Assessment of Potential Toxic Metals in a Ramsar Wetland, Central Mexico and its Self-Depuration through Eichhornia crassipes
Abstract
1. Introduction
2. Materials and Methods
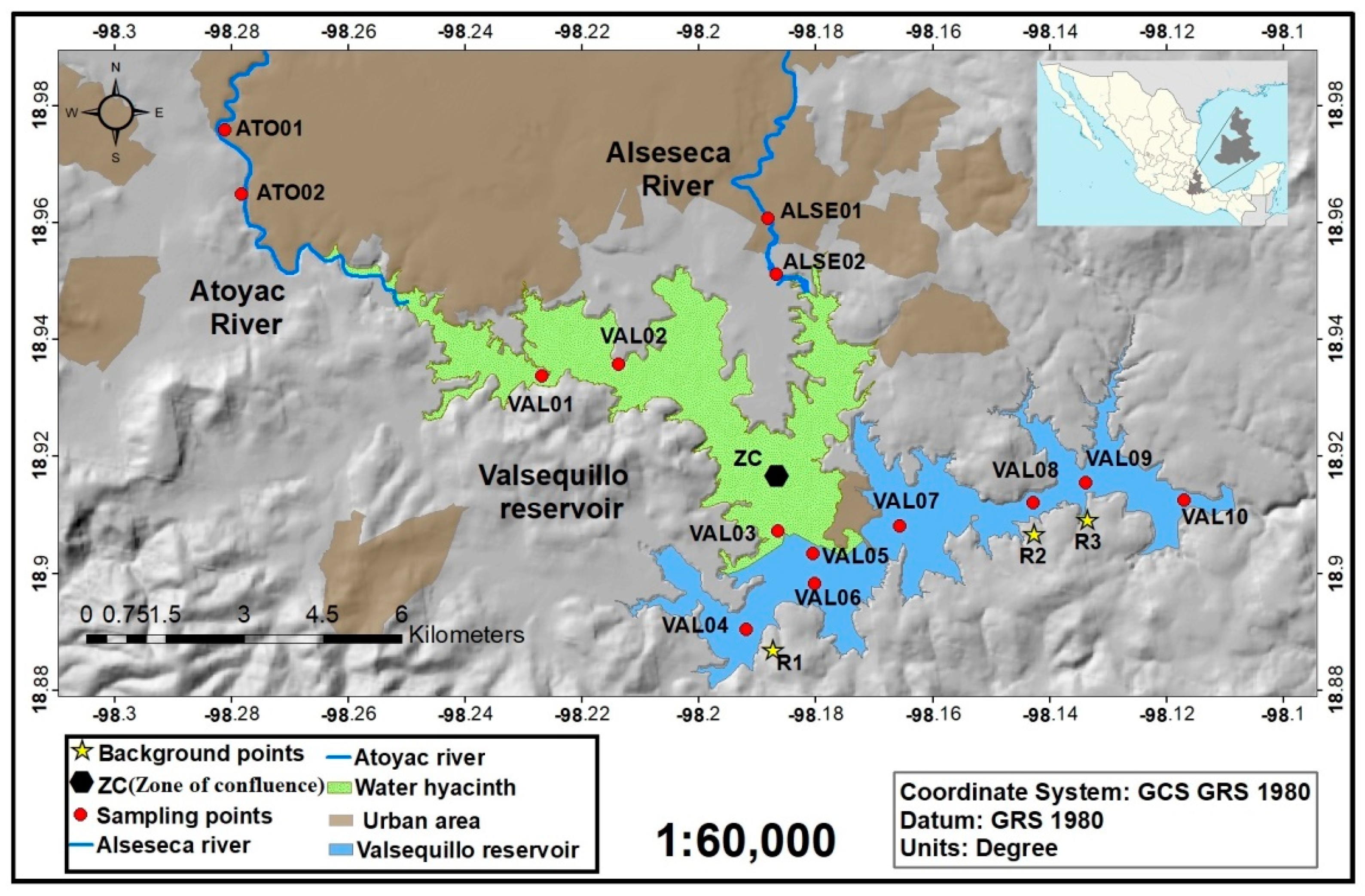
2.1. Study Area
2.2. Geological Settings
2.3. Water
2.3.1. Collection of Water Samples
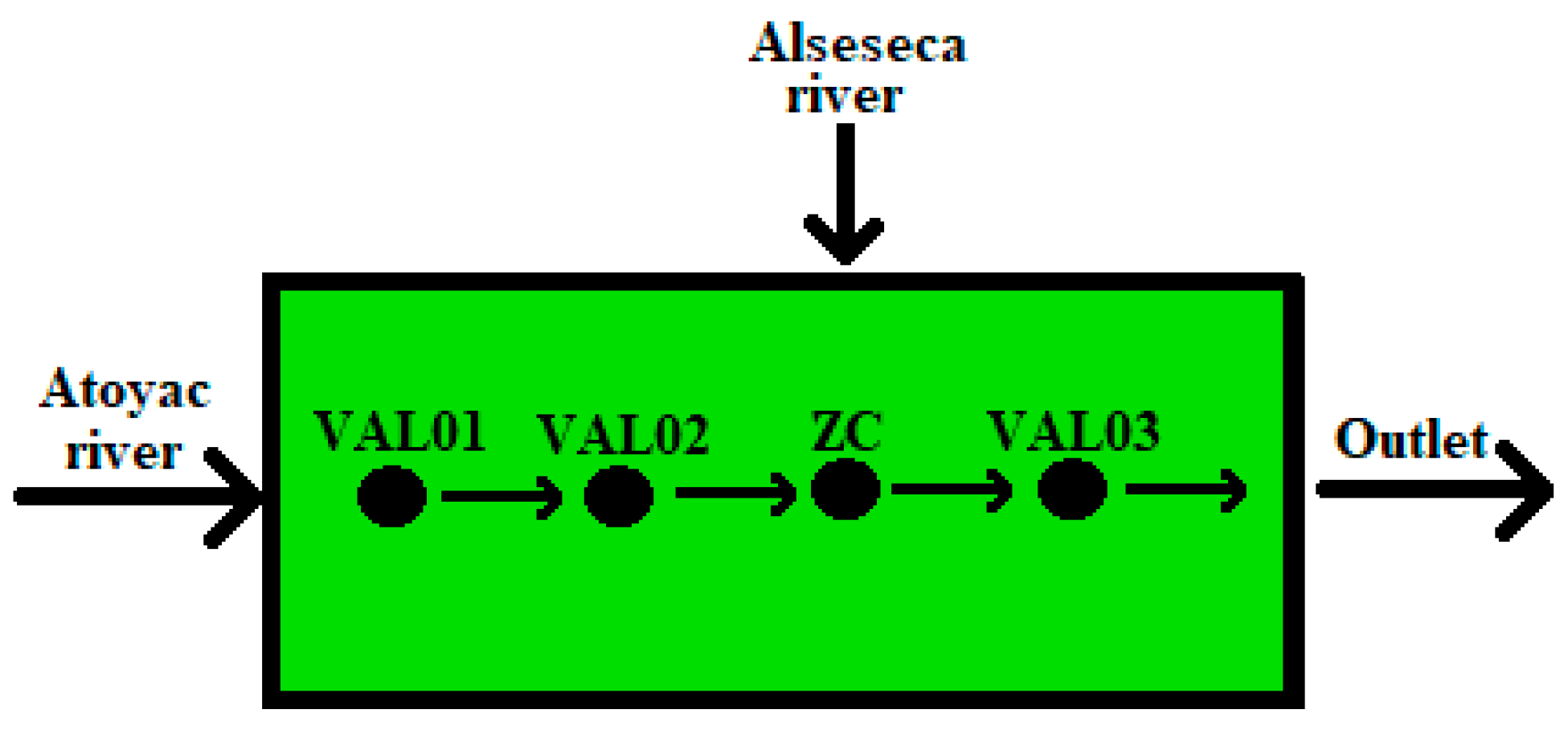
2.3.2. Efficiency of the Removal of Potential Toxic Metals from Water
- RE = Removal efficiency
- CI = Potential toxic metal concentration at the inlet
- CSP = Potential toxic metal concentration at each sampling point
- ZC = Zone of confluence
- C1 = Potential toxic metal concentration in Atoyac River
- C2 = Potential toxic metal concentration in Alseseca River
- WF1 = Water flow in Atoyac River
- WF2 = Water flow in Alseseca River
- CV = Coefficient of variation
- S = Standard deviation of potential toxic metal content
- = Arithmetic mean of potential toxic metal (VAL04–VAL10)
2.4. Water Hyacinth Plants (E. Crassipes)
2.4.1. Collection of Water Hyacinth Plants Samples
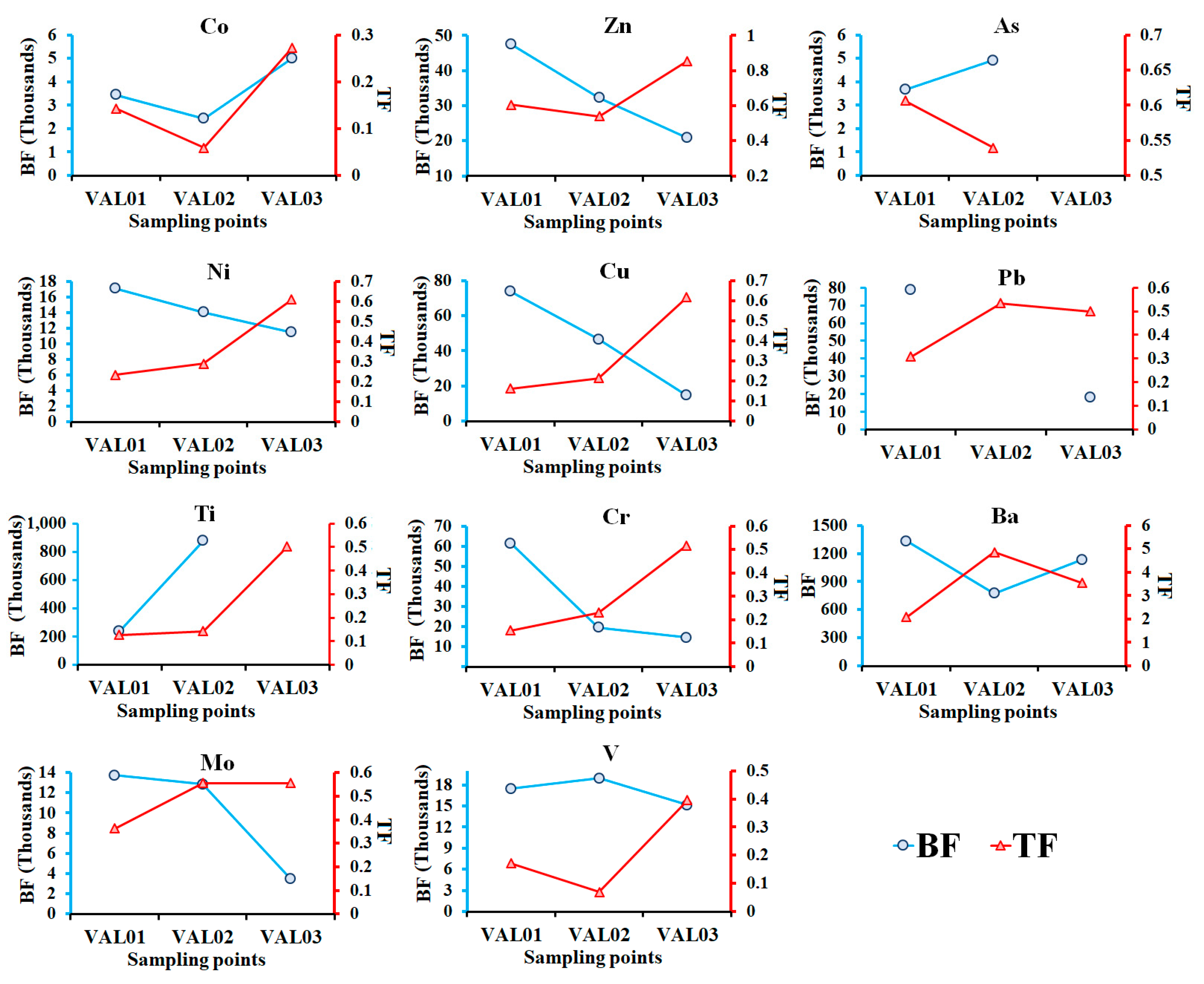
2.4.2. Determination of Bioaccumulation and Translocation Factor
- CW = Concentration levels of each potential toxic metal in water
- CS = Concentration levels of each potential toxic metal in the submerged part of the Water hyacinth
- CA = Concentration levels of each PTM in the aerial part of the Water hyacinth
2.5. Sediments
2.5.1. Collection of Sediments Samples
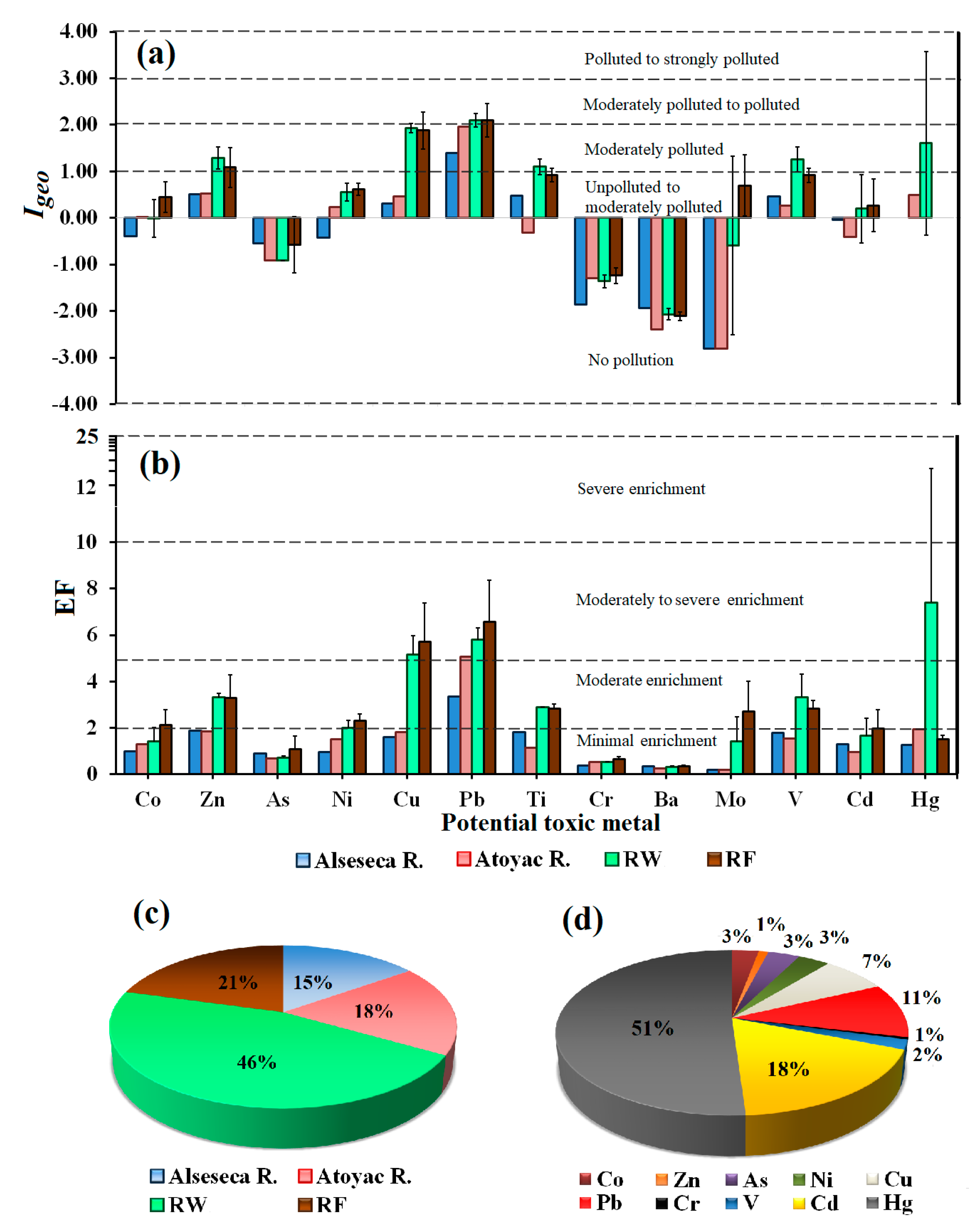
2.5.2. Enrichment Factor (EF)
- CM = is the concentration of a PTM in the sample
- CAl = is the concentration of Aluminum in the sample
- CM ref = is the concentration of a PTM in the natural background value
- CAl ref = is the concentration of a Aluminum in the natural background value
2.5.3. Geoaccumulation Index (Igeo)
- Cn = Is the PTM concentration in sediment sample
- Bn = Is the natural background concentration of PTM n
2.5.4. Potential Ecological Risk Index (PERI)
- E = Monomial potential ecological risk factor
- T = Toxic-response factors or toxicity coefficients (Zn = 1, Cr = V = 2, Co = Ni = Cu = Pb = 5, As = 10, Cd = 30, Hg = 40)
- C = Contamination factor
- Ca = Concentration of PTM in the sediment sample
- Cb = Background value of the PTM in sediments (Metal concentrations in sediment samples R1, R2 and R3).
2.6. Statistical Analyses for Metal Content in Three Matrices Studied
3. Results and Discussion
3.1. Physicochemical Parameters and Potential Toxic Metal (PTMs) in Water
3.2. Potential Toxic Metal in Water Hyacinth Plants
3.3. Potential Toxic Metal in Sediments
4. Conclusions
Future Research
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chatterjee, S.; Datta, S.; Mallick, P.H.; Mitra, A.; Vijay Veer, V.; Mukhopadhyay Kumar, S. Use of wetland plants in bioaccumulation of heavy metals. In Plant-Based Remediation Processes; Kumar, D., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; p. 304. ISBN 978-3-642-35563-9. [Google Scholar]
- Kamari, A.; Yusof, N.; Abdullah, H.; Haraguchi, A.; Abas, M.F. Assessment of heavy metals in water, sediment, anabas testudineus and eichhornia crassipes in a former mining pond in Perak, Malaysia. Chem. Ecol. 2017, 33, 637–651. [Google Scholar] [CrossRef]
- Bradl, H.; Kim, C.; Kramar, U.; Stuben, D. Interaction of heavy metals. In Heavy Metals in the Environment: Origin, Interaction and Remediation; Bradl, H.B., Ed.; Elsevier Academic Press: London, UK, 2005; 269p, ISBN 0-12-088381-3. [Google Scholar]
- Romanova, T.E.; Shuvaeva, O.V.; Belchenko, L.A. Phytoextraction of trace elements by water hyacinth in contaminated area of gold mine tailing. Int. J. Phytoremediation 2015, 1549–7879. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Goyer, R.A.; Waalkes, M.P. Toxic effects of metals. In Casarett and Doull’s—Toxicology: The Basic Science of Poisons; Klaassen, C.D., Ed.; McGraw-Hill Medical: New York, NY, USA, 2008; 1309p, ISBN 0071769250. [Google Scholar]
- Fabbricino, M.; Ferraro, A.; Luongo, V.; Pontoni, L.; Race, M. Soil washing optimization, recycling of the solution, and ecotoxicity assessment for the remediation of Pb-contaminated sites using EDDS. Sustainability 2018, 10, 636. [Google Scholar] [CrossRef]
- Venkatramanan, S.; Chung, S.Y.; Ramkumar, T.; Selvam, S. Ecological risk assessment of selected heavy metals in the surface sediments of three estuaries in the southeastern coast of India. Environ. Earth Sci. 2018, 77, 1–13. [Google Scholar] [CrossRef]
- Vu, C.T.; Lin, C.; Shern, C.C.; Yeh, G.; Le, V.G.; Tran, H.T. Contamination, ecological risk and source apportionment of heavy metals in sediments and water of a contaminated river in Taiwan. Ecol. Indic. 2017, 82, 32–42. [Google Scholar] [CrossRef]
- Gurung, B.; Race, M.; Fabbricino, M.; Komínková, D.; Libralato, G.; Siciliano, A.; Guida, M. Assessment of metal pollution in the Lambro Creek (Italy). Ecotoxicol. Environ. Saf. 2017, 148, 754–762. [Google Scholar] [CrossRef]
- Shruti, V.C.; Rodriguez-Espinosa, P.F.; Escobedo-Urías, D.C.; Martinez-Tavera, E.; Jonathan, M.P. Environmental consequences of metal concentrations in stream sediments of Atoyac river basin, central Mexico: Natural and industrial influences. Int. J. Environ. Chem. Ecol. Geol. Geophys. Eng. 2017, 11, 44–51. [Google Scholar]
- Chai, L.; Li, H.; Yang, Z.; Min, X.; Liao, Q.; Liu, Y.; Men, S.; Yan, Y.; Xu, J. Heavy metals and metalloids in the surface sediments of the Xiangjiang River, Hunan, China: Distribution, contamination, and ecological risk assessment. Environ. Sci. Pollut. Res. 2017, 24, 874–885. [Google Scholar] [CrossRef]
- Hakanson, L. An ecological risk index for aquatic pollution control a sedimentological approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Müller, G. Index of geoaccumulation in sediments of the Rhine River. Geojournal 1969, 2, 108–118. [Google Scholar]
- Rana, V.; Maiti, S.K.; Jagadevan, S. Ecological risk assessment of metals contamination in the sediments of natural urban wetlands in dry tropical climate. Bull. Environ. Contam. Toxicol. 2016, 97, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Wojciechowska, E.; Nawrot, N.; Walkusz-Miotk, J.; Matej-Łukowicz, K.; Pazdro, K. Heavy metals in sediments of urban streams: Contamination and health risk assessment of influencing factors. Sustainability 2019, 11, 563. [Google Scholar] [CrossRef]
- Agunbiade, F.O.; Olu-Owolabi, B.I.; Adebowale, K.O. Phytoremediation potential of Eichornia crassipes in metal-contaminated coastal water. Bioresour. Technol. 2009, 100, 4521–4526. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Maiti, A. The efficiency of Eichhornia crassipes in the removal of organic and inorganic pollutants from wastewater: A review. Environ. Sci. Pollut. Res. 2017, 24, 7921–7937. [Google Scholar] [CrossRef] [PubMed]
- Odjegba, V.J.; Fasidi, I.O. Phytoremediation of heavy metals by Eichhornia crassipes. Environmentalist 2007, 27, 349–355. [Google Scholar] [CrossRef]
- Vymazal, J. Constructed wetlands for treatment of industrial wastewaters: A review. Ecol. Eng. 2014, 73, 724–751. [Google Scholar] [CrossRef]
- Khan, S.; Ahmad, I.; Shah, M.T.; Rehman, S.; Khaliq, A. Use of constructed wetland for the removal of heavy metals from industrial wastewater. J. Environ. Manag. 2009, 90, 3451–3457. [Google Scholar] [CrossRef]
- Zhang, D.Q.; Jinadasa, K.B.S.N.; Gersberg, R.M.; Liu, Y.; Tan, S.K.; Ng, W.J. Application of constructed wetlands for wastewater treatment in tropical and subtropical regions (2000–2013). J. Environ. Sci. 2015, 30, 30–46. [Google Scholar] [CrossRef]
- Chunkao, K.; Nimpee, C.; Duangmal, K. The King’s initiatives using water hyacinth to remove heavy metals and plant nutrients from wastewater through Bueng Makkasan in Bangkok, Thailand. Ecol. Eng. 2012, 39, 40–52. [Google Scholar] [CrossRef]
- Li, Z.; Xiao, H.; Cheng, S.; Zhang, L.; Xie, X.; Wu, Z. A comparison on the phytoremediation ability of triazophos by different macrophytes. J. Environ. Sci. 2014, 26, 315–322. [Google Scholar] [CrossRef]
- Smolyakov, B.S. Uptake of Zn, Cu, Pb, and Cd by water hyacinth in the initial stage of water system remediation. Appl. Geochem. 2012, 27, 1214–1219. [Google Scholar] [CrossRef]
- Melignani, E.; de Cabo, L.I.; Faggi, A.M. Copper uptake by Eichhornia crassipes exposed at high level concentrations. Environ. Sci. Pollut. Res. 2015, 22, 8307–8315. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.R.; Tripathi, B.D. Principle and process of biofiltration of Cd, Cr, Co, Ni & Pb from tropical opencast coalmine effluent. Water Air Soil Pollut. 2007, 180, 213–223. [Google Scholar]
- Hadad, H.R.; Maine, M.a.; Mufarrege, M.M.; Del Sastre, M.V.; Di Luca, G.a. Bioaccumulation kinetics and toxic effects of Cr, Ni and Zn on Eichhornia crassipes. J. Hazard. Mater. 2011, 190, 1016–1022. [Google Scholar] [CrossRef] [PubMed]
- Newete, S.W.; Erasmus, B.F.N.; Weiersbye, I.M.; Byrne, M.J. Sequestration of precious and pollutant metals in biomass of cultured water hyacinth (Eichhornia crassipes). Environ. Sci. Pollut. Res. 2016, 23, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Espinosa, P.F.; Mendoza-Pérez, J.A.; Tabla-Hernandez, J.; Martínez-Tavera, E.; Monroy-Mendieta, M.M. Biodegradation and kinetics of organic compounds and heavy metals in an artificial wetland system (AWS) by using water hyacinths as a biological filter. Int. J. Phytoremediation 2018, 20, 35–43. [Google Scholar] [CrossRef] [PubMed]
- CONAGUA Banco Nacional de Datos de Aguas Superficiales (BANDAS). Available online: http://www.conagua.gob.mx/CONAGUA07/Contenido/Documentos/Portada BANDAS.htm (accessed on 7 December 2017).
- Martinez-Tavera, E.; Rodriguez-Espinosa, P.F.; Shruti, V.C.; Sujitha, S.B.; Morales-Garcia, S.S.; Muñoz-Sevilla, N.P. Monitoring the seasonal dynamics of physicochemical parameters from Atoyac River basin (Puebla), Central Mexico: Multivariate approach. Environ. Earth Sci. 2017, 76, 95. [Google Scholar] [CrossRef]
- Rodriguez-Espinosa, P.F.; Morales-García, S.S.; Jonathan, M.P.; Navarrete-Lopez, M.; Bernal-Campos, A.A.; Gonzales-Cesar, A.; Muñoz-Sevilla, N.P. Servicio Ambiental de la Presa Valsequillo para las cuencas de los Ríos del Atoyac-Zahuapan y Alseseca, Puebla, Tlaxcala, México. Inst Nac Ecol Cambio Clim (INECC). Secr Medio Ambient Recur Na 2011, 1–10. Available online: http://viveatoyac.org.mx/images/Biblioteca%20tem%C3%A1tica/Biblioteca%20sobre%20el%20agua/3g010%20Servicio%20ambiental%20de%20la%20presa%20Valsequillo%20para%20las%20cuencas%20del%20Atoyac%20y%20Alseseca.pdf (accessed on 7 May 2019).
- Traversoni, L.; Vélez, H.; Ruiz Martínez, R.; Carreón, E. Manifestación de impacto ambiental: Remoción de lirio acuático en el embalse de Valsequillo. In Semarnat; 2009. Available online: http://sinat.semarnat.gob.mx/dgiraDocs/documentos/pue/estudios/2009/21PU2009HD056.pdf (accessed on 23 April 2019).
- Flores-Márquez, E.L.; Jiménez-Suárez, G.; Martínez-Serrano, R.G.; Chávez, R.E.; Pérez, D.S. Study of geothermal water intrusion due to groundwater exploitation in the Puebla Valley aquifer system, Mexico. Hydrogeol. J. 2006, 14, 1216–1230. [Google Scholar] [CrossRef]
- Stevens, R.E.; Metcalfe, S.E.; Leng, M.J.; Lamb, A.L.; Sloane, H.J.; Naranjo, E.; González, S. Reconstruction of late Pleistocene climate in the Valsequillo Basin (Central Mexico) through isotopic analysis of terrestrial and freshwater snails. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2012, 319, 16–27. [Google Scholar] [CrossRef]
- Urrutia-Fucugauchi, J.; Trigo-Huesca, A.; Téllez-García, E.; Pérez-Cruz, L.; Méndez-Rivero, F. Volcano-sedimentary stratigraphy in the valsequillo basin, central Mexico inferred from electrical resistivity soundings. Geofis. Int. 2014, 53, 87–94. [Google Scholar] [CrossRef]
- Wang, Q.; Cui, Y.; Dong, Y. Phytoremediation of polluted waters: Potentials and prospects of wetland plants. Acta Biotechnol. 2002, 22, 199–208. [Google Scholar] [CrossRef]
- Branzini, A.; Zubillaga, M.S. Phytostabilization as soil remediation strategy. In Plant-Based Remediation Processes; Dharmendra, K.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; 304p, ISBN 978-3-642-35563-9. [Google Scholar]
- Meitei, M.D.; Prasad, M.N.V. Bioaccumulation of nutrients and metals in sediment, water, and phoomdi from Loktak Lake (Ramsar site), northeast India: Phytoremediation options and risk assessment. Environ. Monit. Assess. 2016, 188, 329. [Google Scholar] [CrossRef] [PubMed]
- Mercado-Borrayo, B.M.; Silke Cram, H.; Rosas Pérez, I.; Hernandez Quiroz, M.; Ponce de Leon Hill, C. Organophosphorus and organochlorine pesticides bioaccumulation by Eichhornia crassipes in irrigation canals in an urban agricultural system. Int. J. Phytoremediation 2015, 17, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.J.M. Accumulators and excluders–Strategies in the response of plants to heavy metals. J. Plant Nutr. 1981, 3, 643–654. [Google Scholar] [CrossRef]
- Reeves, R.D.; Van Der Ent, A.; Baker, A.J.M. Global distribution and ecology of hyperaccumulator plants. In Agromining: Farming for Metals; Van der Ent, A., Echevarria, G., Baker, A.J., Morel, J.L., Eds.; Springer: Cham, Switzerland, 2018; pp. 75–92. ISBN 978-3-319-61898-2. [Google Scholar]
- Hseu, Z.Y.; Chen, Z.S.; Tsai, C.C.; Tsui, C.C.; Cheng, S.F.; Liu, C.L.; Lin, H.T. Digestion methods for total heavy metals in sediments and soils. Water Air Soil Pollut. 2002, 141, 189–205. [Google Scholar] [CrossRef]
- Jahan, S.; Strezov, V. Comparison of pollution indices for the assessment of heavy metals in the sediments of seaports of NSW, Australia. Mar. Pollut. Bull. 2018, 128, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, J.; Liu, R.; Yu, W.; Shen, Z. Spatial variation, environmental risk and biological hazard assessment of heavy metals in surface sediments of the Yangtze River estuary. Mar. Pollut. Bull. 2015, 93, 250–258. [Google Scholar] [CrossRef]
- Mohiuddin, K.M.; Otomo, K.; Ogawa, Y.; Shikazono, N. Seasonal and spatial distribution of trace elements in the water and sediments of the Tsurumi River in Japan. Environ. Monit. Assess. 2012, 184, 265–279. [Google Scholar] [CrossRef]
- Kükrer, S.; Seker, S.; Abaci, Z.T.; Kutlu, B. Ecological risk assessment of heavy metals in surface sediments of northern littoral zone of Lake Çildir, Ardahan, Turkey. Environ. Monit. Assess. 2014, 186, 3847–3857. [Google Scholar] [CrossRef]
- Mamat, Z.; Haximu, S.; Zhang, Z.Y.; Aji, R. An ecological risk assessment of heavy metal contamination in the surface sediments of Bosten Lake, northwest China. Environ. Sci. Pollut. Res. 2016, 23, 7255–7265. [Google Scholar] [CrossRef]
- Kusin, F.M.; Rahman, M.S.A.; Madzin, Z.; Jusop, S.; Mohamat-Yusuff, F.; Ariffin, M.; Mohd Syakirin Md, Z. The occurrence and potential ecological risk assessment of bauxite mine-impacted water and sediments in Kuantan, Pahang, Malaysia. Environ. Sci. Pollut. Res. 2017, 24, 1306–1321. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.E.; Strezov, V.; Davies, P.J.; Wright, I. River sediment quality assessment using sediment quality indices for the Sydney basin, Australia affected by coal and coal seam gas mining. Sci. Total Environ. 2018, 616, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Wang, A.; Kong, L.; He, M. Calculation and application of Sb toxicity coefficient for potential ecological risk assessment. Sci. Total Environ. 2018, 610, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Gárfias, J.; Arroyo, N.; Aravena, R. Hydrochemistry and origins of mineralized waters in the Puebla aquifer system, Mexico. Environ. Earth Sci. 2009, 59, 1789–1805. [Google Scholar] [CrossRef]
- Jackson, L.J. Paradigms of metal accumulation in rooted aquatic vascular plants. Sci. Total Environ. 1998, 7, 223–231. [Google Scholar] [CrossRef]
- Pinto, A.P.; de Varennes, A.; Lopes, E.M.; Teixeira, D.M. Biological approaches for remediation of metal contaminated sites. In Phytoremediation Management of Environmental Contaminants; Ansari, A.A., Gill, S.S., Gill, R., Lanza, G.R., Newman, L., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 3, p. 582. ISBN 978-3-319-40146-1. [Google Scholar]
- Ali, H.; Khan, E.; Sajad, M.A. Phytoremediation of heavy metals—Concepts and applications. Chemosphere 2013, 91, 869–881. [Google Scholar] [CrossRef] [PubMed]
- Dantas-Santos, N.; Gomes, D.L.; Costa, L.S.; Cordeiro, S.L.; Costa, M.S.S.P.; Trindade, E.S.; Franco, C.R.C.; Scortecci, K.C.; Leite, E.L.; Rocha, H.A.O. Freshwater plants synthesize sulfated polysaccharides: Heterogalactans from water hyacinth (Eicchornia crassipes). Int. J. Mol. Sci. 2012, 13, 961–976. [Google Scholar] [CrossRef] [PubMed]
- Tejeda, S.; Zarazúa, G.; Ávila-Pérez, P.; Carapia-Morales, L.; Martínez, T. Total reflection X-ray fluorescence spectrometric determination of elements in water hyacinth from the Lerma River. Spectrochim. Acta Part B 2010, 65, 483–488. [Google Scholar] [CrossRef]
- Upadhyay, A.R.; Mishra, V.K.; Pandey, S.K.; Tripathi, B.D. Biofiltration of secondary treated municipal wastewater in a tropical city. Ecol. Eng. 2007, 30, 9–15. [Google Scholar] [CrossRef]
- Bonanno, G.; Borg, J.A.; Di Martino, V. Levels of heavy metals in wetland and marine vascular plants and their biomonitoring potential: A comparative assessment. Sci. Total Environ. 2017, 576, 796–806. [Google Scholar] [CrossRef]
- Aurangzed, N.; Nisa, S.; Bibi, Y.; Javed, F.; Hussain, F. Phytoremediation potential of aquatic herbs from steel foundry effluent. Braz. J. Chem. Eng. 2014, 31, 881–886. [Google Scholar] [CrossRef]
- Larocque, A.C.L.; Stimac, J.A.; Siebe, C.; Greengrass, K.; Chapman, R.; Mejia, S.R. Deposition of a high-sulfidation Au assemblage from a magmatic volatile phase, Volcán Popocatépetl, Mexico. J. Volcanol. Geotherm. Res. 2008, 170, 51–60. [Google Scholar] [CrossRef]
- Deng, H.; Zhang, J.; Chen, S.; Yang, L.; Wang, D.; Yu, S. Metal release/accumulation during the decomposition of potamogeton crispus in a shallow macrophytic lake. J. Environ. Sci. 2016, 42, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Tripathi, B.D. Effect of aeration and mixed culture of Eichhornia crassipes and Salvinia natans on removal of wastewater pollutants. Ecol. Eng. 2014, 62, 48–53. [Google Scholar] [CrossRef]
- Zimmels, Y.; Kirzhner, F.; Kadmon, A. Effect of circulation and aeration on wastewater treatment by floating aquatic plants. Sep. Purif. Technol. 2009, 66, 570–577. [Google Scholar] [CrossRef]
- Chatterjee, S.; Mitra, A.; Datta, S.; Veer, V. Phytoremediation protocols: An overview. In Plant-Based Remediation Processes; Dharmendra, K.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; 304p, ISBN 978-3-642-35563-9. [Google Scholar]
- Pellegrini, E.; Petranich, E.; Acquavita, A.; Canário, J.; Emili, A.; Covelli, S. Mercury uptake by halophytes in response to a long-term contamination in coastal wetland salt marshes (northern Adriatic Sea). Environ. Geochem. Health 2017, 39, 1–17. [Google Scholar] [CrossRef]
- Wu, X.; Yu, J.; Qiu, H.; Fang, H. Pollution and ecological risk assessment of nutrients associated with deposited sediments collected from roof and road surfaces. Environ. Sci. Pollut. Res. 2018, 25, 8943–8950. [Google Scholar] [CrossRef]

| Sampling Point | DO (mg/L) | Temp (°C) | pH | ORP | Turb (NTU) | EC (µS/cm) |
|---|---|---|---|---|---|---|
| ALSE01 | 2.53 | 22.70 | 8.11 | 143.00 | 138.00 | 2147.20 |
| ALE02 | 2.79 | 23.70 | 6.71 | 135.00 | 135.00 | 1756.20 |
| ATO01 | - | 25.60 | 7.42 | 91.70 | 56.60 | 1187.20 |
| ATO02 | 2.62 | 26.70 | 7.82 | −76.30 | 57.70 | 1167.20 |
| VAL01 | 1.17 | 18.72 | 7.85 | 32.10 | 99.80 | 1271.14 |
| VAL02 | 2.33 | 21.62 | 7.51 | 126.00 | 23.60 | 1096.56 |
| VAL03 | 0.31 | 19.63 | 7.64 | 223.80 | 9.74 | 838.00 |
| VAL04 | 0.79 | 19.12 | 6.81 | 170.30 | 9.10 | 827.00 |
| VAL05 | 2.76 | 21.40 | 7.60 | 149.30 | 8.76 | 837.20 |
| VAL06 | 2.78 | 19.90 | 7.17 | 145.10 | 4.66 | 826.00 |
| VAL07 | 2.25 | 19.91 | 6.89 | 122.10 | 2.90 | 821.00 |
| VAL08 | 0.25 | 20.15 | 7.76 | 150.40 | 1.90 | 816.00 |
| VAL09 | 3.52 | 20.69 | 7.96 | 159.00 | 2.10 | 810.00 |
| VAL10 | 2.30 | 21.97 | 7.66 | 181.90 | 1.27 | 813.00 |
| Physicochemical Parameters (Mean Values ± SD) in Water from Different Zones of the Reservoir | ||||||
| Sampling zone | DO (mg/L) | Temp (°C) | pH | ORP (mV) | Turb (NTU) | EC (µS/cm) |
| Inlet 1 | 2.64 ± 0.13 a | 24.67 ± 1.80 a | 7.51 ± 0.60 a | 73.35 ± 102.28 a | 96.83 ± 45.83 a-c | 1564.45 ± 474.86 a-c |
| RW 2 | 1.26 ± 1.01 a | 19.98 ± 1.48 b-a | 7.66 ± 0.17 a | 127.30 ± 95.85 a | 44.38 ± 48.49 a | 1069.57 ± 217.92 a |
| RF 3 | 2.09 ± 1.16 a | 20.44 ± 0.97 c-a | 7.40 ± 0.44 a | 154.01 ± 19.15 a | 4.38 ± 3.28 c-a | 821.46 ± 9.41 c-a |
| Water (µg/L) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Zone | Co | Zn | As | Ni | Cu | Pb | Ti | Cr | Ba | Mo | V | Hg | Cd |
| Alseseca R. | 0.6 | 51.1 | 2.2 | 7.6 | 1.7 | 0.2 | 8.5 | 4.2 | 89.3 | 0.8 | 8.0 | 1.5 | 0.1 |
| Atoyac R. | 0.4 | 4.8 | 2.8 | 3.9 | 1.2 | 0.1 | 0.4 | 2.0 | 63.1 | 0.6 | 4.8 | 0.8 | 0.0 |
| RW | 1.2 ± 0.8 a | 6.9 ± 2.8 a | 3.4 ± 0.8 a | 4.8 ± 0.7 a | 1.7 ± 0.7 a | 0.9 ± 1.1 a | 3.2 ± 3.1 a | 2.0 ± 0.0 a | 78.9 ± 6.9 a | 1.4 ± 1.1 a | 4.7 ± 1.8 a | 0.6 ± 0.2 a | 0.6 ± 1.0 a |
| RF | 0.8 ± 1.3 a | 2.0 ± 0.0 b | 2.7 ± 1.5 a | 2.8 ± 1.2 a | 0.9 ± 0.9 a | 0.4 ± 1.0 a | 0.4 ± 0.2 b | 2.2 ± 0.6 a | 76.8 ± 4.8 a | 0.8 ± 1.3 a | 2.3 ± 1.4 a | 0.6 ± 0.0 a | 0.5 ± 1.3 a |
| Comparison with Permissible Limits of Water Set by Different Local and International Regulation | |||||||||||||
| Regulation | |||||||||||||
| NOM-001-SEMARNAT-1996 | - | 10000 | 200 | 2000 | 4000 | 500 | - | 1000 | - | - | - | 10 | 200 |
| LFDMA | - | 2000 | 100 | 200 | 200 | 500 | - | 100 | 10# | - | - | - | 10 |
| DCRAX | - | 120 | 50 | 600 | 50 | 30 | - | 50 | - | - | - | 1 | 4 |
| (SQuiRTs) * | 3.0 | 120 | 3.1 + | 52 | 9 | 2.5 | - | 11 ˄ | 3.9 | 34 | 19 | 0.77 | 0.25 |
| Sediments (mg/Kg) | |||||||||||||
| Inlet (both Rivers) | 15.7 ± 2.7 a | 88.7 ± 16.3 a | 3.5 ± 1.0 a | 45. 2 ± 13.2 a | 15.5 ± 3.1 a | 18.7 ± 5.9 a | 2675.0 ± 1081.2 a | 61.5 ± 22.4 a | 76.15 ± 24.6 a | 1.0 ± 0.0 a | 63.5 ± 55.2 a | 1.2 ± 0.5 a | 0.35 ± 0.1 a |
| RW | 18.0 ± 5.3 a | 151.0 ± 23.4 a | 3.0 ± 0.0 a | 68.7 ± 9.3 a | 44.3 ± 3.1 b | 24.3 ± 2.5 a | 5033.3 ± 568 b | 68.7 ± 6.7 a | 79.0 ± 6.9 a | 7.0 ± 5.2 a | 118.3 ± 22 b-inlet | 5.7 ± 7.2 a | 0.5 ± 0.3 a |
| RF | 24.7 ± 5.4 b-inlet | 135.3 ± 41.9 a | 4.3 ± 2.2 a | 71.7 ± 6.3 b-inlet | 44.3 ± 12.4 c | 24.9 ± 6.4 a | 4428.6 ± 442 c | 74.7 ± 8.6 a | 76.8 ± 4.8 a | 12.4 ± 5.8 b-inlet | 92.7 ± 9.4 a | 1.0 ± 0.0 a | 0.5 ± 0.2 a |
| Comparison with Permissible Limits of Sediments Set by International Regulation | |||||||||||||
| (SQuiRTs) 1 | 50.0 | 120.0 | 6.0 | 16.0 | 16.0 | 31.0 | - | 26.0 | - | - | - | 0.2 | 0.6 |
| (SQuiRTs) 2 | - | 98.0 | 10.8 | 19.5 | 28.0 | 37.0 | - | 36.3 | - | - | - | - | 0.6 |
| Sampling Point | Removal Efficiency (Percentage) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ba | Ni | Zn | As | V | Co | Cu | Pb | Mo | Cr | Ti | |
| Inlet | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% |
| VAL01 | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% |
| VAL02 | 0.0% | 0.0% | 10.4% | 0.0% | 21.6% | 0.0% | 25.0% | 62.5% | 0.0% | 0.0% | 100.0% |
| VAL03 | 0.0% | 3.1% | 6.0% | 0.0% | 30.0% | 0.0% | 0.0% | 0.0% | 0.0% | 5.1% | 74.7% |
| Outlet | 0.0% | 41.8% | 71.5% | 19.2% | 65.3% | 18.5% | 51.0% | 65.2% | 54.3% | 5.1% | 51.4% |
| PTM | Potential Toxic Metal Concentration (mg/kg d.w.) | Factors | ||
|---|---|---|---|---|
| Submerged Part | Aerial Part | BF 1 | TF 2 | |
| Co | 18.6 ± 8.6 a | 2.6 ± 1.5 b | 3628 | 0.15 |
| Zn | 249.0 ± 191 a | 159.0 ± 159 a | 33569 | 0.66 |
| As | 9.3 ± 5.6 | BDL | 4298 | - |
| Ni | 69.6 ± 24.0 a | 23.3 ± 4.1 a | 14224 | 0.37 |
| Cu | 74.6 ± 63.6 a | 18.0 ± 7.9 a | 45149 | 0.33 |
| Pb | 37.6 ± 23.5 a | 15.0 ± 6.1 a | - | 0.45 |
| Ti | 900.0 ± 624.4 a | 166.7 ± 57.7 a | 555147 | 0.26 |
| Cr | 63.6 ± 51.6 a | 14.3 ± 5.0 a | 31833 | 0.30 |
| Ba | 86.6 ± 29.6 a | 275.6 ± 38.6 b | 1081 | 3.49 |
| Mo | 9.6 ± 1.5 a | 4.6 ± 0.6 b | 10022 | 0.49 |
| V | 80.6 ± 32.9 a | 15.3 ± 9.0 b | 17184 | 0.21 |
| Cd | 0.4 | BDL | - | - |
| Hg | BDL | BDL | - | - |
| Zn | As | Ni | Cu | Pb | Ti | Cr | Ba | Mo | V | |
|---|---|---|---|---|---|---|---|---|---|---|
| Co | - | - | - | - | - | 0.998 ** | - | - | - | - |
| Zn | 1 | - | - | 0.999 ** | - | - | 0.994 * | - | 0.998 ** | 0.994 * |
| As | 1 | - | - | - | - | - | −0.997 ** | - | - | |
| Ni | 1 | - | - | - | - | - | - | - | ||
| Cu | 1 | - | - | 0.998 ** | - | 1.000 ** | 0.997 ** | |||
| Pb | 1 | - | - | - | - | - | ||||
| Ti | 1 | 0.993 * | - | 0.988 * | 0.994 * | |||||
| Cr | 1 | - | 0.999 ** | 1.000 ** | ||||||
| Ba | 1 | - | - | |||||||
| Mo | 1 | 0.999 ** | ||||||||
| V | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tabla-Hernandez, J.; Rodriguez-Espinosa, P.F.; Mendoza-Pérez, J.A.; Sánchez-Ortíz, E.; Martinez-Tavera, E.; Hernandez-Ramirez, A.G. Assessment of Potential Toxic Metals in a Ramsar Wetland, Central Mexico and its Self-Depuration through Eichhornia crassipes. Water 2019, 11, 1248. https://doi.org/10.3390/w11061248
Tabla-Hernandez J, Rodriguez-Espinosa PF, Mendoza-Pérez JA, Sánchez-Ortíz E, Martinez-Tavera E, Hernandez-Ramirez AG. Assessment of Potential Toxic Metals in a Ramsar Wetland, Central Mexico and its Self-Depuration through Eichhornia crassipes. Water. 2019; 11(6):1248. https://doi.org/10.3390/w11061248
Chicago/Turabian StyleTabla-Hernandez, J., P. F. Rodriguez-Espinosa, J. A. Mendoza-Pérez, E. Sánchez-Ortíz, E. Martinez-Tavera, and A. G. Hernandez-Ramirez. 2019. "Assessment of Potential Toxic Metals in a Ramsar Wetland, Central Mexico and its Self-Depuration through Eichhornia crassipes" Water 11, no. 6: 1248. https://doi.org/10.3390/w11061248
APA StyleTabla-Hernandez, J., Rodriguez-Espinosa, P. F., Mendoza-Pérez, J. A., Sánchez-Ortíz, E., Martinez-Tavera, E., & Hernandez-Ramirez, A. G. (2019). Assessment of Potential Toxic Metals in a Ramsar Wetland, Central Mexico and its Self-Depuration through Eichhornia crassipes. Water, 11(6), 1248. https://doi.org/10.3390/w11061248






