AQUALIFE Software: A New Tool for a Standardized Ecological Assessment of Groundwater Dependent Ecosystems
Abstract
1. Introduction
2. Software Capabilities
2.1. Section 1: Ecological Risk
2.2. Section 2: Hydrological-Hydromorphological Risk
2.3. Section 3: Conservation Priority Index
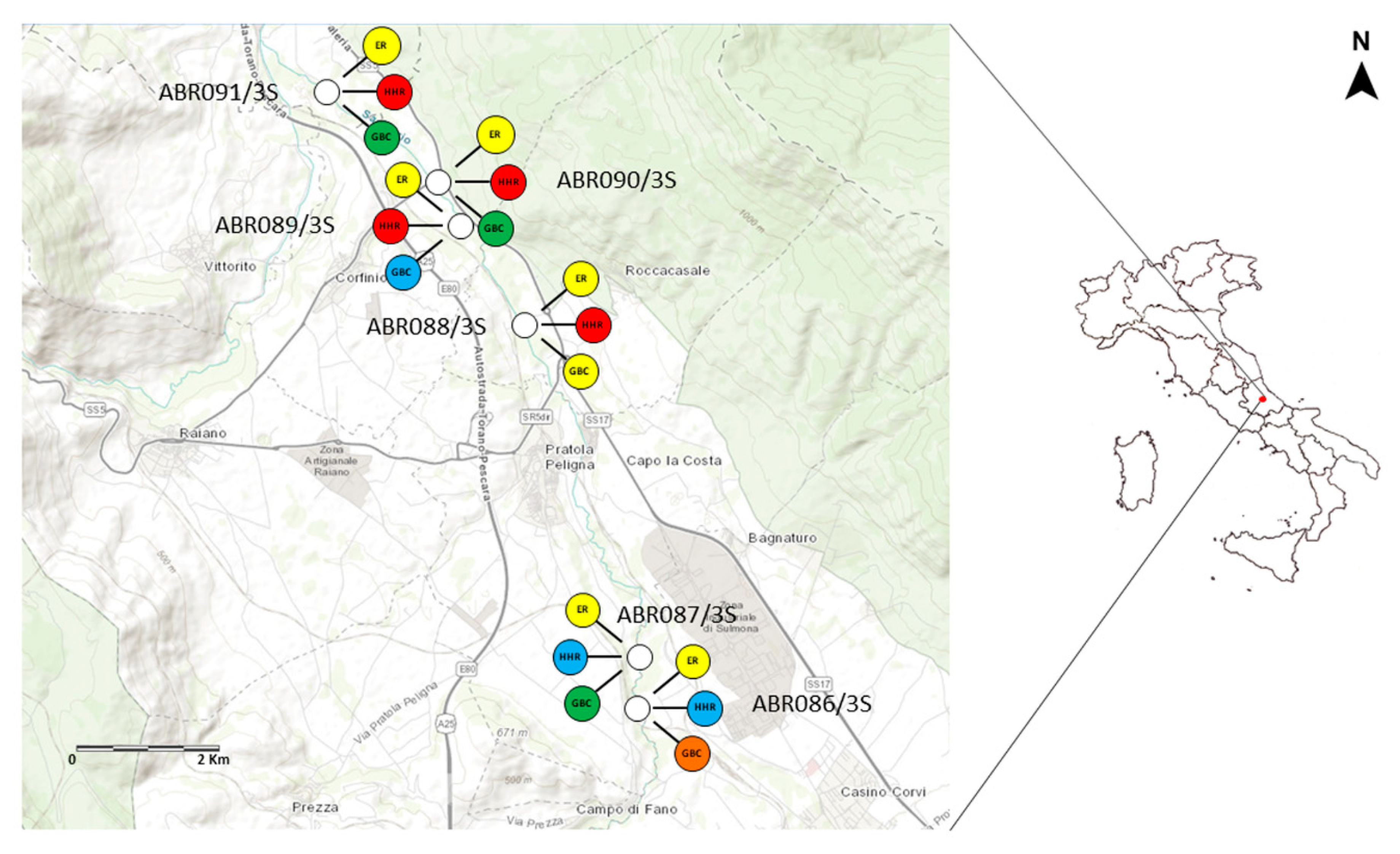
3. Example of Application: The Hyporheic Zone of the River Sagittario
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hatton, T.J.; Evans, R. Dependence of Ecosystems on Groundwater and Its Significance to Australia; CSIRO (Land and Water): Canberra, Australia, 1998. [Google Scholar]
- Eamus, D.; Froend, R. Groundwater-dependent ecosystems: The where, what and why of GDEs. Aust. J. Bot. 2006, 54, 91–96. [Google Scholar] [CrossRef]
- Gibert, J.; Deharveng, L. Subterranean ecosystems: A truncated functional biodiversity. Bioscience 2002, 52, 473–481. [Google Scholar] [CrossRef]
- Humphreys, W.F. Aquifers: The ultimate groundwater-dependent ecosystems. Aust. J. Bot. 2006, 54, 115–132. [Google Scholar] [CrossRef]
- Mammola, S.; Cardoso, P.; Culver, D.C.; Deharveng, L.; Ferreira, R.L.; Fiŝer, C.; Galassi, D.M.P.; Griebler, C.; Halse, S.; Humphreys, W.F.; et al. Scientists’ Warning on the Conservation of Subterranean Ecosystems. Bioscience 2019, 69, 641–650. [Google Scholar] [CrossRef]
- Ferreira, D.; Malard, F.; Dole-Olivier, M.-J.; Gibert, J. Obligate groundwater fauna of France: Diversity patterns and conservation implications. Biodivers. Conserv. 2007, 16, 567. [Google Scholar] [CrossRef]
- Deharveng, L.; Stoch, F.; Gibert, J.; Bedos, A.; Galassi, D.; Zagmajster, M.; Brancelj, A.; Camacho, A.; Fiers, F.; Martin, P.; et al. Groundwater biodiversity in Europe. Freshw. Biol. 2009, 54, 709–726. [Google Scholar] [CrossRef]
- Michel, G.; Malard, F.; Deharveng, L.; Di Lorenzo, T.; Sket, B.; De Broyer, C. Reserve selection for conserving groundwater biodiversity. Freshw. Biol. 2009, 54, 861–876. [Google Scholar] [CrossRef]
- Galassi, D.M.P.; Fiasca, B.; Tosto, D. Patterns of Copepod diversity (Copepoda: Cyclopoida, Harpacticoida) in springs of central Italy: Implications for conservation issues. In Studies on Freshwater Copepoda: A Volume in Honour of Bernard Dussart; Brill: Buckinghamshire, UK, 2011; Volume 16, pp. 199–226. [Google Scholar]
- European Commission. Technical Report on Groundwater Associated Aquatic Ecosystems; Technical Report No. 9; European Commission: Luxembourg, 2015.
- Marmonier, P.; Maazouzi, C.; Baran, N.; Blanchet, S.; Ritter, A.; Saplairoles, M.; Dole-Olivier, M.J.; Galassi, D.M.P.; Eme, D.; Doledec, S.; et al. Ecology-based evaluation of groundwater ecosystems under intensive agriculture: A combination of community analysis and sentinel exposure. Sci. Total Environ. 2018, 613, 1353–1366. [Google Scholar] [CrossRef]
- Jaffé, R.; Prous, X.; Zampaulo, R.; Giannini, T.C.; Imperatriz-Fonseca, V.L.; Maurity, C.; Oliveira, G.; Brandi, I.V.; Siqueira, J.O. Reconciling Mining with the Conservation of Cave Biodiversity: A Quantitative Baseline to Help Establish Conservation Priorities. PLoS ONE 2016, 11. [Google Scholar] [CrossRef]
- EPA. EPA’s Draft Report on the Environment Technical Document; EPA: Washington, DC, USA, 2003.
- Tomlinson, M.; Boulton, A.J.; Hancock, P.J.; Cook, P.G. Deliberate omission or unfortunate oversight: Should stygofaunal surveys be included in routine groundwater monitoring programs? Hydrogeol. J. 2007, 15, 1317. [Google Scholar] [CrossRef]
- Serov, P.; Kuginis, L.; Williams, J.P. Risk Assessment Guidelines for Groundwater Dependent Ecosystems, Volume 1—The Conceptual Framework; NSW Department of Primary Industries, Office of Water: Sydney, Australia, 2012.
- Council of the European Communities. Directive 2000/60/EC of the European Parliament and the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. Eur. Commun. 2000, 327, 1–73. [Google Scholar]
- Council of the European Communities. Directive 2006/118/EC of the European Parliament and of the Council of 12 December 2006 on the protection of groundwater against pollution and deterioration. Off. J. Eur. Union 2006, 372, 19–31. [Google Scholar]
- Millennium Ecosystem Assessment, Ecosystems and Human Well-being: Synthesis; Island Press: Washington, DC, USA, 2005.
- Kløve, B.; Ala-Aho, P.; Bertrand, G.; Boukalova, Z.; Erturk, A.; Goldscheider, N.; Ilmonen, J.; Karakaya, N.; Kupfersberger, H.; Kvaerner, J.; et al. Groundwater dependent ecosystems. Part I: Hydroecological status and trends. Environ. Sci. Policy 2011, 14, 770–781. [Google Scholar] [CrossRef]
- Megdal, S.B.; Gerlak, A.K.; Varady, R.G.; Huang, L.Y. Groundwater Governance in the United States: Common Priorities and Challenges. Groundwater 2015, 53, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Saccò, M.; Blyth, A.; Bateman, P.W.; Hua, Q.; Mazumder, D.; Nicole, W.; Humphreys, W.F.; Laini, A.; Griebler, C.; Grice, K. New light in the dark—A proposed multidisciplinary framework for studying functional ecology of groundwater fauna. Sci. Total Environ. 2019, 662, 963–977. [Google Scholar] [CrossRef]
- Taylor, R.G.; Scanlon, B.; Doll, P.; Rodell, M.; van Beek, R.; Wada, Y.; Longuevergne, L.; Leblanc, M.; Famiglietti, J.S.; Edmunds, M.; et al. Ground water and climate change. Nat. Clim. Chang. 2013, 3, 322–329. [Google Scholar] [CrossRef]
- Phyton Software Foundation (US). Available online: https://www.python.org/ (accessed on 3 October 2019).
- Django Project. Available online: https://www.djangoproject.com (accessed on 3 October 2019).
- Avramov, M.; Schmidt, S.I.; Griebler, C. A new bioassay for the ecotoxicological testing of VOCs on groundwater invertebrates and the effects of toluene on Niphargus inopinatus. Aquat. Toxicol. 2013, 130, 1–8. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Borgoni, R.; Ambrosini, R.; Cifoni, M.; Galassi, D.M.P.; Petitta, M. Occurrence of volatile organic compounds in shallow alluvial aquifers of a Mediterranean region: Baseline scenario and ecological implications. Sci. Total Environ. 2015, 538, 712–723. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Castano-Sanchez, A.; Di Marzio, W.D.; Garcia-Doncel, P.; Nozal Martinez, L.; Galassi, D.M.P.; Iepure, S. The role of freshwater copepods in the environmental risk assessment of caffeine and propranolol mixtures in the surface water bodies of Spain. Chemosphere 2019, 220, 227–236. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Di Marzio, W.D.; Fiasca, B.; Galassi, D.M.P.; Korbel, K.; Iepure, S.; Pereira, J.L.; Reboleira, A.; Schmidt, S.I.; Hose, G.C. Recommendations for ecotoxicity testing with stygobiotic species in the framework of groundwater environmental risk assessment. Sci. Total Environ. 2019, 681, 292–304. [Google Scholar] [CrossRef]
- Calamari, D.; Vighi, M. A proposal to define quality objectives for aquatic life for mixtures of chemical-substances. Chemosphere 1992, 25, 531–542. [Google Scholar] [CrossRef]
- Vighi, M.; Altenburger, R.; Arrhenius, Å.; Backhaus, T.; Bödeker, W.; Blanck, H.; Consolaro, F.; Faust, M.; Finizio, A.; Froehner, K.; et al. Water quality objectives for mixtures of toxic chemicals: Problems and perspectives. Ecotoxicol. Environ. Saf. 2003, 54, 139–150. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Cifoni, M.; Fiasca, B.; Di Cioccio, A.; Galassi, D.M.P. Ecological risk assessment of pesticide mixtures in the alluvial aquifers of central Italy: Toward more realistic scenarios for risk mitigation. Sci. Total Environ. 2018, 644, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Pennington, D. Extrapolating ecotoxicological measures from small data sets. Ecotoxicol. Environ. Saf. 2003, 56, 238–250. [Google Scholar] [CrossRef]
- Backhaus, T.; Faust, M. Predictive Environmental Risk Assessment of Chemical Mixtures: A Conceptual Framework. Environ. Sci. Technol. 2012, 46, 2564–2573. [Google Scholar] [CrossRef]
- Gustavsson, M.; Kreuger, J.; Bundschuh, M.; Backhaus, T. Pesticide mixtures in the Swedish streams: Environmental risks, contributions of individual compounds and consequences of single-substance oriented risk mitigation. Sci. Total Environ. 2017, 598, 973–983. [Google Scholar] [CrossRef]
- ECHA. Chapter, R.10: Characterisation of Dose [Concentration]-Response for Environment. Guidance for the Implementation of REACH—Guidance on Information Requirements and Chemical Safety Assessment; European Chemical Agency: Helskinki, Finland, 2008.
- European Commission. Technical Guidance for Deriving Environmental Quality Standard; European Commission: Brussels, Belgium, 2018.
- ECOTOX Database. Available online: https://cfpub.epa.gov/ecotox/ (accessed on 3 October 2019).
- ECOSAR Software. Available online: http://www.epa.gov/oppt/newchems/tools/21ecosar.htm (accessed on 3 October 2019).
- Galassi, D.M.P.; Fiasca, B.; Di Lorenzo, T.; Montanari, A.; Porfirio, S.; Fattorini, S. Groundwater biodiversity in a chemoautotrophic cave ecosystem: How geochemistry regulates microcrustacean community structure. Aquat. Ecol. 2017, 51, 75–90. [Google Scholar] [CrossRef]
- Rinaldi, M.; Surian, N.; Comiti, F.; Bussettini, M. IDRAIM—Sistema di Valutazione Idromorfologica, Analisi E Monitoraggio dei Corsi d’acqua—Versione Aggiornata 2016; ISPRA: Rome, Italy, 2016. [Google Scholar]
- Brunner, P.; Cook, P.G.; Simmons, C.T. Disconnected Surface Water and Groundwater: From Theory to Practice. Ground Water 2011, 49, 460–467. [Google Scholar] [CrossRef]
- Siergieiev, D.; Ehlert, L.; Reimann, T.; Lundberg, A.; Liedl, R. Modelling hyporheic processes for regulated rivers under transient hydrological and hydrogeological conditions. Hydrol. Earth Syst. Sci. 2015, 19, 329–340. [Google Scholar] [CrossRef]
- Hancock, P.J. The response of hyporheic invertebrate communities to a large flood in the Hunter River, New South Wales. Hydrobiologia 2006, 568, 255–262. [Google Scholar] [CrossRef]
- Arroita, M.; Flores, L.; Larranaga, A.; Martinez, A.; Martinez-Santos, M.; Pereda, O.; Ruiz-Romera, E.; Solagaistua, L.; Elosegi, A. Water abstraction impacts stream ecosystem functioning via wetted-channel contraction. Freshw. Biol. 2017, 62, 243–257. [Google Scholar] [CrossRef]
- Liu, D.S.; Zhao, J.; Chen, X.B.; Li, Y.Y.; Weiyan, S.P.; Feng, M.M. Dynamic processes of hyporheic exchange and temperature distribution in the riparian zone in response to dam-induced water fluctuations. Geosci. J. 2018, 22, 465–475. [Google Scholar] [CrossRef]
- Mathers, K.L.; Hill, M.J.; Wood, P.J. Benthic and hyporheic macroinvertebrate distribution within the heads and tails of riffles during baseflow conditions. Hydrobiologia 2017, 794, 17–30. [Google Scholar] [CrossRef]
- Sawyer, A.H.; Cardenas, M.B.; Bomar, A.; Mackey, M. Impact of dam operations on hyporheic exchange in the riparian zone of a regulated river. Hydrol. Process. 2009, 23, 2129–2137. [Google Scholar] [CrossRef]
- Boano, F.J.; Harvey, W.; Marion, A.; Packman, A.I.; Revelli, R.; Ridolfi, L.; Wörman, A. Hyporheic flow and transport processes: Mechanisms, models, and biogeochemical implications. Rev. Geophys. 2014, 52, 603–679. [Google Scholar] [CrossRef]
- Magliozzi, C.; Grabowski, R.; Packman, A.; Krause, S. Scaling down hyporheic exchange flows: From catchments to reaches. Hydrol. Earth Syst. Sci. Discuss. 2017. [Google Scholar] [CrossRef]
- European Commission. WFD Reporting Guidance 2016. Final Draft 6.0.6; European Commission: Brussels, Belgium, 2016.
- Loheide, S.P.; Booth, E.G. Effects of changing channel morphology on vegetation, groundwater, and soil moisture regimes in groundwater-dependent ecosystems. Geomorphology 2011, 126, 364–376. [Google Scholar] [CrossRef]
- Pacioglu, O.; Shaw, P.; Robertson, A. Patch scale response of hyporheic invertebrates to fine sediment removal in two chalk rivers. Fundam. Appl. Limnol. 2012, 181, 283–288. [Google Scholar] [CrossRef]
- Jones, I.; Growns, I.; Arnold, A.; McCall, S.; Bowes, M. The effects of increased flow and fine sediment on hyporheic invertebrates and nutrients in stream mesocosms. Freshw. Biol. 2015, 60, 813–826. [Google Scholar] [CrossRef]
- Pacioglu, O.; Robertson, A. The invertebrate community of the chalk stream hyporheic zone: Spatio-temporal distribution patterns. Knowl. Manag. Aquat. Ecosyst. 2017, 418, 10. [Google Scholar] [CrossRef][Green Version]
- Hancock, P.J. Human impacts on the stream-groundwater exchange zone. Environ. Manag. 2002, 29, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Merill, L.; Tonjes, D.J. A Review of the Hyporheic Zone, Stream Restoration, and Means to Enhance Denitrification. Crit. Rev. Sci. Technol. 2014, 44, 2337–2379. [Google Scholar] [CrossRef]
- Wondzell, S.M. Effect of morphology and discharge on hyporheic exchange flows in two small streams in the Cascade Mountains of Oregon, USA. Hydrol. Process. 2006, 20, 267–287. [Google Scholar] [CrossRef]
- Fumetti von, S.; Dmitrovic, D.; Pesic, V. The influence of flooding and river connectivity on macroinvertebrate assemblages in rheocrene springs along a third-order river. Fundam. Appl. Limnol. 2017, 190, 251–263. [Google Scholar] [CrossRef]
- Pesic, V.; Dmitrovic, D.; Savic, A.; Milosevic, D.; Zawal, A.; Vukasinovic-Pesic, V.; Fumetti von, S. Application of macroinvertebrate multimetrics as a measure of the impact of anthropogenic modification of spring habitats. Aquat. Conserv. Mar. Freshw. Ecosyst. 2019, 29, 341–352. [Google Scholar] [CrossRef]
- Salameh, E. Over-exploitation of groundwater resources and their environmental and socio-economic implications: The case of Jordan. Water Int. 2008, 33, 55–68. [Google Scholar] [CrossRef]
- Tal, A.; Katz, D. Rehabilitating Israel’s streams and rivers. Int. J. River Basin Manag. 2012, 10, 317–330. [Google Scholar] [CrossRef]
- Siwek, J.; Pociask-Karteczka, J. Springs in South-Central Poland—Changes and threats. Episodes 2017, 40, 38–46. [Google Scholar] [CrossRef]
- Dwire, K.A.; Mellmann-Brown, S.; Gurrieri, J.T. Potential effects of climate change on riparian areas, wetlands, and groundwater-dependent ecosystems in the Blue Mountains, Oregon, USA. Clim. Serv. 2018, 10, 44–52. [Google Scholar] [CrossRef]
- Sabater, F.; Butturini, A.; Mart, E.; Oz, I.; Romaní, A.; Wray, J.; Sabater, S. Effect of Riparian Vegetation Removal on Nutrient Retention in a Mediterranean Stream. J. N. Am. Benthol. Soc. 2000, 19, 609–620. [Google Scholar] [CrossRef]
- Possingham, H.P.; Wilson, K.A. Biodiversity—Turning up the heat on hotspots. Nature 2005, 436, 919–920. [Google Scholar] [CrossRef] [PubMed]
- Funk, S.M.; Fa, J.E. Ecoregion Prioritization Suggests an Armoury Not a Silver Bullet for Conservation Planning. PLoS ONE 2010, 5, e8923. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, P.; Erwin, T.L.; Borges, P.A.V.; New, T.R. The seven impediments in invertebrate conservation and how to overcome them. Biol. Conserv. 2011, 144, 2647–2655. [Google Scholar] [CrossRef]
- Fattorini, S. Use of insect rarity for biotope prioritisation: The tenebrionid beetles of the Central Apennines (Italy). J. Insect Conserv. 2010, 14, 367–378. [Google Scholar] [CrossRef]
- Fattorini, S. Biotope prioritisation in the Central Apennines (Italy): Species rarity and cross-taxon congruence. Biodivers. Conserv. 2010, 19, 3413–3429. [Google Scholar] [CrossRef]
- Fattorini, S.; Cardoso, P.; Rigal, F.; Borges, P.A.V. Use of Arthropod Rarity for Area Prioritisation: Insights from the Azorean Islands. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Kattan, G.H. Rarity and vulnerability: The birds of the Cordillera Central of Colombia. Conserv. Biol. 1992, 6, 64–70. [Google Scholar] [CrossRef]
- Rabinowitz, D. Seven forms of rarity. In The Biological Aspects of Rare Plant Conservation; Synge, H., Ed.; Sinauer Associates: Chichester, UK, 1981. [Google Scholar]
- Rabinowitz, D.; Cairns, S.; Dillon, T. Seven forms of rarity and their frequency in the flora of the British Isles. In Conservation Biology: The Science of Scarcity and Diversity; Soulè, M.E., Ed.; British Ecological Society: London, UK, 1986; pp. 182–204. [Google Scholar]
- Fattorini, S.; Fiasca, B.; Di Lorenzo, T.; Di Cicco, M.; Galassi, D.M.P. A new protocol for assessing the conservation priority of groundwater dependent ecosystems. Aquat. Conserv. Mar. Freshw. Ecosyst. submitted for publication.
- Caschetto, M.; Galassi, D.M.P.; Petitta, M.; Aravena, R. Evaluation of the sources of nitrogen compounds and their influence on the biological communities in the hyporheic zone of the Sagittario River, Italy: An isotopic and biological approach. Ital. J. Geosci. 2017, 136, 145–156. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Cannicci, S.; Spigoli, D.; Cifoni, M.; Baratti, M.; Galassi, D.M.P. Bioenergetic cost of living in polluted freshwater bodies: Respiration rates of the cyclopoid Eucyclops serrulatus under ammonia-N exposures. Fundam. Appl. Limnol. 2016, 188, 147–156. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Cifoni, M.; Lombardo, P.; Fiasca, B.; Galassi, D.M.P. Ammonium threshold values for groundwater quality in the EU may not protect groundwater fauna: Evidence from an alluvial aquifer in Italy. Hydrobiologia 2015, 743, 139–150. [Google Scholar] [CrossRef]
- Scharf, B.; Brunke, M. The recolonization of the river Elbe with benthic and hyporheic Ostracoda (Crustacea) after the reunion of Germany in 1989. Int. Rev. Hydrobiol. 2013, 98. [Google Scholar] [CrossRef]
- Hahn, H.J. The GW-Fauna-Index: A first approach to a quantitative ecological assessment of groundwater habitats. Limnologica 2006, 36, 119–137. [Google Scholar] [CrossRef]
- Fillinger, L.; Hug, K.; Trimbach, A.M.; Wang, H.; Kellermann, C.; Meyer, A.; Bendinger, B.; Griebler, C. The D-A-(C) index: A practical approach towards the microbiological-ecological monitoring of groundwater ecosystems. Water Res. 2019, 163, 114902. [Google Scholar] [CrossRef] [PubMed]
- Korbel, K.; Hose, G.C. The weighted groundwater health index: Improving the monitoring and management of groundwater resources. Ecol. Indic. 2017, 75, 164–181. [Google Scholar] [CrossRef]
- Korbel, K.L.; Hose, G.C. A tiered framework for assessing groundwater ecosystem health. Hydrobiologia 2011, 661, 329–349. [Google Scholar] [CrossRef]
| ER Rating | ER Magnitude | ER Color Ranking |
|---|---|---|
| 0 ≤ RQmix < 1 | no risk | |
| 1 ≤ RQmix < 10 | low risk | |
| 10 ≤ RQmix < 100 | medium risk | |
| 100 ≤ RQmix < 1000 | high risk | |
| RQmix ≥ 1000 | very high risk |
| Relevant Pressures: Names and Descriptions | Impacts: Names and Categories | ||
|---|---|---|---|
| Hydrological alteration Abstraction or flow diversion Dams, barriers, and locks | Presence of transversal works with reduction of discharge flow rates (detention and/or diversion, dams, weirs) | Alteration of discharge flow rates | HHYC-Altered habitats due to hydrological changes |
| Hydromorphological alteration | Presence of transversal works with reduction of discharge flow rates (detention and/or diversion, dams, weirs) limiting sediment and woody debris transport on riverbed | Alteration of the longitudinal connectivity in sediment and woody debris in streams/rivers benthic and hyporheic zones | HMOC-Altered habitats due to morphological changes |
| Abstraction or flow diversion | Presence of solid walls and/or embankments limiting permeability with the alluvial plain of streams and rivers | Alteration of the lateral connectivity in streams/rivers benthic and hyporheic zones | |
| Dams, barriers, and locks | Concreted riverbed, presence of knick points in rivers or channels where the longitudinal (lengthways) slope of the bed suddenly steepens (incision), artificial riverbed aggradation, clogging, overexploitation of aquifers | Alteration of the vertical connectivity river/aquifer | |
| Physical alteration of channel/bed/riparian area/shore | Artificially straightened channel | Morphological alteration of the river stretch and riverbed sediment composition | |
| Groundwater alteration of water level or volume | Removal of perifluvial or riparian vegetation | Increase of riverbank erosion | |
| Large woody debris removal | Large woody debris removal | ||
| Relevant Pressures | Impacts | ||
|---|---|---|---|
| Hydrological alteration Abstraction or flow diversion Dams, barriers, and locks | Presence of transversal works with reduction of discharge flow rates (detention and/or diversion, dams, weirs) downstream the spring emergence | Alteration of discharge flow rate | HHYC–Altered habitats due to hydrological changes |
| Overexploitation of aquifers | Alteration of the vertical connectivity | ||
| Physical alteration of channel/bed/riparian area/shore | Spring used for different purposes or concreted at its emergence | Morphological alteration of spring habitats with loss of the ecotonal nature of GW-fed springs | HMOC–Altered habitats due to morphological changes |
| Riparian vegetation removal | Habitat and microhabitat losses, nutrient dynamics alteration, current velocity alteration | ||
| Impacts | Impact Score |
|---|---|
| Alteration of discharge flow rates | 6 |
| Alteration of the longitudinal connectivity limiting sediment and woody debris transport on riverbed | 4 |
| Alteration of the lateral connectivity | 3 |
| Alteration of the vertical connectivity river/aquifer | 10 |
| Morphological alteration of the river stretch and riverbed sediment composition | 6 |
| Alteration of riparian vegetation | 2 |
| Lowering of woody debris storage in streams/rivers | 1 |
| Impacts | Impact Score |
|---|---|
| Alteration of discharge flow rates | 6 |
| Alteration of the ecological features of the spring (e.g., change from rheocrenic to limnocrenic spring as effect of damming) | 4 |
| Alteration of the vertical connectivity | 10 |
| Morphological alteration of spring habitats with loss of the ecotonal nature of GW-fed springs | 5 |
| Alteration of the riparian vegetation, habitat and microhabitat losses, nutrient dynamics alteration, current velocity alteration | 3 |
| HHR Rating | Risk Ranking | HHR Color Ranking |
|---|---|---|
| HHR < 10 | no risk | |
| HHR ≥ 10 | at risk |
| Dimension | Trait | Score Range |
|---|---|---|
| 1. GEOGRAPHY | Extent of occurrence | 1–7 |
| Degree of endemicity | 1–4 | |
| 2. ECOLOGY | Degree of GW dependence | 1–5 |
| Ecological specialization to GW | 1–3 | |
| Size of ecological niche | 1–5 | |
| Tolerance to variation of climatic parameters | 1–5 | |
| Microhabitat preferences | 1–5 | |
| 3. BIOLOGY | Trophic role | 1–5 |
| Life time span | 1–3 | |
| 4. POPULATION | Frequency | 1–4 |
| Abundance | 1–4 | |
| 5. EVOLUTIONARY HISTORY | Evolutionary origin | 1–3 |
| Phylogenetic rarity | 1–5 |
| GBC Rating | Priority Ranking | GBC Color Ranking |
|---|---|---|
| 0 ≤ GBC ≤ 0.2 | very low priority | |
| 0.2 < GBC ≤ 0.4 | low priority | |
| 0.4 < GBC ≤ 0.6 | medium priority | |
| 0.6 < GBC ≤ 0.8 | high priority | |
| 0.8 < GBC ≤ 1 | very high priority |
| Sampling Site | ER (RQmix) | ER Ranking | HHR | HHR Ranking | SSS | SSS Rescaled | BCC | GBC | Priority Ranking |
|---|---|---|---|---|---|---|---|---|---|
| ABRO86/3S | 58.85 | medium | 6 | no risk | 16 | 1 | 0.333 | 0.667 | high |
| ABRO87/3S | 83.75 | medium | 6 | no risk | 13.8 | 0.421 | 0.324 | 0.372 | low |
| ABRO88/3S | 32.11 | medium | 28 | at risk | 15 | 0.737 | 0.381 | 0.559 | medium |
| ABRO89/3S | 72.10 | medium | 20 | at risk | 12.2 | 0 | 0.344 | 0.172 | very low |
| ABRO90/3S | 51.22 | medium | 16 | at risk | 13.6 | 0.368 | 0.314 | 0.341 | low |
| ABRO91/3S | 49.05 | medium | 15 | at risk | 13.8 | 0.421 | 0.324 | 0.372 | low |
| ER Values | ER Monitoring Measures | HHR Values | HHR Monitoring Measures | GBC Values | Measures |
|---|---|---|---|---|---|
| 0 ≤ RQmix < 1 | SMPs | HHR < 10 | SMPs | 0 ≤ GBC ≤ 0.2 | No PMs |
| 1 ≤ RQmix < 10 | OMPs | HHR ≥ 10 | OMPs | 0.2 < GBC ≤ 0.4 | PMs if RQ ≥ 1000 and HHR ≥ 10 |
| 10 ≤ RQmix < 100 | OMPs | 0.4 < GBC ≤ 0.6 | PMs if RQ > 100 and HHR ≥ 10 | ||
| 100 ≤ RQmix < 1000 | OMPs | 0.6 < GBC ≤ 0.8 | PMs if RQ > 10 and HHR ≥ 10 | ||
| RQmix ≥ 1000 | OMPs | 0.8 < GBC ≤ 1 | PMs if RQ > 1 and/or HHR ≥ 10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strona, G.; Fattorini, S.; Fiasca, B.; Di Lorenzo, T.; Di Cicco, M.; Lorenzetti, W.; Boccacci, F.; Galassi, D.M.P. AQUALIFE Software: A New Tool for a Standardized Ecological Assessment of Groundwater Dependent Ecosystems. Water 2019, 11, 2574. https://doi.org/10.3390/w11122574
Strona G, Fattorini S, Fiasca B, Di Lorenzo T, Di Cicco M, Lorenzetti W, Boccacci F, Galassi DMP. AQUALIFE Software: A New Tool for a Standardized Ecological Assessment of Groundwater Dependent Ecosystems. Water. 2019; 11(12):2574. https://doi.org/10.3390/w11122574
Chicago/Turabian StyleStrona, Giovanni, Simone Fattorini, Barbara Fiasca, Tiziana Di Lorenzo, Mattia Di Cicco, Walter Lorenzetti, Francesco Boccacci, and Diana M. P. Galassi. 2019. "AQUALIFE Software: A New Tool for a Standardized Ecological Assessment of Groundwater Dependent Ecosystems" Water 11, no. 12: 2574. https://doi.org/10.3390/w11122574
APA StyleStrona, G., Fattorini, S., Fiasca, B., Di Lorenzo, T., Di Cicco, M., Lorenzetti, W., Boccacci, F., & Galassi, D. M. P. (2019). AQUALIFE Software: A New Tool for a Standardized Ecological Assessment of Groundwater Dependent Ecosystems. Water, 11(12), 2574. https://doi.org/10.3390/w11122574








