Carabus Population Response to Drought in Lowland Oak Hornbeam Forest
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. The Occurrence of the Carabus Species
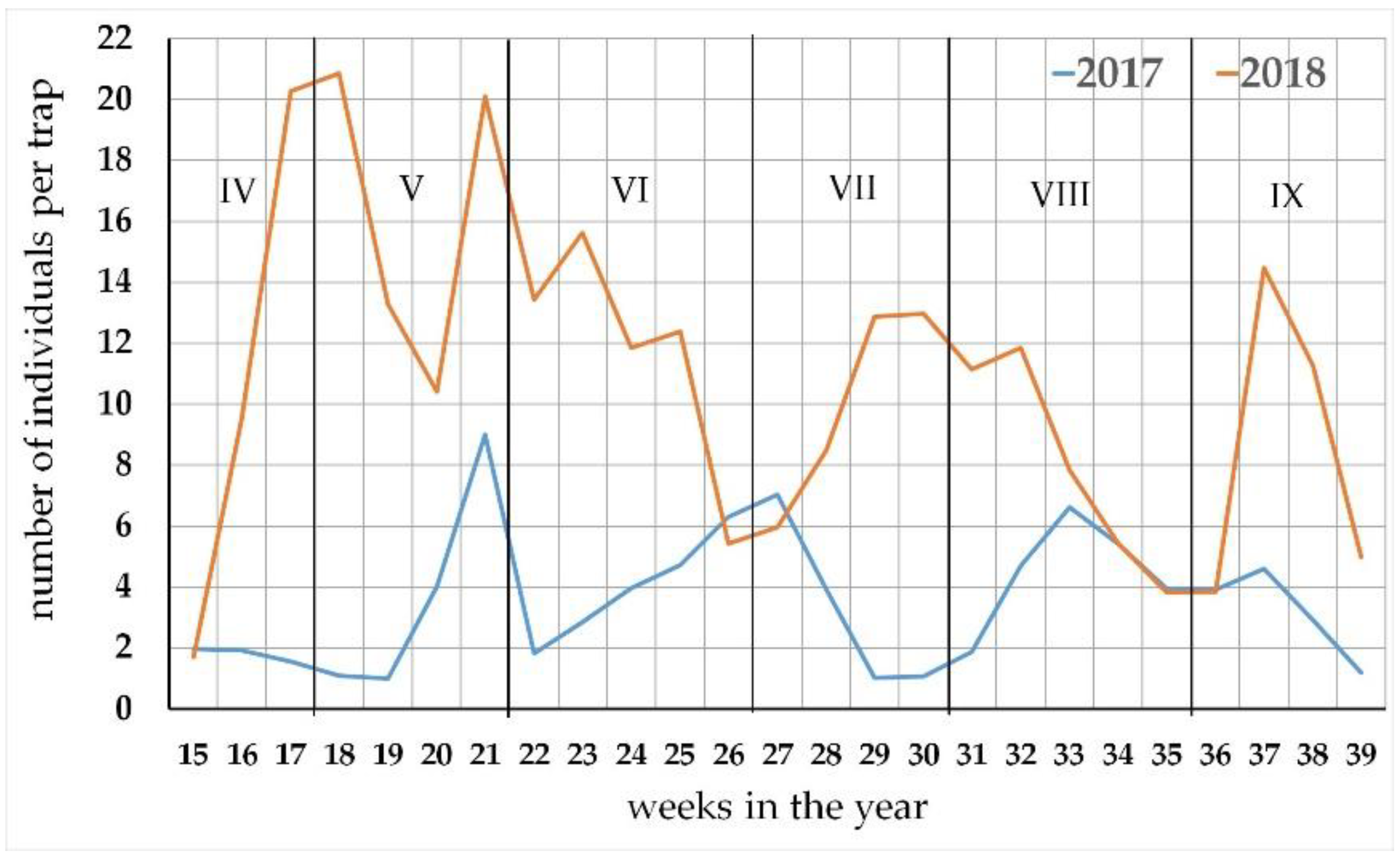
3.2. The Seasonal Activity of the Carabus Species
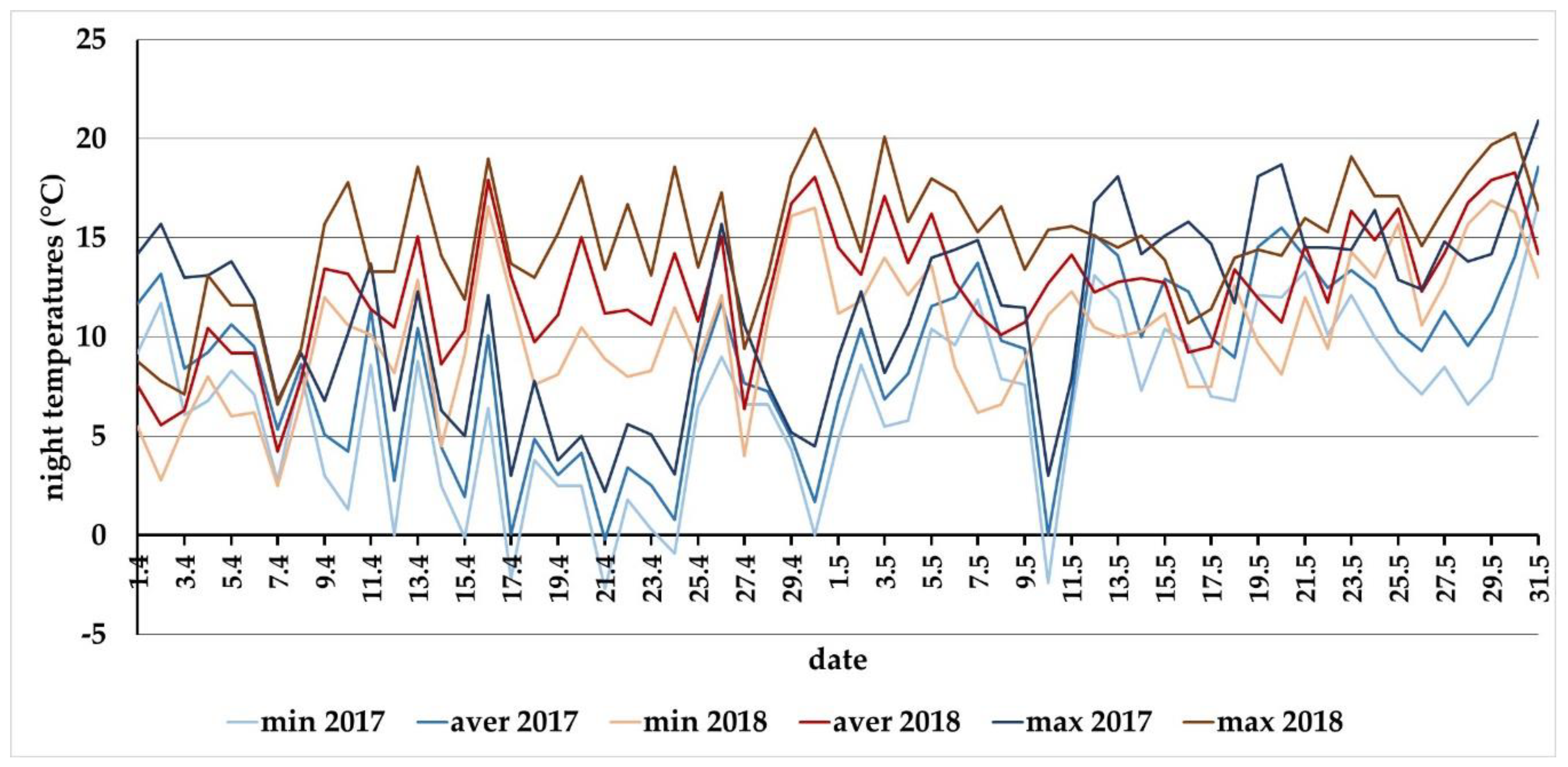
3.3. Meteorological Conditions in 2017 and 2018 for the Locality of Báb
3.4. The Drought Occurrence at the Locality of Báb between 2015 and 2018
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A

References
- Pearce, J.L.; Venier, L.A. The use of ground beetles (Coleoptera: Carabidae) and spiders (Araneae) as bioindicators of sustainable forest management: A review. Ecol. Indic. 2006, 6, 780–793. [Google Scholar] [CrossRef]
- Kotze, D.J.; Brandmayr, P.; Casale, A.; Dauffy-Richard, E.; Dekoninck, W.; Koivula, M.J.; Lövei, G.L.; Mossakowski, D.; Noordijk, J.; Paarmann, W.; et al. Forty years of carabid beetle research in Europe—From taxonomy, biology, ecology and population studies to bioindication, habitat assessment and conservation. ZooKeys 2011, 100, 55–148. [Google Scholar] [CrossRef]
- Niemelä, J.; Langor, D.; Spence, J.R. Effects of clear-cut harvesting on boreal ground-beetle assemblages (Coleoptera: Carabidae) in Western Canada. Conserv. Biol. 1993, 7, 551–561. [Google Scholar]
- Skłodowski, J. Anthropogenic transformation of ground beetle assemblages (Coleoptera: Carabidae) in Białowieża Forest, Poland: From primeval forests to managed woodlands of various ages. Entomol. Fenn. 2006, 11, 296–314. [Google Scholar] [CrossRef]
- Šustek, Z.; Vido, J. Vegetation state and extreme drought as factors determining differentiation and succession of Carabidae communities in forests damaged by a windstorm in the High Tatra Mts. Biologia 2013, 68, 1198–1210. [Google Scholar] [CrossRef]
- Walther, G.; Post, E.; Convey, P.; Menzel, A.; Parmesan, C.; Beebee, T.J.C.; Fromentin, J.; Hoegh-Guldberg, O.; Bairlein, F. Ecological responses to recent climate change. Nature 2002, 416, 389–395. [Google Scholar] [CrossRef]
- Thackeray, S.J.; Sparks, T.H.; Frederiksen, M.; Burthe, S.; Bacon, P.J.; Bell, J.R.; Botham, M.S.; Brereton, T.M.; Bright, P.W.; Carvalho, L.; et al. Trophic level asynchrony in rates of phenological change for marine, freshwater and terrestrial environments. Glob. Chang. Biol. 2010, 16, 3304–3313. [Google Scholar] [CrossRef]
- Thiele, H.U. Carabid Beetles in Their Environments. In A Study on Habitat Selection by Adaptations in Physiology and Behaviour; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1977; p. 369. ISBN 978-3-642-81154-8. [Google Scholar]
- Lovei, G.L.; Sunderland, K.D. Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu. Rev. Entomol. 1996, 41, 231–256. [Google Scholar]
- Loreau, M. Competition in a carabid beetle community: A field experiment. Oikos 1990, 58, 25–38. [Google Scholar]
- IPCC. Climate Change 2001: Impacts, Adaptation and Vulnerability—Contribution of Working Group II to the Third Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2001. [Google Scholar]
- Lehner, B.; Döll, P.; Alcamo, J.; Henrichs, T.; Kaspar, F. Estimating the impact of globalchange on flood and drought risk in Europe: A continental, integrated analysis. Clim. Chang. 2006, 75, 273–299. [Google Scholar] [CrossRef]
- Duan, W.; Hanasaki, N.; Shiogama, H.; Chen, Y.; Zou, S.; Nover, D.; Zhou, B.; Wang, Y. Evaluation and Future Projection of Chinese Precipitation Extremes Using Large Ensemble High-Resolution Climate Simulations. J. Clim. 2017, 32, 2169–2183. [Google Scholar] [CrossRef]
- Haile, G.G.; Tang, Q.; Hosseini-Moghari, S.M.; Liu, X.; Gebremicael, T.G.; Leng, G.; Kassa, A.K.; Xu, X.; Yun, X. Projected impacts of climate change on drought patterns over East Africa. Earth’s Future 2020, 8, e2020EF001502. [Google Scholar] [CrossRef]
- Škvarenina, J.; Tomlain, J.; Hrvoľ, J.; Škvareninová, J. Occurrence of dry and wet periods in altitudinal vegetation stages of West Carpathians in Slovakia: Time-series analysis 1951–2005. In Bioclimatology and Natural Hazards; Střelcová, K., Ed.; Springer: Dordrecht, The Netherlands, 2019; pp. 97–106. [Google Scholar]
- Zlatník, A. Přehled Skupin Typů Geobiocenů Původne Lesních a Křovinných v ČSSR (Overview of the Groups of the Geobiocen Types Originally Forests and Shrubs in ČSSR); Zprávy geografického ústavu ČSAV (Reports of the Institute of geography of the Czechoslovak academy of science), 13; The Institute of Geography of the Czechoslovak Academy of Science: Brno, Czech Republic, 1976; pp. 55–64. (In Czech) [Google Scholar]
- Škvarenina, J.; Tomlain, J.; Hrvoľ, J.; Škvareninová, J.; Nejedlík, P. Progress in dryness and wetness parameters in altitudinal vegetation stages of West Carpathians: Time-series analysis 1951–2007. Idojárás 2009, 113, 47–54. [Google Scholar]
- Atlas Krajiny Slovenskej Republiky (Atlas of the Slovak Republic Landscape). Available online: https://app.sazp.sk/atlassr/ (accessed on 20 October 2020).
- Kurpelová, M.; Coufal, L.; Čulík, J. Agroklimatické podmienky ČSSR (Agroclimatic conditions CSSR); Príroda: Bratislava, Slovakia, 1975; p. 270. [Google Scholar]
- Šiška, B.; Špánik, F. Agroclimatic regionalization of slovak territory in condition of changing climate. Meteorol. Časopis 2008, 11, 61–64. [Google Scholar]
- Šiška, B.; Takáč, J. Drought analyse of agricultural regions as influenced by climatic conditions in the Slovak Republic. Időjárás 2009, 113, 135–143. [Google Scholar]
- Eliáš, P. Species pool of alien plants in the vicinity of Bab’s Forest Research Site, south-west Slovakia. Rosalia 2010, 21, 57–74. [Google Scholar]
- Biskupský, V. (Ed.) Research Project Báb IBP Progress Report II. IBP Report No. 5; Publishing House of the Slovak Academy of Sciences: Bratislava, Slovakia, 1975; p. 526. [Google Scholar]
- Eliáš, P.; Oszlányi, J.; Matušicová, N.; Gerhátová, K.; Halada, L. Oak-Hornbeam Forest in Báb (South-Western Slovakia)—The Former Research Site of International Biological Programme in Slovakia. Životné prostredie 2016, 50, 10–17. [Google Scholar]
- Bab—Slovakia. Available online: https://deims.org/79e10639-dd60-4f30-9c43-7b2bae0f359a (accessed on 5 November 2020).
- Hůrka, K. Střevlíkovití České a Slovenské Republiky, Carabid Beetles of the Czech and Slovak Republic, 1st ed.; Kabourek: Zlín, Czech Republic, 1996; p. 565. ISBN 80-901466-2-7. (In Czech and English). [Google Scholar]
- Šiška, B.; Žilinský, M.; Zuzulová, V. Drought and growing season on Slovakia in climate change conditions. In Hospodaření s Vodou v Krajině, Proceedings of the Conference Hospodaření s Vodou v Krajině, Třeboň, Czech Republic, 13–14 June 2019; Rožnovský, J., Litschmann, T., Eds.; ČHMU: Praha, Czech Republic, 2019; ISBN 978-80-87577-88-2. [Google Scholar]
- Cunev, J.; Šiška, B. Chrobáky (Coleoptera) NPR Bábsky les pri Nitre v podmienkach meniacej sa klímy (Beetles (Coleoptera) NNR Bábsky les near Nitra in the conditions of the changing climate). Rosalia 2006, 18, 155–168, (In Slovak, abstract in English). [Google Scholar]
- Turin, H.; Penev, L.; Casale, A. (Eds.) The Genus Carabus in Europe. A Synthesis; Pensoft Publishers, Sofia-Moscow & European Invertebrate Survey: Leiden, The Netherlands, 2003; p. 512. [Google Scholar]
- Kádár, F.; Fazekas, J.P.; Sárospataki, M.; LÖvei, G. Seasonal dynamics, age structure and reproduction of four Carabus species (Coleoptera: Carabidae) living in forested landscapes in Hungary. Acta Zool. Acad. Sci. Hung. 2015, 61, 57–72. [Google Scholar] [CrossRef]
- Elek, Z.; Howe, A.G.; Enggaard, M.K.; Lovei, G.L. Seasonal dynamics of common ground beetles (Coleoptera: Carabidae) along urbanization gradient near Soro, Zealand, Danmark. Entomol. Fenn. 2017, 28, 27–40. [Google Scholar] [CrossRef]
- Tauber, M.J.; Tauber, C.A.; Masaki, S. Seasonal Adaptations of Insects; Oxford University Press: New York, NY, USA, 1986; ISBN 9780195036350. [Google Scholar]
- Leather, S.R.; Walters, K.F.A.; Bale, J.S. The Ecology of Insect Overwintering; Cambridge University Press: Cambridge, UK, 1993; p. 268. [Google Scholar]
- Danks, H.V. Insect Dormancy: An Ecological Perspective; Biological Survey of Canada (Terrestrial Arthropods): Ottawa, QC, Canada, 1987; p. 439. [Google Scholar]
- Vido, J.; Nalevanková, P.; Valach, J.; Šustek, Z.; Tadesse, T. Drought analyses of the Horné Požitavie region (Slovakia) in the period 1966–2013. Adv. Meteorol. 2019. [Google Scholar] [CrossRef]
- Weber, F. Vergleichende Untersuchungen über das Verhalten von Carabus-Arten in Luftfeuchtigkeitsgefallen. Z. Morph. Okol. 1965, 54, 551–565. [Google Scholar]
- Šustek, Z.; Vido, J.; Škvareninová, J.; Škvarenina, J.; Šurda, P. Drought impact on ground beetle assemblages (Coleoptera, Carabidae) in Norway spruce forests with different management after windstorm damage—A case study from Tatra Mts. (Slovakia). J. Hydrol. Hydromech. 2017, 65, 333–342. [Google Scholar] [CrossRef]
- Šerić Jelaska, L.; Franjević, D.; Jelaska, S.D.; Symondson, W.O.C. Prey detection in carabid beetles (Coleoptera: Carabidae) in woodland ecosystems by PCR analysis of gut contents. Eur. J. Entomol. 2014, 111. [Google Scholar] [CrossRef]
- Cook, A. Behavioural ecology: On doing the right thing, in the right place, at the right time. In The Biology of Terrestrial Molluscs, 1st ed.; Barker, G.M., Ed.; CABI Publishing: Wallingford, CT, USA, 2001; pp. 447–487. [Google Scholar]
- Sulikowska-Drozd, A.; Maltz, T.K. Experimental drought affects the reproduction of the brooding clausiliid Alinda biplicata (Montagu, 1803). J. Molluscan Stud. 2014, 80, 265–271. [Google Scholar] [CrossRef]
- Nicolai, A.; Filser, J.; Lenz, R.; Bertrand, C.; Charrier, M. Adjustment of metabolite composition in the haemolymph to seasonal variations in the land snail Helix pomatia. J. Comp. Physiol. B 2011, 181, 457–466. [Google Scholar]
- Tondoh, J.E. Seasonal changes in earthworm diversity and community structure in Central Côte d’Ivoire. Eur. J. Soil Biol. 2006, 42, S334–S340. [Google Scholar] [CrossRef]
- Lee, K.E. Earthworm Their Ecology and Relationship with Soils and Land Use; Academic Press: Sydney, Australia, 1985; p. 411. [Google Scholar]
- Eggleton, P.; Inward, K.; Smith, J.; Jones, D.T.; Sherlock, M. A six year study of earthworm (Lumbricidae) populations in pasture woodland in southern England shows their responses to soil temperature and soil moisture. Soil Biol. Biochem. 2009, 41, 1857–1865. [Google Scholar]
- Symondson, W.O.C.; Glen, D.; Ives, A.R.; Langdon, C.J. Dynamics of the relationship between a generalist predator and slugs over five years. Ecology 2002, 83, 137–147. [Google Scholar]
- Knapp, M.; Uhnavá, K. Body Size and Nutrition Intake Effects on Fecundity and Overwintering Success in Anchomenus dorsalis (Coleoptera: Carabidae). J. Insect Sci. 2014, 14, 1–6. [Google Scholar] [CrossRef]
- Honěk, A. The effect of temperature on the activity of Carabidae (Coleoptera) in a fallow field. Eur. J. Entomol. 1997, 94, 97–104. [Google Scholar]
- Mapy.cz. Available online: https://sk.mapy.cz/zakladni?x=17.8924454&y=48.3030312&z=18&l=0&base=ophoto (accessed on 4 November 2020).

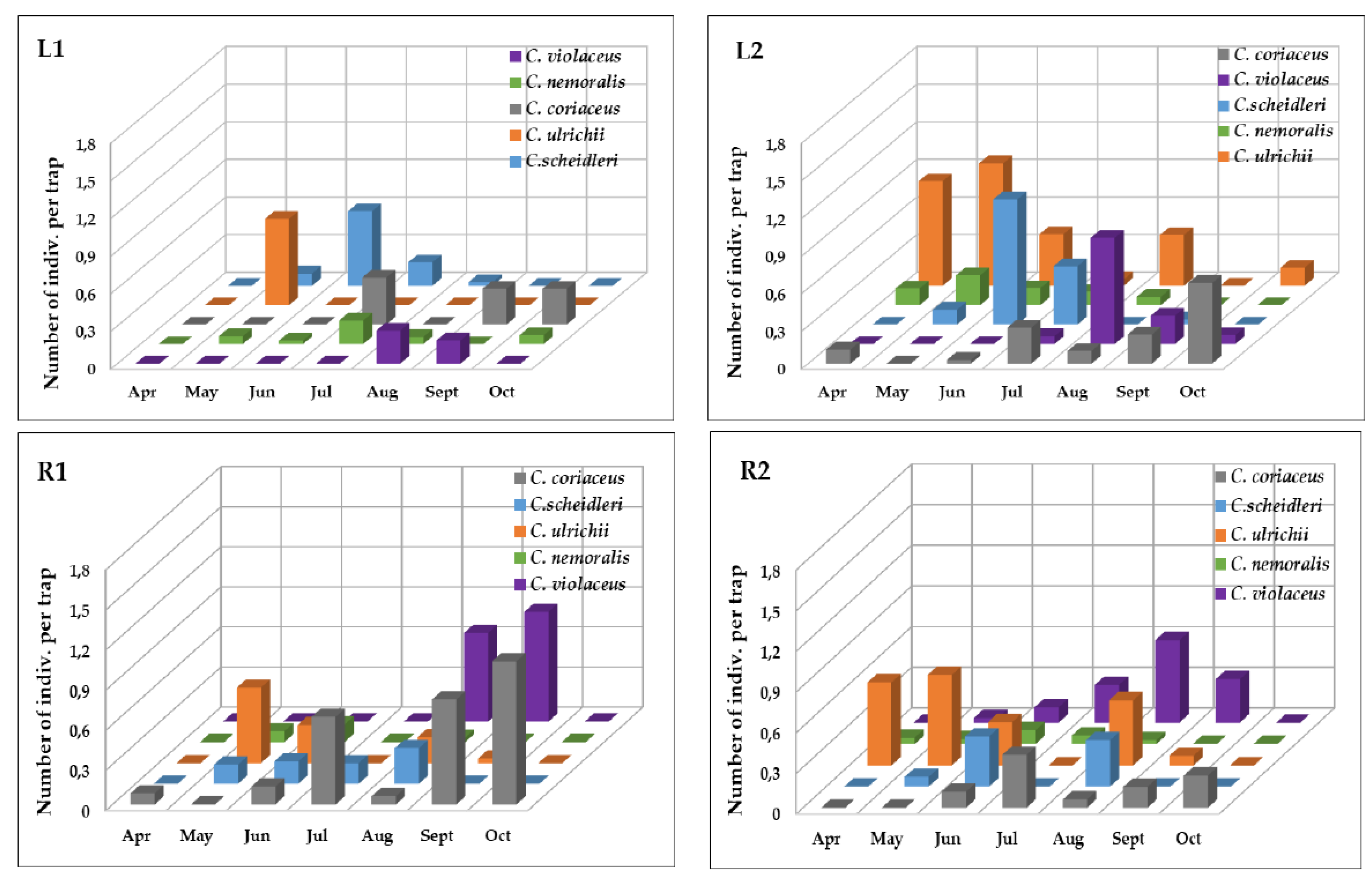
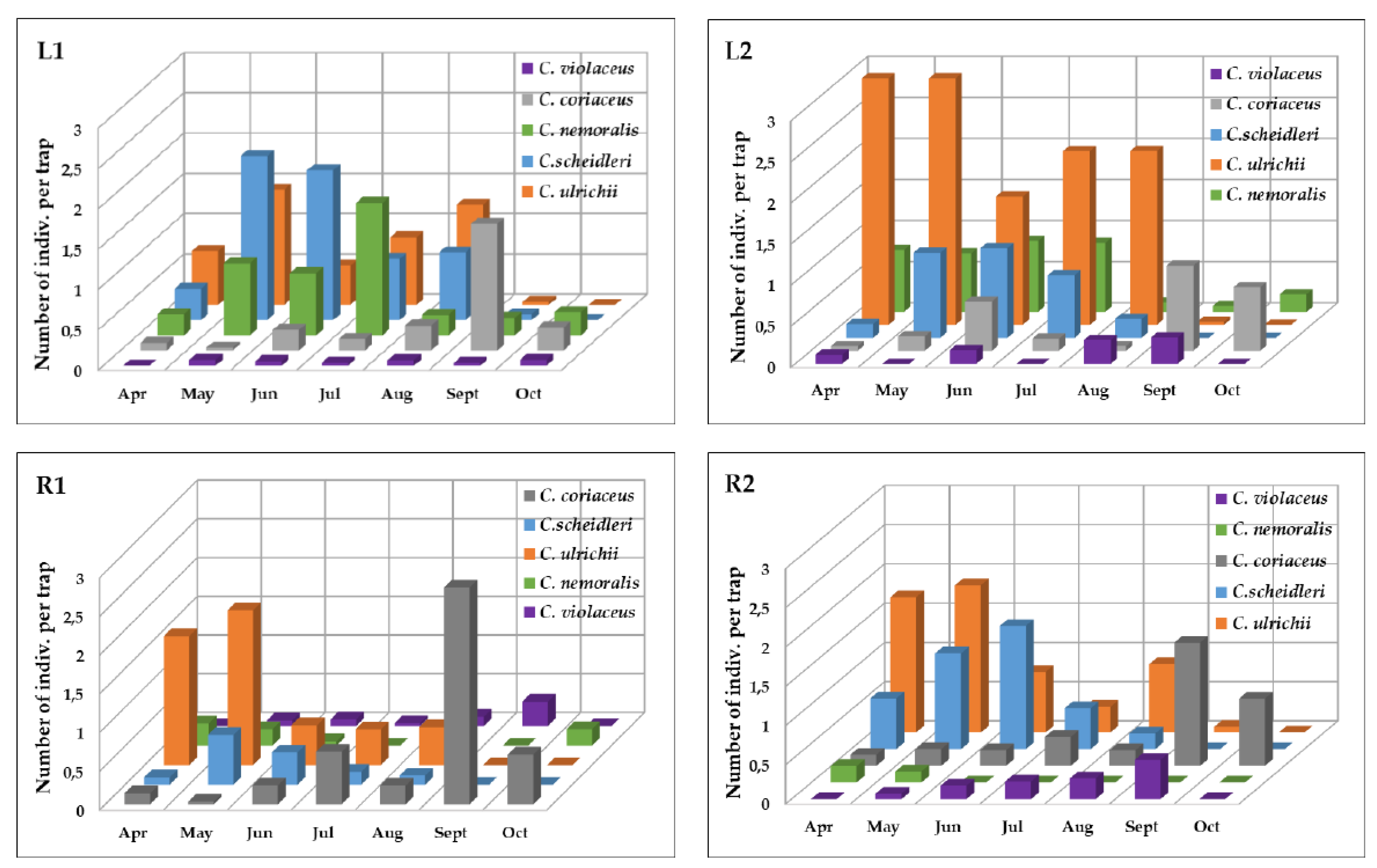

| Index of Dominance (%) | |||||
|---|---|---|---|---|---|
| Species | Total | L1 | L2 | R1 | R2 |
| C. coriaceus | 20.8 | 24.2 | 15.1 | 30.8 | 13.9 |
| C. scheidleri | 23.3 | 24.2 | 26.0 | 25.4 | 16.7 |
| C. ulrichii | 27.4 | 17.7 | 32.3 | 16.2 | 39.6 |
| C. nemoralis | 6.3 | 11.3 | 8.9 | 3.2 | 4.9 |
| C. violaceus | 22.1 | 22.6 | 17.7 | 24.3 | 25.0 |
| Index of Dominance (%) | |||||
|---|---|---|---|---|---|
| Species | Total | L1 | L2 | R1 | R2 |
| C. coriaceus | 22.1 | 14.4 | 11.8 | 30.3 | 21.9 |
| C. scheidleri | 23.4 | 34.6 | 15.9 | 26.2 | 29.6 |
| C. ulrichii | 41.5 | 25.8 | 53.2 | 34.5 | 37.4 |
| C. nemoralis | 7.5 | 23.5 | 15.1 | 4.4 | 2.0 |
| C. violaceus | 5.5 | 1.7 | 4.1 | 4.7 | 9.1 |
| Month | Average Monthly Temperature (T, °C) | Thermal Characteristic | Monthly Precipitation (P, mm) | Precipitation Characteristics | ||||
|---|---|---|---|---|---|---|---|---|
| 2017 | 1961–1990 | ΔT (°C) | 2017 | 1961–1990 | ΔP (%) | |||
| I. | −7.1 | −1.7 | −5.4 | Extremely cold | 0 | 31 | 0 | Extremely dry |
| II. | 1.9 | 0.5 | 1.4 | Warm | 13.2 | 32 | 41 | Very dry |
| III. | 8.4 | 4.7 | 3.7 | Extremely warm | 13.8 | 33 | 42 | Very dry |
| IV. | 9.5 | 10.1 | −0.6 | Normal | 36.2 | 43 | 84 | Normal |
| V. | 16.2 | 14.8 | 1.4 | Warm | 13.8 | 55 | 25 | Extremely dry |
| VI. | 20.8 | 18.3 | 2.5 | Very warm | 24.6 | 70 | 35 | Very dry |
| VII. | 21.3 | 19.7 | 1.6 | Warm | 63.6 | 64 | 99 | Normal |
| VIII. | 22.1 | 19.2 | 2.9 | Very warm | 22.6 | 58 | 39 | Very dry |
| IX. | 14.3 | 15.4 | −1.1 | Cold | 83.4 | 37 | 225 | Extremely wet |
| X. | 10.5 | 10.1 | 0.4 | Normal | 48.4 | 41 | 118 | Normal |
| XI. | 4.4 | 4.9 | −0.5 | Normal | 25.8 | 54 | 48 | Very dry |
| XII. | 0.8 | 0.5 | 0.3 | Normal | 40.0 | 43 | 93 | Normal |
| Month | Average Monthly Temperature (T, °C) | Thermal Characteristic | Monthly Precipitation (P, mm) | Precipitation Characteristics | ||||
|---|---|---|---|---|---|---|---|---|
| 2018 | 1961–1990 | ΔT (°C) | 2018 | 1961–1990 | ΔP (%) | |||
| I. | 1.6 | −1.7 | 3.3 | Very warm | 19 | 31 | 61 | Dry |
| II. | −1.5 | 0.5 | −2.0 | Cold | 33 | 32 | 103 | Normal |
| III. | 2.9 | 4.7 | −1.8 | Cold | 33 | 33 | 100 | Normal |
| IV. | 15.2 | 10.1 | 5.1 | Extremely warm | 5 | 43 | 13 | Extremely dry |
| V. | 18.6 | 14.8 | 3.8 | Extremely warm | 15 | 55 | 27 | Very dry |
| VI. | 20.0 | 18.3 | 1.7 | Warm | 136 | 70 | 195 | Very wet |
| VII. | 21.6 | 19.7 | 1.9 | Warm | 34 | 64 | 53 | Dry |
| VIII. | 22.8 | 19.2 | 3.6 | Extremely warm | 78 | 58 | 134 | Wet |
| IX. | 17.0 | 15.4 | 1.6 | Warm | 99 | 37 | 267 | Extremely wet |
| X. | 12.9 | 10.1 | 2.8 | Very warm | 10 | 41 | 25 | Very dry |
| XI. | 6.5 | 4.9 | 1.6 | Warm | 24 | 54 | 44 | Very dry |
| XII. | 0.4 | 0.5 | −0.1 | Normal | 54 | 43 | 125 | Wet |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šiška, B.; Eliašová, M.; Kollár, J. Carabus Population Response to Drought in Lowland Oak Hornbeam Forest. Water 2020, 12, 3284. https://doi.org/10.3390/w12113284
Šiška B, Eliašová M, Kollár J. Carabus Population Response to Drought in Lowland Oak Hornbeam Forest. Water. 2020; 12(11):3284. https://doi.org/10.3390/w12113284
Chicago/Turabian StyleŠiška, Bernard, Mariana Eliašová, and Ján Kollár. 2020. "Carabus Population Response to Drought in Lowland Oak Hornbeam Forest" Water 12, no. 11: 3284. https://doi.org/10.3390/w12113284
APA StyleŠiška, B., Eliašová, M., & Kollár, J. (2020). Carabus Population Response to Drought in Lowland Oak Hornbeam Forest. Water, 12(11), 3284. https://doi.org/10.3390/w12113284





