3.1. Coagulation/Sedimentation Experiments
Landfill leachate has a COD higher than 5000 mg L
−1 and a BOD
5/COD ratio less than 0.10 (
Table 1). This indicates the presence of persistent organic compounds and non-biodegradable substances. Therefore, effective treatment is required, such as combined physicochemical treatments, excluding biological [
26]. A coagulation/sedimentation process as a first step in the leachate treatment was applied. The effect of some factors such as coagulant dose, pH and slow mixing time on the efficiency of the removal of organic matter for coagulation/sedimentation process were evaluated.
Table 2 presents complete experimental design of coagulation/sedimentation experiments using PAC and FeCl
3·6H
2O coagulant and COD reduction results obtained in this study.
The significant main and interaction effects of factors that influences the COD reduction was determined with ANOVA.
Table 3 presents a summary of ANOVA results for coagulation/sedimentation experiments with PAC coagulant. The most significant factors for COD reduction in order of importance were slow mixing time (C) and coagulant dose (A), according to the high calculated values of F and low values-
p (<0.05). The pH resulted to be non-significant in evaluated experimental range. The self-double-effect of each factor (A
2, B
2 and C
2 terms) were not taken into account because they were non-significant.
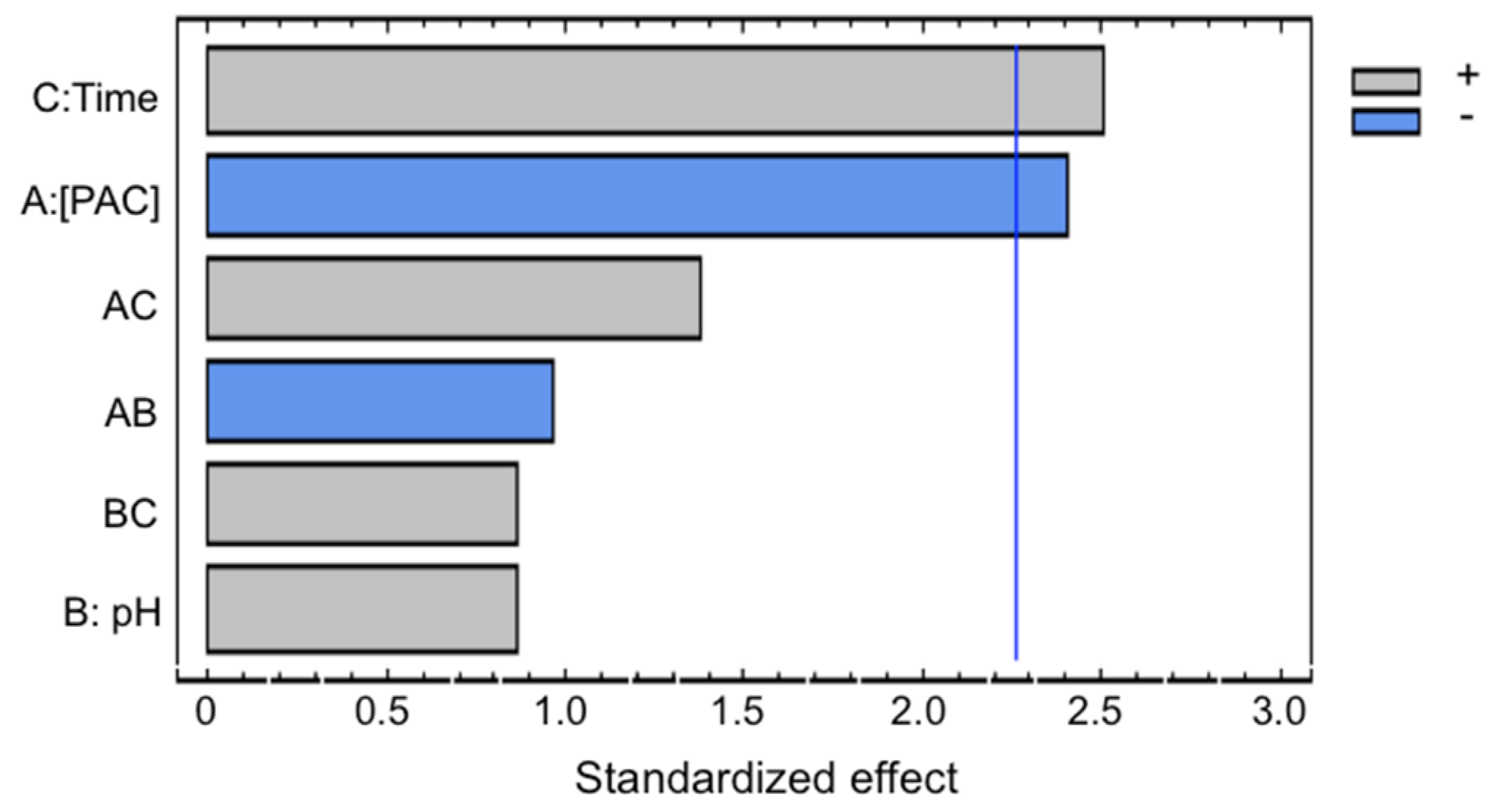
The Pareto diagram represents graphically the standardized effects of each factor and its interactions. The positive (+) or negative (−) effect on the response factor is given by the increase in the level of factor [
27].
Figure 2 presents a Pareto diagram for coagulation/sedimentation experiments with PAC coagulant. The coagulant dose and slow mixing time were the factors with the highest effect over the COD reduction. The coagulant dose presented negative sign, it indicates that the highest COD reduction was obtained at the lowest levels of this factor. The coagulant PAC at low doses can neutralize the surface charge of the particles and allow aggregation but at high doses the effectiveness of the process is reduced [
12]. The increase of coagulant beyond a certain concentration could re-stabilize colloidal particles, forming smaller and weaker floccules through adsorption and bridging due to additional PAC [
14]. The slow mixing time presented positive sign, it indicates that the highest COD reduction was obtained at the highest levels of this factor, this depends on the characteristics of the leachate, being this complex in its components [
28]. The best conditions for COD reduction were obtained. COD reduction of 36.1% were obtained with pH 8, 0.615 g L
−1 of PAC and 20 min of slow mixing, similar to the reported results reported with removal rates of between 25% and 38% [
28].
Table 4 presents a summary of ANOVA results for coagulation/sedimentation experiments with FeCl
3·6H
2O coagulant. The most significant factors for COD reduction in order of importance were pH (A) and the interaction pH-FeCl
3 dose (AB), according to the high calculated values of F and low values-
p (<0.05). The slow mixing time resulted to be non-significant in evaluated experimental range. The self-double-effect of each factor (A
2, B
2 and C
2 terms) were not taken into account because they were non-significant.
Figure 3 presents a Pareto diagram for coagulation/sedimentation experiments with FeCl
3·6H
2O coagulant. The pH and the interaction pH-FeCl
3 dose were the factors with the highest effect over the COD reduction. Both factors presented negative sign, it indicates that the highest COD reduction was obtained at the lowest levels of these factors. The negative effect of pH influenced COD reduction, due to the nature of the leachate, which does not allow for the solubility of iron ions. At pH value > 8 the species Fe(OH)
4− appear, the efficiency to precipitate of which is null according to the coagulation diagrams for FeCl
3, however at pH < 6, appear species such as Fe
3+, Fe
2(OH)
2+, FeOH
2+, which promote the restabilization of colloidal particles [
6].
The conditions of greater COD reduction using FeCl
3·6H
2O coagulant were pH 6, 2.5 g L
−1 of FeCl
3·6H
2O and 30 min of slow mixing, COD reduction of 65.0 % were obtained. These results were consistent with other studies in which FeCl
3·6H
2O showed a higher removal efficiency of organic matter compared with the PAC [
2]. Moradi and Ghanbari [
5] reported similar data for the treatment of leachate with FeCl
3·6H
2O, finding COD removal of 65.0 %. Liu and co-workers [
13] reported optimal results at pH close to 6 and dose of 10 g L
−1 of FeCl
3·6H
2O with COD reduction of 68.7 %, finding that the initial pH and the coagulant dose play an important role. They also point out that it is viable to use coagulation processes for the pre-treatment of leachates. The predominant coagulation mechanisms are charge neutralization, in which the charged hydrolysis species of coagulant can adsorb into the surface of the colloidal particle and destabilize it; and sweep-floc coagulation, the presence of precipitate of ferric hydroxide can physically sweep the colloidal particles from the suspension [
29]. When increasing the dose of FeCl
3·6H
2O, the elimination of COD increases maintaining the pH in 6, as reported by Long and co-workers [
30]. This indicates that FeCl
3·6H
2O presents greater COD removal with pH values close to neutrality. On the contrary, with pH values that oscillate between 8 or more, the COD removal is less, because the Fe
3+ cations allow the colloidal particles to be positively charged, stabilizing the colloids [
31].
With the optimal conditions determined in this study, 36.1 % and 65.0 % COD reduction were obtained using PAC and FeCl3·6H2O coagulants, respectively. Comparing these results, the FeCl3·6H2O presents higher COD reduction with respect to the PAC, this is a great advantage to use FeCl3·6H2O, however it is extremely corrosive and presents higher costs. On the other hand, comparing the results with the conditions obtained with PAC coagulant in this study (pH 8, 0.615 g L−1 of PAC and 20 min of slow mixing, final COD of 3968 mg O2 L−1) and the conditions used in the sanitary landfill (pH 8.0, 1.5 g L−1 of PAC and 30 min of slow mixing, final COD of 4960 mg O2 L−1), an increase of 80% was obtained in the removal of organic matter and the consumption of coagulant was reduced to 59%, maintaining the initial pH of the leachate, therefore, operating costs decrease in this process.
3.2. Solar Photo-Fenton Experiments with PAC Coagulant Pre-Treated
The leachate treated with PAC coagulant increased the removal of organic matter (final COD of 3968 mg O
2 L
−1) compared to the process used in the landfill sanitary (final COD of 4960 mg O
2 L
−1). However, the pre-treated leachate exceeds the legal limit of discharge into natural water streams, according to Colombian legislation the concentration of COD for the final leachate effluent is set at < 180 mg O
2 L
−1 [
32]. Therefore, the combination with photo-oxidation processes is required. The solar photo Fenton process is considered a suitable way for the treatment of leachate. The most important variables related to the rate of removal of COD are the intensity of irradiation, concentration of H
2O
2 and Fe
2+ in the system, pH values and the initial COD, therefore the first three variables were evaluated in this study with the aim to determine the optimal conditions and its effects on COD removal in the pre-treated leachate [
23]. Central composite design (CCD) was used to optimize the three main factors in solar photo Fenton: H
2O
2 dose, Fe
2+ dose and accumulate UV energy. According to conditions reported literature a [H
2O
2]/[Fe
2+] ratio between 5 and 20 was used, considering that the excess of hydrogen peroxide with respect to the amount of iron added is essential to maintain the catalytic character in the chemical reaction [
33]. The pH after the coagulation/sedimentation process was adjusted to a value 3 with addition of H
2 SO
4 to avoid iron precipitation in the form of hydroxide, Fe(OH)
3 [
14].
Table 5 shows the results of complete experimental design and the COD reduction of solar photo-Fenton experiments in pre-treated leachate with PAC coagulant. The highest COD reduction (56.4%) was achieved for the experiment 12, with 13.3 g L
−1 of H
2O
2, 2.56 g L
−1 of Fe
2+ and 122 kJ L
−1 of accumulated radiation. These results are very similar to characterization and detoxification of a mature landfill leachate using combined treatments of coagulation/flocculation and photo-Fenton with Fe
2+ dose of 5.5 mg L
−1 and H
2O
2 dose of 630 mg L
−1, which achieved COD reduction of 56% [
14]. Similarly, combinations of treatments have been tested at landfills, obtaining in the coagulation process a COD reduction of 26% and when combined with the solar photo-Fenton process, a COD reduction of 60% was achieved with accumulated radiation of 165 kJ L
−1 over five days [
7].
The results of the ANOVA are presented in the
Table 6. The factors in order of significance and contribution were: quadratic B, concentration of Fe
2+ (B), quadratic C, accumulated UV energy (C) and quadratic A. All the terms have a p value lower than 0.05 and a high F value.
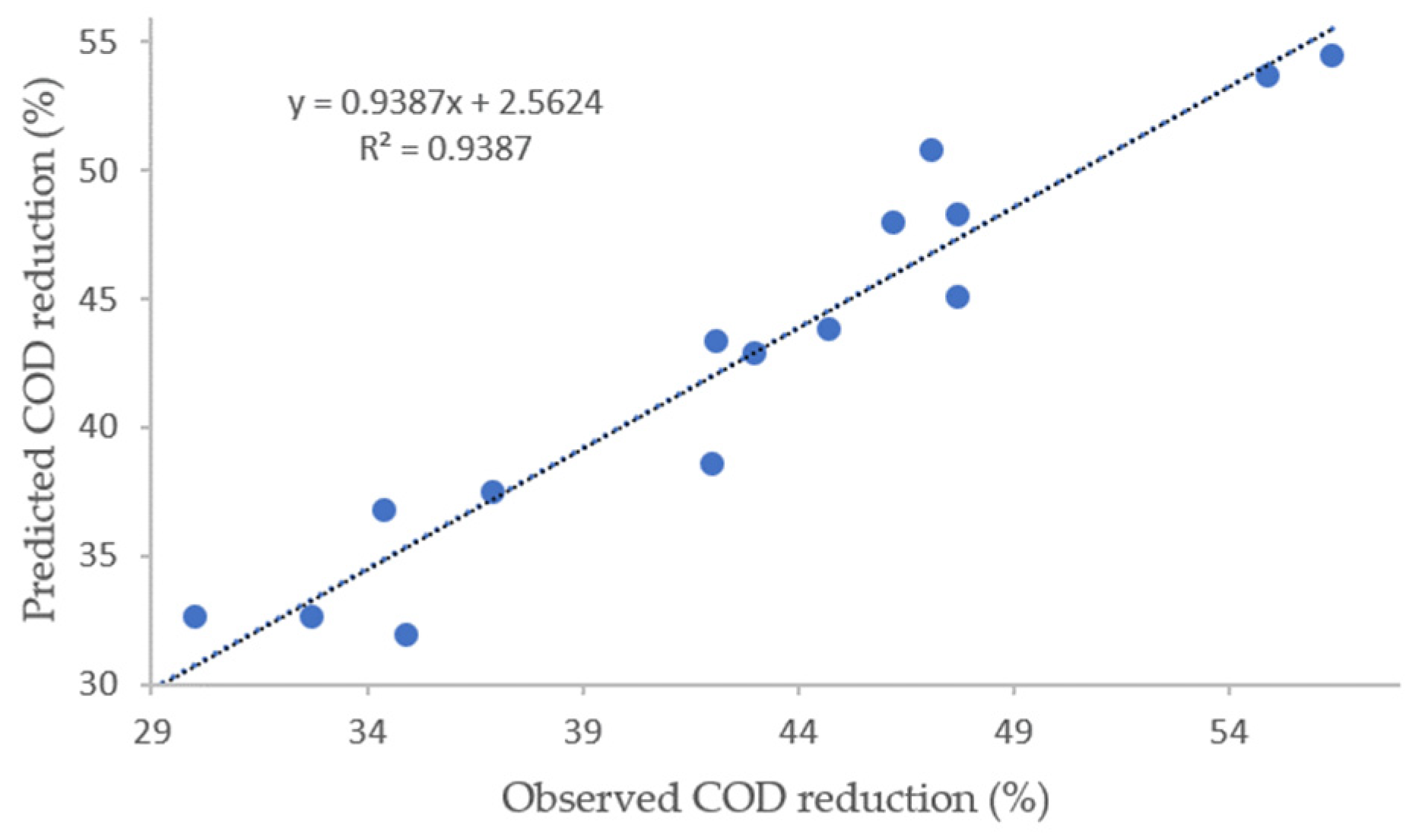
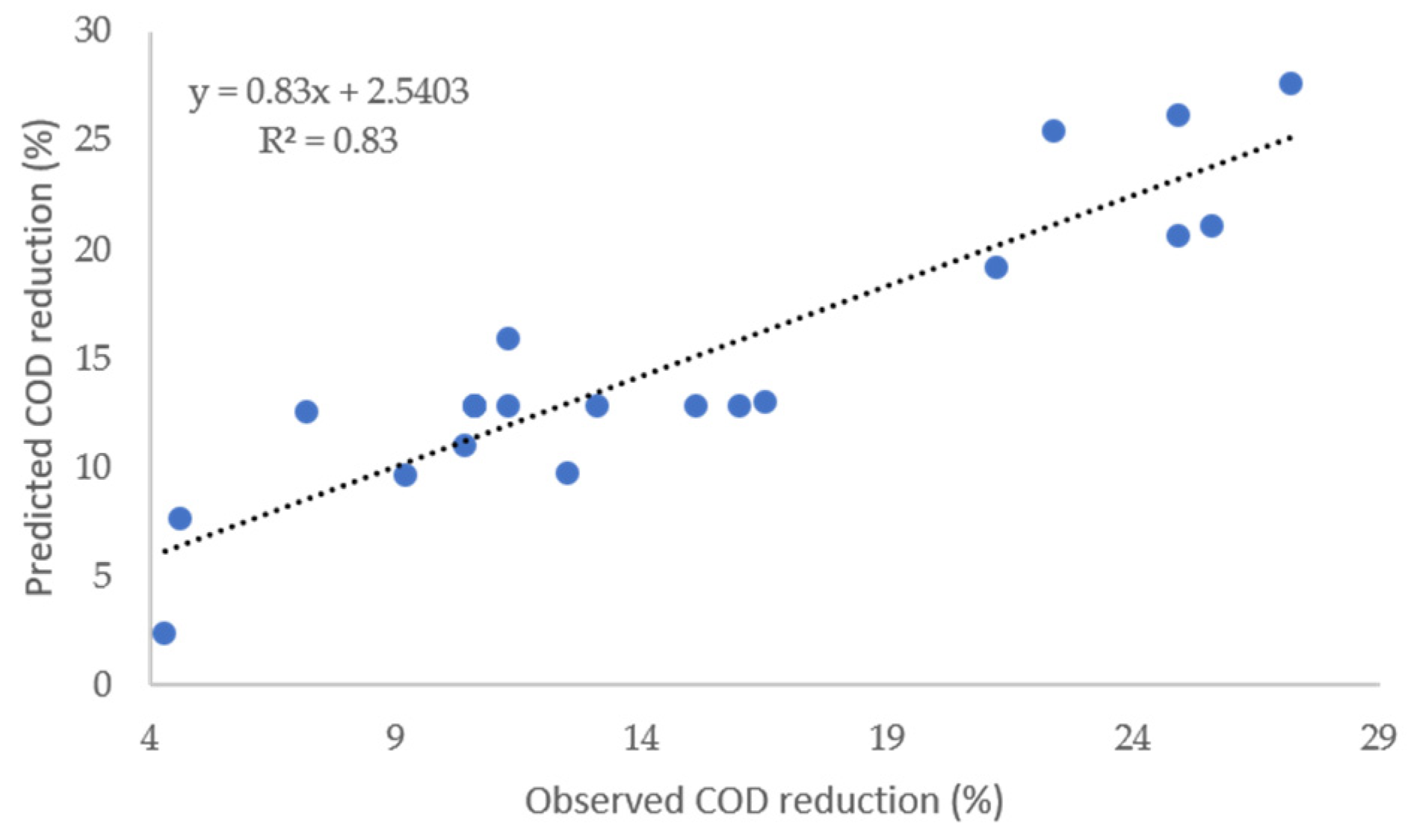
The quality of the fitted model was evaluated based on the determination coefficients, R
2 and R
2adj. The high values of R
2 (93.87%) and R
2adj (84.67%) indicate that Equation (3) present a satisfactory correlation between the model and observed results. Similarity the
Figure 4 demonstrated the concordance between the observed values and the values obtained with the adjusted model.
where, Y is COD reduction, X
1 is H
2O
2 dose, X
2 is Fe
2+ dose and X
3 is accumulate UV energy.
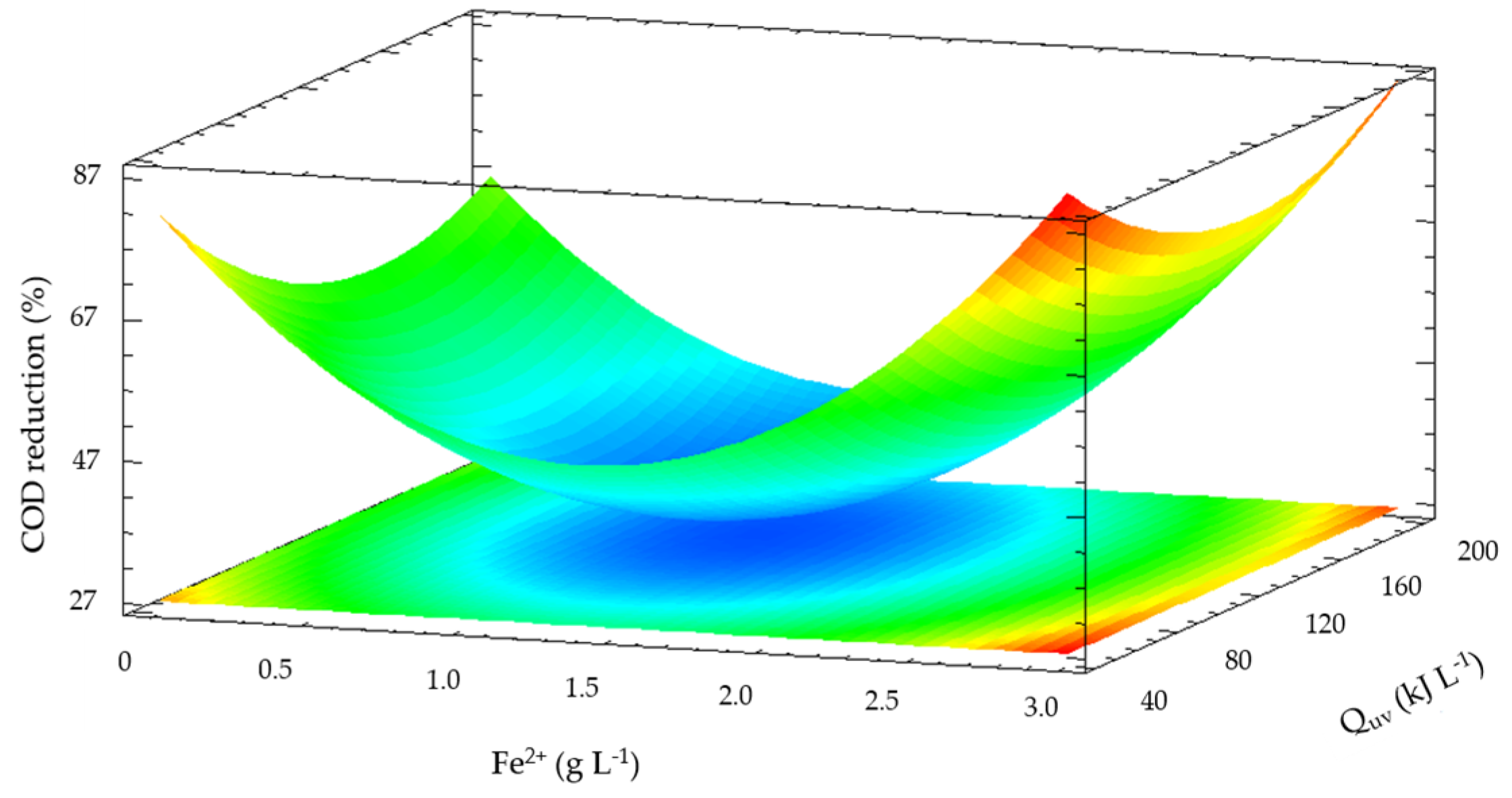
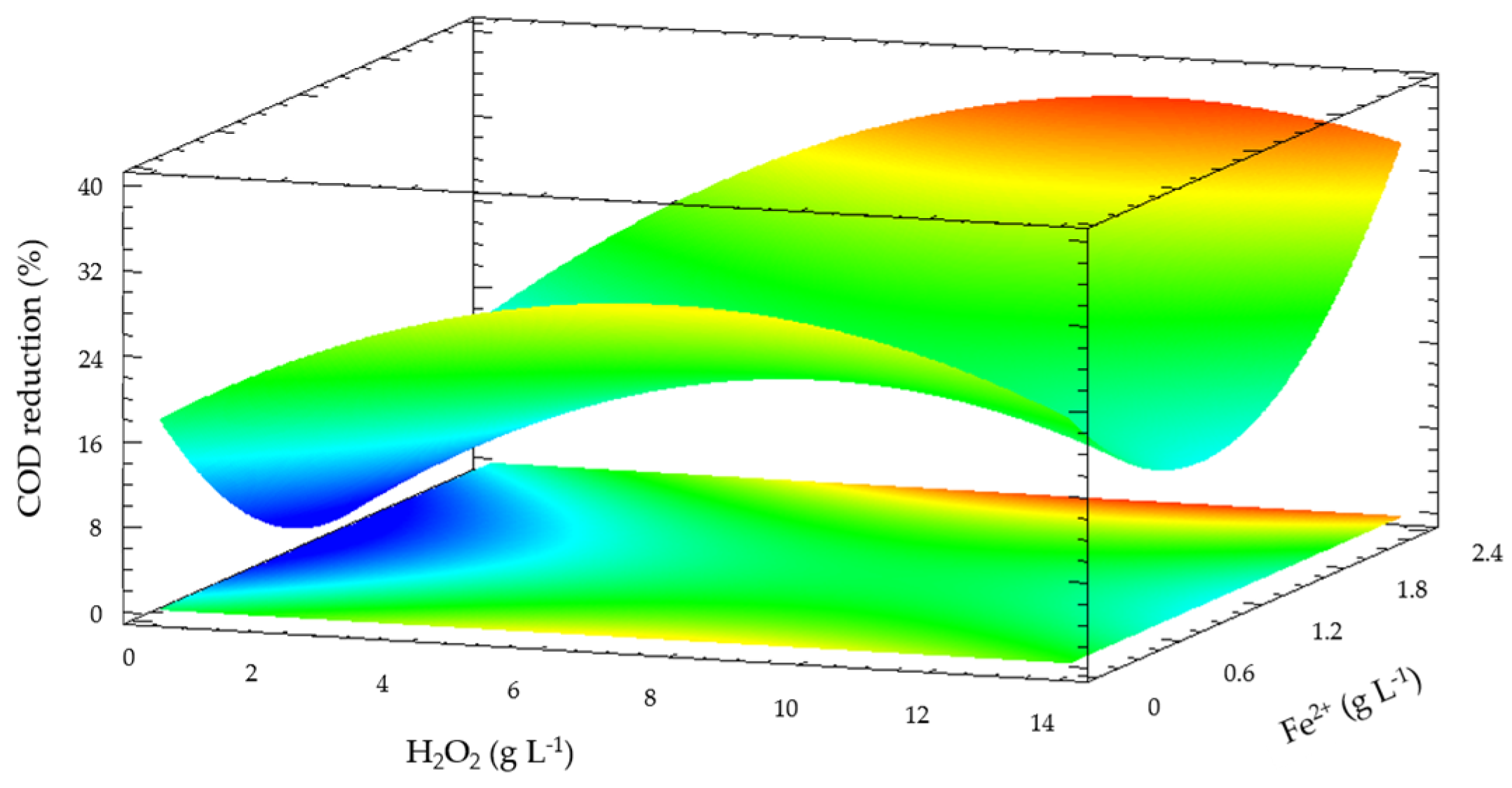
Figure 5 shows the optimization of the operation conditions of solar photo-Fenton experiments in pre-treated leachate with PAC coagulant. The COD reduction predicted obtained in the optimization was 65% with 14.68 g L
−1 of H
2O
2, 2.56 g L
−1 of Fe
2+ and 48 kJ L
−1 of accumulated UV energy. The results show that a higher addition of iron doses and accumulated solar radiation promotes the formation of oxidant species to reduce COD [
34,
35,
36].
3.3. Ferrioxalate-Induced Solar Photo-Fenton Process with FeCl3 Coagulant Pre-Treated
In this case, the efficiency of COD reduction of ferrioxalate-induced solar photo-Fenton process in pre-treated leachate with FeCl
3 coagulant was evaluated. The H
2O
2 dose is determined from the COD of the pre-treated leachate, which was calculated based on the stoichiometric ratio COD:H
2O
2 proposed by Kim and co-workers [
37], where 1 g of COD = 2.125 g of H
2O
2. The dose of Fe
2+ supplied from ferrous sulfate heptahydrate granular (FeSO
4·7H
2O), was obtained from the ratio H
2O
2: Fe
2+, for each 5 a 10 g of H
2O
2 is added 1 g of Fe
2+ and the doses of oxalic acid resulted from the ratio 1 g of Fe
2+ per 3 g of H
2C
2O
4 [
37]. The pH was adjusted with sulfuric acid to 97 % purity according to the predetermined. The experiments were carried out at 167 kJ L
−1 of accumulated UV energy.
The results of COD reduction and complete experimental design of ferrioxalate-induced solar photo-Fenton experiments with a FeCl
3 coagulant pre-treatment are shown in
Table 7. The highest reduction of COD (27.2%) was achieved for Exp. 4, with 10.8 g L
−1 of H
2O
2, 1.97 g L
−1 of Fe
2+, 5.91 g L
−1 of H
2C
2O
4 and pH 4.0.
The ANOVA results show that some factors, except the interactions AC ([H
2O
2]-pH) and BC ([Fe
2+]/[C
2H
2O
4]-pH), are statistically significant (
Table 8). The factors in order of importance and contribution were: pH (C), quadratic B, [Fe
2+]/[C
2H
2O
4] (B), quadratic C, quadratic A, concentration of H
2O
2 (A) and interaction AB ([H
2O
2]−[Fe
2+]/[C
2H
2O
4]). All them terms have a
p value lower than 0.05 and high F value.
The quality of the fitted model was evaluated based on the determination coefficients, R
2 and R
2adj. The high values of R
2 (83.0%) and R
2 adj (77.6%) indicate that Equation (4) present a correlation between the model and observed results. Similarity the
Figure 6 demonstrated the concordance between the observed values and the values obtained with the adjusted model.
where, Y is COD reduction, X
1 is pH, X
2 is H
2O
2 dose and X
3 is Fe
2+ dose.
Figure 7 shows the optimization of the operation conditions of ferrioxalate-induced solar photo-Fenton experiments in pre-treated leachate with FeCl
3·6H
2O coagulant. The COD reduction predicted obtained in the optimization was 37% with 8.95 g L
−1 of H
2O
2, 1.60 g L
−1 of Fe
2+, 4.8 g L
−1 of C
2H
2O
4 and pH 4.0. The results show that a higher addition of iron doses and H
2O
2 doses promotes the formation of oxidant species to reduce COD. This indicates that with sufficient doses of hydrogen peroxide and Fe
2+/C
2H
2O
4 to photocatalytic system, the mineralization of pollutants increases, due to the continuous regeneration of Fe (II) from the photo-reduction of Fe (III) by solar light and the generation of additional free radicals (mainly OH•) due to ferrioxalate photochemistry [
38,
39,
40]. Reducing the pH and increasing the amount of iron in the leachate increases the efficiency of the process to reduce COD, due to the generation of Fe[(C
2O
4)
3]
3−, similar results were reported by Estrada-Arriaga and co-workers [
25]. Low concentrations of Fe
2+ and H
2O
2 generate a decrease in the removal of organic matter, because it decreases the generation of HO• radicals according to Equations (5) and (6) [
41].
3.4. Treatment of Leachate under Optimized Conditions
A preliminary study of the costs per cubic meter of leachate treatment with coagulation followed by solar photo-Fenton process was carried out, in order to compare the costs of the combined treatment and the treatment used in the sanitary landfill located in the Atlantico department, Colombia. The coagulation process is the only treatment used in the treatment of leachates from this sanitary landfill with a removal of organic matter not greater than 20%. Currently, the operational conditions used in the sanitary landfill are 1.5 g L
−1 of PAC, pH 8.0, fast mixing of 300 rpm for 2 min and slow mixing of 40 rpm for 30 min, for a total unitary cost of 2.60 €/m
3 [
42].
(i) Treatment 1: coagulation/sedimentation using PAC followed by solar photo-Fenton process. The treated leachate with the optimal operational conditions of each of the processes (
Section 3.1 and
Section 3.2) obtained a 73% reduction of COD (COD final 1674 mg L
−1, 0.615 g L
−1 of PAC) with a total unitary cost of 3.00 €/m
3. By comparing these results, optimized treatment 1 allowed to substantially improve the reduction of COD in the leachate landfill, with only an increase of 13.3% in the total unitary cost. In addition, the use of the PAC coagulant was reduced by 59%.
(ii) Treatment 2: coagulation/sedimentation using FeCl
3·6H
2O followed by ferrioxalate-induced solar photo-Fenton process. The treated leachate with the optimal operational conditions of each of the processes (
Section 3.1 and
Section 3.3) obtained a 80% reduction of COD (COD final 1240 mg L
−1, 2.5 g L
−1 of FeCl
3·6H
2O) with a total unitary cost of 4.30 €/m
3. By comparing these results, optimized treatment 2 allowed to substantially improve the reduction of COD in the leachate landfill, with only an increase of 39.5% in the total unitary cost.
Treatments 1 and 2 increased the removal of organic matter (final COD of 1674 mg O
2 L
−1 and 1240 mg O
2 L
−1, respectively) compared to the process used in the landfill sanitary (final COD of 4960 mg O
2 L
−1). Under these conditions, the effluent obtained with the alternative treatments presents high values of COD, therefore it cannot be discharged into natural water streams, to Colombian legislation the concentration of COD for the final leachate effluent is set at < 180 mg O
2 L
−1 [
32]. However, the effluent improves enough to be released into public wastewater where the COD legal limit is 2000 mg O
2 L
−1 [
32].
This means that in terms of costs, treatment 1 would be the most competitive to implement in the landfill. However, the PAC coagulant has the disadvantage that the aluminum contents present in the final effluent (Alinicial 1595 mg L−1 and Alfinal 2485 mg L−1) may require an additional process for their removal. Also, in the Caribbean region of Colombia where the sanitary landfill is located and treatment 1 would be implemented, it has a great potential for the use of photo treatment with UV energy, because the climate is tropical dry, it is hot all-year-round, therefore a renewable and sustainable source would take the maximum advantage.