Optimization Study for the Desorption of Methylene Blue Dye from Clay Based Adsorbent Coating
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Solution Used
2.2. Adsorption Study
2.3. Desorption Study
2.4. Optimization Study
2.5. Characterization
3. Results and Discussion
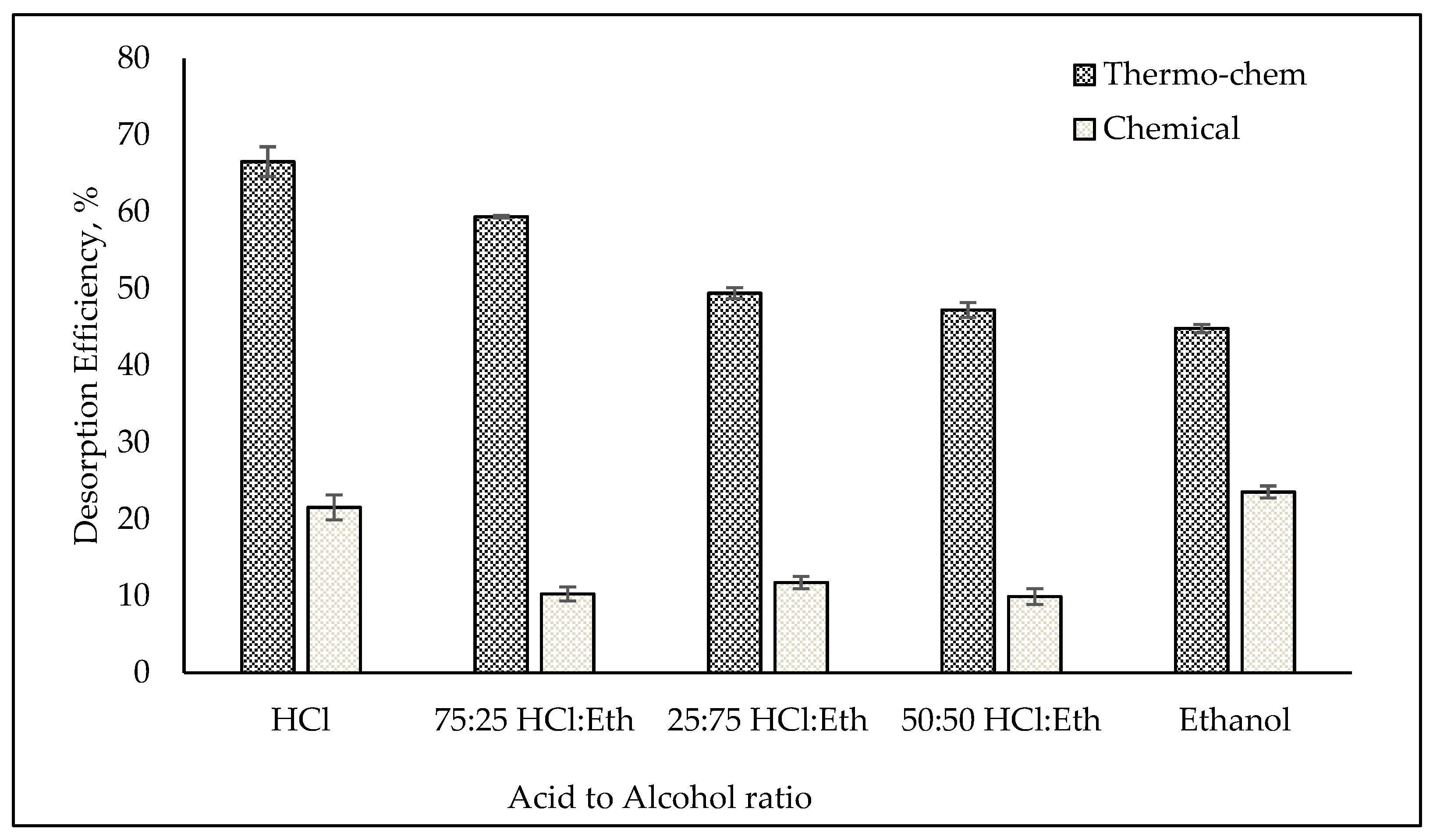
3.1. Desorption Study
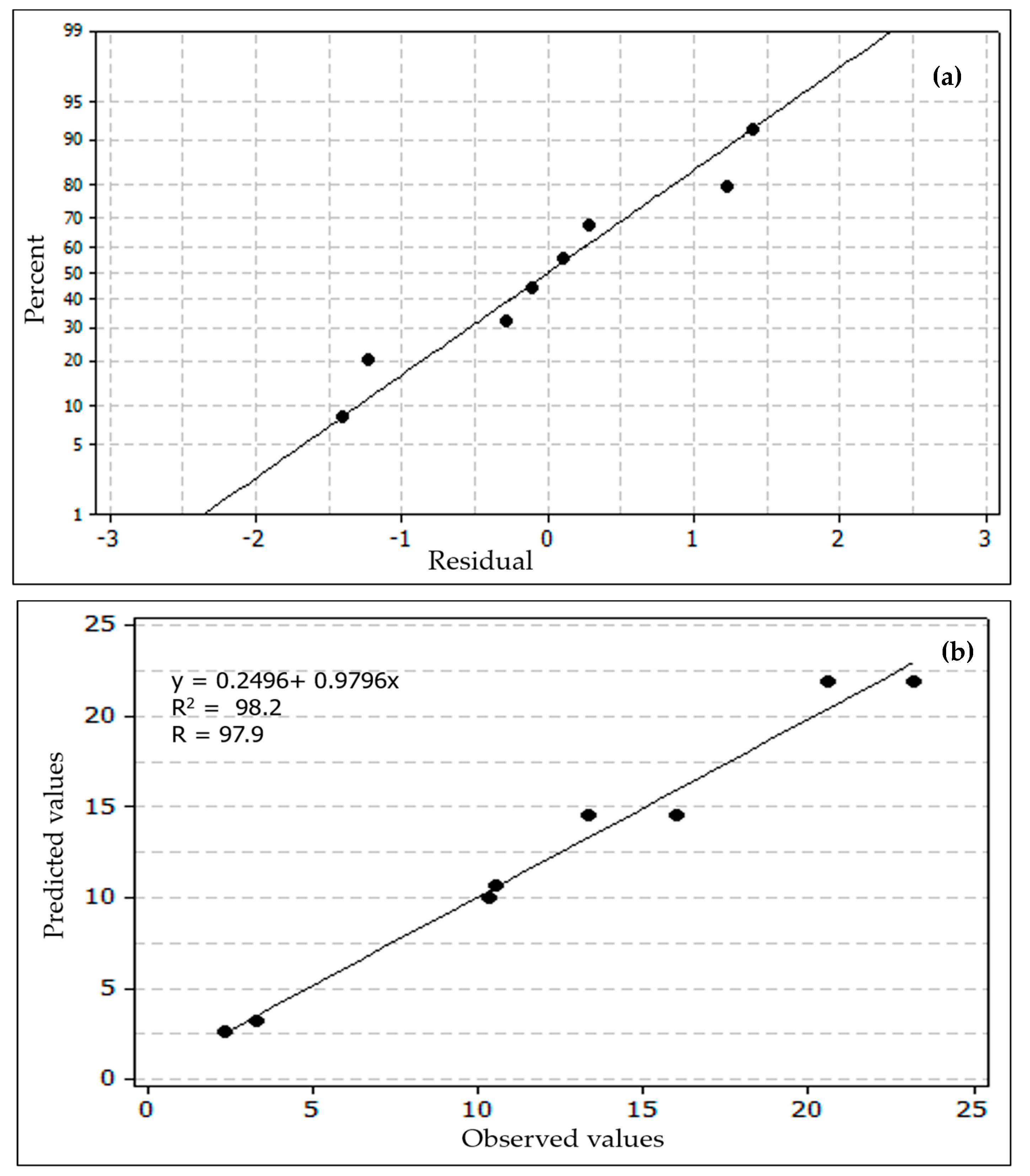
3.2. Optimization Study
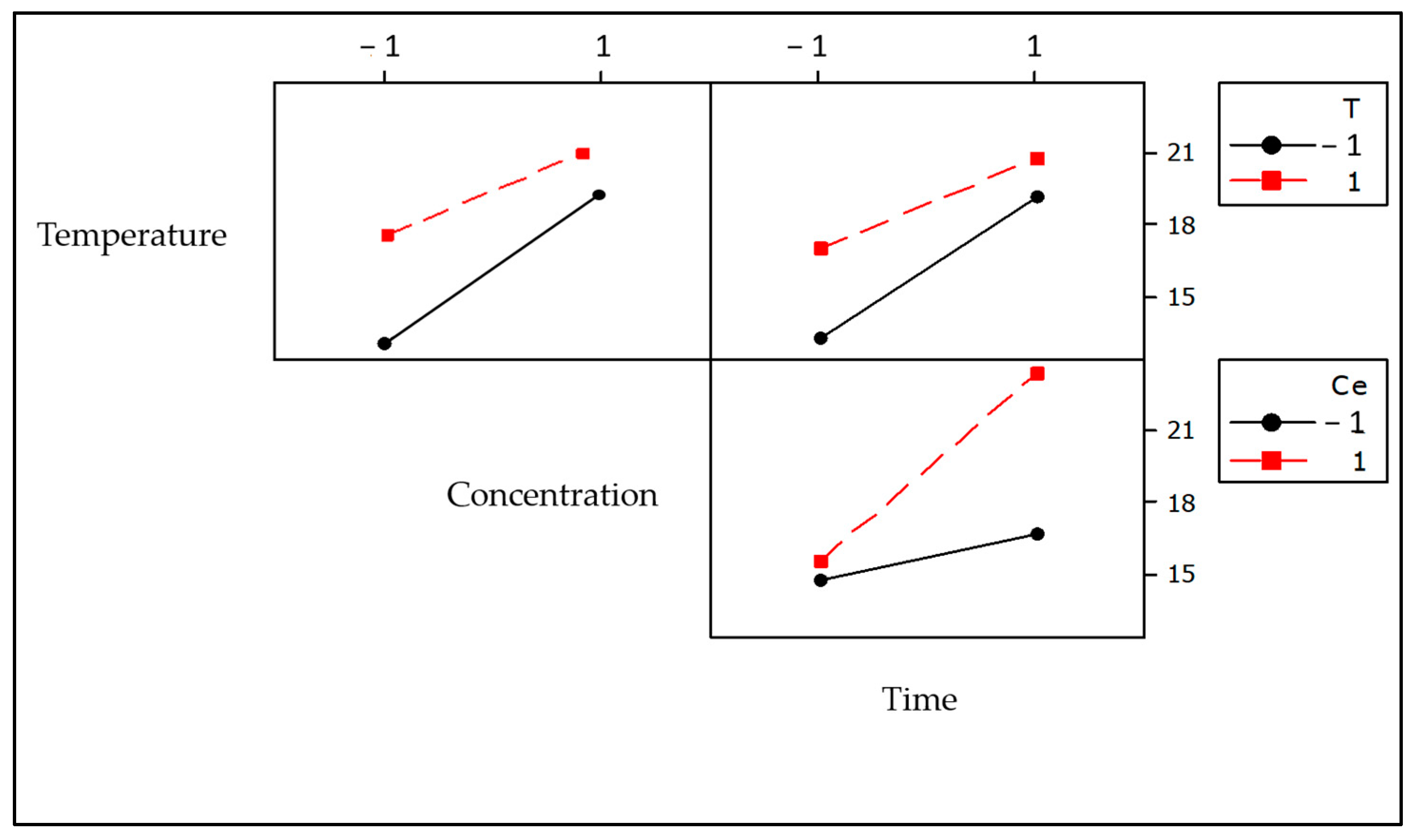
3.2.1. Main Effects
3.2.2. Interaction Plots
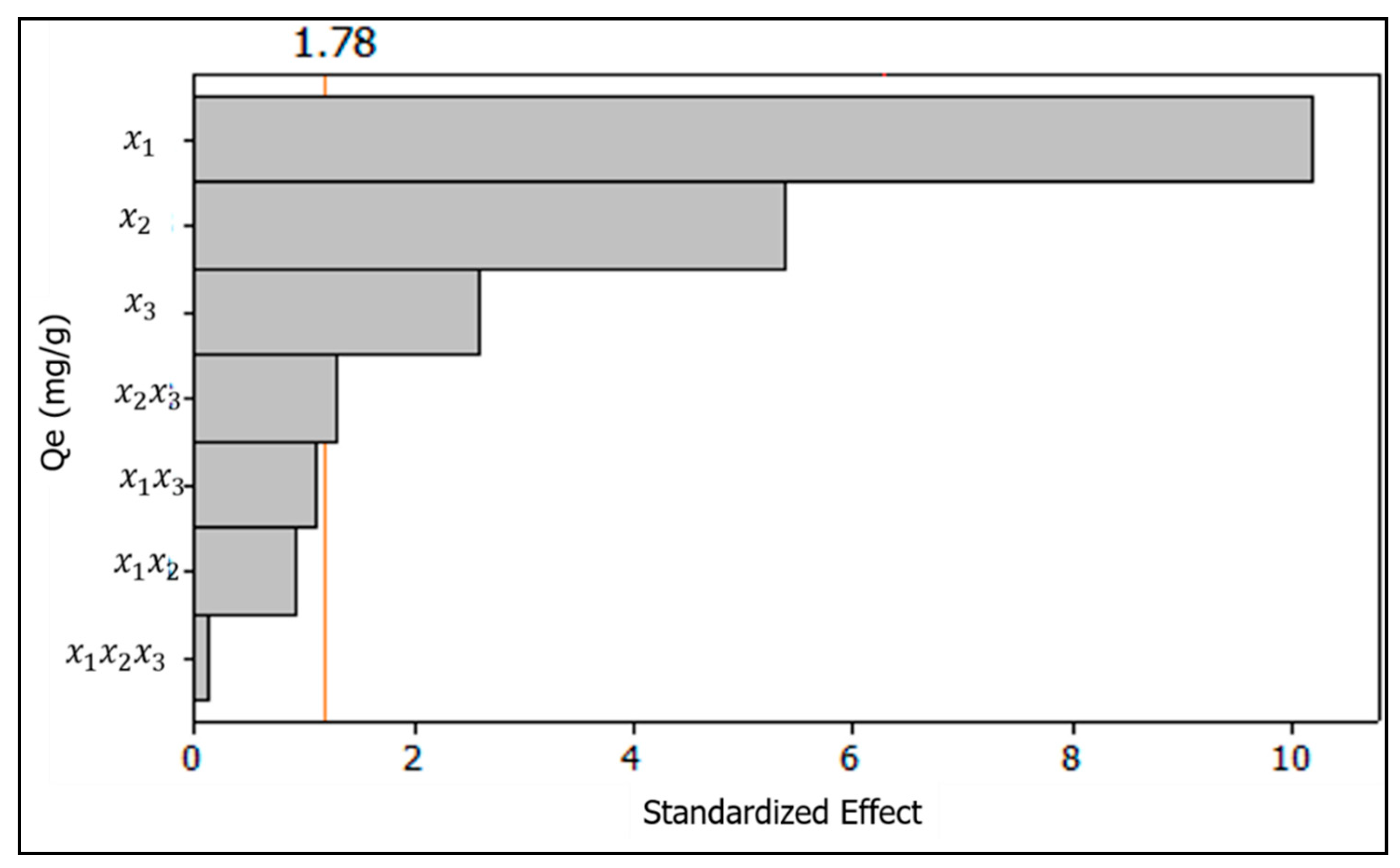
3.2.3. The Pareto Chart
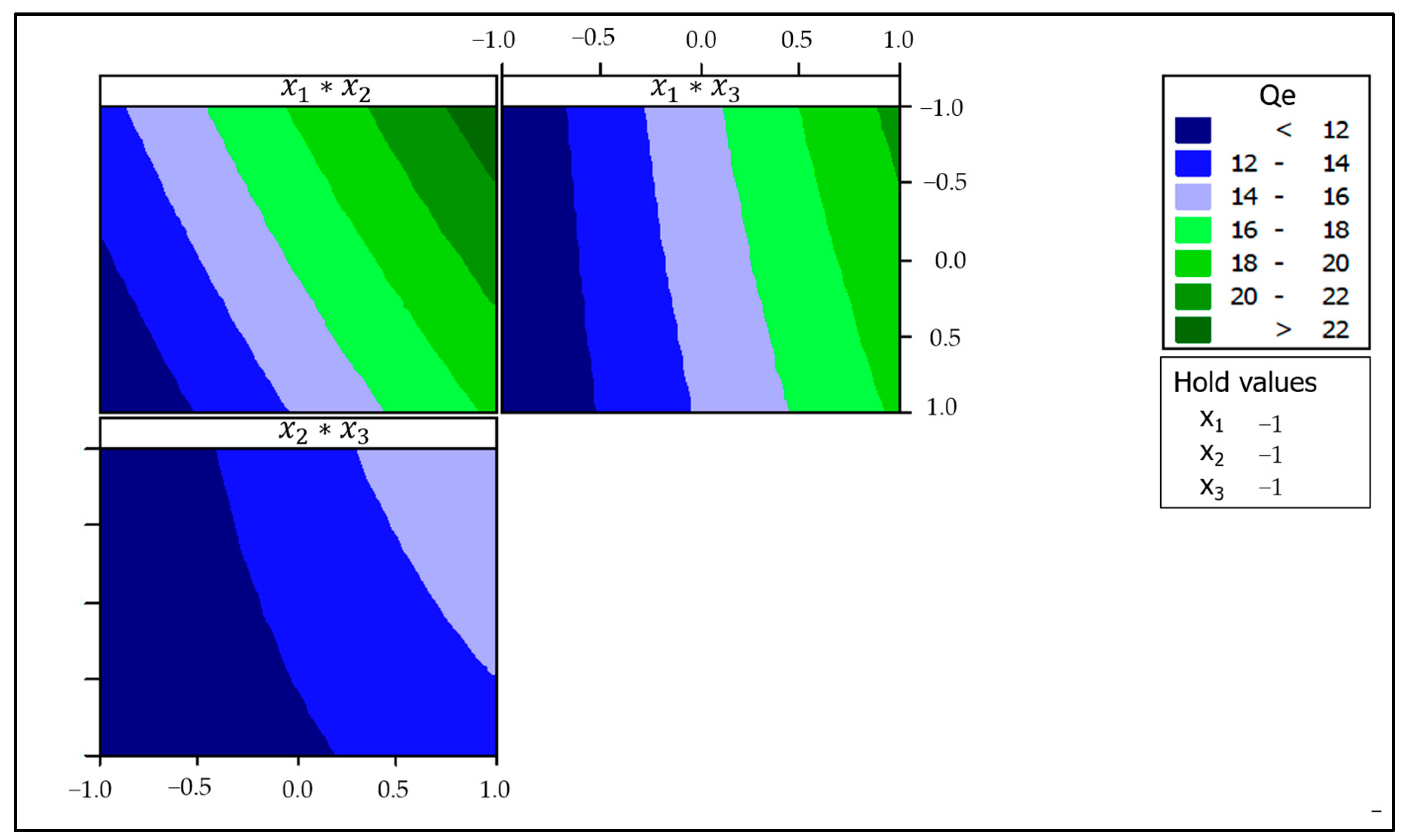
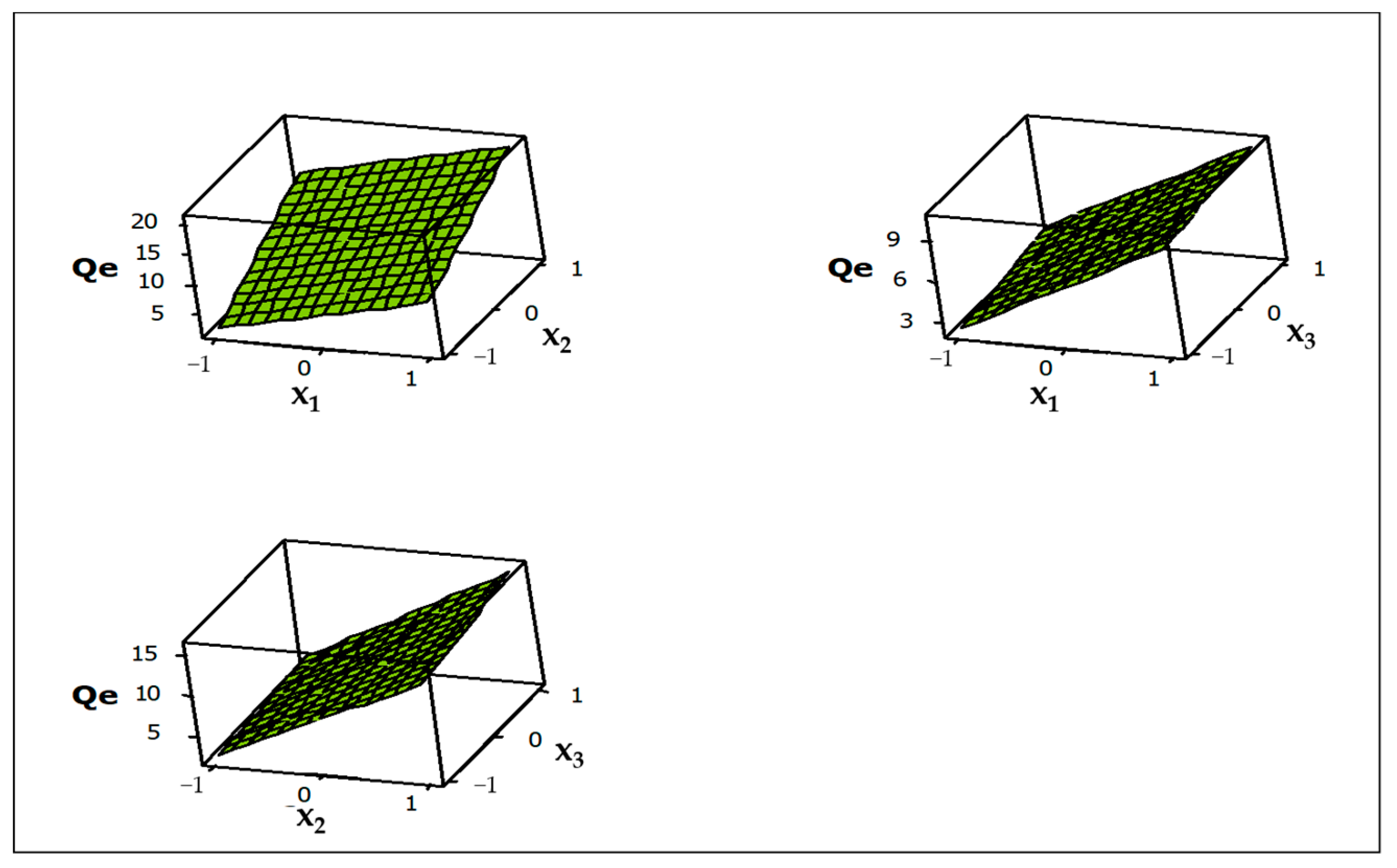
3.2.4. Contour and Surface Plots
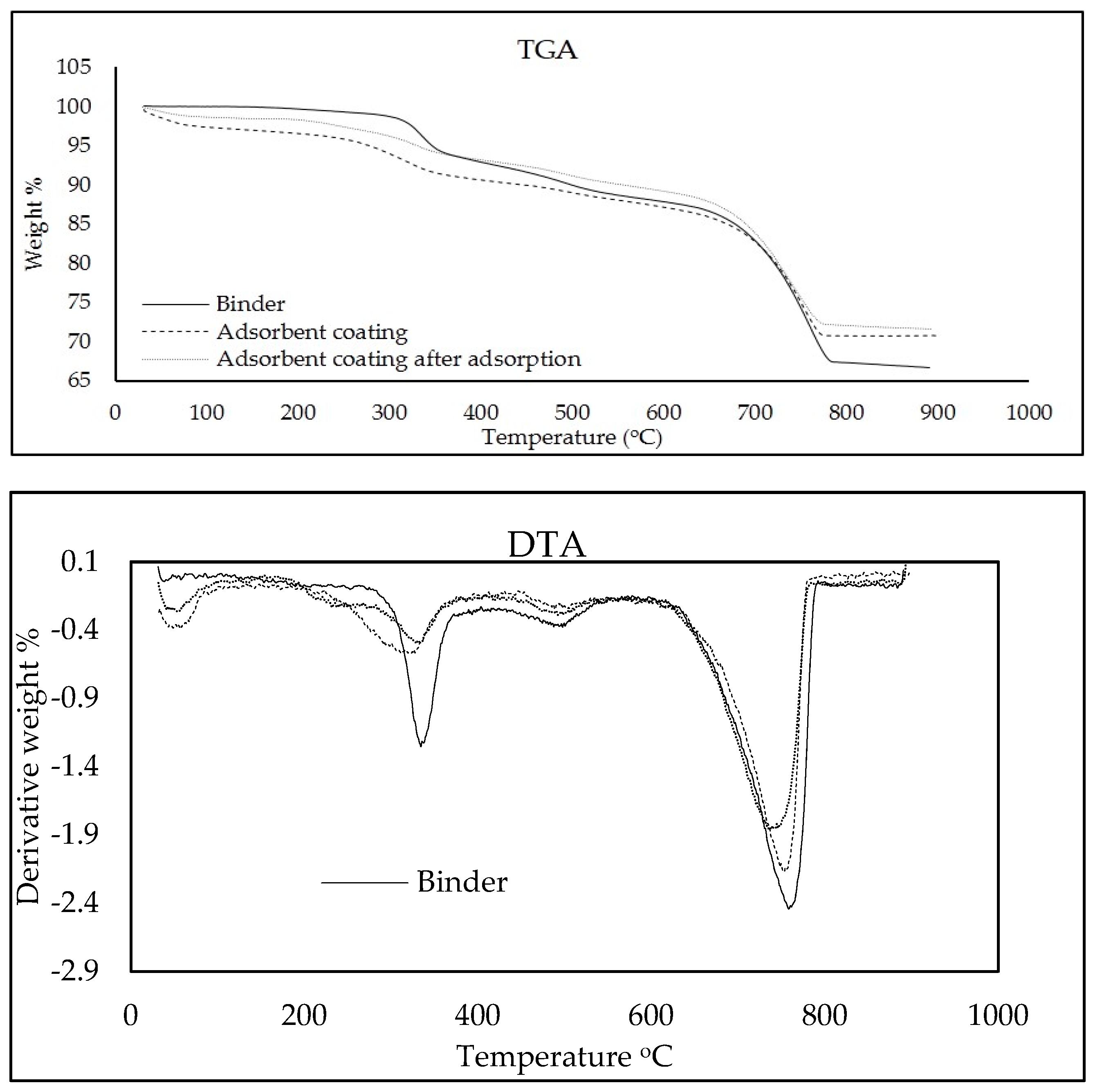
3.3. Characterization
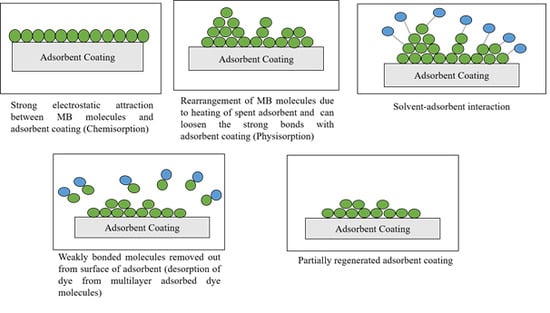
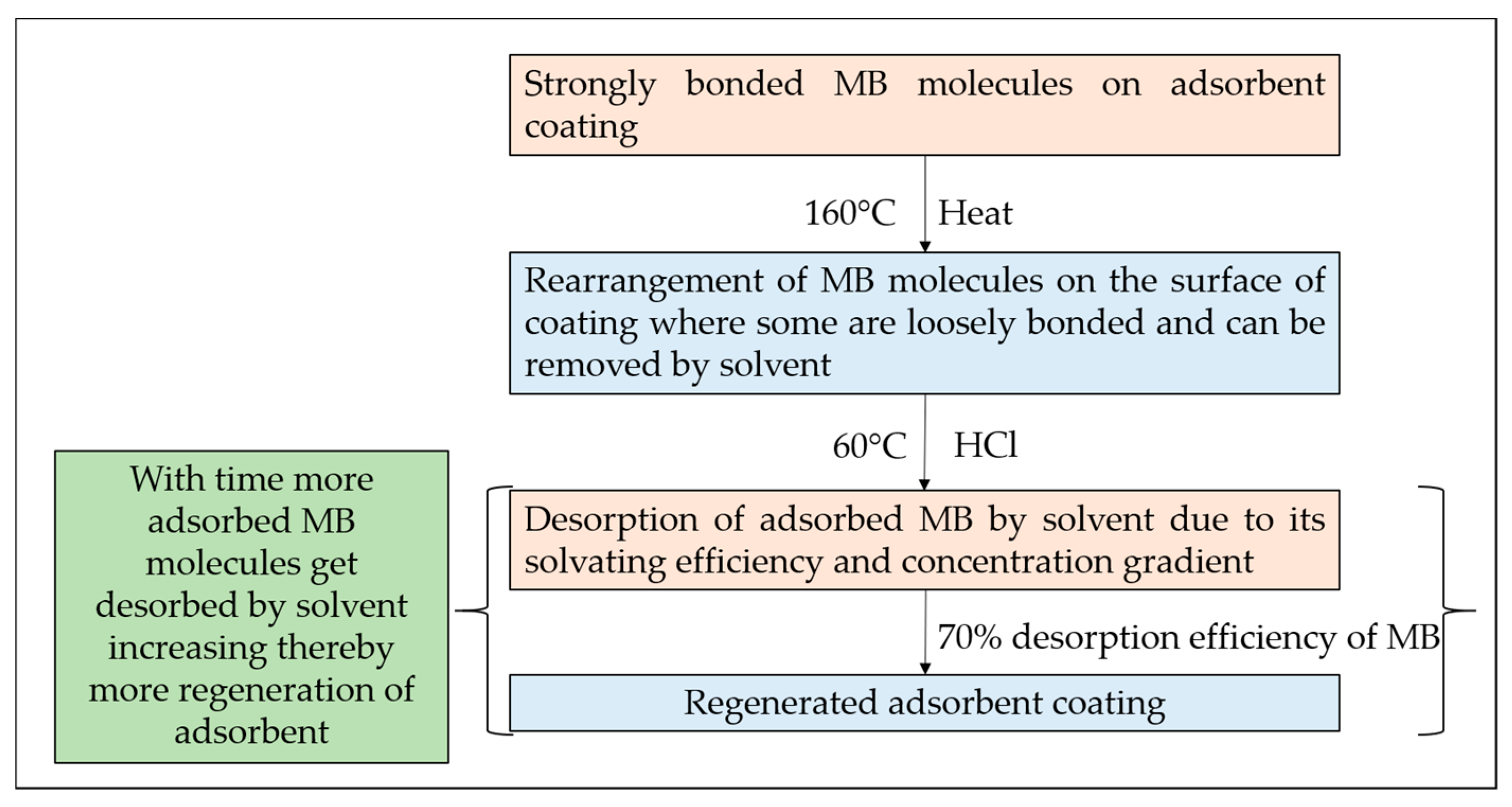
3.4. Desorption Mechanism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mondal, P.; Baksi, S.; Bose, D. Study of environmental issues in textile industries and recent wastewater treatment technology. World Sci. News 2017, 61, 98–109. [Google Scholar]
- Sandin, G.; Peters, G.M. Environmental impact of textile reuse and recycling—A review. J. Clean. Prod. 2018, 184, 353–365. [Google Scholar] [CrossRef]
- Ahmad, T.; Danish, M.; Rafatullah, M.; Ghazali, A.; Sulaiman, O.; Hashim, R.; Ibrahim, M.N.M. The use of date palm as a potential adsorbent for wastewater treatment: A Review. Environ. Sci. Pollut. R. 2012, 19, 1464–1484. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Rafatullah, M.; Ghazali, A.; Sulaiman, O.; Hashim, R. Oil palm biomass based adsorbents for the removal of water pollutants—A review. J. Environ. Sci. Health C 2011, 29, 177–222. [Google Scholar] [CrossRef] [PubMed]
- Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A. Adsorption of methylene blue onto low cost adsorbents: A review. J. Hazard. Mater. 2010, 177, 70–80. [Google Scholar] [CrossRef]
- Vakili, M.; Rafatullah, M.; Salamatinia, B.; Abdullah, A.Z.; Ibrahim, M.H.; Tan, K.B.; Gholami, Z.; Amouzger, P. Application of chitosan and its derivatives as adsorbents for dye removal from water and wastewater: A review. Carbohydr. Polym. 2014, 113, 115–130. [Google Scholar] [CrossRef]
- Ahmad, A.; Mohd-Setapar, S.H.; Chuong, C.S.; Khatoon, A.; Wani, W.A.; Kumar, R.; Rafatullah, M. Recent advances in new generation dye removal technologies: Novel search for approaches to reprocess wastewater. RSC Adv. 2015, 5, 30801–30818. [Google Scholar] [CrossRef]
- Rabia, B.S.; Bullo, S.; Rehman, F. Greener method for the removal of toxic metal ions from the wastewater by application of agricultural waste as an adsorbent. Water 2019, 10, 1316. [Google Scholar]
- Medina-Ramirez, A.; Gamero-Melo, P.; Ruiz-Camacho, B.; Minchaca-Mojica, J.I.; Romero-Toledo, R.; Gamero-Vega, K.Y. Adsorption of aqueous As (III) in presence of coexisting ions by a green Fe-modified W zeolite. Water 2019, 11, 281. [Google Scholar] [CrossRef]
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A critical review on textile wastewater treatments: Possible approaches. J. Environ. Manag. 2016, 182, 351–366. [Google Scholar] [CrossRef]
- Rafatullah, M.; Ahmad, T.; Ghazali, A.; Sulaiman, O.; Danish, M.; Hashim, R. Oil palm biomass as a precursors of activated carbons—A review. Crit. Rev. Environ. Sci. Technol. 2013, 43, 1117–1161. [Google Scholar] [CrossRef]
- Monvisade, P.; Siriphannon, P. Chitosan intercalated montmorillonite: Preparation, characterization and cationic dye adsorption. Appl. Clay Sci. 2009, 42, 427–431. [Google Scholar] [CrossRef]
- Tahir, H.; Sultan, M.; Qadir, Z. Physiochemical modification and characterization of bentonite clay and its application for the removal of reactive dyes. Int. J. Chem. 2013, 5, 19. [Google Scholar] [CrossRef]
- Akl, M.; Youssef, A.; Al-Awadhi, M. Adsorption of acid dyes onto bentonite and surfactant-modified bentonite. J. Anal. Bioanal. Technol. 2013, 4, 3–7. [Google Scholar]
- Azha, S.; Ahmad, A.; Ismail, S. Thin coated adsorbent layer: Characteristics and performance study. Desalin. Water Treat. 2015, 55, 956–969. [Google Scholar] [CrossRef]
- Dunne, S.R.; McKeon, M.J.; Cohen, A.P.; Behan, A.S. Method of Coating Aluminum Substrates with Solid Adsorbent. U.S. Patent US5325916A, 5 July 1994. [Google Scholar]
- Omorogie, M.O.; Babalola, J.O.; Unuabonah, E.I. Regeneration strategies for spent solid matrices used in adsorption of organic pollutants from surface water: A critical review. Desalin. Water Treat. 2016, 57, 518–544. [Google Scholar] [CrossRef]
- Shahadat, M.; Isamil, S. Regeneration performance of clay-based adsorbents for the removal of industrial dyes: A review. RSC Adv. 2018, 8, 24571–24587. [Google Scholar]
- Bingol, D.; Tekin, N.; Alkan, M. Brilliant Yellow dye adsorption onto sepiolite using a full factorial design. Appl. Clay Sci. 2010, 50, 315–321. [Google Scholar] [CrossRef]
- Biswas, S.; Bal, M.; Behera, S.K.; Sen, T.K.; Meikap, B.C. Process optimization study of Zn2+ adsorption on biochar-alginate composite adsorbent by response surface methodology (RSM). Water 2019, 11, 325. [Google Scholar] [CrossRef]
- Alam, M.Z.; Muyibi, S.A.; Toramae, J. Statistical optimization of adsorption processes for removal of 2,4-dichlorophenol by activated carbon derived from oil palm empty fruit bunches. J. Environ. Sci. 2007, 19, 674–677. [Google Scholar] [CrossRef]
- Greluk, M.; Hubicki, Z. Efficient removal of Acid Orange 7 dye from water using the strongly basic anion exchange resin Amberlite IRA-958. Desalination 2011, 278, 219–226. [Google Scholar] [CrossRef]
- Daneshvar, E.; Vazirzadeh, A.; Niazi, A.; Kousha, M.; Naushad, M.; Bhatnagar, A. Desorption of Methylene blue dye from brown macroalga: Effects of operating parameters, isotherm study and kinetic modeling. J. Clean. Prod. 2017, 152, 443–453. [Google Scholar] [CrossRef]
- Chou, K.; Norli, I.; Anees, A. Evaluation of the effect of temperature, NaOH concentration and time on solubilization of palm oil mill effluent (POME) using response surface methodology (RSM). Bioresour. Technol. 2010, 101, 8616–8622. [Google Scholar] [CrossRef] [PubMed]
- Ponnusami, V.; Krithika, V.; Madhuram, R.; Srivastava, S.N. Biosorption of reactive dye using acid-treated rice husk: Factorial design analysis. J. Hazard. Mater. 2007, 142, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Mathialagan, T.; Viraraghavan, T. Biosorption of pentachlorophenol by fungal biomass from aqueous solutions: A factorial design analysis. Environ. Technol. 2005, 6, 571–579. [Google Scholar] [CrossRef] [PubMed]


| Factors | Low Level (–1) | High Level (+1) |
|---|---|---|
| (x1) Temperature (°C) | 30 | 60 |
| (x2) Concentration (mg/L) | 50 | 100 |
| (x3) Contact time (min) | 15 | 150 |
| Heating Temperature (°C) | Desorption Efficiency (%) |
|---|---|
| 150 | 8.03 ± 1.07 |
| 160 | 8.14 ± 0.5 |
| 170 | 8.39 ± 0.67 |
| 190 | 8.99 ± 1.12 |
| Experiment | Coded Values | Real Values | Desorption Efficiency (mg/g) | Predicted Values | ||||
|---|---|---|---|---|---|---|---|---|
| x1 | x2 | x3 | x1 | x2 | x3 | |||
| 1 | 1 | −1 | −1 | 60 | 30 | 15 | 10.293 | 10.016 |
| 2 | −1 | −1 | −1 | 30 | 30 | 15 | 2.302 | 2.606 |
| 3 | 1 | −1 | 1 | 60 | 30 | 150 | 10.493 | 10.610 |
| 4 | −1 | −1 | 1 | 30 | 30 | 150 | 3.293 | 3.200 |
| 5 | 1 | 1 | −1 | 60 | 100 | 15 | 23.169 | 21.923 |
| 6 | 1 | 1 | 1 | 60 | 100 | 150 | 20.560 | 21.950 |
| 7 | −1 | 1 | 1 | 30 | 100 | 150 | 15.965 | 14.540 |
| Predictor | Coef | SE Coef | T | p |
|---|---|---|---|---|
| Constant | −10.031 | 2.499 | −4.10 | 0.026 |
| x1 | 0.247 | 0.0364 | 6.78 | 0.007 |
| x2 | 0.171 | 0.0246 | 6.96 | 0.006 |
| x3 | 0.0062 | 0.017 | 0.36 | 0.007 |
| x2 − x3 | −0.000060 | 0.00023 | −0.26 | 0.011 |
| Source | Seq SS | DF | Adj SS | Adj MS | F Value | p Value |
|---|---|---|---|---|---|---|
| Main effects | 381.740 | 3 | 381.740 | 127.247 | 53.29 | 0.004 |
| Two-way interactions | 0.163 | 1 | 0.163 | 0.163 | 0.07 | 0.008 |
| Regression | 381.903 | 4 | 381.903 | 95.476 | 39.99 | 0.006 |
| Residual error | 7.163 | 3 | 7.163 | 2.388 | ||
| Total | 389.066 | 11 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Momina; Rafatullah, M.; Ismail, S.; Ahmad, A. Optimization Study for the Desorption of Methylene Blue Dye from Clay Based Adsorbent Coating. Water 2019, 11, 1304. https://doi.org/10.3390/w11061304
Momina, Rafatullah M, Ismail S, Ahmad A. Optimization Study for the Desorption of Methylene Blue Dye from Clay Based Adsorbent Coating. Water. 2019; 11(6):1304. https://doi.org/10.3390/w11061304
Chicago/Turabian StyleMomina, Mohd Rafatullah, Suzylawati Ismail, and Anees Ahmad. 2019. "Optimization Study for the Desorption of Methylene Blue Dye from Clay Based Adsorbent Coating" Water 11, no. 6: 1304. https://doi.org/10.3390/w11061304
APA StyleMomina, Rafatullah, M., Ismail, S., & Ahmad, A. (2019). Optimization Study for the Desorption of Methylene Blue Dye from Clay Based Adsorbent Coating. Water, 11(6), 1304. https://doi.org/10.3390/w11061304