Long-Term Water Quality Changes as a Result of a Sustainable Restoration—A Case Study of Dimictic Lake Durowskie
Abstract
1. Introduction
2. Materials and Methods
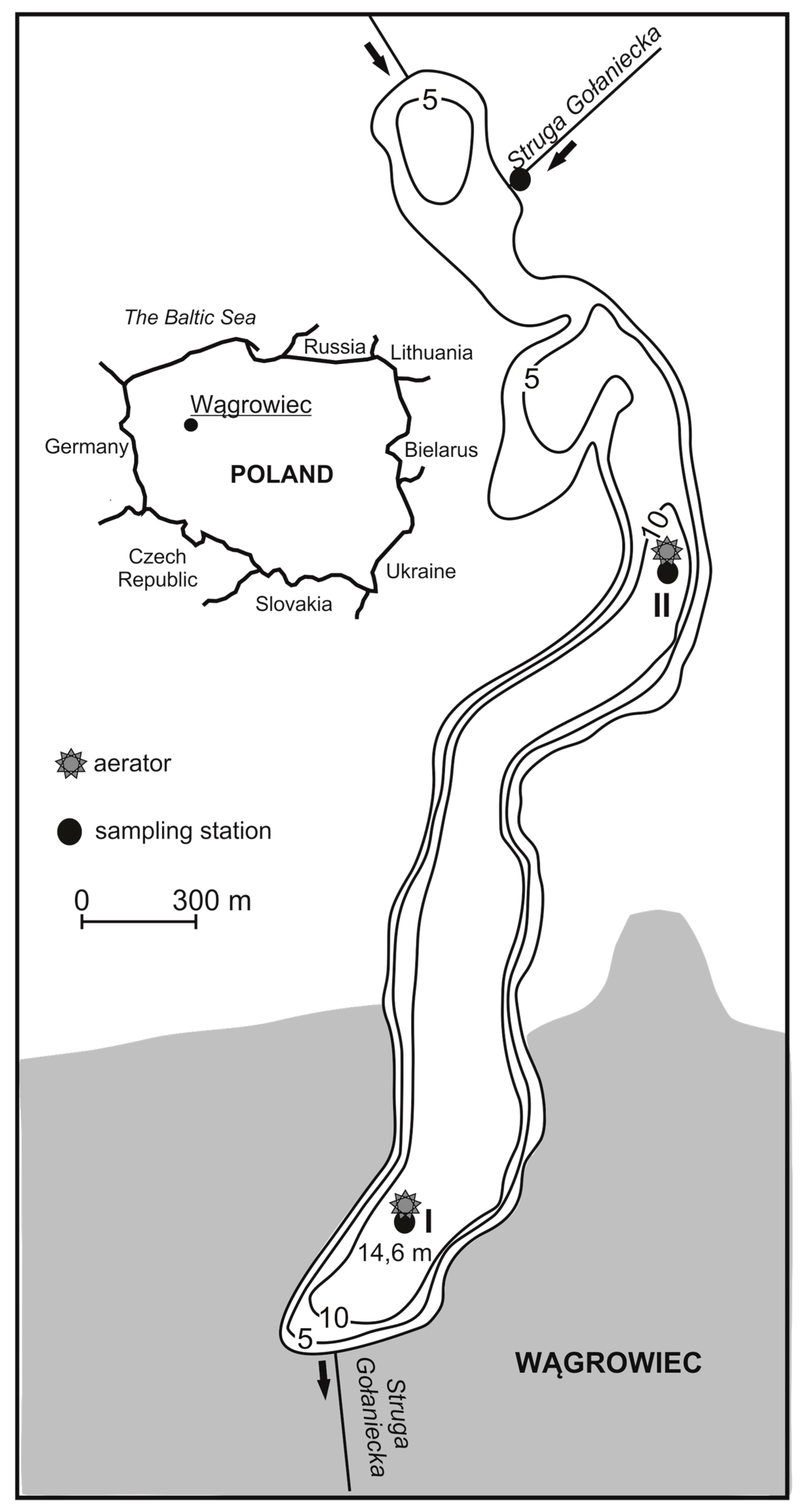
2.1. Study Site
2.2. Methods
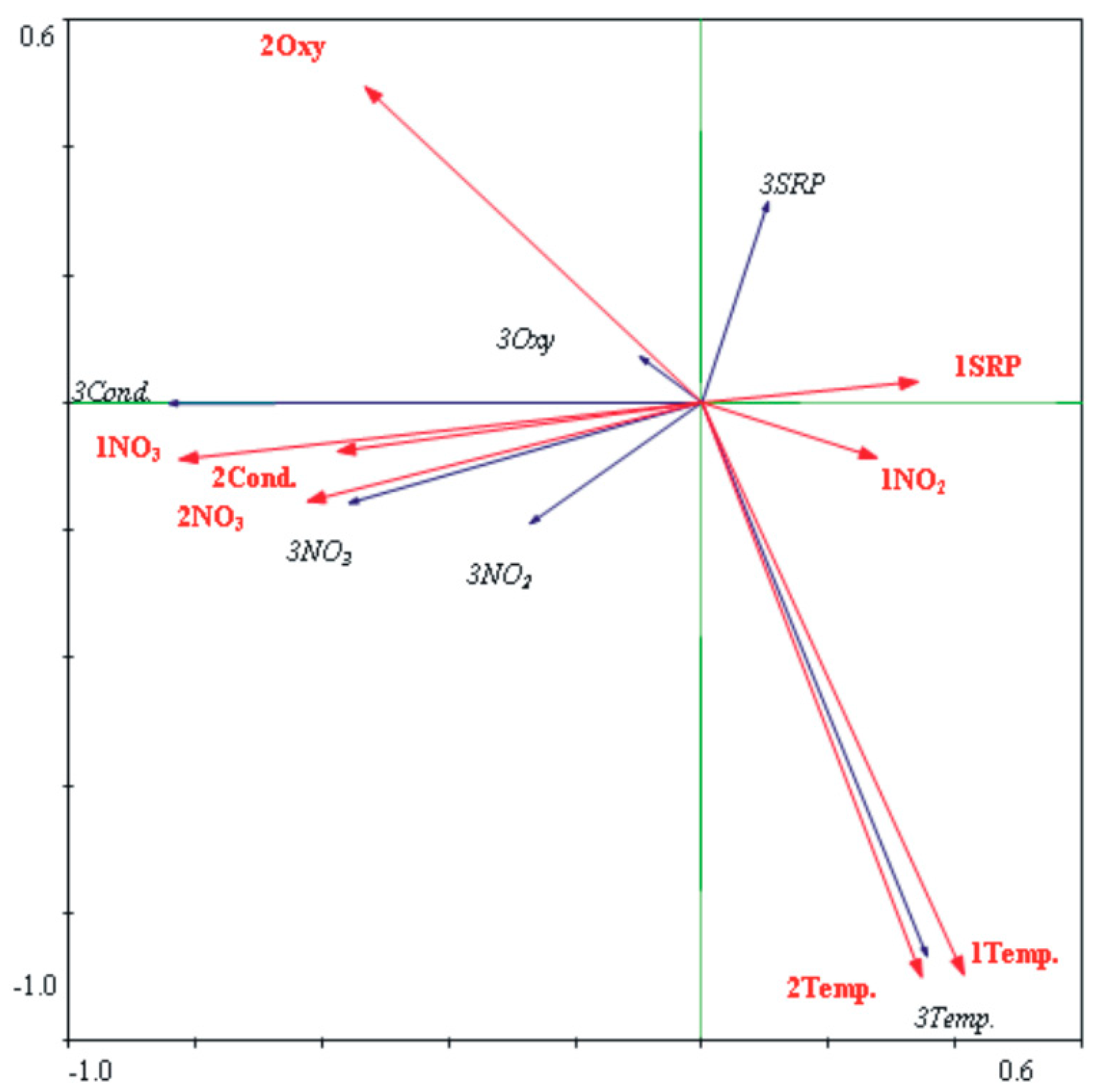
3. Results
3.1. The Quality of Durowskie Lake Waters
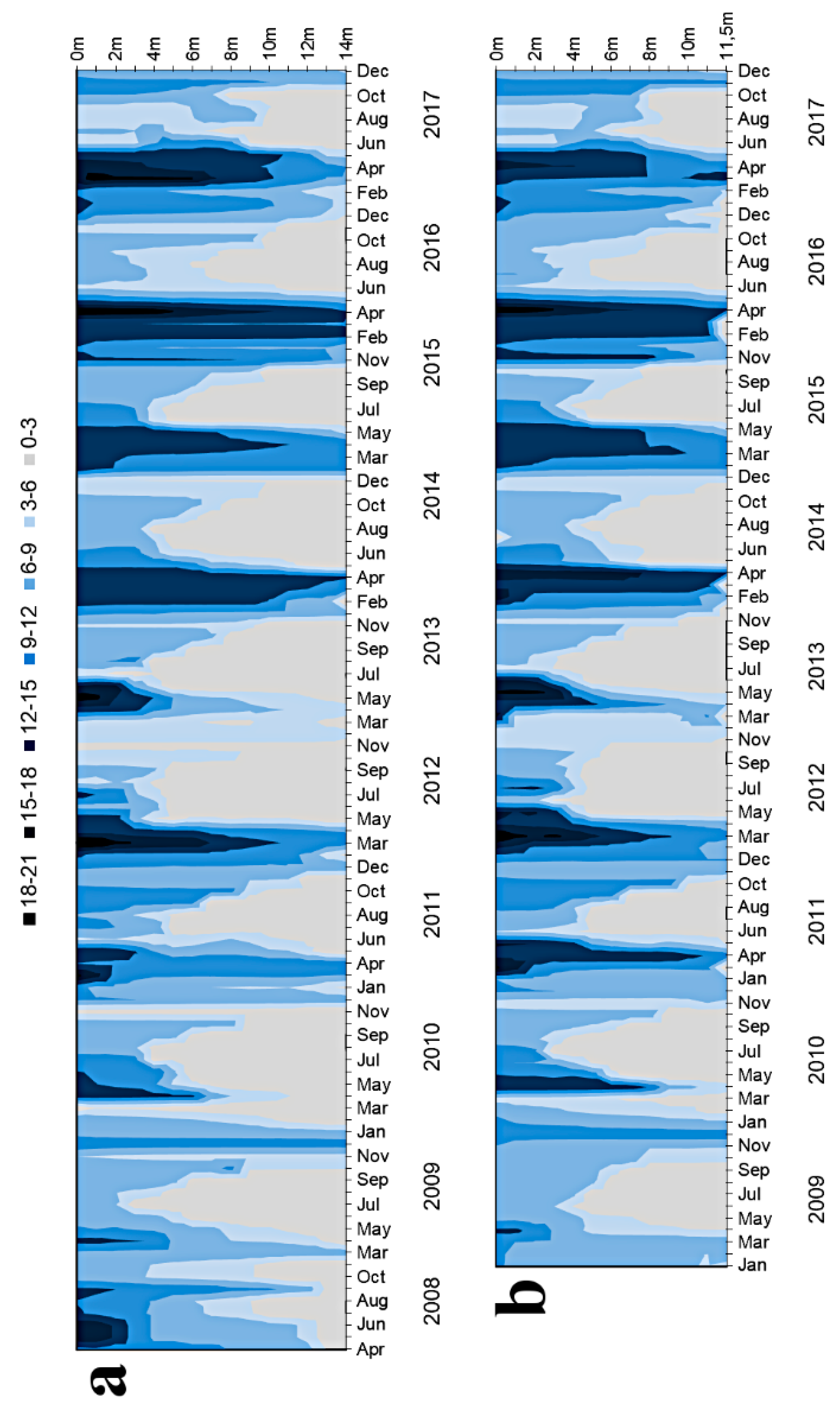
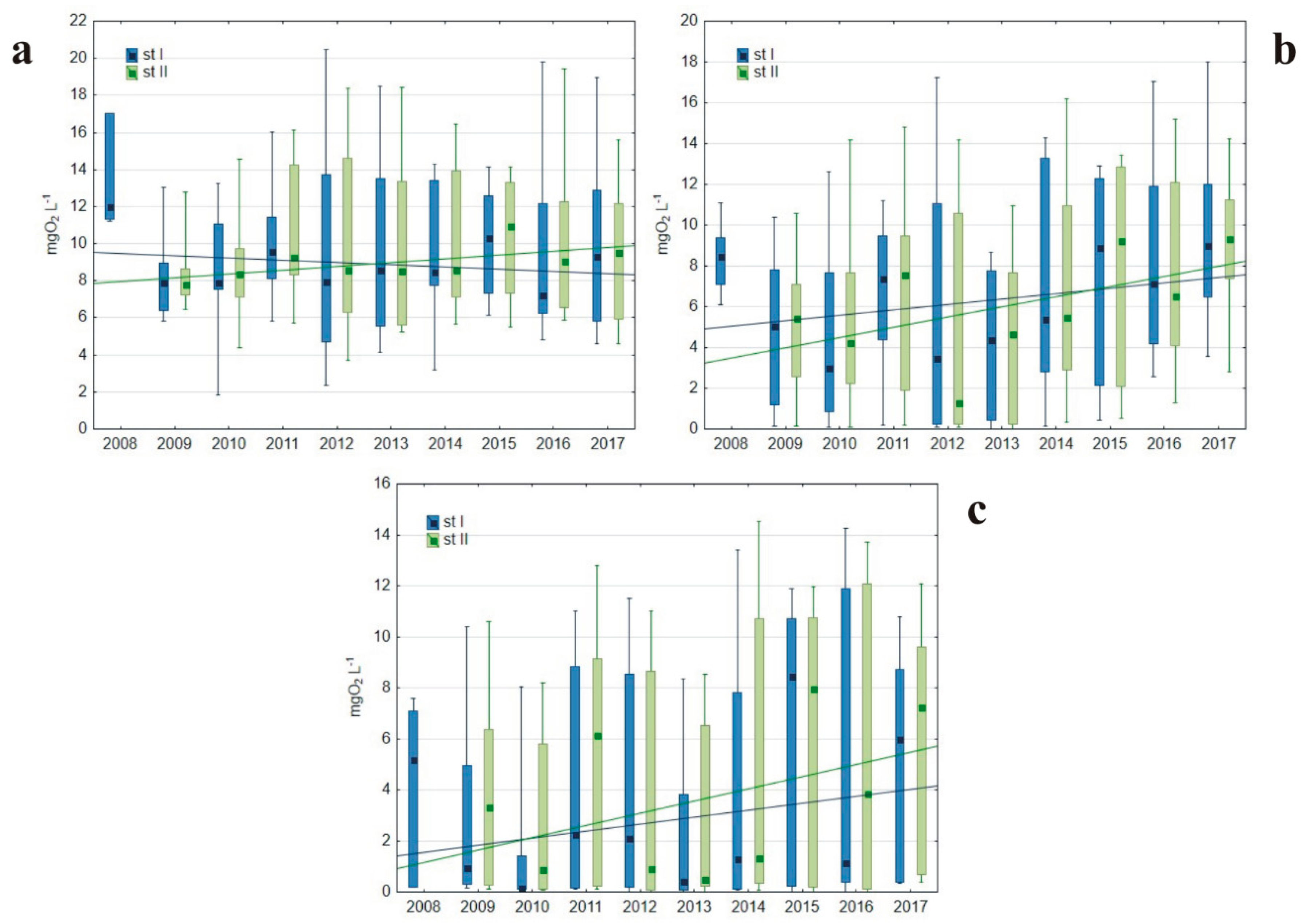
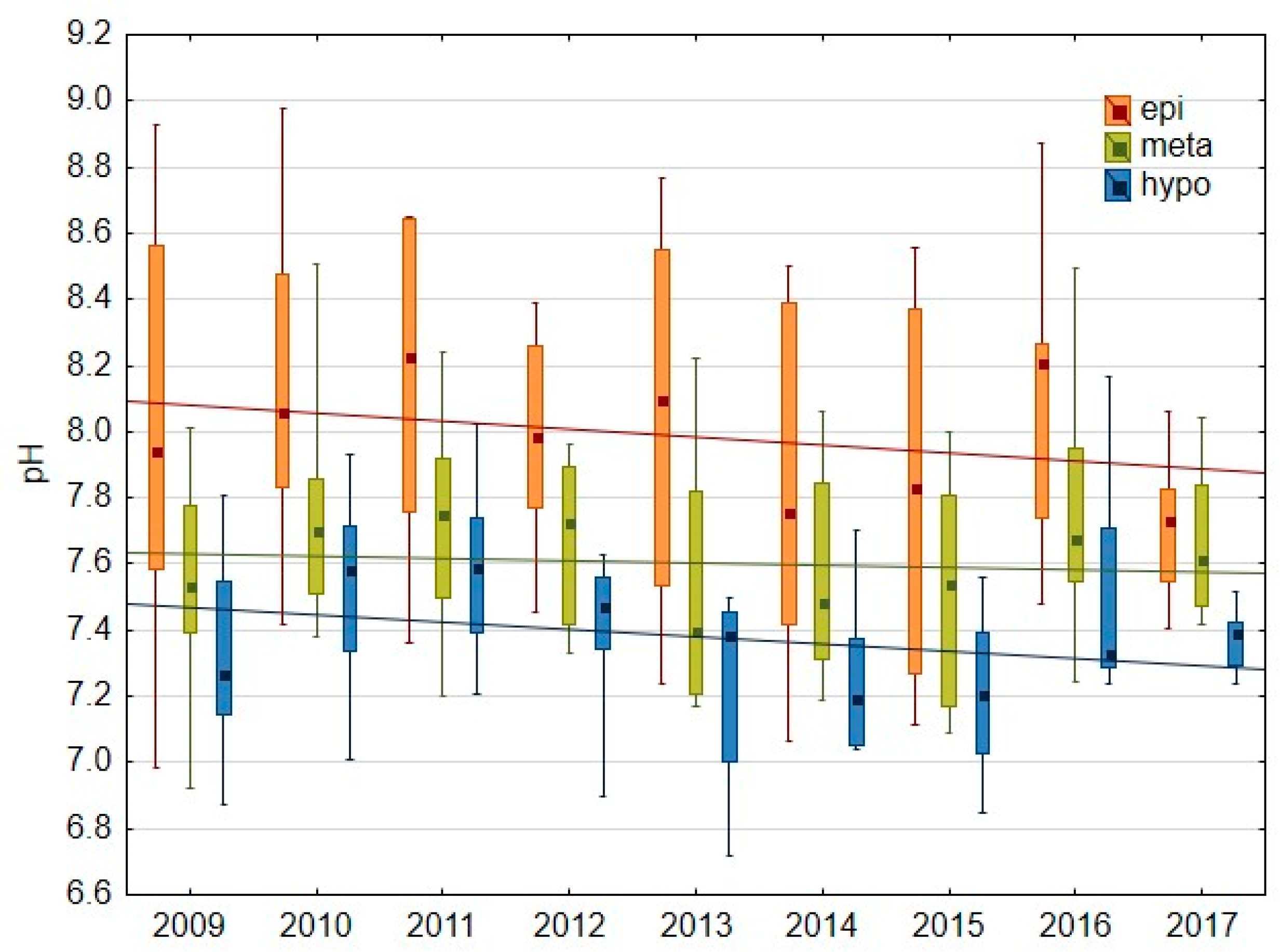
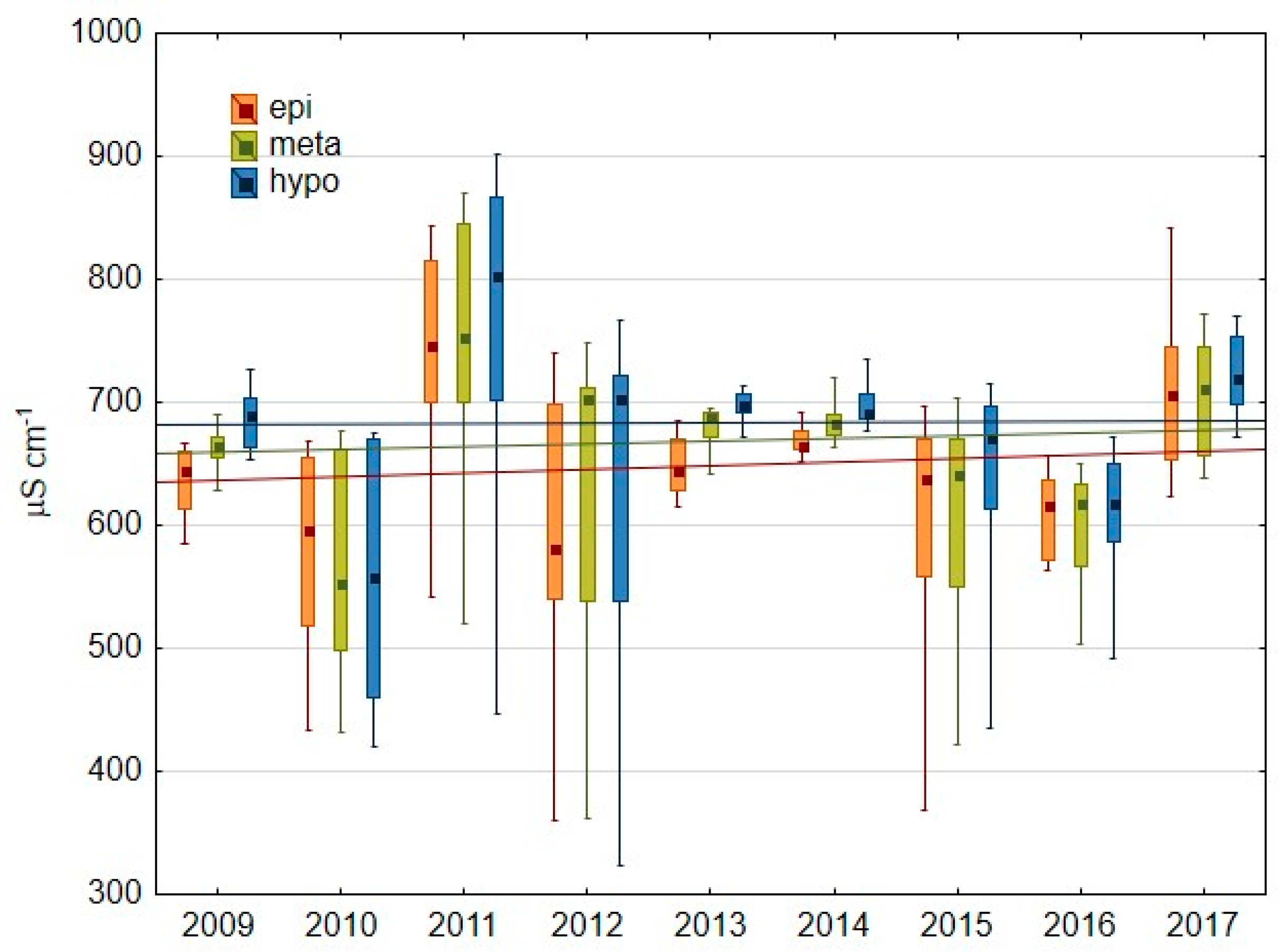
3.1.1. Basic Water Characteristics
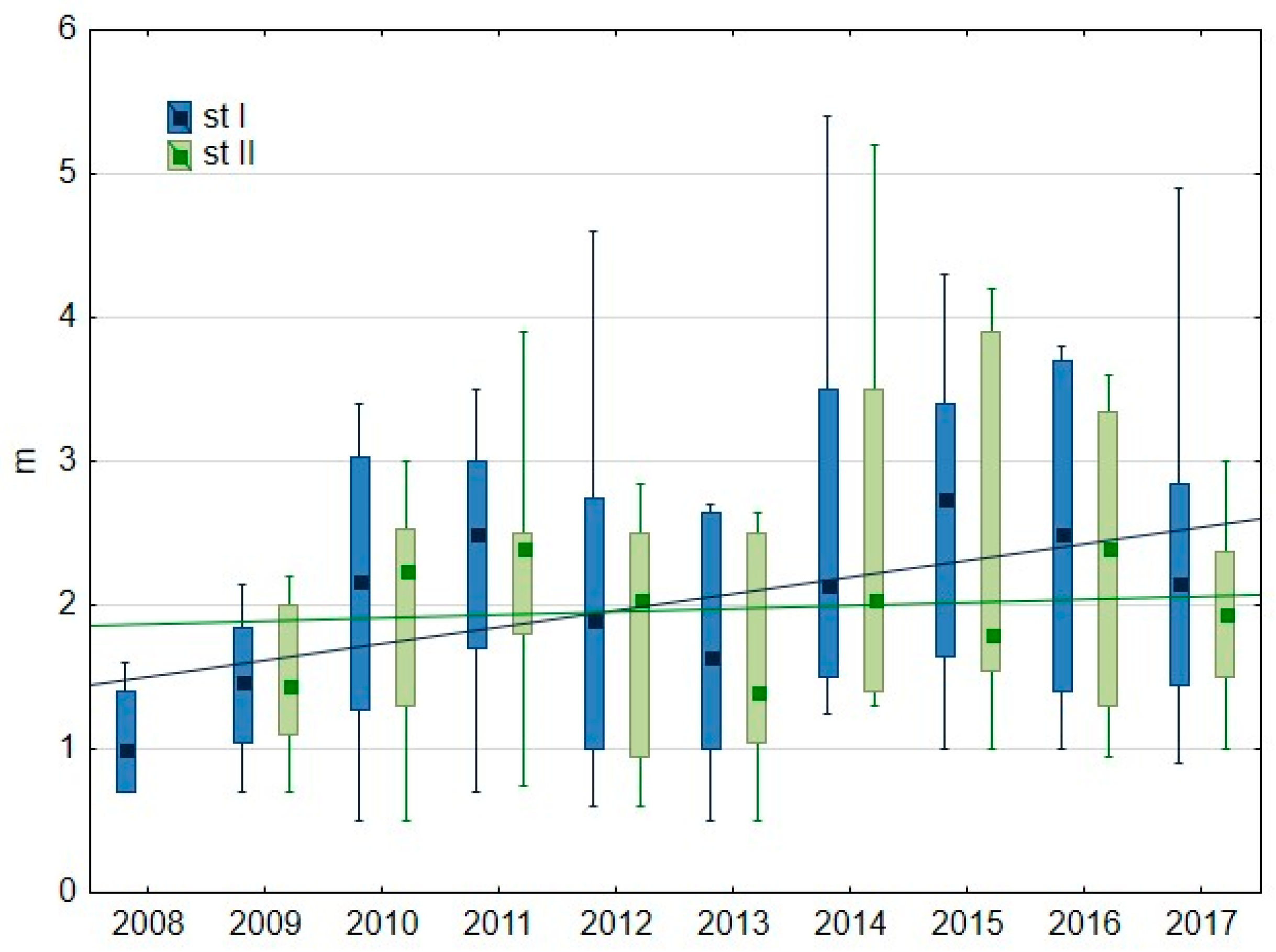
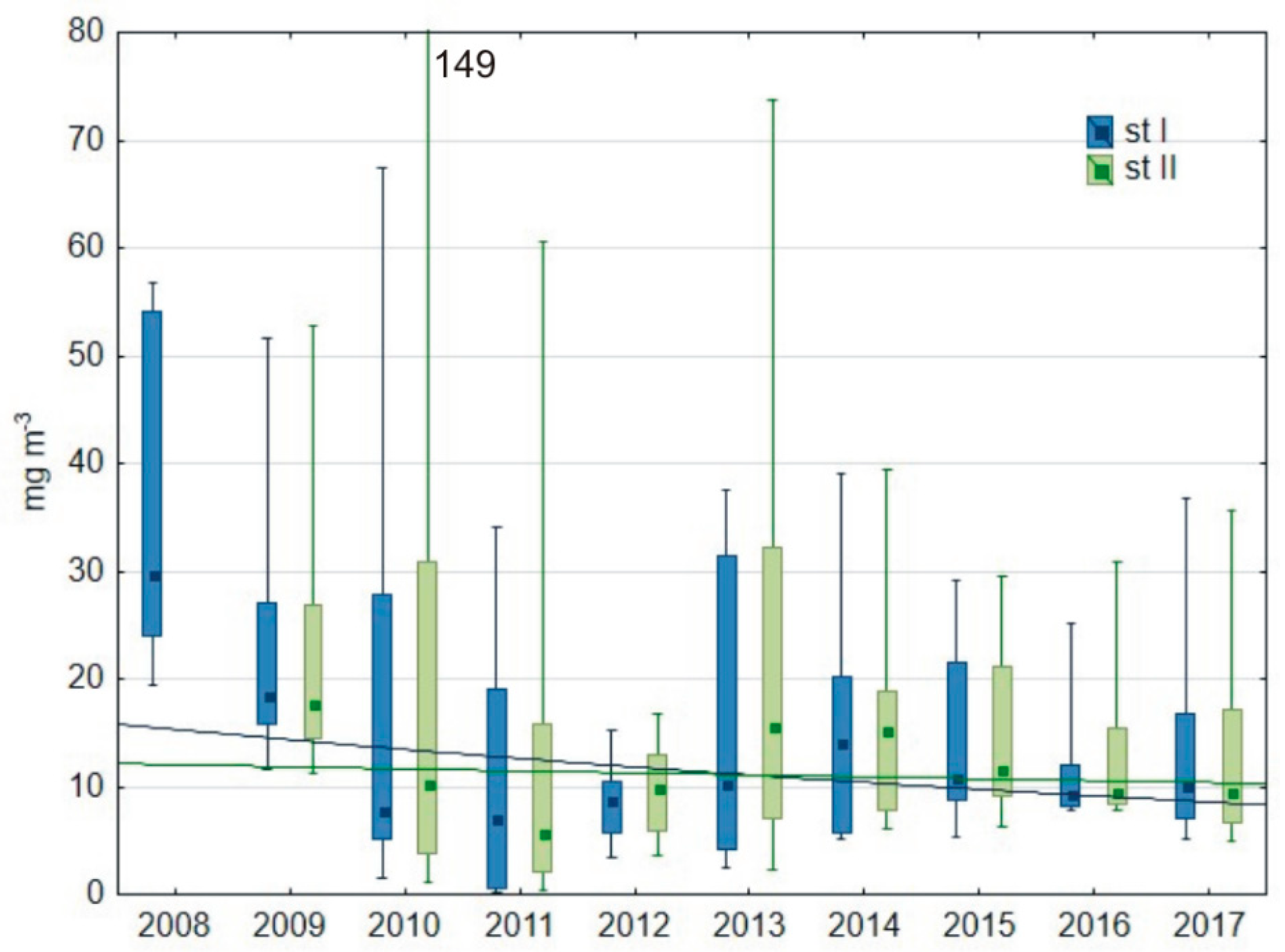
3.1.2. Water Transparency and Chlorophyll-a Content
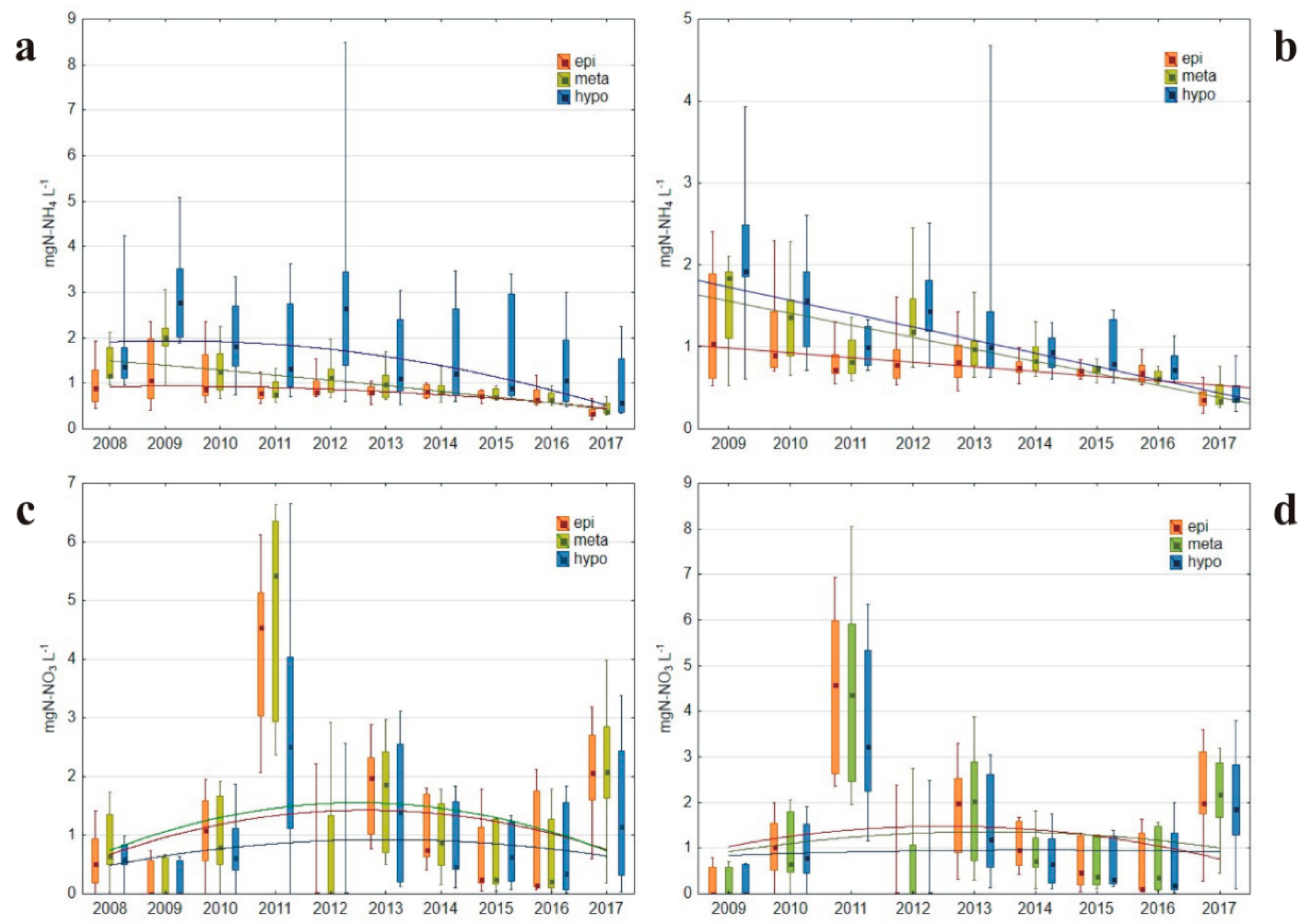
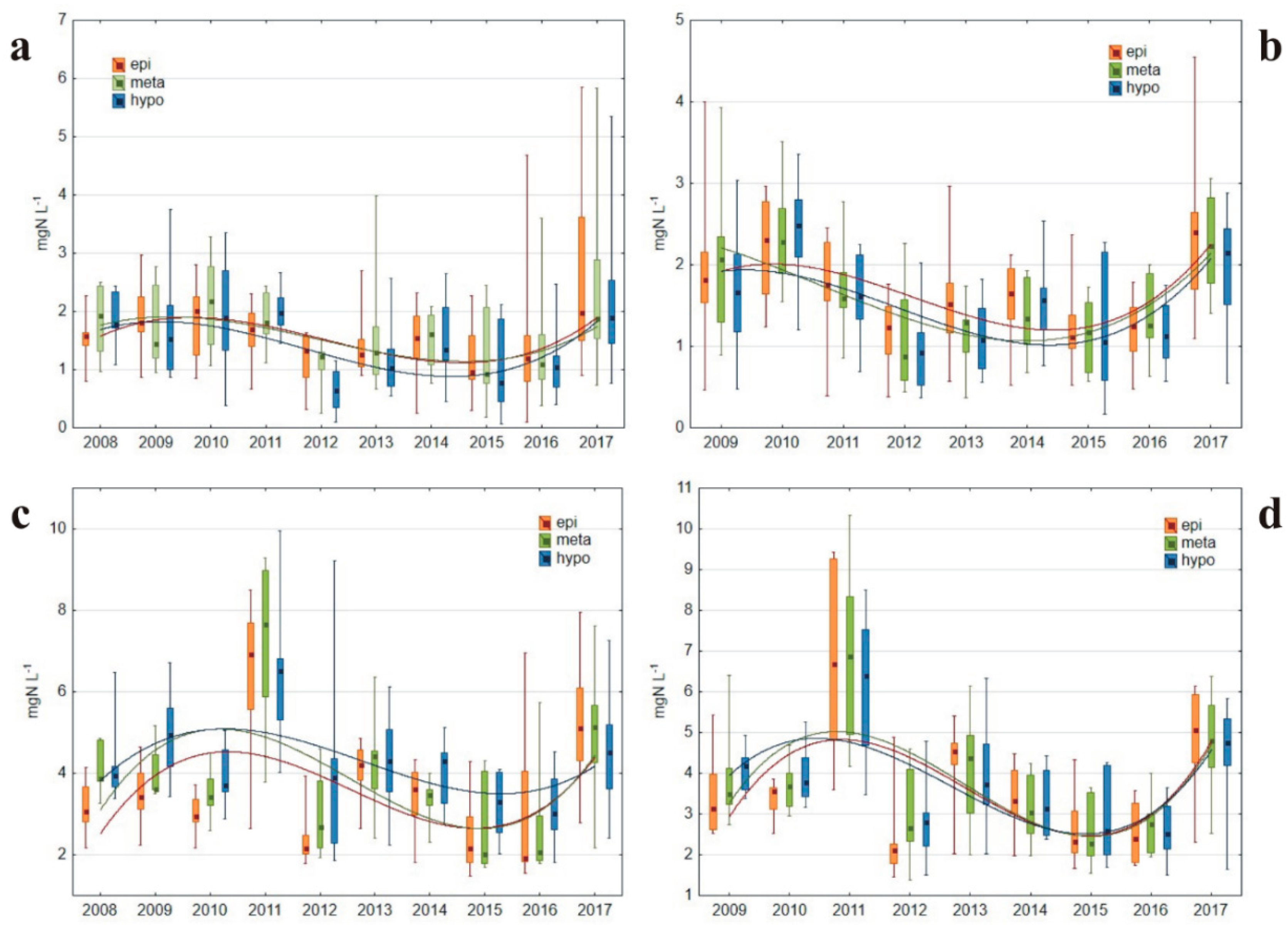
3.1.3. Nitrogen Concentrations
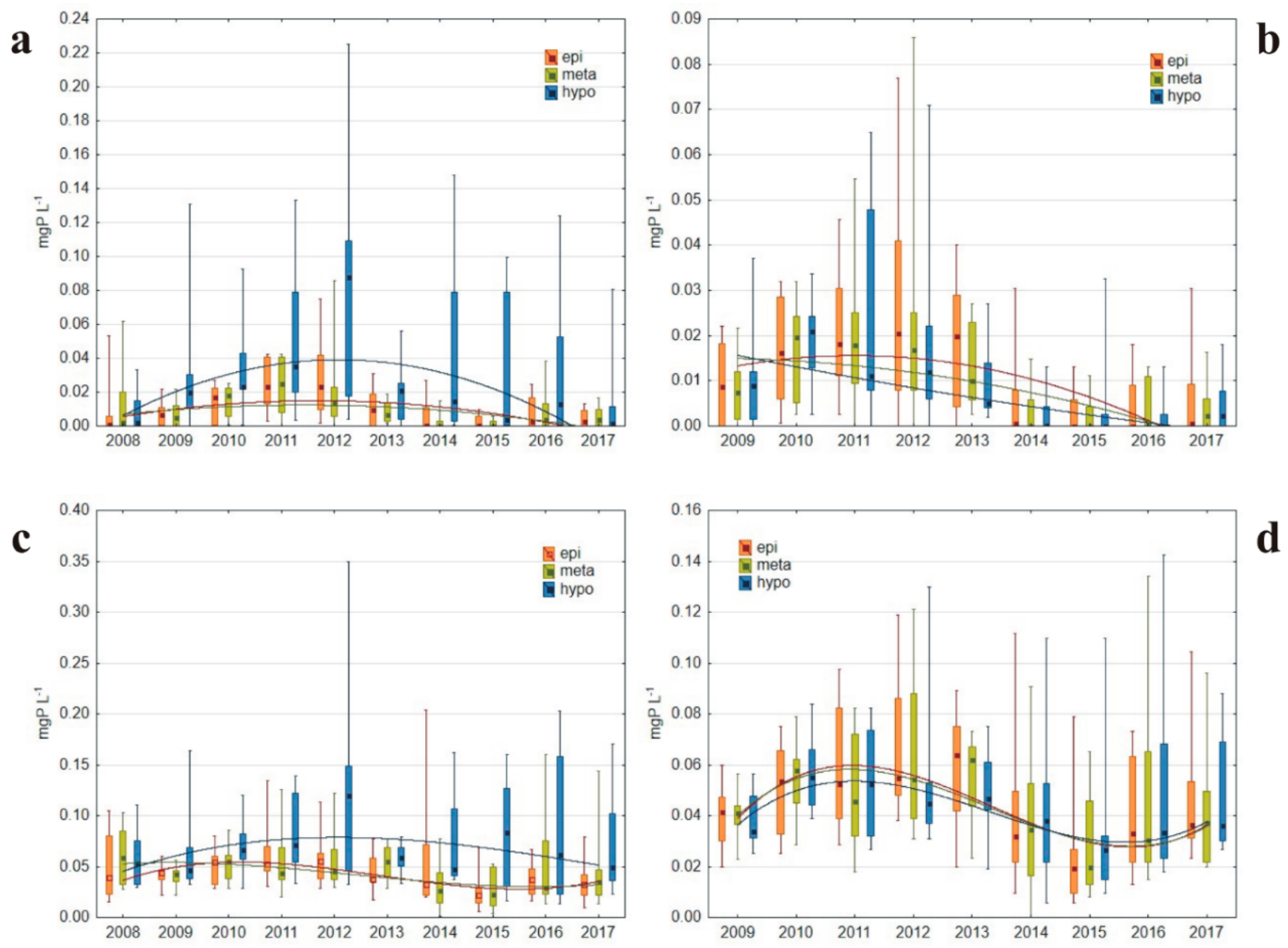
3.1.4. Phosphorus Concentrations
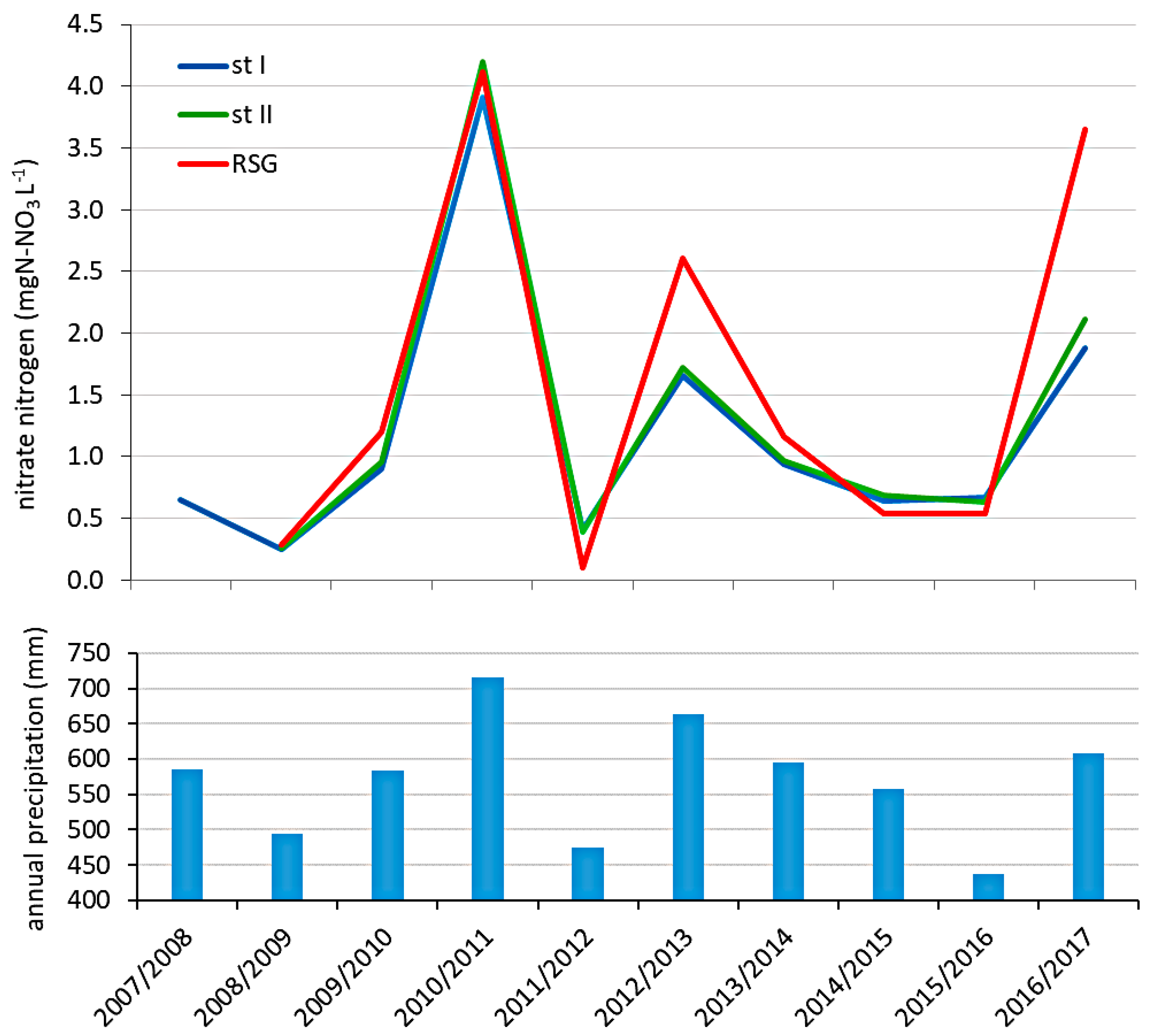
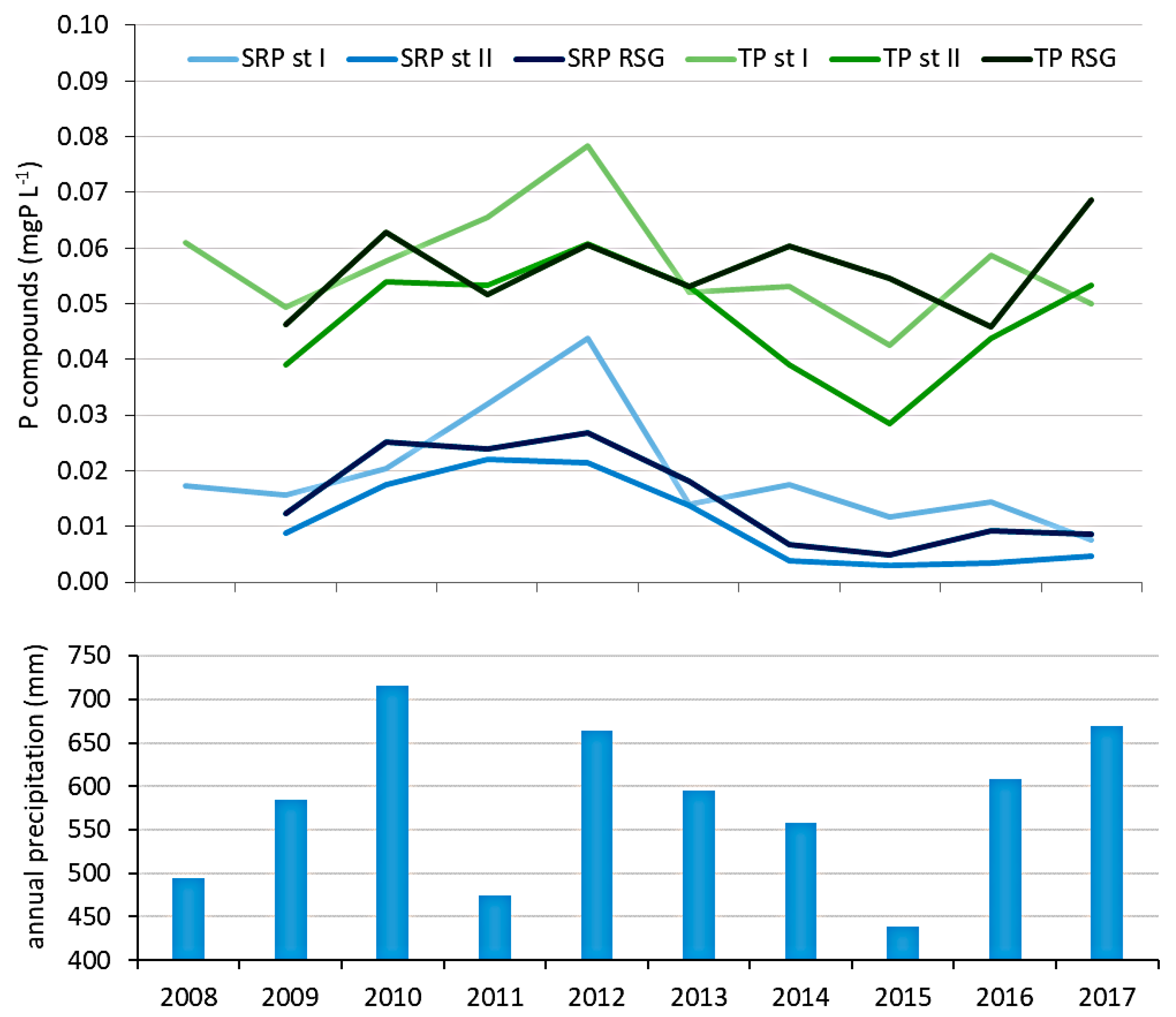
3.2. The Quality of River Struga Gołaniecka Waters
4. Discussion
4.1. Oxygen Conditions As a Result of Hypolimnetic Aeration
4.2. Nitrogen Transformations in Relation to Oxygen and Temperature
4.3. Phosphorus–Oxygen–Chlorophyll-a Interactions
4.4. The Influence of River Struga Gołaniecka on Lake Waters
4.5. Supportive Role of Biomanipulation in Water Quality Improvement
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- EU Directive 2000/60/EC of the European parliament and of the council of 23 October 2000 establishing a framework for community action in the field of water policy. Off. J. Eur. Communities 2000, L327, 1–72.
- European Environment Agency. European Waters. Assessment of Status and Pressures 2018; Report no 7/2018; European Environment Agency: Luxembourg, 2018. [Google Scholar] [CrossRef]
- Verdonschot, P.F.M.; Spears, B.M.; Feld, C.K.; Brucet, S.; Keizler-Vlek, H.; Borja, A.; Elliott, M.; Kernan, M.; Johnson, R.K. A comparative review of recovery processes in rivers, lakes, estuarine and coastal waters. Hydrobiologia 2013, 704, 453–474. [Google Scholar] [CrossRef]
- Annadotter, H.; Cronberg, G.; Aagren, R.; Lundstedt, B.; Nilsson, P.-A.; Ströbeck, S. Multiple techniques for lake restoration. Hydrobiologia 1999, 395/396, 77–85. [Google Scholar] [CrossRef]
- Jeppesen, E.; Sammalkorpi, I. Lakes. In Handbook of Ecological Restoration, Vol. 2, Restoration in Practice; Perrow, M.R., Davy, A.J., Eds.; Cambridge University Press: Cambridge, UK, 2002; pp. 297–324. [Google Scholar]
- Gołdyn, R.; Podsiadłowski, S.; Dondajewska, R.; Kozak, A. The sustainable restoration of lakes—towards the challenges of the Water Framework Directive. Ecohydrol. Hydrobiol. 2014, 14, 68–74. [Google Scholar] [CrossRef]
- Dondajewska, R.; Kozak, A.; Kowalczewska-Madura, K.; Budzyńska, A.; Gołdyn, R.; Podsiadłowski, S.; Tomkowiak, A. The response of a shallow hypertrophic lake to innovative restoration measures—Uzarzewskie lake case study. Ecol. Eng. 2018, 121, 72–82. [Google Scholar] [CrossRef]
- Kowalczewska-Madura, K.; Dondajewska, R.; Gołdyn, R.; Podsiadłowski, S. The influence of restoration measures on phosphorus internal loading from the sediments of a hypereutrophic lake. Environ. Sci. Pollut. Res. 2017, 24, 14417–14429. [Google Scholar] [CrossRef]
- Kowalczewska-Madura, K.; Dondajewska, R.; Gołdyn, R.; Kozak, A.; Messyasz, B. Internal phosphorus loading from the bottom sediments of the dimictic lake during its sustainable restoration. Water Air Soil Pollut. 2018, 229, 280. [Google Scholar] [CrossRef]
- Kowalczewska-Madura, K.; Dondajewska, R.; Gołdyn, R. Internal phosphorus loading in eutrophic lakes in Western Poland. In The Handbook of Environmental Chemistry. Polish River Basins and Lakes—Part I: Hydrology and Hydrochemistry; Korzeniewska, E., Harnisz, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; in press. [Google Scholar]
- Dondajewska, R.; Gołdyn, R.; Kowalczewska-Madura, K.; Kozak, A.; Romanowicz-Brzozowska, W.; Rosińska, J.; Budzyńska, A.; Podsiadłowski, S. Hypertrophic lakes and the results of their restoration in Western Poland. In The Handbook of Environmental Chemistry. Polish River Basins and Lakes—Part II: Biological Status of Water Management; Korzeniewska, E., Harnisz, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; in press. [Google Scholar]
- Jeppesen, E.; Søndergaard, M.; Liu, Z. Lake restoration. In Routledge Handbook of Ecological and Environmental Restoration; Allison, S.K., Murphy, S.D., Eds.; Routlege: London, UK; New York, NY, USA, 2017; pp. 226–242. [Google Scholar]
- O’Sullivan, P.E.; Reynolds, C.S. Lakes Handbook: Lake Restoration and Rehabilitation; Blackwell Publishing: Malden, MA, USA, 2005; Volume 2. [Google Scholar]
- Carpenter, S.R.; Cottingham, K.L. Resilience and restoration of lakes. Ecol. Soc. 1997, 1, 2. [Google Scholar] [CrossRef]
- Gunderson, L.H. Ecological resilience—In theory and application. Annu. Rev. Ecol. Syst. 2000, 31, 425–439. [Google Scholar] [CrossRef]
- Gołdyn, R.; Messyasz, B.; Domek, P.; Windhorst, W.; Hugenschmidt, C.; Nicoara, M.; Plavan, G. The response of Lake Durowskie ecosystem to restoration measures. Carpath. J. Earth Environ. 2013, 8, 43–48. [Google Scholar]
- Podsiadłowski, S.; Osuch, E.; Przybył, J.; Osuch, A.; Buchwald, T. Pulverizing aerator in the process of lake restoration. Ecol. Eng. 2018, 121, 99–103. [Google Scholar] [CrossRef]
- Elbanowska, H.; Zerbe, J.; Siepak, J. Physico-Chemical Water Analyses; AMU Press: Poznań, Poland, 1999. (In Polish) [Google Scholar]
- Wetzel, R.G.; Likens, G.E. Limnological Analyses, 2nd ed.; Springer-Verlag Inc.: New York, NY, USA, 1991. [Google Scholar]
- McQueen, D.J.; Post, J.R.; Mills, E.L. Trophic relationships in freshwater pelagic ecosystems. Can. J. Fish. Aquat. Sci. 1986, 43, 1571–1581. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Kitchell, J.F.; Hodgson, J.R. Cascading trophic interactions and lake productivity. Bioscience 1985, 35, 634–639. [Google Scholar] [CrossRef]
- Taylor, J.M.; Vanni, M.J.; Flecker, A.S. Top-down and bottom-up interactions in freshwater ecosystems: Emerging complexities. In Trophic Ecology: Bottom-Up and Top-Down Interactions Across Aquatic and Terrestrial Systems; Hanley, T.C., La Pierre, K.J., Eds.; Cambridge University Press: Cambridge, UK, 2015; pp. 55–85. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Lauridsen, T.L.; Davidson, T.A.; Liu, Z.; Mazzeo, N.; Trochine, C.; Özkan, K.; Jensen, H.S.; Trolle, D.; et al. Biomanipulation as a restoration tool to combat eutrophication: Recent advances and future challenges. Adv. Ecol. Res. 2012, 47, 411–488. [Google Scholar] [CrossRef]
- Søndergaard, M.; Jensen, J.P.; Jeppesen, E. Role of sediments and internal loading of phosphorus in shallow lakes. Hydrobiologia 2003, 506–509, 135–145. [Google Scholar] [CrossRef]
- Beutel, M.W. Inhibition of ammonia release from anoxic profundal sediments in lakes using hypolimnetic oxygenation. Ecol. Eng. 2006, 28, 271–279. [Google Scholar] [CrossRef]
- Singleton, V.L.; Little, J.C. Designing hypolimnetic aeration and oxygenation systems—A review. Environ. Sci. Technol. 2006, 40, 7512–7520. [Google Scholar] [CrossRef]
- Ekau, W.; Auel, H.; Pörtner, H.-O.; Gilbert, D. Impacts of hypoxia on the structure and processes in pelagic communities (zooplankton, macro-invertebrates and fish). Biogeosciences 2010, 7, 1669–1699. [Google Scholar] [CrossRef]
- Gächter, R.; Müller, B. Why the phosphorus retention of lakes does not necessarily depend on the oxygen supply to their sediment surface. Limnol. Oceanogr. 2003, 48, 929–933. [Google Scholar] [CrossRef]
- Liboriussen, L.; Søndergaard, M.; Jeppesen, E.; Thorsgaard, I.; Grünfeld, S.; Jakobsen, T.S.; Hansen, K. Effects of hypolimnetic oxygenation on water quality: Results from five Danish lakes. Hydrobiologia 2009, 625, 157–172. [Google Scholar] [CrossRef]
- Siwek, H.; Włodarczyk, M.; Czerniawski, R. Trophic state and oxygen conditions of waters aerated with pulverising aerator: The results from seven lakes in Poland. Water 2019, 10, 219. [Google Scholar] [CrossRef]
- Holmroos, H.; Horppila, J.; Laakso, S.; Niemistö, J.; Hietanen, S. Aeration-induced changes in temperature and nitrogen dynamics in a dimictic lake. J. Environ. Qual. 2016, 45, 1359–1366. [Google Scholar] [CrossRef]
- Grochowska, J.; Gawrońska, H. Restoration effectiveness of a degraded lake using multi-annual artificial aeration. Pol. J. Environ. Stud. 2004, 13, 671–691. [Google Scholar]
- Rosińska, J.; Kozak, A.; Dondajewska, R.; Gołdyn, R. Cyanobacterial blooms before and during the restoration process of a shallow urban lake. J. Environ. Manag. 2017, 198, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Rosińska, J.; Kozak, A.; Dondajewska, R.; Kowalczewska-Madura, K.; Gołdyn, R. Water quality response to sustainable restoration measures—Case study of urban Swarzędzkie Lake. Ecol. Indic. 2018, 84, 437–449. [Google Scholar] [CrossRef]
- Burgin, A.J.; Hamilton, S.K. Have we overemphasized the role of denitrification in aquatic ecosystems? A review of nitrate removal pathways? Front. Ecol. Environ. 2007, 5, 89–96. [Google Scholar] [CrossRef]
- Nizzoli, D.; Carraro, E.; Nigro, V.; Viaroli, P. Effect of organic enrichment and thermal regime on denitrification and dissimilatory nitrate reduction to ammonium (DNRA) in hypolimnetic sediments of two lowland lakes. Water Res. 2010, 44, 2715–2724. [Google Scholar] [CrossRef] [PubMed]
- Op den Camp, H.J.M.; Jetten, M.S.M.; Strous, M. Annamox. In Biology of the Nitrogen Cycle; Bothe, H., Ferguson, S.J., Newton, W.E., Eds.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 245–262. [Google Scholar]
- Prepas, E.E.; Burke, J.M. Effects of hypolimnetic oxygenation on water quality in Amiska Lake, Alberta, a deep stratified lake with high internal phosphorus loading rates. Can. J. Fish. Aquat. Sci. 1997, 54, 2111–2120. [Google Scholar] [CrossRef]
- Søndergaard, M.; Wolter, K.D.; Ripl, E. Chemical treatment water and sediments with special references to lakes. In Handbook of Ecological Restoration. Vol. 1. Principles of Restoration; Perrow, M.R., Davy, A.J., Eds.; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Cooke, G.D.; Welch, E.B.; Martin, A.B.; Fulmer, D.G.; Hyde, J.B.; Schrieve, G.D. Effectiveness of Al, Ca and Fe salts to control of internal loading in shallow and deep lakes. Hydrobiologia 1993, 253, 323–335. [Google Scholar] [CrossRef]
- Boers, P.; Van der Does, J.; Quaak, M.; Van der Vlugt, J. Phosphorus fixation with iron (III) chloride: A new method to combat internal phosphorus loading in shallow lakes? Arch. Hydrobiol. 1994, 129, 339–351. [Google Scholar]
- Jaeger, D. Effects of hypolimnetic water aeration and iron-phosphate precipitation on the trophic level of Lake Krupunder. Hydrobiologia 1994, 275–276, 433–444. [Google Scholar] [CrossRef]
- Deppe, T.; Benndorf, J. Phosphorus reduction in shallow hyper-eutrophic reservoir by in-lake dosage of ferrous iron. Water Res. 2002, 36, 4525–4534. [Google Scholar] [CrossRef]
- Hupfer, M.; Lewandowski, J. Oxygen controls the phosphorus release from lake sediments—A long-lasting paradigm in limnology. Int. Rev. Hydrobiol. 2008, 93, 415–432. [Google Scholar] [CrossRef]
- Nygrén, N.A.; Tapio, P.; Horppila, J. Will the oxygen-phosphorus paradigm persist?—Expert views of the future of management and restoration of eutrophic lakes. Environ. Manag. 2007, 60, 947–960. [Google Scholar] [CrossRef] [PubMed]
- Pearl, H.W.; Hall, N.S.; Calandrino, E.S. Controlling harmful cyanobacterial blooms in world experiencing anthropogenic and climate-induced change. Sci. Total Environ. 2011, 409, 1739–1745. [Google Scholar] [CrossRef] [PubMed]
- Napiórkowska-Krzebietke, A.; Dunalska, J.A.; Zębek, E. Taxa-specific eco-sensitivity in relation to phytoplankton bloom stability and ecologically relevant lake state. Acta Oecol. 2017, 81, 10–21. [Google Scholar] [CrossRef]
- Mantzouki, E.; Lürling, M.; Fastner, J.; de Senerpont Domis, L.; Wilk-Woźniak, E.; Koreivienė, J.; Seelen, L.; Teurlincx, S.; Verstijnen, Y.; Krztoń, W.; et al. Temperature effects explain continental scale distribution of cyanobacterial toxins. Toxins 2018, 10, 156. [Google Scholar] [CrossRef] [PubMed]
- Tammeorg, O.; Möls, T.; Niemistö, J.; Holmroos, H.; Horppila, J. The actual role of oxygen deficit in the linkage of the water quality and benthic phosphorus release: Potential implications for lake restoration. Sci. Total Environ. 2017, 599–600, 732–738. [Google Scholar] [CrossRef]
- Saunders, D.L.; Kalff, J. Nitrogen retention in wetlands, lakes and rivers. Hydrobiologia 2001, 443, 205–212. [Google Scholar] [CrossRef]
- Pűtz, K.; Benndorf, J. The importance of pre-reservoirs for the control of eutrophication of reservoirs. Water Sci. Technol. 1998, 37, 317–324. [Google Scholar] [CrossRef]
- Straškraba, M.; Tundisi, J.G.; Duncan, A. State-of-the-art of reservoir limnology and water quality management. Comp. Reserv. Limnol. Water Qual. Manag. 1993, 77, 213–288. [Google Scholar]
| Range of Water Layers (m) | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Station I | epi temp. | 0–4 19.4 | 0–4 20.3 | 0–3 20.9 | 0–3 20.8 | 0–2 22.5 | 0–3 21.3 | 0–3 20.9 | 0–3 20.0 | 0–3 23.1 | 0–3 20.9 |
| meta temp. | 5–8 11.6 | 5–8 13.0 | 4–6 13.9 | 4–7 13.6 | 3–6 16.4 | 4–7 12.5 | 4–8 13.1 | 4–8 14.0 | 4–7 14.1 | 4–7 12.9 | |
| hypo temp. | 9–14 8.1 | 9–14 6.3 | 7–14 6.8 | 8–14 6.6 | 7–14 8.2 | 8–14 5.9 | 9–14 6.9 | 9–14 7.5 | 8–14 6.9 | 8–14 7.7 | |
| Station II | epi temp. | na | 0–4 20.9 | 0–2 20.7 | 0–3 21.2 | 0–2 23.4 | 0–2 21.7 | 0–3 20.5 | 0–3 19.6 | 0–3 23.1 | 0–4 20.2 |
| meta temp. | na | 5–7 13.4 | 3–6 14.6 | 4–6 15.3 | 3–6 16.3 | 3–6 15.4 | 4–6 14.1 | 4–6 15.3 | 4–6 14.3 | 5–6 12.2 | |
| hypo temp. | na | 8–12 7.0 | 7–12 7.6 | 7–12 7.7 | 7–12 8.4 | 7–12 7.1 | 7–12 8.1 | 7–12 8.5 | 7–12 7.5 | 7–12 8.5 | |
| Mean air temperature 1 | 18.9 | 18.3 | 19.2 | 18.7 | 18.6 | 19.2 | 18.9 | 19.6 | 18.9 | 18.7 | |
| Parameter | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 |
|---|---|---|---|---|---|---|---|---|---|
| temperature (°C) | 13.3 22.1 | 9.8 27.1 | 7.8 22.2 | 10.8 25.6 | 15.4 25.7 | 15.2 25.5 | 10.0 20.9 | 12.8 23.9 | 11.0 21.4 |
| oxygen (mgO2 L−1) | 8.2 17.7 | 9.7 16.1 | 10.7 17.5 | 9.5 15.9 | 10.5 16.2 | 9.7 16.8 | 11.0 13.9 | 7.6 18.3 | 9.7 15.7 |
| pH | 8.3 9.2 | 8.4 9.0 | 8.5 8.6 | 8.0 8.8 | 8.3 8.9 | 8.1 8.6 | 7.7 8.5 | 8.2 8.9 | 7.8 8.4 |
| conductivity (μS cm−1) | 685 791 | 587 744 | 716 903 | 566 792 | 700 796 | 710 746 | 647 715 | 598 687 | 820 859 |
| chlorophyll-a (mg m−3) | 26.5 88.9 | 41.9 51.8 | 8.1 54.7 | 25.3 38.0 | 18.6 47.8 | 26.5 45.2 | 28.9 38.5 | 19.5 30.3 | 19.6 28.6 |
| ammonium N (mgN-NH4 L−1) | 0.99 2.59 | 1.24 2.76 | 0.88 2.26 | 1.03 3.03 | 1.02 1.79 | 0.91 2.07 | 0.86 2.37 | 0.97 1.93 | 0.54 1.22 |
| nitrates (mgN-NO3 L−1) | 0 0.88 | 0.77 3.04 | 2.82 10.51 | 0 1.00 | 2.44 5.57 | 1.22 2.72 | 0.34 1.26 | 0.19 1.62 | 2.94 8.08 |
| organic N (mgN L−1) | 1.6 3.9 | 2.0 3.5 | 1.8 3.2 | 1.4 1.8 | 1.1 3.6 | 1.7 2.2 | 1.2 2.3 | 1.3 1.7 | 2.0 3.7 |
| TN (mgN L−1) | 3.7 5.2 | 3.7 4.1 | 5.7 11.8 | 2.5 4.1 | 5.7 7.2 | 4.4 5.0 | 2.6 4.6 | 2.7 4.3 | 5.6 11.1 |
| SRP (mgP L−1) | 0.011 0.025 | 0.025 0.058 | 0.023 0.056 | 0.022 0.069 | 0.019 0.029 | 0.003 0.020 | 0.004 0.011 | 0.006 0.039 | 0.007 0.022 |
| TP (mgP L−1) | 0.050 0.072 | 0.061 0.122 | 0.046 0.080 | 0.049 0.121 | 0.046 0.101 | 0.027 0.067 | 0.030 0.175 | 0.028 0.125 | 0.048 0.200 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dondajewska, R.; Kowalczewska-Madura, K.; Gołdyn, R.; Kozak, A.; Messyasz, B.; Cerbin, S. Long-Term Water Quality Changes as a Result of a Sustainable Restoration—A Case Study of Dimictic Lake Durowskie. Water 2019, 11, 616. https://doi.org/10.3390/w11030616
Dondajewska R, Kowalczewska-Madura K, Gołdyn R, Kozak A, Messyasz B, Cerbin S. Long-Term Water Quality Changes as a Result of a Sustainable Restoration—A Case Study of Dimictic Lake Durowskie. Water. 2019; 11(3):616. https://doi.org/10.3390/w11030616
Chicago/Turabian StyleDondajewska, Renata, Katarzyna Kowalczewska-Madura, Ryszard Gołdyn, Anna Kozak, Beata Messyasz, and Sławek Cerbin. 2019. "Long-Term Water Quality Changes as a Result of a Sustainable Restoration—A Case Study of Dimictic Lake Durowskie" Water 11, no. 3: 616. https://doi.org/10.3390/w11030616
APA StyleDondajewska, R., Kowalczewska-Madura, K., Gołdyn, R., Kozak, A., Messyasz, B., & Cerbin, S. (2019). Long-Term Water Quality Changes as a Result of a Sustainable Restoration—A Case Study of Dimictic Lake Durowskie. Water, 11(3), 616. https://doi.org/10.3390/w11030616









