PFASs in Finnish Rivers and Fish and the Loading of PFASs to the Baltic Sea
Abstract
:1. Introduction
2. Methods
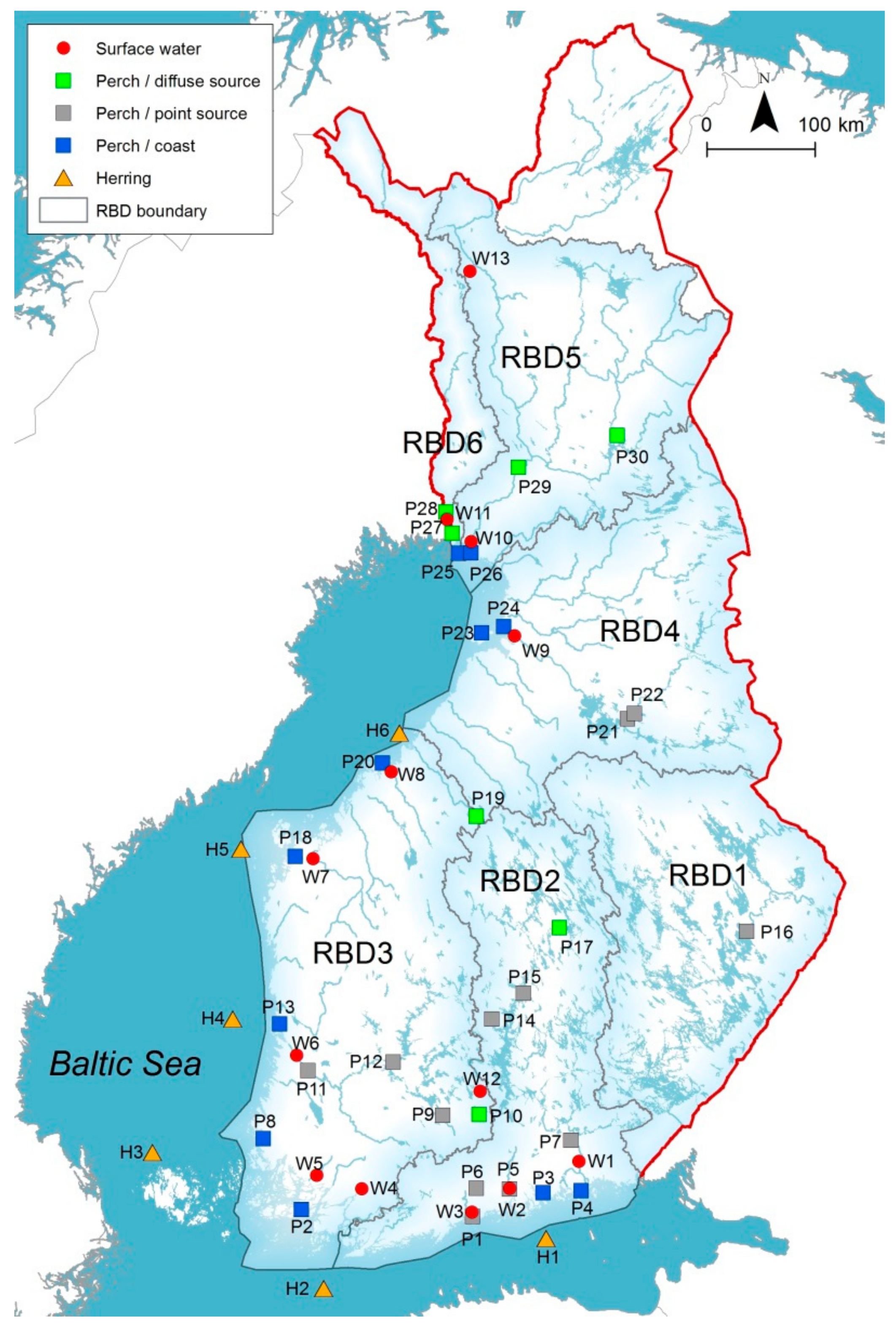
2.1. Selection and Classification of Sampling Sites
2.2. Surface Water Sampling and Sample Preparation
2.3. Fish Sampling and Sample Preparation
2.4. Analytical Methods
2.5. Calculation of Riverine PFAS Load to the Baltic Sea
2.6. Statistical Analysis
3. Results
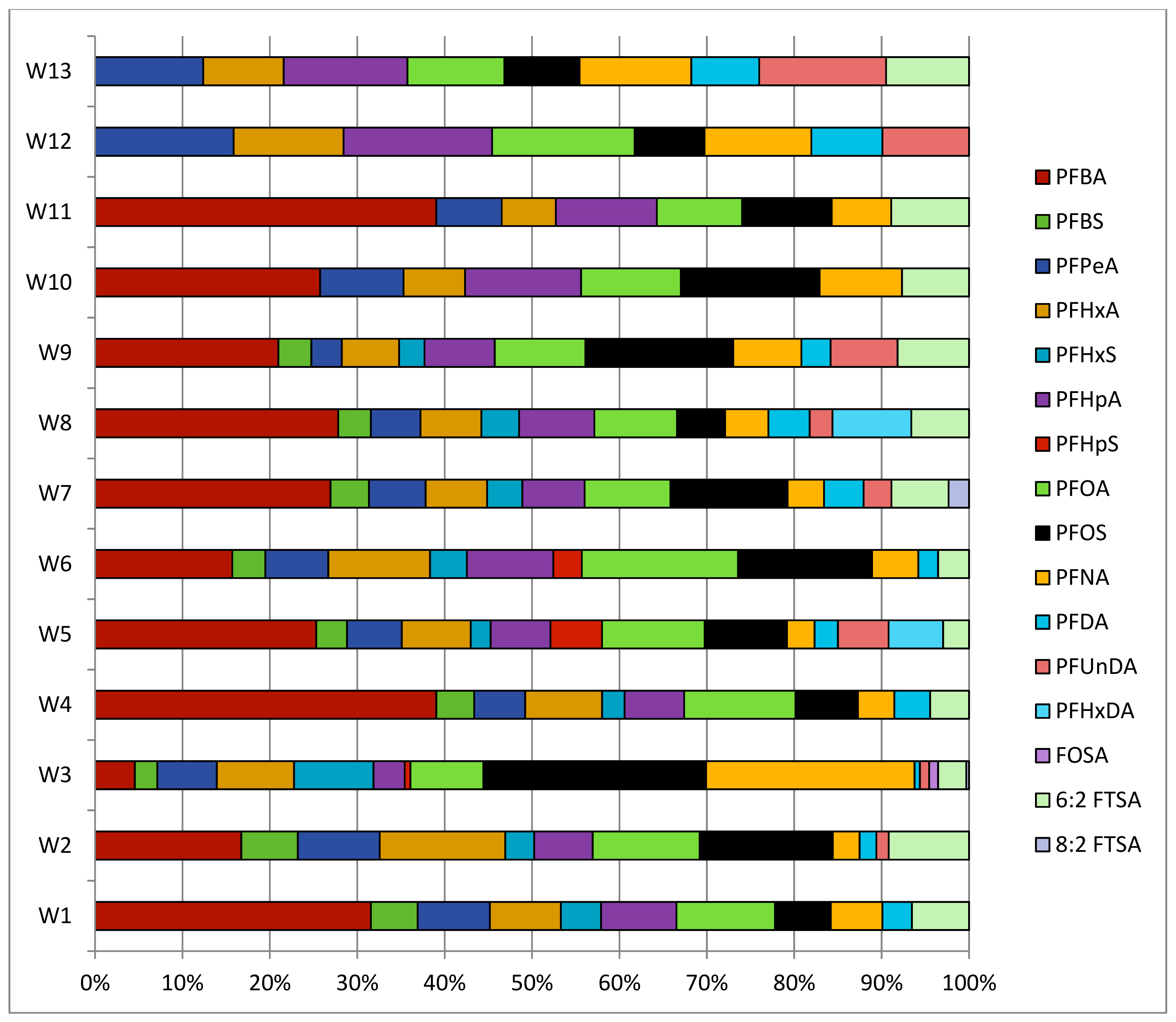
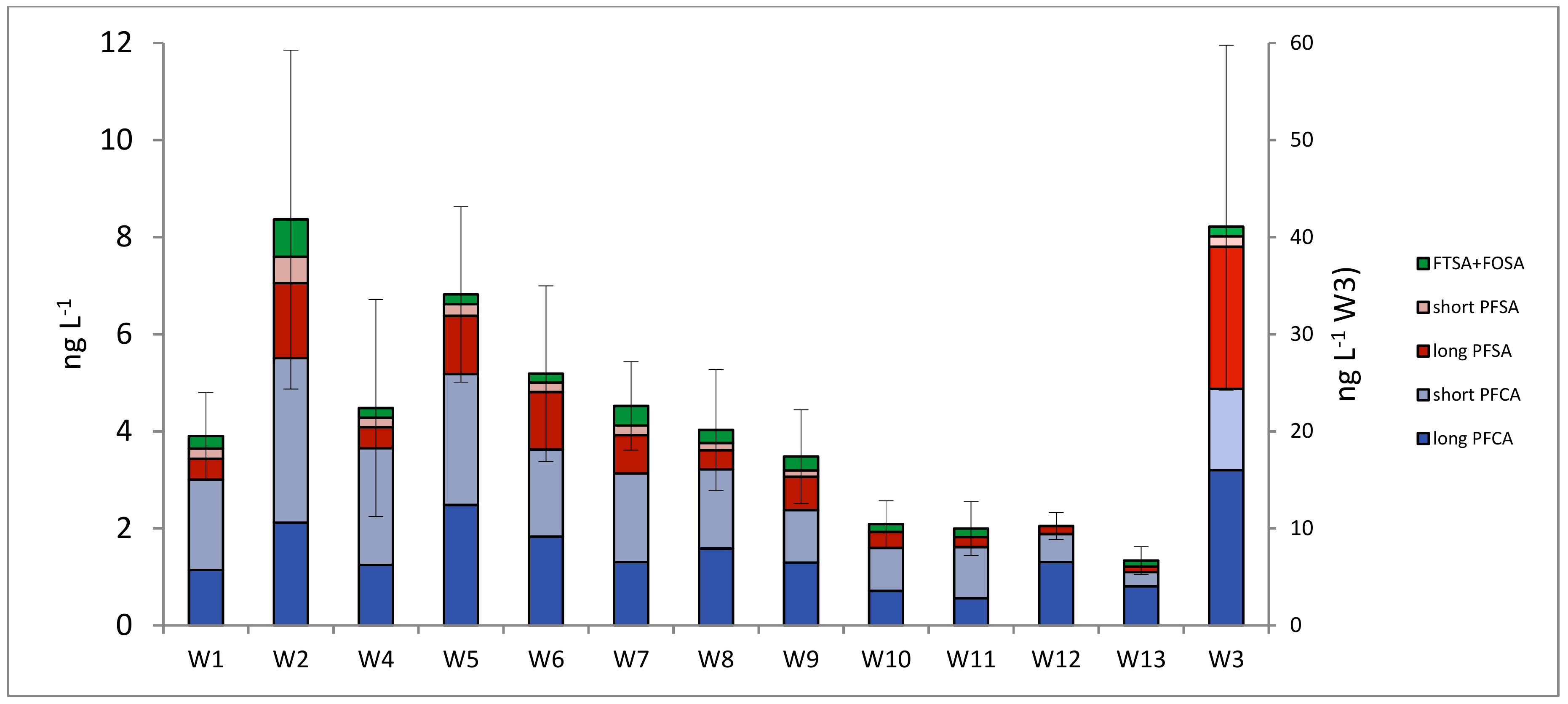
3.1. PFASs in Surface Waters
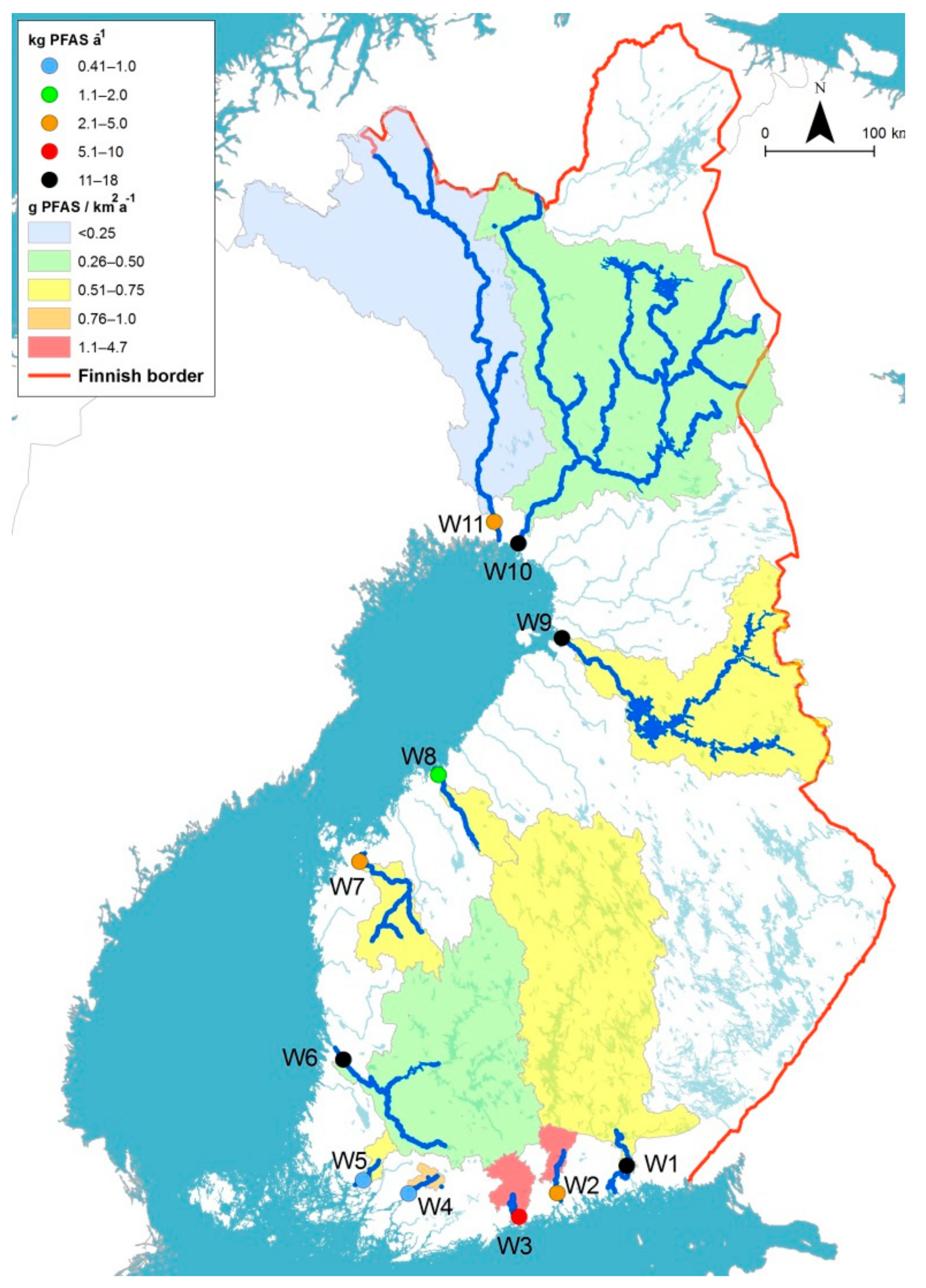
3.2. PFAS Load of the Studied Rivers to the Baltic Sea
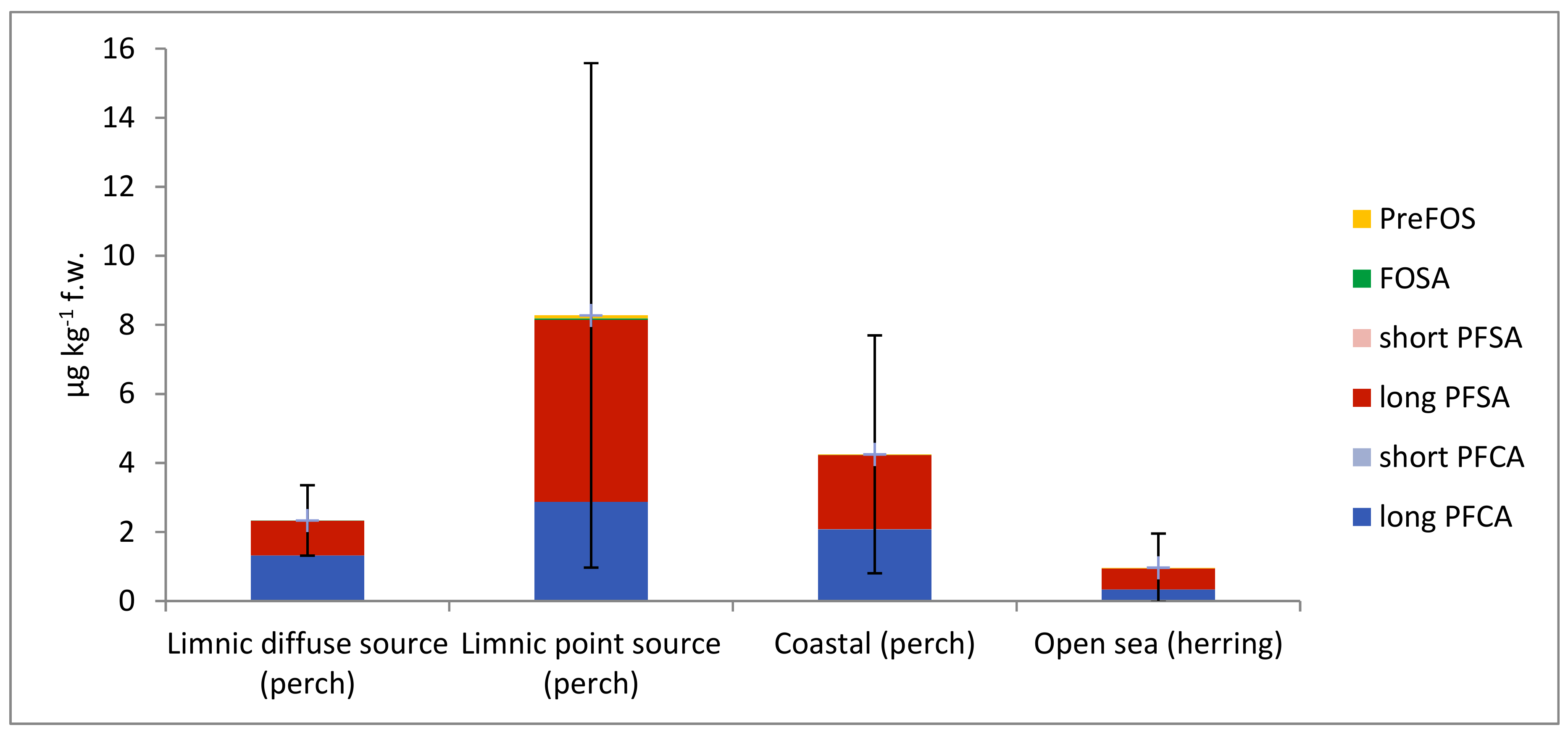
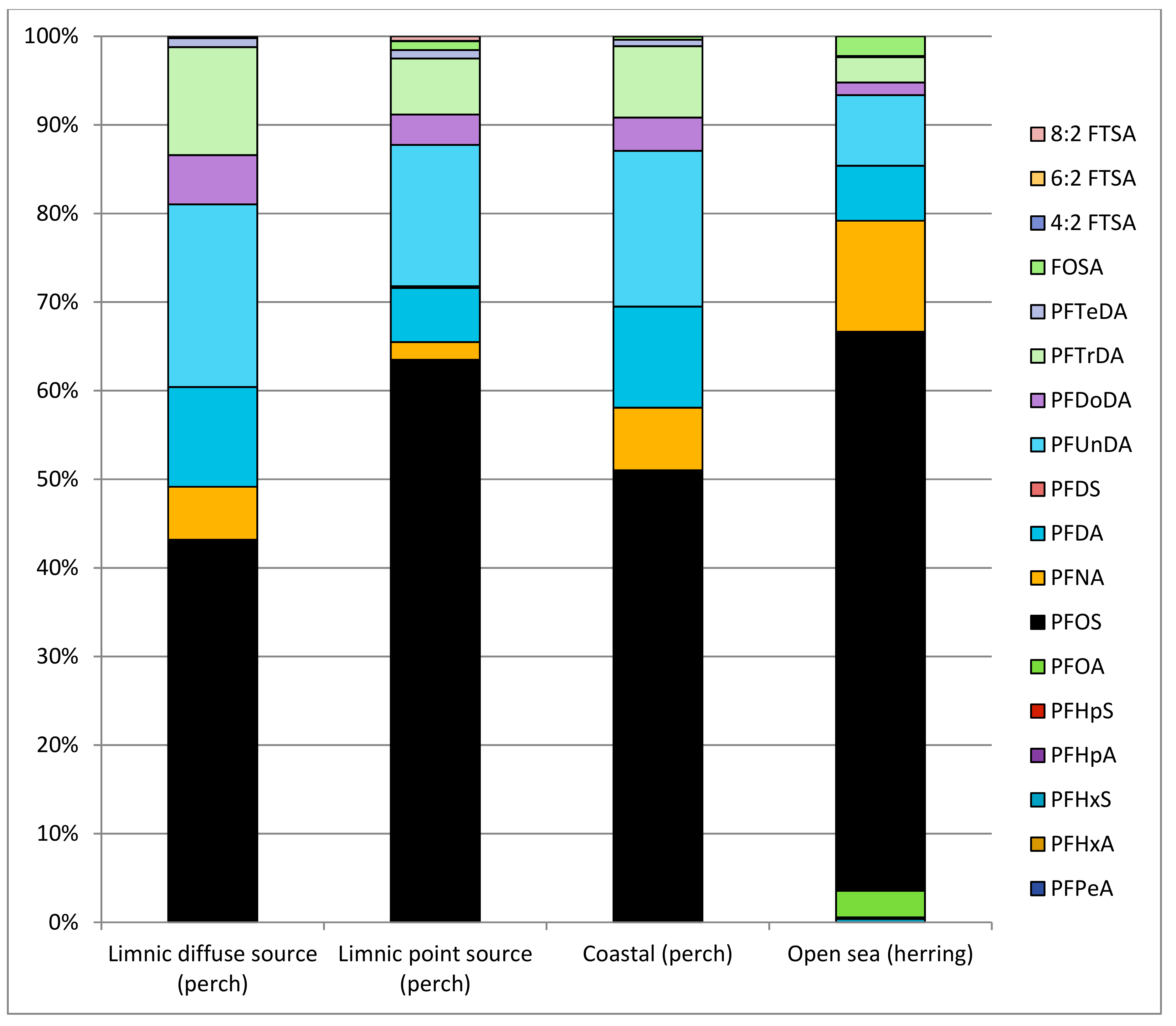
3.3. PFASs in Fish
4. Discussion
4.1. PFASs in Surface Waters
4.2. PFASs in Fish
4.3. PFAS Load to the Baltic Sea
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vaalgamaa, S.; Vähätalo, A.V.; Perkola, N.; Huhtala, S. Photochemical reactivity of perfluorooctanoic acid (PFOA) in conditions representing surface water. Sci. Total Environ. 2011, 409, 3043–3048. [Google Scholar] [CrossRef]
- OECD. Co-Operation on Existing Chemicals—Hazard Assessment of Perfluorooctane Sulfonate and Its Salts, Environment Directorate Joint Meeting of the Chemicals Committee and the Working Party on Chemicals, Pesticides and Biothechnology; Organisation for Economic Co-Operation and Development: Paris, France, 2002. [Google Scholar]
- Ahrens, L. Polyfluoroalkyl compounds in the aquatic environment: A review of their occurrence and fate. J. Environ. Monit. 2011, 13, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Haukås, M.; Berger, U.; Hop, H.; Gulliksen, B.; Gabrielsen, G.W. Bioaccumulation of per- and polyfluorinated alkyl substances (PFAS) in selected species from the Barents Sea food web. Environ. Pollut. 2007, 148, 360–371. [Google Scholar] [CrossRef]
- Ahrens, L.; Rakovic, J.; Axelson, S.; Kallenborn, R. Source Tracking and Impact of Per- and Polyfluoroalkyl Substances at Svalbard–FluorosImpact–Svalbards Miljøvernfond; Swedish University of Agricultural Sciences: Uppsala, Sweden, 2016; p. 17. [Google Scholar]
- AMAP. AMAP Assessment 2016: Chemicals of Emerging Arctic Concern; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2017. [Google Scholar]
- Ahrens, L.; Bundschuh, M. Fate and effects of poly- and perfluoroalkyl substances in the aquatic environment: A Review. Environ. Toxicol. Chem. 2014, 33, 1921–1929. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.; Mabury, S.; Solomon, K.; Muir, D. Bioconcentration and tissue distribution of perfluorinated acids in rainbow trout (Oncorhynchus mykiss). Environ. Toxicol. Chem. 2003, 22, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Scheringer, M.; Trier, X.; Cousins, I.T.; de Voogt, P.; Fletcher, T.; Wang, Z.; Webster, T.F. Helsingør statement on poly- and perfluorinated alkyl substances (PFASs). Chemosphere 2014, 114, 337–339. [Google Scholar] [CrossRef]
- Blum, A.; Balan, S.A.; Scheringer, M.; Trier, X.; Goldenman, G.; Cousins, I.T.; Diamond, M.; Fletcher, T.; Higgins, C.; Lindeman, A.E.; et al. The Madrid statement on poly- and perfluoroalkyl substances (PFASs). Environ. Health Perspect. 2015, 123, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Danish EPA. Short Chain Polyfluoralkyl Substances (PFAS); Environmental Project No. 1707; Danish EPA: København, Denmark, 2015; p. 106. [Google Scholar]
- Jensen, A.A.; Brunn Poulsen, P.; Bossi, R. Survey and Environmental/Health Assessment of Fluorinated Substances in Impregnated Consumer Products and Impregnating Agents; Survey of Chemical Substances in Consumer Products, No. 99; Danish EPA: København, Denmark, 2008; 160p. [Google Scholar]
- Möller, A.; Ahrens, L.; Surm, R.; Westerveld, J.; van der Wielen, F.; Ebinghaus, R.; de Voogt, P. Distribution and sources of polyfluoroalkyl substances (PFAS) in the River Rhine watershed. Environ. Pollut. 2010, 158, 3243–3250. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.C.; Andrews, D.Q.; Lindstrom, A.B.; Bruton, T.A.; Schaider, L.A.; Grandjean, P.; Lohmann, R.; Carignan, C.C.; Blum, A.; Balan, S.A.; et al. Detection of poly- and perfluoroalkyl substances (PFASs) in US drinking water linked to industrial sites, military fire training areas, and wastewater treatment plants. Environ. Sci. Technol. Lett. 2016, 3, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Koponen, J.; Airaksinen, R.; Hallikainen, A.; Vuorinen, P.J.; Mannio, J.; Kiviranta, H. Perfluoroalkyl acids in various edible Baltic, freshwater, and farmed fish in Finland. Chemosphere 2015, 129, 186–191. [Google Scholar] [CrossRef]
- HELCOM. Guidelines for the Annual and Periodical Compilation and Reporting of Waterborne Pollution Inputs to the Baltic Sea (PLC-Water); HELCOM: Helsinki, Finland, 2015. [Google Scholar]
- Nguyen, M.A.; Wiberg, K.; Ribeli, E.; Josefsson, S.; Futter, M.; Gustavsson, J.; Ahrens, L. Spatial distribution and source tracing of per- and polyfluoroalkyl substances (PFASs) in surface water in Northern Europe. Environ. Pollut. 2017, 220, 1438–1446. [Google Scholar] [CrossRef] [PubMed]
- Perkola, N.; Sainio, P. Survey of perfluorinated alkyl acids in Finnish effluents, storm water, landfill leachate and sludge. Environ. Sci. Pollut. Res. 2013, 20, 7979–7987. [Google Scholar] [CrossRef] [PubMed]
- Young, C.J.; Mabury, S.A. Atmospheric Perfluorinated Acid Precursors: Chemistry, Occurrence, and Impacts. In Reviews of Environmental Contamination and Toxicology, Vol 208: Perfluorinated Alkylated Substances; Springer: New York, NY, USA, 2010; Volume 208, pp. 1–109. [Google Scholar]
- Wang, N.; Liu, J.; Buck, R.C.; Korzeniowski, S.H.; Wolstenholme, B.W.; Folsom, P.W.; Sulecki, L.M. 6:2 Fluorotelomer sulfonate aerobic biotransformation in activated sludge of waste water treatment plants. Chemosphere 2011, 8, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Huang, J.; Zhang, K.; Yu, G.; Deng, S.; Wang, B. Stability of 6:2 fluorotelomer sulfonate in advanced oxidation processes: Degradation kinetics and pathway. Environ. Sci. Pollut. Res. 2013, 21, 4634–4642. [Google Scholar] [CrossRef]
- KEMI. Chemical Analysis of Selected Fire-Fighting foams on the Swedish Market 2014; Swedish Chemicals Agency: Stockholm, Sweden, 2015; 22p. [Google Scholar]
- Favreau, P.; Poncioni-Rothlisberger, C.; Place, B.J.; Bouchex-Bellomie, H.; Weber, A.; Tremp, J.; Field, J.A.; Kohler, M. Multianalyte profiling of per- and polyfluoroalkyl substances (PFASs) in liquid commercial products. Chemosphere 2017, 171, 491–501. [Google Scholar] [CrossRef]
- Woldegiorgis, A.; Andersson, J.; Remberger, M.; Kaj, L.; Ekheden, Y.; Blom, L.; Brorsrtöm-Lundén, E. Results from the Swedish National Screening Programme 2005: Subreport 3: Perfluorinated Alkylated Substances (PFAS); IVL Report B1698; Swedish Environmental Research Institute: Stockholm, Sweden, 2006; 4p. [Google Scholar]
- Filipovic, M.; Laudon, H.; McLachlan, M.S.; Berger, U. Mass balance of perfluorinated alkyl acids in a pristine boreal catchment. Environ. Sci. Technol. 2015, 49, 12127–12135. [Google Scholar] [CrossRef] [PubMed]
- Bignert, A.; Danielsson, S.; Faxneld, S.; Nyberg, E. Comments Concerning the National Swedish Contaminant Monitoring Programme in Marine Biota, 2016; Swedish Museum of Natural History: Stockholm, Sweden, 2016. [Google Scholar]
- Guelfo, J.L.; Adamson, D.T. Evaluation of a national data set for insights into sources, composition, and concentrations of per- and polyfluoroalkyl substances (PFASs) in US drinking water. Environ. Pollut. 2018, 236, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Codling, G.; Halsall, C.; Ahrens, L.; Del Vento, S.; Wiberg, K.; Bergknut, M.; Laudon, H.; Ebinghaus, R. The fate of per- and polyfluoroalkyl substances within a melting snowpack of a boreal forest. Environ. Pollut. 2014, 191, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, L.; Hedlund, J.; Dürig, W.; Tröger, R.; Wiberg, K. Screening of PFASs in Groundwater and Surface Water; Rapport 2016:2; Sveriges Lantbruksuniversitet, Institutionen för Vatten Och Miljö: Uppsala, Sweden, 2016; 82p. [Google Scholar]
- Hamid, H.; Loretta, Y.L.; Grace, J.R. Review of the fate and transformation of per- and polyfluoroalkyl substances (PFASs) in landfills. Environ. Pollut. 2018, 235, 74–85. [Google Scholar] [CrossRef]
- European Union. Guidance Document No. 32 on Biota Monitoring (the Implementation of EQSbiota) under the Water Framework Directive; European Union: Luxembourg, Luxembourg, 2014. [Google Scholar]
- Järv, L.; Kiviranta, H.; Koponen, J.; Rantakokko, P.; Ruokojärvi, P.; Radin, M.; Raid, T.; Roots, O.; Simm, M. Persistent organic pollutants in selected fishes of the Gulf of Finland. J. Mar. Syst. 2017, 171, 129–133. [Google Scholar] [CrossRef]
- Faxneld, S.; Danielsson, S.; Nyberg, E. Distribution of PFAS in Liver and Muscle of Herring, Perch, Cod, Eelpout, Arctic Char, and Pike from Limnic and Marine Environments in Sweden; Swedish Museum of Natural History: Stockholm, Sweden, 2014. [Google Scholar]
- Fliedner, A.; Lohmann, N.; Ruedel, H.; Teubner, D.; Wellmitz, J.; Koschorreck, J. Current levels and trends of selected EU Water Framework Directive priority substances in freshwater fish from the German environmental specimen bank. Environ. Pollut. 2016, 216, 866–876. [Google Scholar] [CrossRef] [PubMed]
- Svihlikova, V.; Lankova, D.; Poustka, J.; Tomaniova, M.; Hajslova, J.; Pulkrabova, J. Perfluoroalkyl substances (PFASs) and other halogenated compounds in fish from the upper Labe River basin. Chemosphere 2015, 129, 170–178. [Google Scholar] [CrossRef] [PubMed]
- EFSA CONTAM Panel (EFSA Panel on Contaminants in the Food Chain). Scientific opinion on the risk to human health related to the presence of perfluorooctane sulfonic acid and perfluorooctanoic acid in food. EFSA J. 2018, 16, 5194. [Google Scholar] [CrossRef]
- Filipovic, M.; Berger, U.; McLachlan, M.S. Mass balance of perfluoroalkyl acids in the Baltic Sea. Environ. Sci. Technol. 2013, 47, 4088–4095. [Google Scholar] [CrossRef] [PubMed]
- Vieno, N. Hazardous Substances at Wastewater Treatment Project Report (In Finnish); Vesilaitosyhdistyksen monistesarja nro 34; Finnish Water Utilities Association: Helsinki, Finland, 2014. [Google Scholar]
| Compound | Abbreviation | Category |
|---|---|---|
| Perfluorooctanoic acid | PFOA | Long PFCA |
| Perfluoroheptanoic acid | PFHpA | Long PFCA |
| Perfluorohexanoic acid | PFHxA | Short PFCA |
| Perfluorooctane sulfonic acid | PFOS | Long PFSA |
| Perfluorononaoic acid | PFNA | Long PFCA |
| Perfluoropentanoic acid | PFPeA | Short PFCA |
| Perfluorobutanoic acid | PFBA | Short PFCA |
| Perfluorobutane sulfonic acid | PFBS | Short PFSA |
| Perfluorohexane sulfonic acid | PFHxS | Long PFSA |
| 6:2 Fluorotelomer sulfonic acid | 6:2 FTSA | FTSA |
| Perfluorodecanoic acid | PFDA | Long PFCA |
| Perfluoroundecanoic acid | PFUnDA | Long PFCA |
| Perfluoroheptane sulfonic acid | PFHpS | Long PFSA |
| Perfluorohexadecanoic acid | PFHxDA | Long PFCA |
| 8:2 Fluorotelomer sulfonic acid | 8:2 FTSA | FTSA |
| Perfluorododecanoic acid | PFDoDA | Long PFCA |
| Perfluorotridecanoic acid | PFTrDA | Long PFCA |
| Perfluorotetradecanoic acid | PFTeDA | Long PFCA |
| Perfluorooctadecanoic acid | PFODA | Long PFCA |
| Perfluorodecane sulfonic acid | PFDS | Long PFSA |
| 4:2 Fluorotelomer sulfonic acid | 4:2 FTSA | FTSA |
| N-ethyl perfluorooctane sulfonamide | N-EtFOSA | PreFOS |
| Perfluorooctane sulfonamide | FOSA | PreFOS |
| Sampling Site ID | Name of Water Body | RBD | Catchment Area (km2) | Population in Catchment Area | Average Flow during Sampling Period (m3 s−1) | Number of Samples |
|---|---|---|---|---|---|---|
| W1 | Kymijoki | 2 | 36,300 | 551,000 | 228 | 8 |
| W2 | Porvoonjoki | 2 | 1140 | 96,700 | 7.90 | 8 |
| W3 | Vantaanjoki | 2 | 1670 | 496,948 | 10.5 | 10 |
| W4 | Uskelanjoki | 3 | 514 | 20,600 | 2.50 | 9 |
| W5 | Aurajoki | 3 | 735 | 37,200 | 2.50 | 9 |
| W6 | Kokemäenjoki | 3 | 26,400 | 749,000 | 128 | 9 |
| W7 | Kyrönjoki | 3 | 4810 | 112,000 | 32.1 | 9 |
| W8 | Perhonjoki | 3 | 2410 | 14,300 | 2.90 | 8 |
| W9 | Oulujoki | 4 | 22,700 | 136,000 | 257 | 8 |
| W10 | Kemijoki | 5 | 50,600 | 98,100 | 618 | 8 |
| W11 | Tornionjoki | 6 | 39,400 (the Finnish side of the river) | 15 300 | 492 | 9 |
| W13 | Pallasjoki (background) | 5 | 107 | 0 | n.a. | 4 |
| W12 | Outlet of Lake Valkea–Kotinen (background) | 2 | 0.22 | 0 | n.a. | 5 |
| Substance | Group | Detection Frequency (%) | LoQ (ng L−1) | U (%) | Max. Concentration (Mean) (ng L−1) |
|---|---|---|---|---|---|
| PFOA | Long PFCA | 96 | 0.10 | 25 | 5.4 (0.73) |
| PFHpA | Long PFCA | 91 | 0.10 | 25 | 2.7 (0.44) |
| PFHxA | Short PFCA | 84 | 0.10 | 23 | 5.7 (0.64) |
| PFOS | Long PFSA | 78 | 0.10 | 25 | 26 (1.2) |
| PFNA | Long PFCA | 76 | 0.10 | 29 | 23 (0.97) |
| PFPeA | Short PFCA | 73 | 0.10 | 27 | 4.2 (0.51) |
| PFBA | Short PFCA | 65 | 0.50 | 30 | 5.3 (1.0) |
| PFBS | Short PFSA | 63 | 0.10 | 29 | 1.5 (0.23) |
| PFHxS | Long PFSA | 56 | 0.10 | 25 | 6.4 (0.40) |
| 6:2 FTSA | FTSA | 49 | 0.10 | 25 | 2.7 (0.33) |
| PFDA | Long PFCA | 29 | 0.10 | 24 | 0.52 (0.14) |
| PFUnDA | Long PFCA | 16 | 0.10 | 32 | 1.9 (0.14) |
| PFHpS | Long PFSA | 11 | 0.10 | 22 | 0.40 (0.065) |
| FOSA | PreFOS | 3 | 0.20 | 25 | 0.70 (0.032) |
| PFHxDA | Long PFCA | 2 | 0.25 | 34 | 0.43 (0.061) |
| 8:2 FTSA | FTSA | 2 | 0.10 | 25 | 0.13 (0.018) |
| PFDoDA | Long PFCA | n.d. | 0.50 | 32 | <LoQ |
| PFTrDA | Long PFCA | n.d. | 0.25 | 27 | <LoQ |
| PFTeDA | Long PFCA | n.d. | 0.25 | 30 | <LoQ |
| PFODA | Long PFCA | n.d. | 0.50 | 39 | <LoQ |
| PFDS | Long PFSA | n.d. | 0.20 | 39 | <LoQ |
| 4:2 FTSA | FTSA | n.d. | 0.10 | 25 | <LoQ |
| N-EtFOSA | PreFOS | n.d. | 0.50 | 35 | <LoQ |
| River | PFBA | PFBS | PFPeA | PFHxA | PFHxS | PFHpA | PFOA | PFOS | PFNA | 6:2 FTSA | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|
| W1 | 5.5 | 0.97 | 1.6 | 1.6 | 0.68 | 1.5 | 2.2 | 1.2 | 1.1 | 1.9 | 18 |
| W2 | 0.26 | 0.10 | 0.12 | 0.17 | 0.04 | 0.71 | 0.19 | 0.18 | 0.03 | 0.25 | 2.0 |
| W3 | 0.48 | 0.22 | 0.57 | 0.72 | 0.71 | 0.27 | 0.76 | 2.1 | 1.4 | 0.61 | 7.8 |
| W4 | 0.20 | 0.01 | 0.02 | 0.04 | - | 0.03 | 0.06 | 0.02 | 0.02 | 0.01 | 0.41 |
| W5 | 0.17 | 0.02 | 0.04 | 0.05 | 0.01 | 0.04 | 0.07 | 0.03 | 0.02 | 0.01 | 0.44 |
| W6 | 1.9 | 0.49 | 0.97 | 1.5 | 0.56 | 1.0 | 2.2 | 2.1 | 0.68 | 0.66 | 12 |
| W7 | 1.2 | 0.15 | 0.23 | 0.23 | 0.12 | 0.23 | 0.35 | 0.39 | 0.11 | 0.11 | 3.1 |
| W8 | 0.56 | 0.07 | 0.07 | 0.13 | 0.06 | 0.14 | 0.22 | 0.08 | 0.06 | 0.03 | 1.4 |
| W9 | 2.2 | 0.17 | 0.36 | 1.2 | 0.10 | 1.4 | 2.0 | 1.8 | 1.3 | 1.8 | 12 |
| W10 | 4.4 | - | 0.84 | 0.82 | - | 2.4 | 2.0 | 1.8 | 1.2 | 1.1 | 15 |
| W11 | 0.44 | - | 0.32 | 0.23 | - | 0.61 | 0.61 | 0.33 | 0.25 | 0.56 | 3.4 |
| Total | 17 | 2.2 | 5.1 | 6.7 | 2.3 | 8.1 | 11 | 10 | 6.2 | 7.0 |
| Perch | Herring | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PFAS | Group | LoQ (µg kg−1) | U (%) | n | Detection Frequency | Mean (µg kg−1) | Max (µg kg−1) | n | Detection Frequency | Mean (µg kg−1) | Max. (µg kg−1) |
| PFOS | long PFSA | 0.01 | 25 | 48 | 100% | 3.4 | 18 | 48 | 100% | 0.49 | 1.6 |
| PFUnDA | long PFCA | 0.02 | 24 | 48 | 100% | 1.0 | 8.8 | 48 | 25% | 0.11 | 0.29 |
| PFTrDA | long PFCA | 0.03 | 25 | 48 | 100% | 0.45 | 1.8 | 48 | 10% | 0.06 | 0.09 |
| PFDA | long PFCA | 0.01 | 35 | 48 | 100% | 0.50 | 1.6 | 48 | 38% | 0.08 | 0.25 |
| PFNA | long PFCA | 0.01 | 24 | 48 | 100% | 0.23 | 1.1 | 48 | 60% | 0.18 | 0.56 |
| PFDoDA | long PFCA | 0.03 | 24 | 48 | 100% | 0.23 | 0.52 | 48 | 2% | 0.06 | 0.06 |
| PFTeDA | long PFCA | 0.04 | 25 | 48 | 58% | 0.09 | 0.15 | 48 | n.d. | - | - |
| FOSA | PreFOS | 0.02 | 25 | 18 | 50% | 0.11 | 0.38 | 16 | 44% | 0.04 | 0.09 |
| PFHpS | long PFSA | 0.01 | 29 | 48 | 29% | 0.02 | 0.06 | 48 | n.d. | - | - |
| 8:2 FTSA | FTSA | 0.01 | 25 | 18 | 28% | 0.09 | 0.21 | 16 | n.d. | - | - |
| PFHxS | long PFSA | 0.01 | 22 | 48 | 23% | 0.03 | 0.10 | 48 | 23% | 0.02 | 0.03 |
| 4:2 FTSA | FTSA | 0.01 | 25 | 18 | 22% | 0.01 | 0.01 | 16 | n.d. | - | - |
| PFDS | long PFSA | 0.02 | 27 | 48 | 10% | 0.07 | 0.17 | 48 | n.d. | - | - |
| PFOA | long PFCA | 0.01 | 24 | 48 | 6% | 0.03 | 0.06 | 48 | 40% | 0.07 | 0.28 |
| 6:2 FTSA | FTSA | 0.01 | 25 | 18 | 6% | 0.01 | 0.01 | 16 | n.d. | - | - |
| PFHxA | short PFCA | 0.02 | 24 | 48 | 2% | 0.11 | 0.11 | 48 | n.d. | - | - |
| PFPeA | short PFCA | 0.03 | 25 | 48 | 2% | 0.05 | 0.05 | 0 | n.a. | - | - |
| N-EtFOSA | PreFOS | 0.15 | 30 | 18 | n.d. | - | - | 16 | n.d. | - | - |
| PFBA | short PFCA | 0.04 | 35 | 48 | n.d. | - | - | 16 | n.d. | - | - |
| PFBS | short PFSA | 0.03 | 23 | 48 | n.d. | - | - | 16 | n.d. | - | - |
| PFHxDA | long PFCA | 0.15 | 26 | 48 | n.d. | - | - | 16 | n.d. | - | - |
| PFHpA | long PFCA | 0.01 | 33 | 48 | n.d. | - | - | 48 | 8% | 0.02 | 0.02 |
| PFODA | long PFCA | 0.11 | 31 | 48 | n.d. | - | - | 16 | n.d. | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Junttila, V.; Vähä, E.; Perkola, N.; Räike, A.; Siimes, K.; Mehtonen, J.; Kankaanpää, H.; Mannio, J. PFASs in Finnish Rivers and Fish and the Loading of PFASs to the Baltic Sea. Water 2019, 11, 870. https://doi.org/10.3390/w11040870
Junttila V, Vähä E, Perkola N, Räike A, Siimes K, Mehtonen J, Kankaanpää H, Mannio J. PFASs in Finnish Rivers and Fish and the Loading of PFASs to the Baltic Sea. Water. 2019; 11(4):870. https://doi.org/10.3390/w11040870
Chicago/Turabian StyleJunttila, Ville, Emmi Vähä, Noora Perkola, Antti Räike, Katri Siimes, Jukka Mehtonen, Harri Kankaanpää, and Jaakko Mannio. 2019. "PFASs in Finnish Rivers and Fish and the Loading of PFASs to the Baltic Sea" Water 11, no. 4: 870. https://doi.org/10.3390/w11040870
APA StyleJunttila, V., Vähä, E., Perkola, N., Räike, A., Siimes, K., Mehtonen, J., Kankaanpää, H., & Mannio, J. (2019). PFASs in Finnish Rivers and Fish and the Loading of PFASs to the Baltic Sea. Water, 11(4), 870. https://doi.org/10.3390/w11040870





