Dynamics of Calanus Copepodite Structure during Little Auks’ Breeding Seasons in Two Different Svalbard Locations
Abstract
1. Introduction
2. Materials and Methods
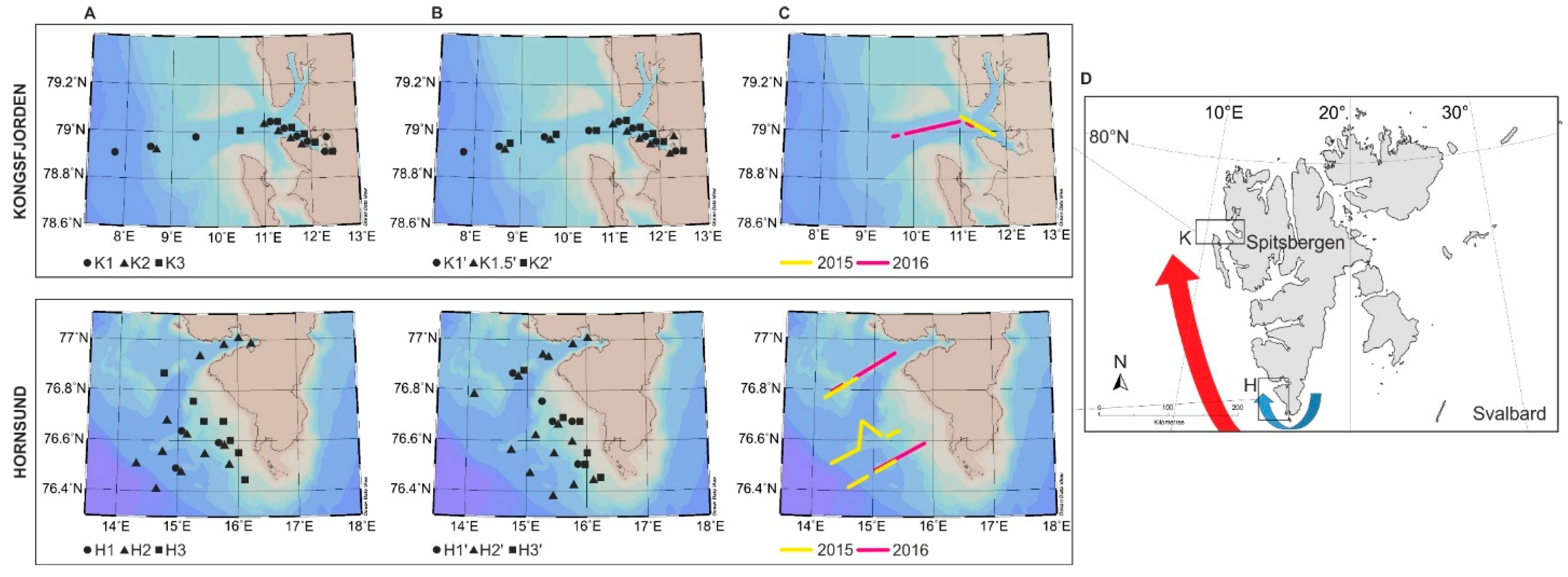
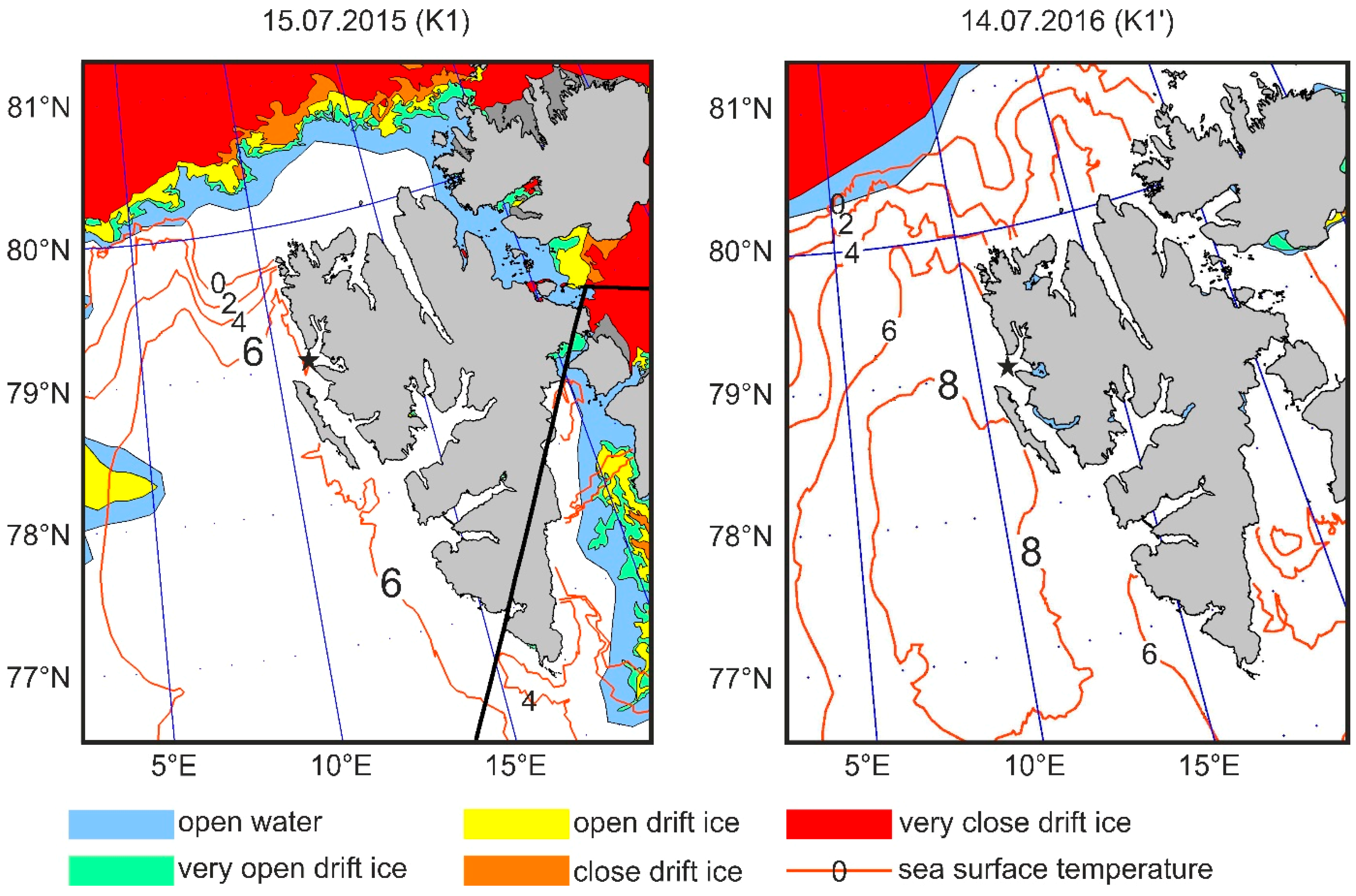
2.1. Study Area
2.2. Sampling Protocol
2.3. Laboratory Analyses
2.4. Temperature Measurements
2.5. LOPC Measurements
2.6. Data Analyses
3. Results
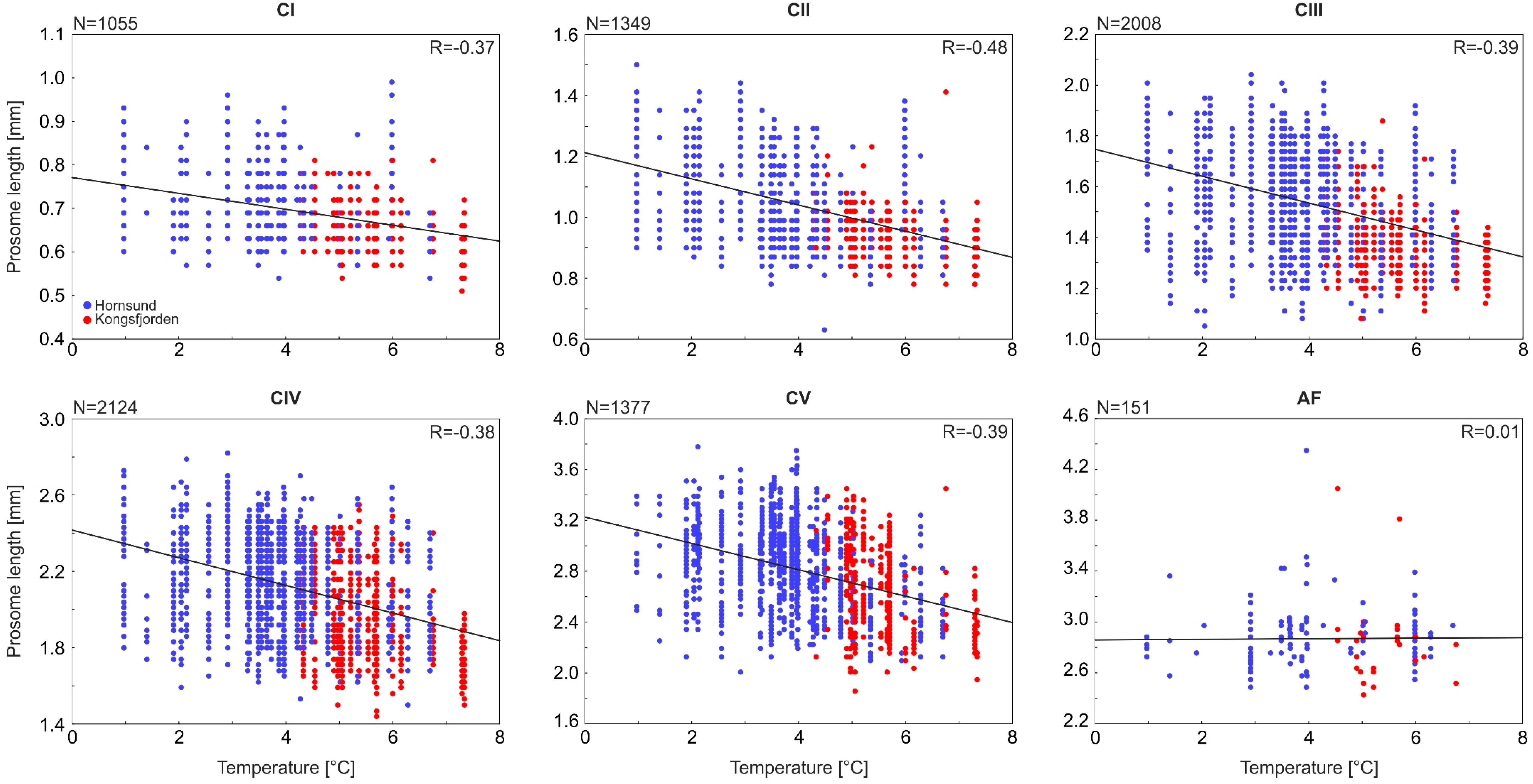
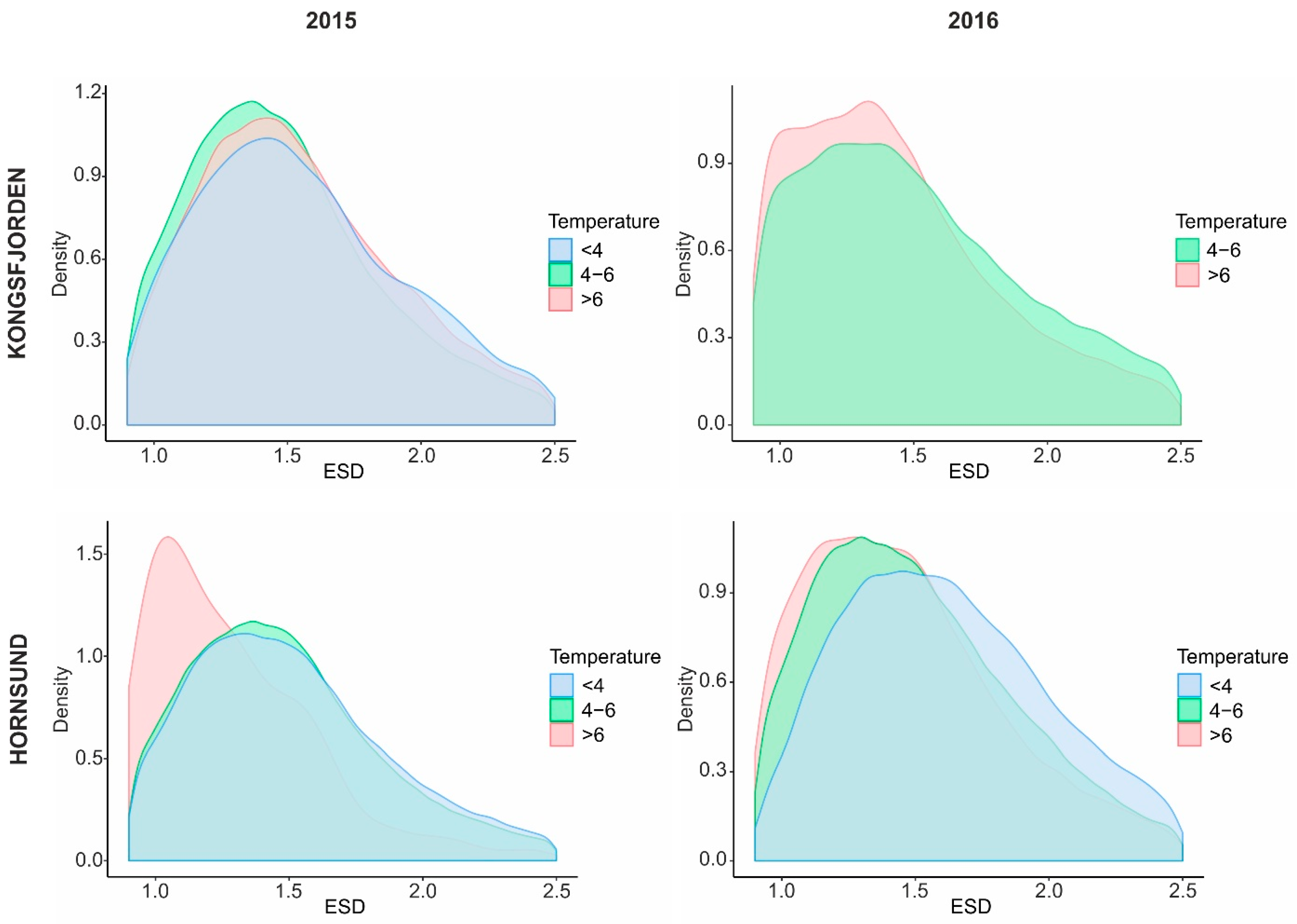
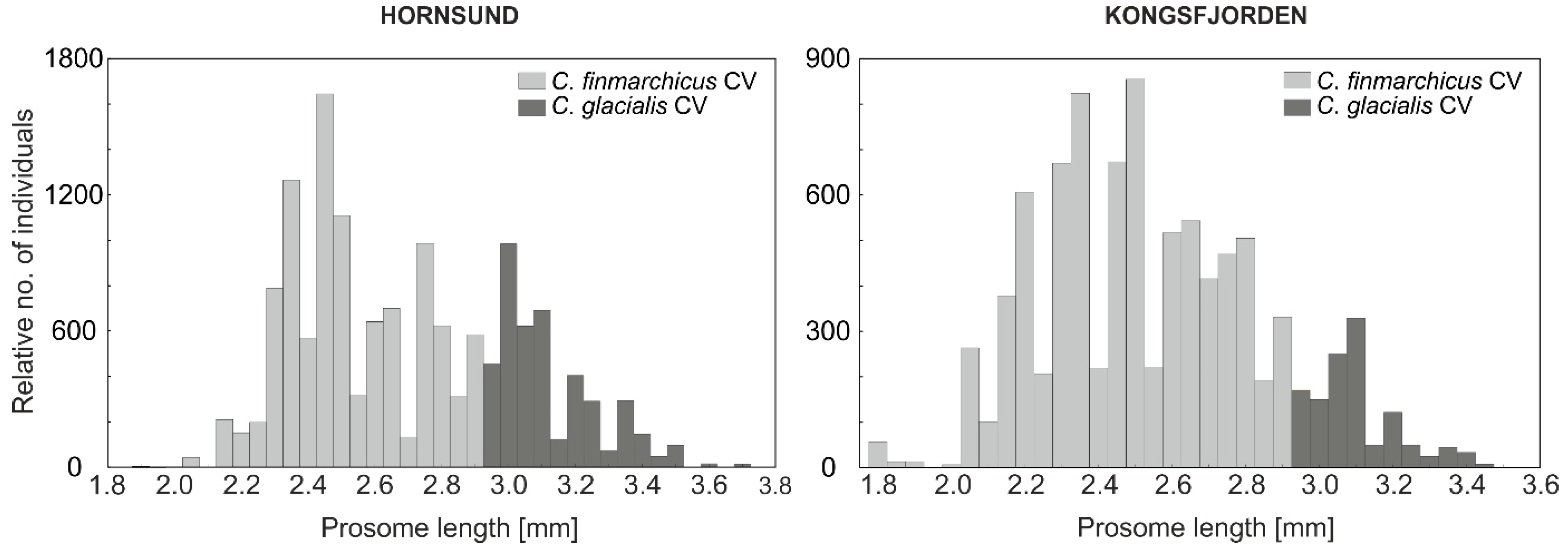
3.1. Size Response of Calanus and Mesozooplankton to Different Seawater Temperatures
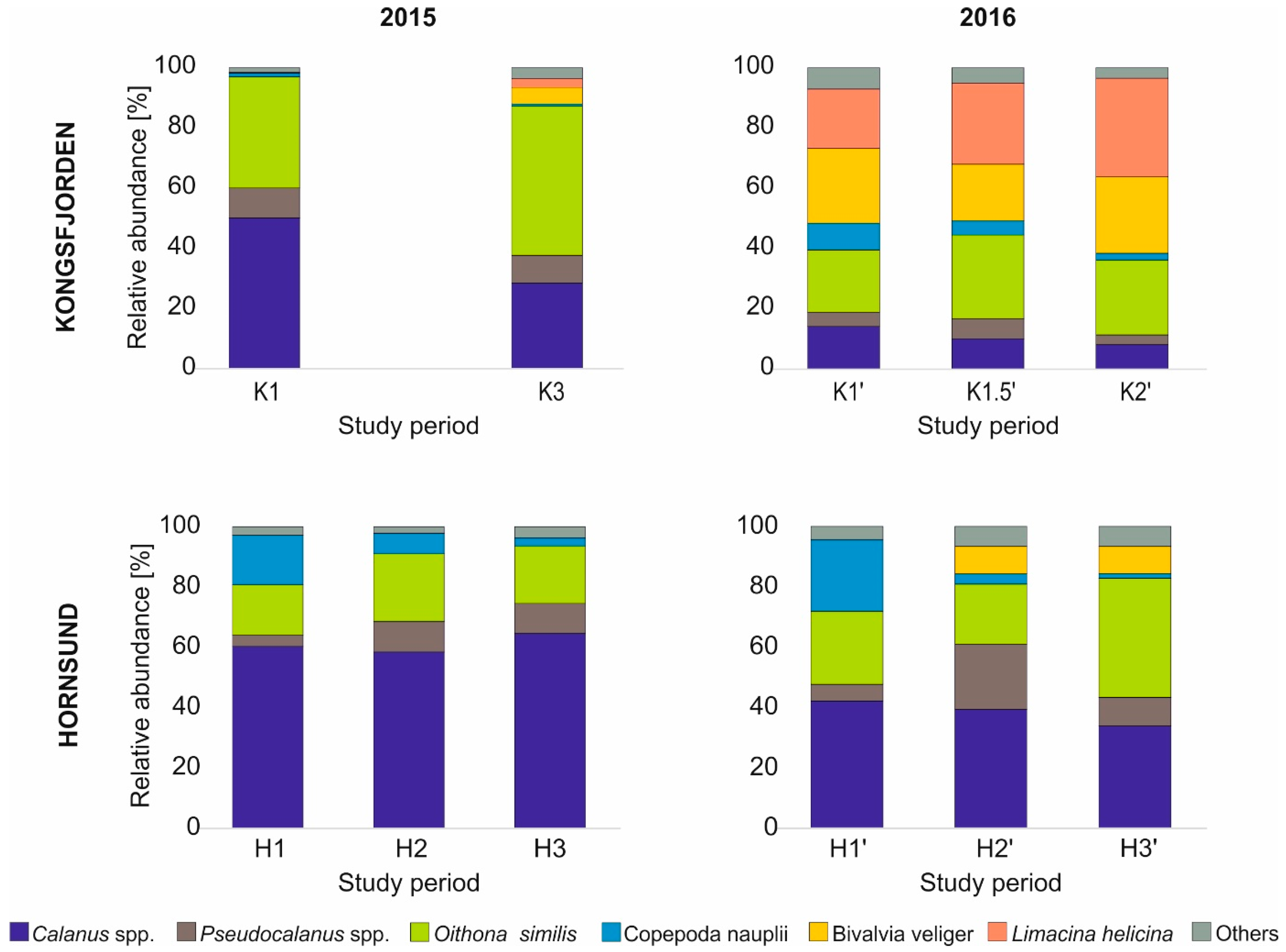
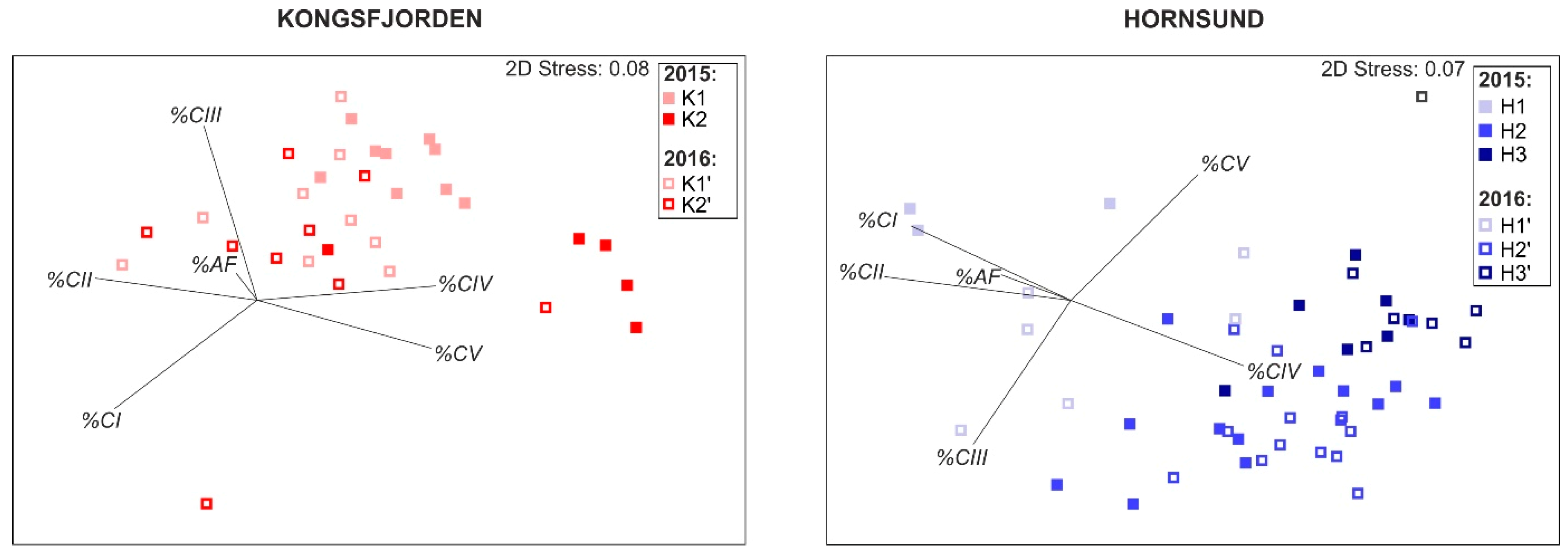
3.2. Zooplankton Community Structure
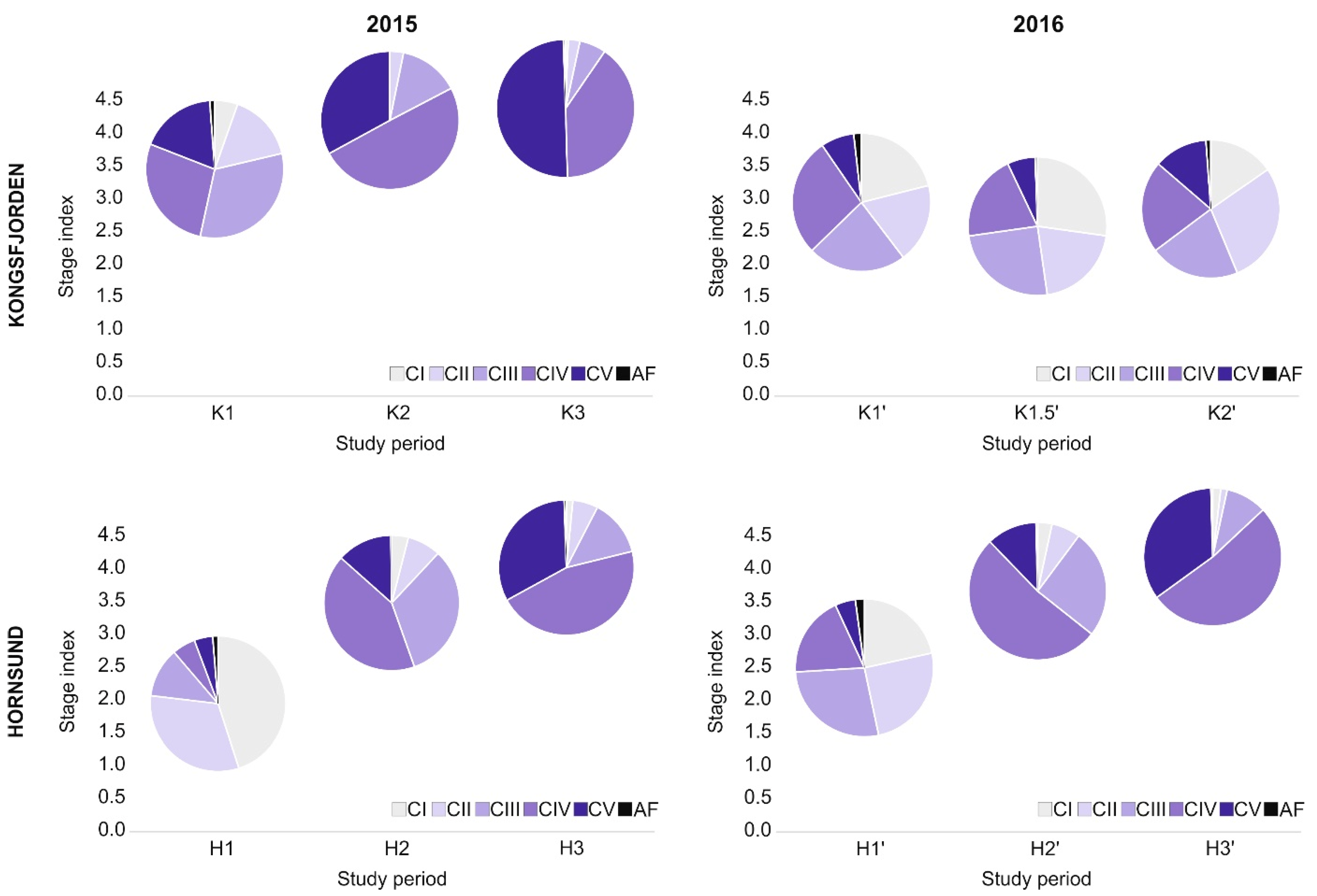
3.3. Calanus Copepodite Structure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Błachowiak-Samołyk, K.; Søreide, J.E.; Kwasniewski, S.; Sundfjord, A.; Hop, H.; Falk-Petersen, S.; Hegseth, E.N. Hydrodynamic control of mesozooplankton abundance and biomass in northern Svalbard waters (79–81°N). Deep Sea Res. Part II Top. Stud. Oceanogr. 2008, 55, 2310–2324. [Google Scholar] [CrossRef]
- Carstensen, J.; Weydmann, A.; Olszewska, A.; Kwasniewski, S. Effects of environmental conditions on the biomass of Calanus spp. in the Nordic Seas. J. Plankton Res. 2012, 34, 951–966. [Google Scholar] [CrossRef]
- Aarflot, J.M.; Skjodal, H.R.; Dalpadado, P.; Skern-Mauritzen, M. Contribution of Calanus species to the mesozooplankton biomass in the Barents Sea. ICES J. Mar. Sci. 2017, 75, 2342–2354. [Google Scholar] [CrossRef]
- Scott, C.L.; Kwasniewski, S.; Falk-Petersen, S.; Sargent, J.R. Lipids and life strategies of Calanus finmarchicus, Calanus glacialis and Calanus hyperboreus in late autumn, Kongsfjorden, Svalbard. Polar Biol. 2000, 23, 510–516. [Google Scholar] [CrossRef]
- Falk-Petersen, S.; Mayzaud, P.; Kattner, G.; Sargent, J. Lipids and life strategy of Arctic Calanus. Mar. Biol. Res. 2009, 5, 18–39. [Google Scholar] [CrossRef]
- Daase, M.; Falk-Petersen, S.; Varpe, Ø.; Darnis, G.; Søreide, J.E.; Wold, A.; Leu, E.; Berge, J.; Philippe, B.; Fortier, L. Timing of reproductive events in the marine copepod Calanus glacialis: A pan-Arctic perspective. Can. J. Fish. Aquat. Sci. 2013, 70, 871–884. [Google Scholar] [CrossRef]
- Melle, W.; Runge, J.; Head, E.; Plourde, S.; Castellani, C.; Licandro, P.; Pierson, J.; Jónasdóttir, S.; Johnson, C.; Broms, C.; et al. The North Atlantic Ocean as habitat for Calanus finmarchicus: Environmental factors and life history traits. Prog. Oceanogr. 2014, 129, 244–284. [Google Scholar] [CrossRef]
- Kubiszyn, A.M.; Piwosz, K.; Wiktor, J.M., Jr.; Wiktor, J.M. The effect of inter-annual Atlantic water inflow variability on the planktonic protist community structure in the West Spitsbergen waters during the summer. J. Plankton Res. 2014, 36, 1190–1203. [Google Scholar] [CrossRef][Green Version]
- Ji, R.; Ashjian, C.J.; Campbell, R.G.; Chen, C.; Gao, G.; Davis, C.S.; Cowles, G.W.; Beardsley, R.C. Life history and biogeography of Calanus copepods in the Arctic Ocean: An individual-based modeling study. Prog. Oceanogr. 2012, 96, 40–56. [Google Scholar] [CrossRef]
- Ackman, R.G. Marine Biogenic Lipids, Fats and Oils; CRC Press: Boca Raton, FL, USA, 1989. [Google Scholar]
- Miller, C.B.; Crain, J.A.; Morgan, C.A. Oil storage variability in Calanus finmarchicus. ICES J. Mar. Sci. 2000, 57, 1786–1799. [Google Scholar] [CrossRef]
- Lee, R.F.; Hagen, W.; Kattner, G. Lipid storage in marine zooplankton. Mar. Ecol. Prog. Ser. 2006, 307, 273–306. [Google Scholar] [CrossRef]
- Mayzaud, P.; Falk-Petersen, S.; Noyon, M.; Wold, A.; Boutoute, M. Lipid composition of the three co-existing Calanus species in the Arctic: Impact of season, location and environment. Polar Biol. 2016, 39, 1819–1839. [Google Scholar] [CrossRef]
- Kwasniewski, S.; Gluchowska, M.; Jakubas, D.; Wojczulanis-Jakubas, K.; Walkusz, W.; Karnovsky, N.; Blachowiak-Samolyk, K.; Cisek, M.; Stempniewicz, L. The impact of different hydrographic conditions and zooplankton communities on provisioning little auks along the west coast of Spitsbergen. Prog. Oceanogr. 2010, 87, 72–82. [Google Scholar] [CrossRef]
- Wassmann, P.; Duarte, C.M.; Agustí, S.; Sejr, M.K. Footprints of climate change in the Arctic marine ecosystem. Glob. Chang. Biol. 2011, 17, 1235–1249. [Google Scholar] [CrossRef]
- Parent, G.J.; Plourde, S.; Turgeon, J. Overlapping size ranges of Calanus spp. off the Canadian Arctic and Atlantic coasts: Impact on species’ abundances. J. Plankton Res. 2011, 33, 1654–1665. [Google Scholar] [CrossRef]
- Gabrielsen, T.M.; Merkel, B.; Søreide, J.E.; Johansson-Karlsson, E.; Bailey, A.; Vogedes, D.; Nygård, H.; Varpe, Ø.; Berge, J. Potential misidentifications of two climate indicator species of the marine arctic ecosystem: Calanus glacialis and C. finmarchicus. Polar Biol. 2012, 35, 1621–1628. [Google Scholar] [CrossRef]
- Nielsen, T.G.; Kjellerup, S.; Smolina, I.; Hoarau, G.; Lindeque, P. Live discrimination of Calanus glacialis and C. finmarchicus females: Can we trust phenological differences? Mar. Biol. 2014, 161, 1299–1306. [Google Scholar] [CrossRef]
- Choquet, M.; Kosobokova, K.; Kwaśniewski, S.; Hatlebakk, M.; Dhanasiri, A.K.S.; Melle, W.; Daase, M.; Svensen, C.; Søreide, J.E.; Hoarau, G. Can morphology reliably distinguish between the copepods Calanus finmarchicus and C. glacialis, or is DNA the only way? Limnol. Oceanogr. Methods 2018, 16, 237–252. [Google Scholar] [CrossRef]
- Renaud, P.E.; Daase, M.; Banas, N.S.; Gabrielsen, T.M.; Søreide, J.E.; Varpe, Ø.; Cottier, F.; Falk-Petersen, S.; Halsband, C.; Vogedes, D.; et al. Pelagic food-webs in a changing Arctic: A trait-based perspective suggests a mode of resilience. ICES J. Mar. Sci. 2018, 75, 1871–1881. [Google Scholar] [CrossRef]
- Campbell, R.; Wagner, M.M.; Teegarden, G.J.; Boudreau, C.A.; Durbin, E.G. Growth and development rates of the copepod Calanus finmarchicus reared in the laboratory. Mar. Ecol. Prog. Ser. 2001, 221, 161–183. [Google Scholar] [CrossRef]
- Wilson, R.J.; Speirs, D.C.; Heath, M.R. On the surprising lack of differences between two congeneric calanoid copepod species, Calanus finmarchicus and C. helgolandicus. Prog. Oceanogr. 2015, 134, 413–431. [Google Scholar] [CrossRef]
- Richardson, A.J.; Schoeman, D.S. Climate impact on plankton ecosystems in the Northeast Atlantic. Science 2004, 305, 1609–1612. [Google Scholar] [CrossRef] [PubMed]
- Lane, P.V.Z.; Llinás, L.; Smith, S.L.; Pilz, D. Zooplankton distribution in the western Arctic during summer 2002: Hydrographic habitats and implications for food chain dynamics. J. Mar. Res. 2008, 70, 97–133. [Google Scholar] [CrossRef]
- Beaugrand, G.; Ibanez, F.; Lindley, J.A.; Reid, P.C. Diversity of calanoid copepods in the North Atlantic and adjacent seas: Species associations and biogeography. Mar. Ecol. Prog. Ser. 2002, 232, 179–195. [Google Scholar] [CrossRef]
- Woodworth-Jefcoats, P.A.; Polovina, J.J.; Drazen, J.C. Climate change is projected to reduce carrying capacity and redistribute species richness in North Pacific pelagic marine ecosystems. Glob. Chang. Biol. 2016, 23, 1000–1008. [Google Scholar] [CrossRef]
- Gluchowska, M.; Dalpadado, P.; Beszczynska-Möller, A.; Olszewska, A.; Ingvaldsen, R.B.; Kwasniewski, S. Interannual zooplankton variability in the main pathways of the Atlantic water flow into the Arctic Ocean (Fram Strait and Barents Sea branches). ICES J. Mar. Sci. 2017, 74, 1921–1936. [Google Scholar] [CrossRef]
- Haug, T.; Bogstad, B.; Chierici, M.; Gjøsæter, H.; Hallfredsson, E.H.; Høines, Å.S.; Hoel, A.H.; Ingvaldsen, R.B.; Jørgensen, L.L.; Knutsen, T.; et al. Future harvest of living resources in the Arctic Ocean north of the Nordic and Barents seas: A review of possibilities and constraints. Fish. Res. 2017, 188, 38–57. [Google Scholar] [CrossRef]
- Hjort, J. Fluctuations in the great fisheries of Northern Europe viewed in the light of biological research. Reun. Cons. Int. Explor. Mer. 1914, 20, 1–228. [Google Scholar]
- Cushing, D.H. Plankton production and year-class strength in fish populations: An update of the match/mismatch hypothesis. Adv. Mar. Biol. 1990, 26, 249–293. [Google Scholar]
- Stenseth, N.C.; Mysterud, A.; Ottersen, G.; Hurrell, J.W.; Chan, K.S.; Lima, M. Ecological effects of climate fluctuations. Science 2002, 297, 1292–1296. [Google Scholar] [CrossRef]
- Both, C.; Bouwhuis, S.; Lessells, C.M.; Visser, M.E. Climate change and population declines in a long-distance migratory bird. Nature 2006, 441, 81–83. [Google Scholar] [CrossRef] [PubMed]
- Hipfner, J.M. Matches and mismatches: Ocean climate, prey phenology and breeding success in a zooplanktivorous seabird. Mar. Ecol. Prog. Ser. 2008, 368, 295–304. [Google Scholar] [CrossRef]
- Nakazawa, T.; Doi, H. A perspective on match/mismatch of phenology in community contexts. Oikos 2012, 121, 489–495. [Google Scholar] [CrossRef]
- Kidawa, D.; Jakubas, D.; Wojczulanis-Jakubas, K.; Stempniewicz, L.; Trudnowska, E.; Boehnke, R.; Keslinka-Nawrot, L.; Błachowiak-Samołyk, K. Parental efforts of an Arctic seabird, the little auk Alle alle, under variable foraging conditions. Mar. Biol. Res. 2015, 4, 349–360. [Google Scholar] [CrossRef]
- Enstipp, R.; Descamps, S.; Fort, J.; Grémillet, D. Almost like a whale-first evidence of suction feeding in a seabird. J. Exp. Biol. 2018, 221, jeb182170. [Google Scholar] [CrossRef] [PubMed]
- Stempniewicz, L.; Darecki, M.; Trudnowska, E.; Błachowiak-Samołyk, K.; Boehnke, R.; Jakubas, D.; Keslinka-Nawrot, L.; Kidawa, D.; Sagan, S.; Wojczulanis-Jakubas, K. Visual prey availability and distribution of foraging little auks (Alle alle) in the shelf waters of West Spitsbergen. Polar Biol. 2013, 36, 949–955. [Google Scholar] [CrossRef]
- Mehlum, F.; Gabrielsen, G.W. The diet of high arctic seabirds in coastal and ice-covered, pelagic areas near the Svalbard archipelago. Polar Res. 1993, 11, 1–20. [Google Scholar] [CrossRef]
- Wojczulanis, K.; Jakubas, D.; Walkusz, W.; Wenneberg, L. Differences in food delivered to chicks by males and females of little auks (Alle alle) on South Spitsbergen. J. Ornithol. 2006, 147, 543–548. [Google Scholar] [CrossRef]
- Jakubas, D.; Wojczulanis-Jakubas, K. Subcolony variation in phenology in breeding parameters in little auk Alle alle. Polar Biol. 2011, 34, 31–39. [Google Scholar] [CrossRef]
- Hovinen, J.E.H.; Wojczulanis-Jakubas, K.; Jakubas, D.; Hop, H.; Berge, J.; Kidawa, D.; Karnovsky, N.J.; Steen, H. Fledging success of little auks in the high Arctic: Do provisioning rates and the quality of foraging grounds matter? Polar Biol. 2014, 37, 665–674. [Google Scholar] [CrossRef]
- Boehnke, R.; Gluchowska, M.; Wojczulanis-Jakubas, K.; Jakubas, D.; Karnovsky, N.J.; Walkusz, W.; Kwasniewski, S.; Błachowiak-Samołyk, K. Supplementary diet components of little auk chicks in two contrasting regions on the west Spitsbergen coast. Polar Biol. 2015, 38, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Boehnke, R.; Balazy, K.; Jakubas, D.; Wojczulanis-Jakubas, K.; Błachowiak-Samołyk, K. Meso-scale variations in little auk chicks diet composition in NW Spitsbergen. Polar Res. 2017, 36, 1409585. [Google Scholar] [CrossRef]
- Møller, E.F.; Johansen, K.L.; Agersted, M.D.; Rigét, F.; Clausen, D.S.; Larsen, J.; Lyngs, P.; Middelbo, A.; Mosbech, A. Zooplankton phenology may explain the North Water polynya’s importance as a breeding area for little auks. Mar. Ecol. Prog. Ser. 2018, 605, 207–223. [Google Scholar] [CrossRef]
- Keslinka, L.K.; Wojczulanis-Jakubas, K.; Jakubas, D.; Neubauer, G. Determinants of the little auk (Alle alle) breeding colony location and size in W and NW coast of Spitsbergen. PLoS ONE 2019, 14, e0212668. [Google Scholar] [CrossRef] [PubMed]
- González-Bergonzoni, I.; Johansen, K.L.; Mosbech, A.; Landkildehus, F.; Jeppesen, E.; Davidson, T.A. Small birds, big effects: The little auk (Alle alle) transforms high Arctic ecosystems. Proc. R. Soc. B Biol. Sci. 2017, 284, 20162572. [Google Scholar] [CrossRef]
- Cottier, F.; Tverberg, V.; Inall, M.; Svendsen, H.; Nilsen, F.; Griffiths, C. Water mass modification in an Arctic fjord through cross-shelf exchange: The seasonal hydrography of Kongsfjorden, Svalbard. J. Geophys. Res. 2005, 110, C12005. [Google Scholar] [CrossRef]
- Prominska, A.; Cisek, M.; Walczowski, W. Kongsfjorden and Hornsund hydrography-comparative study based on a multiyear survey in fjords of west Spitsbergen. Oceanologia 2017, 59, 397–412. [Google Scholar] [CrossRef]
- Saloranta, E.; Svendsen, H. Across the Arctic Front west of Spitsbergen: High-resolution CTD sections from 1998–2000. Polar Res. 2001, 20, 174–184. [Google Scholar] [CrossRef]
- Nilsen, F.; Cottier, F.; Skogseth, R.; Mattson, S. Fjord-shelf exchanges controlled by ice and brine production: The interannual variation of Atlantic Water in Isfjorden, Svalbard. Cont. Shelf Res. 2008, 28, 1838–1853. [Google Scholar] [CrossRef]
- Sakshaug, E.; Johnsen, G.; Kovacs, K. Ecosystem Barents Sea; Tapir Academic Press: Trondheim, Norway, 2009. [Google Scholar]
- Pavlov, A.K.; Tverberg, V.; Ivanov, B.V.; Nilsen, F.; Falk-Petersen, S.; Granskog, M.A. Warming of Atlantic Water in two Spitsbergen fjords over the last century (1912–2009). Polar Res. 2013, 32, 11206. [Google Scholar] [CrossRef]
- Amélineau, F.; Bonnet, D.; Heitz, O.; Mortreux, V.; Harding, A.M.A.; Karnovsky, N.; Walkusz, W.; Fort, J.; Grémillet, D. Microplastic pollution in the Greenland Sea: Background levels and selective contamination of planktivorous diving seabirds. Environ. Pollut. 2016, 219, 1131–1139. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.; Wiebe, L.; Lenz, J.; Skjoldal, H.R.; Huntley, M. ICES Zooplankton Methodology Manual; Academic Press: London, UK, 2000. [Google Scholar]
- Kwasniewski, S.; Hop, H.; Falk-Petersen, S.; Pedersen, G. Distribution of Calanus species in Kongsfjorden, a glacial fjord in Svalbard. J. Plankton Res. 2003, 25, 1–20. [Google Scholar] [CrossRef]
- Herman, A.W. Design and calibration of a new optical plankton counter capable of sizing small zooplankton. Deep-Sea Res. A 1992, 39, 395–415. [Google Scholar] [CrossRef]
- Herman, A.W.; Beanlands, B.; Phillips, E.F. The next generation of Optical Plankton Counter: The Laser-OPC. J. Plankton Res. 2004, 26, 1135–1145. [Google Scholar] [CrossRef]
- Herman, A.W.; Harvey, M. Application of normalized biomass size spectra to laser optical plankton counter net intercomparisons of zooplankton distributions. J. Geophys. Res. 2006, 111, C5. [Google Scholar] [CrossRef]
- Basedow, S.L.; Tande, K.S.; Norrbin, M.F.; Kristiansen, S.A. Capturing quantitative zooplankton information in the sea: Performance test of laser optical plankton counter and video plankton recorder in a Calanus finmarchicus dominated summer situation. Prog. Oceanogr. 2013, 108, 72–80. [Google Scholar] [CrossRef]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; Primer-E Ltd.: Plymouth, UK, 2008. [Google Scholar]
- Clarke, K.R.; Gorley, R.N. Primer; Primer-E Ltd.: Plymouth, UK, 2001. [Google Scholar]
- Anderson, M.J.; Braak, C.J.F. Permutation tests for multi-factorial analysis of variance. J. Stat. Comput. Simul. 2003, 73, 85–113. [Google Scholar] [CrossRef]
- Kwasniewski, S.; Gluchowska, M.; Walkusz, W.; Karnovsky, N.J.; Jakubas, D.; Wojczulanis-Jakubas, K.; Harding, A.M.A.; Goszczko, I.; Cisek, M.; Beszczynska-Möller, A.; et al. Interannual changes in zooplankton on the West Spitsbergen Shelf in relation to hydrography and their consequences for the diet of planktivorous seabirds. J. Mar. Sci. 2012, 69, 890–901. [Google Scholar] [CrossRef]
- Jakubas, D.; Wojczulanis-Jakubas, K.; Boehnke, R.; Kidawa, D.; Błachowiak-Samołyk, K.; Stempniewicz, L. Intra-seasonal variation in zooplankton avialability, chick diet and breeding performance of a high Arctic planktivorous seaird. Polar Biol. 2016, 39, 1547–1561. [Google Scholar] [CrossRef]
- Weydmann, A.; Walczowski, W.; Carstensen, J.; Kwaśniewski, S. Warming of subarctic waters accelerates development of a key marine zooplankton Calanus finmarchicus. Glob. Chang. Biol. 2018, 24, 172–183. [Google Scholar] [CrossRef]
- Jakubas, D.; Wojczulanis-Jakubas, K.; Iliszko, L.M.; Strøm, H.; Stempniewicz, L. Habitat foraging niche of a High Arctic zooplanktivorous seabird in a changing environment. Sci. Rep. 2017, 7, 16203. [Google Scholar] [CrossRef]
- Ottersen, G.; Planque, B.; Belgrano, A.; Post, E.; Reid, P.C.; Stenseth, N.C.H. Ecological effects of the North Atlantic Oscillation. Oecologia 2001, 128, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Beaugrand, G.; Brander, K.M.; Lindley, J.A.; Souissi, S.; Reid, P.C. Plankton effect of cod recruitment in the North Sea. Nature 2003, 426, 661–664. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.; Holleman, L. Warmer springs disrupt the synchrony of oak and winter moth phenology. Proc. R. Soc. B Biol. Sci. 2001, 268, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.J.; Potti, J.; Moreno, J.; Merino, S.; Frias, O. Climate change and fitness components of a migratory bird breeding in the Mediterranean region. Glob. Chang. Biol. 2003, 9, 112. [Google Scholar] [CrossRef]
- Thomas, D.W.; Blondel, J.; Perret, P.; Lambrechts, M.M.; Speakman, J.R. Energetic and fitness costs of mismatching resource supply and demand in seasonally breeding birds. Science 2001, 291, 2598–2600. [Google Scholar] [CrossRef]
- Visser, E.M.; Adriaensen, F.; Van Balen, J.H.; Blondel, J.; Dhondt, A.A.; Van Dongen, S.; Du Feu, C.; Ivankina, E.V.; Kerimov, A.B.; De Laet, J.; et al. Variable responses to large-scale climate change in European Parus populations. Proc. R. Soc. B Boil. Sci. 2003, 270, 367–372. [Google Scholar]
- McKinnon, L.; Picotin, M.; Bolduc, E.; Juillet, C.; Bêty, J. Timing of breeding, peak food availability, and effects of mismatch on chick growth in birds nesting in the High Arctic. Can. J. Zool. 2012, 90, 961–971. [Google Scholar] [CrossRef]
- Mackas, D.L.; Batten, S.; Trudel, M. Effects on zooplankton of a warmer ocean: Recent evidence from the Northeast Pacific. Prog. Oceanogr. 2007, 75, 223–252. [Google Scholar] [CrossRef]
- Bertram, D.F.; Mackas, D.L.; Welch, D.W.; Boyd, W.S.; Ryder, J.L.; Galbraith, M.; Hedd, A.; Morgan, K.; O’Hara, P.D. Variation in zooplankton prey distribution determines marine foraging distributions of breeding Cassin’s Auklet. Deep-Sea Res. Pap. 2017, 129, 32–40. [Google Scholar] [CrossRef]
- Konarzewski, M.; Taylor, J.R.E.; Gabrielsen, G.W. Chick energy requirements and adult energy expenditures of Dovekies (Alle alle). Auk 1993, 110, 603–609. [Google Scholar] [CrossRef]
- Karnovsky, N.J.; Kwasniewski, S.; Weslawski, J.M.; Walkusz, W.; Beszczynska-Møller, A. The foraging behavior of little auks in a heterogeneous environment. Mar. Ecol. Prog. Ser. 2003, 253, 289–303. [Google Scholar] [CrossRef]
- Jakubas, D.; Wojczulanis, K.; Walkusz, W. Response of dovekie to changes in food availability. Waterbirds 2007, 30, 421–428. [Google Scholar] [CrossRef]
- Ragonese, S.; Bianchini, M.L. Growth, mortality and yield-per-recruit of the deep-water shrimp Aristeus antennatus (Crustacea-Aristeidae) of the Strait of Sicily (Mediterranean Sea). Fish. Res. 1996, 26, 125–137. [Google Scholar] [CrossRef]
- Huntley, M.E.; Lopez, M.D.G. Temperature-dependent production of marine copepods: A global synthesis. Am. Nat. 1992, 140, 201–242. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, D. Temperature and organism size—A biological law for ectotherms? Adv. Ecol. Res. 1994, 25, 1–58. [Google Scholar] [CrossRef]
- Angilletta, M.J.; Steury, T.D.; Sears, M.W. Temperature, growth rate, and body size in ectotherms: Fitting pieces of a life-history puzzle. Integr. Comp. Biol. 2004, 44, 498–509. [Google Scholar] [CrossRef]
- Gillooly, J.F.; Brown, J.H.; West, G.B.; Savage, V.M.; Charnov, E.L. Effects of size and temperature on metabolic rate. Science 2001, 293, 2248–2251. [Google Scholar] [CrossRef]
- Daufresne, M.; Lengfellner, K.; Sommer, U. Global warming benefits the small in aquatic ecosystems. Proc. Natl. Acad. Sci. USA 2009, 106, 12788–12793. [Google Scholar] [CrossRef]
- Gardner, J.L.; Peters, A.; Kearney, M.R.; Joseph, L.; Heinsohn, R. Declining body size: A third universal response to warming? Trends Ecol. Evol. 2011, 26, 285–291. [Google Scholar] [CrossRef]
- Banas, N.S.; Møller, E.F.; Nielsen, T.G.; Lisa, B.E. Copepod life strategy and population viability in response to prey timing and temperature: Testing a new model across latitude, time, and the size spectrum. Front. Mar. Sci. 2016, 3, 255. [Google Scholar] [CrossRef]
- Leinaas, H.P.; Jalal, M.; Gabrielsen, T.M.; Hessen, D.O. Inter- and intraspecific variation in body- and genome size in calanoid copepods from temperate and arctic waters. Ecol. Evol. 2016, 6, 5585–5595. [Google Scholar] [CrossRef] [PubMed]
- Daase, M.; Eiane, K. Mesozooplankton distribution in northern Svalbard waters in relation to hydrography. Polar Biol. 2007, 30, 969–981. [Google Scholar] [CrossRef]
- Weydmann, A.; Carstensen, J.; Goszczko, I.; Dmoch, K.; Olszewska, A.; Kwasniewski, S. Shift towards the dominance of boreal species in the Arctic: Inter-annual and spatial zooplankton variability in the West Spitsbergen Current. Mar. Ecol. Prog. Ser. 2014, 501, 41–52. [Google Scholar] [CrossRef]
- Brown, Z.W.; Welcker, J.; Harding, A.M.A.; Walkusz, W.; Karnovsky, N.J. Divergent diving behaviour during short and long trips of a bimodal forager, the little auk Alle alle. J. Avian Biol. 2012, 43, 215–226. [Google Scholar] [CrossRef]
- Gluchowska, M.; Kwasniewski, S.; Prominska, A.; Olszewska, A.; Goszczko, I.; Falk-Petersen, S.; Hop, H.; Weslawski, J.M. Zooplankton in Svalbard fjords on the Atlantic-Arctic boundary. Polar Biol. 2016, 39, 1785–1802. [Google Scholar] [CrossRef]
- Trudnowska, E.; Sagan, S.; Kwasniewski, S.; Darecki, M.; Blachowiak-Samaolyk, K. Fine-scale zooplankton vertical distribution in relation to hydrographic and optical characteristics of the surface waters on the Arctic shelf. J. Plankton Res. 2015, 37, 120–133. [Google Scholar] [CrossRef]
- Willis, K.; Cottier, F.; Kwasniewski, S.; Wold, A.; Falk-Petersen, S. The influence of advection on zooplankton community composition in an Arctic fjord (Kongsfjorden, Svalbard). J. Mar. Syst. 2006, 61, 39–54. [Google Scholar] [CrossRef]
- Lischka, S.; Hagen, W. Seasonal dynamics of mesozooplankton in the Arctic Kongsfjord (Svalbard) during year-round observations from August 1998 to July 1999. Polar Biol. 2016, 39, 1859–1878. [Google Scholar] [CrossRef]
- Espinasse, M.; Halsband, C.; Varpe, Ø.; Gislason, A.; Gudmundsson, K.; Falk-Petersen, S.; Eiane, K. Interannual phenological variability in two North-East Atlantic populations of Calanus finmarchicus. Mar. Biol. Res. 2018, 14, 752–767. [Google Scholar] [CrossRef]
- Søreide, J.E.; Leu, E.; Berge, J.; Graeve, M.; Falk-Petersen, S. Timing of blooms, algal food quality and Calanus glacialis reproduction and growth in a changing Arctic. Glob. Chang. Biol. 2010, 16, 3154–3163. [Google Scholar] [CrossRef]
- Gluchowska, M.; Trudnowska, E.; Goszczko, I.; Kubiszyn, A.M.; Blachowiak-Samolyk, K.; Walczowski, W.; Kwasniewski, S. Variations in the structural and functional diversity of zooplankton over vertical and horizontal environmental gradients en route to the Arctic Ocean through the Fram Strait. PLoS ONE 2017, 12, e0171715. [Google Scholar] [CrossRef] [PubMed]

| Copepodite Stage | Factor | df | MS | Pseudo-F | p | |
|---|---|---|---|---|---|---|
| CI | Fjord | 1 | 1274.8 | 212.98 | 0.001 | 1.40 |
| CII | Fjord | 1 | 5180.9 | 560.51 | 0.001 | 2.43 |
| CIII | Fjord | 1 | 8748.4 | 1086.40 | 0.001 | 2.74 |
| CIV | Fjord | 1 | 5728.8 | 720.28 | 0.001 | 1.98 |
| CV | Fjord | 1 | 2277.5 | 237.43 | 0.001 | 1.43 |
| AF | Fjord | 1 | 1.2 | 0.22 | 0.630 | −0.18 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balazy, K.; Trudnowska, E.; Błachowiak-Samołyk, K. Dynamics of Calanus Copepodite Structure during Little Auks’ Breeding Seasons in Two Different Svalbard Locations. Water 2019, 11, 1405. https://doi.org/10.3390/w11071405
Balazy K, Trudnowska E, Błachowiak-Samołyk K. Dynamics of Calanus Copepodite Structure during Little Auks’ Breeding Seasons in Two Different Svalbard Locations. Water. 2019; 11(7):1405. https://doi.org/10.3390/w11071405
Chicago/Turabian StyleBalazy, Kaja, Emilia Trudnowska, and Katarzyna Błachowiak-Samołyk. 2019. "Dynamics of Calanus Copepodite Structure during Little Auks’ Breeding Seasons in Two Different Svalbard Locations" Water 11, no. 7: 1405. https://doi.org/10.3390/w11071405
APA StyleBalazy, K., Trudnowska, E., & Błachowiak-Samołyk, K. (2019). Dynamics of Calanus Copepodite Structure during Little Auks’ Breeding Seasons in Two Different Svalbard Locations. Water, 11(7), 1405. https://doi.org/10.3390/w11071405






