Abstract
This paper presents the first study of five small lakes located in the city of Murmansk. Field work was carried out during 2018–2019. Water samples were collected using a bathometer, while the sediments were sampled using an Ekman grab and Limnos gravity corer. It was found that the water of the studied lakes in Murmansk belong to the sodium group of the chloride class and to the calcium group of the hydrocarbonate class. Compared to the background level, elevated pH, concentrations of the main cations of alkali and alkaline-earth metals, N compounds, total dissolved solids, and heavy metals were found in the lakes, which indicate exposure to anthropogenic impacts. The sediments of the lakes, composed of organomineral and mineral silts, also have an elevated content of heavy metals compared to the background. The most significant excessive concentrations were found for V, Ni, Sb, Pb, Co, Cr, and W. Based on the calculated pollution load index and geoaccumulation index of the sediments, the studied water bodies in Murmansk can be classified as lakes with heavy and extremely heavy pollution levels. The primary pollution sources are emissions from the Murmansk thermal power plant, coal port, road and, rail transport.
1. Introduction
Global social, political, economic, and environmental problems have become increasingly acute since the late 19th century. Urbanization and associated risks are most significant. As a result, pressure on the environment increases inevitably, and its major constituents, such as water resources, decrease qualitatively [1,2]. Aquatic systems located in technogenic areas are commonly affected by a variety of large-scale human activities from the disturbance of hydrological and heat regimes to water pollution [3,4]. This is especially so in the case of water chemical and phisical properties in urban and industrial territories [5]. One of such territories is Murmansk city, which is located in the northwest of Russia on the hilly coast of Barents Sea (Figure 1). Murmansk is the biggest city beyond the Polar Circle with a population of about 300,000 people and the capital city of the Murmansk region. The history of the city stretches back 103 years.
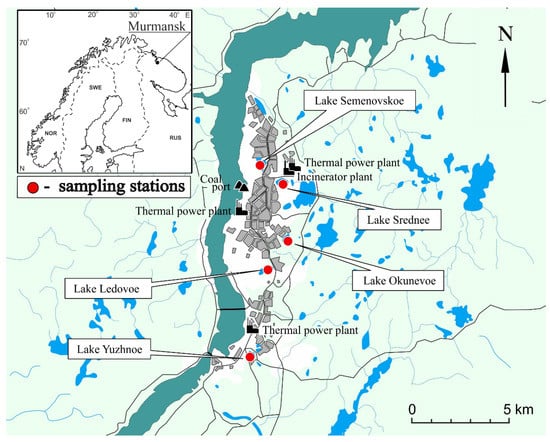
Figure 1.
Map of the Murmansk area showing the lakes studied.
The crucial sources of pollution of Murmansk are emissions from thermal power plants, cars and trains, as well as a coal dusting of the sea trade port. The Murmansk thermal power plant was commissioned in 1934 and used coal as the main fuel source until 1964. Since the 1960s, the main fuel of the thermal power plant of Murmansk has been fuel oil. According to the annual environmental reports for the Murmansk region, emissions of SO2, NO2, suspended substances, heavy metals, and other pollutants from the Murmansk thermal power plant were 14,100 tons in 2017 and 15,310 tons in 2018. Refusal to move away from using fuel oil is the most significant problem for sustainable development of the territory of Murmansk and favorable functioning of aquatic ecosystems of the city.
Besides this, there are potential negative impacts of other large enterprises of the Murmansk region on water ecosystems in Murmansk. This mainly relates to emissions of nickel and nonferrous metallurgy plant complexes in the towns of Monchegorsk and Nikel. Primarily, these factories emit great amounts of Cu, Ni, and S, which are the primary pollutants of the Murmansk and neighboring regions [6]. Also, the long-range transport of different pollutants such as Pb, Cd, Tl, Sb, and Bi from other countries of Europe can cause environmental risks for water environments in Murmansk [7,8,9].
Many scientific studies on the ecological assessment of the current state of natural systems affected by human activities have been conducted. Nevertheless, the special integrated studies of water bodies in urbanized areas are scarce, as are the studies of the urban environment in the Arctic zone, where natural and climatic conditions contribute to the high sensitivity of its ecosystems to the human impact and considerably retard natural restoration after their damage. The purpose of this study is to provide a common environmental analysis of water and surface sediment in five lakes of Murmansk and an assessment of risks linked with pollution of the lakes.
2. Materials and Methods
Murmansk is located on the eastern rocky shore of Kola Bay in the Barents Sea and is one of Russia’s biggest ports. Murmansk has around 20 lakes affected by various human activities. In order to fully assess the impact of the urban environment on lake ecosystems, five lakes were selected for the study (Semenovskoe, Srednee, Okunevoe, Ledovoe, and Yuzhnoe) located in different parts of the city of Murmansk and having great recreational significance for the city’s residents and visitors.
The studied lakes are located in the Barents Sea watershed. The main morphometric characteristics of the lakes are shown in Table 1. Their depths were measured with a Garmin Echomap Plus 42cv (Garmin Ltd., Olathe, KS, USA) echo sounder map plotter. The number of profiles varied with lake size. Echo sounding data were processed and depth maps (Figure 2) compiled using Golden Software Surfer 13 (Golden Software, Golden, CO, USA).

Table 1.
Main morphometric characteristics of Murmansk’s lakes.
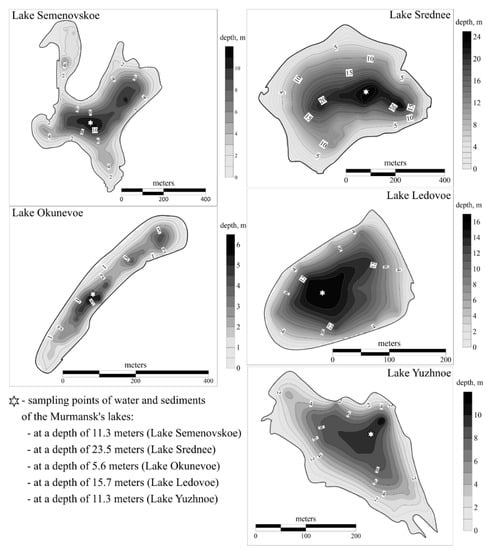
Figure 2.
Maps showing the depths of the researched lakes.
Lake Semenovskoe (68.99080° N and 33.08851° E) is a natural lake situated in the center of Murmansk (Lenin District). It covers an area of 0.213 km2. The lake basin is structurally complex and consists of two main sectors connected by a small strait. There are two adjacent moderately deep to deep stretches with maximum depths of 11.3 and 10.2 m. It is the widest portion of the lake. The second sector is more narrow and shallow. The rocky but low lakeshores are contaminated with household garbage. An annual world winter swimming championship is held on Lake Semenovskoe. This lake is located 1.8 km from the coal port, 2.1 km from Murmansk thermal power plant, and 2.4 km from the incinerator plant.
Lake Srednee (68.98147° N and 33.12422° E) is a natural water body situated in the city center near the eastern boundary of Murmansk’s urbanized environment (Oktyabrsky District). The lake basin displays a simple rounded structure and has a maximum depth of 23.5 m. This value shows that Lake Srednee is the deepest water body described in this paper. The lake covers an area of 0.248 km2. The lake shores are rocky and sandy. The lake is used by locals for recreation, mostly in the summer. Lake Srednee is located 1 km from the Murmansk thermal power plant, 1.4 km from the incinerator plant, and 2.6 km from the coal port.
Lake Ledovoe (68.93313° N and 33.10451° E) is a naturally formed water body located in the city center (Pervomaisky District). The lake basin has a simple round structure and a maximum depth of 15.7 m in its center. The lake covers an area of 0.040 km2. The lake shores are heavily contaminated with household garbage. Lake Ledovoe is located 3.4 km from the Murmansk thermal power plant, 5.6 km from the coal port, and 6.5 km from the incinerator plant.
Lake Okunevoe (68.95012° N and 33.12734° E) is a natural water body located in the center of Murmansk (Pervomaisky District). The lake basin displays an elongated oval structure and has a maximum depth of 5.6 m in its center. The northern sector of the lake also has a deep-water zone. The southern sector is shallow. The lake’s area is 0.048 km2. The lake is surrounded by forest. Its left and right shores are high and rocky, while its north and south shores are paludified. The lake is used by locals for recreation (bathing, fishing, skiing, and camping) in summer and winter. Lake Okunevoe is located 2.9 km from the Murmansk thermal power plant, 4.3 km from the incinerator plant, and 4.4 km from the coal port.
Lake Yuzhnoe (68.88469° N and 33.07660° E) is a natural water body situated near the southern boundary of Murmansk adjacent to the city of Kola. The lake basin has a simple elongated structure and a maximum depth of 11.3 m in the center near the northeastern shore. The lake has an area of 0.053 km2. The steep shores, locally with a concrete fence, are sandy. There is a pier for bathing on the northwestern shore. The lake is used by locals for recreation. Lake Yuzhnoe is located 1.5 km from the Murmansk thermal power plant, 10.8 km from the coal port, and 12 km from the incinerator plant.
Field studies of the lakes were carried out in July 2018 and April and July 2019. Water samples from the surface layer (1 m from the surface) and the bottom layer (1 m from the bottom) of the lakes were collected with a 2-liter plastic bathometer. A total of 18 water samples were taken from the lakes (Semenovskoe-5, Srednee-5, Okunevoe-4, Ledovoe-5, Yuzhnoe-3). Water samples were taken in polyethylene bottles from Nalgene®, the material of which does not have sorbing properties. Prior to use, the bottles were thoroughly washed in the laboratory. When sampling water, the bottles were rinsed twice with lake water, then placed in dark containers and refrigerated (~+4 °C), and in a short time transported to the laboratory. Filtration of water samples was carried out in the laboratory during discharge using a Millipore phase (Sartorius Group, Göttingen, Germany) separation unit from high-density polypropylene through glass and polycarbonate membrane filters of Millipore HVLPO 4700, Schleicher & Schuell ME 25/21 ST, Whatman GF/A (Sartorius Group, Göttingen, Germany) with a pore size of 0.45 μm. The following parameters were determined in unfiltered samples: pH, conductivity, alkalinity, Chemical oxygen demand (CODMn), NH4+, NO3−, PO43+, total phosphorus unfiltered (TPunf), and metals (after acidification with concentrated nitric acid). In the filtered samples, Si, total phosphorus filtered (TPf), color, Cl−, and SO42+ were determined. Water chemistry was determined at the resource sharing center of the Institute of the North Industrial Ecology Problems of Kola Science Center of RAS and Institute of Geology of Karelian Research Centre of RAS using standard methods [10,11]. The water chemistry data from each station were averaged, and the hydrochemical survey data interpretation was based on the averages. For the quality control of the measurements of pH, alkalinity, and concentrations of chlorides, sulfates, alkali, and alkaline-earth metals, the specialized ALPEFORM software (MOLAR project, UCL, London, UK & NIVA, Oslo, Norway) suite was used, including an assessment of ion balance and measured and calculated electrical conductivity. The quality of the laboratory analytical measurements was evidenced by annual international verification [12].
The top layers of the sediments of all lakes were sampled using an Ekman grab (Aquainstrument company, St. Petersburg, Russia) to assess loss on ignition and total content of heavy metals. One averaged sample was taken in July 2018 from each lake as described. This is usually sufficient when studying the sediments of lakes because lake sediments cannot change too quickly. In addition, for assessment of dynamics of an accumulation of total concentration of heavy metals, a sediment core of Lake Semenovskoe was collected in April 2019 using a Limnos gravity cover divided into 1 cm layers through a depth of 38 cm. Furthermore, several cores of surface sediments were collected and divided into 5 cm layers to analyze the mobile chemical forms of the heavy metals.
After collection, all samples were placed into plastic containers, which were labeled and packed in a cooler bag. The samples were delivered to a laboratory, where after measuring the pH of the sediment using a pH-420 millivoltmeter (Akvilon, Moscow, Russia) and an ESK-10610 glass combined electrode (IzmTehnika, Moscow, Russia), the samples were placed in a laboratory refrigerator where they were kept at a temperature of 4 °C.
For further study, the lake sediment samples were removed from refrigeration and dried to an air-dry condition at room temperature, and then to an absolutely dry condition in an oven at a temperature of approximately 105 °C. Laboratory tests were carried out at the Analysis Center of the Institute of Geology of Karelian Research Centre of RAS and at the Institute of Limnology at the Russian Academy of Sciences in St. Petersburg. The heavy metal (Mn, Zn, V, Cr, Co, Pb, Ni, Cu, Mo, Cd, Sb, W) content in the lake sediment samples was measured by the mass spectral method using an XSeries-2 ICP–MS instrument by Thermo Fisher Scientific (Waltham, MA, USA). Loss on ignition (LOI) was measured by weighting after heating the samples to a temperature of 550 °C.
To determine the total content of elements, the sediment samples were decomposed by acid solution using HF, HNO3, and HCl in an open system. For analysis, subsamples of 0.1 g were used. Together with the analyzed and blank samples, one standard (control) sample (bottom sediment from Lake Baikal BIL-1–GSO 7126-94) was prepared. Distilled water and 0.2 M MgCl2 were used as reagents to extract mobile chemical forms of the heavy metals [13].
Statistical processing of the data was done in Microsoft Excel 2007. The results of the study were visualized in Inkscape 0.48.4 (Free Software Foundation, Boston, MA, USA) and CorelDraw Graphics Suite 2019 (Corel Corporation, Ottawa, Canada). The pollution load index (PLI) was used to determine the environmental quality of the sediment [14]:
where CF is the contamination factor (ratio of metal concentration to background metal concentration) and n is the number of metals. The pollution load index can be classified as no pollution (PLI < 1), moderate pollution (1 < PLI < 2), heavy pollution (2 < PLI < 3), and extremely heavy pollution (3 < PLI) [15].
The geoaccumulation index (Igeo) of heavy metals in core sediments of the Lake Semenovskoe was calculated using the formula:
where C is the metal concentration in a given layer and B is the background metal concentration measured in the deepest layer of each sediment core [16]. According to the total pollution index Igeo, there are six pollution levels: uncontaminated (Igeo ≤ 0), uncontaminated to moderately contaminated (0 < Igeo ≤ 1), moderately contaminated (1 < Igeo ≤ 2), moderately to heavily contaminated (2 < Igeo ≤ 3), heavily contaminated (3 < Igeo ≤ 4), heavily to extremely contaminated (4 < Igeo ≤ 5), and extremely contaminated (Igeo > 5).
For both indexes, the concentrations of studied elements in the preindustrial layers of sediments of small lakes in the Republic of Karelia, Russia, were used as the background level [17,18,19].
3. Results and Discussion
3.1. Water Geochemistry
The natural water of the Kola Region is characterized by a low total dissolved solids (TDS) content of 20–30 mg/L and the following pattern of the main ions: HCO3− > SO42− > Cl−; Ca2+ > Na+ > Mg2+ > K+ [20]. In the lakes located close to the Barents Sea, due to the marine aerosols from the Barents Sea, the main ions are distributed as follows: Cl− > HCO3− > SO42−; Na+ > Ca2+ > Mg2+ > K+ [21]. The median pH of the water is 6.5, which lies on the border between the slightly acidic and neutral values.
Over Murmansk’s more than a century-long history, the water chemistry in the city’s lakes has undergone significant changes. The pH value of the lake water in 2018–2019 ranged from 6.6 to 9.34. One of the highest values (almost at the border between the alkaline and strongly alkaline values) was recorded in Lake Ledovoe, which receives water from the Varnichny Stream, running through the densely populated city core. The lake is located alongside the city’s main street—Kola Avenue (Figure 1). High water pH values also were recorded in Lake Yuzhnoe—ranging from 8.02 to 9.11 with an average of 8.42, which is considered alkaline and slightly alkaline. The water of the other lakes had neutral pH values (Table 2). Lake Ledovoe had both the highest pH and the highest TDS value—up to 735 mg/L. Lake Yuzhnoe came second in terms of both pH and TDS values, with TDS ranging from 140 to 371 mg/L with an average of 294 mg/L. TDS values in these lakes were dozens of times higher than the values (20 mg/L on average, which can be considered the background level) recorded in the lakes of the northeastern Murmansk region in the Barents Sea water catchment [21,22]. The lowest TDS values were recorded in Lake Okunevoe and Lake Semenovskoe (63 and 73 mg/L on average, respectively), but these were also above the background level. According to the classification [20], all natural waters by chemical composition are divided in concordance with the prevailing anion (equivalents) into three classes: hydrocarbonate (and carbonate) (HCO3− + CO32−), sulfate (SO42−), and chloride (Cl−). Each class according to the prevailing cation is divided into three groups: calcium (Ca2+), magnesium (Mg2+), and sodium (Na+). According to the classification proposed in [23], the lake water in the city of Murmansk is mainly chloride and sodium. The exceptions are Lake Okunevoe and Lake Srednee, where the lake water is typical for most lakes in the Murmansk region, which have a distribution pattern of the main ions in the calcium group and hydrocarbon class (Table 2). A ternary diagram is constructed for the basic cations and anions, which shows the two groups of lakes by the predominant ions—chloride–sodium and hydrocarbonate–calcium (Figure 3). The studied lakes have supply characteristics in different seasons of the year. In winter, when the lakes are covered with ice, groundwater is the main source of water supply, and in summer, atmospheric waters are the main source. Therefore, the mineralization of water in lakes in winter is more than in summer. In this article, the seasonal features of the chemical composition of the water of the lakes are not considered in detail; another article will be devoted to this.

Table 2.
Average hydrochemical parameters of the Murmansk lakes.
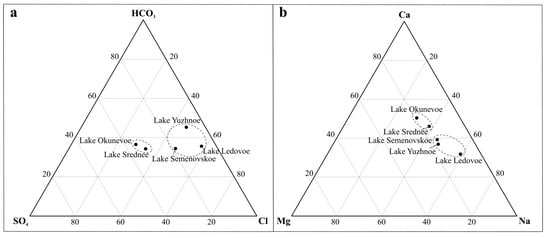
Figure 3.
Chart of the concentration ratio of the prevailing anions (a) and prevailing cations (b) in the chemical composition of the water of the lakes of Murmansk (dotted lines indicate lakes with a single class of water)
The highest content of the nitrogen group (ammonium ion and total nitrogen) was recorded in the water of Lake Ledovoe, which is associated with the influx of wastewater from the city containing high concentrations of nitrogen compounds and with the decomposition of petroleum products, which the sediments of the lake is rich in (Table 2). Oil and petroleum products are known to contain nitrogen in the amount of tenths of a percent [24]. The content of NH4+ in Lake Ledovoe was equal to the content of the main cation K+ in terms of equivalent concentration (248 µeq/L). The highest content of NH4+ in Lake Ledovoe (6390 µgN/L in the bottom layers at a depth of 13 m) was more than 300 times higher than the average content of this ion (16 µgN/L) in the lakes of the northeastern Murmansk region in the Barents Sea catchment [21]. The high levels of NH4+ in the water of Lake Ledovoe are indicative of severe pollution [25]. In the other lakes, the level of ammonium ion was much lower, but still higher than the average level in the lakes of the Barents Sea catchment. The content of nitrate ion in the water of Lake Ledovoe was three orders of magnitude lower (Table 2) than that of ammonium ion, which indicates the presence of a reducing environment and decomposition of plant residues and organic substances. A similar pattern (ammonium ion higher than nitrate ion) was also observed in Lake Yuzhnoe and Lake Semenovskoe, which indicates the development of a reducing environment in the bottom water layers of the lakes. During water sampling in the rather deep Murmansk’s urban lakes (Table 1), summer and winter thermal stratification occurred in July and April, respectively, when the denser (colder in summer and relatively warmer in winter) waters are located in the deep layers of the lake water column. The thermal and density stratification does not allow mixing of the lake water column; therefore, a reducing environment with high NH4+ concentrations is formed in deep water layers as a result of the decomposition of high loads of organic matter. The smell of hydrogen sulfide emanating during the sediment sampling confirms the conclusion about the reducing environment in the deep layers of the water column. Massive development of cyanoprokaryotes was noted in Lake Ledovoe in the summer, which causes an increase in the water pH value. The pH value in the surface water layer in July 2019 was 9.34, and 7.18 in the bottom layer.
High content of phosphorus compounds (phosphate ion and total phosphorus in filtered and unfiltered water) was also observed in the water of Lake Ledovoe (Table 2), which is associated with the influx of urban wastewater containing phosphorus compounds. PO43− in the bottom layers at a depth of 13 m in this lake was up to 65 µgP/L, and total phosphorus was up to 1677 µgP/L, which is one to two orders of magnitude higher than the average (2 and 9 µgP/L, respectively) in the lakes of the northeastern Murmansk region in the Barents Sea catchment [21]. Slightly higher values of these indicators were also recorded in Lake Yuzhnoye and Lake Semenovskoe (Table 2). High phosphorus concentrations (especially PO43−), as well as high NH4+ concentrations, suggests an anoxic hypolimnion in the water of Lake Ledovoe.
The organic matter content in the water of the studied urban lakes (Table 2) was less than the average in the lakes of the northeastern Murmansk region in the Barents Sea catchment [21] 62 Pt°, 9.5 mg/L, and 7.7 mgC/L for Color, CODMn, and TOC, respectively, except for Lake Okunevoe and Lake Ledovoe, where the average value of TOC in the water was not less than the background value. This is probably due to the fact that the studied lakes receive surface runoff from urban areas, to a lesser extent covered by a soil and plant layer compared to natural areas, which reduces the inflow of allochthonous organic matter. This explanation is supported by the observation that the highest organic matter levels, comparable with the above average indicators of the lakes in the Barents Sea catchment, were found in Lake Okunevoe, the least exposed to pollution. Concerning its Color and TOC, Lake Okunevoe has surrounding forest and is similar to the polyhumic forest lakes of the boreal forest regions of Finland, on which much has been published [26,27].
In the water of Lake Ledovoe, the highest concentrations of heavy metals Ni, Cu Zn, Co, Mo, and W were found (Table 3). Judging by the main ions, biogenic elements, and heavy metals, this lake is most exposed to anthropogenic impacts and is the most polluted of all the investigated lakes. In the water of Lake Okunevoe, one of the relatively clean lakes among the studied ones, the highest concentrations of Cd and Pb were found. With rare exceptions, the levels of all heavy metals in the water of urban lakes are higher than the average content in the lakes of the Barents Sea catchment [22], and especially in the lakes of Northern Fennoscandia [28].

Table 3.
Average concentrations (µg/L) of trace elements in water of the Murmansk lakes and lakes of the northeastern part the Barents Sea watershed [22,28].
The highest V level was found in the water of Lake Srednee and Lake Okunevoe (Table 3), the closest to the Murmansk thermal power plant, which is fed with fuel oil containing high concentrations of this element. The highest levels of Cd and Pb in Lake Okunevoe can also be associated with the emissions of the Murmansk thermal power plant.
3.2. Sediments Geochemistry
Sediments in all the studied lakes in Murmansk, with the exception of Lake Yuzhnoe, according to the organic matter content (based on LOI), are classified as organosilicate deposits [29] The sediments of Lake Yuzhnoe are classified as mineral deposits. The highest organic matter levels were recorded in the sediment of Lake Okunevoe (45%) and Lake Semenovskoe (40%). In the sediments of Lake Srednee, the organic matter content was 26% of the total mass, while in Lake Ledovoe and Lake Yuzhnoe, it was 15% and 10%, respectively.
The water pollution level in the lakes of Murmansk, as determined based on a combination of indicators, is also reflected in the condition of the sediments of these water bodies. The total content of almost all heavy metals in the sediments of the lakes of Murmansk exceeds the background level, which is interpreted as the content of the same elements in the preindustrial layers of the sediments of small lakes in the Republic of Karelia, Russia [17,18,19], located to the south of the Murmansk region (Figure 4). Only the background level of Mn is higher or at the same level as in the investigated urban lakes, which may be due to the natural accumulation of this metal in sediments. Concentrations of other heavy metals significantly exceed the background level.
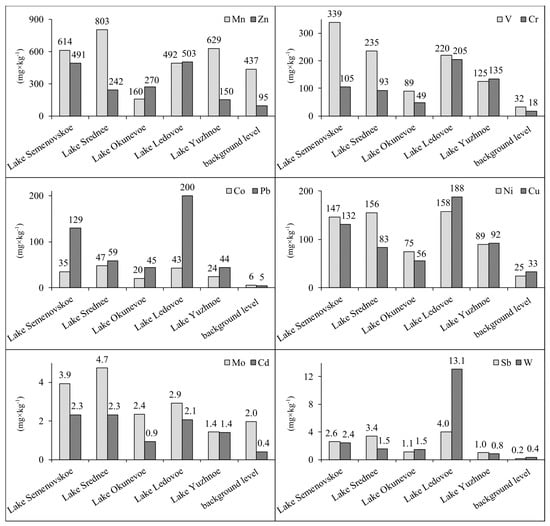
Figure 4.
The total concentration of heavy metals in sediments of lakes of Murmansk in comparison with the background level.
The highest accumulated levels of heavy metals were found in the sediments from Lake Ledovoe, where the background level was exceeded by a factor of 23 for W, 20 for Pb, 17 for Sb, 12 for Cr, 9 for Co, 7 for Ni, 6 for V, 5 for Zn, Cu, and Cd, and 2 for Mo. In the sediments of this lake the pollution load index is 7.6, which corresponds to an extremely heavy pollution level. This is largely due to the presence in the immediate vicinity of the lake of two gas stations, several car repair shops, car dealerships, a parking lot, a granite workshop, a public transit stop, and several garage buildings.
In the sediments of Lake Semenovskoe, located in a recreational area of Murmansk and surrounded by busy streets, the background level was exceeded by a factor of 13 for Pb, 11 for Sb, 10 for V, 8 for Co, 7 for Ni, 6 for Cr, 5 for Cd and Zn, 4 for Cu and W, and 2 for Mo. In the sediments of this lake the pollution load index is 6.0, which corresponds to an extremely heavy pollution level.
In the sediments of Lake Srednee, the background level was exceeded by a factor of 14 for Sb, 10 for Co, 7 for V and Ni, 6 for Pb, 5 for Cd and Cr, 3 for W and Zn, and 2 for Mo and Cu. In the sediments of this lake, the pollution load index is 5.1, which corresponds to an extremely heavy pollution level. Lake Srednee is located near a busy road and thermal power plant, which can explain the high pollution level in the water body.
In the sediments of Lake Yuzhnoe, the background level was exceeded by a factor of 8 for Cr, 5 for Co, 4 for Pb, Ni, Sb, and V, 3 for Cd and Cu, and 2 for Zn. In the sediments of this lake, the pollution load index is 3.1, which corresponds to an extremely heavy pollution level. Local sources of lake pollution can be the gas station, several car repair shops, and the many warehouse buildings located near the lake. On the other hand, the lake is located on the outskirts of Murmansk and therefore is exposed to a lower pollution load compared to lakes Ledovoe, Semenovskoe, and Srednee.
In Lake Okunevoe, located in the center of the city, the background level was exceeded by a factor of 5 for Pb and Sb, 4 for Co, 3 for Ni, Zn, Cr, V, W, and 2 for Cd and Cu. In the sediments of this lake, the pollution load index is 2.6, which corresponds to a heavy pollution level. This is the lowest value of the index among all the lakes studied. This is primarily due to the fact that the lake is surrounded by forest, located away from busy roads, and there is only a camp site, medical facilities, and several garage buildings on its shores.
As indicated in the state report on the state of the environment in the Murmansk region, emissions from Murmansk thermal power plant (TPP) are the main source of air pollution across the region and in the city of Murmansk. In 2017, the power plant emitted 14.1 thousand tons of pollutants and 15.31 thousand tons in 2018. Until 1964, the power plant was coal fired, but in the 1960s the gradual transition of the Murmansk TPP to fuel oil began. Fuel oil is still the main type of fuel used by the Murmansk TPP. Emissions from any fuel oil-based thermal power plant are known to have elevated levels of Fe, V, Ni, Cr, and other elements [30]. This can explain the elevated levels of V, Ni, and Cr in the water and sediments of the urban lakes in Murmansk, especially in lakes Semenovskoe, Srednee, Ledovoe, Okunevoe, located within the direct impact zone of the TPP emissions in the city’s urban core.
On the charts of the vertical distribution of heavy metals in the core sediments of Lake Semenovskoe, it is shown that the concentrations of almost all elements in the upper layers of the sediments are higher than in the lower layers (Figure 5). A particularly sharp increase in metals was noted at a depth of 16 cm. It is here that a smooth increase in the concentrations of V and Ni in the studied sediments begins, which can be taken as the beginning of the operation of a thermal power plant using fuel oil in the 1960s. Given that the company still uses this type of fuel, constantly increasing capacity due to the needs of the city and urban factories, the concentrations of V and Ni are still increasing. Thus, the average sedimentation rate in the lake during the past 55 years is about 2.9 mm per year. It is higher than the average sedimentation rate in the small lakes of the Murmansk region, which varies from 0.03 to 1.25 mm per year. Similar behavior of these metals is noted in other lakes of the city of Murmansk. In addition, similar patterns were previously revealed in the study of sediments of a small urban lake in the Republic of Karelia (Russia), which is also subject to the influence of TPP emissions [31].
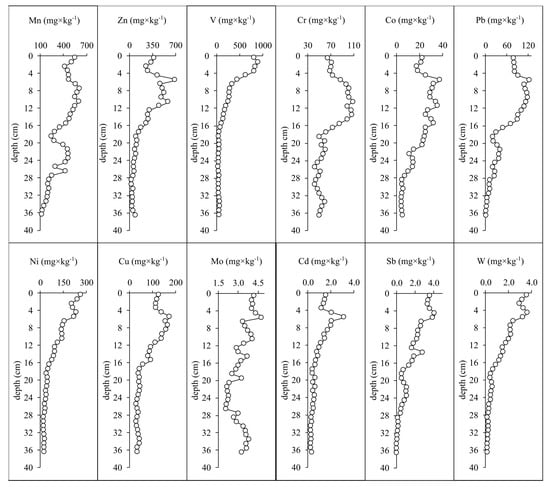
Figure 5.
Vertical distribution of heavy metals in the core sediments of the Lake Semenovskoe.
Significant Pb pollution is observed in almost all lakes, which is due to the use of tetraethyl lead as a gas additive worldwide between the 1930s and 1990s [32,33]. In the recent sediments, the vertical distribution of Pb records the history of anthropogenic impacts on the aquatic environment and on the catchment area of the water body [34,35,36,37]. The vertical distribution of Pb in the core sediments of Lake Semenovskoe confirms the conclusions about the accumulation of lead in this period (Figure 5). The decrease in the concentration of this metal in the uppermost layers of sediments of Lake Semenovskoe reveals a decrease in its release into the environment after the abandonment of leaded gasoline in Russia in 2002.
In addition, Pb can also enter the environment with industrial emissions, and its migration range is hundreds and thousands of kilometers [7,38]. Thus, elevated Pb concentrations, as compared to background (natural) levels, are found in lake sediments in the relatively clean areas of the Murmansk region [6], Russian Karelia [19], and Finland [9]. As noted in numerous studies of the behavior of heavy metals in the environment, emissions from the combustion of fossil coal served as the main source of heavy metals at the beginning of the industrial era development of society [39,40,41,42]. Moreover, the trends in the behavior of these elements in the environment coincide, starting from the end of the 19th century to the present. In addition, metals entered and are entering the atmosphere as a result of activities of enterprises for the extraction and processing of metal ores and the incineration of household waste [43,44]. This may explain the increased accumulation of Sb, Cd, and Zn and other metals in the sediments of the urban lakes in Murmansk.
It should also be noted that one of the key sources of pollution in Murmansk is coal dust originating from the commercial port of Murmansk. Many heavy metals (Sb, Cr, Ni, Co, W, Mo) may be contained in coal and in coal dust as impurities, which become environmental pollutants as a result of atmospheric transport. A further detailed study of the vertical distribution of these metals in sediment core of the urban lakes in Murmansk, together with sediment dating, will make it possible to more accurately detect all sources of pollution of the studied lake ecosystems.
Calculation of the geoaccumulation index (Igeo) reveal uncontaminated and moderate pollution levels for Mo and Mn. For Cu, Zn, and Cd, the index shows moderately and heavily contaminated levels for all the lakes. The highest level of pollution is revealed for Pb, Sb, W, V, and Ni. The most polluted lakes of Murmansk are Ledovoe, Semenovskoe, and Srednee, where the index detects heavily and extremely elevated levels of pollution by Pb, Sb, and V. Analysis of the core sediments from Lake Semenovskoe, the most heavily and extremely contaminated level of the sediments is traced from upper layers to a depth of 18 cm (Figure 6).
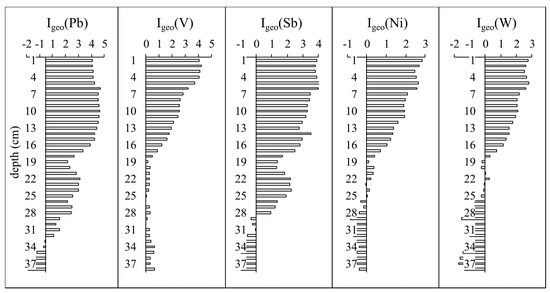
Figure 6.
Values of the geoaccumulation index (Igeo) for Pb, V, Sb, Ni, and W in Lake Semenovskoe sediments.
Increased accumulation of heavy metals in the sediments can initiate secondary pollution of the aquatic environment and the involvement of hazardous chemical elements in biological cycles. For this reason, analyzing the bioavailable forms of metals is an important part of environmental and geochemical studies of urban lakes.
Data on the content of the mobile forms of heavy metals (the sum of water soluble and exchange fractions) in the sediments of the urban lakes in Murmansk demonstrate that the most mobile and bioavailable elements in the lakes are Mn, Co, Zn, and Cd (up to 40% of the total content). However, while in Lake Okunevoe and Lake Srednee there is a gradual increase in content of mobile Co and Zn forms in the more recent sediment, the concentration of mobile forms in the top layers of the core samples (0–5 cm and 5–10 cm) from Lake Semenovskoe decreases significantly to 2–3% (Figure 7). On the other hand, in the sediment from Lake Semenovskoe, the mobility of Cd increases with depth. In Lake Okunevoe there is also a noticeable increase in content of mobile forms of Cd and Ni in the more recent layers of sediment.
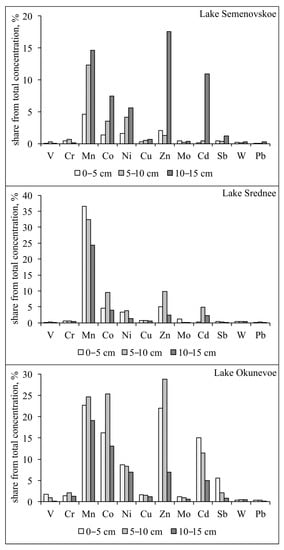
Figure 7.
Share of available (mobile) fractions from total heavy metal concentrations in sediments of Murmansk’s lakes.
V, Cr, Cu, W, Mo, Pb, and Sb are the most strongly associated with residual phases and unbioavailable elements in all studied lakes along the entire depth of the sediment core (not more than 1–3% of the total content). Probably, these elements are bound with organic matter, iron oxides, or finely disseminated mineral particles, which retain pollutants in the sediments of rivers and lakes [2,3,45,46]. Further study of the sediments in the lakes of the city of Murmansk is necessary to give a conclusion concerning this aspect.
4. Conclusions
The following results were obtained in this research of water and sediment of urban lakes of Murmansk:
The water chemistry in the studied urban lakes has undergone significant changes compared to the background water chemistry in the lakes of the Barents Sea catchment. Water pH values are significantly elevated compared to background, which is due to the weathering and decay of urban buildings and structures, as well as the use of reagents for deicing urban roads, and the influx of high concentrations of the main cations of alkali and alkaline-earth metals. Due to anthropogenic impacts, and in particular the influx and decomposition of petroleum products, the content of nitrogen compounds in the water of urban lakes has increased significantly, which has had a detrimental effect on aquatic life. Among the studied lakes, the water of Lake Ledovoe has the highest level of pollution.
In the studied lakes, elevated levels of heavy metals were found in the water and in the top layer (up to 20 cm) of the sediments, as compared to lakes in the cleaner areas of northern Russia. Pb, Sb, V, Cr, and Ni accumulate most actively in the lakes. The pollution load index values for the sediments of the studied lakes reveal an extremely heavy pollution level in lakes Ledovoe (7.6), Semenovskoe (6.0), and Srednee (5.1), and a heavy pollution level in Lake Yuzhnoe (3.1) and Lake Okunevoe (2.6). According to Igeo, the highest level of pollution in the studied lakes was revealed for Pb, Sb, W, Sb, and Ni. Data on the content of the mobile forms of heavy metals in the sediments of the urban lakes in Murmansk demonstrated that the most mobile and bioavailable elements in the lakes are Mn, Co, Zn, and Cd (up to 40% of the total content).
The main sources of pollution, including heavy metals, in the lakes of the city of Murmansk are emissions from the thermal power plant, which is primarily responsible for the elevated levels of V, Ni, and Cr in the water and sediments, and emissions from road and rail transport and the Murmansk coal port. The elevated Pb levels in all of the urban lakes studied are likely associated with the long-term use of tetraethyl Pb as an additive to automotive fuel. Another possible source of the heavy metals entering the aquatic ecosystems of Murmansk may be the long-range atmospheric transport of pollutants from the numerous industrial sites in the Murmansk region and neighboring regions, including cross-border transport.
Author Contributions
Conceptualization, Z.S., V.D., and D.D.; methodology, Z.S., V.D., and A.G.; software, A.C. and E.S.; validation, V.D. and D.D.; investigation and resources, Z.S., D.D., A.G., and A.C.; data curation, Z.S.; writing-original draft preparation, Z.S., V.D., and A.G.; writing-review and editing, V.D. and D.D.; visualization, Z.S., A.C., and E.S.; supervision, V.D.; project administration and funding acquisition, Z.S. All authors have read and agreed to the published version of the manuscript.
Funding
The research is supported by the Russian Science Foundation (project No. 19-77-10007) and Russian Foundation for Basic Research (project No. 18-05-00897). The study of the chemical composition of water and sediments was carried out with a grant from the Russian Science Foundation. Analysis of mobile forms of metals was carried out with a grant from the Russian Foundation for Basic Research.
Acknowledgments
The authors sincerely thank the colleagues from Petrozavodsk and Apatity: V.L. Utitsina, M.V. Ekhova, A.S. Paramonov, Kudryavczeva L.P., Malysheva M.B., and Pavlova A.S. for conducting high-quality analytical studies.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chin, A. Urban transformation of river landscapes in a global context. Geomorphology 2006, 79, 460–487. [Google Scholar] [CrossRef]
- Remor, M.B.; Sampaio, S.C.; deRijk, S.; Boas, M.A.V.; Gotardo, J.T.; Pinto, E.T.; Schardong, F.A. Sediment geochemistry of the urban Lake Paulo Gorski. Int. J. Sediment Res. 2018, 33, 406–414. [Google Scholar] [CrossRef]
- Förstner, U.; Heise, S.; Schwartz, R.; Westrich, B.; Ahlf, W. Historical contaminated sediments and soils at the river basin scale. Examples from the Elbe River catchment area. J. Soils Sediments 2004, 4, 247–260. [Google Scholar] [CrossRef]
- Moiseenko, T.I. Impact of geochemical factors of aquatic environment on the metal bioaccumulation in fish. Geochem. Int. 2015, 53, 213–223. [Google Scholar] [CrossRef]
- Wijaya, A.R.; Ouchi, A.K.; Tanaka, K.; Cohen, M.D.; Sirirattanachai, S.; Shinjo, R.; Ohde, S. Evaluation of heavy metal contents and Pb isotopic compositions in the Chao Phraya River sediments: Implications for anthropogenic inputs from urbanized areas, Bangkok. J. Geochem. Explor. 2013, 126–127, 45–54. [Google Scholar] [CrossRef]
- Dauvalter, V.; Kashulin, V.; Sandimirov, S.; Terentjev, P.; Denisov, D.; Amundsen, P.-A. Chemical composition of lake sediments along a pollution gradient in a Subarctic watercourse. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2011, 46, 1020–1033. [Google Scholar] [CrossRef]
- Norton, S.; Dillon, P.; Evans, R. The history of atmospheric deposition of Cd, Hg and Pb in North America: Evidence from lake and peat bog sediments. Sources, Deposition and Capony Interactions. In V. III, Acidic Precipitation; Springer: New York, NY, USA, 1990; pp. 73–101. [Google Scholar] [CrossRef]
- Bartnicki, J. An Eulerian model for atmospheric transport of heavy metals over Europe: Model description and preliminary results. Water Air Soil Pollut. 1994, 75, 227–263. [Google Scholar] [CrossRef]
- Verta, M.; Tolonen, K.; Simola, H. History of heavy metal pollution in Finland as recorded by lake sediments. Sci. Total Environ. 1998, 87, 1–18. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Dauvalter, V.A.; Lukin, A.A.; Kudryavtseva, L.P.; Ilyashuk, B.P.; Ilyashuk, E.A.; Sandimirov, S.S.; Kagan, L.Y.; Vandysh, O.I.; Sharov, A.N.; et al. Anthropogenic Modifications of the Ecosystem of Lake Imandra; Nauka: Moscow, Russia, 2002; 487p. (In Russian) [Google Scholar]
- Standard Methods for Examination for Water and Wastewater, 20th ed.; Clescerl, L.S., Greenberg, A.E., Eaton, A.D., Eds.; American Public Health Association USA: Washington, DC, USA, 1999; 2671p. [Google Scholar]
- Intercomparison–2016 (2016) 1630: pH, Conductivity, Alkalinity, NO3-N, Cl, SO4, Ca, Mg, Na, K, TOC, Al, Fe, Mn, Cd, Pb, Cu, Ni and Zn. ICP Waters report 129/2016; Report No. 7081,72 p; Norwegian Institute for Water Research: Oslo, Norway, 2016.
- Tessier, A.; Campbell, P.G.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Suresh, G.; Ramasamy, V.; Meenakshisundaram, V.; Venkatachalapathy, R.; Ponnusamy, V. Influence of mineralogical and heavy metal composition on natural radionuclide concentrations in the river sediments. Appl. Radiat. Isot. 2011, 69, 1466–1474. [Google Scholar] [CrossRef]
- Tomlinson, D.; Wilson, J.; Harris, C.; Jeffrey, D. Problems in the assessment of heavy metal levels in estuaries and the formation of a pollution index. Helgoländer. Meeresun 1980, 33, 566–575. [Google Scholar] [CrossRef]
- Müller, G. Heavy metals in the sediments of the Rhine.—Changes since 1971. Umsch. Wiss. Tech. 1979, 79, 778–783. (In German) [Google Scholar]
- Slukovskii, Z.I. Background concentrations of heavy metals and other chemical elements in the sediments of small lakes in the south of Karelia, Russia. Vestn. Mstu 2020, 23, 80–92. [Google Scholar] [CrossRef]
- Medvedev, A.; Slukovskii, Z.; Novitcky, D. Heavy Metals Pollution of Small Urban Lakes Sediments within the Onego Lake Catchment Area. Pol. J. Nat. Sci. 2019, 34, 245–256. [Google Scholar]
- Slukovskii, Z.I.; Medvedev, M.A.; Siroezhko, E.V. Long-range heavy metal aerosols transport as a factor of the formation of the geochemical characteristics of current lake bottom sediments from the southwestern republic of Karelia (exemplified by lake Ukonlampi, Lahdenpohja district). J. Elem. 2020, 25, 125–137. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Rodushkin, I.V.; Dauvalter, V.A.; Kudryavtzeva, L.P. Quality Formation of Surface Waters and Bottom Sediments under Conditions of Anthropogenic Loads on Water Catchments within the Arctic Area; Kola Science Centre RAS: Apatity, Russia, 1996; 263p. (In Russian) [Google Scholar]
- Kashulin, N.A.; Sandimirov, S.S.; Dauvalter, V.A.; Kudryavtzeva, L.P.; Terentjev, P.M.; Denisov, D.B.; Valkova, S.A. Annotated Ecological Catalogue of Lakes in the Murmansk Region: East Area of the Murmansk Region (Basin of the Barents Sea); Kola Science Centre RAS: Apatity, Russia, 2010; 249p. [Google Scholar]
- Bazova, M.M. Specifics of the elemental composition of waters in environments with operating mining and ore-processing plants in the Kola North. Geochem. Int. 2017, 55, 131–143. [Google Scholar] [CrossRef]
- Alekin, O.A. Fundamentals of Hydrochemistry; Hydrometeoizdat: Leningrad, Russia, 1970; 444p. (In Russian) [Google Scholar]
- Bolshakov, G.F. Nitrogen-Organic Compounds of Oil; Nauka: Novosibirsk, Russia, 1988. (In Russian) [Google Scholar]
- Nikanorov, A.M. Hydrochemistry; Gidrometeoizdat: Saint-Petersburg, Russia, 2000; 444p. (In Russian) [Google Scholar]
- Asmala, E.; Carstensen, J.; Räike, A. Multiple anthropogenic drivers behind upward trends in organic carbon concentrations in boreal rivers. Environ. Res. Lett. 2019, 14, 124018. [Google Scholar] [CrossRef]
- Kauppila, T.; Ahokas, T.; Nikolajev-Wikström, L.; Mäkinen, J.; Tammelin, M.H.; Meriläinen, J.J. Aquatic effects of peat extraction and peatland forest drainage: A comparative sediment study of two adjacent lakes in Central Finland. Environ. Earth Sci. 2016, 75, 1473. [Google Scholar] [CrossRef]
- Skjelkvåle, B.L.; Andersen, T.; Fjeld, E.; Mannio, J.; Wilander, A.; Johansson, K.; Jensen, J.P.; Moiseenko, T.I. Heavy Metal Surveys in Nordic Lakes. Concentrations, Geographic Patterns and Relation to Critical Limits. AMBIO 2001, 30, 2–10. [Google Scholar]
- Stankevica, K.; Vincevica-Gaile, Z.; Klavins, M. Freshwater sapropel (gyttja): Its description, properties and opportunities of use in contemporary agriculture. Agron. Res. 2016, 14, 929–947. [Google Scholar]
- Teng, Y.; Ni, S.; Zhang, C.; Wang, J.; Lin, X.; Huang, Y. Environmental geochemistry and ecological risk of vanadium pollution in Panzhihua mining and smelting area, Sichuan, China. Chin. J. Geochem. 2006, 25, 379–385. [Google Scholar] [CrossRef]
- Slukovskii, Z.I.; Svetov, S.A. Geochemical indicators of technogenic pollution of bottom sediments of small rivers in an urbanized environment. Geogr. Nat. Resour. 2016, 37, 32–38. [Google Scholar] [CrossRef]
- Nriagu, J.O. The rise and fall of leaded gasoline. Sci. Total Environ. 1990, 92, 13–28. [Google Scholar] [CrossRef]
- Thomas, V. The elimination of lead in gasoline. Annu. Rev. Energy Environ. 1995, 20, 301–324. [Google Scholar] [CrossRef]
- Keinonen, M. The isotopic composition of lead in man and the environment in Finland 1966–1987: Isotope ratios of lead as indicators of pollutant source. Sci. Total Environ. 1992, 113, 251–268. [Google Scholar] [CrossRef]
- Dauvalter, V.; Kashulin, N. Chalcophile elements (Hg, Cd, Pb, As) in lake Umbozero, Murmansk Province. Water Resour. 2010, 37, 497–512. [Google Scholar] [CrossRef]
- Escobar, J.; Whitmore, T.J.; Kamenov, G.D.; Riedinger-Whitmore, M.A. Isotope record of anthropogenic lead pollution in lake sediments of Florida, USA. J. Paleolimnol. 2013, 49, 237–252. [Google Scholar] [CrossRef]
- Hosono, T.; Alvarez, K.; Kuwae, M. Lead isotope ratios in six lake sediment cores from Japan Archipelago: Historical record of trans-boundary pollution sources. Sci. Total Environ. 2016, 559, 24–37. [Google Scholar] [CrossRef]
- Vinogradova, A.; Kotova, E.; Topchaya, V. Atmospheric transport of heavy metals to regions of the North of the European territory of Russia. Geogr. Nat. Resour. 2017, 38, 78–85. [Google Scholar] [CrossRef]
- McConnell, J.R.; Edwards, R. Coal burning leaves toxic heavy metal legacy in the Arctic. Proc. Natl. Acad. Sci. USA 2008, 34, 12140–12144. [Google Scholar] [CrossRef]
- McConnell, J.R.; Chellman, N.J.; Wilson, A.I.; Stohl, A.; Arienzo, M.M.; Eckhardt, S.; Steffensen, J.P. Pervasive Arctic lead pollution suggests substantial growth in medieval silver production modulated by plague, climate, and conflict. Proc. Natl. Acad. Sci. USA 2019, 116, 14910–14915. [Google Scholar] [CrossRef] [PubMed]
- Krachler, M.; Zheng, J.; Koerner, R.; Zdanowicz, C.; Fisher, D.; Shotyk, W. Increasing atmospheric antimony contamination in the northernhemisphere: Snow and ice evidence from Devon Island, Arctic Canada. J. Environ. Monit. 2006, 7, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Pacyna, J.M.; Pacyna, E.G. An assessment of global and regional emissions of trace metals to the atmosphere from anthropogenic sources worldwide. Environ. Rev. 2001, 9, 269–298. [Google Scholar] [CrossRef]
- Cooke, C.A.; Abbott, M.B. A paleolimnological perspective on industrial-era metal pollution in the central Andes, Peru. Sci. Total Environ. 2008, 393, 262–272. [Google Scholar] [CrossRef]
- Kuwae, M.; Tsugeki, N.K.; Agusa, T.; Toyoda, K.; Tani, Y.; Ueda, S.; Tanabe, S.; Urabe, J. Sedimentary records of metal deposition in Japanese alpine lakes for the last 250 years: Recent enrichment of airborne Sb and In in East Asia. Sci. Total Environ. 2013, 442, 189–197. [Google Scholar] [CrossRef]
- Li, X.; Shen, Z.; Wai, O.W.H.; Li, Y.-S. Chemical forms of Pb, Zn and Cu in the sediment profiles of the Pearl River Estuary. Mar. Pollut. Bull. 2001, 42, 215–223. [Google Scholar] [CrossRef]
- López, D.L.; Gierlowski-Kordesch, E.; Hollenkamp, C. Geochemical Mobility and bioavailability of heavy metals in a lake affected by acid mine drainage: Lake Hope, Vinton County, Ohio. Water Air Soil Pollut. 2010, 213, 27–45. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).