Batch and Column Scale Removal of Cadmium from Water Using Raw and Acid Activated Wheat Straw Biochar
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation and Characterization of Biochar
2.2. Measurement of Point of Zero Charge
2.3. Preparation of Synthetic Wastewater
2.4. Batch Experiment
2.4.1. Effect of Adsorbent Dose and pH
2.4.2. Effect of Initial Cd Concentration and Time
2.4.3. Adsorption and Removal Analysis
2.4.4. Modeling Equilibrium Sorption Data
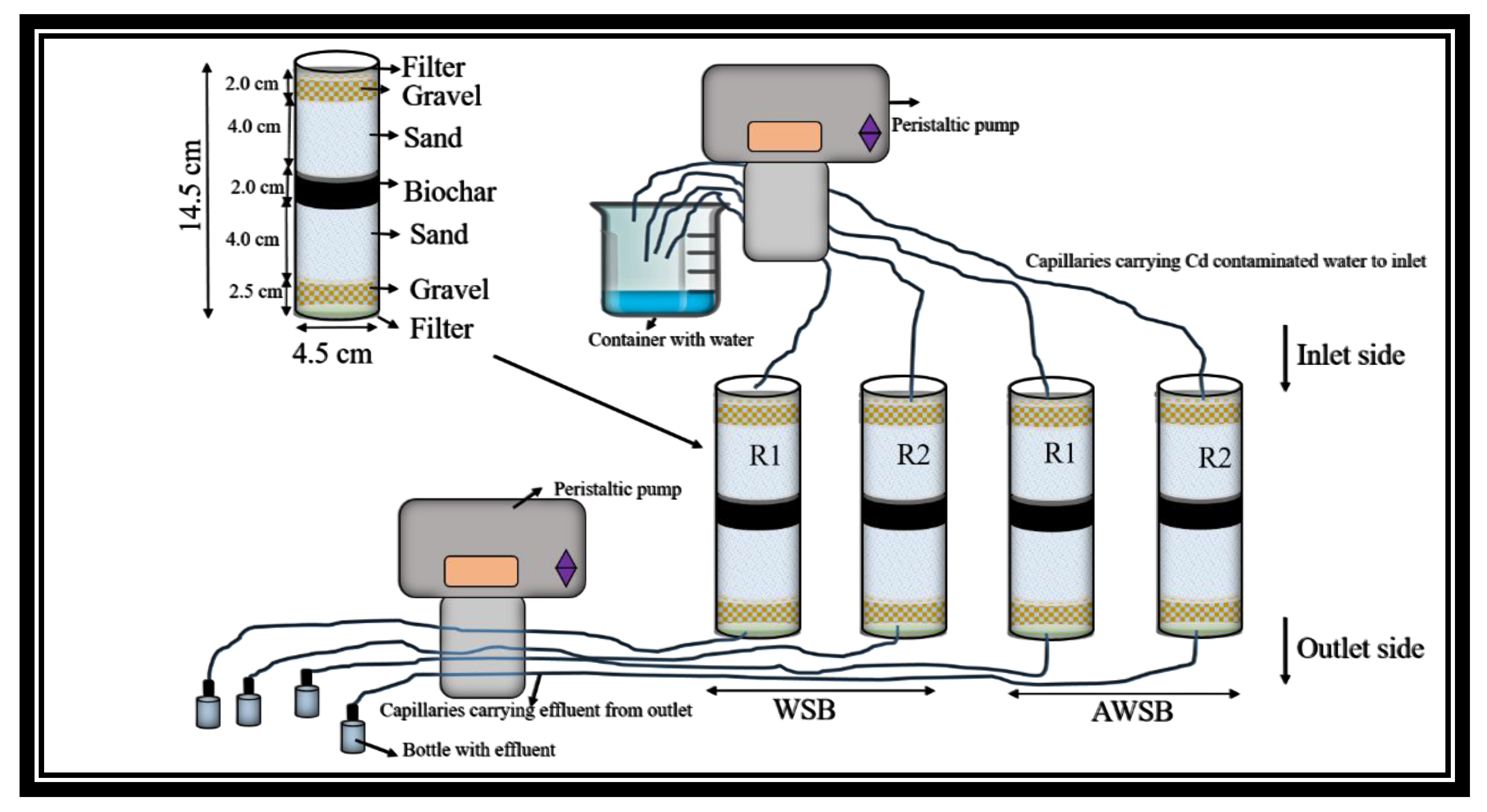
2.5. Laboratory Scale Column Experiments and Reusability of the Biochar
3. Results and Discussion
3.1. Characterization of Biochars
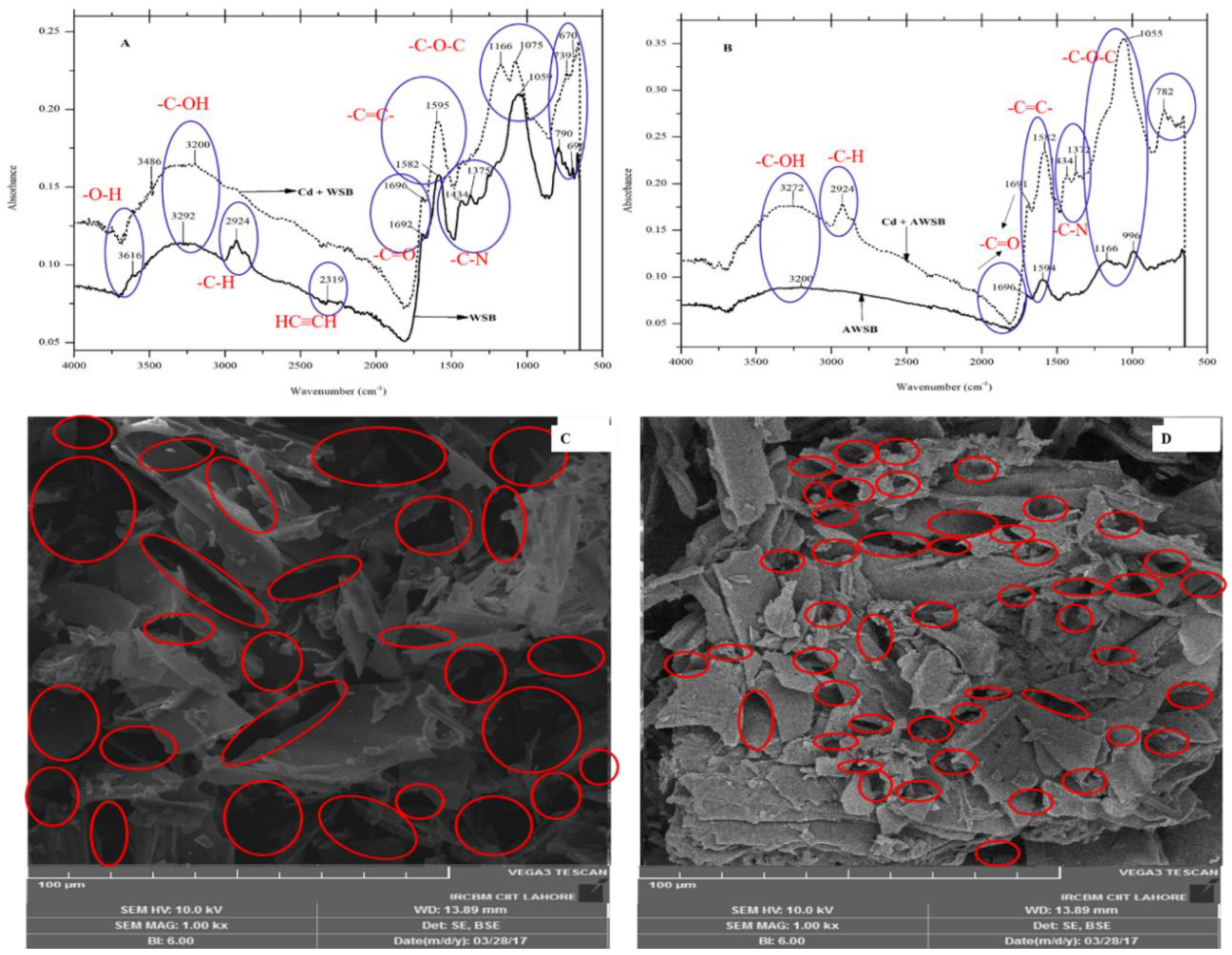
3.1.1. Fourier Transform Infrared Spectra (FTIR)
3.1.2. Scanning Electron Microscopy (SEM) and BET Analysis
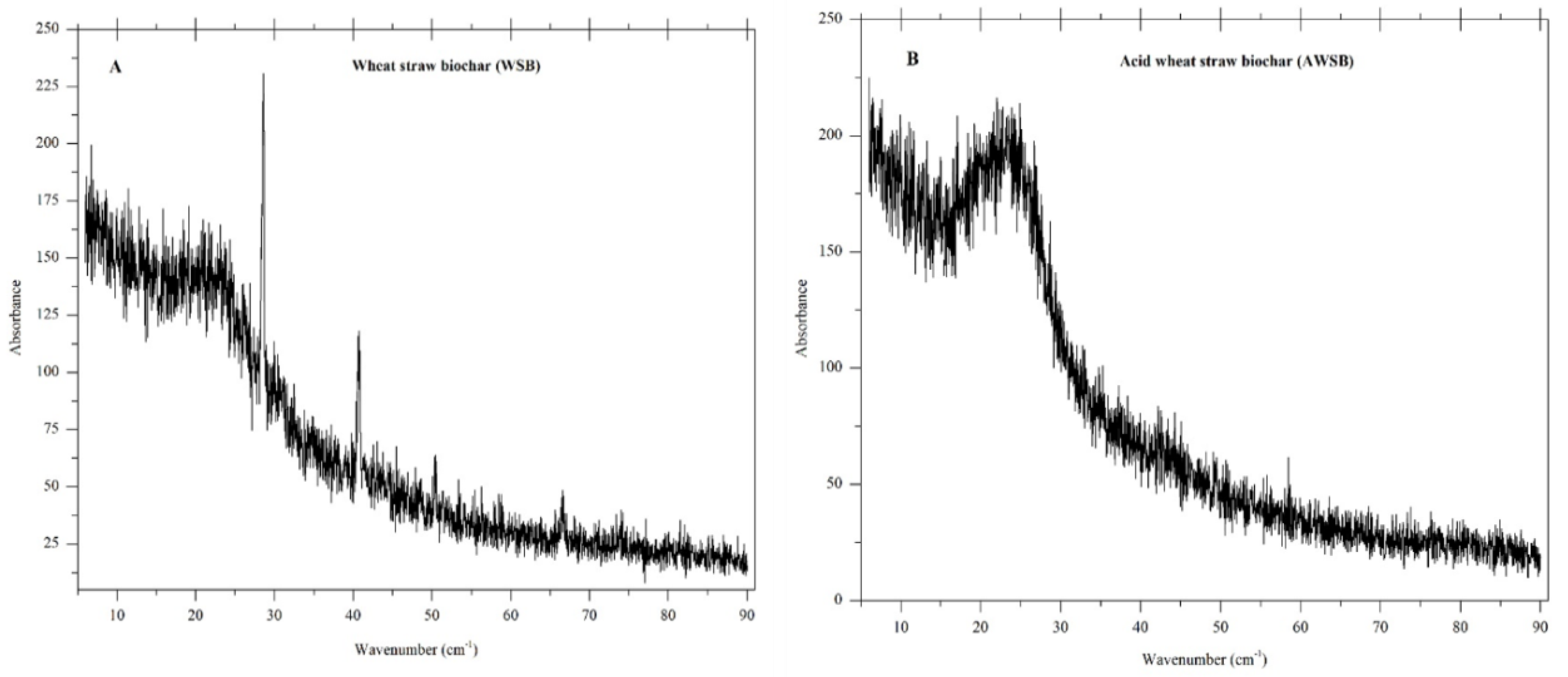
3.1.3. XRD Analysis
3.1.4. Elemental Analysis
3.2. Measurement of PZC
3.3. Optimization for Cd Removal and Adsorption
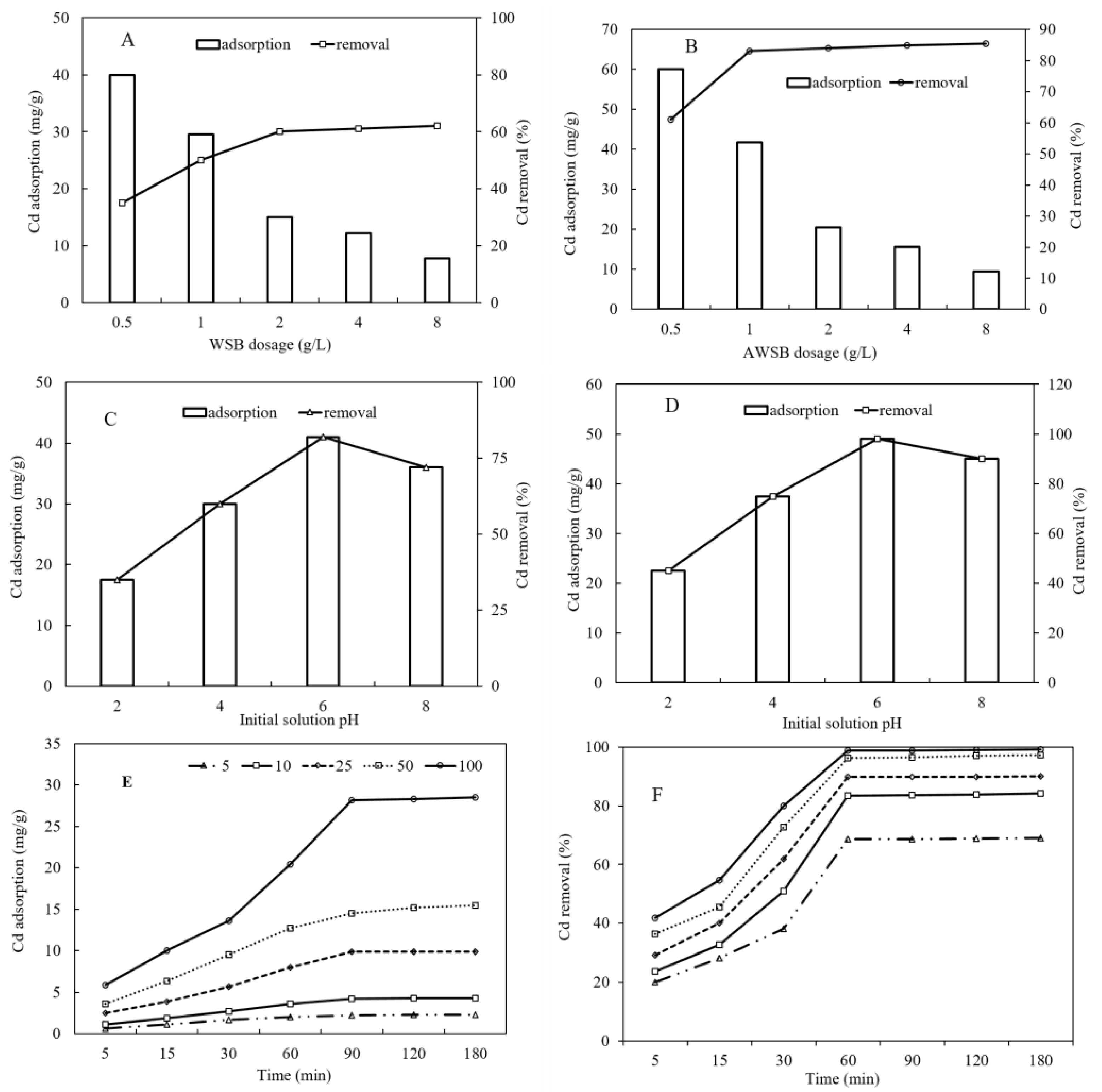
3.3.1. Effect of Adsorbent Dosage and pH
3.3.2. Effect of Initial Cd Concentration and Contact Time
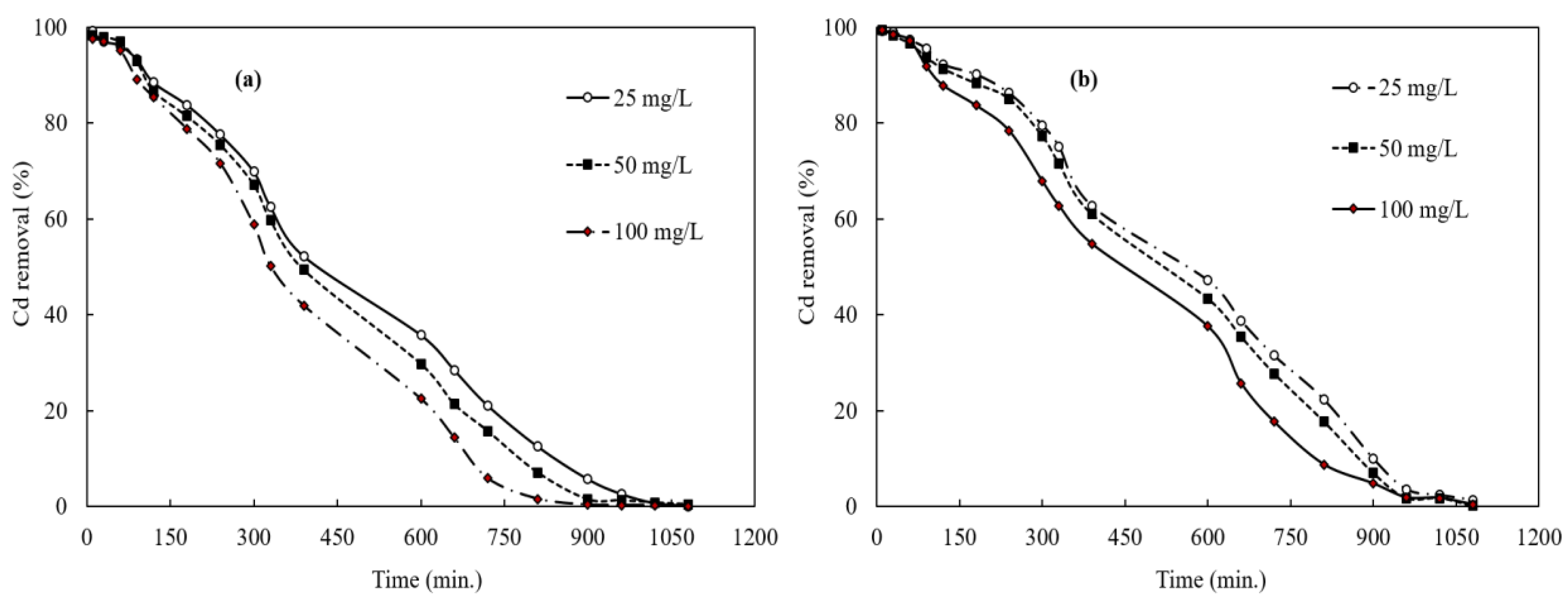
3.4. Cadmium Removal from Laboratory Scale Column Experiments and Reusability of The Biochar
3.5. Study of Adsorption Isotherm Modeling
3.6. Adsorption Kinetics of Cd
3.7. Comparison of Cd Sorption Capacity with Other Adsorbents
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rao, K.; Mohapatra, M.; Anand, S.; Venkateswarlu, P. Review on cadmium removal from aqueous solutions. Int. J. Eng. Sci. Technol. 2010, 2, 81–103. [Google Scholar] [CrossRef]
- Haris, M.R.; Wahab, N.A.; Reng, C.W.; Azahari, B.; Sathasivam, K. The sorption of cadmium (II) ions on mercerized rice husk and activated carbon. Turk. J. Chem. 2011, 35, 939–950. [Google Scholar]
- Sawalha, M.F.; Peralta-Videa, J.R.; Romero-González, J.; Gardea-Torresdey, J.L. Biosorption of Cd (II), Cr (III), and Cr (VI) by saltbush (Atriplex canescens) biomass: Thermodynamic and isotherm studies. J. Colloid Interface Sci. 2006, 300, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Chandra, V.; Park, J.; Chun, Y.; Lee, J.W.; Hwang, I.-C.; Kim, K.S. Water-Dispersible Magnetite-Reduced Graphene Oxide Composites for Arsenic Removal. ACS Nano 2010, 4, 3979–3986. [Google Scholar] [CrossRef] [PubMed]
- Hanigan, D.; Zhang, J.; Herckes, P.; Krasner, S.W.; Chen, C.; Westerhoff, P. Adsorption ofN-Nitrosodimethylamine Precursors by Powdered and Granular Activated Carbon. Environ. Sci. Technol. 2012, 46, 12630–12639. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Min, Q.; Wang, W.; He, L.; Zheng, J. Impact of household social-economic characteristics on the willingness to grow crops: A case study of jasmine growers in Fuzhou based on conservation of the agricultural heritage system. Zhongguo Shengtai Nongye Xuebao/Chin. J. Eco-Agric. 2016, 24, 1714–1721. [Google Scholar]
- Mohan, D.; Pittman, C.U.; Bricka, M.; Smith, F.; Yancey, B.; Mohammad, J.; Steele, P.H.; Alexandre-Franco, M.F.; Gómez-Serrano, V.; Gong, H.; et al. Sorption of arsenic, cadmium, and lead by chars produced from fast pyrolysis of wood and bark during bio-oil production. J. Colloid Interface Sci. 2007, 310, 57–73. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.P.; Wu, S.; Chong, K.-H. Surface modification of a granular activated carbon by citric acid for enhancement of copper adsorption. Carbon 2003, 41, 1979–1986. [Google Scholar] [CrossRef]
- Rivera-Utrilla, J.; Sánchez-Polo, M.; Gómez-Serrano, V.; Álvarez, P.; Alvim-Ferraz, M.; Dias, J.M.; José, R.-U.; Alvarez, P.; Alvim-Ferraz, M.D.C. Activated carbon modifications to enhance its water treatment applications. An overview. J. Hazard. Mater. 2011, 187, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, W.-J.; Zhang, N.; Li, Y.-S.; Jiang, H.; Sheng, G.-P. Polyethylenimine modified biochar adsorbent for hexavalent chromium removal from the aqueous solution. Bioresour. Technol. 2014, 169, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Wang, X.; Yin, D.; Peng, B.; Tan, C.; Liu, Y.; Tan, X.; Wu, S. Efficiency and mechanisms of Cd removal from aqueous solution by biochar derived from water hyacinth (Eichornia crassipes). J. Environ. Manag. 2015, 153, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Crannell, B.S.; Eighmy, T.T.; Krzanowski, J.E.; Eusden, J.D., Jr.; Shaw, E.L.; Francis, C.A. Heavy metal stabilization in municipal solid waste combustion bottom ash using soluble phosphate. Waste Manag. 2000, 20, 135–148. [Google Scholar] [CrossRef]
- Seshadri, B.; Bolan, N.; Choppala, G.; Kunhikrishnan, A.; Sanderson, P.; Wang, H.; Currie, L.; Tsang, D.C.; Ok, Y.; Kim, G.; et al. Potential value of phosphate compounds in enhancing immobilization and reducing bioavailability of mixed heavy metal contaminants in shooting range soil. Chemosphere 2017, 184, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Saleh, T.A.; Sulaiman, K.O.; Al-Hammadi, S.A.; Dafalla, H.; Danmaliki, G.I. Adsorptive desulfurization of thiophene, benzothiophene and dibenzothiophene over activated carbon manganese oxide nanocomposite: With column system evaluation. J. Clean. Prod. 2017, 154, 401–412. [Google Scholar] [CrossRef]
- Sánchez, M.; Lindao, E.; Margaleff, D.; Martínez, O.; Morán, A. Pyrolysis of agricultural residues from rape and sunflowers: Production and characterization of bio-fuels and biochar soil management. J. Anal. Appl. Pyrolysis 2009, 85, 142–144. [Google Scholar] [CrossRef]
- Lima, I.M.; Boykin, D.L.; Klasson, K.T.; Uchimiya, M. Influence of post-treatment strategies on the properties of activated chars from broiler manure. Chemosphere 2014, 95, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Aliabadi, H.M.; Saberikhah, E.; Pirbazari, A.E.; Khakpour, R.; Alipour, H. Triethoxysilylpropylamine modified alkali treated wheat straw: An efficient adsorbent for methyl orange adsorption. Cellul. Chem. Technol. 2018, 52, 129–140. [Google Scholar]
- Edokpayi, J.N.; Odiyo, J.O.; Msagati, T.A.M.; Popoola, E.O. A Novel Approach for the Removal of Lead(II) Ion from Wastewater Using Mucilaginous Leaves of Diceriocaryum eriocarpum Plant. Sustainability 2015, 7, 14026–14041. [Google Scholar] [CrossRef]
- Imran, M.; Suddique, M.; Shah, G.M.; Ahmad, I.; Murtaza, B.; Shah, N.S.; Mubeen, M.; Ahmad, S.; Zakir, A.; Schotting, R.J. Kinetic and equilibrium studies for cadmium biosorption from contaminated water using Cassia fistula biomass. Int. J. Environ. Sci. Technol. 2018, 16, 3099–3108. [Google Scholar] [CrossRef]
- Babarinde, A.; Onyiaocha, G.O. Equilibrium sorption of divalent metal ions onto groundnut (Arachis hypogaea) shell: Kinetics, isotherm and thermodynamics. Chem. Int. 2016, 2, 37–46. [Google Scholar]
- Shah, G.M.; Nasir, M.; Imran, M.; Bakhat, H.F.; Rabbani, F.; Sajjad, M.; Farooq, A.B.U.; Ahmad, S.; Song, L.; Eroglu, E. Biosorption potential of natural, pyrolysed and acid-assisted pyrolysed sugarcane bagasse for the removal of lead from contaminated water. PeerJ 2018, 6, e5672. [Google Scholar] [CrossRef] [PubMed]
- Hesas, R.H.; Arami-Niya, A.; Daud, W.M.; Sahu, J.N. Preparation and Characterization of Activated Carbon from Apple Waste by Microwave-Assisted Phosphoric Acid Activation: Application in Methylene Blue Adsorption. BioResources 2013, 8, 2950–2966. [Google Scholar]
- Suárez-García, F.; Martínez-Alonso, A.; Tascón, J. Pyrolysis of apple pulp: Effect of operation conditions and chemical additives. J. Anal. Appl. Pyrolysis 2002, 62, 93–109. [Google Scholar] [CrossRef]
- Regmi, P.; Moscoso, J.L.G.; Kumar, S.; Cao, X.; Mao, J.; Schafran, G. Removal of copper and cadmium from aqueous solution using switchgrass biochar produced via hydrothermal carbonization process. J. Environ. Manag. 2012, 109, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Huo, X.; Zhu, Y.; Ji, H. Chemisorption and Physical Adsorption Roles in Cadmium Biosorption by Chlamydomonas Reinhardtii. Chin. J. Popul. Resour. Environ. 2010, 8, 54–58. [Google Scholar]
- Deng, Y.; Huang, S.; Laird, D.A.; Wang, X.; Dong, C. Quantitative mechanisms of cadmium adsorption on rice straw- and swine manure-derived biochars. Environ. Sci. Pollut. Res. 2018, 25, 32418–32432. [Google Scholar] [CrossRef] [PubMed]
- Tangjuank, S.; Insuk, N.; Tontrakoon, J.; Udeye, V. Adsorption of lead (II) and cadmium (II) ions from aqueous solutions by adsorption on activated carbon prepared from cashew nut shells. World Acad. Sci. Eng. Technol. 2009, 52, 110–116. [Google Scholar]
- Keiluweit, M.; Nico, P.S.; Johnson, M.G.; Kleber, M. Dynamic Molecular Structure of Plant Biomass-Derived Black Carbon (Biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef]
- Azargohar, R.; Dalai, A. Steam and KOH activation of biochar: Experimental and modeling studies. Microporous Mesoporous Mater. 2008, 110, 413–421. [Google Scholar] [CrossRef]
- Prapagdee, S.; Piyatiratitivorakul, S.; Petsom, A. Activation of Cassava Stem Biochar by Physico-Chemical Method for Stimulating Cadmium Removal Efficiency from Aqueous Solution. Environ. Asia 2014, 7, 60–69. [Google Scholar]
- Takagi, H.; Maruyama, K.; Yoshizawa, N.; Yamada, Y.; Sato, Y. XRD analysis of carbon stacking structure in coal during heat treatment. Fuel 2004, 83, 2427–2433. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, X.; Li, J.; Ma, D.; Han, R. Characterization of bio-char from pyrolysis of wheat straw and its evaluation on methylene blue adsorption. Desalin. Water Treat. 2012, 46, 115–123. [Google Scholar] [CrossRef]
- Al-Degs, Y.; Elbarghouthi, M.; Elsheikh, A.; Walker, G. Effect of solution pH, ionic strength, and temperature on adsorption behavior of reactive dyes on activated carbon. Dye. Pigment. 2008, 77, 16–23. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, T.; Tang, J.; Zheng, Z.; Wang, H.; Shao, Q.; Chen, G.; Li, Z.; Chen, Y.; Zhu, J.; et al. Characteristics and mechanisms of cadmium adsorption from aqueous solution using lotus seedpod-derived biochar at two pyrolytic temperatures. Environ. Sci. Pollut. Res. 2018, 25, 11854–11866. [Google Scholar] [CrossRef] [PubMed]
- Usman, A.; Sallam, A.; Zhang, M.; Vithanage, M.; Ahmad, M.; Al-Farraj, A.; Ok, Y.S.; Abduljabbar, A.; Al-Wabel, M. Sorption process of date palm biochar for aqueous Cd (II) removal: Efficiency and mechanisms. Water Air Soil Pollut. 2016, 227, 449. [Google Scholar] [CrossRef]
- Ahmad, M.; Lee, S.S.; Dou, X.; Mohan, D.; Sung, J.-K.; Yang, J.E.; Ok, Y.S. Effects of pyrolysis temperature on soybean stover-and peanut shell-derived biochar properties and TCE adsorption in water. Bioresour. Technol. 2012, 118, 536–544. [Google Scholar] [CrossRef]
- Ahmad, M.; Moon, D.H.; Vithanage, M.; Koutsospyros, A.; Lee, S.S.; Yang, J.E.; Lee, S.E.; Jeon, C.; Ok, Y.S. Production and use of biochar from buffalo-weed (Ambrosia trifida L.) for trichloroethylene removal from water. J. Chem. Technol. Biotechnol. 2014, 89, 150–157. [Google Scholar] [CrossRef]
- Ahmad, I.; Akhtar, M.J.; Jadoon, I.B.K.; Imran, M.; Ali, S. Equilibrium modeling of cadmium biosorption from aqueous solution by compost. Environ. Sci. Pollut. Res. 2017, 24, 5277–5284. [Google Scholar] [CrossRef]
- Boudrahem, F.; Soualah, A.; Aissani-Benissad, F. Pb(II) and Cd(II) Removal from Aqueous Solutions Using Activated Carbon Developed from Coffee Residue Activated with Phosphoric Acid and Zinc Chloride. J. Chem. Eng. Data 2011, 56, 1946–1955. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Wnetrzak, R.; Leahy, J.J.; Hayes, M.; Kwapinski, W.; Hubicki, Z. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Sallam, A.S.; Al-Omran, A.; El-Naggar, A.H.; Nadeem, M.; Al-Wabel, M.I.; Usman, A.R.; Alenazi, K.K. Chemically Modified Biochar Produced from Conocarpus Wastes: An Efficient Sorbent for Fe(II) Removal from Acidic Aqueous Solutions. Adsorpt. Sci. Technol. 2013, 31, 625–640. [Google Scholar]
- Jiang, K.; Sun, T.-H.; Sun, L.-N.; Li, H.-B. Adsorption characteristics of copper, lead, zinc and cadmium ions by tourmaline. J. Environ. Sci. 2006, 18, 1221–1225. [Google Scholar] [CrossRef]
- Guo, Z.; Liu, X.; Huang, H. Kinetics and Thermodynamics of Reserpine Adsorption onto Strong Acidic Cationic Exchange Fiber. PLoS ONE 2015, 10, e0138619. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Wu, Z.; Manzoli, M.; Jicsinszky, L.; Wu, Z.; Nosyrev, A.E.; Cravotto, G. Adsorptive Recovery of Iopamidol from Aqueous Solution and Parallel Reuse of Activated Carbon: Batch and Flow Study. Ind. Eng. Chem. Res. 2019, 58, 7284–7295. [Google Scholar] [CrossRef]
- Kim, W.-K.; Shim, T.; Kim, Y.-S.; Hyun, S.; Ryu, C.; Park, Y.-K.; Jung, J. Characterization of cadmium removal from aqueous solution by biochar produced from a giant Miscanthus at different pyrolytic temperatures. Bioresour. Technol. 2013, 138, 266–270. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, W.; Yang, Y.; Huang, X.; Wang, S.; Qiu, R. Relative distribution of Pb2+ sorption mechanisms by sludge-derived biochar. Water Res. 2012, 46, 854–862. [Google Scholar] [CrossRef]
- Inyang, M.; Gao, B.; Yao, Y.; Xue, Y.; Zimmerman, A.R.; Pullammanappallil, P.; Cao, X. Removal of heavy metals from aqueous solution by biochars derived from anaerobically digested biomass. Bioresour. Technol. 2012, 110, 50–56. [Google Scholar] [CrossRef]
- Salam, O.E.A.; Reiad, N.A.; Elshafei, M.M. A study of the removal characteristics of heavy metals from wastewater by low-cost adsorbents. J. Adv. Res. 2011, 2, 297–303. [Google Scholar] [CrossRef]
- Guo, W.; Xiong, W.; Gao, S.; Hu, Z.; Liu, H.; Yu, R. Impact of temperature on the isothermal adsorption/desorption of shale gas. Pet. Explor. Dev. 2013, 40, 514–519. [Google Scholar] [CrossRef]
- Sun, Y.; Gao, B.; Yao, Y.; Fang, J.; Zhang, M.; Zhou, Y.; Chen, H.; Yang, L. Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem. Eng. J. 2014, 240, 574–578. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, H.; Cai, J.; Zhang, X.; Zhang, J.; Shao, J. Evaluation and Prediction of Cadmium Removal from Aqueous Solution by Phosphate-Modified Activated Bamboo Biochar. Energy Fuels 2017, 32, 4469–4477. [Google Scholar] [CrossRef]
- Cui, X.; Fang, S.; Yao, Y.; Li, T.; Ni, Q.; Yang, X.; He, Z. Potential mechanisms of cadmium removal from aqueous solution by Canna indica derived biochar. Sci. Total. Environ. 2016, 562, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Goswami, R.; Shim, J.; Deka, S.; Kumari, D.; Kataki, R.; Kumar, M. Characterization of cadmium removal from aqueous solution by biochar produced from Ipomoea fistulosa at different pyrolytic temperatures. Ecol. Eng. 2016, 97, 444–451. [Google Scholar] [CrossRef]
- Xu, X.; Cao, X.; Zhao, L.; Wang, H.; Yu, H.; Gao, B. Removal of Cu, Zn, and Cd from aqueous solutions by the dairy manure-derived biochar. Environ. Sci. Pollut. Res. 2013, 20, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Shen, K.; Long, X.; Wen, J.; Xie, X.; Zeng, X.; Liang, Y.; Wei, Y.; Lin, Z.; Huang, W.; et al. Preparation and Characterization of Biochars from Eichornia crassipes for Cadmium Removal in Aqueous Solutions. PLoS ONE 2016, 11, 0148132. [Google Scholar] [CrossRef] [PubMed]
- Trakal, L.; Bingöl, D.; Pohořelý, M.; Hruška, M.; Komárek, M. Geochemical and spectroscopic investigations of Cd and Pb sorption mechanisms on contrasting biochars: Engineering implications. Bioresour. Technol. 2014, 171, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Liang, C.-F.; Li, T.-Q.; Wang, K.; Huang, H.-G.; Yang, X.-E. Simultaneous removal of cadmium and sulfamethoxazole from aqueous solution by rice straw biochar. J. Zhejiang Univ. Sci. B 2013, 14, 640–649. [Google Scholar] [CrossRef] [PubMed]
- Yakkala, K.; Yu, M.-R.; Roh, H.; Yang, J.-K.; Chang, Y.-Y. Buffalo weed (Ambrosia trifida L. var. trifida) biochar for cadmium (II) and lead (II) adsorption in single and mixed system. Desalin. Water Treat. 2013, 51, 7732–7745. [Google Scholar] [CrossRef]
| Parameters | WSB | AWSB |
|---|---|---|
| Moisture % | 5.83 ± 0.18 | 4.25 ± 0.25 |
| Volatile matters % | 23.23 ± 0.25 | 41.50 ± 0.50 |
| Fixed carbon % | 64.43 ± 0.71 | 44.30 ± 0.50 |
| Ash content % | 12.50 ± 0.50 | 14.23 ± 0.25 |
| C (mg/g) | 74.27 ± 0.70 | 68.23 ± 0.55 |
| H (mg/g) | 5.90 ± 0.18 | 2.17 ± 0.30 |
| O (mg/g) | 14.48 ± 0.50 | 25.50 ± 0.50 |
| P (mg/g) | 0.54 ± 0.11 | 3.69 ± 0.20 |
| K (mg/g) | 10.43 ± 0.25 | 0.30 ± 0.20 |
| Ca (mg/g) | 8.95 ± 0.20 | 3.82 ± 0.20 |
| Mg (mg/g) | 4.01 ± 0.20 | 1.25 ± 0.25 |
| S (mg/g) | 0.63 ± 0.13 | 0.31 ± 0.20 |
| Na (mg/g) | 0.97 ± 0.13 | 0.12 ± 0.08 |
| Fe (mg/g) | 8.20 ± 0.20 | 6.24 ± 0.25 |
| pH | 7.95 ± 0.10 | 3.7 ± 0.13 |
| SBET (m2/g) | 207.33 ± 12.01 | 471.67 ± 17.56 |
| Vt (cm3/g) | 0.12 ± 0.04 | 0.45 ± 0.05 |
| Dp (nm) | 5.94 ± 0.22 | 11.25 ± 0.25 |
| Models | Langmuir | Freundlich | Temkin | Dubinin-Radushkevich | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | KL | Qmax | R2 | KF | n | R2 | KT | B | R2 | qm | kDR | E | R2 |
| WSB | 0.871 | 31.65 | 0.849 | 3.46 | 1.87 | 0.982 | 1.92 | 478.3 | 0.856 | 13.33 | 0.247 | 1.4 | 0.694 |
| AWSB | 0.271 | 74.63 | 0.967 | 15.95 | 2.37 | 0.996 | 12.55 | 255.9 | 0.870 | 32.79 | 0.038 | 3.6 | 0.689 |
| Models | Pseudo-First Order | Pseudo-Second Order | Intra-Particle Diffusion | Elovich | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | k1 | qe | R2 | k2 | qe | R2 | kdif | C | R2 | α | β | R2 |
| WSB | 0.239 | 11.95 | 0.803 | 0.0025 | 16.18 | 0.987 | 0.545 | 5.54 | 0.725 | 4.49 | 0.658 | 0.879 |
| AWSB | 0.514 | 26.38 | 0.743 | 0.0030 | 34.84 | 0.996 | 1.10 | 15.38 | 0.550 | 5.05 | 0.356 | 0.725 |
| Feedstock | Qmax | Reference |
|---|---|---|
| Water hyacinth | 70.30 | [11] |
| KOH activated Cassava stem | 24.88 | [30] |
| Cassava stem | 10.46 | [30] |
| Peanut shells | 32.00 | [37] |
| Phosphate modified activated bamboo | 202.55 | [52] |
| Bamboo | 24.95 | [52] |
| Canna indica | 188.80 | [53] |
| Ipomoea fistulosa | 41.67–72.43 | [54] |
| Dairy manure | 54.60 | [55] |
| Eichornia Crassipes | 49.84 | [56] |
| Wheat straw | 45.00 | [57] |
| Grape husk | 29.20 | [57] |
| Rice straw | 34.13 | [58] |
| Buffalo weed | 11.63 | [59] |
| Wheat straw | 31.65–74.63 | Present study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naeem, M.A.; Imran, M.; Amjad, M.; Abbas, G.; Tahir, M.; Murtaza, B.; Zakir, A.; Shahid, M.; Bulgariu, L.; Ahmad, I. Batch and Column Scale Removal of Cadmium from Water Using Raw and Acid Activated Wheat Straw Biochar. Water 2019, 11, 1438. https://doi.org/10.3390/w11071438
Naeem MA, Imran M, Amjad M, Abbas G, Tahir M, Murtaza B, Zakir A, Shahid M, Bulgariu L, Ahmad I. Batch and Column Scale Removal of Cadmium from Water Using Raw and Acid Activated Wheat Straw Biochar. Water. 2019; 11(7):1438. https://doi.org/10.3390/w11071438
Chicago/Turabian StyleNaeem, Muhammad Asif, Muhammad Imran, Muhammad Amjad, Ghulam Abbas, Muhammad Tahir, Behzad Murtaza, Ali Zakir, Muhammad Shahid, Laura Bulgariu, and Iftikhar Ahmad. 2019. "Batch and Column Scale Removal of Cadmium from Water Using Raw and Acid Activated Wheat Straw Biochar" Water 11, no. 7: 1438. https://doi.org/10.3390/w11071438
APA StyleNaeem, M. A., Imran, M., Amjad, M., Abbas, G., Tahir, M., Murtaza, B., Zakir, A., Shahid, M., Bulgariu, L., & Ahmad, I. (2019). Batch and Column Scale Removal of Cadmium from Water Using Raw and Acid Activated Wheat Straw Biochar. Water, 11(7), 1438. https://doi.org/10.3390/w11071438









