Lower Danube Water Quality Quantified through WQI and Multivariate Analysis
Abstract
1. Introduction
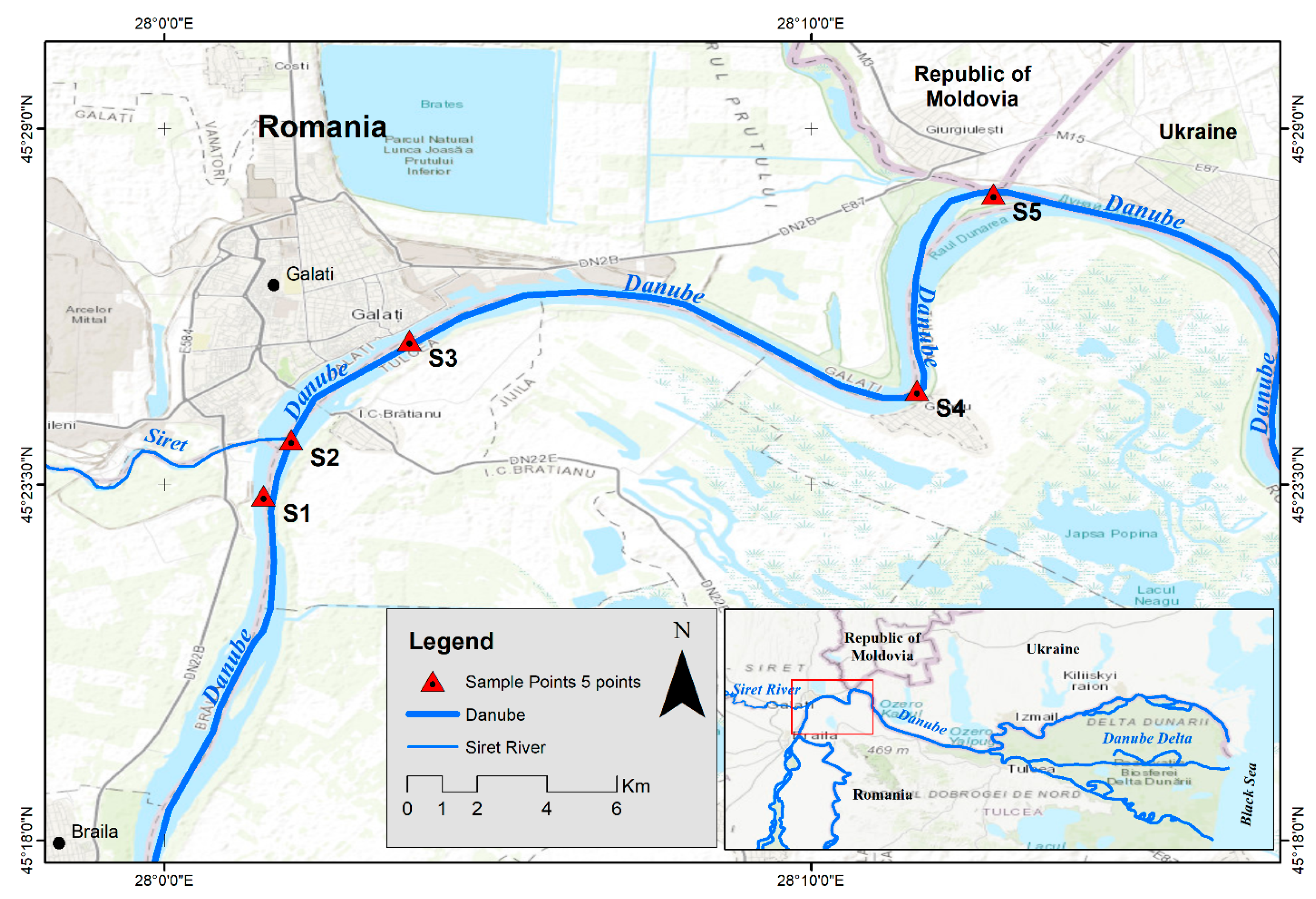
2. Materials and Methods
3. Results and Discussion
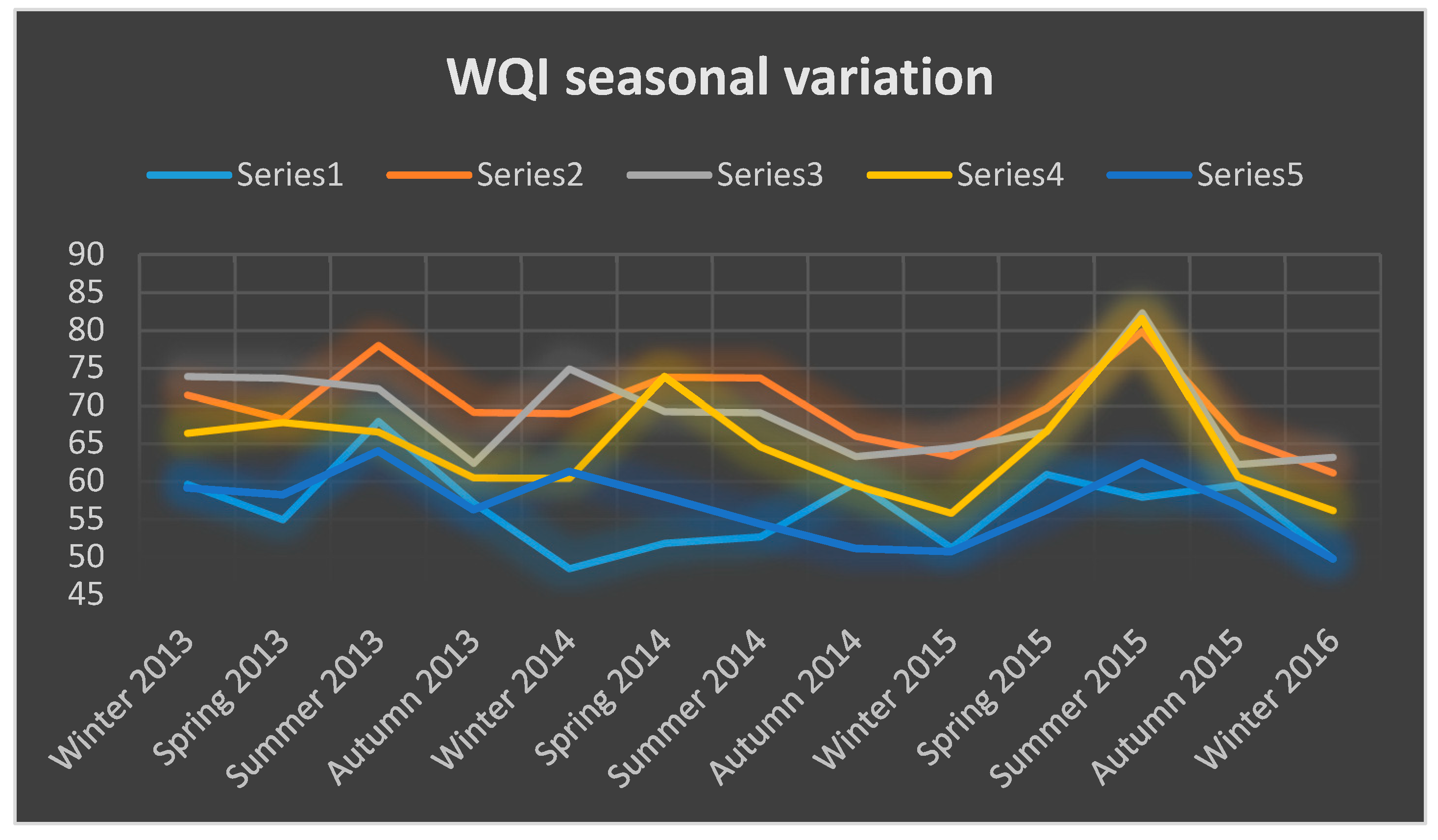
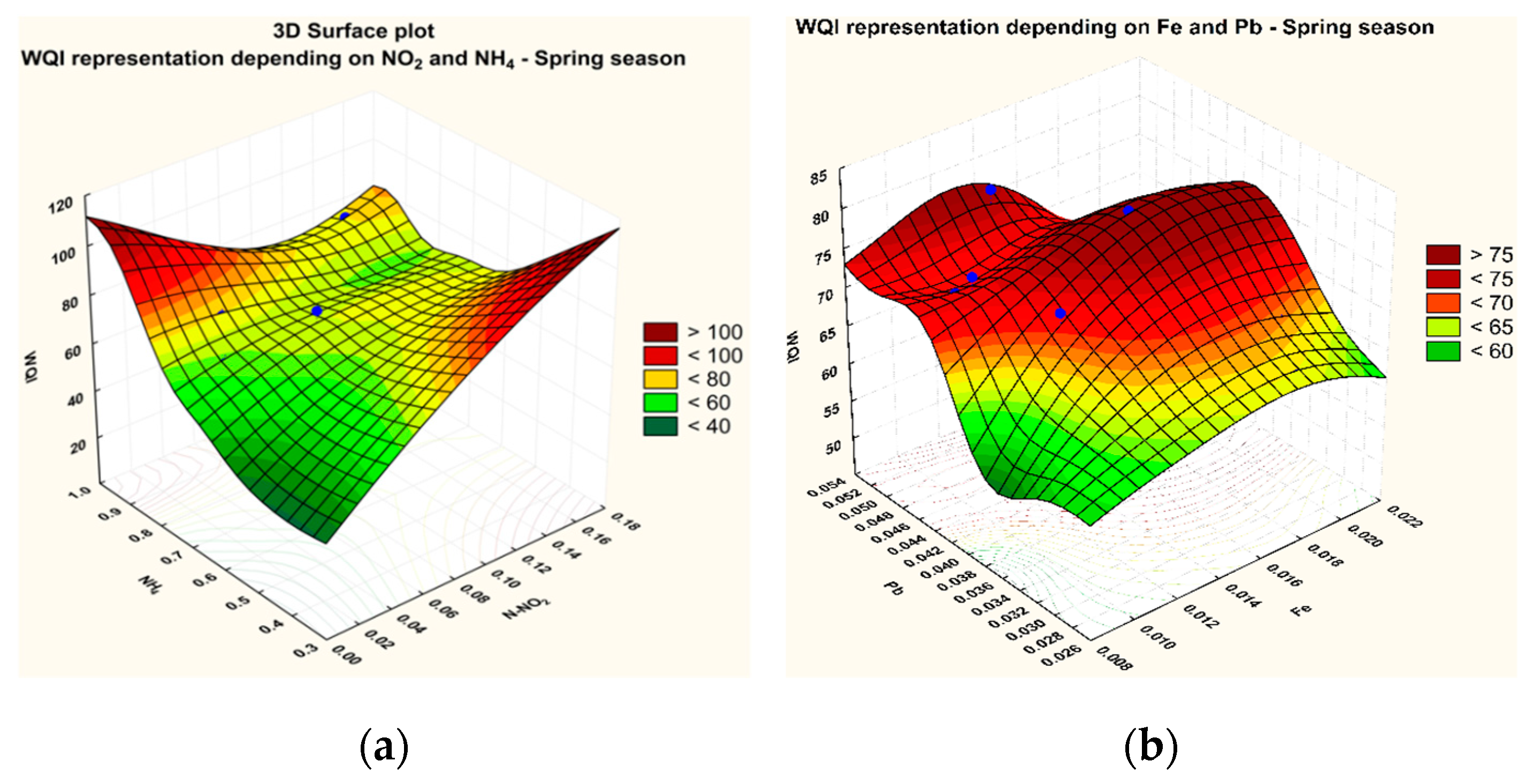
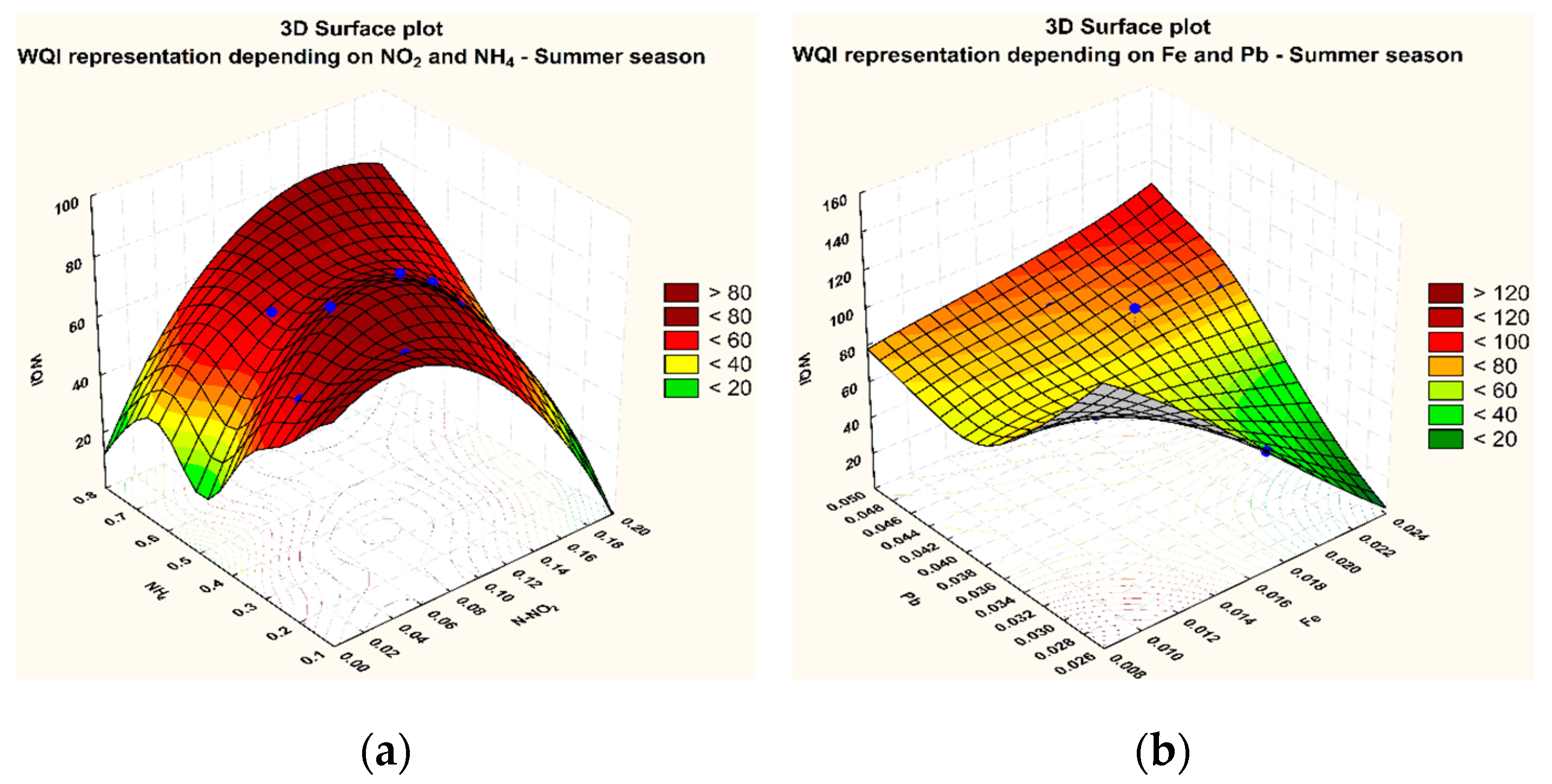
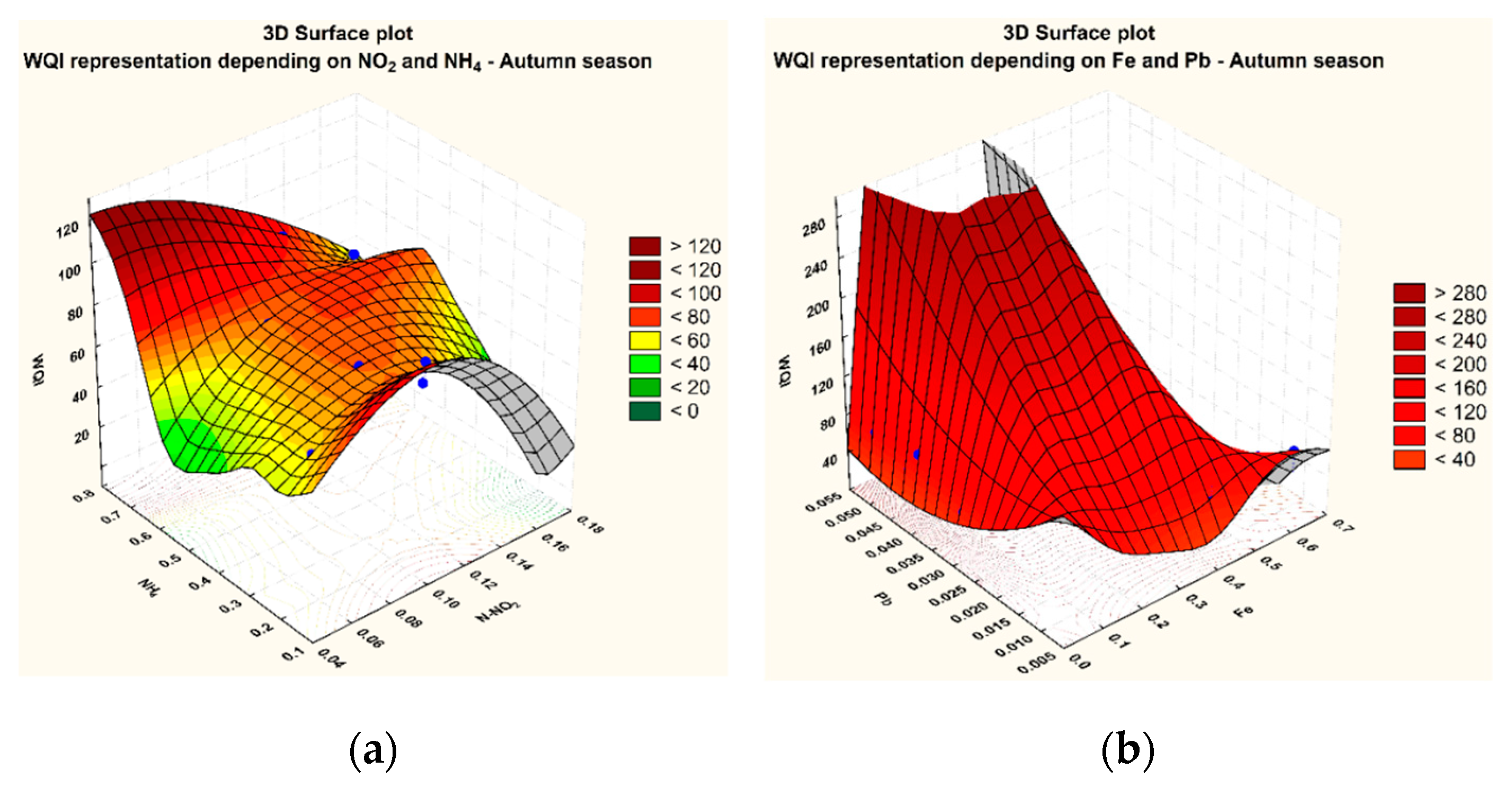
3.1. Variation of The WQI in The Sampling Points
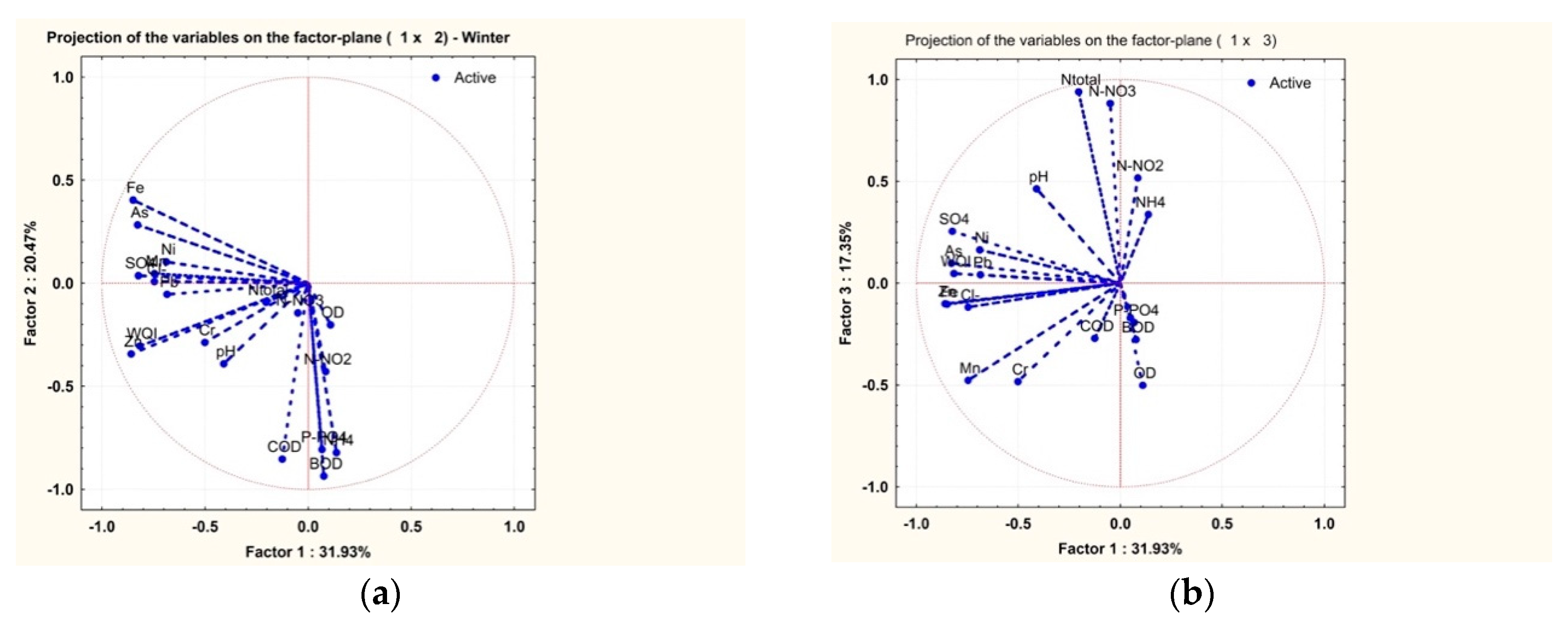
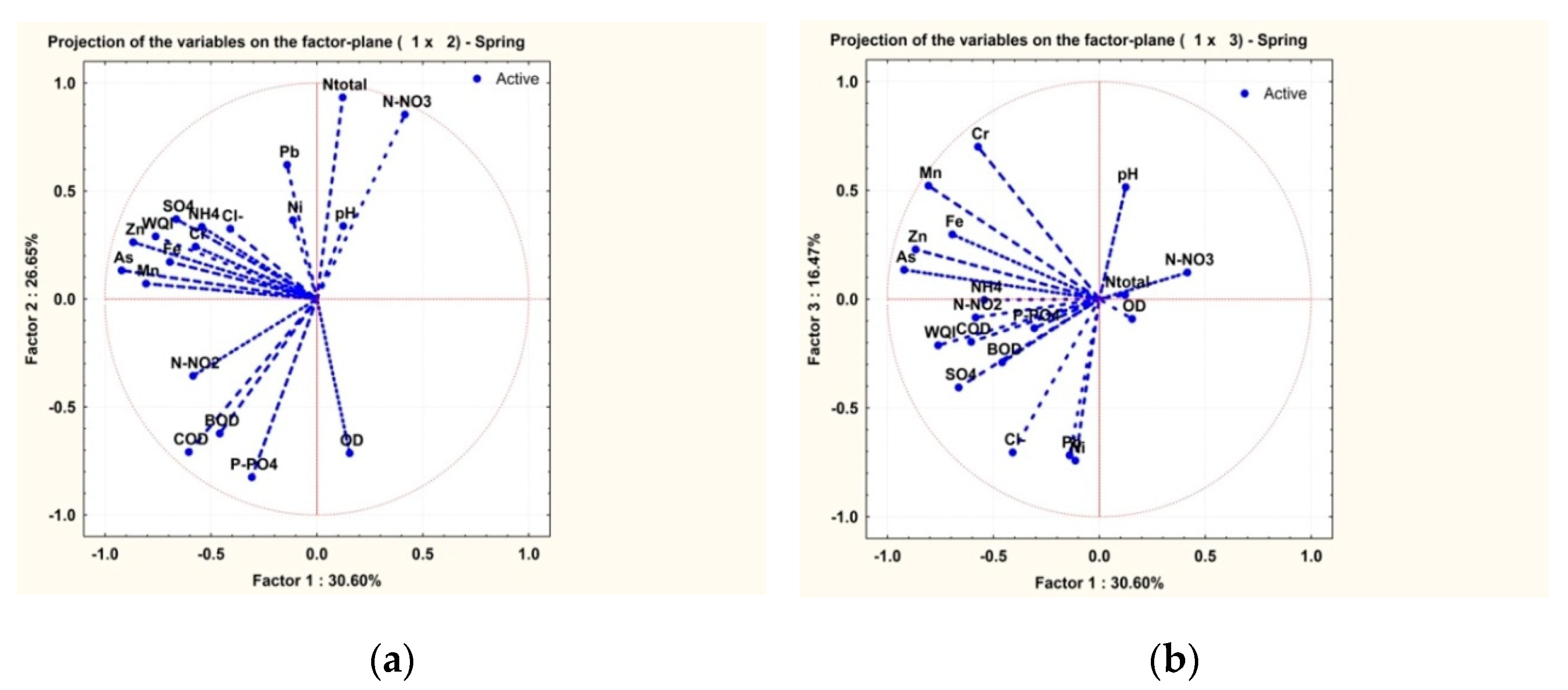
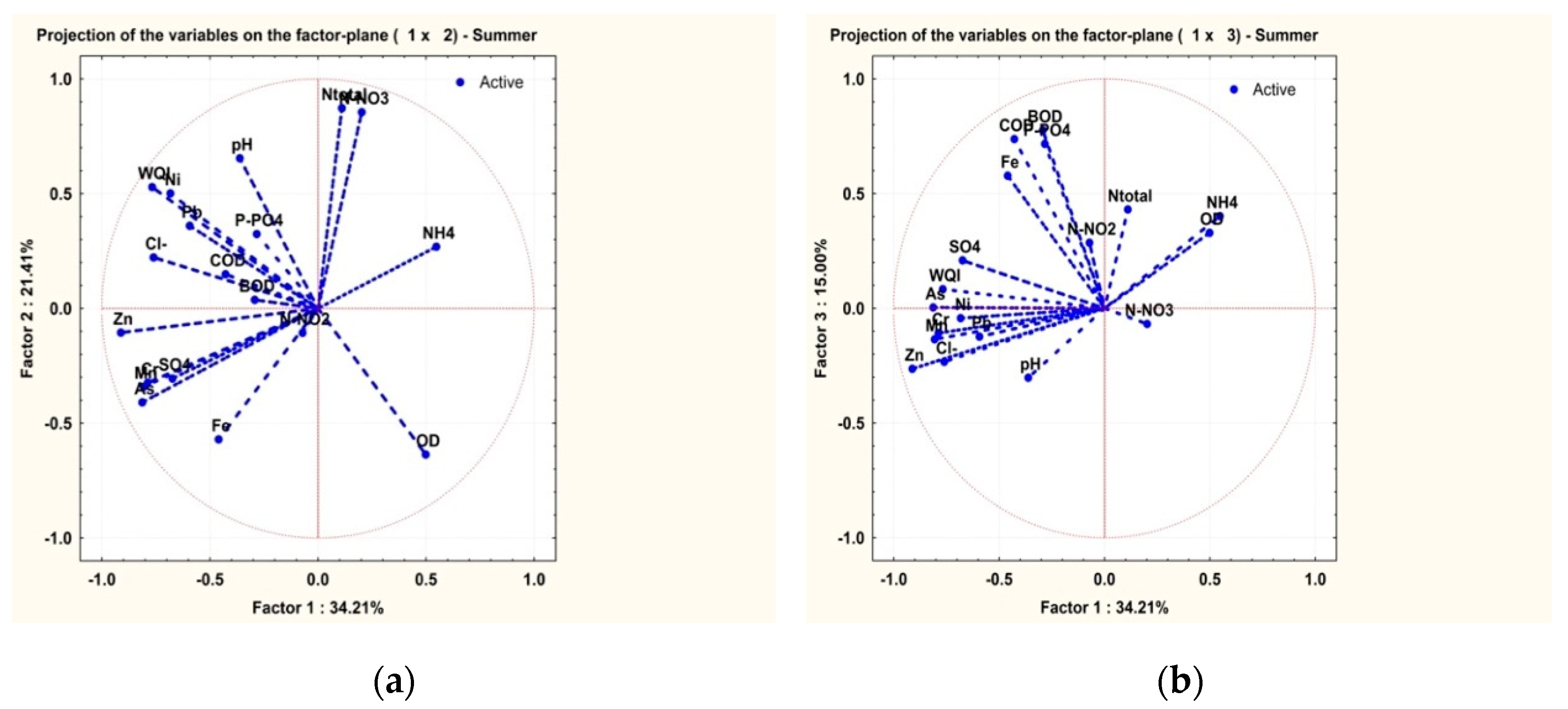
3.2. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bojarczuk, A.; Jelonkiewicz, L.; Lenart-Boron, A. The effect of anthropogenic and natural factors on the prevalence of physicochemical parameters of water and bacterial water quality indicators along the river Białka, southern Poland. Environ. Sci. Pollut. Res. 2018, 25, 10102–10114. [Google Scholar] [CrossRef] [PubMed]
- Iticescu, C.; Georgescu, L.P.; Topa, C.M. Assessing the Danube Water Quality Index in the City of Galati, Romania, Carpath. J. Earth Environ. Sci. 2013, 8, 155–164. [Google Scholar] [CrossRef]
- Karabulut, A.; Egoh, B.N.; Lanzanova, D.; Grizzetti, B.; Bidoglio, G.; Pagliero, L.; Bouraoui, F.; Aloe, A.; Reynaud, A.; Maes, J.; et al. Mapping water provisioning services to supportthe ecosystem–water–food–energy nexus in the Danube river basin. Ecosyst. Serv. 2016, 17, 278–292. [Google Scholar] [CrossRef]
- Voza, D.; Vukovic, M.; Takic, L.; Nikolic, D.; Mladenovic-Ranisavljevic, I. Application of multivariate statistical techniques in the water quality assessment of Danube river, Serbia. Arch. Environ. Protect. 2015, 41, 96–103. [Google Scholar] [CrossRef]
- Stoica, C.; Lucaciu, I.; Nicolau, M.; Vosniakos, F. Monitoring the ecological diversity of the aquatic Danube Delta systems in terms of spatial- temporal relationship. J. Environ. Prot. Ecol. 2012, 13, 476–485. [Google Scholar]
- Caldararu, A.; Rosati, I.; Barbone, E.; Georgescu, L.P.; Iticescu, C.; Basset, A. Implementing European water framework directive: Uncertainty degree of metrics for macroinvertebrates in transitional waters. Environ. Eng. Manag. J. 2010, 9, 1259–1266. [Google Scholar] [CrossRef]
- Iticescu, C.; Murariu, G.; Georgescu, L.P.; Burada, A.; Ţopa, C.M. Seasonal variation of the physico-chemical parameters and Water Quality Index (WQI) of Danube water in the transborder Lower Danube area. Rev. Chim. (Buchar.) 2016, 67, 1843–1849. [Google Scholar]
- Dinka, M.O.; Loiskandl, W.; Ndambuki, J.M. Hydrochemical characterization of various surface water and ground water resources available in Matahara areas, Fantalle Woreda of Oromiya region. J. Hydrol. Reg. Stud. 2015, 3, 444–456. [Google Scholar] [CrossRef]
- El Hawary, A.; Shaban, M. Improving drainage water quality: Constructed wetlands-performance assessment using multivariate and cost analysis. Water Sci. 2018, 32, 301–317. [Google Scholar] [CrossRef]
- Iqbal, M.M.; Shoaib, M.; Agwanda, P.; Lee, J.L. Modeling approach for water-quality management to control pollution concentration: A case study of Ravi River, Punjab, Pakistan. Water 2018, 10, 1068. [Google Scholar] [CrossRef]
- Iqbal, M.M.; Shoaib, M.; Farid, H.U.; Lee, J.L. Assessment of water quality profile using numerical modeling approach in major climate classes of Asia. Int. J. Environ. Res. Public Health 2018, 15, 2258. [Google Scholar] [CrossRef] [PubMed]
- Rakotondrabe, F.; Ndam Ngoupayou, J.R.; Mfonka, Z.; Rasolomanana, E.H.; Nyangono Abolo, A.J.; Ako Ako, A. Water quality assessment in the Bétaré-Oya gold mining area (East-Cameroon): Multivariate Statistical Analysis approach. Sci. Total Environ. 2018, 610–611, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Prasad, M.; Sunitha, V.; Sudharshan Reddy, Y.; Suvarna, B.; Muralidhara Reddy, B.; Ramakrishna Reddy, M. Data on water quality index development for groundwater quality assessment from Obulavaripalli Mandal, YSR district, A.P India. Data Brief 2019, 24, 103846. [Google Scholar] [CrossRef] [PubMed]
- Order 161/2006, The Normative on the Classification of Surface Water Quality in Order to Establish the Ecological s Romanian Ministry of Research and Innovation Tatus of Water Bodies; Official Monitor: Bucharest, Romania, 13 June 2006.
- Directive 2000/60/ec of the European Parliament and of the Council of 23 October 2000. Establishing a Framework for Community Water Policy. Available online: https://eur-lex.europa.eu/legal-content/RO/TXT/?uri=celex%3A32000L0060 (accessed on 16 June 2017).
- Popa, P.; Murariu, G.; Timofti, M.; Georgescu, L.P. Multivariate statistical analyses of water quality of Danube river at Galati, Romania. Environ. Eng. Manag. J. 2018, 17, 491–509. [Google Scholar]
- National Institute of Hydrology and Water Management Daily Reports. Available online: http://www.inhga.ro/diagnoza _si_prognoza_dunare (accessed on 20 May 2019).
- SR ISO 10523, 2009—Water Quality—Determination of pH; ISO: Geneva, Switzerland, 2009.
- SR EN 1899-2:2002—Water Quality—Determination of Biochemical Oxygen Demand after N Days (CBOn). Part 2: Method for Undiluted Samples; ISO: Geneva, Switzerland, 2002.
- SR EN ISO 6878:2005—Water Quality—Determination of Phosphorus. Spectrophotometric Method with Ammonium Molybdate; ISO: Geneva, Switzerland, 2005.
- SR EN ISO 11732:2005—Water Quality—Determination of Ammoniacal Nitrogen. Flow Analysis Method (CFA and FIA) and Spectrometric Detection; ISO: Geneva, Switzerland, 2005.
- ISO 15705:2002—Water Quality—Determination of the Chemical Oxygen Demand Index (ST-COD)—Small-Scale Sealed-Tube Method; ISO: Geneva, Switzerland, 2002.
- SR ISO 6777/1996—Water Quality—Determination of the Nitrogen from Nitrite Determination by Spectrometric Detection; ISO: Geneva, Switzerland, 1996.
- SR ISO 7890-3/2000—Water Quality—Determination of the Nitrogen from Nitrate Determination by Spectrometric Detection; ISO: Geneva, Switzerland, 2000.
- SR EN ISO 11905-1:2003—Water Quality—Determination of Nitrogen Content. Part 1: Oxidative Mineralization Method with Peroxodisulphate; ISO: Geneva, Switzerland, 2003.
- SR ISO 6332:1996/C91:2006—Water Quality—Determination of Iron Content. Spectrometric Method with 1,10—Phenanthroline; ISO: Geneva, Switzerland, 2006.
- SR ISO 9297/2001—Water Quality—Determination of Chloride Content. Titration with Silver Nitrate Using Chromate as Indicator (Mohr Method); ISO: Geneva, Switzerland, 2001.
- STAS 3069-87—Water Quality—Determination of Sulphate Content; ASRO: Bucharest, Romania, 2003.
- SR ISO 10566:2001—Surface Water and Wastewater—Determination of Lead; ISO: Geneva, Switzerland, 2001.
- SR ISO 9174-98—Water Quality—Determination of Cr Total—Spectrometric Method; ISO: Geneva, Switzerland, 1998.
- Clocotici, V. Introduction to Multivariate Statistics, Univ. Al. I. Cuza, Iasi, Faculty of Informatics. 2007. Available online: http://www.docstemplate.com/statistica-multivariata (accessed on 15 September 2016).
- Jianqin, M.; Jingjing, G.; Xiaojie, L. Water quality evaluation model based on principal component analysis and information entropy: Application in Jinshui River. J. Res. Ecol. 2010, 1, 249–252. [Google Scholar] [CrossRef]
- Garizi, A.Z.; Sheikh, V.; Sadoddin, A. Assessment of seasonal variations of chemical characteristics in surface water using multivariate statistical methods. Int. J. Environ. Sci. Technol. 2011, 8, 581–592. [Google Scholar] [CrossRef]
- Kebede, Y.K.; Kebede, T. Application of Principal Component Analysis in Surface Water Quality Monitoring. In Principal Component Analysis—Engineering Applications; Sanguansat, P., Ed.; IntechOpen: London, UK, 2012; pp. 83–100. [Google Scholar]
- Giordano, G. A new dimension in the factorial techniques: The response surface. Stat. Appl. 2006, 18, 359–373. [Google Scholar]
- Jiménez-Contreras, E.; Torres-Salinas, D.; Bailón Moreno, R.; Ruiz Baños, R.; Delgado López-Cózar, E. Response Surface Methodology and its application in evaluating scientific activity. Scientometrics 2008, 79, 201–218. [Google Scholar] [CrossRef]
- Kaurish, F.W.; Younos, T. Developing a standardized water quality index for evaluating surface water quality. J. Am. Water Resour. Assoc. 2007, 3, 533–545. [Google Scholar] [CrossRef]
- Takic, L.M.; Mladenovic-Ranisavljevic, I.I.; Nikolic, V.D.; Nikolic, L.B.; Vukovic, M.V.; Zivkovic, N.V. The assessment of the Danube water quality in Serbia. Adv. Technol. 2012, 1, 58–66. [Google Scholar]
- Water Pollution Control—A Guide to the Use of Water Quality Management Principles; Helmer, R., Hespanhol, I., Eds.; F & FN Spon: London, UK, 1997. [Google Scholar]
- Ayeni, O.; Soneye, A.S.O. Interpretation of surface water quality using principal components analysis and cluster analysis. J. Geogr. Reg. Plan. 2013, 6, 132–141. [Google Scholar] [CrossRef]
- Burada, A.; Topa, C.M.; Georgescu, L.P.; Teodorof, L.; Nastase, C.; Seceleanu-Odor, D.; Iticescu, C. Heavy Metals Environment Accumulation in Somova—Parches Aquatic Complex from the Danube Delta Area. Rev. Chim. (Buchar.) 2015, 66, 48–54. [Google Scholar]
- Ţopa, M.C.; Timofti, M.; Burada, A.; Iticescu, C.; Georgescu, L.P. Danube water quality during and after flood near an urban agglomeration. J. Environ. Prot. Ecol. 2015, 16, 1255–1261. [Google Scholar]
- Pintilie, V.; Ene, A.; Georgescu, L.P.; Moraru, L.P.; Iticescu, C. Measurements of gross alpha and beta activity in drinking water from Galati region, Romania. Rom. Rep. Phys. 2016, 68, 1208–1220. [Google Scholar]
- Rodríguez-Romero, A.J.; Rico-Sánchez, A.E.; Mendoza-Martínez, E.; Gómez-Ruiz, A.; Sedeño-Díaz, J.E.; López-López, E. Impact of Changes of Land Use on Water Quality, from Tropical Forest to Anthropogenic Occupation: A Multivariate Approach. Water 2018, 10, 1518. [Google Scholar] [CrossRef]
- Tyagi, S.; Sharma, B.; Singh, P.; Dobhal, R. Water Quality Assessment in Terms of Water Quality Index. Am. J. Water Resour. 2013, 1, 34–38. [Google Scholar]
- Iticescu, C.; Georgescu, L.P.; Topa, C.; Murariu, G. Monitoring the Danube Water Quality near the Galati City. J. Environ. Prot. Ecol. 2015, 15, 30–38. [Google Scholar]
- Timofti, M.; Popa, P.; Murariu, G.; Georgescu, L.; Iticescu, C.; Barbu, M. Complementary approach for numerical modelling of physicochemical parameters of the Prut River aquatic system. J. Environ. Prot. Ecol. (JEPE) 2016, 1, 53–63. [Google Scholar]
- Chowdhury, R.M.; Muntasir, S.Y.; Hossain, M.M. Water quality index of water bodies along Faridpur-Barisal road in Bangladesh. Glob. Eng. Technol. Rev. 2012, 2, 1–8. [Google Scholar]
- Mena-Rivera, L.; Salgado-Silva, V.; Benavides-Benavides, C.; Coto-Campos, J.M.; Swinscoe, T.H.A. Spatial and Seasonal Surface Water Quality Assessment in a Tropical Urban Catchment: Burío River, Costa Rica. Water 2017, 9, 558. [Google Scholar] [CrossRef]
- Georgescu, P.L.; Voiculescu, M.; Dragan, S.; Timofti, M.; Caldararu, A. Study of spatial and temporal variations of some physic-chemical parameters of Lower Siret River. J. Environ. Prot. Ecol. 2010, 11, 837–844. [Google Scholar]
- Voiculescu, M.; Georgescu, P.L.; Dragan, S.; Timofti, M.; Caldararu, A. Study of anthropogenic effects on the quality of Lower Prut River. J. Environ. Prot. Ecol. 2011, 12, 16–24. [Google Scholar]


| Variable | S1 Min Value | S1 Min Value | S1 Means ± Std.Dev | S2 Min Value | S2 Min Value | S2 Means ± Std.Dev. | S3 Min Value | S3 Min Value | S3 Means ± Std.Dev. | S4 Min Value | S4 Min Value | S4 Means ± Std.Dev. | S5 Min Value | S5 Min Value | S5 Means ± Std.Dev. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH (upH) | 7.82 | 8.82 | 8.30 ± 0.46 | 7.99 | 8.70 | 8.32 ± 0.33 | 7.82 | 8.36 | 8.14 ± 0.19 | 7.87 | 8.34 | 8.11 ± 0.15 | 7.82 | 8.28 | 8.14 ± 0.13 |
| OD (mg·L−1) | 4.78 | 12.00 | 5.65 ± 0.84 | 4.14 | 11.00 | 5.59 ± 1.34 | 4.73 | 9.00 | 5.84 ± 0.96 | 5.62 | 7.60 | 6.24 ± 0.52 | 5.12 | 9.00 | 6.56± 1.81 |
| COD (mg·L−1) | 5.5 | 35 | 19.30 ± 13.62 | 6.00 | 38.00 | 21.10 ± 13.79 | 5.7 | 34 | 16.66 ± 12.41 | 7.2 | 37 | 22.36 ± 11.25 | 6.1 | 26.0 | 15.20 ± 5.88 |
| BOD (mg·L−1) | 1.2 | 7.3 | 4.26 ± 2.70 | 2.10 | 8.30 | 4.63 ± 3.21 | 1.7 | 7.7 | 4.46 ± 2.89 | 1.7 | 6.7 | 4.10 ± 2.26 | 2.0 | 5.2 | 3.40 ± 1.31 |
| N-NH4+ (mg·L−1) | 0.242 | 0.842 | 0.87 ± 0.05 | 0.231 | 0.938 | 0.82 ± 0.12 | 0.248 | 0.799 | 0.82 ± 0.09 | 0.354 | 0.959 | 0.78 ± 0.15 | 0.205 | 0.752 | 0.56 ± 0.20 |
| N-NO2− (mg·L−1) | 0.021 | 0.135 | 0.07 ± 0.04 | 0.035 | 0.148 | 0.10 ± 0.04 | 0.0118 | 0.132 | 0.07 ± 0.04 | 0.024 | 0.178 | 0.11 ± 0.07 | 0.0106 | 0.086 | 0.03 +/- 0.02 |
| N-NO3− (mg·L−1) | 1.98 | 10.21 | 7.37 ± 3.72 | 2.14 | 8.28 | 6.82 ± 2.32 | 2.18 | 9.22 | 7.31 ± 2.73 | 3.30 | 10.32 | 8.55 ± 2.79 | 2.26 | 9.26 | 6.84 ± 3.52 |
| N-total (mg·L−1) | 1.20 | 11.97 | 9.37 ± 2.85 | 3.93 | 10.19 | 8.95 ± 1.31 | 4.51 | 12.06 | 10.21 ± 3.20 | 5.2 | 11.62 | 10.59 ± 0.84 | 4.10 | 10.12 | 7.76 ± 3.14 |
| P-PO43− (mg·L−1) | 0.061 | 0.570 | 0.11 ± 0.05 | 0.054 | 0.74 | 0.18 ± 0.08 | 0.0106 | 0.89 | 0.12 ± 0.08 | 0.093 | 0.93 | 0.21 ± 0.05 | 0.041 | 0.680 | 0.15 ± 0.11 |
| SO42− (mg·L−1) | 98.034 | 131.000 | 112.34 ± 16.91 | 123 | 149 | 143.00 ± 4.36 | 126 | 150 | 135.33 ± 12.70 | 115 | 154 | 122.00 ± 8.66 | 94 | 123 | 109.66 ± 12.58 |
| Cl− (mg·L−1) | 39.00 | 48.00 | 40.00 ± 1.73 | 41 | 59 | 49.33 ± 8.50 | 42 | 62 | 55.00 ± 2.64 | 42 | 52 | 47.66 ± 5.13 | 33 | 45 | 43.00 ± 2.65 |
| Fe-total (mg·L−1) | 0.43 | 0.72 | 0.64 ± 0.0454 | 0.42 | 0.73 | 0.55 ± 0.025 | 0.42 | 0.64 | 0.505 ± 0.03 | 0.32 | 0.43 | 0.41 ± 0.03 | 0.29 | 0.41 | 0.39 ± 0.02 |
| Cr-total (mg·L−1) | 0.027 | 0.040 | 0.030 ± 0.00 | 0.039 | 0.044 | 0.04 ± 0.01 | 0.032 | 0.052 | 0.04 ± 0.01 | 0.039 | 0.049 | 0.04 ± 0.01 | 0.028 | 0.0381 | 0.03 ± 0.01 |
| Pb2+ (μg·L−1) | 6 | 9 | 7.3 ± 1.30 | 7 | 12 | 7.70 ± 0.49 | 6.9 | 13 | 8.80 ± 1.27 | 7.2 | 9.6 | 8.5 ± 0.58 | 6.1 | 8.1 | 7.80 ± 0.30 |
| Ni2+ (μg·L−1) | 12 | 19 | 16.30 ± 3.79 | 10 | 23 | 15.70 ± 5.51 | 14 | 22 | 15.30 ± 2.31 | 10 | 18 | 11.30 ± 1.15 | 9 | 18 | 9.60 ± 051 |
| Mn2+ (μg·L−1) | 95 | 120 | 105.30 ± 12.70 | 87 | 130 | 8.83 ± 1.53 | 80 | 120 | 81.70 ± 1.53 | 30 | 80 | 51.70 ± 21.50 | 25 | 56 | 33.00 ± 7.00 |
| Zn2+ (mg·L−1) | 0.120 | 0.190 | 0.18 ± 0.017 | 0.15 | 0.21 | 0.18 ± 0.03 | 0.14 | 0.191 | 0.18 ± 0.00 | 0.11 | 0.16 | 0.13 ± 0.02 | 0.0123 | 0.13 | 0.10 ± 0.02 |
| As3+ (μg·L−1) | 2 | 4.3 | 3.40 ± 1.21 | 3 | 5 | 3.90 ± 0.15 | 3 | 4.6 | 3.40 ± 0.23 | 2 | 4.2 | 2.6 ± 0.50 | 0.8 | 3.1 | 1.30 ± 0.42 |
| Variable/Monitoring Point | Effect | SS | Degree of Freedom | MS | F | p |
|---|---|---|---|---|---|---|
| pH | ||||||
| S1 | Intercept | 785.05 | 1.00 | 785.05 | 11,988.60 | 0.00 |
| Season | 0.18 | 3.00 | 0.06 | 0.94 | 0.47 | |
| OD | ||||||
| S1 | Intercept | 533.86 | 1.00 | 533.86 | 332.52 | 0.00 |
| Season | 46.22 | 3.00 | 15.40 | 9.59 | 0.01 | |
| COD | ||||||
| S1 | Intercept | 2901.63 | 1.00 | 2901.63 | 24.77 | 0.00 |
| Season | 134.09 | 3.00 | 44.69 | 0.38 | 0.77 | |
| BOD | ||||||
| S1 | Intercept | 130.42 | 1.00 | 130.42 | 37.17 | 0.00 |
| Season | 2.98 | 3.00 | 0.99 | 0.28 | 0.83 | |
| N-NH4+ | ||||||
| S1 | Intercept | 3.16 | 1.00 | 3.16 | 99.75 | 0.00 |
| Season | 0.34 | 3.00 | 0.11 | 3.62 | 0.06 | |
| N-NO2− | ||||||
| S1 | Intercept | 0.06 | 1.00 | 0.06 | 45.55 | 0.00 |
| Season | 0.01 | 3.00 | 0.00 | 2.96 | 0.09 | |
| N-NO3− | ||||||
| S1 | Intercept | 221.96 | 1.00 | 221.96 | 46.18 | 0.00 |
| Season | 38.74 | 3.00 | 12.91 | 2.69 | 0.12 | |
| Ntotal | ||||||
| S1 | Intercept | 415.25 | 1.00 | 415.25 | 77.11 | 0.00 |
| Season | 51.43 | 3.00 | 17.14 | 3.18 | 0.08 | |
| P-PO43− | ||||||
| S1 | Intercept | 0.41 | 1.00 | 0.41 | 11.06 | 0.01 |
| Season | 0.07 | 3.00 | 0.02 | 0.65 | 0.60 | |
| SO42− | ||||||
| S1 | Intercept | 167,332.10 | 1.00 | 167,332.10 | 1160.99 | 0.00 |
| Season | 152.50 | 3.00 | 50.80 | 0.35 | 0.78 | |
| Error | 1153.00 | 8.00 | 144.1 | |||
| Cl− | ||||||
| S1 | Intercept | 22,883.08 | 1.00 | 22,883.08 | 4228.44 | 0.00 |
| Season | 81.19 | 3.00 | 27.06 | 5.00 | 0.03 | |
| Fe-total | ||||||
| S1 | Intercept | 4.22 | 1.00 | 4.22 | 1988.02 | 0.00 |
| Season | 0.06 | 3.00 | 0.02 | 9.55 | 0.01 | |
| Cr-total | ||||||
| S1 | Intercept | 0.01 | 1.00 | 0.01 | 2823.36 | 0.00 |
| Season | 0.00 | 3.00 | 0.00 | 9.59 | 0.01 | |
| Pb2+ | ||||||
| S1 | Intercept | 0.00 | 1.00 | 0.00 | 691.69 | 0.00 |
| Season | 0.00 | 3.00 | 0.00 | 0.83 | 0.51 | |
| Error | 0.00 | 8.00 | 0.00 | |||
| Ni2+ | ||||||
| S1 | Intercept | 0.00 | 1.00 | 0.00 | 420.76 | 0.00 |
| Season | 0.00 | 3.00 | 0.00 | 0.41 | 0.75 | |
| Mn2+ | ||||||
| S1 | Intercept | 0.14 | 1.00 | 0.14 | 1479.18 | 0.00 |
| Season | 0.00 | 3.00 | 0.00 | 2.01 | 0.19 | |
| Zn2+ | ||||||
| S1 | Intercept | 0.32 | 1.00 | 0.32 | 612.13 | 0.00 |
| Season | 0.00 | 3.00 | 0.00 | 2.05 | 0.18 | |
| As3+ | ||||||
| S1 | Intercept | 0.00 | 1.00 | 0.00 | 223.16 | 0.00 |
| Season | 0.00 | 3.00 | 0.00 | 0.18 | 0.90 |
| Variable/Monitoring Point | Effect | SS | Degree of Freedom | MS | F | p |
|---|---|---|---|---|---|---|
| OD | ||||||
| S1 | Intercept | 533.86 | 1.00 | 533.86 | 332.51 | 0.00 |
| Season | 46.23 | 3.00 | 15.41 | 9.59 | 0.01 | |
| N-NH4+ | ||||||
| S2 | Intercept | 3.54 | 1.00 | 3.55 | 160.30 | 0.00 |
| Season | 0.36 | 3.00 | 0.12 | 5.42 | 0.02 | |
| S3 | Intercept | 2.56 | 1.00 | 2.56 | 80.75 | 0.00 |
| Season | 0.39 | 3.00 | 0.13 | 4.14 | 0.05 | |
| N-NO2− | ||||||
| S2 | Intercept | 0.11 | 1.00 | 0.11 | 133.88 | 0.00 |
| Season | 0.01 | 3.00 | 0.00 | 3.30 | 0.04 | |
| S3 | Intercept | 0.07 | 1.00 | 0.068978 | 83.12 | 0.00 |
| Season | 0.01 | 3.00 | 0.003806 | 4.58 | 0.05 | |
| N-NO3− | ||||||
| S3 | Intercept | 258.26 | 1.00 | 258.26 | 81.13 | 0.00 |
| Season | 41.84 | 3.00 | 13.95 | 4.38 | 0.04 | |
| Cl− | ||||||
| S1 | Intercept | 22,883.08 | 1.00 | 22,883.08 | 4228.44 | 0.00 |
| Season | 81.19 | 3.00 | 27.06 | 5.001 | 0.03 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iticescu, C.; Georgescu, L.P.; Murariu, G.; Topa, C.; Timofti, M.; Pintilie, V.; Arseni, M. Lower Danube Water Quality Quantified through WQI and Multivariate Analysis. Water 2019, 11, 1305. https://doi.org/10.3390/w11061305
Iticescu C, Georgescu LP, Murariu G, Topa C, Timofti M, Pintilie V, Arseni M. Lower Danube Water Quality Quantified through WQI and Multivariate Analysis. Water. 2019; 11(6):1305. https://doi.org/10.3390/w11061305
Chicago/Turabian StyleIticescu, Catalina, Lucian P. Georgescu, Gabriel Murariu, Catalina Topa, Mihaela Timofti, Violeta Pintilie, and Maxim Arseni. 2019. "Lower Danube Water Quality Quantified through WQI and Multivariate Analysis" Water 11, no. 6: 1305. https://doi.org/10.3390/w11061305
APA StyleIticescu, C., Georgescu, L. P., Murariu, G., Topa, C., Timofti, M., Pintilie, V., & Arseni, M. (2019). Lower Danube Water Quality Quantified through WQI and Multivariate Analysis. Water, 11(6), 1305. https://doi.org/10.3390/w11061305







