Modified Local Soil (MLS) Technology for Harmful Algal Bloom Control, Sediment Remediation, and Ecological Restoration
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Sites
2.3. Preparation of Modified Local Soil
2.4. Treatment and Sample Analysis
3. Results
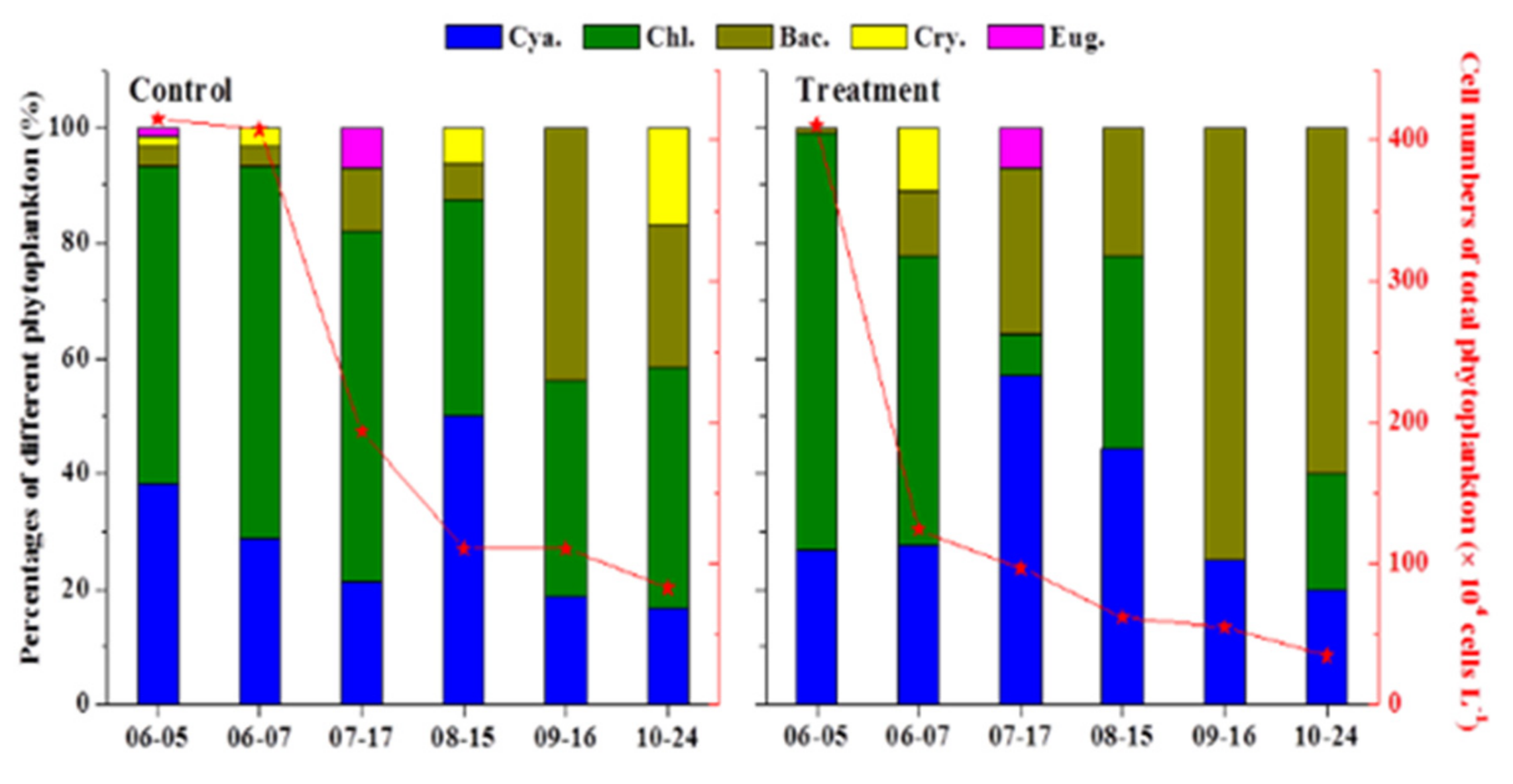
3.1. Water Quality Improvement and Algal Bloom Control
3.1.1. Emergent HABs Removal
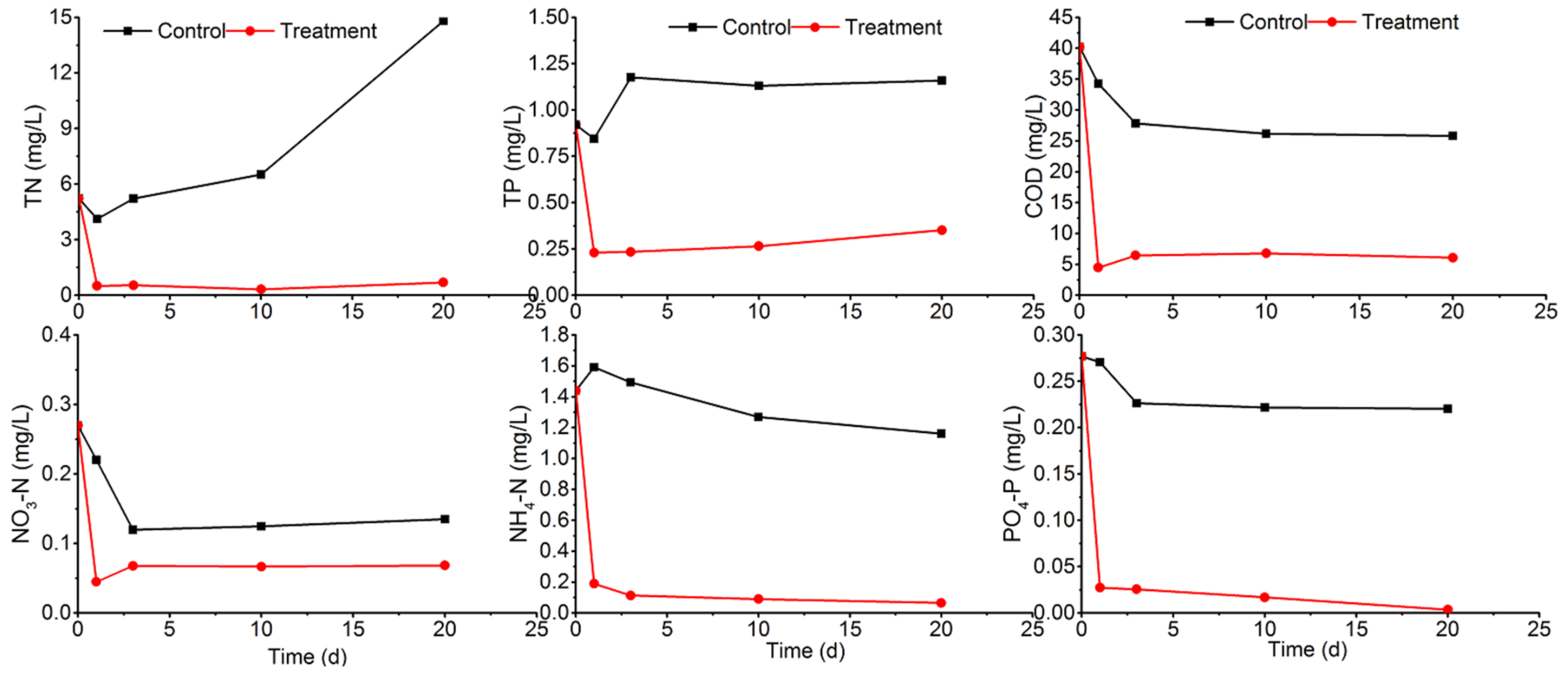
3.1.2. Short-Term Water Quality Improvement
3.1.3. Mid-Term Water Quality Improvement
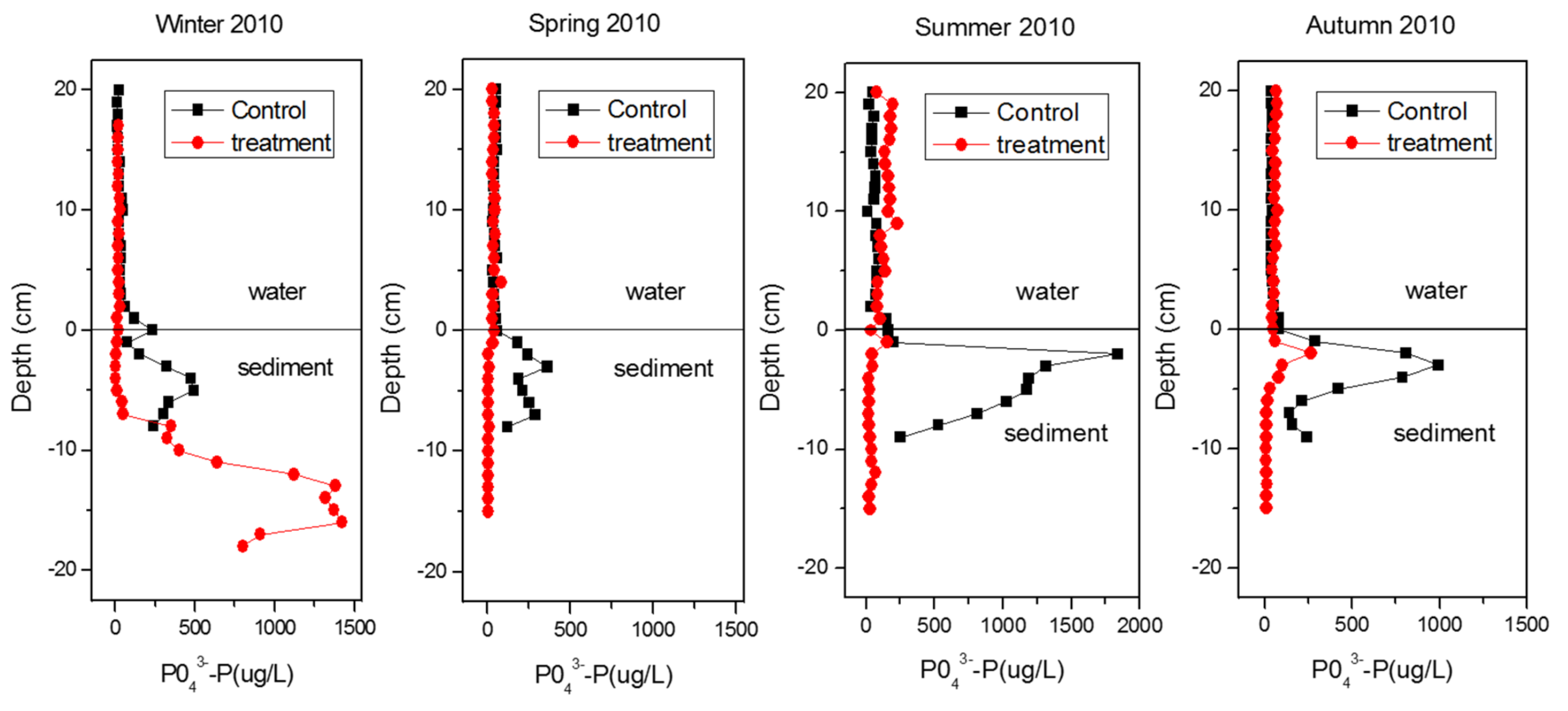
3.2. Sediment Remediation
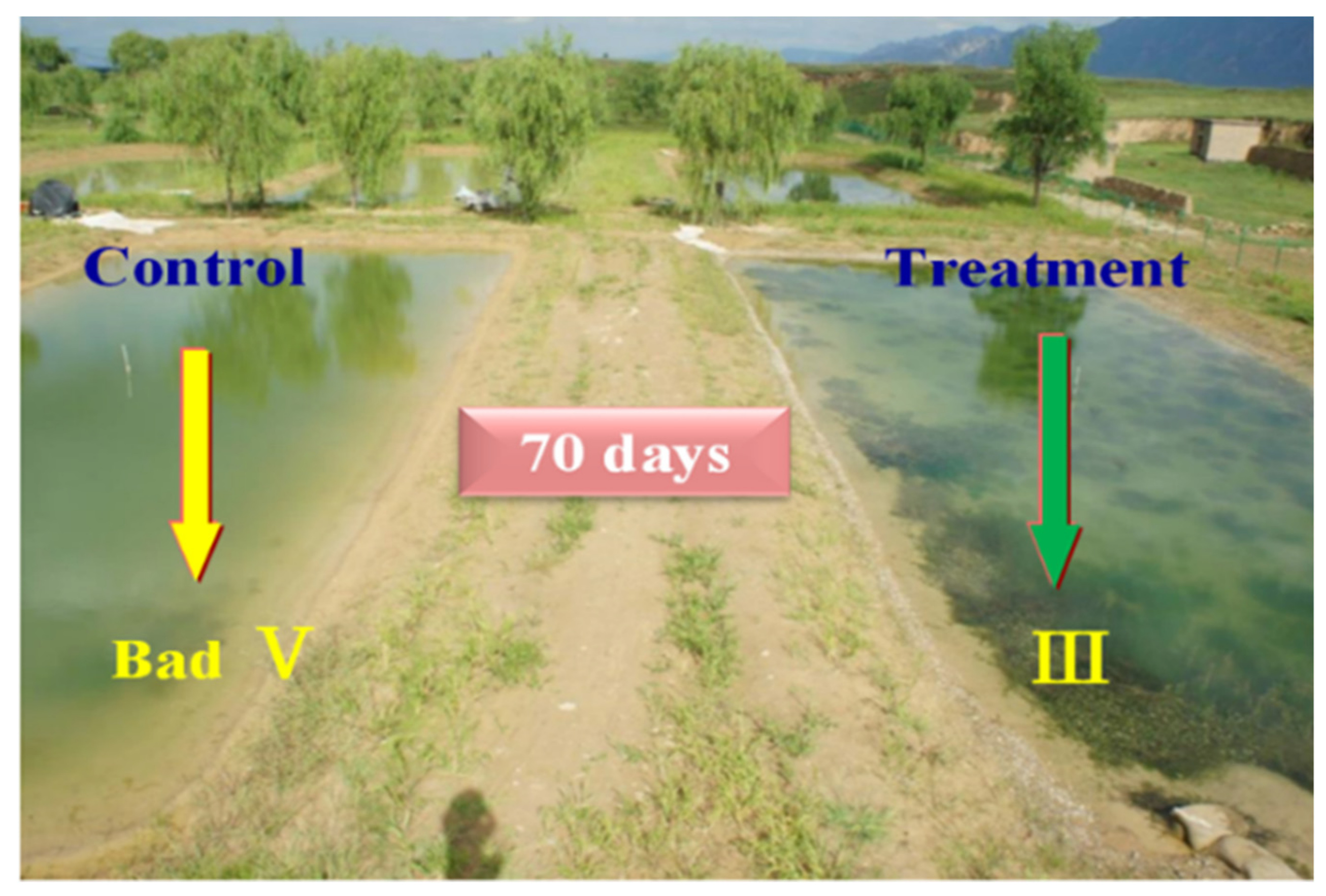
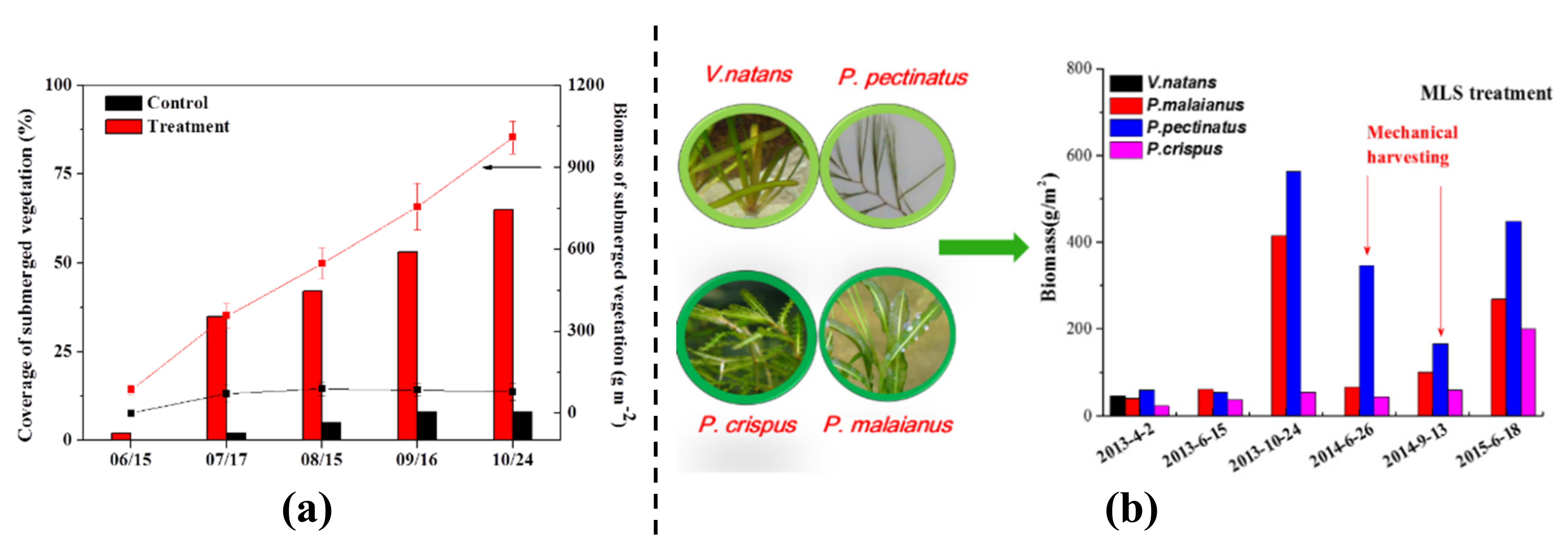
3.3. Long Term Ecological Restoration
4. Disscussion
4.1. Water Quality
4.2. Sediment Remediation
4.2.1. Shallow Waters
4.2.2. Deep Waters
4.3. Ecological Restoration
4.4. Cost Analysis
4.5. Ecological Safety
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Pan, G.; Lyu, T.; Mortimer, R. Comment: Closing phosphorus cycle from natural waters: Re-capturing phosphorus through an integrated water-energy-food strategy. J. Environ. Sci. 2018, 375–376. [Google Scholar] [CrossRef] [PubMed]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Controlling eutrophication: Nitrogen and phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Khare, Y.; Naja, G.M.; Stainback, G.A.; Martinez, C.J.; Paudel, R.; van Lent, T. A phased assessment of restoration alternatives to achieve phosphorus water quality targets for Lake Okeechobee, Florida, USA. Water 2019, 11, 327. [Google Scholar] [CrossRef]
- Lürling, M.; Mackay, E.; Reitzel, K.; Spears, B.M. Editorial–A critical perspective on geo-engineering for eutrophication management in lakes. Water Res. 2016, 97. [Google Scholar] [CrossRef]
- Mackay, E.B.; Maberly, S.C.; Pan, G.; Reitzel, K.; Bruere, A.; Corker, N.; Douglas, G.; Egemose, S.; Hamilton, D.; Hatton-Ellis, T. Geoengineering in lakes: Welcome attraction or fatal distraction? Inland Waters 2014, 4, 349–356. [Google Scholar] [CrossRef]
- Spears, B.M.; Maberly, S.C.; Pan, G.; Mackay, E.; Bruere, A.; Corker, N.; Douglas, G.; Egemose, S.; Hamilton, D.; Hatton-Ellis, T. Geo-engineering in lakes: A crisis of confidence? Environ. Sci. Technol. 2014. [Google Scholar] [CrossRef]
- Pan, G.; Krom, M.D.; Zhang, M.; Zhang, X.; Wang, L.; Dai, L.; Sheng, Y.; Mortimer, R.J. Impact of suspended inorganic particles on phosphorus cycling in the Yellow River (China). Environ. Sci. Technol. 2013, 47, 9685–9692. [Google Scholar] [CrossRef]
- Pan, G.; Zhang, M.-M.; Chen, H.; Zou, H.; Yan, H. Removal of cyanobacterial blooms in Taihu Lake using local soils. I. Equilibrium and kinetic screening on the flocculation of Microcystis aeruginosa using commercially available clays and minerals. Environ. Pollut. 2006, 141, 195–200. [Google Scholar] [CrossRef]
- Pan, G.; Chen, J.; Anderson, D.M. Modified local sands for the mitigation of harmful algal blooms. Harmful Algae 2011, 10, 381–387. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhang, H.; Pan, G. Flocculation of cyanobacterial cells using coal fly ash modified chitosan. Water Res. 2016, 97, 11–18. [Google Scholar] [CrossRef]
- Biswas, B.; Warr, L.N.; Hildera, E.F.; Goswami, N.; Rahman, M.M.; Churchman, J.G.; Vasilev, K.; Pan, G.; Naidub, R. Biocompatible functionalisation of nanoclays for improved environmental remediation. Chem. Soc. Rev. 2019. (under revision). [Google Scholar]
- Zou, H.; Pan, G.; Chen, H.; Yuan, X. Removal of cyanobacterial blooms in Taihu Lake using local soils II. Effective removal of Microcystis aeruginosa using local soils and sediments modified by chitosan. Environ. Pollut. 2006, 141, 201–205. [Google Scholar] [CrossRef]
- Pan, G.; Zou, H.; Chen, H.; Yuan, X. Removal of harmful cyanobacterial blooms in Taihu Lake using local soils III. Factors affecting the removal efficiency and an in situ field experiment using chitosan-modified local soils. Environ. Pollut. 2006, 141, 206–212. [Google Scholar] [CrossRef]
- Shi, W.; Tan, W.; Wang, L.; Pan, G. Removal of Microcystis aeruginosa using cationic starch modified soils. Water Res. 2016, 97, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Pan, G. A universal method for flocculating harmful algal blooms in marine and fresh waters using modified sand. Environ. Sci. Technol. 2013, 47, 4555–4562. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Pan, G. Harmful algal blooms mitigation using clay/soil/sand modified with xanthan and calcium hydroxide. J. Appl. Phycol. 2012, 24, 1183–1189. [Google Scholar] [CrossRef]
- Dai, L.; Pan, G. The effects of red soil in removing phosphorus from water column and reducing phosphorus release from sediment in Lake Taihu. Water Sci. Technol. 2014, 69, 1052–1058. [Google Scholar] [CrossRef]
- Pan, G.; Dai, L.; Li, L.; He, L.; Li, H.; Bi, L.; Gulati, R.D. Reducing the recruitment of sedimented algae and nutrient release into the overlying water using modified soil/sand flocculation-capping in eutrophic lakes. Environ. Sci. Technol. 2012, 46, 5077–5084. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Pan, G. Simultaneous removal of harmful algal blooms and microcystins using microorganism-and chitosan-modified local soil. Environ. Sci. Technol. 2015, 49, 6249–6256. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Yang, B.; Wang, D.; Chen, H.; Tian, B.-H.; Zhang, M.-L.; Yuan, X.-Z.; Chen, J. In-lake algal bloom removal and submerged vegetation restoration using modified local soils. Ecol. Eng. 2011, 37, 302–308. [Google Scholar] [CrossRef]
- Zhang, H.; Shang, Y.; Lyu, T.; Chen, J.; Pan, G. Switching harmful algal blooms to submerged macrophytes in shallow waters using geo-engineering methods: Evidence from a 15N tracing study. Environ. Sci. Technol. 2018, 52, 11778–11785. [Google Scholar] [CrossRef]
- Pan, G.; He, G.; Zhang, M.; Zhou, Q.; Tyliszczak, T.; Tai, R.; Guo, J.; Bi, L.; Wang, L.; Zhang, H. Nanobubbles at hydrophilic particle–Water interfaces. Langmuir 2016, 32, 11133–11137. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Miao, X.; Pan, G. Microwave-induced interfacial nanobubbles. Langmuir 2016, 32, 11147–11154. [Google Scholar] [CrossRef]
- Zhang, H.; Lyu, T.; Bi, L.; Tempero, G.; Hamilton, D.P.; Pan, G. Combating hypoxia/anoxia at sediment-water interfaces: A preliminary study of oxygen nanobubble modified clay materials. Sci. Total Environ. 2018, 637, 550–560. [Google Scholar] [CrossRef]
- Wang, L.; Miao, X.; Lyu, T.; Pan, G. Quantification of oxygen nanobubbles in particulate matters and potential applications in remediation of anaerobic environment. Am. Chem. Sci. Omega 2018, 3, 10624–10630. [Google Scholar] [CrossRef]
- Shi, W.; Pan, G.; Chen, Q.; Song, L.; Zhu, L.; Ji, X. Hypoxia remediation and methane emission manipulation using surface oxygen nanobubbles. Environ. Sci. Technol. 2018, 52, 8712–8717. [Google Scholar] [CrossRef] [PubMed]
- Conley, D.J.; Carstensen, J.; Vaquer-Sunyer, R.; Duarte, C.M. Ecosystem thresholds with hypoxia. Hydrobiologia 2009, 629, 21–29. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association (APHA): Washington, DC, USA, 2005. [Google Scholar]
- Hu, H. The Freshwater Algae of China: Systematics, Taxonomy and Ecology; Science Press: Beijing, China, 2006. [Google Scholar]
- Oladoja, N.A.; Pan, G. Modification of local soil/sand with Moringa oleifera extracts for effective removal of cyanobacterial blooms. Sustain. Chem. Pharm. 2015, 2, 37–43. [Google Scholar] [CrossRef]
- Douglas, G.; Hamilton, D.; Robb, M.; Pan, G.; Spears, B.; Lurling, M. Guiding principles for the development and application of solid-phase phosphorus adsorbents for freshwater ecosystems. Aquat. Ecol. 2016, 50, 385–405. [Google Scholar] [CrossRef]
- Wang, L.; Pan, G.; Shi, W.; Wang, Z.; Zhang, H. Manipulating nutrient limitation using modified local soils: A case study at Lake Taihu (China). Water Res. 2016, 101, 25–35. [Google Scholar] [CrossRef]
- Yuan, X.-Z.; Pan, G.; Chen, H.; Tian, B.-H. Phosphorus fixation in lake sediments using LaCl3-modified clays. Ecol. Eng. 2009, 35, 1599–1602. [Google Scholar] [CrossRef]
- Xu, R.; Zhang, M.; Mortimer, R.J.; Pan, G. Enhanced phosphorus locking by novel lanthanum/aluminum–hydroxide composite: Implications for eutrophication control. Environ. Sci. Technol. 2017, 51, 3418–3425. [Google Scholar] [CrossRef]
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic shifts in ecosystems. Nature 2001, 413, 591–596. [Google Scholar] [CrossRef]
- Pan, G.; Krom, M.D.; Herut, B. Adsorption−desorption of phosphate on airborne dust and riverborne particulates in East Mediterranean seawater. Environ. Sci. Technol. 2002, 36, 3519–3524. [Google Scholar] [CrossRef]
- Parkyn, S.M.; Hickey, C.W.; Clearwater, S.J. Measuring sub-lethal effects on freshwater crayfish (Paranephrops planifrons) behaviour and physiology: Laboratory and in situ exposure to modified zeolite. Hydrobiologia 2011, 661, 37–53. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, H.; Pan, G. Ecotoxicological assessment of flocculant modified soil for lake restoration using an integrated biotic toxicity index. Water Res. 2016, 97, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef]
- Mucci, M.; Noyma, N.P.; de Magalhaes, L.; Miranda, M.; van Oosterhout, F.; Guedes, I.A.; Huszar, V.L.; Marinho, M.M.; Lürling, M. Chitosan as coagulant on cyanobacteria in lake restoration management may cause rapid cell lysis. Water Res. 2017, 118, 121–130. [Google Scholar] [CrossRef]



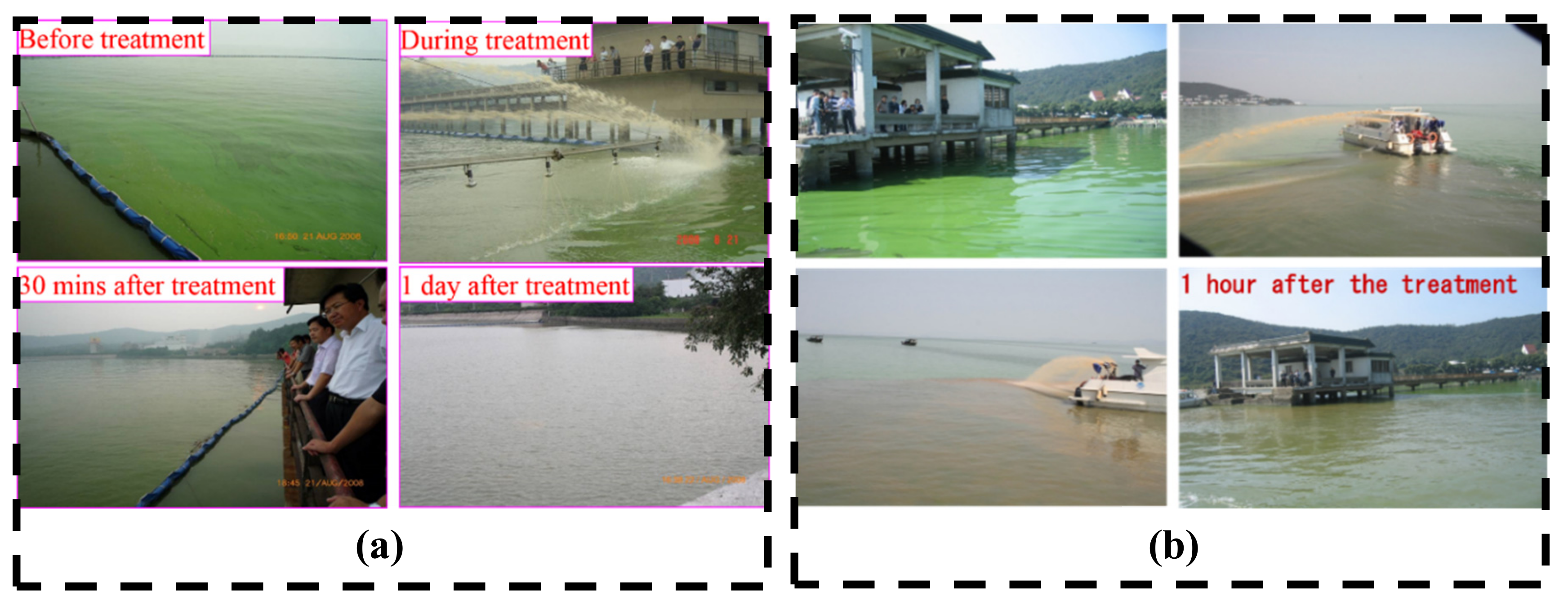




| Project | Time | Site | MLS Material | Monitoring | Duration |
|---|---|---|---|---|---|
| 1 | 2008 | Lake Tai- Meiliang Bay | chitosan MLS | Water (visually) | 1 day |
| 2 | 2009 | Lake Tai- Mashan Bay | chitosan MLS | Water (visually) | 1 day |
| 3 | 2012 | Lake Tai- Tanxi Bay | chitosan MLS | Water | 20 days |
| 4 | 2010 | Lake Tai- Shiba Bay | chitosan MLS | Sediment | 1 year |
| 5 | 2012 | Cetian Reservoir | chitosan-PAC MLS | Water Ecology | 70 days 3 years |
| pH | DO | TN | TP | COD | NH4+-N | |
|---|---|---|---|---|---|---|
| mg/L | ||||||
| Treated Pond | 9 | 10.8I | 0.8III | 0.08II | 14.5II | 0.4II |
| Std. | 0.4 | 1.4 | 0.3 | 0.03 | 2.1 | 0.2 |
| Control Pond | 8.9 | 5.9II | 3.1 bV | 0.5 bV | 58 bV | 1.1IV |
| Std. | 0.5 | 0.9 | 1.8 | 0.3 | 8.5 | 0.3 |
| Particle Materials | Flocculation by MLS1 | Capping4 | Extra O2 Supply5 | Operation | ||
|---|---|---|---|---|---|---|
| Types | Price ($/t) | Chitosan MLS2 (k$/km2) | Chitosan-PAC MLS3 (k$/km2) | Price (k$/km2) | O2 nanobubble Modification (k$/km2) | Mechanic Spray (k$/km2) |
| Kaolinite | 135 | 38 | 59 | 2024 | - | 1.5 |
| Local soil | 60 | 35 | 56 | 900 | - | 1.5 |
| zeolite | 120 | - | - | 1799 | 30 | 1.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, G.; Miao, X.; Bi, L.; Zhang, H.; Wang, L.; Wang, L.; Wang, Z.; Chen, J.; Ali, J.; Pan, M.; et al. Modified Local Soil (MLS) Technology for Harmful Algal Bloom Control, Sediment Remediation, and Ecological Restoration. Water 2019, 11, 1123. https://doi.org/10.3390/w11061123
Pan G, Miao X, Bi L, Zhang H, Wang L, Wang L, Wang Z, Chen J, Ali J, Pan M, et al. Modified Local Soil (MLS) Technology for Harmful Algal Bloom Control, Sediment Remediation, and Ecological Restoration. Water. 2019; 11(6):1123. https://doi.org/10.3390/w11061123
Chicago/Turabian StylePan, Gang, Xiaojun Miao, Lei Bi, Honggang Zhang, Lei Wang, Lijing Wang, Zhibin Wang, Jun Chen, Jafar Ali, Minmin Pan, and et al. 2019. "Modified Local Soil (MLS) Technology for Harmful Algal Bloom Control, Sediment Remediation, and Ecological Restoration" Water 11, no. 6: 1123. https://doi.org/10.3390/w11061123
APA StylePan, G., Miao, X., Bi, L., Zhang, H., Wang, L., Wang, L., Wang, Z., Chen, J., Ali, J., Pan, M., Zhang, J., Yue, B., & Lyu, T. (2019). Modified Local Soil (MLS) Technology for Harmful Algal Bloom Control, Sediment Remediation, and Ecological Restoration. Water, 11(6), 1123. https://doi.org/10.3390/w11061123









