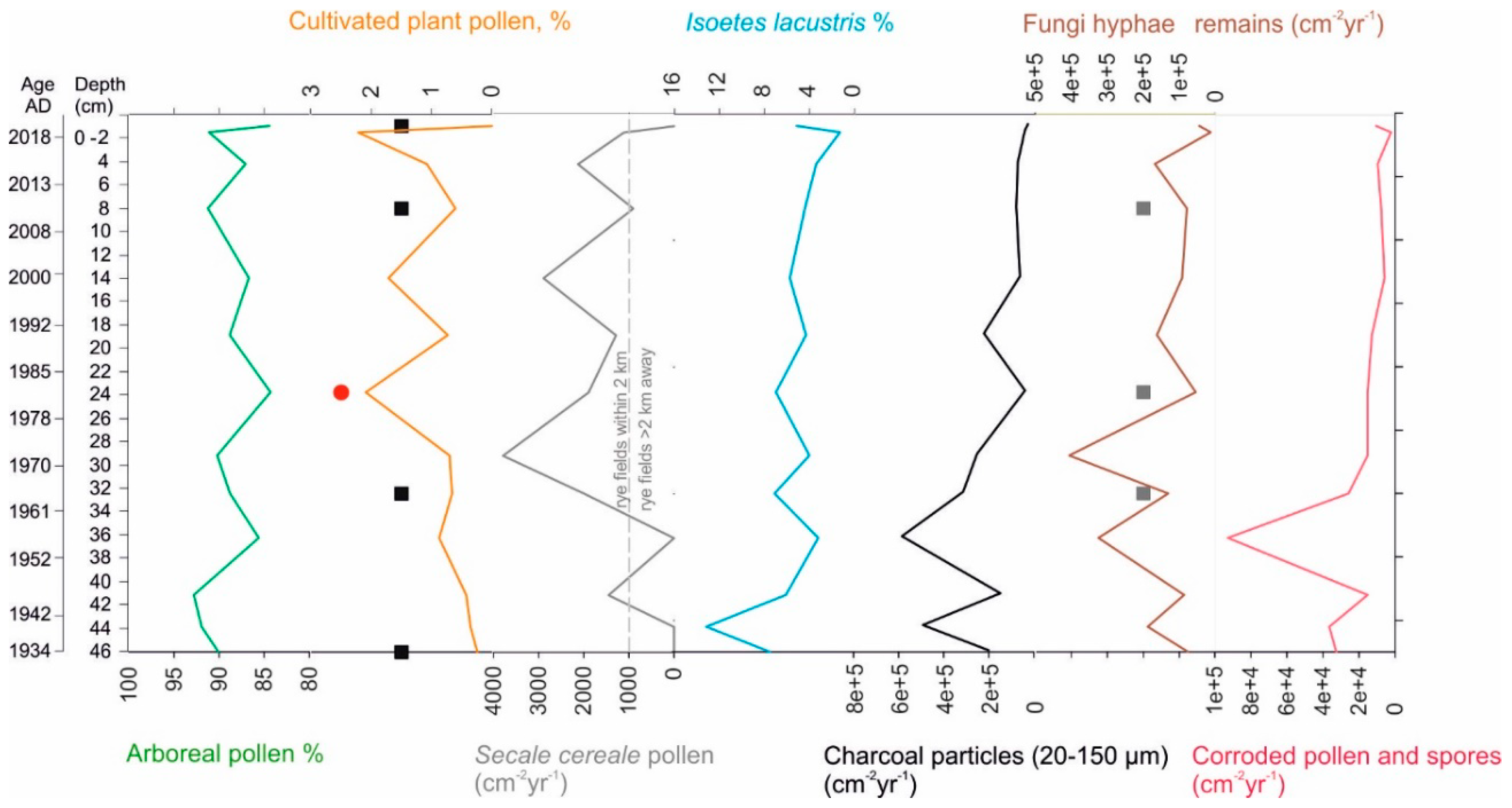
2.2.4. Pollen and Non-Pollen Palynomorphs
2.2.5. Diatom Analysis
2.2.6. Cladocera Analysis
2.2.7. Chironomidae Analysis
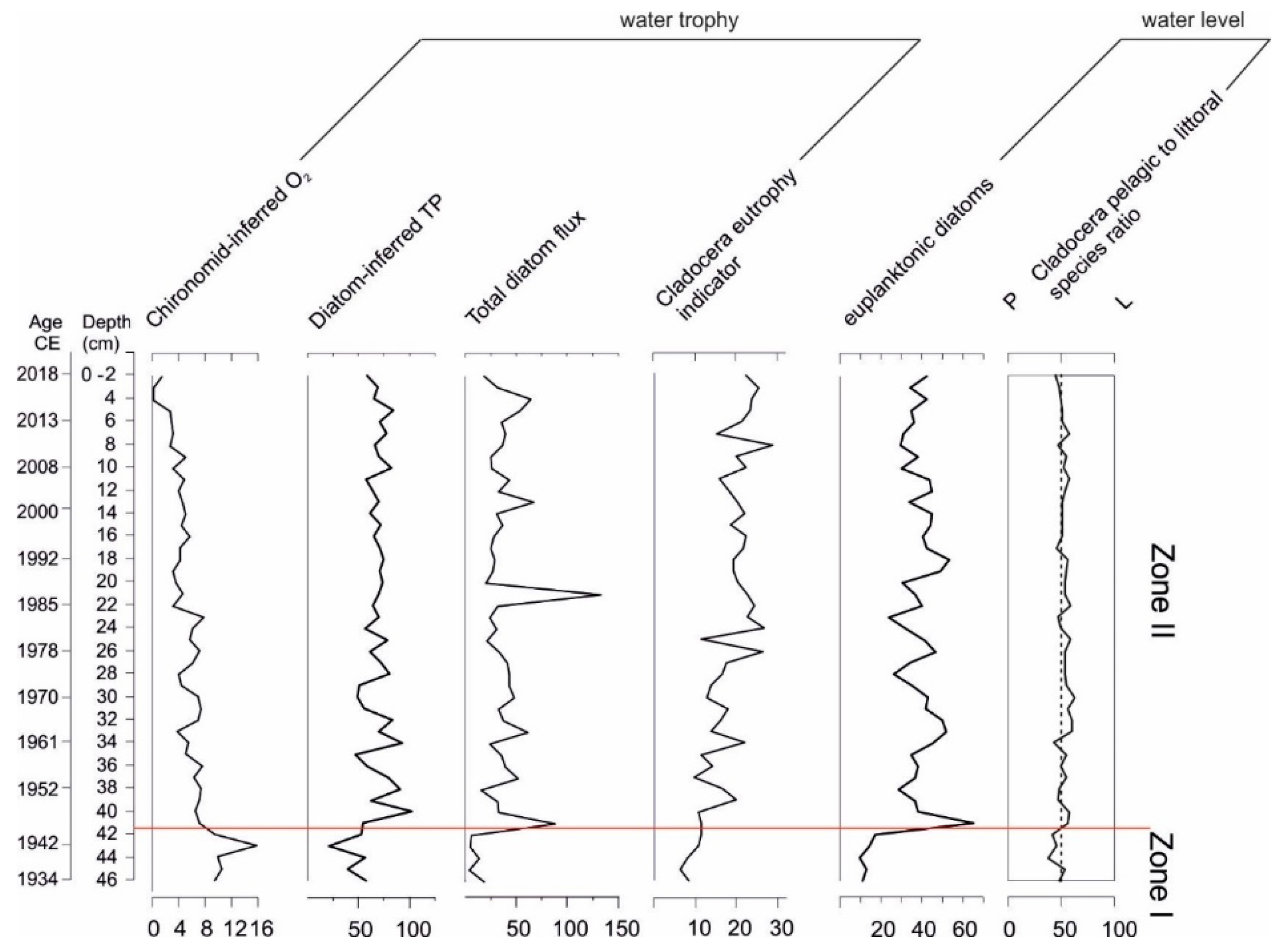
3. Results
3.1. Core Chronology
3.2. Sediment Composition
3.3. Pollen and Non-Pollen Palynomorphs
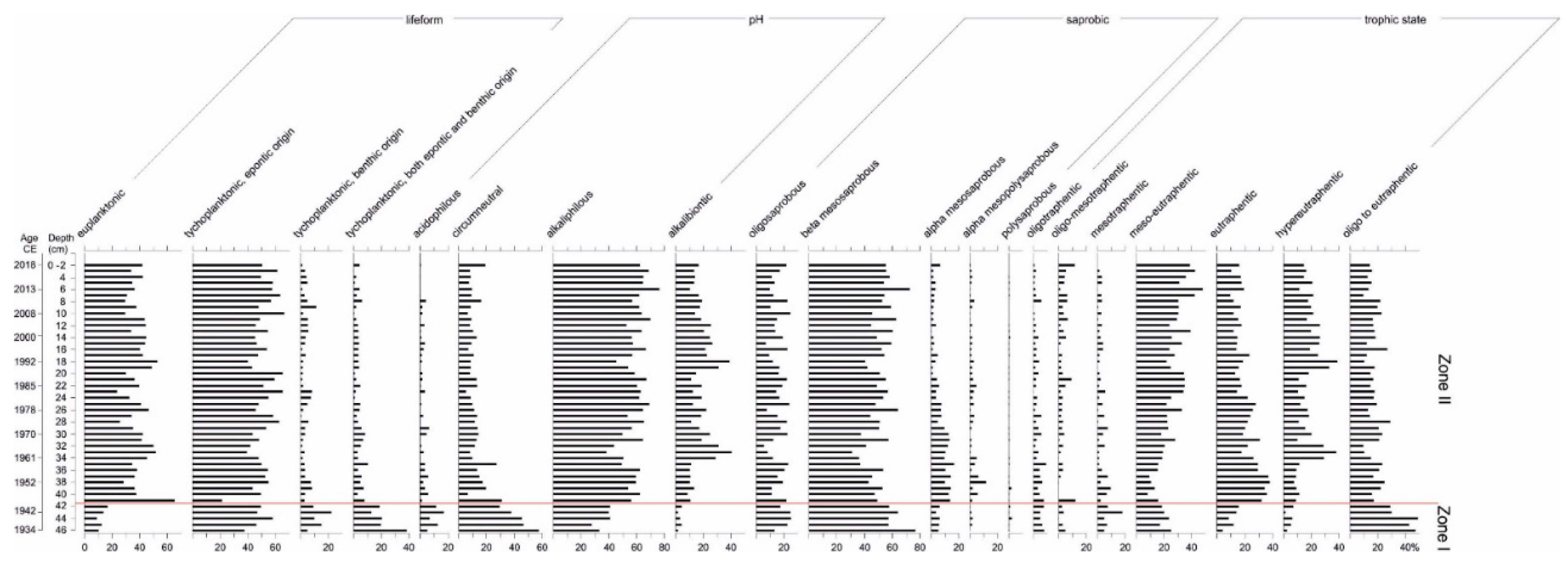
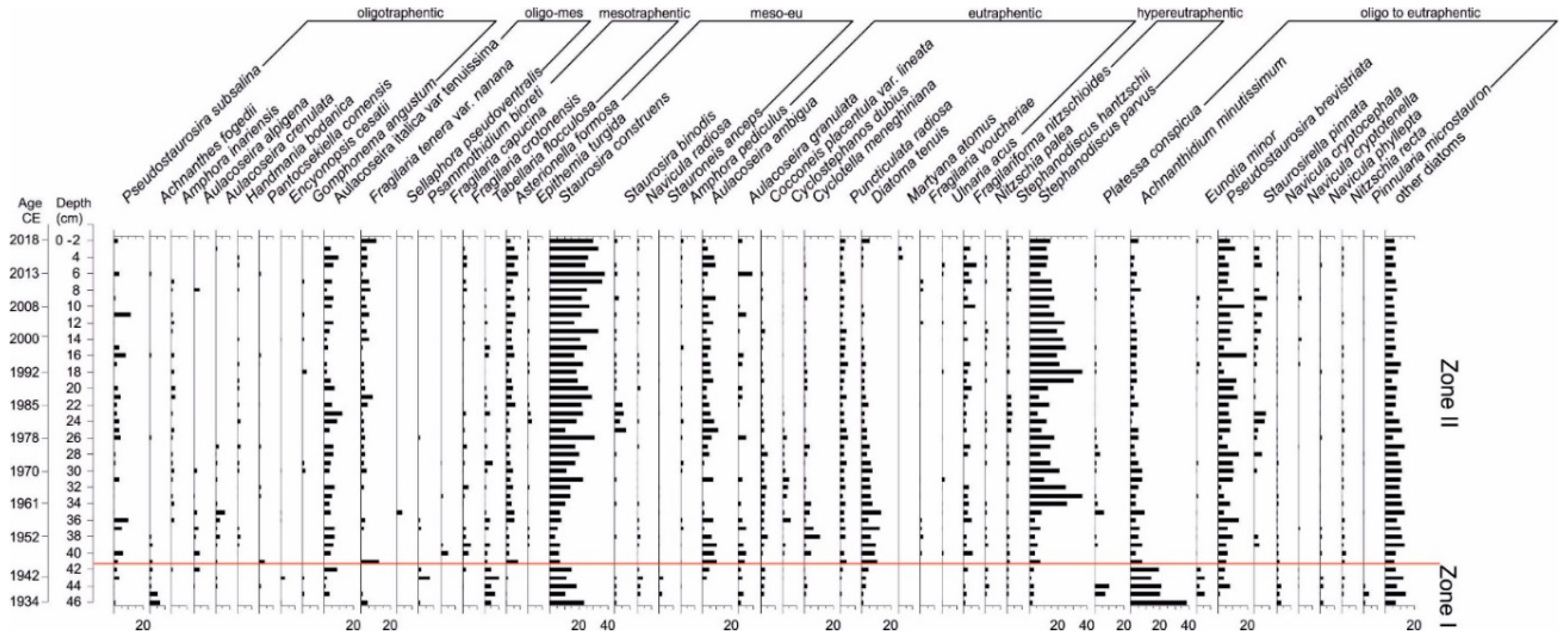
3.4. Diatom Analysis
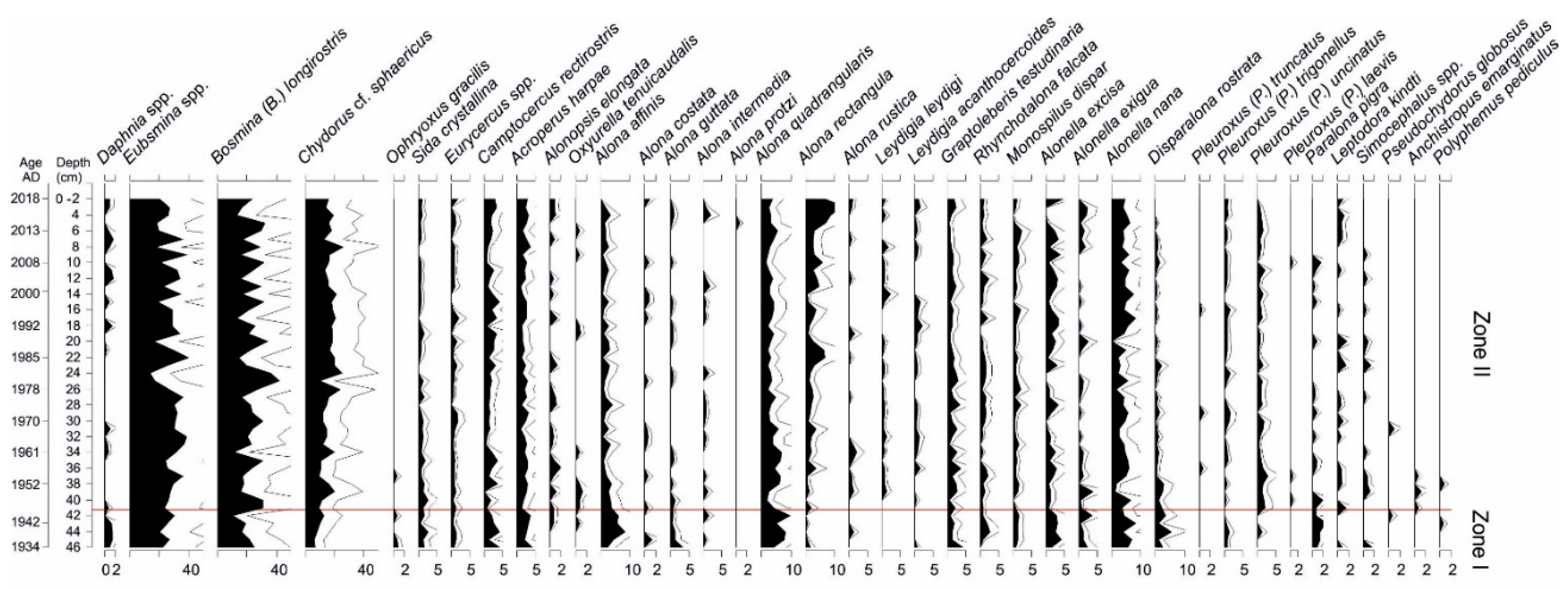
3.5. Cladocera Analysis
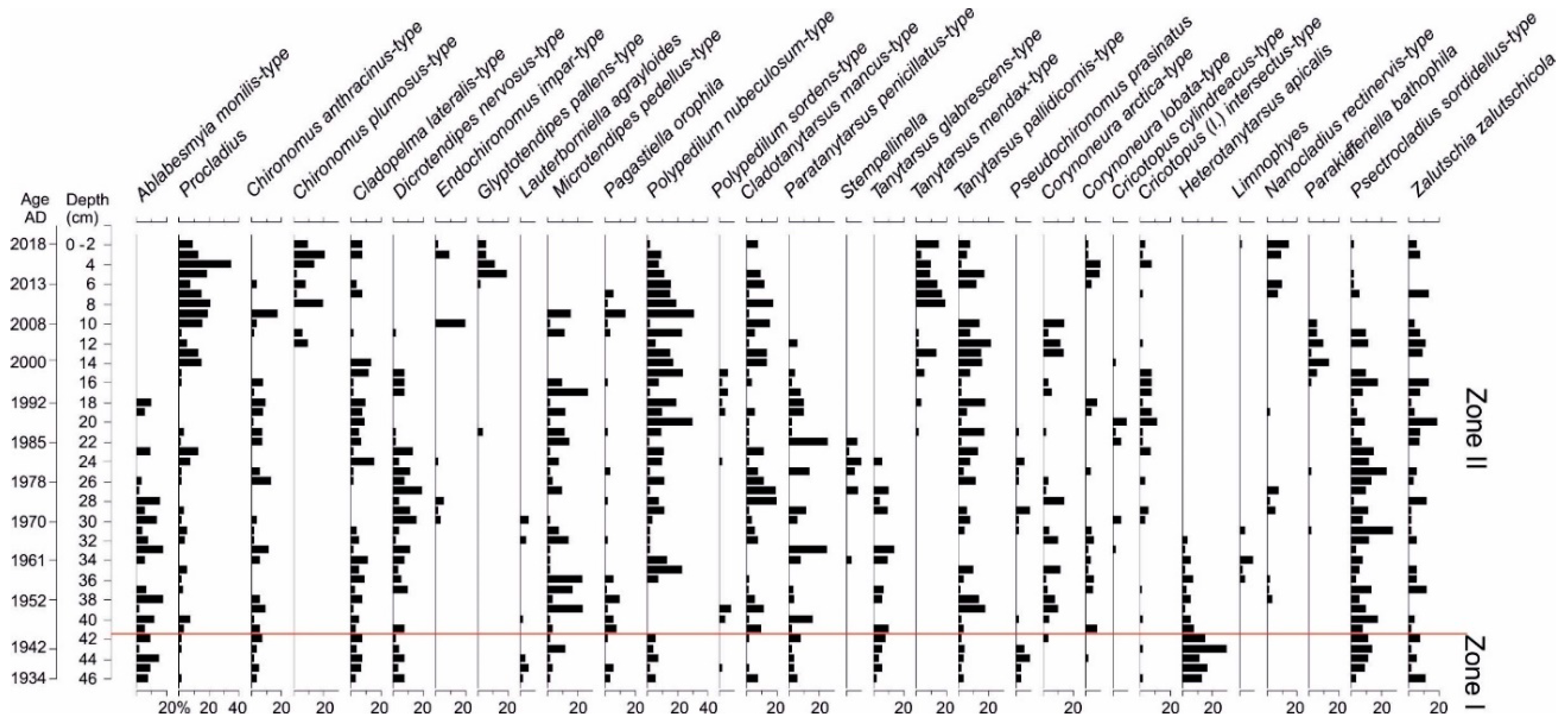
3.6. Chironomidae Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haas, M.; Baumann, F.; Castella, D.; Haghipour, N.; Reusch, A.; Strasser, M.; Eglinton, T.I.; Dubois, N. Roman-driven cultural eutrophication of Lake Murten, Switzerland. Earth Planet Sci. Lett. 2019, 505, 110–117. [Google Scholar] [CrossRef]
- Wassmann, P. Cultural eutrophication: Perspectives and prospects. In Drainage Basin Nutrient Inputs and Eutrophication: An Integrated Approach; Wassmann, P., Olli, K., Eds.; University of Tromsø: Tromsø, Norway, 2005; pp. 224–234. [Google Scholar]
- Hasler, A.D. Eutrophication of lakes by domestic drainage. Ecology 1947, 28, 383–395. [Google Scholar] [CrossRef]
- Elmgren, R.; Larsson, U. Eutrophication in the Baltic Sea area: Integrated coastal management issues. In Science and Integrated Coastal Management; von Bodungen, B., Turner, R.K., Eds.; University Press: Berlin, Germany, 2001; pp. 15–35. [Google Scholar]
- Smol, J.P. Pollution of Lakes and Rivers. A Paleoenvironmental Perspective, 2nd ed.; Blackwell Publishing Ltd.: Oxford, UK, 2008; pp. 1–396. [Google Scholar]
- Yuksel, I.; Arman, H.; Ceribasi, G. Water Resources Management of Hydropower and Dams as Sustainable Development. In Proceedings of the International Conference on Sustainable Systems and the Environment (ISSE), Sharjah, UAE, 23–24 March 2011. [Google Scholar]
- Chen, J.; Qian, H.; Gao, Y.; Wang, H.; Zhang, M. Insights into hydrological and hydrochemical processes in response to water replenishment for lakes in arid regions. J. Hydrol. 2020, 581. [Google Scholar] [CrossRef]
- Dziluma, M. Riga Water and the Riga Water Supply Museum; Riga Water: Riga, Latvia, 2003; pp. 1–86. [Google Scholar]
- Jansons, E. Rīgas Dzeramais ūdens un tā Resursu Aizsardzība. Baltezeru un to Apkaimes Izpēte; Rīgas Ūdens: Rīga, Latvija, 1997; pp. 1–53. [Google Scholar]
- Krutofala, T.; Levins, I. Pazemes ūdeņu Atradnes “Baltezers”, Iecirkņa “Akoti” Pase. Pazemes ūdeņu Ekspluatācijas Krājumu Novērtējums. Atradne “Baltezers”, iecirknis “Akoti”. Gruntsūdens Horizonts; Latvijas Vides, ģeoloģijas un Meteoroloģijas Aģentūra: Rīga, Latvija, 2006; pp. 1–38. [Google Scholar]
- Pastors, A. Sekšu ezers. In Latvijas Daba: Enciklopēdija; Kavacs, G., Ed.; Latvijas Enciklopēdija: Rīga, Latvija, 1995; Volume 5, pp. 12–13. [Google Scholar]
- Bauze-Krastiņš, M.G. Garkalnes novada teritorijas plānojums 2009–2021. Gadam. Paskaidrojuma Raksts; Garkalnes novads: Rīga, Latvija, 2009; pp. 1–169. [Google Scholar]
- Latvijas Klimats. Latvijas Vides, ģeoloģijas un Meteoroloģijas Centrs. Available online: https://www.meteo.lv/lapas/laika-apstakli/klimatiska-informacija/latvijas-klimats/latvijas-klimats?id=1199&nid=562 (accessed on 10 March 2020).
- Natural Data Management System OAK, Meža Valsts Reģistra Informācija. Available online: http://ozols.daba.gov.lv/pub/Life/ (accessed on 10 March 2020).
- Zarina, D. Sekšu Ezera, Sudrabezera un Venču Ezera Ekoloģiskais Stāvoklis. Master’s Thesis, Latvijas Universitāte, Riga, Latvija, 2014. [Google Scholar]
- Tylmann, W.; Bonk, A.; Goslar, T.; Wulf, S.; Grosjean, M. Calibrating 210Pb dating results with varve chronology and independent chronostratigraphic markers: Problems and implications. Quat. Geochronol. 2016, 32, 1–10. [Google Scholar] [CrossRef]
- Rose, N. A method for the selective removal of inorganic ash particles from lake sediments. J. Paleolimnol. 1990, 4, 61–68. [Google Scholar] [CrossRef]
- Alliksaar, T. Spatial and temporal variability of the distribution of spherical fly-ash particles in sediments in Estonia. Ph.D. Thesis, Tallinn Pedagogical University, Tallinn, Estonia, 2000. [Google Scholar]
- Hedges, J.I.; Eglinton, G.; Hatcher, P.G.; Kirchman, D.L.; Arnosti, C.; Derenne, S.; Evershed, R.P.; Kögel-Knabner, I.; de Leeuw, J.W.; Littke, R.; et al. The molecularly-uncharacterized component of non-living organic matter in natural environments. Org. Geochem. 2000, 31, 945–958. [Google Scholar] [CrossRef]
- Masiello, C.A. New directions in black carbon organic geochemistry. Mar. Chem. 2004, 92, 201–213. [Google Scholar] [CrossRef]
- Stivrins, N.; Wulf, S.; Wastegård, S.; Lind, E.M.; Allisaar, T.; Gałka, M.; Andersen, T.J.; Heinsalu, A.; Seppä, H.; Veski, S. Detection of the Askja AD 1875 cryptotephra in Latvia, Eastern Europe. J. Quat. Sci. 2016, 31, 437–441. [Google Scholar] [CrossRef]
- Blaauw, M. Methods and code for ‘classical’ age-modelling of radiocarbon sequences. Quat. Geochronol. 2010, 5, 512–518. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation: Vienna, Austria, 2018. [Google Scholar]
- Heiri, O.; Lotter, A.F.; Lemcke, G. Loss on ignition as a method for estimating organic and carbonate content in sediments, reproducibility and comparability of results. J. Paleolimnol. 2001, 25, 101–110. [Google Scholar] [CrossRef]
- Berglund, B.E.; Ralska-Jasiewiczowa, M. Pollen analysis and pollen diagrams. In Handbook of Holocene Palaeoecology and Palaeohydrology; Berglund, B., Ed.; John Wiley & Sons: Chichester, UK, 1986; pp. 455–484. [Google Scholar]
- Stockmarr, J. Tablets with spores used in absolute pollen analysis. Pollen Spores 1971, 13, 615–621. [Google Scholar]
- Fægri, K.; Iversen, J. Textbook of Pollen Analysis; John Wiley & Sons: Chichester, UK, 1986. [Google Scholar]
- Miola, A. Tools for non-pollen palynomorphs (NPPs) analysis: A list of quaternary NPP types and reference literature in English language (1972–2011). Rev. Palaeobot. Palynol. 2012, 186, 142–161. [Google Scholar] [CrossRef]
- Sweeney, C.A. A key for the identification of stomata of the native conifers of Scandinavia. Rev. Palaeobot. Palynol. 2004, 128, 281–290. [Google Scholar] [CrossRef]
- Finsinger, W.; Tinner, W. New insights on stomata analysis of European conifers 65 years after the pioneering study of Werner Trautmann (1953). Veg. Hist. Archaeobotany 2019. [Google Scholar] [CrossRef]
- Grimm, E.C. TILIA Software; Illinois State Museum, Research and Collection Center: Springfield, IL, USA, 2012. [Google Scholar]
- Battarbee, R.W. Diatom analysis. In Handbook of Holocene Paleoecology and Paleohydrology; Berglund, B.E., Ed.; John Wiley and Sons Ltd.: Chichester, UK, 1986; pp. 527–570. [Google Scholar]
- Battarbee, R.W.; Kneen, M.J. The use of electronically counted microsphere sin absolute diatom analysis. Limnol. Oceanogr. 1982, 27, 184–188. [Google Scholar] [CrossRef]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 4. Achnanthaceae. In Süsswasserflora von Mitteleuropa, 3rd ed.; Ettl, H., Gärtner, G., Gerloff, J., Heyning, H., Mollenhauer, D., Eds.; Fisher: Stuttgardt, Germany, 2011; Volume 2, p. 437. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 1. Naviculaceae. In Süsswasserflora von Mitteleuropa, 4th ed.; Ettl, H., Gerloff, J., Heyning, H., Mollenhauer, D., Eds.; Fisher: Stuttgardt, Germany, 2010; Volume 2, p. 876. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 2. Ephitemiaceae. Bacillariaceae. Surirellaceae. In Süsswasserflora von Mitteleuropa, 4th ed.; Ettl, H., Gerloff, J., Heyning, H., Mollenhauer, D., Eds.; Fisher: Stuttgardt, Germany, 2008; Volume 2, p. 596. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 3. Centrales. Fragilariaceae. Eunotiaceae. In Süsswasserflora von Mitteleuropa, 3rd ed.; Ettl, H., Gerloff, J., Heyning, H., Mollenhauer, D., Eds.; Fisher: Stuttgardt, Germany, 2008; Volume 2, pp. 1–577. [Google Scholar]
- Lange-Bertalot, H.; Hofmann, G.; Werum, M. Diatomeen im Süßwasser-Benthos von Mitteleuropa; A.R.G. Gantner Verlag, K.G.: Ruggell, Liechtenstein, 2011; pp. 1–908. [Google Scholar]
- Lange-Bertalot, H.; Metzeltin, D. Indicators of Oligotrophy. 800 taxa representative of three ecologically distinct lake types. In Iconographia Diatomologica: Annotated Diatom Micrographs; Lange-Bertalot, H., Ed.; A.R.G. Gantner Verlag, K.G.: Ruggel, Liechtenstein, 1996; pp. 1–390. [Google Scholar]
- Lange-Bertalot, H.; Bąk, M.; Witkowski, A.; Tagliaventi, N. Eunotia and related genera. In Diatoms of Europe; A.R.G. Gantner Verlag, K.G.: Ruggell, Liechtenstein, 2011; Volume 6, pp. 1–747. [Google Scholar]
- AlgaeBase. Available online: https://www.algaebase.org/ (accessed on 19 February 2020).
- Lecointe, C.; Coste, M.; Prygiel, J. “Omnidia”: Software for taxonomy, calculation of diatom indices and inventories management. In Twelfth International Diatom Symposium; Springer: Dordrecht, The Netherlands, 1993; pp. 509–513. [Google Scholar]
- Denys, L. A Check-List of the Diatoms in the Holocene Deposits of the Western Belgian Coastal Plain with a Survey of their Apparent Ecological Requirements; Service Geologique de Belgique: Brussels, Belgium, 1991; Volume 1, pp. 1–2. [Google Scholar]
- Van Dam, H.; Mertens, A.; Sinkeldam, J. A coded checklist and ecological indicator values of freshwater diatoms from the Netherlands. Neth. J. Aquat. Ecol. 1994, 28, 117–133. [Google Scholar]
- Juggins, S. Version 1.0. User Guide. The European Diatom Database; Newcastle University: Newcastle upon Tyne, UK, 2001. [Google Scholar]
- Szeroczyńska, K.; Sarmaja-Korjonen, K. Atlas of Subfossil Cladocera from Central and Northern Europe; Friends of Lower Vistula Society: Świecie, Poland, 2007; pp. 1–84. [Google Scholar]
- Kurek, J.; Korosi, J.B.; Jeziorski, A.; Smol, J.P. Establishing reliable minimum count sizes for cladoceran subfossils sampled from lake sediments. J. Paleolimnol. 2010, 44, 603–612. [Google Scholar] [CrossRef]
- Juggins, S. C2 Version 1.5. User Guide. Software for Ecological and Palaeoecological Data Analysis and Visualisation; Newcastle University: Newcastle upon Tyne, UK, 2007. [Google Scholar]
- Flössner, D. Die Haplopoda und Cladocera (ohne Bosminidae) Mitteleuropas; Backhuys Publishers: Leiden, The Netherlands, 2000; p. 428. [Google Scholar]
- Brooks, S.J.; Langdon, P.G.; Heiri, O. The Identification and Use of Palaearctic Chironomidae Larvae in Palaeoecology; Quaternary Research Association: Edinburgh, UK, 2007. [Google Scholar]
- Luoto, T.P.; Nevalainen, L. Inferring reference conditions of hypolimnetic oxygen for deteriorated Lake Mallusjärvi in the cultural landscape of Mallusjoki, southern Finland using fossil midge assemblages. Water Air Soil Pollut. 2011, 217, 663–675. [Google Scholar] [CrossRef]
- Luoto, T.P.; Salonen, V.P. Fossil midge larvae (Diptera: Chironomidae) as quantitative indicators of late-winter hypolimnetic oxygen in southern Finland: A calibration model, case studies and potentialities. Boreal Environ. Res. 2010, 15, 1–18. [Google Scholar]
- Meltsov, V.; Poska, A.; Odgaardd, B.V.; Sammula, M.; Kull, T. Palynological richness and pollen sample evenness in relation to local floristic diversity in southern Estonia. Rev. Palaeobot. Palynol. 2011, 166, 344–351. [Google Scholar] [CrossRef]
- Stivrins, N.; Kołaczek, P.; Reitalu, T.; Seppä, H.; Veski, S. Phytoplankton response to the environmental and climatic variability in a temperate lake over the last 14,500 years in eastern Latvia. J. Paleolimnol. 2015, 54, 103–119. [Google Scholar] [CrossRef]
- Schindler, D.W. The Dilemma of Controlling Cultural Eutrophication of Lakes. Proc. R. Soc. B 2012, 279, 4322–4333. [Google Scholar] [CrossRef] [PubMed]
- Lampert, W.; Sommer, U. Limnoecology; Oxford University Press: Oxford, UK, 1997; pp. 1–382. [Google Scholar]
- Schindler, D.W. Recent advances in the understanding and management of eutrophication. Limnol. Oceanogr. 2006, 51, 356–363. [Google Scholar] [CrossRef]
- Leira, M.; Cantonati, M. Effects of water-level fluctuations on lakes: An annotated bibliography. Hydrobiologia 2008, 613, 171–184. [Google Scholar] [CrossRef]
- Koralay, N.; Kara, O. Effects of Soil Erosion on Water Quality and Aquatic Ecosystem in a Watershed. In Proceedings of the 1st International Congress on Agricultural Structures and Irrigation, Antalya, Turkey, 26–28 September 2018; pp. 20–29. [Google Scholar]
- Kuptsch, P. Die Cladoceren der Umgegend von Riga. Arch. Hydrobiol. 1927, 18, 273–315. [Google Scholar]
- Hakkari, L. Zooplankton species as indicators of environment. Aqua Fenn. 1972, 1, 46–54. [Google Scholar]
- Lyche, A. Cluster analysis of plankton community structure in 21 lakes along a gradient of trophy. Verh. Internat. Verein Limnol. 1990, 24, 586–591. [Google Scholar] [CrossRef]
- Urtane, L. Cladocera as indicators of lake types and trophic state in Latvian lakes. Ph.D. Thesis, University of Latvia, Riga, Latvia, 1998. [Google Scholar]
- Walseng, B.; Halvorsen, G. Littoral microcrustaceans as indices of trophy. Verh. Internat. Verein Limnol. 2005, 29, 827–829. [Google Scholar] [CrossRef]
- Jensen, T.C.; Dimante-Deimantovica, I.; Schartau, A.K.; Walseng, B. Cladocerans respond to differences in trophic state in deeper nutrient poor lakes from Southern Norway. Hydrobiologia 2013, 715, 101–112. [Google Scholar] [CrossRef]
- Umja Ezera Dabas Aizsardzības Plāns. Available online: https://www.daba.gov.lv/upload/File/DAPi_apstiprin/DL_Ummis-06.pdf (accessed on 20 March 2020).
- Susko, U. (Latvian Fund for Nature, Riga, Latvia). Personal communication, 2020.
- Sloka, N. Latvijas PSR Dzīvnieku Noteicējs. Latvijas Kladoceru (Cladocera) Fauna un Noteicējs; P. Stučkas Latvijas valsts universitāte: Rīga, Latvia, 1981; p. 146. [Google Scholar]
- Eutrophication of Waters, Monitoring, Assessment and Control; OECD (Organisation for Economic Cooperation and Development): Paris, France, 1982.
- Andrén, E. Changes in the Composition of the Diatom Flora During the Last Century Indicate Increased Eutrophication of the Oder Estuary, South-western Baltic Sea. Estuar. Coast. Shelf Sci. 1999, 48, 665–676. [Google Scholar] [CrossRef]
- Andrén, E.; Clarke, A.L.; Telford, R.J.; Wesckström, K.; Vilbaste, S.; Aigars, J.; Conley, D.; Johnsen, T.; Juggins, S.; Korhola, A. Defining Reference Conditions for Coastal Areas in the Baltic Sea; TemaNord Series; Nordic Council of Ministers: Copenhagen, Denmark, 2007. [Google Scholar]
- Andrén, E.; Shimmield, G.; Brand, T. Environmental changes of the last three centuries indicated by siliceous microfossil records from the southwestern Baltic Sea. Holocene 1999, 9, 25–38. [Google Scholar] [CrossRef]
- Weckström, K. Assessing recent eutrophication in coastal waters of the Gulf of Finland (Baltic Sea) using subfossil diatoms. J. Paleolimnol. 2006, 35, 571–592. [Google Scholar] [CrossRef]
- Milecka, K.; Bogaczewicz-Adamczak, B. Zmiany żyzności trofii w ekosystemach miękkowodnych jezior Borów Tucholskich. Przegl. Geol 2006, 54, 81–86. [Google Scholar]
- Christensen, K.K.; Sand-Jensen, K. Precipitated iron and manganese plaques restrict root uptake of phosphorus in Lobelia dortmanna. Can. J. Bot. 1998, 76, 2158–2163. [Google Scholar]
- Battarbee, R.W.; Simpson, G.L.; Shilland, E.M.; Flower, R.J.; Kreiser, A.; Yang, H.; Clarke, G. Recovery of UK lakes from acidification: An assessment using combined palaeoecological and contemporary diatom assemblage data. Ecol. Indic. 2014, 37, 365–380. [Google Scholar] [CrossRef]
- Battarbee, R.W.; Jones, V.J.; Flower, R.J.; Cameron, N.G.; Bennion, H.; Carvalho, L.; Juggins, S. Diatoms. In Tracking Environmental Change Using Lake Sediments. Terrestrial, Algal, and Siliceous Indicators; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwier Academic Publishers: Norwell, MA, USA, 2001; Volume 3, pp. 155–202. [Google Scholar]
- Battarbee, R.W.; Charles, D.F.; Dixit, S.S.; Renberg, I. Diatoms as indicators of surface water acidity. In The Diatoms: Applications for the Environmental and Earth Sciences; Stoermer, E.F., Smol, J.P., Eds.; Cambridge University Press: Cambridge, UK, 1999; pp. 85–127. [Google Scholar]
- Battarbee, R.W.; Charles, D.F. Diatom-based pH reconstruction studies of acid lakes in Europe and North America: A synthesis. Water Air Soil Pollut. 1986, 30, 347–354. [Google Scholar] [CrossRef]
- Klavins, M.; Kokorite, I.; Jankevica, M.; Mazeika, J.; Rodinov, V. Trace elements in sediments of lakes in Latvia. In Proceedings of the 4th WSEAS International Conference on Energy and Development—Environment—Biomedicine, Corfu Island, Greece, 14–16 July 2011. [Google Scholar]
- Kļaviņš, M.; Briede, A.; Kļaviņa, I.; Rodinov, V. Metals in sediments of lakes in Latvia. Environ. Int. 1995, 21, 451–458. [Google Scholar] [CrossRef]
- Liiv, M.; Alliksaar, T.; Amon, L.; Freiberg, R.; Heinsalu, A.; Reitalu, T.; Saarse, L.; Seppä, H.; Stivrins, N.; Tõnno, I.; et al. Late glacial and early Holocene climate and environmental changes in the eastern Baltic area inferred from sediment C/N ratio. J. Paleolimnol. 2019, 61, 1–16. [Google Scholar] [CrossRef]
- Terasmaa, J.; Puusepp, L.; Marzecová, A.; Vandel, E.; Vaasma, T.; Koff, T. Natural and human-induced environmental changes in Eastern Europe during the Holocene: A multi-proxy palaeolimnological study of a small Latvian lake in a humid temperate zone. J. Paleolimnol. 2013, 49, 663–678. [Google Scholar] [CrossRef]
- Meyers, P.A.; Ishiwatari, R. Lacustrine organic geochemistry—An overview of indicators of organic matter sources and diagenesis in lake sediments. Org. Geochem. 1993, 20, 867–900. [Google Scholar] [CrossRef]
- Meyers, P.A.; Teranes, J.L. Sediment Organic Matter. In Tracking Environmental Change Using Lake Sediments: Physical and Geochemical Methods, Developments in Paleoenvironmental Research; Last, W.M., Smol, J.P., Eds.; Springer: Dordrecht, The Netherlands, 2001; pp. 239–269. [Google Scholar]
- Buzajevs, V.; Levina, N.; Levins, I. Pazemes ūdeņu bilance un kvalitāte Baltezera ūdensgūtnēs; Valsts Ģeoloģijas Dienests: Riga, Latvia, 1997; pp. 1–56. [Google Scholar]
- Sarmaja-Korjonen, K. Correlation of fluctuations in cladoceran planktonic: Littoral ratio between three cores from a small lake in southern Finland: Holocene water-level changes. Holocene 2001, 11, 53–63. [Google Scholar] [CrossRef]
- Cooper, J.T.; Steinitz-Kannan, M.; Kallmeyer, D.E. Bacillariophyta: The Diatoms. In Algae: Source of Treatment; Ripley, M.G., Ed.; American Water Works Association: Denver, CO, USA, 2010; pp. 207–215. [Google Scholar]
- Zelnik, I.; Balanč, T.; Toman, M. Diversity and Structure of the Tychoplankton Diatom Community in the Limnocrene Spring Zelenci (Slovenia) in Relation to Environmental Factors. Water 2018, 10, 361. [Google Scholar] [CrossRef]
- Dimante-Deimantovica, I. (Latvian Institute of Aquatic Ecology, Riga, Latvia). Personal communication, 2020.
- Ardiles, V.; Alcocer, J.; Vilaclara, G.; Oseguera, L.A.; Velasco, L. Diatom fluxes in a tropical, oligotrophic lake dominated by large-sized phytoplankton. Hydrobiologia 2012, 679, 77–90. [Google Scholar] [CrossRef]
- Romero, O.E.; Thunell, R.C.; Astor, Y.; Varela, R. Seasonal and interannual dynamics in diatom production in the Cariaco Basin, Venezuela. Deep-Sea Res. 2009, 56, 571–581. [Google Scholar] [CrossRef]
- Ryves, D.B.; Jewson, D.H.; Sturm, M.; Battarbee, R.W.; Flower, R.J.; Mackay, A.W.; Granin, N.G. Quantitative and qualitative relationships between planktonic diatom communities and diatom assemblages in sedimenting material and surface sediments in Lake Baikal, Siberia. Limnol. Oceanogr. 2003, 48, 1643–1661. [Google Scholar] [CrossRef]
- Ceirans, A. Zooplankton indicators of trophy in Latvian lakes. In Acta Universitatis Latviensis. Biology; University of Latvia: Riga, Latvia, 2007; Volume 723, pp. 61–69. [Google Scholar]
- Veski, S.; Amon, L.; Heinsalu, A.; Reitalu, T.; Saarse, L.; Stivrins, N.; Vassiljev, J. Lateglacial vegetation dynamics in the eastern Baltic region between 14,500 and 11,400 cal yr BP: A complete record since the Bolling (GI-1e) to the Holocene. Quat. Sci. Rev. 2012, 40, 39–53. [Google Scholar] [CrossRef]
- Kołaczek, P.; Zubek, S.; Błaszkowski, J.; Mleczko, P.; Włodzimierz, M. Erosion or plant succession—How to interpret the presence of arbuscular mycorrhizal fungi (Glomeromycota) spores in pollen profiles collected from mires. Rev. Palaeobot. Palynol. 2013, 189, 29–37. [Google Scholar] [CrossRef]
- Wang, L.; Mackay, A.W.; Leng, M.J.; Rioual, P.; Panizzo, V.N.; Lu, H.; Gu, Z.; Chu, G.; Han, J.; Kendrick, C.P. Influence of the ratio of planktonic to benthic diatoms on lacustrine organic matter δ13C from Erlongwan maar lake, northeast China. Org. Geochem. 2013, 54, 62–68. [Google Scholar] [CrossRef]
- Wolin, J.A.; Stone, J.R. Diatoms as Indicators of Water-Level Change in Freshwater Lakes. In The Diatoms Applications to the Environmental and Earth Sciences; Stoermer, E.F., Smol, J.P., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 174–185. [Google Scholar]
- Carmignani, J.R.; Roy, A.H. Ecological impacts of winter water level drawdowns on lake littoral zones: A review. Aquat. Sci. 2017, 79, 803–824. [Google Scholar] [CrossRef]
- Magny, M.; Arnaud, F.; Billaud, Y.; Marguet, A. Lake-level fluctuations at Lake Bourget (eastern France) around 4500–3500 cal. a BP and their palaeoclimatic and archaeological implications. J. Quat. Sci. 2012, 27, 494–502. [Google Scholar] [CrossRef]
- Theuerkauf, E.J.; Braun, K.N.; Nelson, D.M.; Kaplan, M.; Vivirito, S.; Williams, J.D. Coastal geomorphic response to seasonal water-level rise in the Laurentian Great Lakes: An example from Illinois Beach State Park, USA. J. Great Lakes Res. 2019, 45, 1055–1068. [Google Scholar] [CrossRef]
- Holmer, M.; Storkholm, P. Sulphate reduction and sulphur cycling in lake sediments: A review. Freshw. Biol. 2001, 46, 431–451. [Google Scholar] [CrossRef]
- Couture, R.M.; Fischer, R.; Van Cappellen, P.; Gobeil, C. Non-steady state diagenesis of organic and inorganic sulfur in lake sediments. Geochim. Cosmochim. Acta 2016, 194, 15–33. [Google Scholar] [CrossRef]
- Aasif, A.; Nilli, A.; Aasif, L.; Hema, A.; Rayees, S. Diatom Diversity and Organic Matter Sources in Water Bodies around Chennai, Tamil Nadu, India. MOJ Ecol. Environ. Sci. 2017, 2, 141–146. [Google Scholar]
- Rosińska, A.; Rakocz, K. Rola biodegradowalnej materii organicznej w procesie dezynfekcji wody. Inż. Ochr. Środ. 2013, 16, 511–521. [Google Scholar]
- Brodersen, K.P.; Quinlan, R. Midges as palaeoindicators of lake productivity, eutrophication and hypolimnetic oxygen. Quat. Sci. 2006, 25, 1995–2012. [Google Scholar] [CrossRef]
- Timrots, P. (Riga Water Supply Museum, Riga, Latvia). Personal communication, 2019.
- Brodersen, K.P.; Pedersen, O.; Lindegaard, C.; Hamburger, K. Chironomids (Diptera) and oxy-regulatory capacity: An experimental approach to paleolimnological interpretation. Limnol. Oceanogr. 2004, 49, 1549–1559. [Google Scholar]
- Kacalova, O.; Laganovsla, R. Zivju Barības Bāze Latvijas PSR Ezeros; Latvijas PSR Zinātņu akadēmijas izdevniecība: Rīga, Latvija, 1961; pp. 17–23. [Google Scholar]
- Bledzki, L.A.; Rybak, J.I. Freshwater Crustacean Zooplankton of Europe Cladocera and Copepoda (Calanoida, Cyclopoida), Key to Species Identification, with Notes on Ecology, Distribution, Methods and Introduction to Data Analysis; Springer: New York, NY, USA, 2016; pp. 1–918. [Google Scholar]
- Lielā un Mazā Baltezera Eitroficēšanās Izpēte. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwiPufXQp7_oAhXpxIsKHfMmDOcQFjAAegQIBRAB&url=https%3A%2F%2Fwww.ezeri.lv%2Fblog%2FDownloadAttachment%3Fid%3D347&usg=AOvVaw1qjWh6fVLl6-JGxc-_EbzG (accessed on 10 March 2020).
- Balode, M.; Purina, I.; Strake, S.; Purvina, S.; Pfeifere, M.; Barda, I.; Povidisa, K. Toxic cyanobacteria in the lakes located in Rīga (the capital of Latvia) and its surroundings: Present state of knowledge. Afr. J. Mar. Sci. 2006, 28, 225–230. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zawiska, I.; Dimante-Deimantovica, I.; Luoto, T.P.; Rzodkiewicz, M.; Saarni, S.; Stivrins, N.; Tylmann, W.; Lanka, A.; Robeznieks, M.; Jilbert, T. Long-Term Consequences of Water Pumping on the Ecosystem Functioning of Lake Sekšu, Latvia. Water 2020, 12, 1459. https://doi.org/10.3390/w12051459
Zawiska I, Dimante-Deimantovica I, Luoto TP, Rzodkiewicz M, Saarni S, Stivrins N, Tylmann W, Lanka A, Robeznieks M, Jilbert T. Long-Term Consequences of Water Pumping on the Ecosystem Functioning of Lake Sekšu, Latvia. Water. 2020; 12(5):1459. https://doi.org/10.3390/w12051459
Chicago/Turabian StyleZawiska, Izabela, Inta Dimante-Deimantovica, Tomi P. Luoto, Monika Rzodkiewicz, Saija Saarni, Normunds Stivrins, Wojciech Tylmann, Anna Lanka, Martins Robeznieks, and Tom Jilbert. 2020. "Long-Term Consequences of Water Pumping on the Ecosystem Functioning of Lake Sekšu, Latvia" Water 12, no. 5: 1459. https://doi.org/10.3390/w12051459
APA StyleZawiska, I., Dimante-Deimantovica, I., Luoto, T. P., Rzodkiewicz, M., Saarni, S., Stivrins, N., Tylmann, W., Lanka, A., Robeznieks, M., & Jilbert, T. (2020). Long-Term Consequences of Water Pumping on the Ecosystem Functioning of Lake Sekšu, Latvia. Water, 12(5), 1459. https://doi.org/10.3390/w12051459





