Assessing the Impact of Climate Change on Groundwater Quality of the Shallow Coastal Aquifer of Eastern Dahomey Basin, Southwestern Nigeria
Abstract
1. Introduction
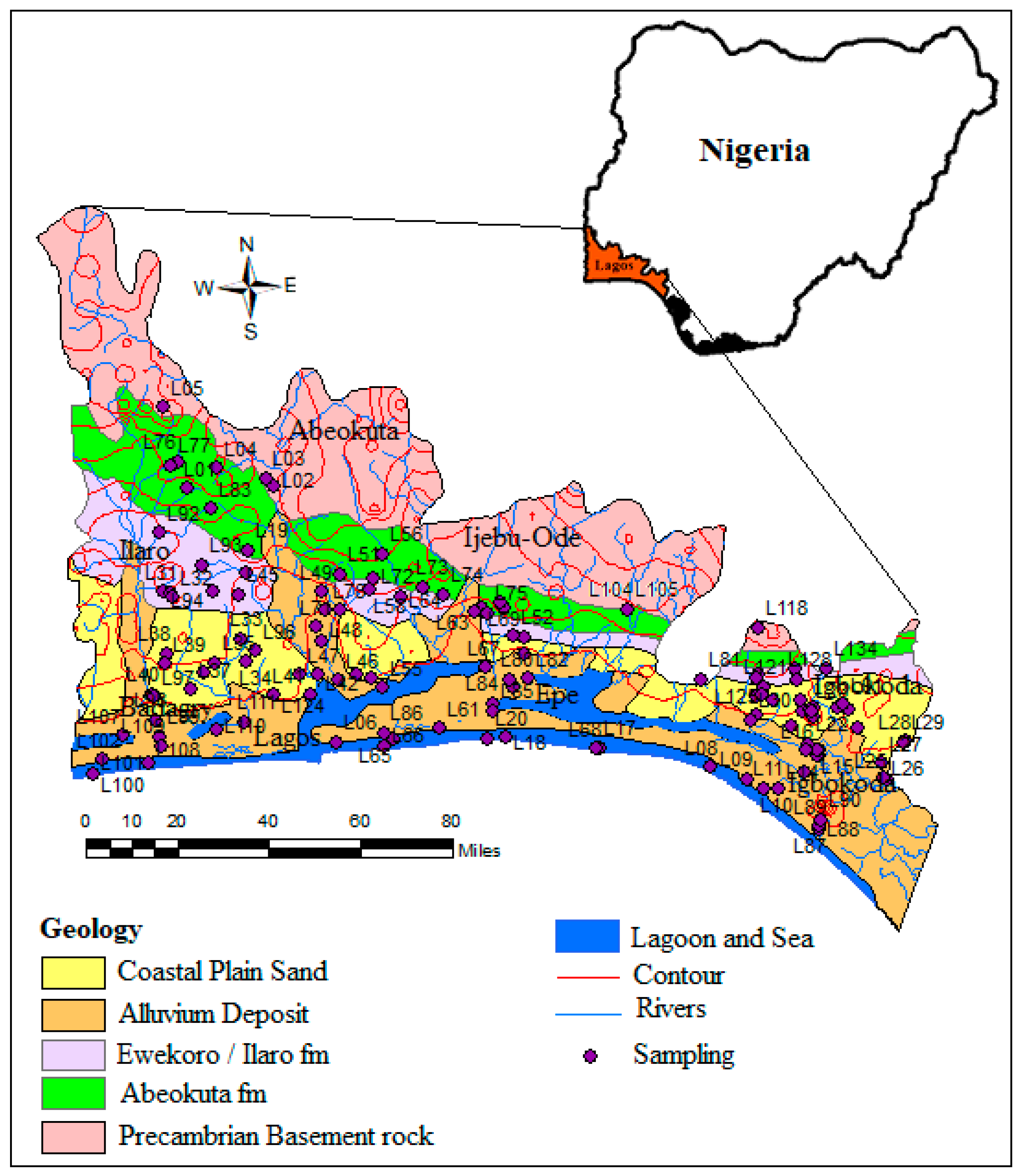
2. Description of the Study Area
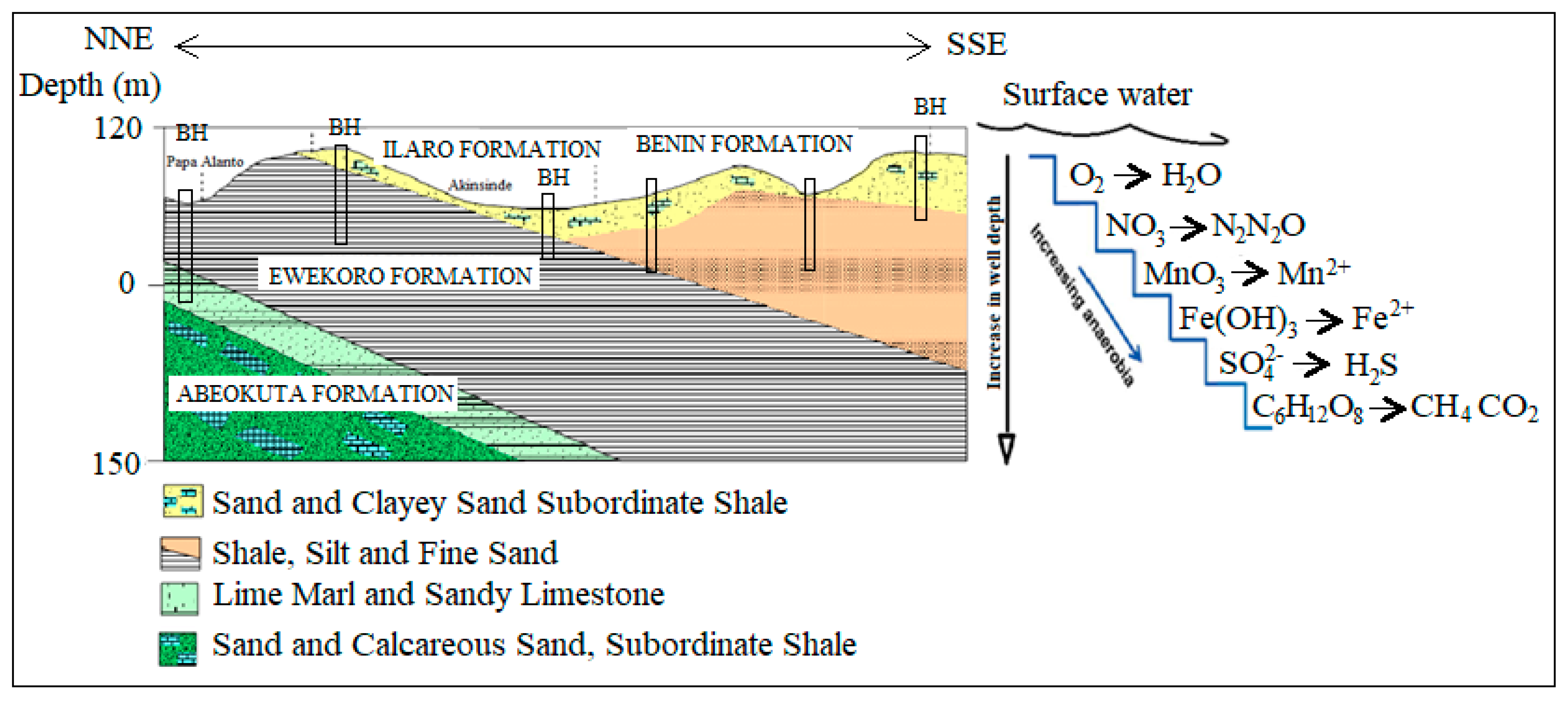
2.1. Geology and Hydrogeology
2.1.1. Geology
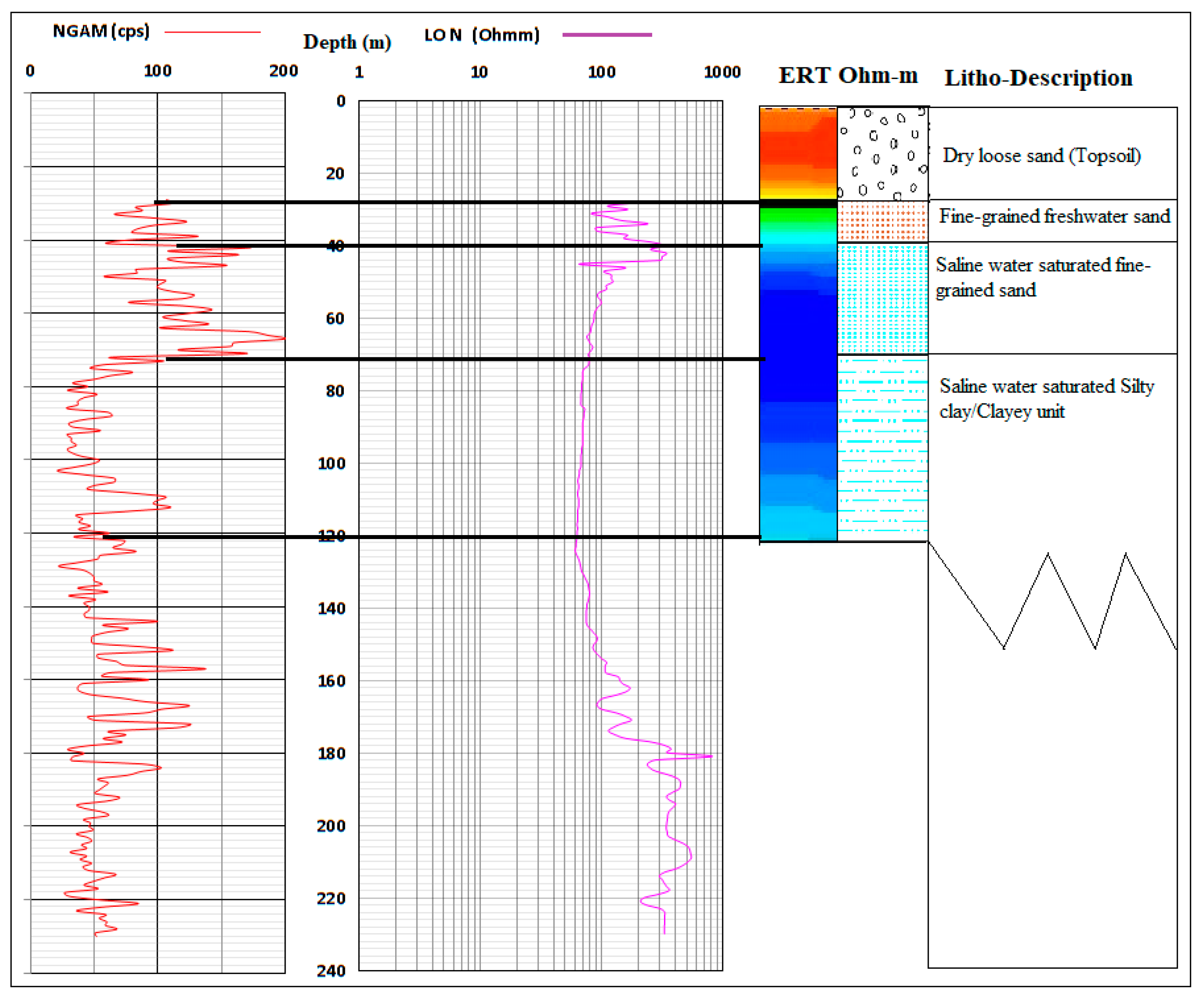
2.1.2. Hydrogeology
3. Methodology
3.1. Field Physicochemical Measurement
3.2. Laboratory Analysis
3.3. Data Evaluation and Statistical Analysis
4. Results and Discussion
4.1. Physicochemical characterisation and desciption
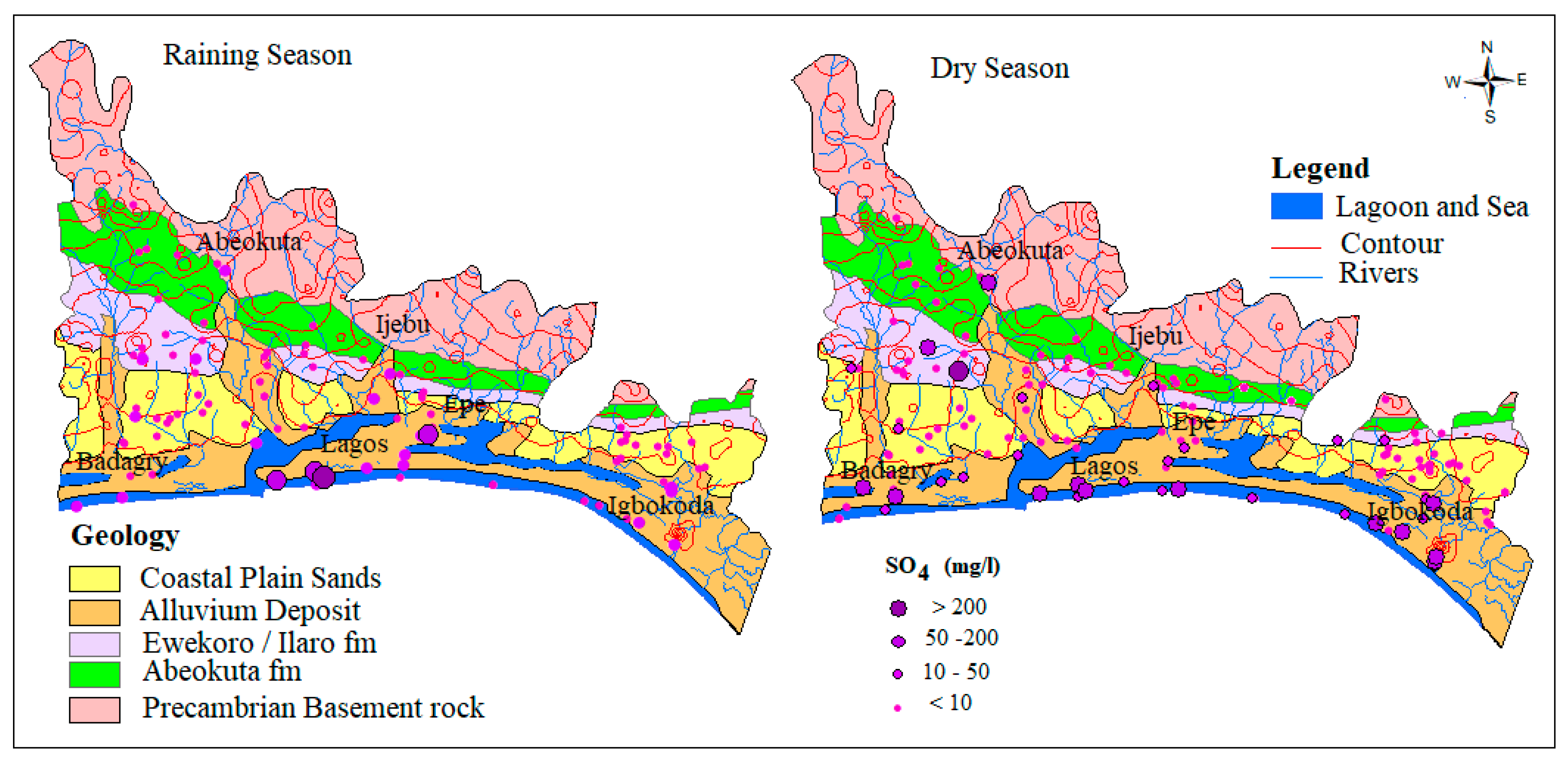
4.1.1. Sulfate (SO42−)
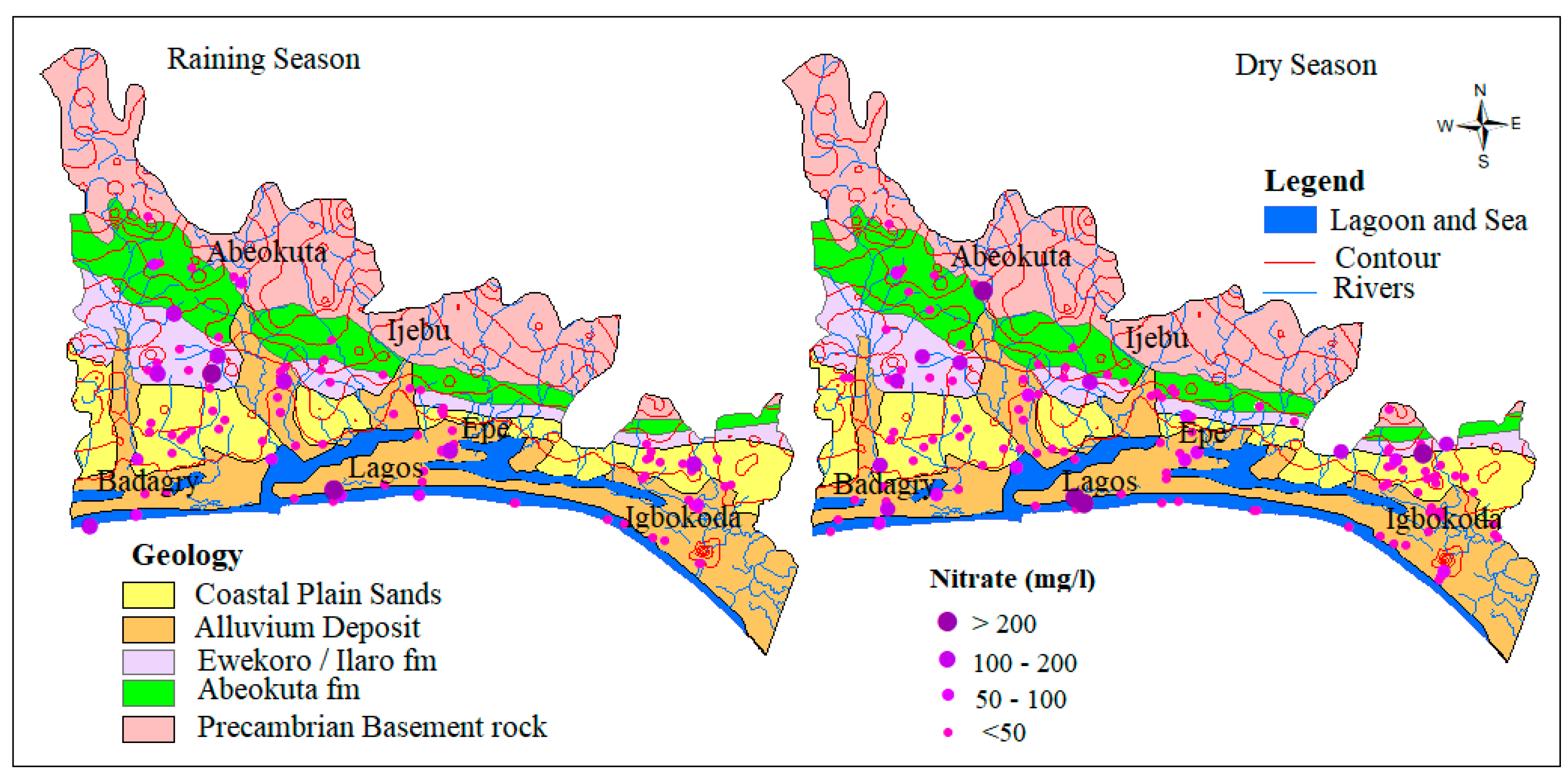
4.1.2. Nitrate (NO3−)
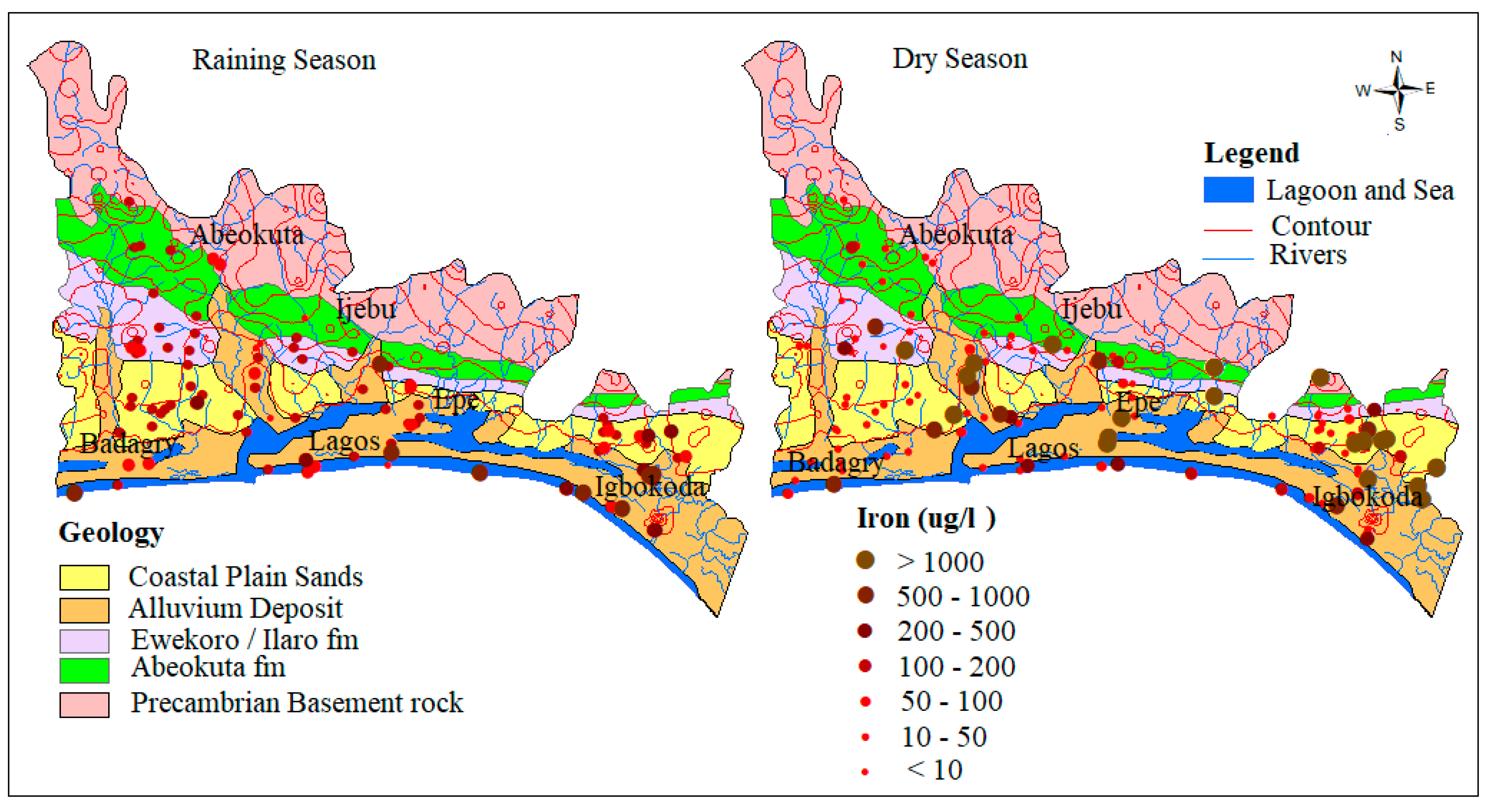
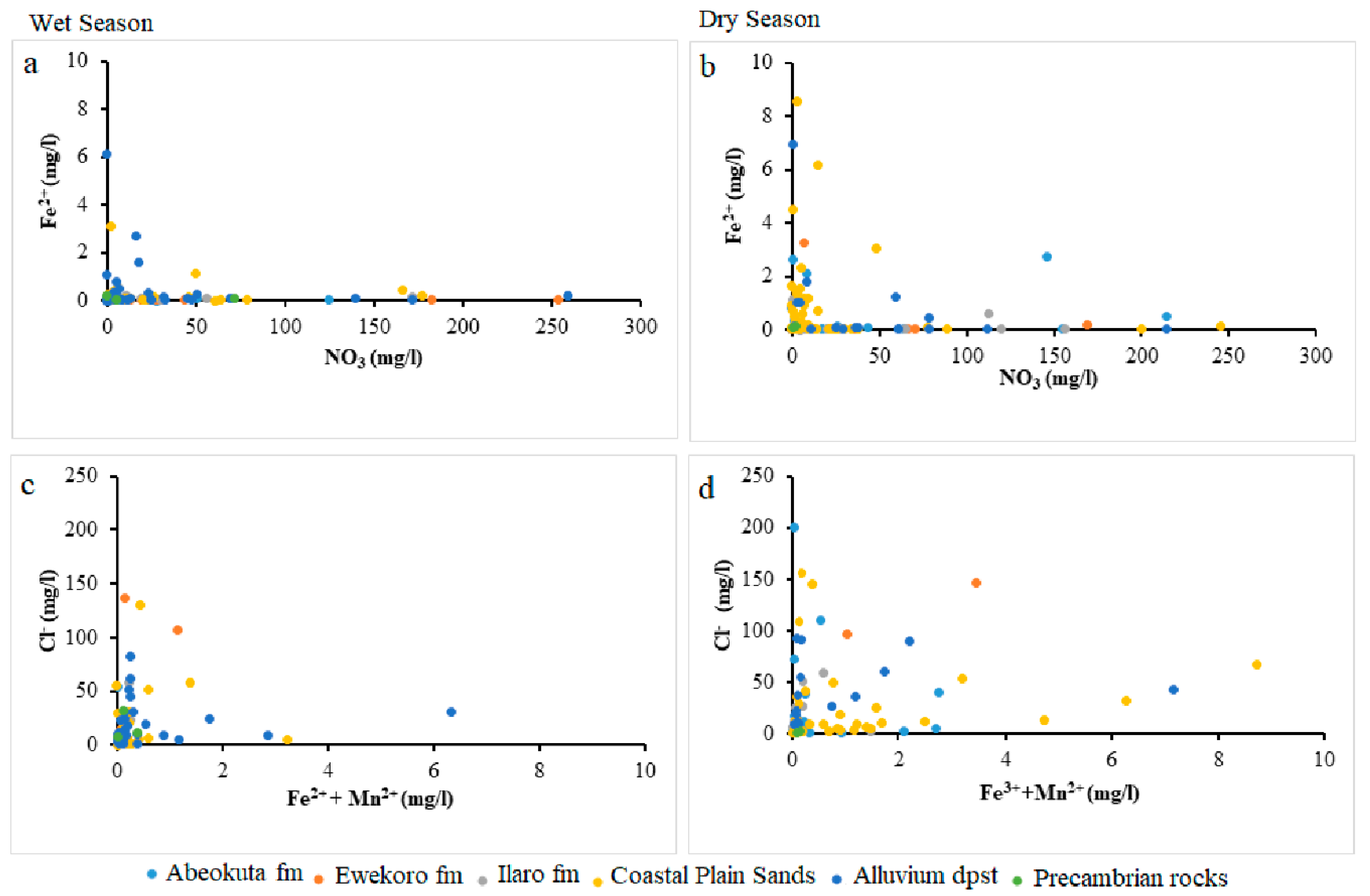
4.1.3. Iron (Fe)
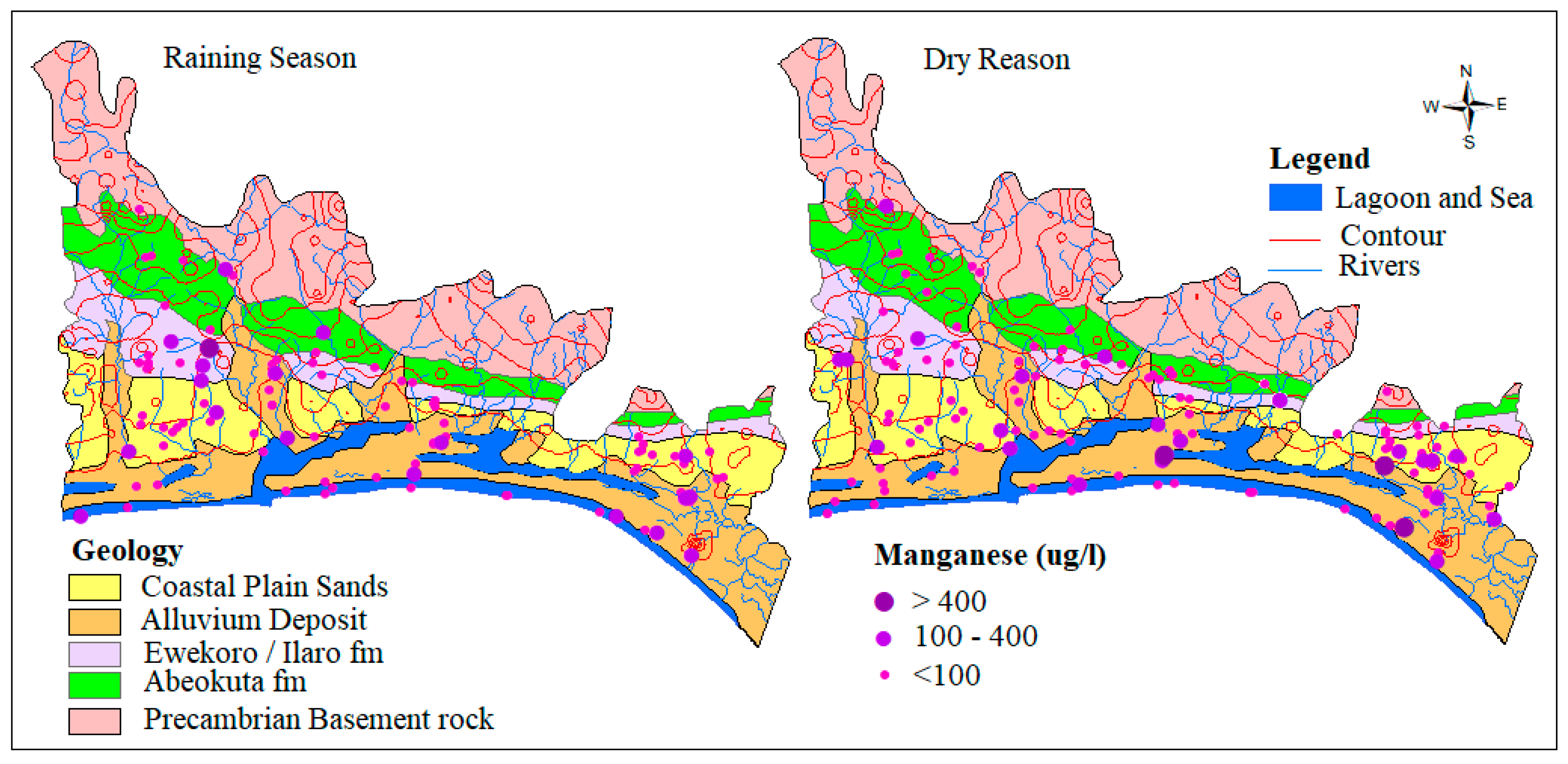
4.1.4. Manganese (Mn)
4.2. Seasonal Effect of Climate Change on Biogeochemical Processes
Multiple Linear Regression Model
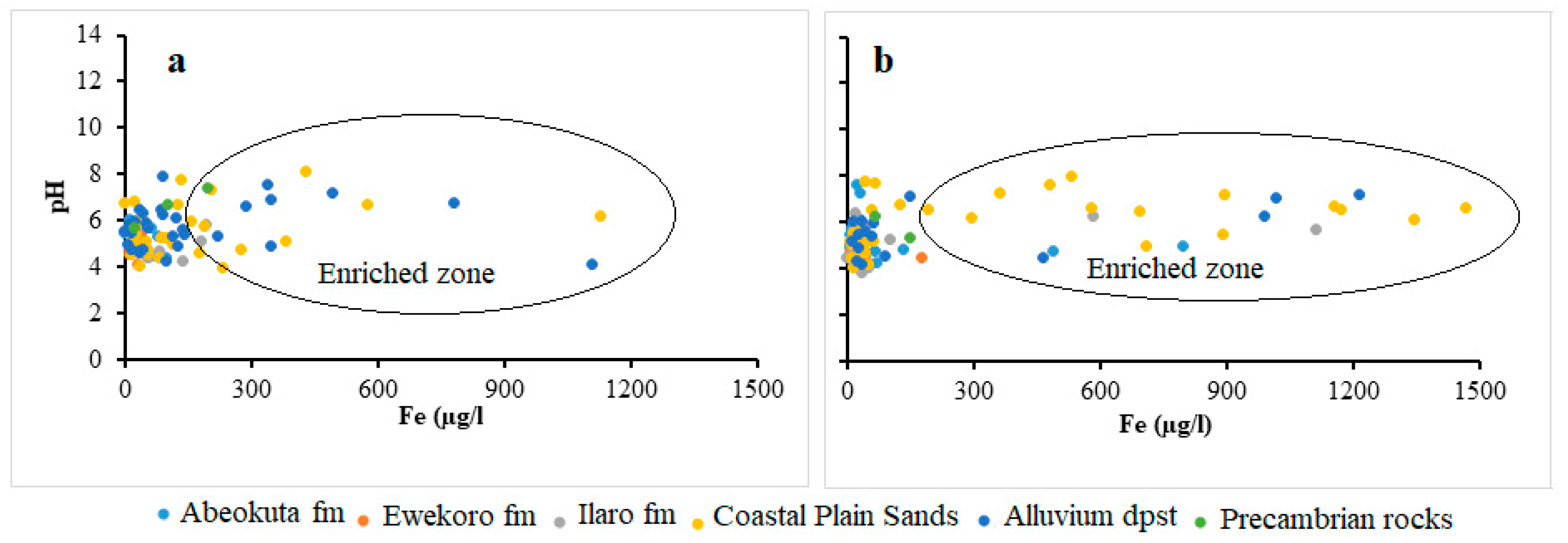
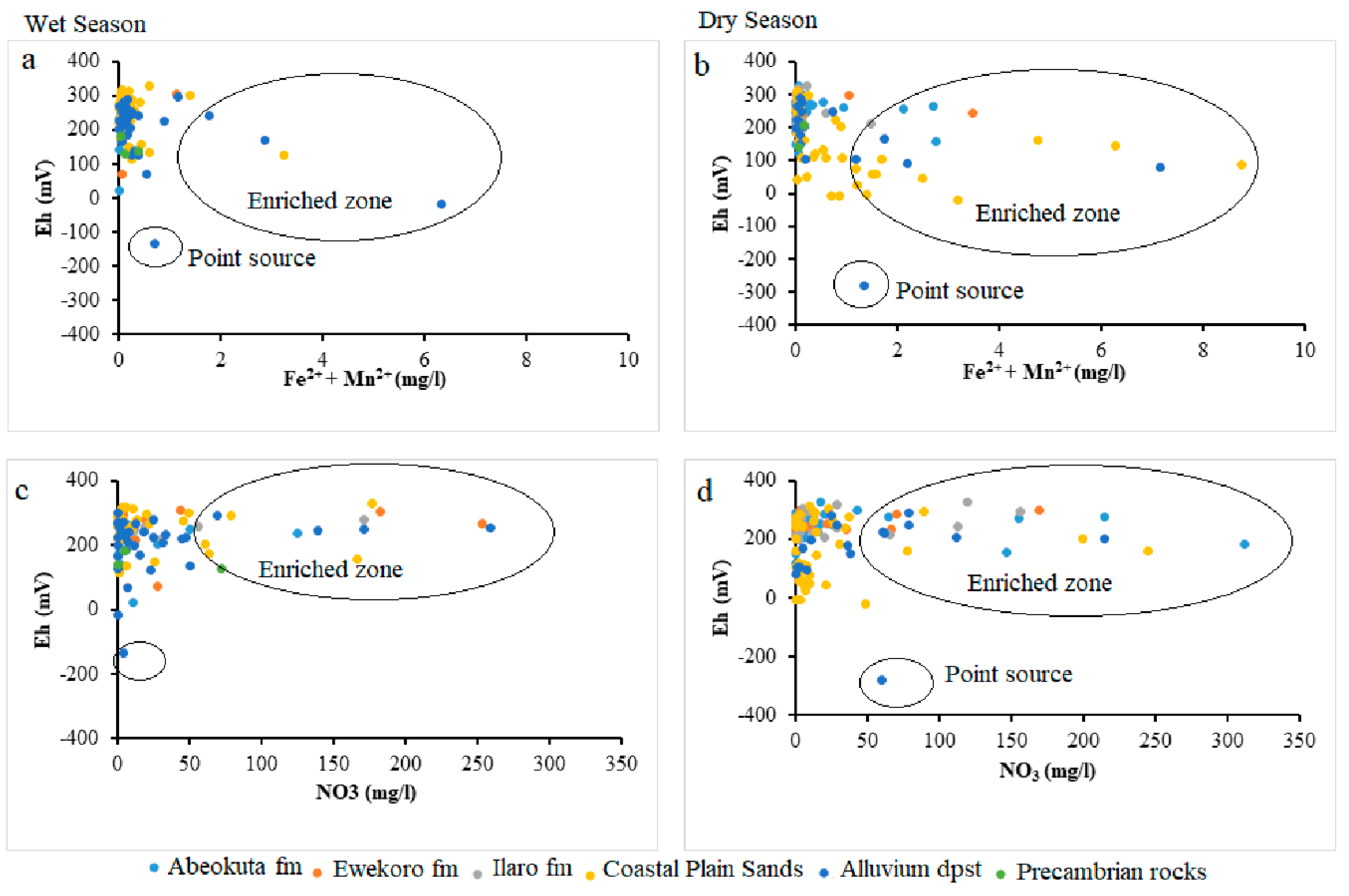
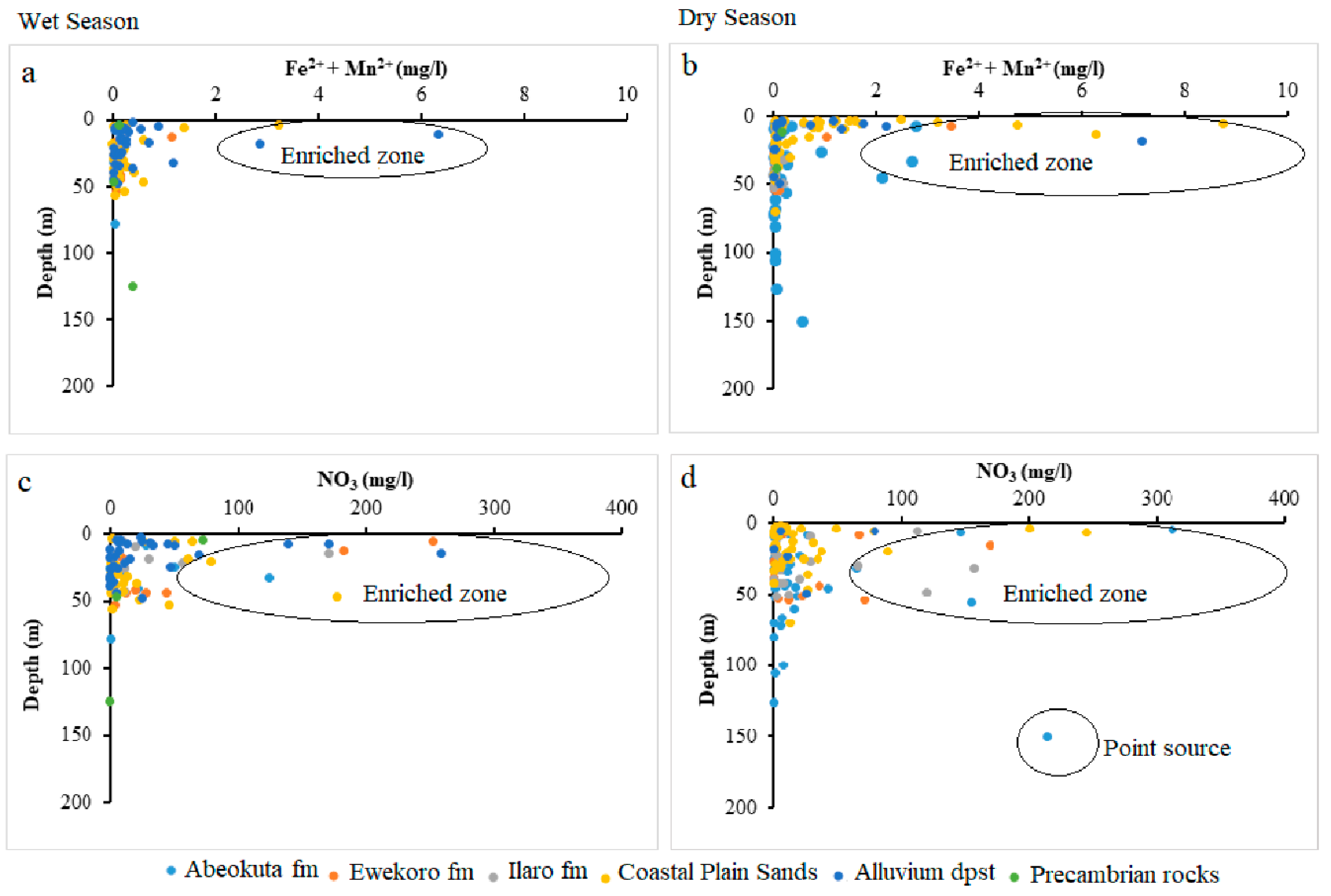
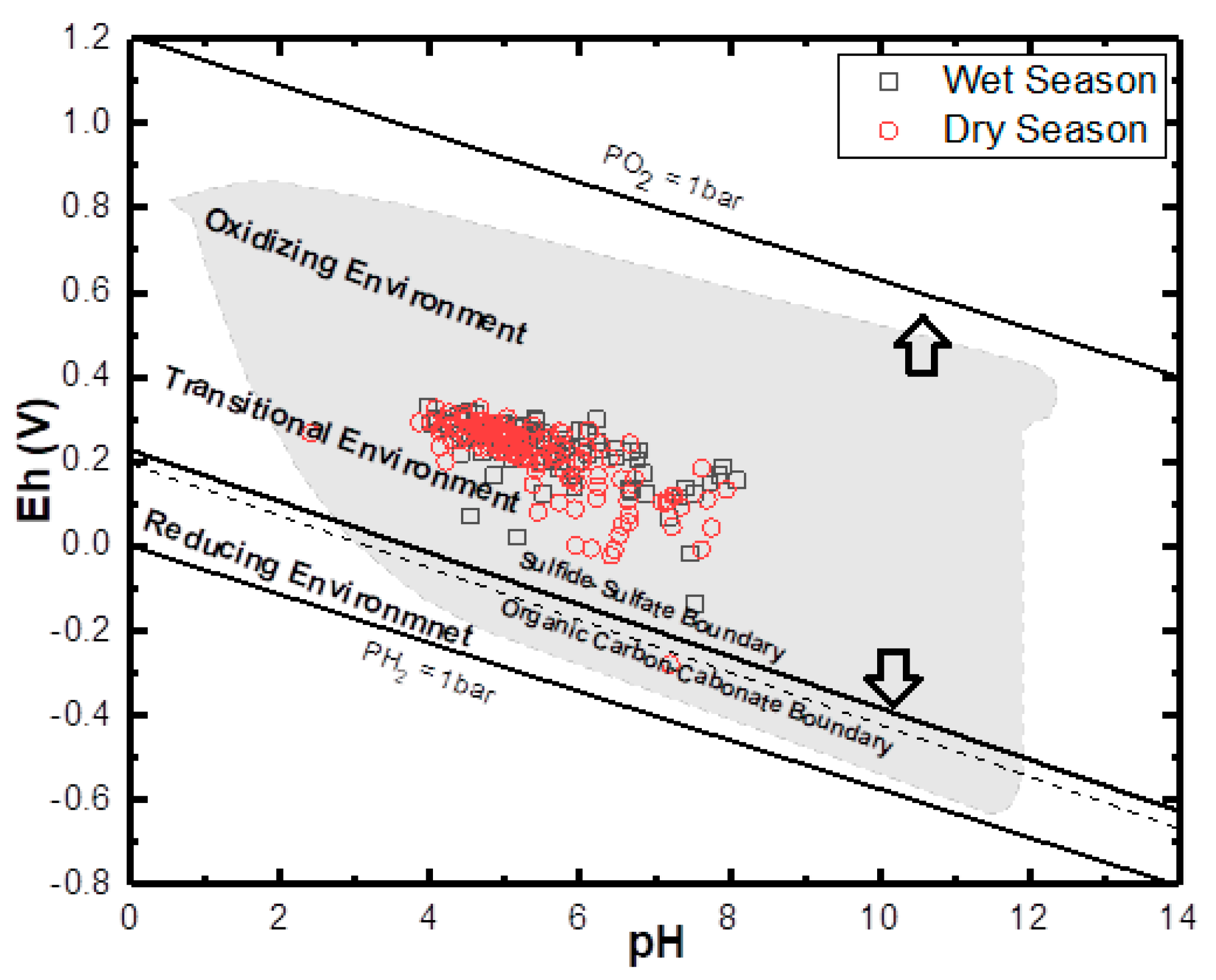
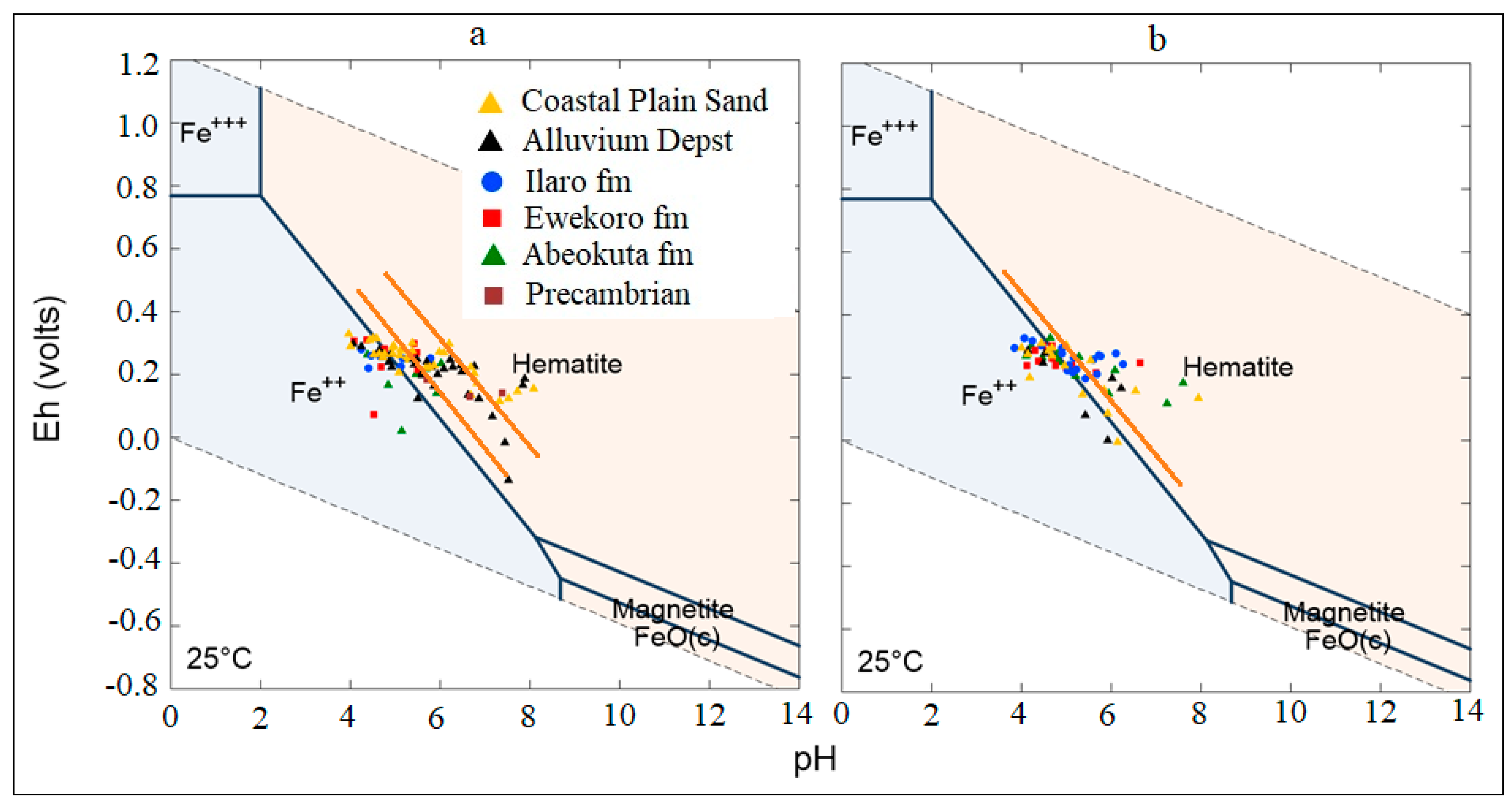
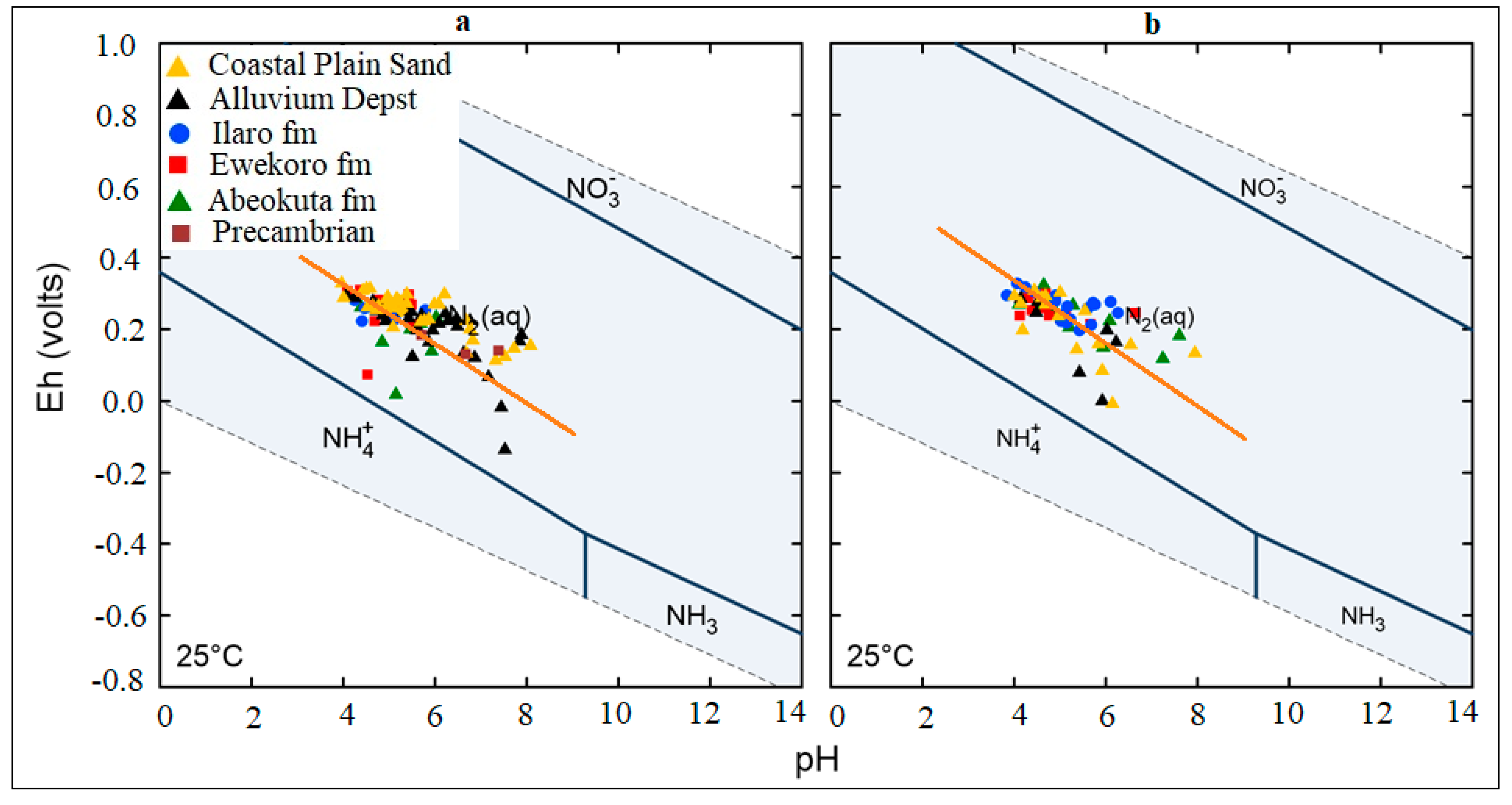
4.3. Analysis of Biogeochemical Processes of Redox Reaction in Groundwater of EDB
4.4. Implication on the Groundwater Quality and Human Health
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Omosuyi, G.O.; Oseghale, A. Groundwater vulnerability assessment in shallow aquifers using geoelectric and hydrogeologic parameters at Odigbo, Southwestern Nigeria. Am. J. Sci. Ind. Res. 2012, 501–512. [Google Scholar] [CrossRef]
- Adegbola, R.; Oseni, S.; Majolagbe, A. The impact assessment of oil spillage and trace metal assessment on groundwater in Baruwa, Lagos Southwest and Nigeria. Int. Sci. Investig. J. 2013, 2, 12–32. [Google Scholar]
- Oyelami, A.C.; Ojo, O.A.; Aladejana, .J.A.; Agbede, .O.O. Assessing the effect of a dumpsite on groundwater quality: A case study of aduramigba estate within osogbo metropolis. J. Environ. Earth Sci. 2013, 3, 120–131. [Google Scholar]
- Edet, A. Hydrogeology and groundwater evaluation of a shallow coastal aquifer, southern Akwa Ibom State (Nigeria). Appl. Water Sci. 2016, 7, 2397–2412. [Google Scholar] [CrossRef]
- Snyder, M.; Taillefert, M.; Ruppel, C. Redox zonation at the saline-influenced boundaries of a permeable surficial aquifer: Effects of physical forcing on the biogeochemical cycling of iron and manganese. J. Hydrol. 2004, 296, 164–178. [Google Scholar] [CrossRef]
- Holden, A.A.; Haque, S.E.; Mayer, K.U.; Ulrich, A.C. Biogeochemical processes controlling the mobility of major ions and trace metals in aquitard sediments beneath an oil sand tailing pond: Laboratory studies and reactive transport modeling. J. Contam. Hydrol. 2013, 151, 55–67. [Google Scholar] [CrossRef]
- Matiatos, I. Nitrate source identification in groundwater of multiple land-use areas by combining isotopes and multivariate statistical analysis: A case study of Asopos basin (Central Greece). Sci. Total Environ. 2016, 541, 802–814. [Google Scholar] [CrossRef]
- Delpla, I.; Jung, A.; Baures, E.; Clement, M.; Thomas, O. Impacts of climate change on surface water quality in relation to drinking water production. Environ. Int. 2009, 35, 1225–1233. [Google Scholar] [CrossRef]
- Ojolowo, S.; Wahab, B. Municipal solid waste and flooding in Lagos metropolis, Nigeria: Deconstructing the evil nexus. J. Geogr. Reg. Plan. Full 2017, 10, 174–185. [Google Scholar]
- Ayolabi, E.A.; Epelle, E.S.; Lucas, O.B.; Ojo, A. Geophysical and geochemical site investigation of eastern part of Lagos metropolis, southwestern Nigeria. Arab. J. Geosci. 2014, 8, 7445–7453. [Google Scholar] [CrossRef]
- IPCC. Climate Change 1995. The Science of Climate Cliange; The Press Syndicate of the University of Cambridge: Camebridge, UK, 1995; Volume 53. [Google Scholar]
- Whitehead, P.G.; Wilby, R.L.; Battarbee, R.W.; Kernan, M.; Wade, A.J. A review of the potential impacts of climate change on surface water quality a review of the potential impacts of climate change on surface water quality. Hydrol. Sci. J. ISSN 2009, 54, 101–123. [Google Scholar] [CrossRef]
- Guasch, H.; Serra, A.; Corcoll, N.; Bonet, B.; Leira, M. Metal ecotoxicology in fluvial biofilms: Potential influence of water scarcity. Hdb. Env. Chem. 2010, 41–53. [Google Scholar] [CrossRef]
- Ezekwe, C.I.; Edoghotu, M.I. Water quality and environmental health indicators in the Andoni River estuary, Eastern Niger Delta of Nigeria. Environ. Earth Sci. 2015, 74, 6123–6136. [Google Scholar] [CrossRef]
- Cann, K.F.; Thomas, D.; Salmon, R.; Wyn-Jones, A.; Kay, D. Extreme water-related weather events and waterborne disease. Epidemiol. Infect. 2013, 141, 671–686. [Google Scholar] [CrossRef]
- Kasei, R.A. Modelling Impacts of Climate Change on Water Resources in the Volta Basin, West Africa. Ph.D. Thesis, Mathematisch-Naturwissenschaftlichen Fakultät, Rheinischen Friedrich-Wilhelms-Universität Bonn, Bonn, Germany, 2009. [Google Scholar]
- Babanyara, Y.Y.; Usman, H.A.; Saleh, U.F. An overview of urban poverty and environmental problems in Nigeria. J. Hum. Ecol. 2010, 31, 135–143. [Google Scholar] [CrossRef]
- Serdeczny, O.; Adams, S.; Baarsch, F.; Coumou, D.; Robinson, A.; Hare, W.; Schaeffer, M.; Perrette, M.; Reinhardt, J. Climate change impacts in Sub-Saharan Africa: From physical changes to their social repercussions. Reg. Environ. Chang. 2016, 1–16. [Google Scholar] [CrossRef]
- Pfeiffer, M.; Batbayar, G.; Hahn-tomer, D.K.S. Investigating arsenic (As) occurrence and sources in ground, surface, waste and drinking water in northern Mongolia. Environ. Earth Sci. 2015, 73, 649–662. [Google Scholar] [CrossRef]
- Priyantha Ranjan, S.; Kazama, S.; Sawamoto, M. Effects of climate and land use changes on groundwater resources in coastal aquifers. J. Environ. Manag. 2006, 80, 25–35. [Google Scholar] [CrossRef]
- Linzey, D. Atlantic climate adaptation solutions association saltwater intrusion and climate change. 2011. Available online: http://www.gov.pe.ca/photos/original/cle_WA1.pdf (accessed on 1 January 2020).
- Hiyama, T.; Babiker, I.S.; Mohamed, M.A.A. Groundwater as a Key for Adaptation to Changing Climate and Society; Springer: Tokyo, Japan, 2014; ISBN 978-4-431-54967-3. [Google Scholar]
- Unsal, B.; Yagbasan, O.; Yazicigil, H. Assessing the impacts of climate change on sustainable management of coastal aquifers. Environ. Earth Sci. 2014, 72, 2183–2193. [Google Scholar] [CrossRef]
- Nistor, M.-M.; Dezsi, Ş.; Cheval, S.; Baciu, M. Climate change effects on groundwater resources: A new assessment method through climate indices and effective precipitation in Beliş district, Western Carpathians. Meteorol. Appl. 2016, 23, 554–561. [Google Scholar] [CrossRef]
- Ayolabi, E.A.; Folorunso, A.F.; Kayode, O.T. Integrated geophysical and geochemical methods for environmental assessment of municipal dumpsite system. Int. J. Geosci. 2013, 4, 850–862. [Google Scholar] [CrossRef]
- Hashmi, M.Z.; Yu, C.; Shen, H.; Duan, D. Concentrations and human health risk assessment of selected heavy metals in surface water of the siling reservoir watershed in Zhejiang Province, China. Pol. J. Environ. Stud. 2014, 23, 801–811. [Google Scholar]
- Bieroza, M.Z.; Heathwaite, A.L. Seasonal variation in phosphorus concentration—Discharge hysteresis inferred from high-frequency in situ monitoring. J. Hydrol. 2015, 524, 333–347. [Google Scholar] [CrossRef]
- Nick, H.M.; Raoof, A.; Centler, F.; Thullner, M.; Regnier, P. Reactive dispersive contaminant transport in coastal aquifers: Numerical simulation of a reactive Henry problem. J. Contam. Hydrol. 2013, 145, 90–104. [Google Scholar] [CrossRef]
- Offodile, M.E. The Hydrogeology of Coastal Areas of Southeastern states of Nigeria. J. Min. Geol. 1971, 14, 94–101. [Google Scholar]
- Adegoke, O.S.; Omatsola, M.E. Tectonic evolution and cretaceous stratigraphy of the dahomey basin. Niger. J. Min. Geol. 1981, 18, 130–137. [Google Scholar]
- Jones, H.A.; Hockey, R. The geology of part of southwestern Nigeria. GSN Bull. 1964, 31, 87. [Google Scholar]
- Kogbe, C.A. The cretaceous and paleogene sediments of Southern Nigeria. In Geology of Nigeria, 2nd ed.; Kogbe, C.A., Ed.; Rock View (Nig) Ltd.: Owerri, Nigeria, 1976; pp. 273–282. [Google Scholar]
- Reyment, R.A. Aspects of Geology of Nigeria; Ibadan University Press: Ibadan, Nigeria, 1965; p. 145. [Google Scholar]
- Longe, E.O.; Malomo, S.; Olorunniwo, M.A. Hydrogeology of Lagos metropolis. J. Afr. Earth Sci. 1987, 6, 163–174. [Google Scholar] [CrossRef]
- Adeoti, L.; Alile, O.M.; Uchegbulam, O. Geophysical investigation of saline water intrusion into freshwater aquifers: A case study of Oniru, Lagos State. Sci. Res. Essays 2010, 5, 248–259. [Google Scholar]
- Fatoba, J.O.; Omolayo, S.D.; Adigun, E.O. Using geoelectric soundings for estimation of hydraulic characteristics of aquifers in the coastal area of Lagos, southwestern Nigeria. Int. Lett. Nat. Sci. 2014, 11, 30–39. [Google Scholar] [CrossRef]
- Faleye, E.T. Olorunfemi aquifer characterization and groundwater potential assessment of the sedimentary basin of ondo state 1 2. Ife J. Sci. 2015, 17, 429–439. [Google Scholar]
- Offodile, M.E. Hydrogeology: Groundwater Study and Development in Nigeria, 3rd ed.; Mecon Geology and Engineering Services Ltd.: Jos, Nigeria, 2014; ISBN 978-30956-41. [Google Scholar]
- Adhikary, P.P.; Chandrasekharan, H.; Chakraborty, D.; Kumar, B.; Yadav, B.R. Statistical approaches for hydrogeochemical characterization of groundwater in West Delhi, India. Environ. Monit. Assess. 2009, 154, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-L.; Wang, Y.-S.; Sun, C.-C.; Wang, H.; Dong, J.-D.; Yin, J.-P.; Han, S.-H. Identification of coastal water quality by statistical analysis methods in Daya Bay, South China Sea. Mar. Pollut. Bull. 2010, 60, 852–860. [Google Scholar] [CrossRef]
- Amangabara, G.T.; Ejenma, E. Groundwater quality assessment of Yenagoa and between 2010 and 2011. Resour. Environ. 2012, 2, 20–29. [Google Scholar] [CrossRef]
- Thilagavathi, R.; Chidambaram, S.; Thivya, C.; Prasanna, M.V.; Keesari, T.; Pethaperumal, S. Assessment of groundwater chemistry in layered coastal aquifers using multivariate statistical analysis. Sustain. Water Resour. Manag. 2017, 3, 55–69. [Google Scholar] [CrossRef]
- Boughariou, E.; Bahloul, M.; Jmal, I.; Allouche, N.; Makni, J.; Khanfir, H.; Bouri, S. Hydrochemical and statistical studies of the groundwater salinization combined with MODPATH numerical model: Case of the Sfax coastal aquifer, Southeast Tunisia. Arab. J. Geosci. 2018, 11, 69. [Google Scholar] [CrossRef]
- Ganiyu, S.A.; Badmus, B.S.; Olurin, O.T.; Ojekunle, Z.O. Evaluation of seasonal variation of water quality using multivariate statistical analysis and irrigation parameter indices in Ajakanga area, Ibadan, Nigeria. Appl. Water Sci. 2018, 8, 35. [Google Scholar] [CrossRef]
- White, J.K.; Beaven, R.P.; Powrie, W.; Knox, K. Leachate recirculation in a landfill: Some insights obtained from the development of a simple 1-D model. Waste Manag. 2011, 31, 1210–1221. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, F.; Zhang, Q.; Li, J.; Liu, Q. Tracing nitrate pollution sources and transformation in surface- and ground-waters using environmental isotopes. Sci. Total Environ. 2014, 490, 213–222. [Google Scholar] [CrossRef]
- Larsen, G.R. Determination of Coastal Ground and Surface Water Processes and Character by Use of Hydrochemistry and Stable Isotopes, Fraser Coast, Queensland. Ph.D. Thesis, Queensland University of Technology, Brisbane City, Australia, 2012. [Google Scholar]
- Russak, A.; Sivan, O.; Yechieli, Y. Trace elements (Li, B, Mn and Ba) as sensitive indicators for salinization and freshening events in coastal aquifers. Chem. Geol. 2016, 441, 35–46. [Google Scholar] [CrossRef]
- Wu, Y.; Prulho, R.; Brigante, M.; Dong, W.; Hanna, K.; Wu, Y.; Prulho, R.; Brigante, M.; Dong, W.; Hanna, K. Activation of persulfate by Fe (III) species: Implications for 4-tert-butylphenol degradation to cite this version: HAL Id: hal-01438110. J. Hazard. Mater. 2017. [Google Scholar] [CrossRef]
- Capaccioni, B.; Didero, M.; Paletta, C.; Didero, L. Saline intrusion and refreshening in a multilayer coastal aquifer in the Catania Plain (Sicily, Southern Italy): Dynamics of degradation processes according to the hydrochemical characteristics of groundwaters. J. Hydrol. 2005, 307, 1–16. [Google Scholar] [CrossRef]
- Fan, A.M.; Steinberg, V.E. Health implications of nitrate and nitrite in drinking water: An update on methemoglobinemia occurrence and reproductive and developmental toxicity. Regul. Toxicol. Pharmacol. 1996, 23, 35–43. [Google Scholar] [CrossRef] [PubMed]
| Param | Wet Season | Dry Season | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Min | Max | Aver | Stdev | N | Min | Max | Aver | Stdev | N | |
| Elev | −1.00 | 230.0 | 46.3 | 39.8 | 96 | −1.0 | 230 | 48.2 | 41.5 | 134 |
| pH | 3.97 | 8.10 | 5.57 | 1.00 | 96 | 3.85 | 8.0 | 5.5 | 0.9 | 134 |
| EC | 0.00 | 12000 | 295 | 1219 | 96 | 5.50 | 10009 | 348 | 995 | 134 |
| TDS | 0.00 | 8500 | 201 | 863 | 96 | 2.30 | 6750 | 232 | 668 | 134 |
| ORP | −136 | 330 | 222 | 74 | 96 | −280 | 326 | 208 | 92.1 | 134 |
| Sal | 0.00 | 5000 | 137 | 509 | 96 | 14 | 5070 | 173 | 502 | 134 |
| Temp | 25.5 | 34.6 | 29.4 | 1.7 | 96 | 26.6 | 37.7 | 31.2 | 1.7 | 134 |
| SWL | 0.20 | 67.0 | 18.3 | 15.4 | 96 | 0.3 | 123 | 18.8 | 19.2 | 134 |
| Cl | 0.10 | 18970 | 218 | 1934 | 96 | 0.9 | 1289 | 31.6 | 116 | 134 |
| SO4 | 0.10 | 2211 | 37.9 | 245 | 96 | 0.3 | 576 | 17.5 | 56.1 | 134 |
| NO3 | 0.02 | 259 | 32.2 | 54 | 96 | 0.3 | 312 | 30.3 | 54.4 | 134 |
| Br | 0.00 | 40.8 | 0.46 | 4.2 | 96 | 0.01 | 21.7 | 0.3 | 1.9 | 134 |
| Fe | 0.00 | 6.10 | 0.3 | 0.8 | 96 | 0.01 | 136 | 1.5 | 11.9 | 134 |
| Mn | 0.00 | 1.11 | 0.1 | 0.1 | 96 | 0.00 | 27.7 | 0.3 | 2.4 | 134 |
| Parameters | Coefficient | Std. Error | z | p-Value | |
|---|---|---|---|---|---|
| pH(−1) | 0.2715 | 0.0516 | 5.265 | <0.0001 | *** |
| const | 6.1009 | 0.9010 | 6.771 | <0.0001 | *** |
| SO42 | 0.0192 | 0.0036 | 5.291 | <0.0001 | *** |
| Cl | −0.0022 | 0.0004 | −5.188 | <0.0001 | *** |
| NO3 | −0.0022 | 0.0008 | −3.195 | 0.0014 | *** |
| Fe | 0.2721 | 0.0833 | 3.269 | 0.0011 | *** |
| Mn | −0.0052 | 0.4067 | −0.01280 | 0.9898 | |
| EC | 7.48261 × 10−5 | 4.67708 × 10−5 | 1.600 | 0.1096 | |
| Temp | −0.0777 | 0.0287 | −2.711 | 0.0067 | *** |
| Parameters | Coefficient | Std. Error | z | p-Value | |
|---|---|---|---|---|---|
| pH(−1) | 0.05621 | 0.0449 | 1.252 | 0.2106 | |
| const | 4.9667 | 0.3383 | 14.68 | <0.0001 | *** |
| Sal | 4.81623 × 10−5 | 0.0002 | 0.2280 | 0.8196 | |
| Temp | 0.0108 | 0.00301 | 3.594 | 0.0003 | *** |
| SO4 | 0.0022 | 0.0020 | 1.076 | 0.2819 | |
| Cl | 0.0007 | 0.0011 | 0.6099 | 0.5419 | |
| NO3 | −0.0057 | 0.0019 | −2.948 | 0.0032 | *** |
| Fe | 0.0125 | 0.0827 | 0.1508 | 0.8801 | |
| Mn | −0.0636 | 0.4079 | −0.1559 | 0.8761 | |
| SWL | −0.0173335 | 0.0056 | −3.100 | 0.0019 | *** |
| Parameters | Coefficient | Std. Error | z | p-Value | |
|---|---|---|---|---|---|
| ORP(−1) | 0.3181 | 0.0989 | 3.215 | 0.0013 | *** |
| const | 0.4747 | 0.6421 | 0.7393 | 0.4598 | |
| Sal | −0.2739 | 0.0872 | −3.142 | 0.0017 | *** |
| Temp | 5.6701 | 4.7094 | 1.204 | 0.2286 | |
| SWL | 1.0481 | 0.5535 | 1.894 | 0.0583 | * |
| SO42 | 0.4348 | 0.5958 | 0.7298 | 0.4655 | |
| Cl | −0.0530 | 0.0689 | −0.7689 | 0.4420 | |
| NO3 | 0.1468 | 0.1004 | 1.462 | 0.1437 | |
| Fe | −22.0104 | 10.2563 | −2.146 | 0.0319 | ** |
| Mn | −23.3314 | 31..8584 | −0.7323 | 0.4640 |
| Parameters | Coefficient | Std. Error | z | p-Value | |
|---|---|---|---|---|---|
| ORPmV(−1) | 0.3596 | 0.03501 | 10.27 | <0.0001 | *** |
| const | 143.197 | 11.3490 | 12.62 | <0.0001 | *** |
| Sal | −0.0624 | 0.0087 | −7.178 | <0.0001 | *** |
| Temp | −0.2611 | 0.1027 | −2.542 | 0.0110 | ** |
| SWL | 0.9631 | 0.2057 | 4.684 | <0.0001 | *** |
| SO4 | 0.1294 | 0.0708 | 1.828 | 0.0675 | * |
| Cl | −0.2583 | 0.0347 | −7.446 | <0.0001 | *** |
| NO3 | 0.4514 | 0.0970 | 4.655 | <0.0001 | *** |
| Fe | −2.6130 | 1.4991 | −1.743 | 0.0814 | * |
| Mn | −0.8053 | 1.5327 | −0.5254 | 0.5993 |
| WHO Standard Classification | Wet Season Groundwater Samples N = 96 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameters | pH | EC | TDS | Cl | SO4 | NO3 | Fe | Mn | |
| Samples within WHO Limits | No. of Sample | 18 | 95 | 95 | 94 | 95 | 78 | 90 | 77 |
| % Sample | 18.75 | 98.96 | 98.96 | 97.9 | 98.96 | 81.3 | 93.8 | 80.2 | |
| Samples above WHO Permissible Limits | No. of Sample | 78 | 1 | 1 | 2 | 1 | 18 | 6 | 19 |
| % Sample | 81.1 | 1 | 1 | 2.1 | 1 | 18.8 | 6.3 | 19.8 | |
| Dry Season Groundwater Samples N = 134 | |||||||||
| Samples within WHO Limits | No. of Sample | 21 | 123 | 123 | 130 | 133 | 108 | 10 | 109 |
| % Sample | 15.8 | 92.3 | 92.3 | 99.6 | 99.6 | 82.4 | 7.5 | 81.8 | |
| Samples above WHO Permissible Limits | No. of Sample | 113 | 11 | 11 | 1 | 1 | 23 | 123 | 25 |
| % Sample | 84.2 | 7.7 | 7.7 | 0.4 | 0.4 | 17.6 | 92.5 | 18.2 | |
| WHO Permissible Std | 6.5–8.5 | 750 | 500 | 200 | 200 | 50 | 0.01 | 0.1 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aladejana, J.A.; Kalin, R.M.; Sentenac, P.; Hassan, I. Assessing the Impact of Climate Change on Groundwater Quality of the Shallow Coastal Aquifer of Eastern Dahomey Basin, Southwestern Nigeria. Water 2020, 12, 224. https://doi.org/10.3390/w12010224
Aladejana JA, Kalin RM, Sentenac P, Hassan I. Assessing the Impact of Climate Change on Groundwater Quality of the Shallow Coastal Aquifer of Eastern Dahomey Basin, Southwestern Nigeria. Water. 2020; 12(1):224. https://doi.org/10.3390/w12010224
Chicago/Turabian StyleAladejana, Jamiu A., Robert M. Kalin, Philippe Sentenac, and Ibrahim Hassan. 2020. "Assessing the Impact of Climate Change on Groundwater Quality of the Shallow Coastal Aquifer of Eastern Dahomey Basin, Southwestern Nigeria" Water 12, no. 1: 224. https://doi.org/10.3390/w12010224
APA StyleAladejana, J. A., Kalin, R. M., Sentenac, P., & Hassan, I. (2020). Assessing the Impact of Climate Change on Groundwater Quality of the Shallow Coastal Aquifer of Eastern Dahomey Basin, Southwestern Nigeria. Water, 12(1), 224. https://doi.org/10.3390/w12010224






